Expression of the morphogen Shh and its receptors is robustly regulated in the embryonic and adult mammalian brain following maternal or adult-onset thyroid hormone perturbations.
Abstract
Thyroid hormone is important for development and plasticity in the immature and adult mammalian brain. Several thyroid hormone-responsive genes are regulated during specific developmental time windows, with relatively few influenced across the lifespan. We provide novel evidence that thyroid hormone regulates expression of the key developmental morphogen sonic hedgehog (Shh), and its coreceptors patched (Ptc) and smoothened (Smo), in the early embryonic and adult forebrain. Maternal hypo- and hyperthyroidism bidirectionally influenced Shh mRNA in embryonic forebrain signaling centers at stages before fetal thyroid hormone synthesis. Further, Smo and Ptc expression were significantly decreased in the forebrain of embryos derived from hypothyroid dams. Adult-onset thyroid hormone perturbations also regulated expression of the Shh pathway bidirectionally, with a significant induction of Shh, Ptc, and Smo after hyperthyroidism and a decline in Smo expression in the hypothyroid brain. Short-term T3 administration resulted in a significant induction of cortical Shh mRNA expression and also enhanced reporter gene expression in Shh+/LacZ mice. Further, acute T3 treatment of cortical neuronal cultures resulted in a rapid and significant increase in Shh mRNA, suggesting direct effects. Chromatin immunoprecipitation assays performed on adult neocortex indicated enhanced histone acetylation at the Shh promoter after acute T3 administration, providing further support that Shh is a thyroid hormone-responsive gene. Our results indicate that maternal and adult-onset perturbations of euthyroid status cause robust and region-specific changes in the Shh pathway in the embryonic and adult forebrain, implicating Shh as a possible mechanistic link for specific neurodevelopmental effects of thyroid hormone.
Thyroid hormone deficiencies during critical periods of brain development result in profound structural as well as functional abnormalities (1–4). Several studies suggest that the critical period in which thyroid hormone exerts dramatic effects on neuronal structure appears to be predominantly during early postnatal life (1). However, maternally derived thyroid hormone, transported via the placenta, is known to alter neural progenitor proliferation, differentiation, and migration within the developing embryo, which expresses thyroid hormone receptor (TR) isoforms before the onset of fetal thyroid hormone synthesis (5–11). Furthermore, thyroid hormone is known to exert effects on neuronal and glial progenitor proliferation, survival, and maturation within the adult brain (12–18). Clinical evidence indicates that early gestational deprivation of thyroid hormone, as well as adult-onset hypothyroid status, result in cognitive and functional neurological impairments, with the early onset effects associated with more severe consequences (1, 3, 19). Taken together, these studies provide evidence of a role for thyroid hormone in modulating structure and function in the mammalian brain throughout life.
Thyroid hormone effects are mediated through the transcriptional regulation of target genes via distinct TR isoforms, encoded by the TRα and TRβ genes (20). Several thyroid hormone-responsive genes, including laminin, transient axonal glycoprotein-1, reelin, neurogenic differentiation, rabconnectin 3/neurogranin, and Dab-1, have been implicated in mediating thyroid hormone effects on neurodevelopment (21–24). Although many thyroid hormone-responsive genes are sensitive to thyroid hormone perturbations only during critical periods, there are relatively fewer target genes that are regulated by thyroid hormone across the lifespan (25–27). Given that thyroid hormone influences neural and glial progenitors in both the developing as well as mature nervous system, we hypothesized that the expression of developmental morphogens, which retain a powerful influence on these progenitors across the lifespan, may be regulated by thyroid hormone. Sonic hedgehog (Shh), a major developmental morphogen, is thought to be a key regulator of neural and oligodendroglial progenitors across development and into adulthood (28–31). Shh mediates its biological effects via its membrane-associated receptors smoothened (Smo) and patched (Ptc, Ptch1), such that Shh binding to Ptc relieves the inhibitory influence of Ptc on Smo, thus activating the downstream cascade (32). Strikingly, during amphibian metamorphosis, thyroid hormone is reported to exert its metamorphic effects within the gastrointestinal system via influencing Xenopus hedgehog (Xhh) expression, indicating that this developmental signaling pathway can be targeted by thyroid hormone in vertebrates (33). We hypothesized that perturbations of thyroid hormone status may influence expression of Shh, or its receptors Ptc and Smo, both in the embryonic and adult mammalian brain. Here, we provide novel evidence that perturbations of maternal and adult thyroid hormone status results in a robust and bidirectional regulation of the expression of the Shh signaling cascade in the embryonic and adult rodent brain, respectively, suggesting that this pathway may mediate some of the important neurodevelopmental effects of thyroid hormone perturbations.
