Highlights
► Novel whole brain sagittal organotypic vibrosection in vitro model. ► Survival of cholinergic and dopaminergic neurons. ► Capillaries in whole brain sections in vitro. ► Long nerve fiber growth with neurotracing (Mini-Ruby). ► Striato-nigral varicosities.
Keywords: Organotypic, Whole brain cultures, Dopaminergic, Cholinergic, Vascular
Abstract
In vitro cell culture models are of enormous importance in neuroscience research and organotypic brain slices are found to be a potent model very close to the in vivo situation. Brain slices can be cultured as single slices or as co-slices. However, there is need to culture whole brain sections, containing the complex functional architecture. The aim of the present study was to develop and characterize whole brain sagittal slice cultures (200 μm organotypic vibrosections) from postnatal day 8 rats. We show that sagittal vibrosections can be cultured for several weeks and they maintain survival of cholinergic and dopaminergic neurons, as well as a strong capillary network. Partly long-distance cortico-striatal and cortico-hippocampal nerve fibers were found using Mini-Ruby neurotracing. Dopaminergic nerve fibers extended from the mesencephalon, but in the striato-nigral tract and in the striatum only strong dense varicosities were found. The model also allows to study pathological triggers, such as e.g. hydrogen peroxide markedly increased propidiumiodide-positive nuclei in the hippocampus. In conclusion, our novel model provides an easy potent whole sagittal brain culture system that allows to study cholinergic and dopaminergic neurons together but also in close interaction with all other cells of the brain and with capillaries. It will be a great challenge in future to use this model to re-construct whole pathways. This vibrosection model may partly represent a close adult in vivo situation, which allows to study neurodegeneration and neuroprotection of cholinergic and dopaminergic neurons, which plays an important role in Alzheimer's and Parkinson's disease, respectively.
1. Introduction
In vitro cell culture models are of particular importance in Neuroscience Research. While single cell cultures are widely used, the organotypic brain slice model is so far the best model being closest to an in vivo situation. The organotypic brain slice contains all cells present in the brain. Hence, the individual cells are arranged in close contact and do not lose density-dependent regulatory mechanisms, three-dimensional architecture as well as tissue-specific transport and diffusion probabilities. The slice model allows to maintain the survival of different cell types, the cytoarchitecture of the tissue and the connections between cells and neuronal properties. The basic cellular and connective organization of the donor brain regions are well preserved, thus the slice cultures provide an easily accessible experimental model for studies of toxic, degenerative and plastic regenerative or developmental changes in the brain (Gähwiler et al., 1997; Lossi et al., 2009).
The organotypic brain slice model has been introduced by Crain et al. (1982) using spinal cord-dorsal root ganglia and was optimized and characterized by Gähwiler and Hefti (1984) as roller tube cultures. The technique was modified as a permanent culture model on membrane inserts (Stoppini et al., 1991; Buchs et al., 1993) and is meanwhile used by several research groups (Ostergaard et al., 1990; Ostergaard, 1993; Gähwiler et al., 1990; Robertson et al., 1997). Slices can be cultured as single slices from a respective brain area, or as a co-culture where two functional related brain slices are connected. The culturing of organotypic brain slices on membranes is well established in our research group (Humpel et al., 1995; Schatz et al., 1999, 2000; Weis et al., 2001; Humpel and Weis, 2002). We have strong experience in culturing mesencephalic dopaminergic neurons (Schatz et al., 1999), cholinergic basal nucleus of Meynert (nBM) neurons (Weis et al., 2001), dorsal raphe serotonergic neurons (Hochstrasser et al., 2011), or cells from cortical or hippocampal slices. Moreover, co-cultures of the nBM together with the cortex (Humpel and Weis, 2002) and the ventral mesencephalon (vMes) together with the dorsal striatum (dStr) (Schatz et al., 1999) provide an excellent system to study interaction between two brain regions. Recently, we also improved this slice model and provided a large slice containing 4 functionally related brain areas (Ullrich and Humpel, 2009a,b). Such slices allow to study developmental aspects, survival of neurons, neurodegeneration or the effect of toxins or trophic molecules.
However, a disadvantage of single organotypic brain slices is the loss of interaction between different neuronal/astroglial systems and capillaries of the whole brain structure. In the present study we aim to develop and characterize whole brain sagittal organotypic vibrosection cultures to culture dopaminergic and cholinergic neurons together. Thus, we demonstrate the culture of whole brain sagittal vibrosections from postnatal day 8 rats which maintain dopaminergic and cholinergic neurons, as well as a complex capillary network and long nerve fiber growth.
2. Material and methods
2.1. Organotypic vibrosection cultures
For vibrosection cultures postnatal day 8 Sprague Dawley rats were used. All experiments conformed to Austrian guidelines on the ethical use of animals and all efforts were made to minimize the number of animals used and their suffering. The animals were rapidly sacrificed, the brains dissected and sagittally cut (Fig. 1A and F). The brains were glued (Glue Loctite 404 or 401) onto the chuck of a water cooled vibratome Leica VT1000A, and triggered close to a commercial shave racer (Fig. 1B). Under aseptic conditions, 200 μm vibrosections were cut and collected in sterile medium. The organotypic vibrosections were carefully placed onto a sterile 0.4 μm pore membrane (Millipore HTTP02500) (Fig. 1C and E), which was then placed into a 0.4 μm membrane insert (Millipore PICM03050) within a 6-well plate (Fig. 1D). Vibrosections were cultured in 6-well plates (Greiner) at 37 °C and 5% CO2 with 1.2 ml/well of the following culture medium: 50% MEM/HEPES (Gibco), 25% heat inactivated horse serum (Gibco/Lifetech, Austria), 25% Hanks’ solution (Gibco), 2 mM NaHCO3 (Merck, Austria), 6.5 mg/ml glucose (Merck, Germany), 2 mM glutamine (Merck, Germany), pH 7.2. Vibrosections were incubated for 2 weeks and medium was changed twice a week. The sections attach to the membranes after 2 weeks in vitro. Vibrosections were cultured with or without 10 ng/ml nerve growth factor (NGF) and/or glial cell line-derived neurotrophic factor (GDNF). At the end of the experiment, vibrosections were fixed for 3 h at 4 °C in 4% paraformaldehyde (PAF)/10 mM phosphate buffered saline (PBS) and then stored at 4 °C in PBS until use.
Fig. 1.
