Abstract
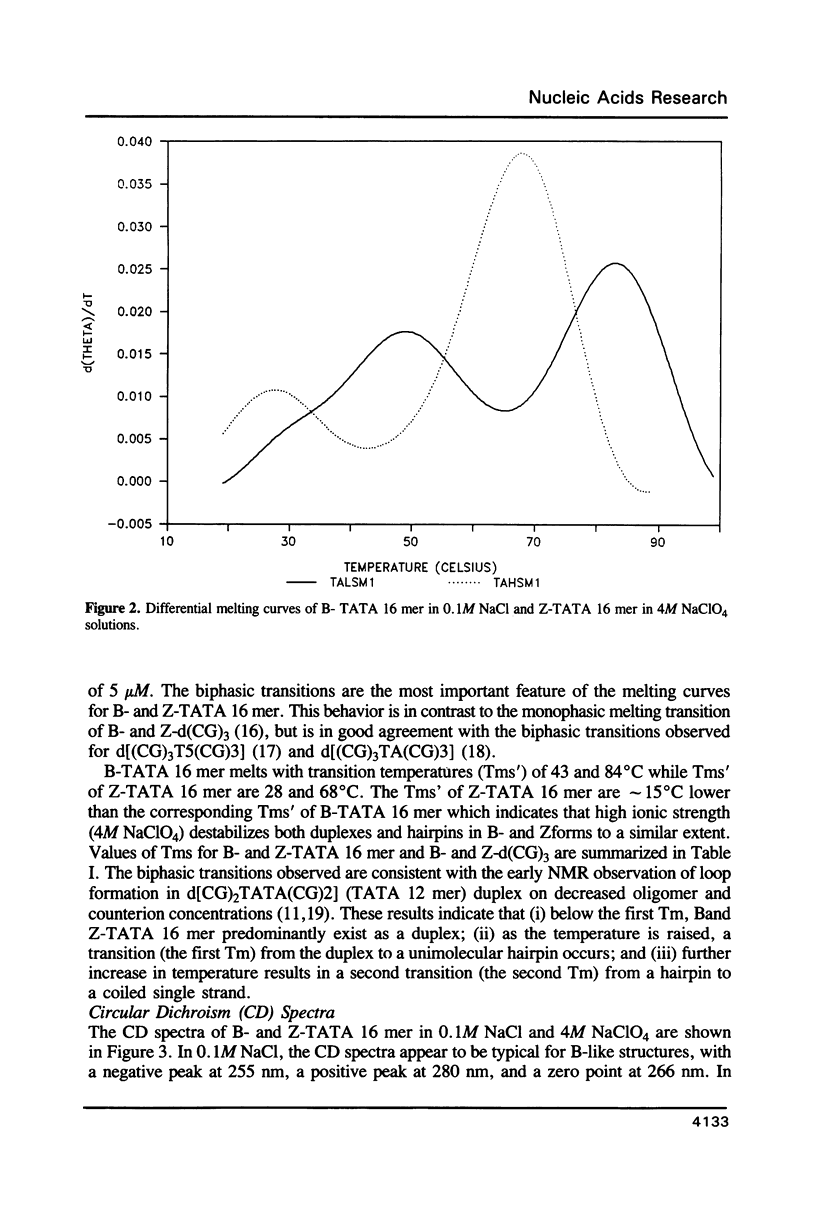
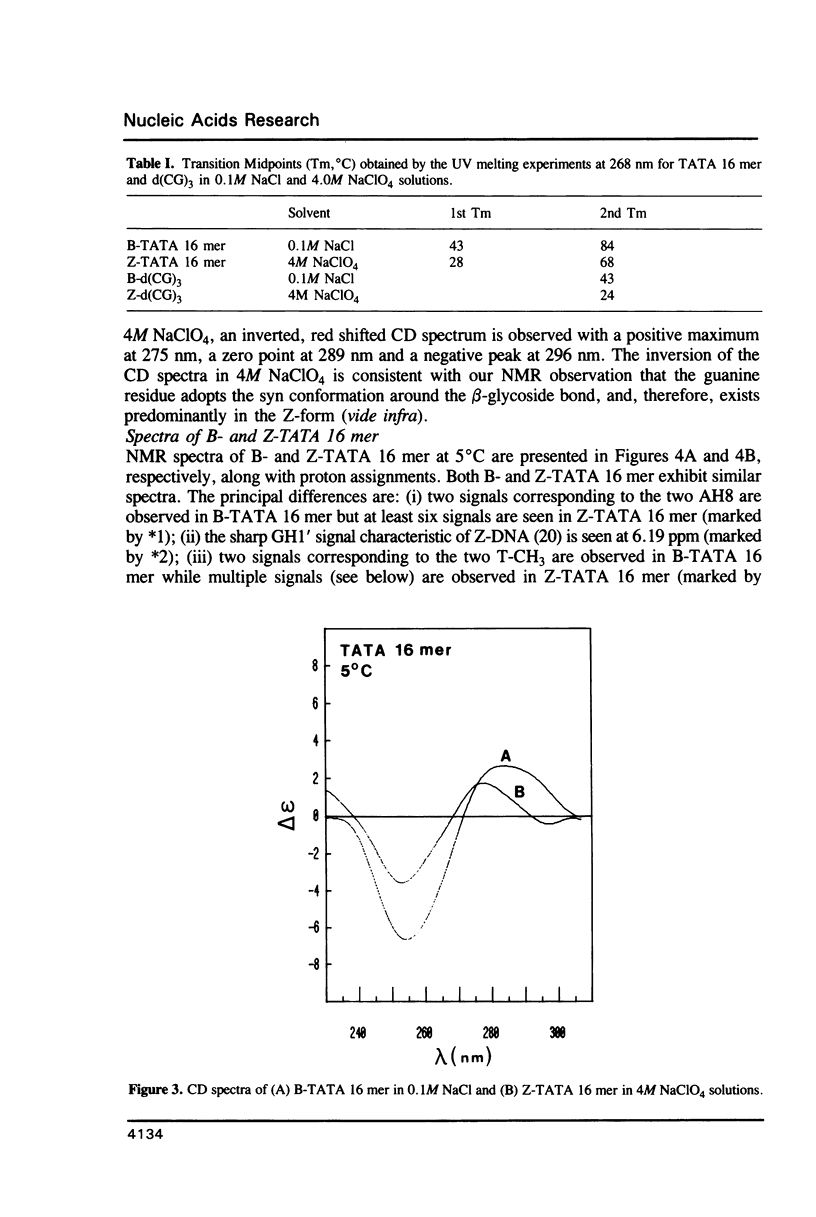
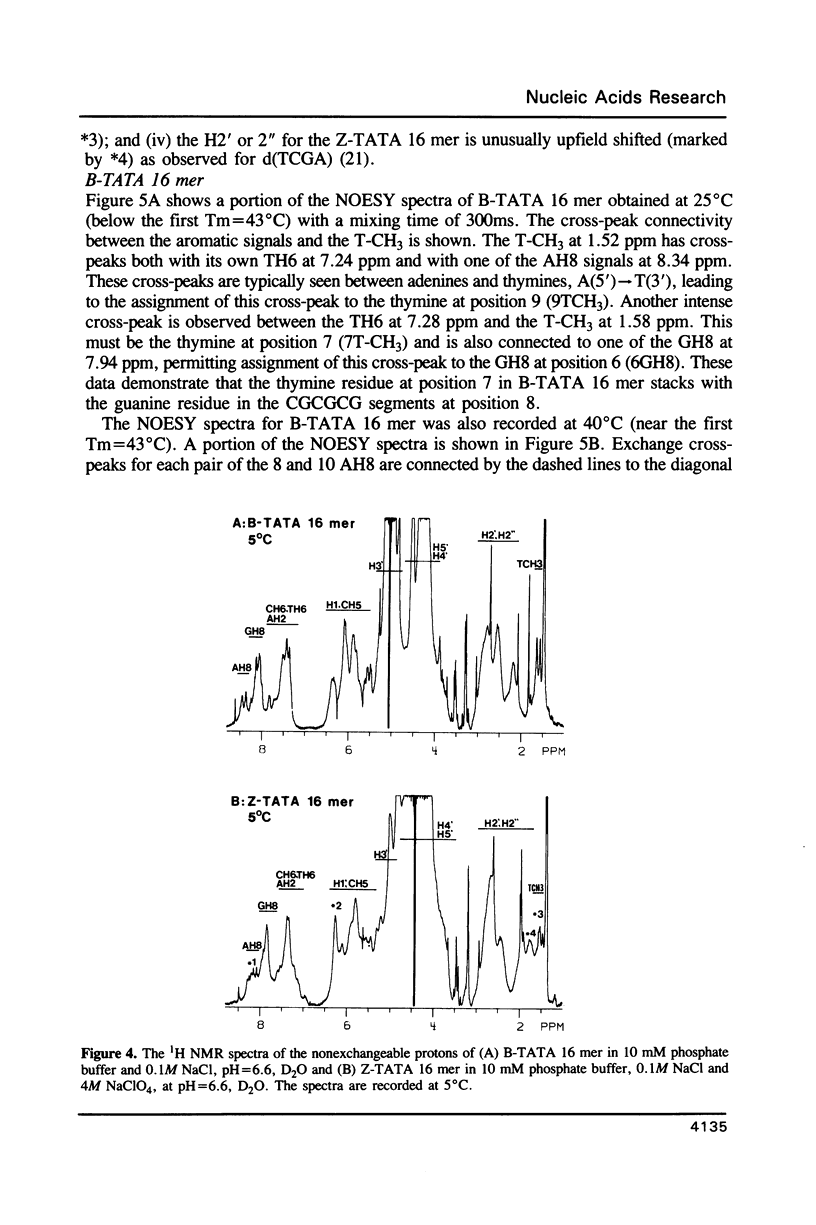
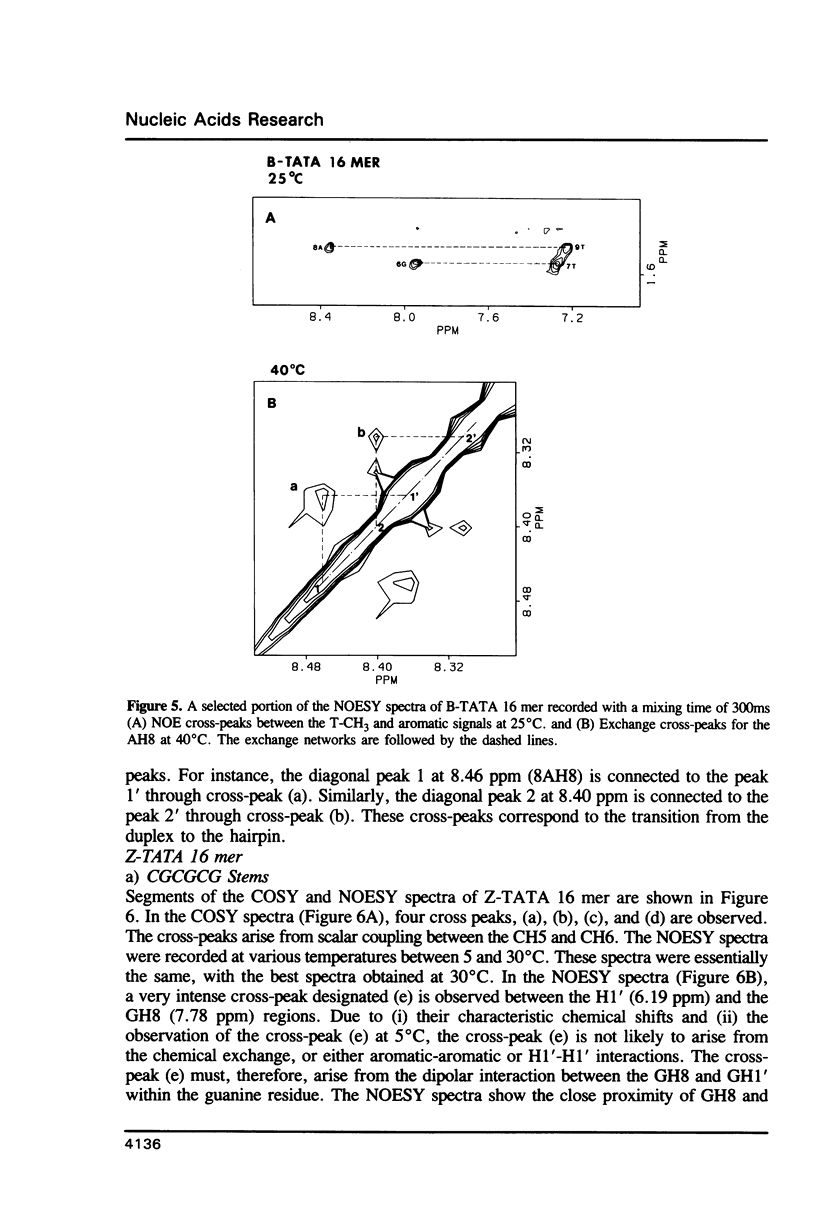
The conformation and dynamics of a DNA oligomer, d[(CG)3TATA(CG)3], in 4M NaClO4 (Z-TATA 16 mer) have been studied by 1H NMR. The principal results of our investigation are: (i) at low temperature d[(CG)3TATA(CG)3] exists as duplexes in both low (0.1M NaCl) and high (4M NaClO4) ionic strength solutions; (ii) CGCGCG segments undergo the B-to-Z transition in 4M NaClO4; (iii) even in 4M NaClO4 the TATA box exhibits non-Z-structures and possesses multiple conformations which are slowly exchanging on the NMR chemical shift difference time scale; and, (iv) the Z-type structure of the CGCGCG segments induced in 4M NaClO4 is more conformationally mobile than its B-type counterpart in 0.1M NaCl on the nanosecond time scale.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Dickerson R. E., Drew H. R. Structure of a B-DNA dodecamer. II. Influence of base sequence on helix structure. J Mol Biol. 1981 Jul 15;149(4):761–786. doi: 10.1016/0022-2836(81)90357-0. [DOI] [PubMed] [Google Scholar]
- Drew H., Takano T., Tanaka S., Itakura K., Dickerson R. E. High-salt d(CpGpCpG), a left-handed Z' DNA double helix. Nature. 1980 Aug 7;286(5773):567–573. doi: 10.1038/286567a0. [DOI] [PubMed] [Google Scholar]