Materials and Methods
Animal treatment paradigms
Sprague Dawley rats (220–270 g) bred in our animal-breeding colony were used for all experiments. Animals were group housed and maintained on a 12-h light, 12-h dark cycle with access to food and water ad libitum. All animal procedures were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and approved by the Tata Institute of Fundamental Research and Columbia University Institutional Animal Ethics Committees. Shh+/LacZ mice were generated by A. Kottmann through the targeting of an internal ribosome entry site LacZ construct into the 3′ untranslated region of the Shh gene keeping the Shh coding sequence intact (34, 35).
To address the influence of maternal hypothyroidism on the Shh signaling cascade in the embryo, female rats were subjected to sham surgery or thyroidectomized (n = 4–5 dams per group) and allowed recovery for a week. Thyroidectomized and control dams were set up for mating with the day of vaginal plug being considered as d 0.5. After detection of a vaginal plug, 6-n-propyl-2-thiouracil (PTU) (0.05%; Sigma, St. Louis, MO) was added to the drinking water of thyroidectomized dams till sacrifice. Embryos were harvested from hypothyroid and control animals at embryonic d (e)13.5 (n = 11–15 embryos per group). All embryos used in these studies were age matched. Embryos harvested from hypothyroid dams showed decreased body size, and to rule out differences due to a possible developmental lag, all embryos used in the study were also somite matched. To address the influence of maternal hyperthyroidism, pregnant dams received T3 (1 mg/kg) or vehicle (0.02 n NaOH) injections ip from the day the vaginal plug was observed until killing at e13.5 (n = 7 embryos per group derived from three to four dams per group). After decapitation, the embryonic head was frozen on dry ice and stored at −70 C.
To address the influence of adult-onset hypothyroidism on Shh pathway expression, male rats received the goitrogens 2-mercapto-1-methylimidazole (MMI) (Sigma) or PTU (n = 4/group). MMI (0.025%) or PTU (0.05%) in drinking water for a period of 28 and 21 d, respectively, whereas controls received normal drinking water. To address the influence of adult-onset hyperthyroidism, T3 (0.5 mg/kg) or vehicle (0.02 n NaOH) was sc administered daily for 10 d with animals killed 2 h after the last T3 injection (n = 5–6/group). To address the influence of acute T3 treatment, adult male rats received a single sc injection of T3 (0.5 mg/kg) or vehicle (0.02 n NaOH) and were killed 3 h later [n = 3–5/group for in situ hybridization and n = 7–10/group for chromatin immunoprecipitation (ChIP)]. All animals were killed via rapid decapitation, and brains were fresh frozen on dry ice and stored at −70 C until use.
To examine the influence of short-duration thyroid hormone treatment, Shh+/LacZ reporter mice were injected sc twice daily with T3 (0.5 mg/kg) or vehicle (0.02 n NaOH) for 2 d and killed 2 d after the last T3 injection (n = 5/group) via transcardial perfusion. The brains were postfixed in 4% paraformaldehyde and free-floating sections (30 μm) obtained using a vibratome (TPI, St. Louis, MO).
Trunk blood was collected from all animals at the time of killing and serum T3 levels determined using the commercially available RIA kit (RIAK-4/4A; BRIT, Mumbai, India) as previously described (36). Serum T3 levels are shown in Supplemental Table 1, published on The Endocrine Society's Journals Online web site at http://endo.endojournals.org.
Primary cortical cultures
Cortical neurons were isolated from rat embryos (e17.5). After removal of meninges, the cleaned cortices were placed in trypsin-EDTA (Invitrogen, Carlsbad, CA) for 15 min followed by washes with cold HEPES buffered Hank's balanced salt solution (Invitrogen) and dissociated in culture medium (Neurobasal medium supplemented with 2% B27 supplement, 0.5 mm l-glutamine, 5 U/ml penicillin, and 5 U/ml streptomycin) (Invitrogen). Cells were plated on Poly-d-lysine (Sigma) coated 35-mm dishes at a density of 106 cells/dish. Neurons were allowed to attach and extend processes for 9 d in vitro before initiating treatment with 20 nm T3 (Sigma) for 3 h. Cells were harvested 3 h after T3 treatment and processed for RNA extraction, with all assays performed in triplicate.
Quantitative PCR (qPCR)
RNA purification, cDNA synthesis, and qPCR were performed as described previously (37). In brief, total RNA was isolated using Tri Reagent (Sigma), according to the manufacturer's protocol. The RNA was quantified using Nanodrop (Eppendorf, Hamburg, Germany) and 2 μg of RNA per sample was used to prepare cDNA using the Quantitect RT kit (QIAGEN, Valencia, CA). cDNA was amplified in a Realplex mastercycler (Eppendorf) and visualized using a SYBR Green kit (Applied Biosystems, Foster City, CA). Hypoxanthine phosphoribosyl transferase (Hprt) was used as an endogenous housekeeping gene control. To compare the expression of Hprt and target genes, the comparative threshold cycle (CT) method was used as described previously (38). ΔCT = absolute CT value − endogenous CT value; and ΔΔCT = ΔCT T3 treatment − ΔCT control. Data are fold change ± sem compared with control. Primer sequences used are described in Supplemental Table 2.
