Abstract
Objective. To define the role of IL-10 in lupus pathogenesis, and to understand the immunological mechanisms underlying resistance vs susceptibility to lupus disease induction by dendritic cells (DCs) and dying cells.
Methods. Groups of IL-10-deficient and normal C57BL/6 mice were injected with syngenic DCs that had ingested necrotic cells prepared by either freeze–thaw cycle (DC/necF/T) or heat shock (DC/necH/S) procedures, or with DC or necrotic cells alone, or with PBS only. Disease development, including proteinuria and renal pathological changes, was monitored. Levels of autoantibodies against different lupus-associated nuclear antigens were measured by ELISAs, and IC deposition in the kidneys was confirmed by immunostaining.
Results. No significant proteinuria was detected in the mice. However, striking renal pathological changes typical of IC-mediated GN were consistently observed in the DC/necF/T-treated IL-10−/− mice. These included glomerular hypercellularity and macrophage infiltration, renal IC deposition, circulating kidney-reactive autoantibodies and the presence of immunoglobulin G2 isotype-specific antibody complexes in the diseased kidneys. We demonstrated further that host-derived IL-10 was primarily responsible for protecting against the induction of pathogenic Th1 type of autoantibody responses in the mice.
Conclusion. IL-10 protects against the induction of lupus-like renal end-organ damage by down-regulating pathogenic Th1 responses.
Keywords: IL-10, Autoimmunity, Lupus, Dendritic cells, Dying cells
Introduction
SLE is a complex autoimmune disorder and the aetiology is still far from clear. The disease is characterized immunologically by the production of autoantibodies against a variety of self-tissue antigens, particularly of the nuclear components [1]. Development of IC-mediated kidney disease may result in renal failure, which is a severe complication and major cause of death in SLE [1, 2]. While B cells are understandably very important in the process, the induction of autoantibodies especially those of pathogenic isotypes [3–6] is largely T-cell dependent and antigen driven [1, 7]. The questions have been how these harmful immune responses are initiated, perpetuated and, importantly, why they are under control in normal individuals [8–11].
Emerging evidence indicates that processing and presentation of autoantigens acquired from dying cells by dendritic cells (DCs) are involved in the development of lupus disease [4, 12, 13]. The findings may provide and explain immunologically the initial trigger of autoimmune responses, but more complex mechanisms appear to be involved in the regulation of the disease induction and development, which are evidently governed by multiple and genetically determined disease predisposing or protective factors [14]. We have previously demonstrated that injections of DCs loaded with syngenic necrotic cells (DC/nec) induced strong ANA responses in mouse models [4]. The treatment not only accelerated disease progression in a spontaneous lupus mouse model (MRL/lpr strain), but also induced a full-blown lupus-like disease in the wild-type (W/T) control MRL/+ mice. Another intriguing finding from the study was that, in contrast to the susceptible Murphy Roths Large (MRL) strains, similarly treated C57BL/6 mice were fully protected from the disease induction despite high levels of circulating anti-dsDNA antibodies. A pathogenic Th1 type of response was found to be closely associated with the disease induction. Importantly, the results suggested that differences in the resistance vs susceptibility to disease induction could be explained by the presence or absence of a feedback regulatory mechanism in which IL-10 was likely to play an important role [4].
IL-10 is a potent immunosuppressive cytokine. It plays an essential role in dampening down overt immune responses, especially the pro-inflammatory Th1 type of response, and suppresses autoimmunity [15–17]. The cytokine is known to inhibit DC maturation and functions, including their ability to produce IL-12 essential for driving Th1-cell differentiation [18]. The protective role of IL-10 has been previously demonstrated in several Th1-driven autoimmune diseases, including RA [19] and IBD [20]. The role of IL-10 in SLE is, however, still very controversial. Elevated serum levels and enhanced in vitro production of IL-10 have been reported in patients with SLE [21, 22]. Since IL-10 is also a cofactor for B-cell growth, as well as a switching factor for IgG1, IgG3 and IgA antibody isotypes, the enhanced IL-10 expression, particularly its close association with disease activity, has been interpreted as being pathogenic in nature [22, 23]. In support of this notion, results from several studies in SLE patients and their relatives indicated an association of IL-10 promoter polymorphism (high IL-10 expression) with the disease [24, 25], although the findings varied in studies on different geographical populations [26]. Conclusions drawn from one clinical trial in a group of six SLE patients in Mexico reported 9 years ago did also suggest a beneficial effect of anti-IL-10 treatment for the disease [27], but so far there has been no further report to confirm these findings.
Results from studies in mice also suggest that the IL-10 pathway is involved in murine lupus, but conflicting findings have been reported too as to whether IL-10 may play a disease-promoting or protective role [28–30]. While injection of anti-IL-10 antibodies was shown to delay disease activity in NZB/W F1 lupus-prone mice [28], an acceleration of disease onset and severity was observed in MRL/lpr mice deficient in IL-10 gene [29]. Genetically, while no linkage of IL-10 gene polymorphism to disease susceptibility in the mouse has been reported to date, polymorphism of the murine IL-10 receptor alpha chain (Il10ra) of low expression has recently been shown to be associated with the autoimmune phenotype in both of the NZW and MRL mouse strains [31].
The present study therefore was designed to define the role of IL-10 in lupus pathogenesis. By employing mice with an otherwise resistant background (C57BL/6), but deficient in their IL-10 gene (IL-10−/−), we tried to understand the immunological and molecular mechanisms underlying resistance vs susceptibility to lupus disease induction.
Materials and methods
Mice
IL-10 knockout mice (H-2b, strain B6.129P2-Il10tm1Cgn/J, stock no. 002251, generation: N10F33, JAXR Mice Database-002251 B6.129P2-Il10<tm1Cgn>/J), and W/T control C57BL/6 mice (H-2b), were purchased from The Jackson Laboratory (Bar Harbor, ME, USA) and set up as breeders. The breeders of IL-10−/− strain have generally a poor breeding efficiency, due to development of spontaneous IBD [32]. These mice and animals undergoing experimental procedures were maintained in individual ventilation cages (IVCs). All experiments involving live animals were carried out strictly according to the protocol under licences approved by the Committee on the Use of Live Animals in Teaching and Research (CULATR 761-03, 913-04), Hong Kong, and by the UK Government Home Office (PIL70/5946).
