Abstract
Aim
Occurrence of ocular motor cranial nerve palsies (OMCNP), following stroke, has not been reported in relation to the type of OMCNP seen and in relation to brain area affected by stroke. The aim of this study was to identify all patients referred with suspected visual impairment to establish the presence and type of OMCNP.
Methods
Prospective, observation study with standardised referral and assessment forms across 20 sites. Visual assessment included visual acuity measurement, visual field assessment, ocular alignment, and movement and visual inattention assessment. Multicentre ethics approval and informed patient consent was obtained.
Results
In total, 915 patients were recruited with mean age of 69.18 years (SD 14.19). Altogether, 498 patients (54%) were diagnosed with ocular motility abnormalities. Of these, 89 patients (18%) had OMCNP. Unilateral third nerve palsy was present in 23 patients (26%), fourth nerve palsy in 14 patients (16%), and sixth nerve palsy in 52 patients (58%). Out of these, 44 patients had isolated OMCNP and 45 had OMCNP combined with other ocular motility abnormalities. Location of stroke was reported mainly in cerebellum, brain stem, thalamus, and internal and external capsules. Treatment was provided for each case including prisms, occlusion, typoscope, scanning exercises, and refraction.
Conclusions
OMCNP account for 18% of eye movement abnormalities in this stroke sub-population. Sixth CNP was most common, followed by third and fourth CNP. Half were isolated and half combined with other eye movement abnormality. Most were treated with prisms or occlusion. The reported brain area affected by stroke was typically the cerebellum, brain stem, and diencephalic structures.
Keywords: ocular motor cranial nerve palsy, stroke, ocular motility, treatment
Introduction
Ocular motor cranial nerve palsies (OMCNP) are well-recognised ocular motility abnormality. The prevalence of third, fourth, and sixth nerve palsies has been documented in large population studies, which have found a higher proportion of sixth nerve palsies followed by third and then fourth nerve palsies.1, 2, 3, 4 Causes of OMCNP include head trauma, space-occupying lesion (including neoplasm and aneurysm), and vascular lesions, with a number due to undetermined reasons. Vascular causes have been reported to include diabetes, hypertension, and atherosclerosis, accounting for 20% of third nerve palsies, 18.6% of fourth nerve palsies, and 17.7% of sixth nerve palsies.3
There is a limited literature regarding the occurrence of OMCNP in a defined stroke population. Two studies have reported OMCNP following stroke in addition to other pathologies comprising acquired brain injury.5, 6 Ciuffreda6 reported that accommodation and vergence deficits were most common in transient ischaemic attack, but strabismus and CNP were more frequent following stroke. Pedersen7 reported that ocular motility abnormalities following stroke were inclusive of gaze palsy and nystagmus, and as well as of OMCNP. Details of individual ocular motor cranial nerve involvement, clinical features, and associations with the palsy plus outcome have not been identified.
The purpose of this study is to review a defined stroke sub-population (those with identified visual impairment) to determine the prevalence of OMCNP, and identify the associations and outcomes with these palsies inclusive of ocular and non-ocular features.
Materials and methods
The design of this study was a prospective multicentre observational case cohort study. The Vision In Stroke group consists of local investigators from 20 UK hospital trusts, who are responsible for assessing stroke patients and collecting patient data. The data are collated centrally at the University of Liverpool. The study has multicentre ethics approval via the National Research Ethics Service and is being undertaken in accordance with the Tenets of Helsinki. The recruitment period for this study was from May 2006 to April 2009.
The target population was stroke patients suspected of having a visual difficulty. Referrals could be made from in-patient wards, rehabilitation units, community services, or outpatient clinics. Patients were given an information sheet and recruited after informed, written consent. Patients were excluded if they were unable to consent because of cognitive impairment, unwilling to consent, if their diagnosis was that of transient ischaemic attack, or other pathology such as aneurysm or intracranial tumour, or if they were discharged without vision assessment.