In situ hybridization
In situ hybridization was carried out as previously described (39, 40). Cryostat-cut sections were thaw mounted onto Probe-on plus slides (Electron Microscopy Sciences, Hatfield, PA). Slides were fixed, acetylated, and dehydrated before storage at −70 C. Smo, Ptc, and Shh cRNA probes were generated from transcription competent pGEM-4Z plasmids provided by Martial Ruat (Centre National de la Recherche Scientifique, France). Antisense cRNA probes were transcribed using 35S-labeled uridine-5′-triphosphate (Amersham, Buckinghamshire, UK). Slides were incubated for 18–20 h at 60 C in hybridization buffer containing 35S-uridine-5′-triphosphate-labeled riboprobes at a concentration of 1 × 106 cpm/150 μl. Within an individual experiment, all slides were exposed to the same batch of riboprobe. After hybridization, slides were subjected to ribonuclease A (20 μg/ml) treatment, followed by stringent washes in decreasing concentrations of saline sodium citrate. Slides were air dried and exposed to Biomax film (Kodak, New York, NY) for 3 wk. All slides within a single experiment were exposed to the same film to reduce variability that may arise during autoradiographic film development. Sense riboprobes, or a ribonuclease (40 μg/ml at 37 C for 30 min) prehybridization wash, did not yield significant hybridization (data not shown), confirming the specificity of the signal observed.
Levels of Shh, Ptc, and Smo transcripts were analyzed using the Macintosh-based Scion Image software (Scion, Frederick, MD). To correct for nonlinearity, 14C standards were used for calibration. An equivalent area was outlined for each sample, and optical density measurements (6–8) from both sides of three to four individual sections from each animal were analyzed, from which the mean value was calculated. Shh mRNA expression in embryonic brain was quantitated in the ventral telecephalon (VT), zona limitans intrathalamica (Zli), and hypothalamus (Hyp). Shh transcript expression in the adult brain was analyzed in the vertical limb of the diagonal band (VDB), cingulate cortex, cortical layer V, medial region and lateral region of striatum, and dentate gyrus (DG) subfield of the hippocampus. Smo and Ptc mRNA expression was determined in the embryonic brain within the VT, Zli, Hyp, and cortex. Smo and Ptc mRNA expression was determined in the adult brain within the cortex, medial and lateral striatum, and subventricular zone (SVZ) lining the lateral ventricles and the DG.
Immunofluorescence
Immunofluorescence staining for β-galactosidase was performed on sections from Shh+/LacZ animals. In brief, sections were incubated with primary antibody: goat anti-β-galactosidase (1:250; AbD Serotec, Kidlington, UK) for 3 d at 4 C and were then incubated with a secondary antibody: Alexa Fluor 488-conjugated antigoat (1:250; Invitrogen) for 2 h. Quantitative analysis for β-galactosidase immunopositive cells was performed by an experimenter blind to the treatment conditions, and only those cells that were strongly immunopositive were counted as β-galactosidase positive cells. Double immunofluorescence experiments for β-galactosidase with the neuronal marker neuronal nuclei (NeuN), the chondroitin sulfate proteoglycan NG2, the 2′,3′-cyclic nucleotide 3′-phosphodiesterase RIP, or the glial fibrillary acidic protein (GFAP) were carried out as previously described (35). In brief, sections were incubated with primary antibody cocktails of goat anti-β-galactosidase with mouse anti-NeuN (1:500; Millipore Corp., Bedford, MA) or mouse anti-RIP (1:10; Developmental Studies Hybridoma Bank, Iowa City, IA) along with rabbit anti-NG2 (1:250; Millipore Corp.) or rabbit anti-GFAP (1:250; Millipore Corp.). After incubation with primary antibody for 3 d at 4 C, sections were washed and incubated with a cocktail of secondary antibodies: Alexa Fluor 488-conjugated antigoat (1:250); rhodamine-conjugated antimouse IgG (1:500; Millipore Corp.), and Cy-5-conjugated antirabbit IgG (1:500; Millipore Corp.) for 2 h. Sections were mounted onto slides with Vectashield (Vector Laboratories, Burlingame, CA) and analyzed using a Zeiss Axioplan2 confocal laser scanning microscope (Zeiss, Oberkochen, Germany). Double immunofluorescence experiments on primary cortical neurons for all TR antibodies with the neuronal marker microtubule-associated protein (MAP)-2 were carried out as described above using a combination of all the TR antibodies, i.e. goat anti-TRα, TRβ1, and TRβ2 (1:200 each; Santa Cruz Biotechnology, Inc., Santa Cruz, CA) along with mouse anti-MAP-2 (1:1000; Sigma). Secondary antibodies used were Alexa Fluor 488-conjugated antigoat (1:400) and rhodamine-conjugated antimouse IgG (1:500). Nuclei were counterstained using Hoechst 33342 (Invitrogen).