DC generation and uptake of necrotic cells in vitro
DCs were prepared as previously described [4]. Briefly, bone marrow precursor cells from 6- to 8-week-old mice were cultured in medium containing murine GM-CSF (20 ng/ml; Invitrogen) and murine IL-4 (5 ng/ml; PeproTech) for 6 days. Two types of necrotic cell were prepared from syngeneic mouse splenocytes, and cell necrosis was induced by either a freezing (−80°C, 12 min) and thawing (37°C) cycle (NecF/T), or by a heat-shock procedure (56°C, 1 h) (NecH/S), respectively. Cell necrosis was confirmed by trypan blue (1%, Sigma), or by propidium iodide (PI), staining. DCs were then co-cultured overnight with the necrotic cells (1 : 5, DC : necrotic cells) in vitro and the efficiency of dying cell uptake by DC assessed as described previously [4]. DC purity and functional phenotypes before and after co-culturing with dying cells were analysed by flow cytometry using antibodies to DC surface makers (MHC class II, CD11c, CD80, CD86, CD40; BD Pharmingen).
Immunization protocol
A total of 1 × 106 DCs loaded with either NecF/T (DC/necF/T) or NecH/S (DC/necH/S) cells of the same strain, or DC alone (DC); or 5 × 106 necrotic cells alone (Nec), were injected in PBS intravenously into individual mice. In selected experiments, for comparison, the W/TDC/nec or IL-10−/− DC/nec cells were also injected reciprocally into the W/T and IL-10−/−-recipient mice. Three injections were given at 14-day intervals starting at the age of 7−9 weeks. At 12–16 weeks post-injection (5–6 months of age), the mice were sacrificed and renal pathological changes assessed by histological and immunocytochemical examinations. The in vivo model was repeated in principle five times with consistent results, and the repeated experiments were started freshly with naïve and age- and sex-matched animals, and monitored throughout until the experimental end points.
Due to the poor breeding efficiency, however, the number of age- and sex-matched mice available for each of the experiments, and repeated experiments, was often limited and difficult to predict. In addition, IL-10−/− mice with signs of enterocolitis [32] were excluded at initial immunization. Therefore, each experiment had to be designed based on availability of the mice with often small group size, and priority of the experimental groups (i.e. key groups only in some in vivo experiments). A total of 58 W/T and 72 IL-10−/− mice were tested in groups, in five experiments, by the in vivo model. These included mice of the two strains injected with either DC/necF/T (W/T: n = 22, IL-10−/−: n = 27), DC/necH/S (W/T: n = 12, IL-10−/−: n = 16), DC (W/T: n = 11, IL-10−/−: n = 11) or NecF/T (W/T: n = 3, IL-10−/−: n = 3); and the control groups (Control) were strain-, age- and sex-matched mice injected with PBS only or no injection (W/T: n = 10, IL-10−/−: n = 15), respectively. Also, during the experiments, some of the IL-10−/− mice that developed severe enterocolitis had to be put down for ethical reasons. When this occurred during a relatively early stage of the experiments, they were excluded from the quantitative pathological assessments mentioned below (see supplementary tables 1 and 2, available as supplementary data at Rheumatology Online).
Renal pathological assessments
Proteinuria was assessed weekly using Haema–combistix (Bayer Diagnostics), graded according to the manufacturer’s instructions. Sixteen weeks after the initial DC immunizations, mice were killed and kidney tissues were either snap-frozen, or fixed in Bouin’s solution for at least 2 h, transferred into 70% ethanol and processed into paraffin. For histological examinations, the paraffin-embedded kidney sections were stained with periodic acid–Schiff (PAS) reagent and scored for GN. Glomerular cellularity was graded in a blinded fashion as follows: Grade 0, normal; Grade 1, hypercellularity involving >50% of the glomeruli tuft in 25–50% of glomeruli; Grade 2, hypercellularity involving >50% of the glomeruli tuft in 50–75% of glomeruli; Grade 3, hypercellularity involving >75% of the glomeruli or crescents in >25% of glomeruli; and Grade 4, severe proliferative GN in >90% of glomeruli. For consistency, the grading was all done blindly by an experienced renal pathologist, H.T.C. (second author), in the Hammersmith Hospital, London.
For detection of renal IC deposition, FITC-conjugated goat polyclonal antibodies against mouse IgG (1/200 dilution; Sigma-Aldrich), IgG1 (1/100 dilution; Serotec, STAR81F), IgG2b (1/50 dilution; Serotec, STAR83F) and C3 (1/100 dilution; Cappel, #55500), and a rat anti-mouse CD68 antibody (1/200 dilution), were used on the frozen tissue sections. For IgG2ab subclass detection, a biotinylated goat anti-mouse IgG2ab antibody (1/50 dilution; BD Pharmingen #553504) was added as the primary antibody, followed by FITC-conjugated streptavidin (1/100 dilution; Vector, #SA5001). In selected experiments, the staining intensity was quantified as previously described and expressed as arbitrary fluorescence units (AFU) [33].
Serological analyses
Blood samples were collected every 2 weeks, and serum levels of anti-dsDNA Abs (IgG, IgG1, IgG2ab, IgG2b) were measured by ELISA as described previously [4]. Briefly, microtitre plates coated with type I calf thymus DNA (Sigma) were treated with S1 nuclease (3 U/ml, Invitrogen) to digest ssDNA. Serial diluted serum samples were added and bound Abs detected with alkaline phosphatase (AP)-conjugated goat anti-mouse IgG (Sigma), IgG1, IgG2ab or IgG2b (Southern Biotech. Associates) detecting antibodies. The results were expressed as arbitrary ELISA units (AEU) relative to a standard positive control using pooled sera from aged (5–6 months) and terminally ill MRL/lpr lupus mice. The pooled MRL/lpr sera were stored at −80°C as aliquots and used as a standard positive control throughout the study.
To determine the presence of kidney reactive autoantibodies, serum samples doubling diluted (1 : 5 to 1 : 80) were applied to the kidney sections prepared from normal W/T mice (12 week old, female), and incubated for 1 h at room temperature (RT). Bound antibodies (Abs) were detected with fluorescein-conjugated IgG Fc-specific anti-mouse Ab (1/200, Sigma), and analysed using an Olympus BX4 fluorescence microscope (Olympus).
Statistics
The non-parametric Mann–Whitney test and the Student’s t-test were used for statistical analysis of results from the in vivo and in vitro experiments, respectively. Results from the in vivo experiments were also reanalysed and confirmed by the one-way non-parametric analysis of variance (ANOVA) test. P ≤ 0.05 (*) was considered statistically significant; ≤0.01 (**) highly statistically significant; and ≤0.001 (***) very highly statistically significant.
