Abstract
Inhibition of the small GTPase RhoA attenuates the development of pulmonary edema and restores positive alveolar fluid clearance in a murine model of Pseudomonas aeruginosa pneumonia. Activated protein C (aPC) blocks the development of an unfavorably low ratio of small GTPase Rac1/RhoA activity in lung endothelium through endothelial protein C receptor (EPCR)/protease-activated receptor–1 (PAR-1)–dependent signaling mechanisms that include transactivating the sphingosine-1–phosphate (S1P) pathway. However, whether aPC's cytoprotective effects can attenuate the development of pulmonary edema and death associated with P. aeruginosa pneumonia in mice remains unknown. Thus, we determined whether the normalization of a depressed ratio of activated Rac1/RhoA by aPC would attenuate the P. aeruginosa–mediated increase in protein permeability across lung endothelial and alveolar epithelial barriers. Pretreatment with aPC significantly reduced P. aeruginosa–induced increases in paracellular permeability across pulmonary endothelial cell and alveolar epithelial monolayers via an inhibition of RhoA activation and a promotion of Rac1 activation that required the EPCR–PAR-1 and S1P pathways. Furthermore, pretreatment with aPC attenuated the development of pulmonary edema in a murine model of P. aeruginosa pneumonia. Finally, a cytoprotective-selective aPC mutant, aPC-5A, which lacks most of aPC's anticoagulant activity, reproduced the protective effect of wild-type aPC by attenuating the development of pulmonary edema and decreasing mortality in a murine model of P. aeruginosa pneumonia. Taken together, these results demonstrate a critical role for the cytoprotective activities of aPC in attenuating P. aeruginosa–induced lung vascular permeability and mortality, suggesting that cytoprotective-selective aPC-5A with diminished bleeding risks could attenuate the lung damage caused by P. aeruginosa in critically ill patients.
Keywords: protein C, Pseudomonas aeruginosa, RhoA, Rac1, acute lung injury
Clinical Relevance
We previously showed that inhibition of the small GTPase RhoA attenuates the development of pulmonary edema and restores positive alveolar fluid clearance in a murine model of P. aeruginosa pneumonia. Activated protein C (aPC) blocks the development of an unfavorably low ratio of small GTPase Rac1/RhoA activity in lung endothelium. However, it remains unknown whether aPC's cytoprotective effects can attenuate the development of pulmonary edema and death associated with P. aeruginosa pneumonia. Here we found that the cytoprotective activities of aPC play a critical role in attenuating P. aeruginosa–induced lung vascular permeability and mortality, suggesting that cytoprotective-selective aPC, with a diminished risk for bleeding, could attenuate lung damage caused by P. aeruginosa in critically ill patients.
Pseudomonas aeruginosa is an opportunistic pathogen that causes lethal pneumonia in immunocompromised individuals and in critically ill patients (1, 2). The high mortality of patients who develop P. aeruginosa pneumonia is associated with the development of acute lung injury (ALI), characterized by the flooding of airspaces with protein-rich edema. P. aeruginosa pneumonia is also characterized by a shift in the balance between procoagulant and anticoagulant/fibrinolytic activity in the alveoli, followed by intra-alveolar fibrin deposition. Early procoagulant activity in the alveolar space may exert beneficial effects by decreasing the protein leakage through the alveolar–capillary barrier caused by P. aeruginosa. However, excessive fibrin deposition may cause deleterious effects by increasing the lung inflammatory response and vascular permeability (3).
The protein C pathway is one of the major natural anticoagulant pathways, and thus regulates the activity of the coagulation system by inactivating factors Va and VIIIa and of the fibrinolytic pathway through its neutralization of plasminogen activator inhibitor–1 (PAI-1) activity (4, 5). In addition to its anticoagulant activity, activated protein C (aPC) also expresses indirect inflammatory properties by inhibiting the formation of thrombin. Indeed, thrombin triggers the release of chemokines and increases the surface expression of various adhesion molecules via the activation of its own receptor, protease-activated receptor–1 (PAR-1). aPC also limits the proinflammatory effect of fibrin by inhibiting PAI-1. Beyond these indirect protective effects via interference with different coagulation mediators, aPC exerts direct anti-inflammatory and antiapoptotic effects through interactions with endothelial cells and leukocytes via binding to the endothelial protein C receptor (EPCR) (5). In particular, aPC mediates direct protective effects on lung endothelial barrier function through cytoskeletal reorganization, secondary to the activation of the sphingosine-1–phosphate (S1P) pathway. By activating the S1P pathway, aPC restores a physiologic, elevated ratio of Rac1/RhoA activity, which prevents the formation of actin–myosin stress fibers and prevents an increase in paracellular permeability across the lung endothelial barrier caused by injurious agents (4–7).
Several clinical studies demonstrated the importance of the protein C pathway in critically ill patients with infectious and noninfectious ALI. For example, plasma and airspace concentrations of protein C are decreased in patients with acute lung injury or pneumonia, and these concentrations have prognostic significance for adverse outcomes in these patients (8). Furthermore, normal fibrinolytic mechanisms are impaired in the alveolar compartment in patients with P. aeruginosa pneumonia, and elevated concentrations of plasminogen activator inhibitor (PAI-1) in the plasma and pulmonary edema fluid are predictive of mortality in patients with P. aeruginosa pneumonia (9). Therefore, the correlation between low protein C concentrations and elevated PAI-1 concentrations may play an important role in the pathogenesis of infectious ALI.
Previous in vivo studies showed that P. aeruginosa causes the development of severe alveolar pulmonary edema in rodents through the Type III secretion system and the activation of several Toll-like receptors (10–12). These signaling pathways result in the activation of the small GTPase RhoA. Accordingly, ExoS and ExoT, two cytotoxins from the Type III secretion system, are responsible for the P. aeruginosa–mediated increase in protein permeability across lung endothelial cell monolayers via an inhibition of Rac1 and an activation of RhoA (13). In an in vivo study, we found that the inhibition of RhoA attenuates the increase in lung endothelial and alveolar epithelial permeability to protein, the development of pulmonary edema, and the inhibition of alveolar fluid clearance in a murine model of P. aeruginosa pneumonia (14). aPC favorably elevates the ratio of Rac1/RhoA activity in the lung endothelium, apparently by activating the S1P pathway through EPCR/PAR-1–dependent mechanisms, and indirectly by preventing the formation of thrombin and its detrimental effects on the barrier properties of the lung endothelium and alveolar epithelium (4). However, it remains unknown whether and by which mechanisms aPC would attenuate the development of pulmonary edema and death associated with P. aeruginosa pneumonia in mice.
Thus, we first determined that the inhibition of RhoA activation by aPC attenuated the P. aeruginosa–mediated increase in protein permeability across the lung endothelial and alveolar epithelial barriers. aPC significantly blunted the P. aeruginosa–induced increase in paracellular permeability across endothelial cell and alveolar epithelial monolayers via an elevation of the Rac1/RhoA activity ratio, which required the activation of the EPCR–PAR-1 and S1P pathways. Second, we found that pretreatment with aPC attenuated the development of pulmonary edema in a murine model of P. aeruginosa pneumonia. Finally, we demonstrated that a cytoprotective-selective aPC mutant with minimal anticoagulant activity reproduced the protective effects of wild-type aPC by attenuating the development of pulmonary edema and decreasing mortality in a murine model of P. aeruginosa pneumonia. Taken together, these results demonstrate a critical role for the cytoprotective activities of aPC in attenuating P. aeruginosa–induced lung vascular permeability.