ChIP assay
ChIP was carried out as described previously (41). Briefly, bilateral cortices were dissected, fixed to cross-link the DNA and the bound proteins. The tissue was dounce homogenized, sonicated, and immunoprecipitated using a pan-acetylation histone 3 (H3) or pan-acetylation histone 4 (H4) antibody (1 μg; Cell Signaling Technology, Beverly, MA). After reverse cross-linking and chromatin precipitation, qPCR analysis was performed within upstream regions of the Shh gene. Putative TR binding sites were analyzed for the 5′ upstream sequence of the rat Shh gene from −6500 to the transcriptional start site using AliBaba 2.1 (http://www.gene-regulation.com/pub/programs.html). Of the several putative TRα and TRβ binding sites identified, a region containing both putative TRα and TRβ binding sites was amplified, and a second region from −184 to the transcriptional start site was amplified. We also performed qPCR analysis to examine possible enrichment of acetylated histones H3 (AcH3) and H4 (AcH4) within upstream regions of the Ptc and Smo genes. In each sample, the results were normalized to a region amplified from the glyceraldehyde-3-phosphate dehydrogenase promoter. Primer sequences used in ChIP experiments are described in Supplemental Table 2.
Statistical analysis
Results were subjected to statistical analysis using Student's unpaired t test for experiments with two groups and one-way ANOVA for experiments with three groups (Prism; GraphPad, San Diego, CA). When two groups compared exhibited unequal variances, statistical analysis was performed using a Student's unpaired t test with a Welch correction. Differences were considered to be statistically significant at P < 0.05.
Results
Maternal hypothyroidism decreases Shh signaling cascade expression in the embryonic rat brain
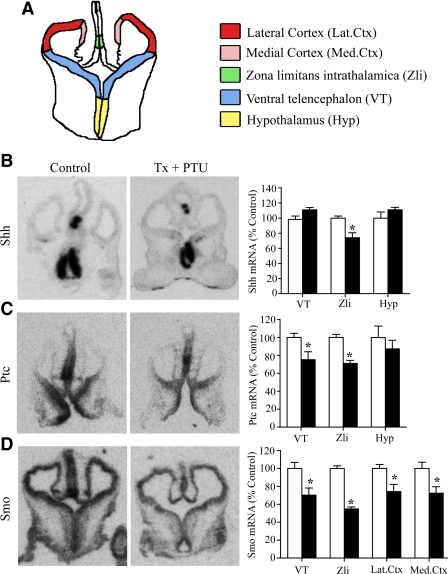
Shh is expressed in ventral signaling areas within the Hyp and ventral telencephalon, where it is critical for the development of the ventral forebrain, specifying cell fate choice, proliferation, and survival (42–44). Shh is also expressed within the Zli, where it regulates the development of adjacent dorsal and ventral thalamic structures (45–48). Although most studies have focused on the consequences of hypothyroidism at late embryonic and postnatal stages in the rodent brain, the fetus that expresses TRs before the onset of fetal thyroid hormone synthesis is exposed to maternal thyroid hormone transported via the placenta from the earliest stages of embryonic forebrain development. We analyzed the consequences of maternal hypothyroidism on the regulation of Shh mRNA and its receptors in the early embryonic brain. E13.5 embryos harvested from hypothyroid dams exhibited a significant down-regulation of Shh mRNA in the Zli (Fig. 1B). Expression of Shh in the ventral telencephalon and the Hyp remained unchanged in these embryos. In addition, Ptc mRNA expression in the embryonic brain was also significantly down-regulated in the ventral telencephalon and in the Zli (Fig. 1C). The Shh signaling receptor, Smo, displayed a down-regulation in a more wide-spread manner, in areas that are responsive to secreted Shh, including the entire thalamus, the ventral telencephalon, and the developing embryonic neocortex (Fig. 1D). Our results indicate that maternal hypothyroidism significantly decreases expression of Shh and its coreceptors Ptc and Smo in the embryonic rodent brain.
Fig. 1.