- Hall K., Cruz P., Tinoco I., Jr, Jovin T. M., van de Sande J. H. 'Z-RNA'--a left-handed RNA double helix. Nature. 1984 Oct 11;311(5986):584–586. doi: 10.1038/311584a0. [DOI] [PubMed] [Google Scholar]
- Haniford D. B., Pulleyblank D. E. Facile transition of poly[d(TG) x d(CA)] into a left-handed helix in physiological conditions. Nature. 1983 Apr 14;302(5909):632–634. doi: 10.1038/302632a0. [DOI] [PubMed] [Google Scholar]
- Ikuta S., Chattopadhyaya R., Dickerson R. E. Reverse-phase polystyrene column for purification and analysis of DNA oligomers. Anal Chem. 1984 Oct;56(12):2253–2256. doi: 10.1021/ac00276a063. [DOI] [PubMed] [Google Scholar]
- Ikuta S., Chattopadhyaya R., Ito H., Dickerson R. E., Kearns D. R. NMR study of a synthetic DNA hairpin. Biochemistry. 1986 Aug 26;25(17):4840–4849. doi: 10.1021/bi00365a018. [DOI] [PubMed] [Google Scholar]
- Jaworski A., Hsieh W. T., Blaho J. A., Larson J. E., Wells R. D. Left-handed DNA in vivo. Science. 1987 Nov 6;238(4828):773–777. doi: 10.1126/science.3313728. [DOI] [PubMed] [Google Scholar]
- Kilpatrick M. W., Klysik J., Singleton C. K., Zarling D. A., Jovin T. M., Hanau L. H., Erlanger B. F., Wells R. D. Intervening sequences in human fetal globin genes adopt left-handed Z helices. J Biol Chem. 1984 Jun 10;259(11):7268–7274. [PubMed] [Google Scholar]
- Klevit R. E., Wemmer D. E., Reid B. R. 1H NMR studies on the interaction between distamycin A and a symmetrical DNA dodecamer. Biochemistry. 1986 Jun 3;25(11):3296–3303. doi: 10.1021/bi00359a032. [DOI] [PubMed] [Google Scholar]