Patients with suspected visual difficulty were identified using a screening form. Subsequently, this was used as the referral form to the Orthoptic service. A standardised investigation sheet was used for the eye assessment consisting of identification of known pre-existent ocular pathology, symptoms and signs, investigation of visual field, ocular motility, and perceptual aspects. Visual acuity was assessed at near and distance fixation with Snellen or logMAR acuity tests. Assessment of ocular alignment and motility consisted of cover test, evaluation of saccadic, smooth pursuit and vergence eye movements, retinal correspondence (Bagolini glasses), fusional vergence (20D or fusional range), stereopsis (Frisby near test), prism cover test, and lid and pupil function. Perceptual deficits were recorded after questioning of the patient and/or carers and relatives. Inattention was assessed by means of a combination of assessments including line bisection, Albert's test, cancellation tests, and memory tests using verbal description and drawing. Stroke details were recorded from patient notes, accounting for stroke laterality, type, and area involved. Ocular treatment details were recorded along with outcome.
Results were inputted to the statistical package SPSS version 15 (Copyright IBM Corporation, Somer, NY, USA). Pearson χ2-test was undertaken to analyse cross-tabulations of results for CNP vs factors such as location and laterality of stroke, associated ocular and non-ocular signs, and symptoms and outcome. Non-parametric assessment (Wilcoxon test) was undertaken to compare categorical data.
Results
General demographics
Overall, 1345 patients were referred for visual assessment for this study. Out of these, 915 patients were recruited and 430 patients were excluded, the latter mainly because of the inability to consent. Of the 915 patients recruited, 59% were male and 41% female. Mean age at onset of stroke was 69.18 years (range 1–94: SD 14.19 years). Median duration from onset of stroke to initial eye examination was 22 days (0–2543 days); the mean of 40.84 (SD 141.28) days being skewed by three outliers (patients referred a number of years after the stroke onset). Stroke lesion was right sided in 48.9% (ie, right-sided brain damage), left sided in 37.7%, and bilateral in 13.4%. Infarcts accounted for 84.5% including thrombosis and embolism. Haemorrhagic strokes accounted for the remainder.
Ocular motor cranial nerve palsies
Of 915 patients, ocular motility abnormalities were recorded in 498 (54%). Of these 498 patients, 89 had OMCNP (18%: 10% of the overall stroke cohort). Unilateral third nerve palsy was present in 23 patients (26%), unilateral fourth nerve palsy in 14 patients (16%), and unilateral sixth nerve palsy in 52 patients (58%). Of the third nerve palsies, 19 were partial or complete, one was a nuclear palsy, and three were associated with Weber's syndrome.
Location of stroke
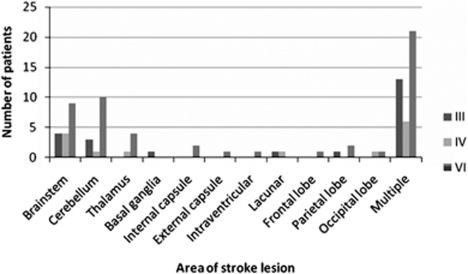
The area of brain involved by the stroke was cortical in 40%, brainstem and/or cerebellum in 47%, and mixed in 13%. Cortical areas included the frontal, parietal and occipital lobes, internal and external capsule, intraventricular area, basal ganglia, and thalamus; either in isolation or as multiple involved areas (Figure 1). The stroke lesion was right sided in 42%, left sided in 38%, and bilateral in 20%. There was no significant difference in laterality or location of stroke lesion, and presence of OMCNP in this subgroup of ocular motility abnormality (χ2-test).
Figure 1.
Location of stroke. The most usual area for location of stroke lesion causing cranial nerve palsy was the brainstem or cerebellum.