Maternal hypothyroidism decreases Shh signaling cascade expression in the embryonic rat brain. Rat e13.5 pups were obtained from thyroidectomized females that were administered PTU (Tx + PTU), and levels of Shh, Ptc, and Smo mRNA were determined by in situ hybridization. Shown is a schematic representation of a coronal section through the embryonic rat brain with a color code depicting the regions quantitated (A) and representative autoradiographs of Shh (B), Ptc (C), and Smo (D) mRNA in embryos derived from control and hypothyroid (Tx + PTU) female dams. Maternal hypothyroidism resulted in a significant decrease in Shh mRNA in the Zli with no change in the VT or Hyp (B). Ptc and Smo mRNA were significantly down-regulated in several forebrain regions (namely the VT, Zli, Lat.Ctx and Med.Ctx) of e13.5 embryos derived from hypothyroid dams (C and D). Results are expressed as a percentage of control and are the mean ± sem (n = 11–15 embryos per group derived from four to five dams in each group). *, P < 0.05 compared with embryos derived from control dams (Student's unpaired t test). White bars, Embryos derived from vehicle-treated dams; black bars, embryos derived from Tx + PTU-treated dams.
Maternal hyperthyroidism enhances the expression of Shh in the embryonic rat brain
In contrast to the decreased Shh ligand expression in embryos derived from hypothyroid dams, an opposite effect was seen in embryos obtained from hyperthyroid dams. These embryos exhibited a significant up-regulation in Shh mRNA levels in the major signaling centers of the ventral telencephalon, Zli, and in the hypothalamic region (Fig. 2A). However, we did not observe any change in the expression of the Shh receptors, Ptc and Smo (Fig. 2, B and C). Our results indicate that Shh signaling from embryonic ventral forebrain signaling centers is likely to be enhanced with maternal hyperthyroidism due to a robust up-regulation of the ligand.
Fig. 2.
Maternal hyperthyroidism enhances the expression of Shh in the embryonic rat brain. Rat e13.5 pups were derived from vehicle-treated and chronic T3-treated dams, and the levels of Shh, Ptc, and Smo mRNA were determined by in situ hybridization. Shown are representative autoradiographs showing Shh mRNA expression (A) in the VT, Zli, and Hyp in e13.5 embryos derived from vehicle or T3-treated dams. Maternal hyperthyroidism resulted in a significant induction in Shh mRNA in the VT, Zli, as well as Hyp, of the embryonic brain (A). Ptc and Smo mRNA remained unaltered in embryos derived from maternally hyperthyroid dams (B and C). Results are expressed as a percentage of control and are the mean ± sem (n = 7/group derived from three to four dams in each group). *, P < 0.05 when compared with control (Student's unpaired t test). White bars, embryos derived from vehicle-treated dams; black bars, embryos derived from chronic T3-treated dams.
Adult-onset hypothyroid status selectively decreases Smo mRNA in the DG subfield of the hippocampus
Shh continues to be expressed within specific regions in the adult mammalian brain, including the neocortex, striatum, and the VDB (49). Although Ptc expression is widespread in the adult brain, the signaling coreceptor Smo is predominantly expressed in the adult neurogenic niches, namely the SVZ lining the lateral ventricles and in the DG subfield of the hippocampus (49). Adult hypothyroid status resulted in no change in the expression of Shh mRNA (Supplemental Fig. 1). The expression of Ptc mRNA as well remained unaltered in adult hypothyroid animals (Supplemental Fig. 1). In contrast, adult-onset hypothyroidism significantly down-regulated the expression of the signaling receptor Smo in the DG (Fig. 3B). Interestingly, hypothyroid animals did not show any change in Smo expression within the other major neurogenic niche of the SVZ (Fig. 3B).
Fig. 3.
Adult-onset hypothyroidism decreases Smo mRNA expression in the DG subfield of the hippocampus. Adult male rats were rendered hypothyroid by administration of MMI for 28 d, or PTU for 21 d, and the levels of Smo mRNA were determined by in situ hybridization. Shown are schematic representations of coronal sections through the ventricular zone and hippocampus of the adult rodent brain highlighting the SVZ and the DG region of the hippocampus, respectively (A). Smo mRNA was significantly and selectively down-regulated in the DG after adult-onset hypothyroidism compared with vehicle-treated controls (B). Results are expressed as a percentage of control and are the mean ± sem (n = 4/group). *, P < 0.05 compared with control (one-way ANOVA, Bonferroni post hoc test). White bars, Vehicle-treated controls; gray bars, MMI-treated animals; black bars, PTU-treated animals.