- Kłysik J., Stirdivant S. M., Larson J. E., Hart P. A., Wells R. D. Left-handed DNA in restriction fragments and a recombinant plasmid. Nature. 1981 Apr 23;290(5808):672–677. doi: 10.1038/290672a0. [DOI] [PubMed] [Google Scholar]
- Marky L. A., Blumenfeld K. S., Kozlowski S., Breslauer K. J. Salt-dependent conformational transitions in the self-complementary deoxydodecanucleotide d(CGCAATTCGCG): evidence for hairpin formation. Biopolymers. 1983 Apr;22(4):1247–1257. doi: 10.1002/bip.360220416. [DOI] [PubMed] [Google Scholar]
- Mirau P. A., Behling R. W., Kearns D. R. Internal motions in B- and Z-form poly(dG-dC).poly(dG-dC): 1H NMR relaxation studies. Biochemistry. 1985 Oct 22;24(22):6200–6211. doi: 10.1021/bi00343a026. [DOI] [PubMed] [Google Scholar]
- Nordheim A., Lafer E. M., Peck L. J., Wang J. C., Stollar B. D., Rich A. Negatively supercoiled plasmids contain left-handed Z-DNA segments as detected by specific antibody binding. Cell. 1982 Dec;31(2 Pt 1):309–318. doi: 10.1016/0092-8674(82)90124-6. [DOI] [PubMed] [Google Scholar]
- Nordheim A., Rich A. The sequence (dC-dA)n X (dG-dT)n forms left-handed Z-DNA in negatively supercoiled plasmids. Proc Natl Acad Sci U S A. 1983 Apr;80(7):1821–1825. doi: 10.1073/pnas.80.7.1821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel D. J., Kozlowski S. A., Hare D. R., Reid B., Ikuta S., Lander N., Itakura K. Conformation, dynamics, and structural transitions of the TATA box region of self-complementary d[(C-G)n-T-A-T-A-(C-G)n] duplexes in solution. Biochemistry. 1985 Feb 12;24(4):926–935. doi: 10.1021/bi00325a018. [DOI] [PubMed] [Google Scholar]
- Pohl F. M., Jovin T. M. Salt-induced co-operative conformational change of a synthetic DNA: equilibrium and kinetic studies with poly (dG-dC). J Mol Biol. 1972 Jun 28;67(3):375–396. doi: 10.1016/0022-2836(72)90457-3. [DOI] [PubMed] [Google Scholar]
- Reid D. G., Salisbury S. A., Brown T., Williams D. H. Conformations of two duplex forms of d(TCGA) in slow-exchange equilibrium characterized by NMR. Biochemistry. 1985 Jul 30;24(16):4325–4332. doi: 10.1021/bi00337a011. [DOI] [PubMed] [Google Scholar]
- Summers M. F., Byrd R. A., Gallo K. A., Samson C. J., Zon G., Egan W. Nuclear magnetic resonance and circular dichroism studies of a duplex--single-stranded hairpin loop equilibrium for the oligodeoxyribonucleotide sequence d(CGCGATTCGCG). Nucleic Acids Res. 1985 Sep 11;13(17):6375–6386. doi: 10.1093/nar/13.17.6375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan Z. K., Ikuta S., Huang T., Dugaiczyk A., Itakura K. Solid-phase synthesis of polynucleotides. VIII: A simplified synthesis of oligodeoxyribonucleotides. Cold Spring Harb Symp Quant Biol. 1983;47(Pt 1):383–391. doi: 10.1101/sqb.1983.047.01.045. [DOI] [PubMed] [Google Scholar]