Schematic of the preparation of whole brain sagittal organotypic vibrosections. Postnatal day 8 rats were dissected sagittally (A) and fixed at the vibratome close to a razor knife (B). A semipermeable 0.4 μm pore membrane was prepared (C, arrow) and a sectioned organotypic vibrosection was carefully placed on this membrane and put into a semipermeable membrane insert (D), and incubated in a 6-well plate at 37 °C and 5% CO2 (E). (F) shows a cresyl violet stained overview of a whole sagittal brain organotypic vibrosection cultured for 2 weeks. Scale bar = 2600 μm.
2.2. Immunohistochemistry
Immunohistochemistry was performed as previously described (Weis et al., 2001). All incubations were performed free floating for 2 days including 0.1% Triton, such that the antibodies can penetrate from both sides into the vibrosections. Vibrosections were washed 30 min with 0.1% Triton/PBS (T-PBS) at room temperature and pre-treated 20 min with 20% methanol/1% H2O2/PBS. After thorough rinsing, the vibrosections were blocked with 20% horse serum/0.2% BSA/T-PBS and then incubated for 2 days at 4 °C with primary antibodies against choline acetyltransferase (ChAT, 1:750, Millipore AB144P, raised against human placental enzyme), tyrosine hydroxylase (TH, 1:500, Millipore AB152, from rat pheochromocytoma) or laminin (1:500, Sigma L9393, purified from the basement membrane of Englebreth Holm-Swarm mouse sarcoma) or calbindin (1:200, abcam ab66185, raised against full length purified native cow protein). Then the vibrosections were washed again with PBS and incubated with secondary biotinylated anti-goat (ChAT) or anti-rabbit (laminin, TH) or anti-mouse (Calbindin) antibodies (1:200, Vector Lab., USA) for 1 h at room temperature. Following further washing steps with PBS, vibrosections were incubated in an avidin-biotin complex solution (ABC-Elite Vectastain reagent Vector Lab.) for 1 h. After being washed with 50 mM Tris-buffered saline (TBS), the signal was detected by using 0.5 mg/ml 3,3′-diaminobenzidine (DAB) including 0.003% H2O2 as a substrate in Tris-buffered saline. The vibrosections were mounted on glass slides, air dried and coverslipped with Entellan (Merck, Germany). Unspecific staining was defined by omitting the primary antibody. For fluorescence immunohistochemistry the methanol pre-treatment was omitted and as a secondary antibody Alexa-488 (Invitrogen, 1:400) was used. To label nuclei, organotypic vibrosections were incubated with 4,6-diamidino-2-phenylindole (DAPI, 1:10,000; Sigma) for 30 min. Immunolabelling was visualized with a Leica DMIRB fluorescence inverse microscope equipped with an Apple computer and Openlab software.
2.3. Triple immunohistochemical staining
Organotypic vibrosections were first processed for ChAT immunohistochemistry and stained with DAB-nickel giving a black color. Sections were then washed in PBS, blocked with the avidin/biotin blocking kit (Vector SP-2001), and then processed for TH immunohistochemistry. Dopaminergic neurons were stained with Vector SG substrate (SK-470) giving a grey color. Sections were then again washed in PBS and blocked with the avidin/biotin blocking kit and were then processed for calbindin immunohistochemistry and stained with the Vector VIP substrate (SK-4600) giving a violet color.
2.4. Propidiumiodide staining
To analyze cell death, organotypic vibrosections were treated with or without 450 mM H2O2 for 3 days, then incubated for 30 min at 37 °C with 2 μg/ml propidiumiodide (PI), washed with PBS and stained with DAPI for 20 min (Hochstrasser et al., 2011). After washing, sections were immediately analyzed under a fluorescence microscope, then fixed and further processed.
2.5. In situ hybridization
In situ hybridization was performed as described (Humpel et al., 1993). As a control, rats were decapitated and the brains frozen, sectioned (14 μm) with a cryostat (Leica) and thawed onto slides (ProbeOn™ slides, Fisher Biotech, USA). Organotypic vibrosections were taken fresh, postfixed in 4% paraformaldehyde for 15 min at 4 °C, washed 3 × 5 min in 0.1 M autoclaved PBS, incubated in 100% ethanol for 5 min and then airdried. Antisense oligonucleotides complementary to the ChAT gene were labelled at the 3′ end with [α-35S]dATP using terminal deoxyribonucleotidyl transferase (NEN, DuPont, Austria) and purified on Quiagen columns. Brains/vibrosections were hybridized at 42 °C overnight in a humidified chamber with 75 μl per section of the hybridization solution (50% formamide, 4xSSC, 0.02% polyvinyl-pyrrolidone, 0.02% Ficoll, 0.02% bovine serum albumin, 10% dextrane sulfate, 0.5 mg/ml sheared salmon sperm DNA, 1% sarcosyl (N-lauroyl sarcosine), 0.02 M NaPO4 (pH 7.0), 50 mM dithiotreitol) containing 1 × 107 CPM/ml probe. Sections were subsequently rinsed, washed four times (15 min each) at 54 °C in 1xSSC, cooled to room temperature, dehydrated through 70%, 90% and 99% ethanol and air dried. Sections were exposed to X-ray film for 1 week or dipped in Kodak NTB-2 photo emulsion, exposed for 4 weeks at −20 °C, developed, fixed, and lightly counterstained with cresyl violet. Sections were analyzed using light and dark-field microscopy and photographed.
2.6. Acetylcholinesterase (AChE) histochemistry
Vibrosections were subjected to AChE-histochemistry as reported earlier (Humpel and Weis, 2002). Briefly, vibrosections were washed 3 × 10 min in 100 mM maleate buffer (pH 6.0) and then incubated in a fresh solution of 0.03 mM acetylthiocholine iodide, 0.05 mM K3Fe(CN)6(III), 0.3 mM CuSO4, 0.05 mM sodium citrate in 100 mM maleate (pH 6.0) for 1.5 h. Sections were then thoroughly rinsed in PBS and developed in a 0.04% DAB/0.3% nickel ammonium sulfate/0.003% H2O2/Tris–HCl pH 7.6 solution for 20 min. Sections were then postfixed for 3 h with 4%PAF and mounted on glass slides.
2.7. Mini-Ruby tracing
After 2 weeks in culture, a few crystals of Mini-Ruby (Invitrogen, D3312) were placed directly onto the surface of the sections (cortex or striatum or vMes) using the tip of a small needle as described in detail by us (Ullrich and Humpel, in press). The sections were incubated for 3 h or 3 days, then fixed in 4% PAF for 3 h and then transferred to glass slides. Sections were observed under the fluorescence microscope using a Y3 filter (excitation 535/50 nm, emission 610/75 nm). Some vibrosections were also subjected to TH immunohistochemistry using Alexa 488 antibodies (Invitrogen).