Results
To test the hypothesis that IL-10 plays a protective role against lupus disease induction, groups of IL-10−/− and W/T control mice (7–9 weeks old, female) were injected intravenously with syngenic necrotic cells loaded DCs (DC/nec), three times at bi-weekly intervals using a protocol we described previously [4]. Two types of necrotic cell, prepared by either the freeze–thaw cycle (DC/necF/T) or heat-shock procedure (DC/necH/S), were used and compared in the present study (see Materials and methods section). Experimental control groups included mice injected with DCs alone, necrotic cells alone or PBS only. Disease development was subsequently monitored throughout the experiments.
Mice deficient in IL-10 showed increased susceptibility to DC/nec-induced renal disease
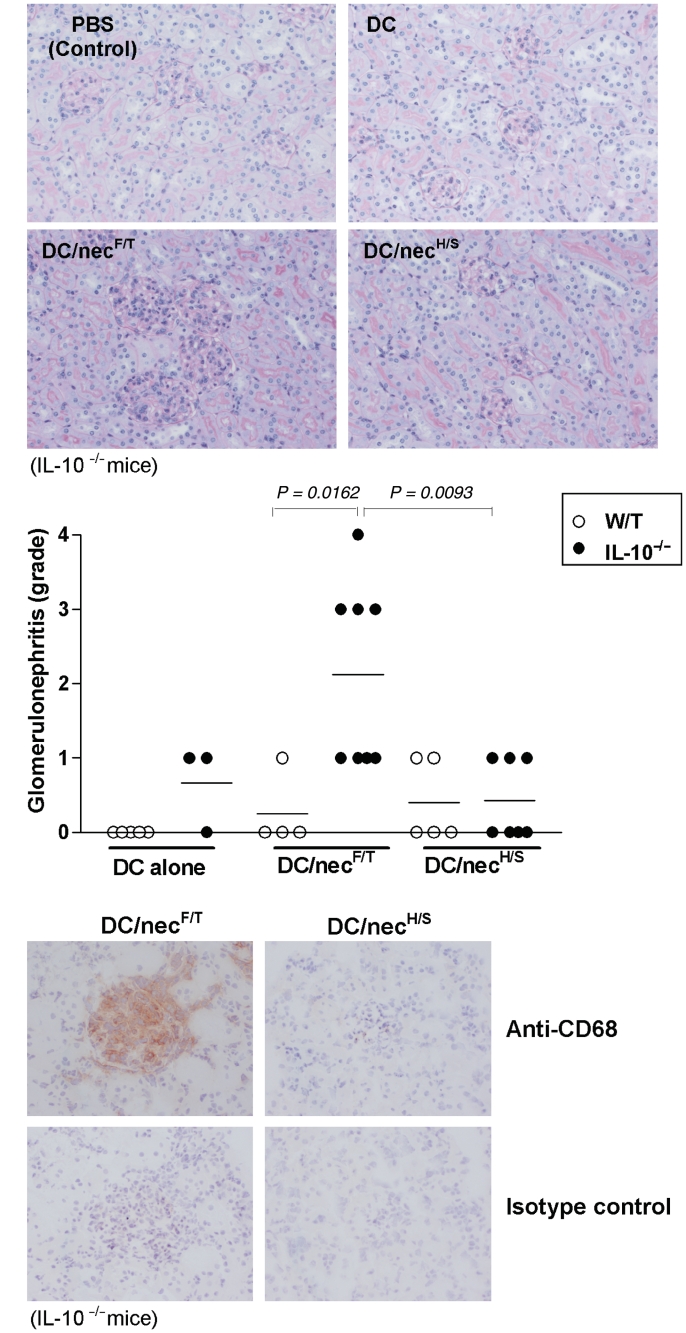
There was no significant proteinuria detected in any of the experimental mouse groups during and after the course of treatment (supplementary figure 1, available as supplementary data at Rheumatology Online). However, upon examining the kidneys at the end of each of three repeated experiments (4 months after the first DC injection), severe renal pathological changes typical of the IC-mediated GN were consistently observed in the IL-10−/− mice treated with DC/necF/T but not with DC/necH/S, DCs or necrotic cells alone or PBS only; or in any of the similarly treated W/T control mouse groups. The pathological changes included increased glomerular size, glomerular hypercellularity, narrowing of capillary lumens (Fig. 1A) and macrophage (CD68+) infiltration (Fig. 1B). Results from quantitative analysis (individually and blindly scored 0–4) confirmed that glomerular hypercellularity was significantly higher (3- to 5-fold the mean scores) in the DC/necF/T-treated IL-10−/− mice (Fig. 1A, dot plots) as compared with mice of other treatment groups.
Fig. 1.
Renal pathological assessments. Groups of W/T and IL-10−/− mice (7–9 weeks of age, female) were injected three times, and at bi-weekly intervals, with 1 × 106 syngenic DCs that had been preloaded with necrotic cells prepared either by the freeze–thaw (DC/necF/T) or heat-shock (DC/necH/S) procedure (see Materials and methods section), or with DC alone (DC) or PBS alone (PBS). At 16-week post-injection (6 months of age), all mice were sacrificed and renal pathological changes assessed by histological and immunocytochemical examination. (A) Kidney histological changes: PAS-stained kidney sections showing representative micrographs for each of the four experimental groups. Glomerular cellularity was scored (0–4, see Materials and methods section) for individual mice and shown as dot plots. (B) Glomerular macrophage infiltration: immunostaining of kidneys for CD68 (brown) showing extensive macrophage infiltration in the affected glomeruli of DC/necF/T—but not DC/necH/S-treated IL-10−/− mice. Original magnifications ×20 in (A), and ×40 in (B). The quantitative data (dot plots) shown in (A) (randomly selected only from repeated experiments) were results pooled from three experiments (for details, see supplementary table 2, available as supplementary data at Rheumatology Online). Statistical differences between groups were determined by the non-parametric Mann–Whitney test, and the P-values indicated in the graphs.