Symptoms
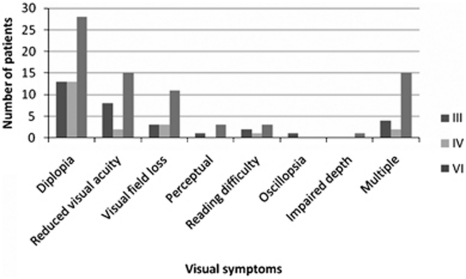
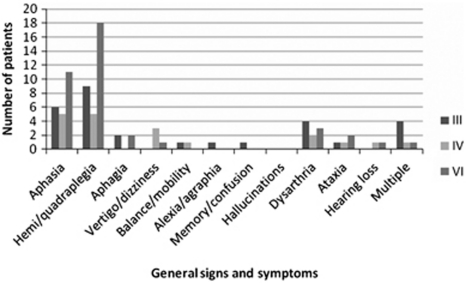
Symptoms reported at presentation were ocular and non-ocular. The most common ocular symptom was diplopia in 54 patients (61%), followed by blurred vision in 27 patients (30%) and visual field loss (20%) (Figure 2). General signs and symptoms are outlined in Figure 3. The most commonly reported was haemiplegia/quadriplegia in 32 patients (36%) followed by aphasia/dysphasia in 22 patients (25%).
Figure 2.
Visual symptoms associated with cranial nerve palsies. Most common visual symptoms included diplopia and blurred vision.
Figure 3.
General signs and symptoms. Aphasia and haemiplegia were the most common general signs and symptoms.
Associations
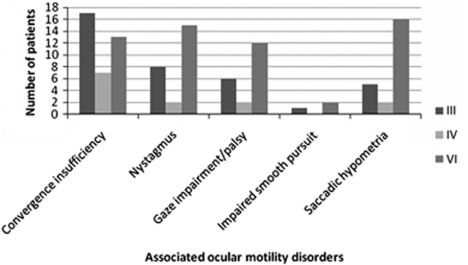
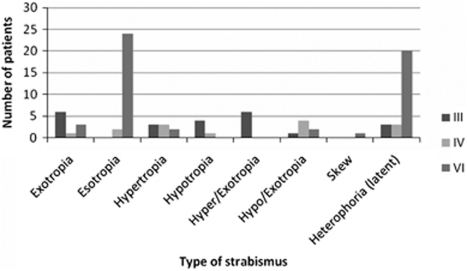
At assessment, OMCNPs were found to occur as an isolated ocular motility abnormality in 44 patients (49%) and were combined with other ocular motility abnormalities in the remaining cases. These associated ocular motility disturbances included convergence insufficiency, nystagmus, and saccadic dysmetria (Figure 4). Strabismus was detected on cover test and the type of strabismus was typical of that expected with each OMCNP type, that is, vertical deviation and exotropia in third nerve palsy, vertical deviation in fourth nerve palsy, and esotropia in sixth nerve palsy (Figure 5). Presence of manifest strabismus was significantly higher in third and fourth nerve palsies than in sixth nerve palsy (P=0.02, Wilcoxon test).
Figure 4.
Ocular motility disorders associated with cranial nerve palsy.
Figure 5.
Strabismus types associated with cranial nerve palsy. Esotropia was more prevalent in sixth nerve palsy.
Treatment
Each patient received therapy options related to their symptoms (Table 1), most frequently involving use of prisms or occlusion to alleviate diplopia. Advice included guidance on maximising eye movements and avoiding positions of gaze associated with diplopia.
Table 1. Management options for cranial nerve palsy.
| Third nerve palsy | Fourth nerve palsy | Sixth nerve palsy | |
|---|---|---|---|
| Prisms | 5 | 7 | 18 |
| Occlusion | 11 | 5 | 14 |
| Refraction | 2 | 1 | 3 |
| Typoscope | 1 | 2 | |
| Head posture | 1 | ||
| Low vision aids | 1 | ||
| Peli prisms | 2 | ||
| Advice | 16 | 4 | 39 |
| Multiple | 4 | 2 | 6 |
Review
In total, 49 patients were followed up on review assessment (Table 2). Complete recovery was noted in 22.5% and partial improvement in 43%. No improvement with static ocular motility findings was noted in 33%, and one patient showed deterioration of ocular motility.