Materials and Methods
Please see the online supplement for additional details.
Reagents
All cell culture media were prepared by the University of California at San Francisco Cell Culture Facility, using deionized water and analytical grade reagents.
Cell Culture
Bovine pulmonary arterial endothelial cells (BPAECs) (all passages < 8; catalogue number CCL-209; American Type Culture Collection, Manassas, VA) and human microvascular lung endothelial cells (HMVEC-Ls) (all passages < 8; catalogue number CC-0264; American Type Culture Collection) were cultured as previously described. Primary rat alveolar epithelial Type II (ATII) cells were isolated as described elsewhere (15).
Preparation of P. aeruginosa
The wild-type, PAK strain of Pseudomonas aeruginosa K was a gift from Stephen Lory, PhD (Harvard Medical School, Boston, MA). The preparation of P. aeruginosa was performed as described elsewhere (13).
Measurement of Transendothelial and Transepithelial Albumin Flux
Transendothelial and transepithelial albumin flux was measured as described elsewhere (15).
RhoA and Rac1 Activity Assay
The activity of RhoA in lung endothelial and alveolar epithelial cell monolayers was measured using a luminescence-based G-LISA RhoA activation kit, according to the manufacturer's instructions (Cytoskeleton, Inc., Denver, CO).
Immunofluorescence for Visualization of Actin Stress Fibers
Immunofluorescence for the visualization of actin stress fibers was performed as previously described (13).
Western Blot Analyses
Western blot analyses from cell homogenates were performed as described elsewhere (15).
Cell Viability Assay
Cell viability was measured by the Alamar Blue assay after exposure to various experimental conditions, as described elsewhere (13).
Mice
Wild-type C57BL/6 mice were purchased from Jackson Laboratories (Bar Harbor, ME). All animal experiments were approved by the Institutional Animal Care and Use Committee of the University of California at San Francisco.
Model of Pneumonia
Mice were anesthetized and instilled with P. aeruginosa (50 μl of phosphate-buffered saline containing 1 × 107 colony-forming units of P. aeruginosa), as reported previously (14).
Lung Vascular Permeability and Alveolar Fluid Clearance Measurements, Bronchoalveolar Lavage, and Bacterial Cultures from the Lungs, Spleen, and Blood
These measurements were performed as described elsewhere (13, 15).
BAL KC and Lung Myeloperoxidase Measurements
An ELISA assay for keratinocyte-derived cytokine (KC) was performed according to the manufacturer's protocol. Myeloperoxidase (MPO) activity was measured with the murine MPO kit HK210 (Cell Sciences, Canton, MA).
Survival Protocol
Twenty wild-type mice were randomly assigned to two groups (untreated mice, n = 10; 5A-aPC–treated mice, n = 10). Mice were exposed to P. aeruginosa, as already described. All mice received 1 ml of subcutaneous saline to prevent dehydration. Mortality in the two groups was monitored for 60 hours. Survival time was defined as the time between instillation and death.
Statistical Analysis
All data are summarized as mean ± SD. For the statistical analysis, we used Statview version 5.0 (SAS, Inc., Chicago, IL) and MedCalc version 7.2.0.2 (MedCalc Software, Inc, Maria Kerke, Belgium). The normal distribution was verified using the Kolmogorov-Smirnov test. Because all series of data were normally distributed, one-way ANOVA and the Fisher exact t test were used to compare experimental groups with control groups. P < 0.05 was considered statistically significant. A Kaplan-Meier analysis was used to compare survival between the two experimental groups of mice.
Results
aPC Attenuates P. aeruginosa–Induced Increases in Paracellular Permeability by Favorably Increasing the Ratio of Rac1/RhoA Activity in Lung Endothelial and Alveolar Epithelial Cell Monolayers
We previously reported that the increase in transendothelial albumin flux induced by P. aeruginosa was blocked by the inhibition of the small GTPase RhoA (14) or of RhoA activated kinase (ROCK), the immediate downstream effector of RhoA (13). Other investigators showed that aPC inhibits the activation and activity of RhoA in lung endothelial cells, and increases the activity of Rac1 (6). Thus, the first series of experiments was designed to determine the effect of aPC on the P. aeruginosa–mediated increase in permeability across BPAEC monolayers. The results showed that exposure to P. aeruginosa caused a significant increase in the paracellular permeability across BPAEC monolayers (Figure 1A). aPC (0.01–1.0 μg/ml) caused a dose-dependent inhibition of the increased permeability induced by P. aeruginosa across these cell monolayers (Figure 1A). The protective effect of aPC was associated with an elevated ratio of Rac1/RhoA activity and an inhibition of P. aeruginosa–induced formation of actin stress fibers in BPAEC monolayers (Figures 1B–1D). We also examined the effects of aPC on the P. aeruginosa–induced increases in permeability across human microvascular lung endothelial cell monolayers. The results also showed that exposure to P. aeruginosa caused a significant increase in paracellular permeability across HMVEC monolayers (Figure 1E), and that this protective effect was associated with a significant increase in the ratio of Rac1/RhoA activity in these cell monolayers (Figure 1F and G). These effects of aPC involved both an inhibition of RhoA activation and an augmentation of Rac1 activity. In similar related experiments using primary cultures of rat alveolar epithelial Type II cell monolayers, we found a comparable protective effect of aPC against the increase in permeability induced by P. aeruginosa (Figure 2A), associated with an augmentation in the ratio of Rac1/RhoA activity (Figures 2B and 2C). Exposure to P. aeruginosa did not affect the viability of the three primary cell types used for these experiments (data not shown). Taken together, these results demonstrate a protective role for aPC's activation of Rac1, linked to the inhibition of P. aeruginosa–dependent increases in protein permeability across both the lung endothelial and alveolar epithelial barriers, attributable to the activation of RhoA.
Figure 1.
Activated protein C (aPC) attenuates P. aeruginosa–induced increase in permeability via an inhibition of the small GTPase RhoA in bovine pulmonary endothelial and human microvascular lung endothelial cell monolayers. Bovine pulmonary artery cell (BPAEC) and human microvascular lung endothelial cell (HMVEC) monolayers were treated with the wild-type P. aeruginosa strain (PAK) (bacterial to bovine cell ratio, 1:2) or its vehicle. Cell monolayers were treated with recombinant human aPC (0.01–1.0 μg/ml) or its vehicle for 1 hour before stimulation with P. aeruginosa (PAK) or its vehicle. (A) aPC decreased PAK-induced permeability in a dose-dependent manner in BPAEC monolayers. (B) Pretreatment with aPC inhibited PAK-induced increase in RhoA activity in BPAEC monolayers. (C) aPC increased the ratio of Rac1/RhoA activity in BPAEC monolayers. (D) Pretreating cells with aPC inhibited PAK-induced formation of actin stress fibers in BPAEC monolayers. (E) aPC decreased PAK-induced permeability. (F) Pretreatment with aPC inhibited PAK-induced increase in RhoA activity in HMVEC monolayers. (G) aPC increased the ratio of Rac1/RhoA activity in HMVEC monolayers. All experiments were performed at least in triplicate, and repeated four times. Results are shown as mean ± SD. *P < 0.05 vs. control samples. **P < 0.05 vs. PAK-treated cell monolayers.