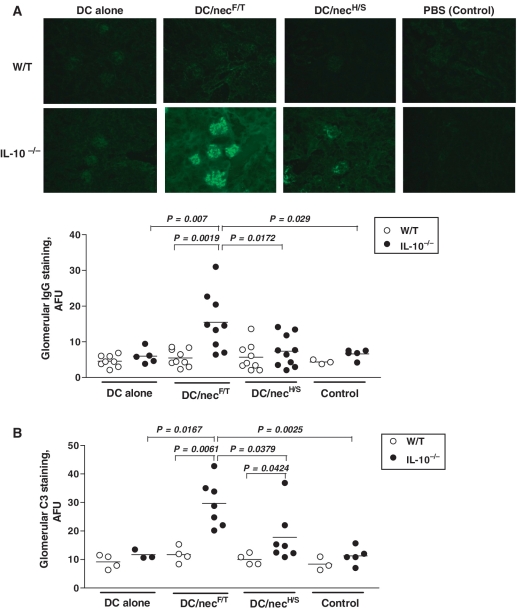
Lupus GN is typically characterized by IC deposition. To determine whether the glomerular lesions were mediated by ICs in the DC/necF/T-treated IL-10−/− mice, frozen kidney sections were assessed for IgG and C3 deposition by IF staining. Prominent glomerular IgG deposits were detected in the DC/necF/T-treated IL-10−/− mice (Fig. 2A, dot plots, n = 9). Although some glomerular IgG deposits were also detected in the IL-10−/− mice treated with DC/necH/S (n = 10) or DC alone (n = 5), the intensity of the staining was significantly lower when compared with DC/necF/T-treated IL-10−/− mice (Fig. 2A). No significant renal IgG deposition was observed in mice treated with NecF/T alone (data not shown). Similar findings were obtained for renal C3 deposition (Fig. 2B). Results from quantitative analysis also revealed that the intensity of C3 deposition in the DC/necF/T-treated IL-10−/− mice was the highest among all the experimental groups (Fig. 2B, dot plots). In contrast, and consistent with our previous findings [4], no or very low levels of glomerular IgG and C3 deposits were detected in the similarly treated W/T control groups (Fig. 2A and B, open circles). The quantitative measurements mentioned above were used where necessary for confirmation purpose. These were done on tissues from selective and representative experiments only. However, such quantification had been repeated at least once, on tissues from different experiments, with consistent results. The data shown were pooled from three (Figs 1A and 2A) and two (Fig. 2B) experiments, respectively. For CD68 staining, as a marker for macrophage infiltration, hence additional supporting evidence of renal inflammation, the kidney sections selected were the ones with high GN score (≥3).
Fig. 2.
Renal IC deposition: kidney tissue samples were from mice of the different treatment groups described in Fig. 1. Immunostaining for mouse IgG (A) and C3 (B) on frozen kidney sections revealed severe glomerular deposits but only in DC/necF/T-treated IL-10−/− mice. The scattered dot plots compare the relative levels of renal IgG and C3 deposits, as quantified by the IF method (see Materials and methods section). Each symbol represents a single mouse and the horizontal bars are the mean values for each of the four treatment groups of each mouse strain. In these experiments, the donor cells and recipient mice were of the same strain, i.e. W/T and IL-10−/− mice, respectively. Original magnification ×20 (A). The quantitative data (dot plots) shown in (A) and in (B) were results pooled from three or two repeated experiments, respectively (for details see supplementary tables 3 and 4, available as supplementary data at Rheumatology Online). Statistical differences between groups are determined by the non-parametric Mann–Whitney test, and the P-values indicated in the graphs. The results were also reanalysed and reconfirmed by the non-parametric one-way ANOVA test to be highly significant among all of the experimental groups (P < 0.0001); or among the four differently treated IL-10−/− (P < 0.01), but not W/T control (P > 0.05), groups of the same strain.
Serum ANA responses induced in DC/nec-treated mice
The presence of circulating autoantibodies against nuclear constituents, anti-dsDNA in particular, is the hallmark of lupus disease [1]. We have previously demonstrated that injection of syngenic DC/nec could induce strong anti-dsDNA antibody responses in both normal and lupus-prone mice, but disease induction was closely associated with the types or isotypes of the autoantibodies so induced [4]. To understand the mechanism underlying disease induction in IL-10−/− mice, serum levels of autoantibodies against a range of different classical lupus-associated nuclear antigens were quantified. Significant levels of anti-dsDNA IgG antibodies were detected in the sera of all DC-injected mouse groups, of both W/T and IL-10−/− strains. Surprisingly, the relative levels induced by DC/necF/T treatment in the IL-10−/− mice appeared to be similar, or even lower (e.g. for anti-histone and anti-SM/nRNP), when compared with those detected in the DC alone or DC/necH/S-treated IL-10−/−, or the corresponding W/T control, disease-free mice (supplementary figure 2, available as supplementary data at Rheumatology Online). The serum anti-dsDNA antibody levels in the mice treated with necrotic cells alone were also similar to those in the PBS-treated or untreated control mice (data not shown). In other words, no significant difference in the serum levels of anti-ssDNA, anti-chromatin, anti-histone and anti-SM/nRNP autoantibodies between the diseased and disease-free mouse groups was detected. Further isotypic analysis of the serum autoantibodies (supplementary figure 3, available as supplementary data at Rheumatology Online) and total IgG (data not shown) also failed to reveal any significant difference that could otherwise explain the disease induction in the DC/necF/T-treated IL-10−/− mice.
Characterization of renal reactive autoantibodies in DC/necF/T-treated IL-10−/− mice
There may be different explanations for the lack of correlation between the serum levels of autoantibodies quantified and the renal disease development observed above. For example, it could be due to different types or alternative specificities of the autoantibodies induced, or deposition of the pathogenic autoantibodies onto the tissues thus removing them from circulation.
To examine whether DC/necF/T treatment in IL-10−/− mice induced antibodies specific for renal antigens, the presence and levels of circulating renal reactive autoantibodies in these mice were also determined. For the immunoreactivity assay, frozen kidney tissue sections prepared from normal C57BL/6 mice were used as the substrate. The results showed that sera from DC/necF/T-treated IL-10−/− mice contained strong reactivities against antigens present in the normal kidneys, but primarily on the Bowman’s capsule and peritubular capillary network (supplementary figure 4, available as supplementary data at Rheumatology Online). Although the staining pattern was different from the typical glomerular staining observed on tissues taken directly from the diseased mice (Fig. 2A), these results together suggested that DC/necF/T treatment preferentially induced various nephrophilic autoantibodies in IL-10−/− (but not similarly treated W/T) mice, which could be responsible for causing the renal damage observed.