Table 2. Recovery of cranial nerve palsy.
| Third nerve palsy | Fourth nerve palsy | Sixth nerve palsy | |
|---|---|---|---|
| Complete recovery | 2 | 1 | 8 |
| Improvement | 8 | 2 | 11 |
| Static | 6 | 2 | 8 |
| Deterioration | 1 |
Discussion
Of our entire stroke cohort with visual difficulties, 10% (89 patients) were documented as having OMCNP. This equates to a report of 10% prevalence of OMCNP in a cerebrovascular population (all patients were ambulatory outpatients with vision-related symptoms) vs a prevalence of 7% in a transient ischaemic attack population.6 A further study reported a prevalence of 9% in a population with brain damage comprising stroke patients predominantly, but also head injury and other conditions.5
In our patients with OMCNP, the most frequently documented was sixth nerve palsy (58%), followed by third nerve palsy (26%) and fourth nerve palsy (16%). This may reflect the nature of vascular blood supply to the cranial nerves in the brainstem, in addition to their infranuclear pathways, with exposure of the sixth nerve pathway to a greater number of arteries along its pathway.
The nuclear, fascicular, and proximal infranuclear pathways for the third, fourth, and sixth cranial nerves are supplied by the vertebrobasilar arterial system.8 The vertebral arteries join to form the basilar artery. Branches of the basilar artery supplying the medulla and lower pons include the posterior inferior cerebellar artery and anterior inferior cerebellar artery. Branches supplying the pons include the labyrinthine artery, paramedian pontine artery, and circumferential pontine artery. Branches supplying the pons and lower midbrain include the superior cerebellar artery. Those supplying the midbrain include the posterior cerebral artery, posterior communicating artery, and quadrigeminal artery. The distal and orbital pathways for these cranial nerves are supplied by branches from the internal carotid arteries including the middle cerebral artery and its branches: anterior choroidal artery and posterior choroidal artery.8
Specifically, the sixth cranial nerve pathway is supplied with branches of the basilar artery including the anterior inferior cerebellar artery, posterior inferior cerebellar artery, pontomedullary artery, and accessory arteries in the pons and clivus region. The distal pathway is supplied by the internal auditory artery, anterolateral artery, and tentorial artery.9, 10
The third cranial nerve pathway is supplied by branches of the basilar artery including the superior cerebellar arteries, posterior cerebral arteries, mesencephalic perforating arteries, collicular, and accessory arteries in the midbrain; the thalamoperforating arteries supplemented by the superior cerebellar artery, posterior communicating artery, and posterior cerebral artery in the proximal nerve pathway; and inferior cavernous sinus arteries, medial posterior choroidal artery, and tentorial arteries in the distal nerve pathway.11, 12
The fourth cranial nerve pathway is either in close association or in contact with branches of the basilar artery in the midbrain including the superior cerebellar artery, vernian artery, and collicular artery. It is supplied by posterior cerebral artery and posterior communicating artery in its proximal pathway and by the internal carotid artery, medial posterior choroidal artery, and tentorial arteries in the distal pathway.13, 14
Posterior circulation strokes leading to isolated mesencephalic infarcts have been associated with nuclear or fascicular third nerve palsy (Weber's syndrome, Benedict's syndrome) and contralateral motor deficits. Distal territory infarcts of the mesecephalon, thalamus, medial temporal, and occipital lobes have been reported with nuclear and fascicular third nerve palsy (Claude's syndrome), whereas middle territory infarcts of the pons and anterior inferior cerebellar artery have been associated with horizontal gaze palsy, INO, one and a half syndrome, vertical gaze palsy, fourth nerve palsy, Horner's syndrome, and skew deviation.15
It is clear that the cranial nerves are fed by a comprehensive network of arterial blood vessels, and thus are susceptible to vascular compromise. Ciuffreda6 stated that the three cranial nerves responsible for innervating the extraocular muscles are particularly vulnerable to localised lesions and disturbances at the level of the brainstem cranial nerve nuclei as well as in the cavernous sinus just before innervating the extra ocular muscles. Only 10% of our entire stroke cohort had OMCNPs and this may be explained by the overlap between arteries that supply the brainstem and infranuclear pathways along with variations in collateral circulation.16
Ocular motility abnormalities following stroke have been reported as typically associated with brainstem and/or cerebellar lesions.16 OMCNPs may be nuclear, fascicular, or infranuclear in type, depending on the location of the pathological lesion. In our cohort of patients with OMCNP, there was a tendency for these nerve palsies to occur more in brainstem/cerebellum strokes (47%) than in cortical strokes (40%), although this was not significant. Thirteen percent were mixed locations, that is, the lesion involved both cortical and brainstem or cerebellar areas. Of note, some areas listed on brain imaging reports as the site of stroke were areas not usually associated with the pathway of these cranial nerves, such as the frontal lobe and intraventricular areas. A limitation was that the location of stroke on imaging scans was determined from the radiology reports and not by a physician's review of the scans in associated with the clinical signs and symptoms. It is therefore assumed in these cases that the brain imaging reports detailed the main site of infarct or haemorrhage. However, there may be extension of the stroke to other non-reported areas in which the ocular motor cranial nerve pathway may be involved with related vascular compromise of the blood supply to the cranial nerves.