Figure 2.
aPC attenuates P. aeruginosa–induced increase in paracellular permeability via an inhibition of small GTPase RhoA in rat alveolar epithelial Type II (ATII) cell monolayers. Rat ATII cell monolayers were treated with wild-type P. aeruginosa strain PAK (bacterial to rat cell ratio, 1:2) or its vehicle. Cell monolayers were treated with recombinant human aPC (1.0 μg/ml) or its vehicle for 1 hour before stimulation with P. aeruginosa (PAK) or its vehicle. (A) aPC decreased PAK-induced permeability in a dose-dependent manner in ATII cell monolayers. (B) Pretreatment with aPC inhibited PAK-induced increase in RhoA activity in ATII cell monolayers. (C) aPC increased the ratio of Rac1/RhoA activity in ATII cell monolayers. All experiments were performed at least in triplicate, and repeated four times. Results are mean ± SD, *P < 0.05 vs. control samples. **P < 0.05 vs. PAK-treated cell monolayers.
aPC Attenuation of P. aeruginosa–Induced Increases in Lung Endothelial Permeability Requires EPCR–PAR-1 and S1P Pathways and PI3K-Dependent Mechanisms
Previous studies showed that the cytoprotective effect of aPC on the lung endothelium is mediated via an EPCR-dependent activation of the thrombin receptor, PAR-1. The S1P pathway, which increases endothelial barrier integrity by a reorganization of the cytoskeleton, is also required to mediate the protective effects of aPC in the lung endothelium (6, 7). Thus, we determined the requirements for EPCR and PAR-1 in mediating the protective effect of aPC on the P. aeruginosa–mediated increase in permeability across BPAEC monolayers. The results showed that blocking EPCR or PAR-1 with their respective blocking antibodies inhibited the protective effect of aPC on the barrier function of BPAEC monolayers exposed to P. aeruginosa (Figure 3A). This inhibition of aPC's barrier-protective effects was associated with blocking aPC's ability to blunt the activation of RhoA and to promote the activation of Rac1 in BPAEC monolayers exposed to P. aeruginosa (Figure 3B). We next demonstrated that aPC caused the phosphorylation of sphingosine-1 kinase (SK1) in BPAEC monolayers (Figure 3C). Furthermore, pretreating cells with 10 nM of the SK1 inhibitor, dimethylsphingosine, inhibited the protective effect of aPC on the barrier function of BPAEC monolayers exposed to P. aeruginosa (Figure 3D), whereas pretreatment with a specific agonist for the S1P1 receptor, SEW2871, significantly attenuated the damaging effect of P. aeruginosa on the barrier function of BPAEC monolayers (Figure 3E). Because aPC transactivates the S1P receptor, S1P1, apparently through the activation of the PI3-kinase–Akt pathway (6), experiments were designed to test the role of the PI3–kinase pathway in mediating the protective effect of aPC on the barrier function of BPAEC monolayers exposed to P. aeruginosa. The results showed that pretreating BPAEC monolayers with a specific inhibitor of all isoforms of PI3-kinase, N-(2,3-Dihydro-7,8-dimethoxyimidazo [1,2-c]quinazolin-5-yl)-3-pyridinecarboxamide (PIK-90), completely abolished the barrier-protective effects of aPC on BPAEC monolayers (Figure 3F). Taken together, these results demonstrate that the protective role of aPC on the barrier function of the lung endothelium exposed to P. aeruginosa is mediated by EPCR–PAR-1 and S1P pathways and PI3-kinase–dependent mechanisms.
Figure 3.
aPC attenuation of P. aeruginosa–induced increase in paracellular permeability is mediated via the endothelial protein C receptor (EPCR) and protease-activated receptor 1 (PAR-1) pathway, and the sphingosine-1–phosphate (S1P) and the phosphoinositide-3 kinase (PI3K) pathway, in bovine pulmonary endothelial cell monolayers. Bovine pulmonary artery cell (BPAEC) monolayers were treated with wild-type P. aeruginosa strain PAK (bacterial to bovine cell ratio, 1:2) or its vehicle. Cell monolayers were treated with recombinant human aPC (1.0 μg/ml) or its vehicle for 1 hour before stimulation with P. aeruginosa (PAK) or its vehicle. Some cell monolayers were also treated with a blocking antibody (Ab) to either EPCR or PAR-1 or their respective isotype control antibodies for 1 hour before stimulation with P. aeruginosa (PAK). (A) aPC (1.0 μg/ml) decreased PAK-induced permeability across BPAEC monolayers, but this effect was reversed with antibody blockade of either EPCR or PAR-1. (B) Blocking EPCR or PAR-1 receptors completely blocked the ability of aPC pretreatment (1.0 μg/ml) to inhibit the PAK-mediated increase in RhoA activity in BPAEC monolayers. Results are mean ± SD. *P < 0.05 vs. control samples. **P < 0.05 vs. PAK + aPC–treated BPAEC monolayers. (C) aPC (1.0 μg/ml) caused a significant increase in the phosphorylation of SK1. *P < 0.05 vs. control samples. (D) aPC decreased PAK-induced permeability, but this effect was blocked by pretreatment of cells with an SK1 inhibitor, dimethylsphingosine (DMS). *P < 0.05 vs. PAK-treated cell monolayers. **P < 0.05 vs. PAK + aPC–treated cell monolayers. (E) Pretreatment with a specific agonist for S1P1 receptor SEW2871 for 1 hour before exposure to PAK significantly attenuated PAK-mediated increase in paracellular permeability. *P < 0.05 vs. control samples. **P < 0.05 vs. PAK-treated cell monolayers. (F) Cell monolayers were treated with recombinant human aPC (1.0 μg/ml), a specific PI3K inhibitor N-(2,3-Dihydro-7,8-dimethoxyimidazo [1,2-c]quinazolin-5-yl)-3-pyridinecarboxamide (PIK-90) and/or their vehicle for 1 hour before stimulation with P. aeruginosa (PAK) or its vehicle. Results are mean ± SD. *P < 0.05 vs. control samples. **P < 0.05 vs. PAK + aPC–treated cell monolayers. For A–F, results are mean ± SD. Experiments were performed at least in triplicate, and repeated four times.
Cytoprotective-Selective aPC Mutants Inhibit P. aeruginosa–Induced Increases in Lung Endothelial Permeability
Because our initial results (Figures 1–3) demonstrated that the barrier-protective effects of aPC required EPCR and PAR-1, we tested the hypothesis that aPC mutants with normal cell signaling activities but less than 10% of normal anticoagulant activity (designated as cytoprotective-selective aPC mutants) can provide lung endothelium barrier-protective effects. Such aPC mutants, remarkably, can reduce mortality in murine models of sepsis (16). The results showed that pretreatment with the cytoprotective-selective human 3K3A-aPC mutant significantly attenuated the increased permeability induced by P. aeruginosa across BPAEC and HMVEC monolayers (Figures 4A and 4B). The inhibition of 3K3A-aPC's signaling effects on EPCR or PAR-1 with their respective blocking antibodies inhibited the barrier-protective effect of 3K3A-aPC on BPAEC monolayers exposed to P. aeruginosa (Figures 4C and 4D). Taken together, these results demonstrate that non-anticoagulant human mutant 3K3A-aPC retains its ability to protect the barrier function of the lung endothelium exposed to P. aeruginosa, and that the 3K3A-aPC mutant's protective effect is mediated by an EPCR/PAR-1–dependent mechanism.