2.8. Analysis, quantification and statistics
The number of microscopically detectable immunoreactive neurons was counted in the striatum, nBM or vMes and visualized under a 20× objective. The areas were identified by the respective immunohistochemical stainings. Multistatistical analysis was obtained by one way ANOVA, followed by Fisher PLSD posthoc test by comparing controls against the respective treatments, where p < 0.05 represents statistical significance.
3. Results
3.1. Cholinergic neurons
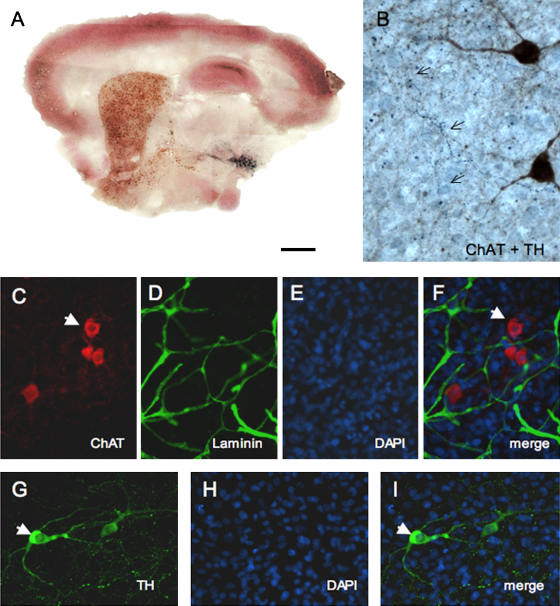
Whole brain organotypic vibrosections were incubated with NGF for 2 weeks and were strongly stained by immunohistochemistry for ChAT (Fig. 2A). Large cholinergic neurons were found in the basal nucleus of Meynert (Table 1; Fig. 2B and C) and smaller cholinergic interneurons were seen in the whole striatum (Table 1; Fig. 2D). ChAT+ like immunoreactivity was strongly seen in the whole cortex and hippocampus (Fig. 2A and F). ChAT+ neurons were also identified by fluorescence immunohistochemistry and nuclear DAPI revealed the cytoplasmic staining (Fig. 5C–F). A strong AChE+ fiber network was found in the hippocampus (Fig. 2F) and the cortex (Fig. 2G). The cholinergic nerve fibers were strongly stained and also displayed several intensive varicosities being either ChAT+ (Fig. 2B) or AChE+ (Fig. 2G). Some vibrosections were incubated for up to 12 weeks with NGF and did not differ in their expression of cholinergic neurons (data not shown). The addition of recombinant NGF was important, because sections cultured without NGF contained nearly no ChAT+ nBM neurons (Table 1), which was significantly increased when NGF was added (Table 1). In the striatum several ChAT+ neurons were detectable without NGF, but again the number increased when sections were incubated with NGF (Table 1). The number of ChAT+ nBM and striatal neurons was doubled when vibrosections were incubated with both NGF and GDNF (Table 1).
Fig. 2.
Immunohistochemistry for cholinergic neurons stained by choline acetyltransferase (ChAT, A–E) and histochemistry for acetylcholinesterase (AChE, F–G). (A) shows an overview of a whole brain organotypic vibrosection cultured for 2 weeks with nerve growth factor. Cholinergic neurons were nicely stained in the basal nucleus of Meynert (nBM; A–C) and striatum (str, A and D). Note enhanced ChAT-like immunoreactivity in cortex (A) and hippocampus (hip, A and E). AChE-positive nerve fibers were strongly stained in the hippocampus (F) and in cortex (ctx, G). (B) and (G) show a cholinergic ChAT+ (B) or AChE+ (G) nerve fiber with large intensive varicosities (arrows). Scale bar in (A) = 1600 μm, 35 μm (B and G), 170 μm (C and D), and 120 μm (E and F).
Table 1.
Number of cholinergic and dopaminergic neurons in sagittal vibrosections after 2 weeks in culture.
| nBM (ChAT+) | Str (ChAT+) | vMes (TH+) | |
|---|---|---|---|
| Minus | 2 ± 1 (5) | 30 ± 5 (5) | 40 ± 21 (5) |
| NGF | 81 ± 15 (5)*** | 121 ± 22 (5) p = 0.07 | 40 ± 14 (5) ns |
| GDNF | 3 ± 0.2 (5) ns | 41 ± 6 (5) ns | 65 ± 26 (5)** |
| NGF + GDNF | 165 ± 11 (25)*** | 382 ± 23 (26)*** | 158 ± 24 (15)** |
Sagittal vibrosections were cultured for 2 weeks with or without 10 ng/ml nerve growth factor (NGF) or glial cell-line derived neurotrophic factor (GDNF) or combinations, then stained for choline acetyltransferase (ChAT+) or tyrosine hydroxylase (TH+) and the number of neurons was counted in the basal nucleus of Meynert (nBM), striatum (Str) or ventral mesencephalon (vMes). Values are given as mean ± SEM neurons/brain area, the values in parenthesis give the number of analyzed vibrosections. Statistical analysis was performed by one way ANOVA with a subsequent Fisher PLSD posthoc test; ns, not significant.
p < 0.01.
p < 0.001.
Fig. 5.
Co-localization of dopaminergic and cholinergic neurons and capillaries in organotypic sagittal vibrosections using chromogenic substrate (A and B) or fluorescence labelling (C–I). (A) shows a triple immunohistochemical staining for cholinergic neurons (DAB-nickel, black), dopaminergic neurons (SG, grey) and calbindin (VIP, violet). (B) shows TH+ nerve fibers (arrows, blue) crossing ChAT+ neurons (brown) in the basal nucleus of Meynert. (C)–(F) show co-localization of cholinergic ChAT+ neurons (arrow, C) with laminin+ capillaries (D). (G)–(I) show cytoplasmic staining of TH+ dopaminergic neurons (arrow, G). Nuclei were stained with DAPI (E, H). (F) and (I) show the merged pictures. Scale bar in (A) = 1200 μm, 30 μm (B) and 60 μm (C–I).