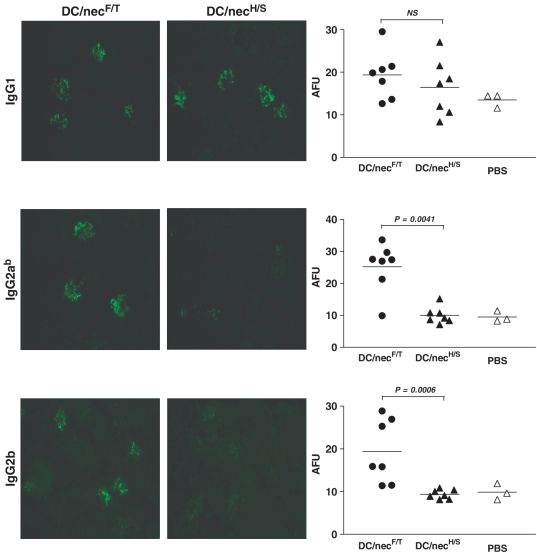
To determine whether certain pathogenic autoantibody subclasses were involved in the disease induction, isotypic analysis of the autoantibodies present in the affected kidneys was also carried out. In agreement with our previous findings in MRL mice [4], high frequencies and intensity of the IgG2ab (C57BL/6 allotype) and IgG2b-containing IC deposits, in addition to those of the IgG1 isotype, were detected but only in the glomeruli of DC/NecF/T-treated IL-10−/− mice (Fig. 3). This was in contrast to what was observed in the DC/NecH/S-treated group, being solely of the IgG1 subclass (Fig. 3, DC/NecH/S). Due to its long half life, high affinity towards complement binding and activation of certain Fcγ receptors, FcγIV in particular [34], the IgG2 subclass is believed to be the most pathogenic autoantibody isotype [35]. The results confirm therefore again the importance of Th1-mediated responses in the lupus disease induction [4], and that IL-10 plays an essential protective role in preventing the pathogenic nephrophilic autoantibody responses in the mice.
Fig. 3.
Characterization of renal reactive autoantibodies—isotypic analysis of the renal ICs. The antibody isotypes of renal Ig deposits in the DC/necF/T- and DC/necH/S-treated IL-10−/− mice were assessed by immunostaining using FITC-conjugated antibodies specific for mouse IgG1, IgG2ab (C57/BL6 allotype specific Abs) and IgG2b, respectively. Photomicrographs representative of each of the two treatment groups (DC/necF/T, DC/necH/S) are shown. The intensity of the staining was quantified (AFU, dot plots). Each symbol represents a single mouse, the horizontal bar is the mean value for each group, and the data shown were results pooled from two repeated experiments (for details see supplementary table 5, available as supplementary data at Rheumatology Online). Control group: PBS-treated mice. Original magnification: ×20; statistical differences between groups were determined by the non-parametric Mann–Whitney test, and the P-values indicated in the graphs.
A dominant role of host-derived IL-10 in controlling the pathogenic Th1 type of nephrophilic autoantibody response
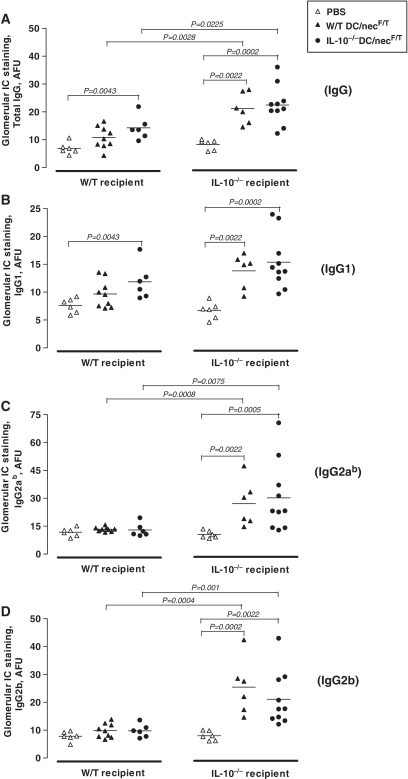
The above data point, therefore, to an essential role of IL-10 in controlling DC/nec-induced renal disease development. A further important question is about the source of IL-10, specifically refering to the role of DC-derived vs host-derived IL-10, in the mechanism underlying disease resistance. To verify this, W/T or IL-10−/− DCs pulsed with NecF/T were respectively and reciprocally injected into both W/T and IL-10−/− mice. As shown in Fig. 4, substantial glomerular IC deposits were evident in both groups of the IL-10−/− recipent mice injected with either W/T or IL-10−/− DC/necF/T. The frequencies of glomeruli positive for IgG (Fig. 4A), IgG1 (Fig. 4B), IgG2a (Fig. 4C) and IgG2b (Fig. 4D) were all significantly higher in the DC/necFT-treated IL-10−/− mice as compared with the PBS-treated control group. In the W/T recipient mouse groups however, although higher levels of IgG- and IgG1-positive glomeruli were also evident especially in the IL-10−/− DC/necF/T-treated group (Fig. 4A and B), only background levels of IgG2a- and IgG2b-positive glomeruli were detectable in the DC/necF/T-treated groups similar to those observed in the PBS-treated control mice (Fig. 4C and D). These further findings, therefore, indicated that host-derived endogenous IL-10 was primarily and overwhelmingly responsible for protection against the disease, possibly by affecting different stages of disease induction and development in our model. One possibility is that the injected DC could have been taken up by host DC to prime autoreactive T cells. However, considering the relatively very small number of DCs adoptively transferred per mouse against the number of host cells, which could potentially produce the cytokine (i.e. endogenous IL-10), it was not surprising to see that the differences between mice injected with W/TDC/nec and IL-10−/−DC/nec were small. Nevertheless, the level of IgG deposits did appear relatively higher in the IL-10−/−DC/nec-treated, as compared with the W/TDC/nec-treated, recipient mice of the same strain, indicating that the lack of DC-derived IL-10 could have also contributed to the disease induction possibly during the initiation phase.
Fig. 4.
A dominant role of host-derived IL-10 in controlling the pathogenic Th1 type of nephrophilic autoantibody responses. To further assess the roles of host vs DC-derived IL-10 on renal disease, recipient mice of the two mouse strains were injected with donor cells (DC/necF/T) from either W/T or IL-10−/− mice respectively and reciprocally. The animals were sacrificed 4 months after the first injection and kidney sections were stained for mouse IgG (A), IgG1 (B), IgG2ab (C) and IgG2b (D), and similarly quantified as described in Figs 2 and 3. The results shown are quantitative data (dot plots) pooled from three repeated experiments (for details see supplementary Table 6, available as supplementary data at Rheumatology Online). Statistical differences between groups were determined by the non-parametric Mann–Whitney test, and the P-values of groups having statistical significance are indicated in the graphs.