Figure 4.
Cytoprotective-selective 3K3A-aPC attenuates P. aeruginosa–induced increase in paracellular permeability via EPCR and PAR-1 in bovine pulmonary artery and human microvascular lung endothelial cell monolayers. Bovine pulmonary artery endothelial cell (BPAEC) and human microvascular lung endothelial cell (HMVEC) monolayers were treated with wild-type P. aeruginosa strain PAK (bacterial to bovine/human cell ratio, 1:2) or its vehicle. Cell monolayers were treated with recombinant human cytoprotective-selective 3K3A-aPC (1.0 μg/ml) or its vehicle for 1 hour before stimulation with P. aeruginosa (PAK) or its vehicle. (A and B) 3K3A-aPC decreased PAK-induced permeability across bovine and human lung endothelial cell monolayers. *P < 0.05 vs. control samples. **P < 0.05 vs. PAK-treated cell monolayers. (C and D) Cytoprotective-selective 3K3A-aPC decreased PAK-induced permeability, but this effect was reversed by blocking either EPCR or PAR-1 across bovine and human lung endothelial cell monolayers. *P < 0.05 vs. control samples. **P < 0.05 vs. PAK ± aPC–treated cell monolayers. For A–D, results are mean ± SD. Each experiment was repeated at least four times.
aPC Decreases Pulmonary Edema and Prolongs Survival through Its Cytoprotective Activities in a Lethal Model of P. aeruginosa Pneumonia in Mice
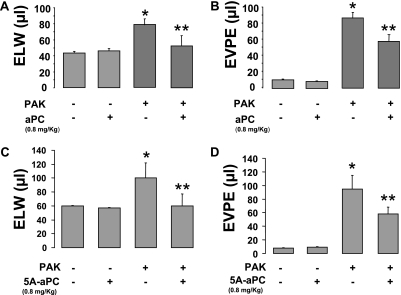
Experiments were designed to determine whether murine wild-type aPC and the murine cytoprotective-selective aPC mutant, 5A-aPC, in which five residues are replaced by alanine residues (16, 17) (0.8 mg/kg, intraperitoneal, given 1 hour before exposure of the airspace to P. aeruginosa or its vehicle), would attenuate the development of pulmonary edema and lung vascular permeability to protein in a murine model of P. aeruginosa pneumonia. The results showed that instillation of P. aeruginosa into the airspace caused the development of pulmonary edema (excess lung water), and significantly increased the lung's endothelial permeability to protein in mice. These outcomes were both greatly diminished by pretreating the animals with wild-type murine aPC or the cytoprotective-selective murine 5A-aPC mutant (0.8 mg/kg intraperitoneal) (Figures 5A–5D). We found that pretreating mice with 5A-aPC (0.8 mg/kg, intraperitoneal) also significantly attenuated the accumulation of protein-rich alveolar edema in the distal airspace of the lung (Figure 6A), and restored positive alveolar fluid clearance after the instillation of P. aeruginosa into the airspace of mice, without affecting the alveolar innate immune response necessary for the eradication of P. aeruginosa from the lung (Figure 6B). Indeed, pretreating mice with 5A-aPC (0.8 mg/kg, intraperitoneal) did not decrease the total cell and neutrophil counts, BALF concentrations of KC, and lung MPO activity, 4 hours after exposure to P. aeruginosa (Figures 7A–7D). We also measured bacterial clearance from the lungs and the bacterial count in the blood and spleens of mice treated with 5A-aPC or its vehicle. The results showed no change in bacterial clearance from the lungs (4.1 × 108 CFU/g tissue ± 0.2 versus 3.9 × 108 ± 0.2 CFU/g tissue, not significant), but a significant decrease in the number of bacteria in the bloodstream (4.1 × 102 CFU/100 μl ± 0.3 versus 0.5 × 102 ± 0.2 CFU/100 μl, P < 0.05) and spleen (8.3 × 104 ± 0.3 CFU/g tissue versus 2.5 × 104 ± 0.2 CFU/g tissue, P < 0.05) in mice treated with 5A-aPC, compared with mice pretreated with its vehicle. Finally, we demonstrated that 5A-aPC significantly decreased mortality in a lethal model of P. aeruginosa pneumonia in mice. Indeed, mice treated with cytoprotective-selective 5A-aPC demonstrated 40% survival at 60 hours after exposure of the airspace to P. aeruginosa, whereas mice treated with the control vehicle all died within 28 hours after the onset of P. aeruginosa pneumonia (P < 0.05) (Figure 7E). Taken together, these results demonstrate the protective effect of a cytoprotective-selective 5A-aPC mutant in attenuating the breakdown of the alveolar–capillary barrier and in decreasing mortality in a murine model of P. aeruginosa pneumonia.
Figure 5.
Pretreatment with cytoprotective-selective 5A-aPC reduces pulmonary edema in a murine model of P. aeruginosa pneumonia. C57BL/6 mice were treated with wild-type murine aPC or cytoprotective-selective murine 5A-aPC (0.8 mg/kg, intraperitoneal) or their vehicle, 1 hour before exposure of airspace to P. aeruginosa or its vehicle for 4 hours (n = 5 in each group). (A and C) Pretreatment with wild-type aPC or 5A-aPC significantly reduced pulmonary edema induced by P. aeruginosa. (B and D) Pretreatment with wild-type aPC or 5A-aPC significantly reduced lung vascular protein permeability induced by P. aeruginosa. ELW, extravascular lung water; EVPE, lung endothelial permeability to protein. Results are mean ± SD. *P < 0.05 vs. control samples. **P < 0.05 vs. mice treated with airspace P. aeruginosa and aPC vehicle.
Figure 6.
Pretreatment with cytoprotective-selective 5A-aPC attenuates alterations in alveolar epithelial permeability and fluid transport in a murine model of P. aeruginosa pneumonia. C57BL/6 mice were treated with murine 5A-aPC (0.8 mg/kg, intraperitoneal) or its vehicle, 1 hour before exposure of airspace to P. aeruginosa or its vehicle for 4 hours (n = 5 in each group). (A and B) Pretreatment with 5A-aPC attenuates P. aeruginosa-induced alteration in alveolar epithelial permeability. AFC, alveolar fluid clearance; BAL, bronchoalveolar lavage. Results are presented as mean ± SD. *P < 0.05 vs. control samples. **P < 0.05 vs. mice treated with airspace P. aeruginosa and 5A-aPC vehicle.
Figure 7.