Presenting symptoms were varied. General associated symptoms have been reported as dyspraxia and dysarthria16 and in our study, haemiplegia and aphasia were most commonly documented. The most frequent ocular symptom was diplopia (61%). A further 24% complained of multiple visual symptoms including diplopia, blurred vision, visual field impairment, and reading difficulty. Thirty percent complained of blurred vision as an isolated symptom, which, in some cases, may relate to a genuine reduction in visual acuity, but in others, may relate to blurring of vision due to ocular misalignment; in the latter cases, images are not completely separate, thus they result in blurred rather than double vision. Twenty percent complained of visual field loss. The association of visual field loss with CNP was related to stroke that involved areas of the brain in which the infranuclear cranial nerve pathway was associated with the visual pathway, such as diencephalic areas and the region of the cavernous sinus.
Diplopia commonly relates to the presence of strabismus or asymmetrical ocular motility on gaze away from the primary ‘straight-ahead' position and may cause nausea, imbalance, or impaired depth perception.17 It has been listed as one of the typical symptoms of strokes affecting the brainstem and reported in 38% of one brainstem stroke population.16 However, it is not a characteristic consequence of cortical stroke.5 We have previously reported a lack of symptomatic diplopia in stroke survivors with acquired isolated strabismus.18 However, strabismus in this current study was not isolated, but due to ocular motor cranial nerve palsy, and a high proportion of patients complained of this symptom, regardless of the type of palsy.
The type of strabismus largely related to the type of palsy. Third nerve palsy was frequently associated with divergent and vertical deviations, which were manifest in 87% of cases. Fourth nerve palsy was most associated with vertical deviations, which were manifest in 79% of cases. Sixth nerve palsy was typically associated with convergent deviations, which were manifest in 62% of cases, which was significantly less than patients with third or fourth nerve palsy. The increased frequency of manifest deviations in third and fourth nerve palsies may relate to the presence of vertical deviations in which small vertical fusion ranges result in difficulty to maintain control of the vertical deviation. Larger divergent fusional vergence may help to compensate the convergent deviation in sixth nerve palsy, resulting in greater ability to maintain binocular single vision.
Almost half of OMCNPs occurred as isolated ocular motility disturbances. The remainder was combined with other ocular motility abnormalities, particularly vergence impairment, nystagmus, and saccadic hypometria. Vergence abnormalities have been reported previously as being documented frequently following stroke.6 However, it is unknown whether the convergence insufficiency preceded the stroke or occurred subsequent to the stroke and its effect on the ocular motor system. Given the rate at which reading impairment is noted in stroke populations,19 it is important to check the presence of convergence insufficiency, as it is readily treatable. Equally it is important to check for saccadic abnormalities, nystagmus, and gaze defects because of the impact of poor saccadic control, oscillopsia, and defective gaze on activities of daily living and overall rehabilitation.17, 20
Treatment for visual symptoms comprised mostly occlusion and prisms, which were targeted at alleviating diplopia.17 Advice was commonly provided in relation to the adoption of a compensatory head posture, use of visual scanning strategies, undertaking scanning eye exercises to improve gaze and saccadic eye movements, plus reading strategies. Just over half of patients were followed for review, which constitutes a limitation of this study. For those patients who attended for review, a good outcome was achieved in 65.5% defined by either complete recovery or improvement in ocular motility. Similarly, Rush2 reported a recovery rate of 71% for CNPs caused by vascular events, although it is not specified whether recovery was complete or partial.