3.2. Dopaminergic neurons
Dopaminergic neurons in whole brain organotypic vibrosections were incubated with GDNF for 2 weeks and were stained by immunohistochemistry for tyrosine hydroxylase (Fig. 3A). A large number of TH+ neurons (Table 1) were seen in the ventral mesencephalon (Fig. 3A–C) with intensive varicosities (Fig. 3B) and only a few TH+ neurons were found in the cortex (Fig. 3J). The TH+ positive nerve fibers extended from the vMes to the striatum (Fig. 3C), however, after approx. 1 mm the nerve fibers were not clearly to follow up (Fig. 3D, G, and H), but instead several TH+ positive varicosities were seen (Fig. 3D and I). These TH+ varicosities extended along the striato-nigral pathway (Fig. 3E, H, and I) and were also seen in the striatum (Fig. 3F). Partly some TH+ nerve fibers were seen crossing the nBM (Fig. 5B). TH+ neurons were also identified by fluorescence immunohistochemistry and nuclear DAPI revealed the cytoplasmic staining (Fig. 5G–I). Some sections were cultured for up to 12 weeks with GDNF and did not differ in their expression of dopaminergic neurons (data not shown). When vibrosections were incubated without recombinant GDNF approx. 40 TH+ neurons were found, which was slightly increased when incubated with GDNF (Table 1). Again the number of TH+ vMes neurons was markedly enhanced when vibrosections were incubated with both NGF and GDNF together (Table 1).
Fig. 3.
Immunohistochemistry for dopaminergic neurons stained by tyrosine hydroxylase (TH). (A) shows an overview of a whole brain sagittal organotypic vibrosection cultured for 2 weeks with glial cell-line derived neurotrophic factor. Dopaminergic neurons were nicely stained in the ventral mesencephalon (vMes, A–C) and displayed strong intensive varicosities (B, arrows). The striato-nigral TH+ positive nerve fibers projected from the vMes to the striatum (C), and were visible as long nerve fibers and as strong varicosities approx. 1 mm apart (D and G, box (I) in A). Along the striato-nigral pathway only a few nerve fibers were found and a high density of strong TH+ varicosities was seen (E and H, box (II) in A). In the striatum no nerve fibers but a high density of TH+ varicosities was visible (F and I, box (III) in A). Some few TH+ neurons were also found in the cortex (J). Scale bar in (A) = 1700 μm, 60 μm (B), 870 μm (C), 65 μm (D–F), 13 μm (G–I) and 140 μm (J).
3.3. Brain capillaries
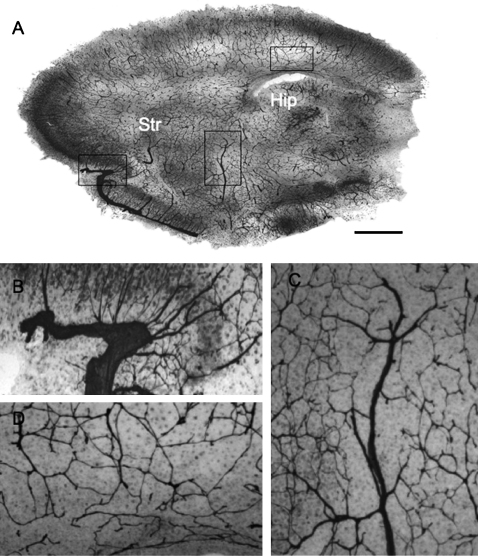
Brain capillaries were stained by laminin immunohistochemistry and were distributed all over the organotypic vibrosections cultured for 2 weeks (Fig. 4A). Strong large brain vessels (Fig. 4B and C) as well as small fine capillary processes were identified (Fig. 4D). Laminin+ capillaries were identified by fluorescence immunohistochemistry and were distributed all over the organotypic vibrosection, also in closer connection to cholinergic neurons (Fig. 5B–D).
Fig. 4.
Immunohistochemistry for vascular brain capillaries stained by laminin. (A) shows an overview of a whole brain organotypic vibrosection cultured for 2 weeks. (B)–(D) show higher magnifications from (A), as outlined in boxes, showing large brain capillaries at the brain border (B), within the brain (C) or a dense small capillary network (D). Str, striatum; Hip, hippocampus. Scale bar in (A) = 1600 μm, 200 μm (B), 340 μm (C), and 280 μm (D).
3.4. In situ hybridization
In situ hybridization showed clear ChAT mRNA expression in adult rats in the striatum and nBM (Fig. 6A) or septum (Fig. 6B). When organotypic vibrosections were processed for ChAT mRNA expression, a distinct staining was observed in the same areas (striatum, nBM) (Fig. 6C). Dark field microscopy showed that the black grains were directly located over cresyl violet counterstained cells (Fig. 6D).
Fig. 6.
In situ hybridization for cholinergic neurons (ChAT mRNA) in adult brains (A, B, and D) and in 2-week old organotypic vibrosections incubated with NGF (C). (A) and (C) show X-ray films exposed for 1 week, and (B) shows a dipped slide at dark-field and (D) at bright field. Note black silver grains over cresyl violet counterstained cells (D). Str, striatum; nBM, basal nucleus of Meynert. Scale bar in (A) = 2000 μm, 700 μm (B), 3000 μm (C), 85 μm (D).
3.5. Effects of hydrogen peroxide
In order to test organotypic vibrosections for a pathological dysfunction, we exposed the organotypic vibrosections to 450 mM H2O2 for 3 days. Clear DAPI positive nuclei were found in both controls and hydrogen peroxide treated organotypic vibrosections (Fig. 7A and D). However, while nearly no propidiumiodide+ nuclei were seen in controls, there was a dramatically enhanced number of propidiumiodide+ nuclei in hydrogen peroxide-exposed organotypic vibrosections in the hippocampus (Fig. 7B and E). Control organotypic vibrosections displayed strong calbindin staining in hippocampus (Figs. 1G and C and 7), while in hydrogen peroxide treated organotypic vibrosections the immunostaining for calbindin was markedly reduced (Fig. 7F).
Fig. 7.
H2O2-induced (450 mM for 3 days) alterations in the hippocampus of organotypic sagittal vibrosections stained for DAPI (A and D), propidiumiodide (B and E; PI) and calbindin (C and F). Note enhanced PI staining and reduced calbindin staining after hydrogen peroxide. DG, dentate gyrus; CA1, pyramidal cell layer CA1. Scale bar in (F) = 500 μm.
3.6. Mini-Ruby positive neurotracing
When Mini-Ruby was applied onto the striatum (Fig. 8A), several nerve fibers were seen projecting caudal to the mesencephalon (Fig. 8B), however, the fibers did not reach from the striatum to the mesencephalon. However, a long cortico-striatal nerve fiber was seen after application onto the striatum (Fig. 8C). When Mini-Ruby was applied onto the cortex, a long cortico-hippocampal fiber could be followed (Fig. 8D). When Mini-Ruby was applied onto the mesencephalon, several nerve fibers projecting frontal to the striatum were observed (Fig. 8E), and a few of them also co-localized with TH immunoreactivity representing dopaminergic nerve fibers (Fig. 8F–H).