Pretreatment with cytoprotective-selective 5A-aPC does not attenuate neutrophil recruitment into the airspace, and does not decrease lung myeloperoxidase activity or BAL fluid levels of keratinocyte-derived cytokine (KC), but decreases mortality in a murine model of P. aeruginosa pneumonia. (A–D) C57BL/6 mice were treated with non-anticoagulant murine 5A-aPC (0.8 mg/kg intraperitoneal) or its vehicle, 1 hour before exposure of airspace to P. aeruginosa or its vehicle for 8 hours (n = 5 in each group). BAL was performed, cells were counted, and myeloperoxidase (MPO) activity and KC levels were measured as described in Materials and Methods. For all experiments in A–D, results are shown as mean ± SD. (E) C57BL/6 mice were treated twice with murine 5A-aPC (0.8 mg/kg, intraperitoneal) or its vehicle, 1 hour before and 12 hours after exposure of airspace to P. aeruginosa or its vehicle. Kaplan-Meier survival analysis was performed (n = 10 mice in each experimental group). Mice treated with 5A-aPC had 40% survival at 60 hours after exposure of airspace to P. aeruginosa, whereas all mice treated only with control vehicle died within 28 hours after onset of P. aeruginosa pneumonia (P < 0.05).
Discussion
We previously reported that the inhibition of the small GTPase RhoA attenuated the development of pulmonary edema, and restored positive alveolar fluid clearance in a murine model of P. aeruginosa pneumonia (14). aPC can restore a physiologically favorable ratio of elevated Rac1/RhoA activity in the lung endothelium exposed to inflammatory mediators by activating the S1P pathway through an EPCR/PAR-1–dependent mechanism (6, 7). In experiments to define whether the cytoprotective effects of aPC would attenuate the development of pulmonary edema associated with P. aeruginosa pneumonia in mice, we made four key findings. First, we found that aPC inhibited the P. aeruginosa–dependent increase in protein permeability across both the lung endothelial and alveolar epithelial barriers. Second, this protective effect of aPC was not dependent on its anticoagulant activity, but was affected by the restoration of a favorably increased ratio of Rac1/RhoA activity in the lung endothelium. Third, these protective effects required two recognized aPC receptors, EPCR and PAR-1, as well as the sphingosine-1–phosphate pathway. Fourth, a cytoprotective-selective aPC mutant that retains the cytoprotective function of wild-type aPC but has lost more than 92% of its anticoagulant activity attenuated the breakdown of the alveolar–capillary barrier and decreased mortality, without inhibiting the lung innate immune response, in a murine model of P. aeruginosa pneumonia.
Previous experimental and clinical studies showed that P. aeruginosa causes the development of severe alveolar pulmonary edema in animals and humans through the Type III secretion system and the activation of several Toll-like receptors (10–12). These signaling pathways result in the activation of the small GTPase RhoA, which plays a critical role in mediating the increase in paracellular permeability across the lung endothelial barrier (18). Until recently, the mechanisms by which P. aeruginosa increased lung vascular permeability were poorly understood. Our laboratory demonstrated that ExoS and ExoT, two cytotoxins from the Type III secretion system, are responsible for the P. aeruginosa–mediated increase in protein permeability across lung endothelial cell monolayers via the inhibition of Rac1, combined with the activation of RhoA (13). In a subsequent in vivo study, we found that the inhibition of RhoA attenuated the increase in lung endothelial and alveolar epithelial permeability to protein and the development of pulmonary edema, and further, that this restored positive alveolar fluid clearance in a murine model of P. aeruginosa pneumonia (14). Thus, we hypothesized that the mediators decreasing the activity of RhoA in the lung cells constituting the alveolar–capillary barrier may attenuate the lung damage caused by P. aeruginosa. The protein C pathway is traditionally described as an anticoagulant system because of its proteolytic cleavage of activated coagulation factors V and VIII and its profibrinolytic activity via the direct or indirect inhibition of two major inhibitors of fibrinolysis, namely, PAI-1 and the thrombin activatable fibrinolysis inhibitor (4, 5). This anticoagulant pathway, however, also expresses direct and indirect anti-inflammatory properties, in part via its inhibition of RhoA activity. Indeed, by inhibiting the formation of thrombin, aPC prevents the detrimental effects of this coagulation protein on lung endothelial and epithelial permeability that are mediated by the activation of RhoA and the corresponding inhibition of Rac1 (19–21). aPC can also inhibit the activity of RhoA via mechanisms that involve activation of S1P pathway, and that require the aPC receptors EPCR and PAR-1 (6, 7). Thus, the first objective of this study was to determine whether pretreatment with aPC in vitro would attenuate the increase in protein permeability across the lung endothelial and alveolar epithelial barriers caused by exposure to P. aeruginosa. Our results demonstrated that the protective effect of aPC on the barrier function of both the lung endothelium and alveolar epithelium was mediated by the favorable normalization of the Rac1/RhoA activity ratio. Indeed, P. aeruginosa inhibited the activity of Rac1 while it increased the activity of RhoA in both cell types. When cell monolayers were pretreated with aPC and then exposed to the bacteria, a reversal of these deleterious effects occurred in these two key small GTPases, with a restoration of high Rac1 activity and a corresponding inhibition of the RhoA activity.
What are the signaling pathways implicated in the P. aeruginosa–mediated increase in protein permeability across the lung endothelial and alveolar epithelial barriers? Although our results demonstrate a critical role for the activation of RhoA in mediating the deleterious effect of P. aeruginosa on the alveolar–capillary membrane, the signaling pathways that control changes in protein permeability via the activation of RhoA are likely to be different for the two components of this barrier. For example, we previously reported an important role for TGF-β1 signaling in mediating the breakdown of the alveolar–epithelial barrier associated with acute lung injury (22). One of the major mechanisms mediating the activation of TGF-β1 in the distal lung epithelium is the activation of RhoA by IL-1β or thrombin agonists, resulting in a αvβ6–integrin–mediated activation of TGF-β1 in alveolar epithelial cells (15). Furthermore, TGF-β1 mediates an increase in protein permeability and an inhibition of baseline and c-AMP–dependent vectorial fluid transport across the alveolar–epithelial barrier in experimental rodent models of acute lung injury (23). Finally, we recently demonstrated that P. aeruginosa increased protein permeability across rat alveolar epithelial Type II cell monolayers, an effect that was inhibited by a specific inhibitor of RhoA or a soluble Type II receptor for TGF-β1 (13, 14). The reversal of the activity ratio of Rac1 to RhoA by aPC attenuated the P. aeruginosa–induced increase in protein permeability across ATII cell monolayers, in accordance with our previous work, and is likely to be at least in part mediated by the TGF-β1 signaling pathway. In contrast, although PAR-1 and EPCR are expressed by alveolar epithelial cells (19, 24), the signaling pathways implicated in the activation of Rac1 and the subsequent inhibition of RhoA activity by aPC remain unknown, and will be examined in future studies.
In the lung endothelium, our results demonstrate that the barrier-enhancing effect of aPC on lung endothelium exposed to P. aeruginosa was dependent on its coreceptors, EPCR and PAR-1. Nonetheless, in principle, aPC may also prevent the formation of thrombin and fibrin, and would thus inhibit their known deleterious effects on lung vascular permeability. Additional experiments demonstrated the importance of the S1P and the PI3–kinase pathways in causing the favorable normalization of the Rac1/RhoA activity ratio in these cell monolayers. aPC not only attenuates coagulation, but its biological activities include anti-inflammatory, antiapoptotic, and endothelial-protective properties (4, 5). For example, pretreating confluent HUVEC monolayers with aPC decreases thrombin-induced protein permeability across these monolayers (7). Blocking sphingosine–kinase 1 and the S1P1receptor, two main components of the S1P pathway, by short, interfering RNA transfection reduced the barrier-protective effect of aPC (7). In another study, Finigan and colleagues demonstrated direct evidence of cytoskeletal reorganization induced by aPC in human pulmonary artery endothelial cells (6). In these cells, pretreatment with aPC attenuated the formation of actin stress fibers induced by thrombin. These actin stress fibers play an important role in hyperpermeability to protein across the lung endothelium (18). aPC's barrier-protective effects thus require S1P, its receptor, S1P1, EPCR, PAR-1, and an activation of the PI3–kinase pathway (6, 7). Taken together, these previous findings and our present data indicate an important role for the EPCR–PAR-1–S1P pathways in mediating the protective effect of aPC against the breakdown of the lung endothelial barrier caused by P. aeruginosa.