One further limitation of this study was that a large proportion of patients were excluded from analysis because of lack of consent. The ethics approval for this study allowed capture of an ocular diagnosis for non-recruited patients and 92 patients (21.4%) had ocular motility abnormalities. However, ethics approval did not allow us to obtain anymore detailed information than a general category of diagnosis.
Conclusions
Ten percent of our stroke population had OMCNP (18% of those with ocular motility abnormalities). The symptom of diplopia was frequently reported and predominantly treated with prisms or occlusion. Sixth nerve palsy was more common than third and fourth nerve palsies. However, the presence of manifest strabismus was significantly less in sixth nerve palsy. Almost half of OMCNPs in this study were isolated and the remainder was associated with additional ocular motility abnormalities, including vergence deficit, nystagmus, gaze palsy, and saccadic hypometria. OMCNPs were due to both cortical and brainstem strokes, and there was no significant difference for presence of nerve palsy dependent on the location or laterality of stroke.

Acknowledgments
We acknowledge Alison Price, Birmingham: Sandwell and West Birmingham NHS Trust and Linda Walker, Burnley: East Lancashire Hospitals NHS Trust for their involvement with this study.
Appendix
VIS Group:
David Wright: Altnagelvin: Altnagelvin Hospitals HHS Trust; Darren Brand: Ayr: NHS Ayrshire and Arran; Carole Jackson: Bath: Royal United Hospitals Bath NHS Trust; Shirley Harrison: Bury: Bury PCT; Carla Eccleston: Derby: Derby Hospitals NHS Trust; Tallat Maan: Durham: Durham and Darlington Hospitals NHS Foundation Trust; Claire Scott: Ipswich: Ipswich Hospital NHS Trust; Linda Vogwell: Gloucester: Gloucestershire Hospitals NHS Foundation Trust; Sarah Peel: Jersey: St Helier General Hospital; Leonie Robson: Lincoln: United Lincolnshire Hospitals NHS Trust; Nicola Akerman: Nottingham: University Hospital NHS Trust; Caroline Dodridge: Oxford: Oxford Radcliffe Hospitals NHS Trust; Claire Howard: Salford: Salford Primary Care Trust; Tracey Shipman: Sheffield: Sheffield Teaching Hospitals NHS Foundation Trust; Una Sperring: Swindon: Swindon and Marlborough NHS Trust; Sue Yarde: Taunton: Taunton and Somerset NHS Trust; Fiona Rowe: Warrington: Warrington and Halton Hospitals NHS Foundation Trust; Sonia MacDiarmid: Wigan: Wrightington, Wigan and Leigh NHS Trust; Cicely Freeman: Worcester: Worcestershire Acute Hospitals NHS Trust.
The authors declare no conflict of interest.
Footnotes
This article is based on a study first reported in the Transactions of the International Strabismological Association, Istanbul, Turkey, 2010, in press.
References
- Rucker CW. Paralysis of the third, fourth and sixth cranial nerves. Am J Ophthalmol. 1958;46:787–794. doi: 10.1016/0002-9394(58)90989-9. [DOI] [PubMed] [Google Scholar]
- Rush JA, Younge BR. Paralysis of cranial nerves III, IV and VI. Cause and prognosis in1000 cases. Arch Ophthalmol. 1981;99:76–79. doi: 10.1001/archopht.1981.03930010078006. [DOI] [PubMed] [Google Scholar]
- Rucker CW. The causes of paralysis of the third, fourth and sixth cranial nerves. Am J Ophthalmol. 1966;61:1293–1298. doi: 10.1016/0002-9394(66)90258-3. [DOI] [PubMed] [Google Scholar]
- Richards BW, Jones FR, Younge BR. Causes and prognosis in 4278 cases of paralysis of the oculomotor, trochlear and abducens cranial nerves. Am J Ophthalmol. 1992;113:489–496. doi: 10.1016/s0002-9394(14)74718-x. [DOI] [PubMed] [Google Scholar]
- Fowler MS, Wade DT, Richardson AJ, Stein JF. Squints and diplopia seen after brain damage. J Neurol. 1996;243:86–90. doi: 10.1007/BF00878537. [DOI] [PubMed] [Google Scholar]
- Ciuffreda KJ, Kapoor N, Rutner D, Suchoff IB, Han ME, Craig S. Occurrence of oculomotor dysfunctions in acquired brain injury: a retrospective analysis. Optometry. 2007;78:155–161. doi: 10.1016/j.optm.2006.11.011. [DOI] [PubMed] [Google Scholar]
- Pedersen RA, Troost BT. Abnormalities of gaze in cerebrovascular disease. Stroke. 1981;12:251–254. doi: 10.1161/01.str.12.2.251. [DOI] [PubMed] [Google Scholar]
- Kouka N.Central nervous system blood supply . www.brain101.info . Accessed 10 September 2010.