- Wang A. H., Quigley G. J., Kolpak F. J., Crawford J. L., van Boom J. H., van der Marel G., Rich A. Molecular structure of a left-handed double helical DNA fragment at atomic resolution. Nature. 1979 Dec 13;282(5740):680–686. doi: 10.1038/282680a0. [DOI] [PubMed] [Google Scholar]
- Wemmer D. E., Chou S. H., Hare D. R., Reid B. R. Duplex-hairpin transitions in DNA: NMR studies on CGCGTATACGCG. Nucleic Acids Res. 1985 May 24;13(10):3755–3772. doi: 10.1093/nar/13.10.3755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H. M., Dattagupta N., Crothers D. M. Solution structural studies of the A and Z forms of DNA. Proc Natl Acad Sci U S A. 1981 Nov;78(11):6808–6811. doi: 10.1073/pnas.78.11.6808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xodo L. E., Manzini G., Quadrifoglio F., van der Marel G. A., van Boom J. H. Oligodeoxynucleotide folding in solution: loop size and stability of B-hairpins. Biochemistry. 1988 Aug 23;27(17):6321–6326. doi: 10.1021/bi00417a018. [DOI] [PubMed] [Google Scholar]
- Xodo L. E., Manzini G., Quadrifoglio F., van der Marel G. A., van Boom J. H. Thermodynamic behaviour of the heptadecadeoxynucleotide d(CGCGCGTTTTTCGCGCG) forming B and Z hairpins in aqueous solution. Nucleic Acids Res. 1986 Jul 11;14(13):5389–5398. doi: 10.1093/nar/14.13.5389. [DOI] [PMC free article] [PubMed] [Google Scholar]