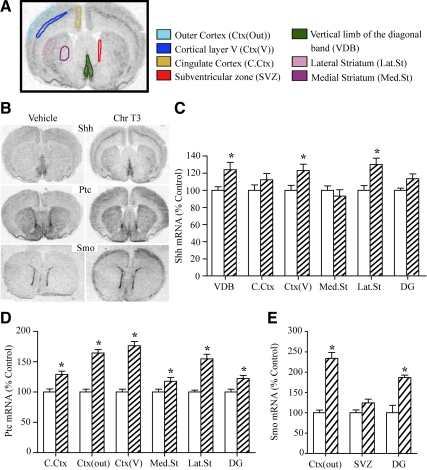
Shh mRNA is significantly up-regulated in the adult rat brain in response to chronic T3 administration
Chronic T3 administration in adulthood resulted in a significant up-regulation of Shh mRNA in several brain regions, including the cortex, VDB, and lateral striatum (Fig. 4, B and C). Further, Ptc mRNA expression was also significantly up-regulated in several regions of the adult rodent brain (Fig. 4, B and D). In addition, mRNA expression of the Shh signaling receptor Smo was also increased in the DG (Fig. 4, B and E). Strikingly, Smo expression was also robustly enhanced in superficial layers of neocortex in hyperthyroid brains, whereas Smo mRNA signal was almost undetectable in euthyroid controls using in situ hybridization (Fig. 4, B and E).
Fig. 4.
Shh mRNA is significantly up-regulated in the adult rat brain in response to chronic T3 administration. Adult male rats received chronic T3 (Chr T3) administration for a period of 10 d to render them hyperthyroid, and the levels of Shh, Ptc, and Smo mRNA were determined by in situ hybridization. Shown is a schematic representation of a coronal section of the adult rat brain with a color code depicting the regions quantitated (A). Also shown are representative autoradiographs of Shh, Ptc, and Smo mRNA expression in coronal sections of the adult brains from vehicle and chronic T3-treated male rats (B). Adult-onset hyperthyroidism resulted in a robust and significant increase in Shh expression in the VDB, cortex, and Lat.St (C). Chronic T3 treatment significantly increased the expression of Ptc (D) and Smo (E) mRNA in the adult rat brain. Results are expressed as a percentage of control and are the mean ± sem (n = 5–6/group). *, P < 0.05 compared with control (Student's unpaired t test). White bars, Vehicle-treated controls; hatched bars, chronic T3-treated animals.
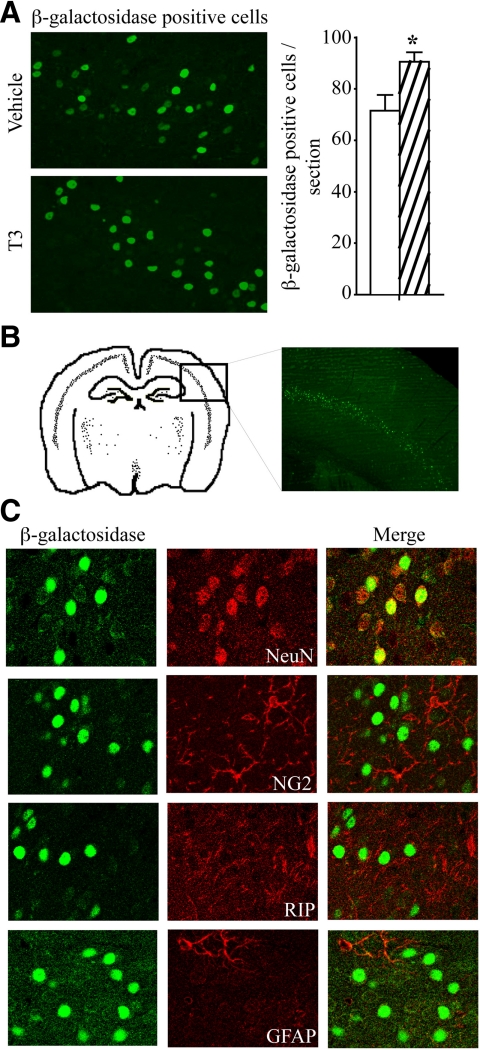
Subchronic T3 administration enhances neocortical β-galactosidase positive cell numbers in Shh+/LacZ mice
Short-duration T3 treatment in Shh+/LacZ mice resulted in a significant increase in the number of β-galactosidase immunopositive cells within the neocortex (Fig. 5A). These results are consistent with the hypothesis that Shh is a thyroid hormone-responsive gene in the adult mammalian brain. To gain an understanding of the cell types that express Shh within the neocortex (Fig. 5B), we performed double labeling experiments for β-galactosidase with neuronal and glial markers. Cells expressing β-galactosidase colocalized with the neuronal marker NeuN (Fig. 5C), indicating presence of the reporter in cortical neurons. We did not observe any β-galactosidase expression within oligodendroglial cells expressing NG2 or RIP or in GFAP-immunopositive astrocytes (Fig. 5C).