- Marinkovic SV, Gibo H, Stimec B. The neurovascular relationships and the blood supply of the abducent nerve: surgical anatomy of its cisternal segment. Neurosurgery. 1994;34:1017–1026. [PubMed] [Google Scholar]
- Yousry I, Camelio S, Wiesmann M, Schmid UD, Moriggl B, Brückmann H, et al. Detailed magnetic resonance imaging anatomy of the cisternal segment of the abducent nerve: Dorello's canal and neurovascular relationships and landmarks. J Neurosurg. 1999;91 (2:276–283. doi: 10.3171/jns.1999.91.2.0276. [DOI] [PubMed] [Google Scholar]
- Marinkovic S, Gibo H. The neurovascular relationships and the blood supply of the oculomotor nerve: the microsurgical anatomy of its cisternal segment. Surg Neurol. 1994;42:505–516. doi: 10.1016/0090-3019(94)90081-7. [DOI] [PubMed] [Google Scholar]
- Cahill M, Bannigan J, Eustace P. Anatomy of the infranuclear blood supply to the intracranial oculomotor nerve. Br J Ophthalmol. 1996;80:177–181. doi: 10.1136/bjo.80.2.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marinkovic S, Gibo H, Zelic O, Nikodijevic I. The neurovascular relationships and the blood supply of the trochlear nerve: surgical anatomy of its cisternal segment. Neurosurgery. 1996;38:161–169. doi: 10.1097/00006123-199601000-00036. [DOI] [PubMed] [Google Scholar]
- Yousry I, Moriggl B, Dieterich M, Naidich TP, Schmid UD, Yousry T. MR anatomy of the proximal cisternal segment of the trochlear nerve: neurovascular relationships and landmarks. Radiology. 2002;223:31–38. doi: 10.1148/radiol.2231010612. [DOI] [PubMed] [Google Scholar]
- Kumral E, Bayulkem G, Akyol A, Yunten N, Sirin H, Sagduyn A. Mesencephalic and associated posterior circulation infarcts. Stroke. 2002;33:2224–2231. doi: 10.1161/01.str.0000027438.93029.87. [DOI] [PubMed] [Google Scholar]
- Teasell R, Foley N, Doherty T, Finestone H. Clinical characteristics of patients wth brainstem strokes admitted to a rehabilitation unit. Arch Phys Med Rehabil. 2002;83:1013–1016. doi: 10.1053/apmr.2002.33102. [DOI] [PubMed] [Google Scholar]
- Khan S, Leung E, Jay WM. Stroke and visual rehabilitation. Top Stroke Rehabil. 2008;15:27–36. doi: 10.1310/tsr1501-27. [DOI] [PubMed] [Google Scholar]
- Rowe FJ. VIS group UKThe profile of strabismus in stroke survivors. Eye. 2010;24:682–685. doi: 10.1038/eye.2009.138. [DOI] [PubMed] [Google Scholar]
- Rowe FJ.VIS group UKReading difficulty after stroke: ocular and non ocular causes Int J Stroke 2011. e-pub ahead of print 1 March 2011; doi: 10.1111/j.1747-4949.2011.00583.x [DOI]
- Rowe FJ. VIS group UKVisual impairment following stroke. Do stroke patients require vision assessment. Age and Ageing. 2009;38:188–193. doi: 10.1093/ageing/afn230. [DOI] [PubMed] [Google Scholar]