Fig. 8.
Mini-Ruby neurotracing (red) of 2-week old vibrosections. Mini-Ruby was applied (*) either onto the striatum (A–C), or the cortex (D) or the ventral mesencephalon (vMes, E–H) and slices were analyzed after 3 h. (A) shows a co-staining of Mini-Ruby labelled cells in the striatum (red) and TH+ neurons in the vMes (green). (B) shows Mini-Ruby positive striatal nerve fibers growing caudal. (C) shows a large Mini-Ruby positive cortico-striatal neuron (arrows) and (D) shows a large Mini-Ruby labelled cortico-hippocampal neuron (arrows). (E)–(H) show mesencephalic Mini-Ruby positive cells and nerve fibers (E). The box in E shows co-localization of Mini-Ruby with tyrosine hydroxylase (TH) (see F–H). Note a TH+ nerve fiber (F, green, arrow), which co-localizes (H) with a Mini-Ruby labelled nerve fiber (G, red). Scale bar in A = 1170 μm (A), 290 μm (B), 185 μm (C), 160 μm (D), 200 μm (E–H).
4. Discussion
The present study demonstrates, that whole brain sagittal vibrosections can be cultured for several weeks from postnatal rats and maintain survival of cholinergic and dopaminergic neurons, as well as a strong capillary network, including long nerve fiber growth.
Cholinergic neurons in vibroslices. Cell death of cholinergic neurons is the central hallmark in Alzheimer's disease. Cholinergic neurons are located in distinct areas in the brain and neurons located in the septum/diagonal band of Broca project to the hippocampus, while neurons located in the basal nucleus of Meynert innervate the whole cortex. In the striatum the cholinergic neurons are mainly large interneurons. Already in 1983, Keller et al. (1983) reported that AChE histochemistry shows the presence of cholinergic cells and nerve fibers in organotypic cultures of septum and hippocampus. It is well established that forebrain cholinergic neurons develop axons that rapidly grow into the cerebral cortex in postnatal rat organotypic cultures (Baratta et al., 1996). We have also extensively studied cholinergic neurons in single organotypic brain slices of the striatum and nBM (Humpel et al., 1995; Humpel and Weis, 2002; Weis et al., 2001). The enzyme ChAT can be used as a key enzyme to label cholinergic neurons and the number of ChAT+ neurons strongly correlates with the survival rate. We have previously shown, that cholinergic neurons survive in co-slices of the nBM and cortex and cholinergic (AChE+) nerve fibers grew into the cortex (Humpel and Weis, 2002). We are aware of one study where horizontal organotypic slices from postnatal days 5 rats have been prepared and cholinergic and dopaminergic neurons were characterized (Thomas et al., 1998). Our present study shows now for the first time, culturing of sagittal vibrosections and a detailed characterization. For technical reasons sagittal slices were sectioned more laterally, thus no septal cholinergic neurons were cultured. The survival of the striatal as well as nBM neurons in the presence of NGF was very high and did not differ from previous studies.
Dopaminergic neurons in vibroslices. Cell death of dopaminergic neurons is the central hallmark in Parkinson‘s disease. Dopaminergic neurons are located in the ventral mesencephalon and neurons of the substantia nigra project into the dorsal striatum (nigro-striatal pathway), while neurons of the ventral tegmental area project to the ventral striatum (meso-limbic pathway). Organotypic brain slices of the vMes and striatum are well established and there are several exciting papers describing the nigro-striatal pathway in slices. Franke et al. (2003) reported that in vMes/striatum co-slices an extensive fiber bridge was observed in the co-cultures, and that TH+ neurons develop their typical innervation pattern. Snyder-Keller et al. (2001) showed that the striatal patch/matrix organization was maintained in organotypic slice cultures taken from E19-P4 rats. We ourselves showed in previous work, that co-slices of the vMes-striatum exhibit a large number of surviving dopamine neurons in the presence of GDNF and that an intense fiber innervation is seen in striatal slices (Schatz et al., 1999; Zassler et al., 2003, 2005). Similarly to ChAT, we have used the well established key enzyme TH to label dopaminergic neurons (Schatz et al., 1999). In the present study we show for the first time that dopamine neurons survive in sagittal vibrosections and can be cultured together with striatal and nBM cholinergic neurons. The number of dopamine neurons incubated with GDNF in vibrosections was not different to neurons found in single slices.
Role of growth factors NGF and GDNF. The addition of NGF or GDNF is necessary in this system, because the slices do not produce enough endogenous growth factors to support the survival of the neurons. We have previously shown that slices contain less than 3 pg NGF/mg tissue, which is insufficient to support the cholinergic phenotype (Weis et al., 2001). In addition, our previous studies have shown that GDNF supports the survival of dopaminergic neurons in our organotypic brain slices (Schatz et al., 1999). It is well established that GDNF increases the survival of dopamine neurons and also enhances the number of dopaminergic processes innervating the striatal and cortical but not hippocampal brain slices (Jaumotte and Zigmond, 2005). In order to investigate the number of cholinergic and dopaminergic neurons with or without these two growth factors, we performed experiments, and show that cholinergic nBM and less striatal neurons are highly dependent on NGF. Dopaminergic neurons in the vMes also survived well, however, their number increased when sections were incubated with GDNF. Interestingly, the combination of NGF and GDNF markedly increased the survival of cholinergic and dopaminergic neurons, clearly pointing that a complex interaction of the neurons is necessary for their survival. Such an interaction can efficiently be studied in our sagittal vibrosection model.