Discussion
Based on and continued from our previous work [4], the present study aimed to understand the immunological and molecular mechanisms underlying resistance vs susceptibility to lupus disease induction by DCs and dying cells. We have previously demonstrated how injection of DCs loaded with syngeneic necrotic cells could induce strong autoantibody responses leading to the development of a lupus-like disease in the susceptible MRL mouse strain. Intriguingly, similar treatment failed to induce disease expression in the C57BL/6 mouse strain despite high levels of circulating autoantibodies. A potent feedback regulatory mechanism, which could prevent the induction of pathogenic types of autoantibodies, appeared to be responsible for protecting these mice from the DC/nec-induced tissue pathology [4]. We demonstrated further here that, in the absence of IL-10, even mice with the resistant background could be rendered susceptible to the disease induction. Although full-blown lupus disease was not observed, these mice developed typical lupus-like IC-mediated renal end-organ damage, pointing to an essential protective role of IL-10 in the regulatory mechanism.
The key issues being addressed here are about the precise role of IL-10 in lupus pathogenesis, and how the present study in animal models may advance our understanding of the human disease. Although it is a general consensus that IL-10 is involved in lupus pathogenesis, with the IL-10 gene being in one of the lupus-susceptible loci identified on human chromosome 1 [36], the conclusions drawn from many previous studies have been conflicting as to whether IL-10 is playing a disease-protective or promoting role [21, 22, 24–30]. Serum IL-10 level is evidently elevated in SLE patients, which also correlates with disease activity [21, 22], but the question is about the true causal relationship. In mice, while some earlier work seemed to suggest a disease-promoting activity of IL-10 in the NZB/W mice [28], more recent findings including ours pointed to a potentially protective role of this very cytokine in MRL mouse strains [4, 29]. It is possible that the disease mechanisms, hence the roles of IL-10, may be very different in the two spontaneous lupus mouse models. However, the present study showing disease induction in mice with an otherwise non-autoimmune background (C57BL/6), yet deficient in their IL-10 gene, appears to suggest otherwise.
An alternative explanation is that IL-10 may play different roles depending on the stage of disease development. The cytokine is known to be functionally pleiotropic. While it suppresses Th1-mediated inflammatory responses, it can also promote B-cell growth and mediate antibody isotype switching (IgG1, IgG3, IgA1) [17]. The present study, by using IL-10-deficient mice on the resistant background, allowed us to elucidate the role of IL-10 particularly in the disease initiation phase. The findings are largely in agreement with what has been previously reported in lupus-prone MRL/lpr IL-10−/− mice [29], that IL-10 is protective at the early stage of disease development. However, the effects may be very different from those observed in lupus patients, who are understandably already in the established phase of clinical disease. Since IL-10 is a key cytokine that limits tissue injury during inflammatory responses [37], high levels of IL-10 detected in the SLE patients may be the result of tissue inflammation or a bystander product of chronic end-organ damage. Consequently, overt production of IL-10 may favour disease development by promoting autoreactive B-cell hyperactivity during the clinical stage of SLE [22, 23]. IL-10 could therefore have potentially conflicting dual effects on blocking disease induction vs late stage disease perpetuation.
Cytokines are, however, known to operate in a very complex regulatory network. The elevation of IL-10 expression, for example, was also found to be associated with increased IFN-γ expression in SLE patients [23, 38], and in NZB/W F1 lupus-prone mice [39]. In agreement with our previous findings in MRL mouse strains [4], the presence and deposition of certain pathogenic isotypes of the autoantibodies, i.e. IgG2a and IgG2b, which require IFN-γ for the isotype switch, was found to be closely associated with the development of glomerular C3 deposition and renal end-organ damage in DC/necF/T-treated IL-10−/−C57BL/6 mice. The low levels of IgG deposits detected in the kidneys of non-diseased IL-10−/− mice (e.g. DC/necH/S-treated group) could also be well explained by the presence of IgG1, yet lacking the more pathogenic IgG2a or IgG2b, isotype (Fig. 3), which also confirmed our previous findings in W/T mice [4].
Based on our observations, we conclude that IL-10 may protect against disease induction and development in different stages of the disease development. We showed here in vivo evidence that the endogenous host-derived IL-10 was most crucial (Fig. 4), whose presence could understandably have significant protective effects on different stages of disease development. By comparing directly DCs generated from W/T and IL-10−/− mice, however, we also tried to understand whether and how the DC-derived IL-10 could contribute to the protective mechanism particularly during the disease initiation phase. The pathogenic potential of NecF/T, in contrast to NecH/S, could be explained by the extent of necrotic cell death and the release of certain dying cell-derived soluble factors (Ling GS et al., data not published). Importantly, we demonstrated here that in the absence of IL-10, the NecF/T-conditioned DCs could act as a potent inducer of high levels of the pathogenic IgG2a/2b responses and deposits, which are a good marker of Th1 response, in the DC/necF/T-treated IL-10−/− mice (Figs 3, 4C and D). In contrast, the NecH/S-conditioned DCs induced a Th2 type of response against self-Ags, as illustrated by IgG1-only deposition in the kidneys. Such a response, although sustained well in the IL-10 deficient mice (Figs 3 and 4B), was not pathogenic enough to induce local inflammation (Fig. 1A and B). These findings are particularly important, because B-cell isotype switching, which takes place in the secondary lymphoid organs, is known to be T-cell dependent. Further studies should also be carried out in order to understand how local tissue-derived vs systemic IL-10 may be involved, or differentially involved, in controlling local pro-inflammatory responses responsible for the end-organ damage observed. Since IL-10 is best known for its potent immunosuppressive effects on Th1 responses, the enhanced IL-10 production and IL-10 promoter activity previously observed either in lupus patients or animal models may simply reflect a desperate but failed attempt of the immune system to down-regulate pathogenic Th1 responses. Clarification of such a causal relationship is thus vital in the rational design of therapeutic approaches for patients.
Lupus incidence requires complex interplay of multiple genetic and environmental factors [40]. Genetic background is also a crucial determinant of the severity and disease phenotype in murine lupus. For example, the MRL background contains the susceptibility loci necessary for full disease penetrance. Therefore, MRL mice homozygous for the lpr mutation of the Fas gene (MRL-lpr/lpr) or treated with DC/nec developed full-blown clinical lupus disease. In contrast, the same Fas mutation in mice with the the C57BL/6 background (B6/lpr), or being subjected to DC/nec insults, failed to develop clinical lupus disease [4, 41]. This variation in disease penetrance suggests that the C57BL/6 background contains lupus-resistance loci, which protect the mice from devastating organ injury, even when the peripheral tolerance mechanisms are disrupted. Although our data clearly demonstrate that IL-10 prevented the induction of lupus-like disease in C57BL/6 mice by dampening Th1 responses, full-blown expression of SLE was not observed in the DC/nec-treated IL-10−/−C57BL/6 mice, and none of the animals showed renal failure or lupus-related early mortality. Therefore, other intrinsic factor(s) in the C57BL/6 background that control disease development may also exist, which require further elucidation. The direct relevance of our findings to human SLE, and to other lupus mouse models, requires of course further studies. Nevertheless, the present study has provided evidence indicating an essential protective role of IL-10 against the induction and development of lupus disease, in mice with a non-autoimmune background in particular.