Despite our progress in understanding the mechanisms behind the cytoprotective effects of the protein C pathway, many questions remain unanswered. The first question concerns the apparent paradox that both thrombin and aPC bind to the PAR-1 receptor, but may exert opposite effects on paracellular permeability across the vascular endothelium. However, Russo and colleagues (25) provided additional information about this apparent paradox. Those investigators showed that when aPC signaling and endothelial barrier-protective effects were abolished in cells lacking caveolin-1, thrombin signaling remained intact (25). These finding suggest that the compartmentalization of PAR-1 in caveolae is critical for aPC signaling to Rac1 activation and endothelial barrier protection. These investigators also found that aPC induces PAR-1 phosphorylation and desensitizes endothelial cells to thrombin signaling, but promotes limited receptor cleavage and internalization, even after prolonged exposure (25). These data explain, at least in part, the differential effects of aPC and thrombin on endothelial barrier function, even though both mediators bind to PAR-1. These results are important to a further understanding of the complex interactions between thrombin and aPC, but strong experimental evidence indicates that aPC possesses additional anti-inflammatory properties. For example, a specific monoclonal antibody that blocks the activation of protein C by thrombin leads to 100% lethality in lethal dose 50 (LD50) models of animal sepsis (26). These results suggest that aPC may exert additional cytoprotective effects, possibly related to the inhibition of neutrophil activation and the reduction of the endothelial cell expression of adhesion molecules (4). Second, the importance of S1P signaling as a critical link between inflammation and coagulation was only recently addressed in the landmark paper by Niessen and colleagues (27). Although both thrombin and aPC signal via PAR-1, they exert opposite effects on the modulation of endothelial cell permeability. Niessen and colleagues (27) convincingly demonstrated that the PAR-1 cleaved by thrombin or by the aPC–EPCR complex causes the coupling of S1P to different receptors. Signaling triggered via the EPCR–aPC–PAR-1 axis is mediated through the S1P1 receptor, whereas the thrombin–PAR-1 axis is coupled to the S1P3 receptor. Furthermore, the differential effects of aPC and thrombin on vascular permeability could be recapitulated in vivo with small molecule agonists and antagonists for S1P1 or S1P3 (27). Nevertheless, the role of different S1P receptors, and particularly S1P1, in modulating lung vascular permeability is still not fully understood. One recent study indicated that the enhancing effect of S1P1 on the barrier function of lung endothelium is largely dependent on the concentration and route of administration of its agonists (28). In the present study, we found that the inhibition of sphingosine kinase-1 or PI3-kinase by specific inhibitors completely blocked the protective effect of aPC against the P. aeruginosa–mediated increase in lung endothelial permeability. In contrast, treating lung endothelial cell monolayers with a S1P1 agonist partly reproduced the protective effect of aPC on the barrier function of the lung barrier, indicating that the S1P pathway plays an important role in mediating the protective effect of aPC against the breakdown of the lung endothelial barrier by P. aeruginosa.
Because the protective effect of aPC against the P. aeruginosa–mediated increase in lung endothelial permeability required aPC receptors and thus appeared dependent on its cytoprotective effects, we next determined whether aPC mutants (human 3K-aPC or murine 5A-aPC) that have lost more than 92% of their anticoagulant activity, but have retained normal cell signaling activities (29), would provide the same protective effects as observed with wild-type aPC. We found that these cytoprotective-selective aPC mutants not only inhibited the deleterious effects of P. aeruginosa, but also protected against alveolar flooding edema and decreased mortality in a murine model of P. aeruginosa pneumonia. These results are the first to indicate that the protective effects of aPC in an experimental model of P. aeruginosa pneumonia are mediated by the cytoprotective, EPCR–PAR-1–dependent activities of this protein. This study extends to P. aeruginosa lung injury the previously described concept that a non-anticoagulant aPC mutant can be as effective as wild-type aPC in reducing mortality after an intravenous Escherichia coli endotoxin challenge in mice, and in enhancing the survival of mice subjected to the polymicrobial peritoneal sepsis triggered by a colonic stent implantation (16). Conversely, the administration of a recombinant aPC variant with greatly diminished cytoprotective functions, but with full anticoagulant function, demonstrated no therapeutic benefit in reducing mortality in a murine E. coli endotoxemia model (17).
The effects of wild-type aPC in protecting against acute lung injury varied in different reports. Earlier studies reported a protective effect of aPC on lung inflammation after the administration of E. coli endotoxin (30). Further experimental evidence supported the use of aPC as a potential therapeutic agent in acute lung injury. For example, aPC given systemically or by inhalation protected against ventilator-induced lung injury (31, 32). aPC also improved pulmonary function in ovine acute lung injury from smoke inhalation and sepsis (33). aPC, administered to donor airways, attenuated ischemia–reperfusion injury after experimental lung transplantation in rats (34). In contrast, other experimental studies failed to identify a clear anti-inflammatory or protective effect of aPC on acute lung injury caused by hyperoxia or oleic acid (35, 36). In addition, a recent Phase II trial evaluating the effects of aPC in patients with acute lung injury without sepsis was halted because no beneficial effects were evident (37). In experimental models of P. aeruginosa pneumonia, two different research groups reported that aPC failed to show a protective effect on lung function, despite the correction of airspace procoagulant activity induced by the instillation of P. aeruginosa (38, 39). The difference between the results of these earlier studies and our work can be attributed to several reasons. First, in previous studies, a large dose of aPC was given either by multiple intravenous boluses or continuous intravenous infusion. In contrast, we only administered one intraperitoneal bolus of wild-type or non-anticoagulant mutant aPC. In both earlier studies, aPC reversed the procoagulant activity of the distal airspace induced by the presence of P. aeruginosa. The formation of fibrin within the airspaces was shown to be an important mechanism that limits the dissemination of bacteria within the lung and into the bloodstream. Second, in one previous study, rats treated with a continuous intravenous infusion of aPC had more severe lung injury than the animals that received P. aeruginosa alone. This deleterious effect of aPC could also be partly explained by the findings of Sammani and colleagues (28). In their study, the administration of a large dose of an agonist of the S1P1 receptor would increase, but not inhibit, the increase in lung vascular permeability induced by E. coli endotoxin (28). Because the activation of the S1P1 receptor is likely to mediate some of the cytoprotective mechanisms of aPC, we speculate that increasing the dose of aPC (or of its cytoprotective-selective mutants) may be deleterious to lung vascular permeability. Additional studies will be needed for a better understanding of the relative importance of S1P pathways in mediating the protective effects of aPC. Recent studies showed that aPC can also signal through the apolipoprotein E receptor 2 in monocyte-like human cells, and can thereby activate protective PI3K–Akt pathways (40). However, it remains unknown whether the signaling activated by this new aPC receptor would play a role in the protective effects of aPC against bacterial lung injury, as observed in the present study.