Age of the pups. Normally organotypic brain slices are taken from postnatal rats, however, also embryonic stages have been reported. Plenz and Kitai (1996) developed cortex-striatum-vMes (triple) organotypic cultures from rat postnatal days 0–2 brain and modified the “roller tube technique” by embedding slices in a plasma/thrombin clot onto a Millicell membrane on a coverslip. Organotypic slice cultures from vMes, striatum, hippocampus and cerebellum were prepared from late fetal (E21) to P7 rats and cultured for 3–60 days by the roller tube technique (Ostergaard et al., 1990). The aim for our group is to develop a model which more likely reflects the adult and not the developing situation and which is very close to in vivo. Thus a dissection stage of older animals has the big advantage that the main pathways are fully developed. Of course, it would be the best way to culture organotypic brain slices from adult rats, however, so far only acute slices of adult rats can be cultured for a few hours. Similarly, culturing of slices older as postnatal days 12–14 is not possible. We noticed that this is highly dependent on the properties of the slices to flatten during culturing, and older slices do not easily flatten. This is a real obstacle because also thinner vibrosections of older animals do not exhibit good results so far. For these reasons, we use postnatal day 8 rats, which give us good results for neuronal survival, and also represents a nearly fully developed neuronal network. Such cultures can be easily cultured for several weeks (up to 12 weeks in this study), but can be cultured even longer, as shown in a previous study e.g. for up to 50 months (Marksteiner and Humpel, 2008). Such a long-time culture model may provide a near adult in vivo situation.
Neuronal connectivity. One important reason to culture whole brain slices is to study and characterize the long-distance nerve fiber growth and connectivity between neuronal populations and brain areas. In our whole brain sagittal vibrosections we found cholinergic nerve fibers in the hippocampus and cortex stained by ChAT and AChE. However, only a limited number of nerve fibers could be followed back to the nBM. In fact, we found several long-distance nerve fibers e.g. between the striatum and the cortex or the hippocampus which could be followed up after Mini-Ruby tracing. The organotypic brain slice model is a 3-dimensional model and it is evident that nerve fibers can only be followed up, if they extend close to the surface of the brain sections. More advanced 2-photon-confocal microscopy may help to visualize and follow up also nerve fibers deeper in the brain slice. Another technical problem could also be that the tracer or the antibodies do not penetrate deeper into the slices, and limit the stainings of nerve fiber tracts.
The striato-nigral tract is of special interest, because it degenerates in Parkinson's disease. It has been reported that outgrowth of dopamine fibers from the vMes occurred irrespective of the age of the donor of rats, and a pronounced innervation of dopamine nerve fibers into the striatum has been seen (Ostergaard et al., 1990). The distance between the vMes and striatum was between 0.5 and 2.0 mm at the end of the culturing, thus the TH+ fibers from the vMes could extend over a long distance and it was reported that the maximum distance covered between vMes-striatum co-slices was 5.7 mm (Ostergaard et al., 1990). In our whole brain sagittal vibrosection model, we could clearly stain dopaminergic neurons and nerve fibers extending to the striatum. However, approx. 1 mm apart, these nerve fibers were no longer visible, but a high density of TH+ varicosities was seen along the nigro-striatal tract. Only here and there a few shorter TH+ fibers could be observed. In the striatum, only a high density of TH+ varicosities was visible. The staining of these TH+ varicosities was specific, because no staining was seen outside the nigro-striatal tract and outside the striatum. Of course the question arises if these TH+ varicosities represent functional connections between the vMes and striatum. Unfortunately, we cannot answer it at this point. The easiest explanation would be that the nerve fibers are axotomized and degenerate (Wallerian degeneration) and only large dense axonal stumps (proximal) survive, while the rest of the axon (distal) degenerates. However, after axotomy the nerve fibers should degenerate fast and disappear within hours or a few days. Since our vibrosections were cultured for 2 weeks it is fully unclear why these TH+ varicosities do not disappear, thus it seems possible that these varicosities represent active boutons of a TH+ nerve fiber. The vibrosection model is a complex 3-dimensional model and it is clear that the activity of the neurons and nerve fibers are regulated by other systems. It has been shown that e.g. glutamate, acting on group I mGluRs, plays an important trophic role for the development of the nigro-striatal dopamine pathway (Plenz and Kitai, 1998) and it may be possible that the cortico-striatal glutamatergic input regulates the dopamine neurons. More work on studying these TH+ varicosities is necessary to understand the spatial organization of varicosities along axons. Shepherd and Raastad (2003) have extensively characterized such axonal varicosities along parallel fibers in the cerebellum and discuss common principles underlying the placement of both varicosities and synapses along axons.
Laminin. Brain capillaries constitute the blood–brain barrier and innervate all areas of the brain. We have previously shown, that organotypic brain slices contain a strong network of laminin- or RECA-1 positive brain capillaries (Moser et al., 2003, 2004). Laminin is a well-established basement membrane marker and stains excellently the vascular structures of the brain (Eriksdotter-Nilsson et al., 1986; Jucker et al., 1996; Sixt et al., 2001). Previously we have also demonstrated a strong network of laminin-positive brain capillaries in slices composed of four brain slices (Ullrich and Humpel, 2009b). Furthermore we have shown, that brain capillaries grow from one side to another between the nBM and cortex when stimulated with thapsigargin (Ullrich and Humpel, 2009b). In the organotypic vibrosection model we observed an intense capillary network all over the brain section, from large brain capillaries to smaller structures in deeper brain regions. We have previously demonstrated that the presence of capillaries is important as synaptic targets for cholinergic neurons and that capillaries survive without any circulation (Moser et al., 2003). Thus, although the capillaries are not functional any longer and do not display any blood flow, it is likely, that they express and secrete a cocktail of different molecules, which indeed may also influence e.g. the nerve fiber innervations. Thus our organotypic vibrosection model may allow to study vascular connections, re-growth of capillaries and interaction with other (e.g. astroglial) cells in the brain. And indeed such vascular interactions may directly influence nerve fiber growth or act as a barrier or may release inhibitory molecules. We have recently shown that capillaries are present in a 4-brain area model and that capillaries can re-grow together between specific areas when stimulated with thapsigargin (Ullrich and Humpel, 2009b).
Advantages and disadvantages of this model. While several groups have shown exciting results with survival of cholinergic or dopaminergic neurons or nerve fiber innervation into target areas, this is the first detailed study of a whole brain sagittal model to our knowledge. A sagittal vibrosection model offers more likely the chance to study a 3-dimensional network of related neurons, such as e.g. dopaminergic and cholinergic neurons and their related pathways. In addition, this model allows to investigate the complex interaction with astrocytes and capillaries, but also with microglia. The model is easy to establish, can be cultured for several weeks and thus may represent a more advanced adult stage. Technically, this model allows principally the use of all histochemical methods, such as immunohistochemistry including triple or quadruplicate co-localizations, but also in situ hybridization to detect mRNAs or tracing of cells e.g. with Mini-Ruby. In this model stimuli which play a role in neurodegeneration of dopaminergic and cholinergic neurons can be easily studied, but also growth factors, which may counteract this cell death. This may directly have an impact to learn more about processes occurring in Alzheimer's or Parkinson's disease. As an example for such a pathological assay, we tested alterations after hydrogen peroxide treatment, which reflects a fast and simple tool to induce propodiumiode+ nuclei of cells which undergo cell death. In addition the propodiumiode+ staining was accompanied by decreased calbindin staining in the hippocampus pointing to calcium-induced changes.