Fig. 5.
Subchronic T3 treatment enhances the number of β-galactosidase immunopositive cells within the neocortex of Shh+/LacZ mice. Shown are representative images of β-galactosidase expressing cells in the cortex after short-duration T3 treatment over 2 d in Shh+/LacZ mice. T3 treatment significantly increased the number of cells that were strongly immunopositive for β-galactosidase in layer V of cortex (A). Results are expressed as a percentage of vehicle-treated control and are the mean ± sem (n = 5/group). *, P < 0.05 compared with control (Student's unpaired t test). Shown is a schematic representation (B) of a coronal section illustrating β-galactosidase immunopositive cells in Shh+/LacZ mice. The boxed area is enlarged to show a representative area indicating the presence of several strongly immunopositive β-galactosidase immunopositive cells in the neocortex of T3-treated Shh+/LacZ mice. Double immunofluorescence experiments revealed the colocalization of β-galactosidase with the mature neuronal marker NeuN but not with the astroglial (GFAP) or oligodendrocytic (RIP or NG2) markers examined (C).
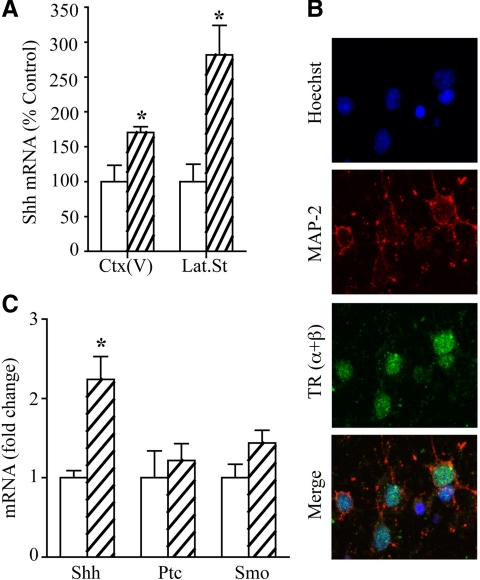
Acute T3 administration induces the expression of Shh mRNA in the adult brain and cortical neurons in vitro
Next, we examined whether acute T3 treatment influences Shh expression in vivo. Acute T3 administration resulted in a significant up-regulation of Shh mRNA in layer V of cortex, as well as the lateral striatum of adult rats, suggesting a rapid transcriptional regulation of Shh mRNA by T3 (Fig. 6A). To address direct effects of T3 on cortical neuronal Shh mRNA levels, we performed qPCR analysis on primary cortical neurons in vitro that were treated with 20 nm T3 for 3 h. Primary cortical cultures significantly up-regulated Shh mRNA in response to acute T3 treatment, supporting a direct effect of T3 on Shh expression (Fig. 6C). Cortical neurons were found to express TRs in vitro as confirmed by immunohistochemistry experiments (Fig. 6B).
Fig. 6.
Acute T3 treatment increases Shh mRNA expression in vivo and in vitro. Adult male rats received a single injection of T3, and the levels of Shh mRNA were determined 3 h later by in situ hybridization. Acute T3 treatment resulted in a significant increase in Shh mRNA expression in the cortex and Lat.St (A). Results are expressed as a percentage of control and are the mean ± sem (n = 3–5/group). *, P < 0.05 compared with vehicle-treated controls (Student's unpaired t test). Ctx(V), Cortical layer V. Shh, Ptc, and Smo mRNA was determined by qPCR from T3-treated primary cortical neuron cultures, isolated from e17.5 pups that were grown in vitro for 9 d before acute treatment with 20 nm T3 for 3 h. Shown are representative images of MAP-2 immunopositive primary cortical neurons expressing all TR isoforms (B) with their nuclei counterstained with Hoechst 33342. Acute T3 treatment of cortical cultures resulted in a significant increase in Shh mRNA expression, whereas Ptc and Smo mRNA remained unchanged as revealed by qPCR analysis (C). All genes were normalized to the housekeeping gene Hprt. Results are expressed as fold change and are the mean ± sem (n = 3–5/group). *, P < 0.05 compared with vehicle-treated controls (Student's unpaired t test).