In conclusion, we report that aPC significantly reduced the P. aeruginosa–induced increase in paracellular permeability across endothelial cell and alveolar epithelial monolayers via an inhibition of RhoA activity and via an activation of Rac1 activity, as mediated by EPCR–PAR-1–S1P pathways. Furthermore, pretreatment with aPC, by virtue of its cytoprotective properties, attenuated the development of pulmonary edema and decreased mortality in a murine model of P. aeruginosa pneumonia. These outcomes were associated with a decrease in the systemic dissemination of P. aeruginosa, without inhibiting neutrophil activity in lung tissue in response to the airspace instillation of these bacteria. Further studies will determine whether the administration of cytoprotective-selective aPC mutants by repeated bolus dosing or by inhalation may offer a new potential adjuvant therapy to treat the lung injury induced by P. aeruginosa, an infection associated with high mortality in critically ill patients.
Supplementary Material
Acknowledgments
We are grateful for the technical assistance of Ms. Xiao Xu (Scripps Research Institute).
Footnotes
This work was supported in part by National Institutes of Health grants RO1 GM- GM086416 (J.-F.P.), HL031950 (J.H.G.), and HL052246 (J.H.G.), and by an American Lung Association Senior Research Training Fellowship and T32 GM008440 (J.R.).
This article has an online supplement, which is accessible from this issue's table of contents at www.atsjournals.org
Originally Published in Press as DOI: 10.1165/rcmb.2010-0397OC on January 21, 2011
Author Disclosure: J.H.G. received financial reimbursement for consultancies from ZZ Biotech, LLC, and his university has a patent pending with ZZ Biotech, LLC. None of the other authors has a financial relationship with a commercial entity that has an interest in the subject of this manuscript.
References
- 1.Rello J, Allegri C, Rodriguez A, Vidaur L, Sirgo G, Gomez F, Agbaht K, Pobo A, Diaz E. Risk factors for ventilator-associated pneumonia by Pseudomonas aeruginosa in presence of recent antibiotic exposure. Anesthesiology 2006;105:709–714 [DOI] [PubMed] [Google Scholar]
- 2.Rello J, Rue M, Jubert P, Muses G, Sonora R, Valles J, Niederman MS. Survival in patients with nosocomial pneumonia: impact of the severity of illness and the etiologic agent. Crit Care Med 1997;25:1862–1867 [DOI] [PubMed] [Google Scholar]
- 3.Gunther A, Mosavi P, Heinemann S, Ruppert C, Muth H, Markart P, Grimminger F, Walmrath D, Temmesfeld-Wollbruck B, Seeger W. Alveolar fibrin formation caused by enhanced procoagulant and depressed fibrinolytic capacities in severe pneumonia: comparison with the acute respiratory distress syndrome. Am J Respir Crit Care Med 2000;161:454–462 [DOI] [PubMed] [Google Scholar]
- 4.Neyrinck AP, Liu KD, Howard JP, Matthay MA. Protective mechanisms of activated protein C in severe inflammatory disorders. Br J Pharmacol 2009;158:1034–1047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mosnier LO, Zlokovic BV, Griffin JH. The cytoprotective protein C pathway. Blood 2007;109:3161–3172 [DOI] [PubMed] [Google Scholar]
- 6.Finigan JH, Dudek SM, Singleton PA, Chiang ET, Jacobson JR, Camp SM, Ye SQ, Garcia JG. Activated protein C mediates novel lung endothelial barrier enhancement: role of sphingosine 1–phosphate receptor transactivation. J Biol Chem 2005;280:17286–17293 [DOI] [PubMed] [Google Scholar]
- 7.Feistritzer C, Riewald M. Endothelial barrier protection by activated protein C through PAR1-dependent sphingosine 1–phosphate receptor–1 crossactivation. Blood 2005;105:3178–3184 [DOI] [PubMed] [Google Scholar]
- 8.Ware LB, Matthay MA, Parsons PE, Thompson BT, Januzzi JL, Eisner MD. Pathogenetic and prognostic significance of altered coagulation and fibrinolysis in acute lung injury/acute respiratory distress syndrome. Crit Care Med 2007;35:1821–1828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Song Y, Lynch SV, Flanagan J, Zhuo H, Tom W, Dotson RH, Baek MS, Rubio-Mills A, Singh G, Kipnis E, et al. Increased plasminogen activator inhibitor–1 concentrations in bronchoalveolar lavage fluids are associated with increased mortality in a cohort of patients with Pseudomonas aeruginosa. Anesthesiology 2007;106:252–261 [DOI] [PubMed] [Google Scholar]
- 10.Roy-Burman A, Savel RH, Racine S, Swanson BL, Revadigar NS, Fujimoto J, Sawa T, Frank DW, Wiener-Kronish JP. Type III protein secretion is associated with death in lower respiratory and systemic Pseudomonas aeruginosa infections. J Infect Dis 2001;183:1767–1774 [DOI] [PubMed] [Google Scholar]
- 11.Hauser AR, Cobb E, Bodi M, Mariscal D, Valles J, Engel JN, Rello J. Type III protein secretion is associated with poor clinical outcomes in patients with ventilator-associated pneumonia caused by Pseudomonas aeruginosa. Crit Care Med 2002;30:521–528 [DOI] [PubMed] [Google Scholar]
- 12.Miao EA, Mao DP, Yudkovsky N, Bonneau R, Lorang CG, Warren SE, Leaf IA, Aderem A. Innate immune detection of the Type III secretion apparatus through the NLRC4 inflammasome. Proc Natl Acad Sci USA 2010;107:3076–3080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ganter MT, Roux J, Su G, Lynch SV, Deutschman CS, Weiss YG, Christiaans SC, Myazawa B, Kipnis E, Wiener-Kronish JP, et al. Role of small GTPases and alphaVbeta5 integrin in Pseudomonas aeruginosa–induced increase in lung endothelial permeability. Am J Respir Cell Mol Biol 2009;40:108–118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Carles M, Lafargue M, Goolaerts A, Roux J, Song Y, Howard M, Weston D, Swindle JT, Hedgpeth J, Burel-Vandenbos F, et al. Critical role of the small GTPase RhoA in the development of pulmonary edema induced by Pseudomonas aeruginosa in mice. Anesthesiology 2010;113:1134–1143 [DOI] [PubMed] [Google Scholar]
- 15.Ganter MT, Roux J, Miyazawa B, Howard M, Frank JA, Su G, Sheppard D, Violette SM, Weinreb PH, Horan GS, et al. Interleukin-1beta causes acute lung injury via alphaVbeta5 and alphaVbeta6 integrin-dependent mechanisms. Circ Res 2008;102:804–812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kerschen EJ, Fernandez JA, Cooley BC, Yang XV, Sood R, Mosnier LO, Castellino FJ, Mackman N, Griffin JH, Weiler H. Endotoxemia and sepsis mortality reduction by non-anticoagulant activated protein C. J Exp Med 2007;204:2439–2448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mosnier LO, Zampolli A, Kerschen EJ, Schuepbach RA, Banerjee Y, Fernandez JA, Yang XV, Riewald M, Weiler H, Ruggeri ZM, et al. Hyperantithrombotic, noncytoprotective Glu149Ala-activated protein C mutant. Blood 2009;113:5970–5978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Komarova Y, Malik AB. Regulation of endothelial permeability via paracellular and transcellular transport pathways. Annu Rev Physiol 2010;72:463–493 [DOI] [PubMed] [Google Scholar]