Nerve fiber growth is a well controlled process and nerve fibers in co-cultures partly grew in all directions, not fully representing an in vivo situation. Similarly, transplantation of neurons into the brain causes a regulated highly controlled nerve fiber growth to specialized areas, but also in vivo long-distance growth of fibers and nerve fiber re-construction failed. Thus, further studies on fiber-reconstruction in this model will be a great challenge to fully characterize the nigro-striatal pathway and also other neuronal pathways (e.g. cortico-striatal). A combination with tracing studies and electrophysiology may also in addition proof a functional interconnection between different brain areas located far away. Such a sagittal model may offer a chance to re-construct and culture at least partly a brain in vitro.
Taken together, our novel organotypic vibrosection model provides an easy and potent whole sagittal brain culture system that allows to study cholinergic and dopaminergic neurons together but also in close interaction with all other cells of the brain and with capillaries. It will be a great challenge in future to use this model to re-construct whole pathways. This novel whole brain vibrosection model may allow to study neurodegeneration and neuroprotection of cholinergic and dopaminergic neurons, which plays an important role in Alzheimer's and Parkinson's disease, respectively.
Acknowledgements
This study was supported by the Austrian Science Funds (SFB F04405-B19). We thank Ursula Kirzenberger-Winkler for excellent technical assistance.
References
- Baratta J., Marienhagen J.W., Ha D., Yu J., Robertson R.T. Cholinergic innervation of cerebral cortex in organotypic slice cultures: sustained basal forebrain and transient striatal cholinergic projections. Neuroscience. 1996;72:1117–1132. doi: 10.1016/0306-4522(95)00603-6. [DOI] [PubMed] [Google Scholar]
- Buchs P.A., Stoppini L., Muller D. Structural modifications associated with synaptic development in area CA1 of rat hippocampal organotypic cultures. Brain Res Dev. 1993;71:81–91. doi: 10.1016/0165-3806(93)90108-m. [DOI] [PubMed] [Google Scholar]
- Crain S.M., Crain B., Peterson E.R. Development of cross-tolerance to 5-hydroxytryptamine in organotypic cultures of mouse spinal cord-ganglia during chronic exposure to morphine. Life Sci. 1982;31:241–247. doi: 10.1016/0024-3205(82)90584-7. [DOI] [PubMed] [Google Scholar]
- Eriksdotter-Nilsson M., Björklund H., Olson L. Laminin immunohistochemistry: a simple method to visualize and quantitate vascular structures in the mammalian brain. J Neurosci Methods. 1986;17:275–286. doi: 10.1016/0165-0270(86)90128-7. [DOI] [PubMed] [Google Scholar]
- Franke H., Schelhorn N., Illes P. Dopaminergic neurons develop axonal projections to their target areas in organotypic co-cultures of the ventral mesencephalon and the striatum/prefrontal cortex. Neurochem Int. 2003;42:431–439. doi: 10.1016/s0197-0186(02)00134-1. [DOI] [PubMed] [Google Scholar]
- Gähwiler B.H., Capogna M., Debanne D., McKinney R.A., Thompson S.M. Organotypic slice cultures: a technique has come of age. Trends Neurosci. 1997;20:471–477. doi: 10.1016/s0166-2236(97)01122-3. [DOI] [PubMed] [Google Scholar]
- Gähwiler B.H., Hefti F. Guidance of acetylcholinesterase-containing fibers by target tissue in co-cultured brain slices. Neuroscience. 1984;40:235–243. doi: 10.1016/0306-4522(84)90088-5. [DOI] [PubMed] [Google Scholar]
- Gähwiler B.H., Tietschin L., Knöpfel T., Enz A. Continuous presence of nerve growth factor is required for maintenance of cholinergic septal neurons in organotypic slice cultures. Neuroscience. 1990;36:27–31. doi: 10.1016/0306-4522(90)90348-8. [DOI] [PubMed] [Google Scholar]
- Hochstrasser T., Ullrich C., Sperner-Unterweger B., Humpel C. Inflammatory stimuli reduce survival of serotonergic neurons and induce neuronal expression of indoleamine 2,3-dioxygenase in rat dorsal raphe nucleus organotypic brain slices. Neuroscience. 2011;184:128–138. doi: 10.1016/j.neuroscience.2011.03.070. [DOI] [PubMed] [Google Scholar]
- Humpel C., Johansson M., Marksteiner J., Saria A., Strömberg I. Mesencephalic grafts increase preprotachykinin-A mRNA expression in striatal grafts in an in oculo co-graft model. Regul Pept. 1995;56:9–17. doi: 10.1016/0167-0115(95)00122-r. [DOI] [PubMed] [Google Scholar]
- Humpel C., Weis C. Nerve growth factor and cholinergic CNS neurons studied in organotypic brain slices. J Neural Transm. 2002;62:253–263. doi: 10.1007/978-3-7091-6139-5_23. [DOI] [PubMed] [Google Scholar]
- Humpel C., Wetmore C., Olson L. Regulation of brain-derived neurotrophic factor messenger RNA and protein at the cellular level in pentylenetetrazol-induced epileptic seizures. Neuroscience. 1993;53:909–918. doi: 10.1016/0306-4522(93)90476-v. [DOI] [PubMed] [Google Scholar]
- Jaumotte J.D., Zigmond M.J. Dopaminergic innervation of forebrain by ventral mesencephalon in organotypic slice co-cultures: effects of GDNF. Brain Res Mol. 2005;134:139–146. doi: 10.1016/j.molbrainres.2004.11.018. [DOI] [PubMed] [Google Scholar]
- Jucker M., Tian M., Norton D.D., Sherman C., Kusiak J.W., Laminin α2 is a component of brain capillary basement membrane: reduced expression in dystrophic dy mice. Neuroscience. 1996;71:1153–1161. doi: 10.1016/0306-4522(95)00496-3. [DOI] [PubMed] [Google Scholar]
- Keller F., Rimvall K., Waser P.G. Choline acetyltransferase in organotypic cultures of rat septum and hippocampus. Neurosci Lett. 1983;42:273–280. doi: 10.1016/0304-3940(83)90274-4. [DOI] [PubMed] [Google Scholar]