Supplementary data
Supplementary data are available at Rheumatology Online.
Acknowledgements
We wish to thank Dr Talat Malik, Dr Rose L Kirsten and Mr Chin-Kin Lo and many other colleagues for their assistance both experimentally and technically during the study. We would like to thank Dr Dan Chen (London School of Economics), Dr Andrea Cortini and Dr Talat Malik (Imperial College London) for their advice on statistical data analysis. We would also like to thank Dr A. Neil Barclay in the MRC Cellular Immunology Unit, Sir William Dunn School of Pathology, Oxford for the cytokines and antibody reagents provided. The authors declare no financial conflict of interest.
Funding: The work was supported by grants to FP Huang from the Hong Kong Research Grant Council (RGC, HKU7580/06M) and the Arthritis Research Campaign UK (arc UK, 18523).
Disclosure statement: The authors have declared no conflicts of interest.
References
- 1.Rahman A, Isenberg DA. Systemic lupus erythematosus. N Engl J Med. 2008;358:929–39. doi: 10.1056/NEJMra071297. [DOI] [PubMed] [Google Scholar]
- 2.Collings LA, Waters MFR, Poulter LW. The involvement of dendritic cells in the cutaneous lesions associated with tuberculoid and lepromatous leprosy. Clin Exp Immunol. 1985;62:458. [PMC free article] [PubMed] [Google Scholar]
- 3.Peng SL, Moslehi J, Craft J. Roles of interferon-gamma and interleukin-4 in murine lupus. J Clin Invest. 1997;99:1936–46. doi: 10.1172/JCI119361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ma L, Chan KW, Trendell-Smith NJ, et al. Systemic autoimmune disease induced by dendritic cells that have captured necrotic but not apoptotic cells in susceptible mouse strains. Eur J Immunol. 2005;35:3364–75. doi: 10.1002/eji.200535192. [DOI] [PubMed] [Google Scholar]
- 5.Peng SL, Szabo SJ, Glimcher LH. T-bet regulates IgG class switching and pathogenic autoantibody production. Proc Natl Acad Sci USA. 2002;99:5545–50. doi: 10.1073/pnas.082114899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baudino L, Azeredo da Silveira S, Nakata M, Izui S. Molecular and cellular basis for pathogenicity of autoantibodies: lessons from murine monoclonal autoantibodies. Springer Semin Immunopathol. 2006;28:175–84. doi: 10.1007/s00281-006-0037-0. [DOI] [PubMed] [Google Scholar]
- 7.Seery JP, Wang EC, Cattell V, Carroll JM, Owen MJ, Watt FM. A central role for alpha beta T cells in the pathogenesis of murine lupus. J Immunol. 1999;162:7241–8. [PubMed] [Google Scholar]
- 8.Herrmann M, Zoller OM, Hagenhofer M, Voll R, Kalden JR. What triggers anti-dsDNA antibodies? Mol Biol Rep. 1996;23:265–7. doi: 10.1007/BF00351179. [DOI] [PubMed] [Google Scholar]
- 9.Madaio MP, Hodder S, Schwartz RS, Stollar BD. Responsiveness of autoimmune and normal mice to nucleic acid antigens. J Immunol. 1984;132:872–6. [PubMed] [Google Scholar]
- 10.Wun HL, Leung DT, Wong KC, Chui YL, Lim PL. Molecular mimicry: anti-DNA antibodies may arise inadvertently as a response to antibodies generated to microorganisms. Int Immunol. 2001;13:1099–107. doi: 10.1093/intimm/13.9.1099. [DOI] [PubMed] [Google Scholar]
- 11.Isenberg D, Shoenfeld Y. Autoantibodies, idiotypes, anti-idiotypes and autoimmunity. Acta Haematol. 1986;76:95–100. doi: 10.1159/000206029. [DOI] [PubMed] [Google Scholar]
- 12.Frisoni L, McPhie L, Colonna L, et al. Nuclear autoantigen translocation and autoantibody opsonization lead to increased dendritic cell phagocytosis and presentation of nuclear antigens: a novel pathogenic pathway for autoimmunity? J Immunol. 2005;175:2692–701. doi: 10.4049/jimmunol.175.4.2692. [DOI] [PubMed] [Google Scholar]
- 13.Crispin JC, Martinez A, Alcocer-Varela J. Quantification of regulatory T cells in patients with systemic lupus erythematosus. J Autoimmun. 2003;21:273–6. doi: 10.1016/s0896-8411(03)00121-5. [DOI] [PubMed] [Google Scholar]
- 14.Fairhurst AM, Wandstrat AE, Wakeland EK. Systemic lupus erythematosus: multiple immunological phenotypes in a complex genetic disease. Adv Immunol. 2006;92:1–69. doi: 10.1016/S0065-2776(06)92001-X. [DOI] [PubMed] [Google Scholar]
- 15.Fiorentino DF, Zlotnik A, Vieira P, et al. IL-10 acts on the antigen-presenting cell to inhibit cytokine production by Th1 cells. J Immunol. 1991;146:3444–51. [PubMed] [Google Scholar]
- 16.Elenkov IJ, Chrousos GP. Stress hormones, proinflammatory and antiinflammatory cytokines, and autoimmunity. Ann NY Acad Sci. 2002;966:290–303. doi: 10.1111/j.1749-6632.2002.tb04229.x. [DOI] [PubMed] [Google Scholar]
- 17.Moore KW, de Waal Malefyt R, Coffman RL, O’Garra A. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol. 2001;19:683–765. doi: 10.1146/annurev.immunol.19.1.683. [DOI] [PubMed] [Google Scholar]
- 18.Corinti S, Albanesi C, la Sala A, Pastore S, Girolomoni G. Regulatory activity of autocrine IL-10 on dendritic cell functions. J Immunol. 2001;166:4312–8. doi: 10.4049/jimmunol.166.7.4312. [DOI] [PubMed] [Google Scholar]
- 19.Aletaha D, Landewe R, Karonitsch T, et al. Reporting disease activity in clinical trials of patients with rheumatoid arthritis: EULAR/ACR collaborative recommendations. Ann Rheum Dis. 2008;67:1360–4. doi: 10.1136/ard.2008.091454. [DOI] [PubMed] [Google Scholar]