- 19.Jenkins RG, Su X, Su G, Scotton CJ, Camerer E, Laurent GJ, Davis GE, Chambers RC, Matthay MA, Sheppard D. Ligation of protease-activated receptor 1 enhances alpha(V)beta6 integrin-dependent TGF-beta activation and promotes acute lung injury. J Clin Invest 2006;116:1606–1614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xu MY, Porte J, Knox AJ, Weinreb PH, Maher TM, Violette SM, McAnulty RJ, Sheppard D, Jenkins G. Lysophosphatidic acid induces alphaVbeta6 integrin-mediated TGF-beta activation via the LPA2 receptor and the small G protein G alpha(Q). Am J Pathol 2009;174:1264–1279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Su G, Hodnett M, Wu N, Atakilit A, Kosinski C, Godzich M, Huang XZ, Kim JK, Frank JA, Matthay MA, et al. Integrin alphaVbeta5 regulates lung vascular permeability and pulmonary endothelial barrier function. Am J Respir Cell Mol Biol 2007;36:377–386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pittet JF, Griffiths MJ, Geiser T, Kaminski N, Dalton SL, Huang X, Brown LA, Gotwals PJ, Koteliansky VE, Matthay MA, et al. TGF-beta is a critical mediator of acute lung injury. J Clin Invest 2001;107:1537–1544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Roux J, Carles M, Koh H, Goolaerts A, Ganter MT, Chesebro BB, Howard M, Houseman BT, Finkbeiner W, Shokat KM, et al. Transforming growth factor beta1 inhibits cystic fibrosis transmembrane conductance regulator–dependent cAMP-stimulated alveolar epithelial fluid transport via a phosphatidylinositol 3–kinase–dependent mechanism. J Biol Chem 2010;285:4278–4290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang L, Bastarache JA, Wickersham N, Fang X, Matthay MA, Ware LB. Novel role of the human alveolar epithelium in regulating intra-alveolar coagulation. Am J Respir Cell Mol Biol 2007;36:497–503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Russo A, Soh UJ, Paing MM, Arora P, Trejo J. Caveolae are required for protease-selective signaling by protease-activated receptor–1. Proc Natl Acad Sci USA 2009;106:6393–6397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Taylor FB, Jr, Chang A, Esmon CT, D'Angelo A, Vigano-D'Angelo S, Blick KE. Protein C prevents the coagulopathic and lethal effects of Escherichia coli infusion in the baboon. J Clin Invest 1987;79:918–925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Niessen F, Schaffner F, Furlan-Freguia C, Pawlinski R, Bhattacharjee G, Chun J, Derian CK, Andrade-Gordon P, Rosen H, Ruf W. Dendritic cell PAR1–S1P3 signalling couples coagulation and inflammation. Nature 2008;452:654–658 [DOI] [PubMed] [Google Scholar]
- 28.Sammani S, Moreno-Vinasco L, Mirzapoiazova T, Singleton PA, Chiang ET, Evenoski CL, Wang T, Mathew B, Husain A, Moitra J, et al. Differential effects of S1P receptors on airway and vascular barrier function in the murine lung. Am J Respir Cell Mol Biol 2010;43:394–402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mosnier LO, Yang XV, Griffin JH. Activated protein C mutant with minimal anticoagulant activity, normal cytoprotective activity, and preservation of thrombin activable fibrinolysis inhibitor–dependent cytoprotective functions. J Biol Chem 2007;282:33022–33033 [DOI] [PubMed] [Google Scholar]
- 30.van der Poll T, Levi M, Nick JA, Abraham E. Activated protein C inhibits local coagulation after intrapulmonary delivery of endotoxin in humans. Am J Respir Crit Care Med 2005;171:1125–1128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Finigan JH, Boueiz A, Wilkinson E, Damico R, Skirball J, Pae HH, Damarla M, Hasan E, Pearse DB, Reddy SP, et al. Activated protein C protects against ventilator-induced pulmonary capillary leak. Am J Physiol Lung Cell Mol Physiol 2009;296:L1002–L1011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Maniatis NA, Letsiou E, Orfanos SE, Kardara M, Dimopoulou I, Nakos G, Lekka ME, Roussos C, Armaganidis A, Kotanidou A. Inhaled activated protein C protects mice from ventilator-induced lung injury. Crit Care 2010;14:R70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Maybauer MO, Maybauer DM, Fraser JF, Traber LD, Westphal M, Enkhbaatar P, Cox RA, Huda R, Hawkins HK, Morita N, et al. Recombinant human activated protein C improves pulmonary function in ovine acute lung injury resulting from smoke inhalation and sepsis. Crit Care Med 2006;34:2432–2438 [DOI] [PubMed] [Google Scholar]
- 34.Hirayama S, Cypel M, Sato M, Anraku M, Liaw PC, Liu M, Waddell TK, Keshavjee S. Activated protein C in ischemia–reperfusion injury after experimental lung transplantation. J Heart Lung Transplant 2009;28:1180–1184 [DOI] [PubMed] [Google Scholar]
- 35.Looney MR, Esmon CT, Matthay MA. Role of coagulation pathways and treatment with activated protein C in hyperoxic lung injury. Thorax 2009;64:114–120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Richard JC, Bregeon F, Leray V, Le Bars D, Costes N, Tourvieille C, Lavenne F, Devouassoux-Shisheboran M, Gimenez G, Guerin C. Effect of activated protein C on pulmonary blood flow and cytokine production in experimental acute lung injury. Intensive Care Med 2007;33:2199–2206 [DOI] [PubMed] [Google Scholar]
- 37.Liu KD, Levitt J, Zhuo H, Kallet RH, Brady S, Steingrub J, Tidswell M, Siegel MD, Soto G, Peterson MW, et al. Randomized clinical trial of activated protein C for the treatment of acute lung injury. Am J Respir Crit Care Med 2008;178:618–623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Choi G, Hofstra JJ, Roelofs JJ, Florquin S, Bresser P, Levi M, van der Poll T, Schultz MJ. Recombinant human activated protein C inhibits local and systemic activation of coagulation without influencing inflammation during Pseudomonas aeruginosa pneumonia in rats. Crit Care Med 2007;35:1362–1368 [DOI] [PubMed] [Google Scholar]
- 39.Robriquet L, Collet F, Tournoys A, Prangere T, Neviere R, Fourrier F, Guery BP. Intravenous administration of activated protein C in Pseudomonas-induced lung injury: impact on lung fluid balance and the inflammatory response. Respir Res 2006;7:41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yang XV, Banerjee Y, Fernandez JA, Deguchi H, Xu X, Mosnier LO, Urbanus RT, de Groot PG, White-Adams TC, McCarty OJ, et al. Activated protein C ligation of APOER2 (LRP8) causes Dab1-dependent signaling in U937 cells. Proc Natl Acad Sci USA 2009;106:274–279 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.