- Lossi L., Alasia S., Salio C., Merighi A. Cell death and proliferation in acute slices and organotypic cultures of mammalian CNS. Prog Neurobiol. 2009;88:221–245. doi: 10.1016/j.pneurobio.2009.01.002. [DOI] [PubMed] [Google Scholar]
- Marksteiner J., Humpel C. Beta-amyloid expression, release and extracellular deposition in aged rat brain slices. Mol Psychiatry. 2008;13:939–952. doi: 10.1038/sj.mp.4002072. [DOI] [PubMed] [Google Scholar]
- Moser K.V., Reindl M., Blasig I., Humpel C. Brain capillary endothelial cells proliferate in response to NGF, express NGF receptors and secrete NGF after inflammation. Brain Res. 2004;1017:53–60. doi: 10.1016/j.brainres.2004.05.013. [DOI] [PubMed] [Google Scholar]
- Moser K.V., Schmidt-Kastner R., Hinterhuber H., Humpel C. Brain capillaries and cholinergic neurons persist in organotypic brain slices in the absence of blood flow. Eur J Neurosci. 2003;18:85–94. doi: 10.1046/j.1460-9568.2003.02728.x. [DOI] [PubMed] [Google Scholar]
- Ostergaard K., Schou J.P., Zimmer J. Rat ventral mesencephalon grown as organotypic slice cultures and co-cultured, hippocampus, and cerebellum. Exp Brain Res. 1990;82:547–565. doi: 10.1007/BF00228796. [DOI] [PubMed] [Google Scholar]
- Ostergaard K. Organotypic slice cultures of rat striatum, I. A histochemical and immunohistochemical study of acetylcholinesterase, choline acetyltransferase, glutamate decarboxylase and GABA. Neuroscience. 1993;53:679–693. doi: 10.1016/0306-4522(93)90616-n. [DOI] [PubMed] [Google Scholar]
- Plenz D., Kitai S.T. Organotypic cortex–striatum–mesencephalon cultures: the nigrostriatal pathway. Neurosci Lett. 1996;209:177–180. doi: 10.1016/0304-3940(96)12644-6. [DOI] [PubMed] [Google Scholar]
- Plenz D., Kitai S.T. Regulation of the nigrostriatal pathway by metabotropic glutamate receptors during development. J Neurosci. 1998;18:4133–4144. doi: 10.1523/JNEUROSCI.18-11-04133.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robertson R.T., Baratta J., Kageyama G.H., Ha D.A., Yu J. Specificity of attachment and neurite outgrowth of dissociated basal forebrain cholinergic neurons seeded on to organotypic slice cultures of forebrain. Neuroscience. 1997;80:741–752. doi: 10.1016/s0306-4522(97)00067-5. [DOI] [PubMed] [Google Scholar]
- Schatz D.S., Kaufmann W.A., Saria A., Humpel C. Dopamine neurons in a simple GDNF-treated meso-striatal organotypic co-culture model. Exp Brain Res. 1999;127:270–280. doi: 10.1007/s002210050796. [DOI] [PubMed] [Google Scholar]
- Schatz D.S., Kaufmann W.A., Schuligoi R., Humpel C., Saria A. 3,4-Methylen-edioxymetamphetamine (Ecstasy) induces c-fos-like protein and mRNA in rat organotypic dorsal striatal slices. Synapse. 2000;36:75–83. doi: 10.1002/(SICI)1098-2396(200004)36:1<75::AID-SYN8>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- Shepherd G.M., Raastad M. Axonal varicosity distributions along parallel fibers: a new angle on a cerebellar circuit. Cerebellum. 2003;2:110–130. doi: 10.1080/14734220310011407. [DOI] [PubMed] [Google Scholar]
- Sixt M., Engelhardt B., Pausch F., Hallmann R., Vendler O., Sorokin L.M. Endothelial cell laminin isoforms, laminins 8 and 10, play decisive roles in T cell recruitment across the blood–brain barrier in experimental autoimmune encephalomyelities. J Cell Biol. 2001;153:933–945. doi: 10.1083/jcb.153.5.933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder-Keller A., Costantini L.C., Graber D.J. Development of striatal patch/matrix organization in organotypic co-cultures of perinatal striatum, cortex and substantia nigra. Neuroscience. 2001;103:97–109. doi: 10.1016/s0306-4522(00)00535-2. [DOI] [PubMed] [Google Scholar]
- Stoppini L., Buch P.A., Muller D. A simple method for organotypic cultures of nervous tissue. J Neurosci Methods. 1991;37:173–182. doi: 10.1016/0165-0270(91)90128-m. [DOI] [PubMed] [Google Scholar]
- Thomas M.P., Webster W.W., Norgren R.B., Jr., Monaghan D.T., Morrisett R.A. Survival and functional demonstration of interregional pathways in fore/midbrain slice explant cultures. Neuroscience. 1998;85:615–626. doi: 10.1016/s0306-4522(97)00646-5. [DOI] [PubMed] [Google Scholar]
- Ullrich C, Humpel C. Mini-Ruby is rapidly taken up by neurons and astrocytes in organotypic brain slices. Neurochem Res, in press. [DOI] [PMC free article] [PubMed]
- Ullrich C., Humpel C. Rotenone induces cell death of cholinergic neurons in an organotypic co-culture brain slice model. Neurochem Res. 2009;34:2147–2153. doi: 10.1007/s11064-009-0014-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ullrich C., Humpel C. The pro-apoptotic substance thapsigargin selectively stimulates re-growth of brain capillaries. Curr Neurovasc Res. 2009;6:171–180. doi: 10.2174/156720209788970063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weis C., Marksteiner J., Humpel C. Nerve growth factor and glial cell line-derived neurotrophic factor restore the cholinergic phenotype in organotypic brain slices of the basal nucleus of Meynert. Neuroscience. 2001;102:129–138. doi: 10.1016/s0306-4522(00)00452-8. [DOI] [PubMed] [Google Scholar]
- Zassler B., Dechant G., Humpel C. Urea enhances the nerve growth factor-induced neuroprotective effect on cholinergic neurons in organotypic rat brain slices. Neuroscience. 2005;130:317–323. doi: 10.1016/j.neuroscience.2004.09.010. [DOI] [PubMed] [Google Scholar]
- Zassler B., Weis C., Humpel C. Tumor necrosis factor-alpha triggers cell death of sensitized potassium chloride-stimulated cholinergic neurons. Mol Brain Res. 2003;113:78–85. doi: 10.1016/s0169-328x(03)00092-5. [DOI] [PubMed] [Google Scholar]