- 20.Fedorak RN, Gangl A, Elson CO, et al. Recombinant human interleukin 10 in the treatment of patients with mild to moderately active Crohn’s disease. The Interleukin 10 Inflammatory Bowel Disease Cooperative Study Group. Gastroenterology. 2000;119:1473–82. doi: 10.1053/gast.2000.20229. [DOI] [PubMed] [Google Scholar]
- 21.Houssiau FA, Lefebvre C, Vanden Berghe M, Lambert M, Devogelaer JP, Renauld JC. Serum interleukin 10 titers in systemic lupus erythematosus reflect disease activity. Lupus. 1995;4:393–5. doi: 10.1177/096120339500400510. [DOI] [PubMed] [Google Scholar]
- 22.Llorente L, Richaud-Patin Y, Wijdenes J, et al. Spontaneous production of interleukin-10 by B lymphocytes and monocytes in systemic lupus erythematosus. Eur Cytokine Netw. 1993;4:421–7. [PubMed] [Google Scholar]
- 23.Csiszar A, Nagy G, Gergely P, Pozsonyi T, Pocsik E. Increased interferon-gamma (IFN-gamma), IL-10 and decreased IL-4 mRNA expression in peripheral blood mononuclear cells (PBMC) from patients with systemic lupus erythematosus (SLE) Clin Exp Immunol. 2000;122:464–70. doi: 10.1046/j.1365-2249.2000.01369.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Eskdale J, Wordsworth P, Bowman S, Field M, Gallagher G. Association between polymorphisms at the human IL-10 locus and systemic lupus erythematosus. Tissue Antigens. 1997;49:635–9. doi: 10.1111/j.1399-0039.1997.tb02812.x. [DOI] [PubMed] [Google Scholar]
- 25.Chong WP, Ip WK, Wong WH, Lau CS, Chan TM, Lau YL. Association of interleukin-10 promoter polymorphisms with systemic lupus erythematosus. Genes Immun. 2004;5:484–92. doi: 10.1038/sj.gene.6364119. [DOI] [PubMed] [Google Scholar]
- 26.Beebe AM, Cua DJ, de Waal Malefyt R. The role of interleukin-10 in autoimmune disease: systemic lupus erythematosus (SLE) and multiple sclerosis (MS) Cytokine Growth Factor Rev. 2002;13:403–12. doi: 10.1016/s1359-6101(02)00025-4. [DOI] [PubMed] [Google Scholar]
- 27.Llorente L, Richaud-Patin Y, Garcia-Padilla C, et al. Clinical and biologic effects of anti-interleukin-10 monoclonal antibody administration in systemic lupus erythematosus. Arthritis Rheum. 2000;43:1790–800. doi: 10.1002/1529-0131(200008)43:8<1790::AID-ANR15>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 28.Ishida H, Muchamuel T, Sakaguchi S, Andrade S, Menon S, Howard M. Continuous administration of anti-interleukin 10 antibodies delays onset of autoimmunity in NZB/W F1 mice. J Exp Med. 1994;179:305–10. doi: 10.1084/jem.179.1.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yin Z, Bahtiyar G, Zhang N, et al. IL-10 regulates murine lupus. J Immunol. 2002;169:2148–55. doi: 10.4049/jimmunol.169.4.2148. [DOI] [PubMed] [Google Scholar]
- 30.Blenman KR, Duan B, Xu Z, et al. IL-10 regulation of lupus in the NZM2410 murine model. Lab Invest. 2006;86:1136–48. doi: 10.1038/labinvest.3700468. [DOI] [PubMed] [Google Scholar]
- 31.Qi ZM, Wang J, Sun ZR, et al. Polymorphism of the mouse gene for the interleukin 10 receptor alpha chain (Il10ra) and its association with the autoimmune phenotype. Immunogenetics. 2005;57:697–702. doi: 10.1007/s00251-005-0036-7. [DOI] [PubMed] [Google Scholar]
- 32.Kuhn R, Lohler J, Rennick D, Rajewsky K, Muller W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75:263–74. doi: 10.1016/0092-8674(93)80068-p. [DOI] [PubMed] [Google Scholar]
- 33.Robson MG, Cook HT, Botto M, et al. Accelerated nephrotoxic nephritis is exacerbated in C1q-deficient mice. J Immunol. 2001;166:6820–8. doi: 10.4049/jimmunol.166.11.6820. [DOI] [PubMed] [Google Scholar]
- 34.Nimmerjahn F, Bruhns P, Horiuchi K, Ravetch JV. FcgammaRIV: a novel FcR with distinct IgG subclass specificity. Immunity. 2005;23:41–51. doi: 10.1016/j.immuni.2005.05.010. [DOI] [PubMed] [Google Scholar]
- 35.Theofilopoulos AN, Dixon FJ. Murine models of systemic lupus erythematosus. Adv Immunol. 1985;37:269–390. doi: 10.1016/s0065-2776(08)60342-9. [DOI] [PubMed] [Google Scholar]
- 36.Tsao BP. Lupus susceptibility genes on human chromosome 1. Int Rev Immunol. 2000;19:319–34. doi: 10.3109/08830180009055502. [DOI] [PubMed] [Google Scholar]
- 37.Mocellin S, Panelli MC, Wang E, Nagorsen D, Marincola FM. The dual role of IL-10. Trends Immunol. 2003;24:36–43. doi: 10.1016/s1471-4906(02)00009-1. [DOI] [PubMed] [Google Scholar]
- 38.Hagiwara E, Gourley MF, Lee S, Klinman DK. Disease severity in patients with systemic lupus erythematosus correlates with an increased ratio of interleukin-10: interferon-gamma-secreting cells in the peripheral blood. Arthritis Rheum. 1996;39:379–85. doi: 10.1002/art.1780390305. [DOI] [PubMed] [Google Scholar]
- 39.Enghard P, Langnickel D, Riemekasten G. T cell cytokine imbalance towards production of IFN-gamma and IL-10 in NZB/W F1 lupus-prone mice is associated with autoantibody levels and nephritis. Scand J Rheumatol. 2006;35:209–16. doi: 10.1080/03009740500417791. [DOI] [PubMed] [Google Scholar]
- 40.Rhodes B, Vyse TJ. General aspects of the genetics of SLE. Autoimmunity. 2007;40:550–9. doi: 10.1080/08916930701510657. [DOI] [PubMed] [Google Scholar]
- 41.Santiago-Raber ML, Laporte C, Reininger L, Izui S. Genetic basis of murine lupus. Autoimmun Rev. 2004;3:33–9. doi: 10.1016/S1568-9972(03)00062-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.