Thyroid hormone treatment is associated with enhanced histone acetylation of the Shh promoter
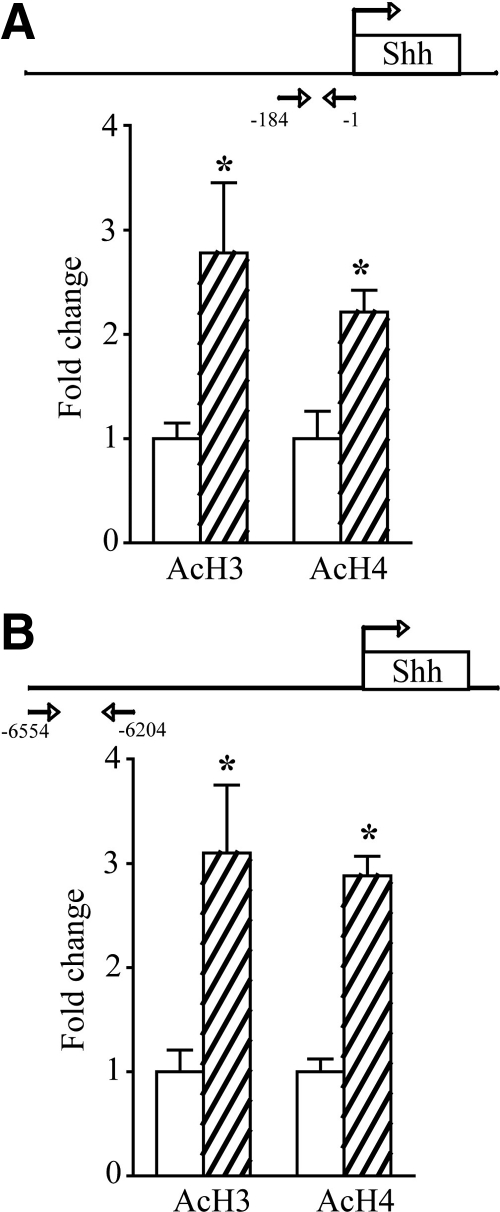
To investigate whether the rapid transcriptional up-regulation of Shh after acute T3 treatment in vivo is associated with enhanced histone acetylation at the Shh promoter, we carried out ChIP assays for AcH3 and AcH4 within upstream regions of the Shh gene in cortical tissue. After ChIP, we performed qPCR analysis for upstream regions of the Shh gene that contained putative thyroid hormone response elements (TREs) (−6423 to −6413 bp and −6270 to −6261 bp) and also within the first 200 bp from the transcriptional start site. Our results revealed that neocortical tissue derived from T3-treated animals had significantly enhanced acetylation of both histone H3 and H4 at putative TRE-containing regions upstream of the Shh gene and close to the transcriptional start site in the Shh promoter (Fig. 7, A and B).
Fig. 7.
Acute T3 treatment increases histone acetylation within upstream regulatory regions of the Shh gene in the adult rat cortex. ChIP assays were performed for pan histone H3 and H4 acetylation changes within gene regulatory sequences from −184 bp to the transcriptional start site of the Shh gene. Cortical tissue derived from T3-treated animals had significantly enhanced acetylation of histone H3 and H4 (AcH3 and AcH4) close to the transcriptional start site within the Shh promoter after acute T3 treatment (A). ChIP analysis within upstream regions of the Shh gene that contained putative TREs (−6554 to −6204 bp) revealed that cortical tissue derived from T3-treated animals had increased acetylation of both histone H3 and H4 within these regulatory sequences as well (B). Results are expressed as fold change and are the mean ± sem (n = 7–10/group). *, P < 0.05 compared with vehicle-treated controls (Student's unpaired t test).
We next examined whether acute T3 treatment influenced acetylation within the first 200 bp from the transcriptional start site of both the Ptc and Smo genes. We observed no change in the enrichment of AcH3 and AcH4 within Ptc or Smo gene upstream regulatory regions. AcH3 at the Ptc and Smo promoter, fold change (Ptc): control, 1 ± 0.39; T3 treated, 0.75 ± 0.44; fold change (Smo): control, 1 ± 0.24; T3 treated, 1.41 ± 0.40. AcH4 at the Ptc and Smo promoter, fold change (Ptc): control, 1 ± 0.34; T3 treated, 1.31 ± 0.52; fold change (Smo): control, 1 ± 0.22; T3 treated, 0.97 ± 0.35. Data are the mean ± sem (P > 0.05, Student's t test). Taken together, these data indicate that the enhanced expression of Shh mRNA in the cortex is accompanied by significant increases in histone acetylation levels within Shh, but not Ptc or Smo, transcriptional regulatory sequences.
Discussion
Thyroid hormone is known to exert profound effects on neurodevelopment and retains a powerful influence on plasticity within the mature brain, including key effects on adult hippocampal neurogenesis (13, 15, 50, 51). Although several thyroid hormone-responsive genes, such as reelin, Dab-1, and Neurogenic differentiation, have been hypothesized to mediate the neurodevelopmental influences of thyroid hormone, most of these genes are transcriptionally responsive to thyroid hormone only during select developmental time windows (22, 52). In contrast, there are relatively few genes that retain thyroid hormone responsivity across the life span, for example neuroendocrine specific protein and octamer transcription factor-1, which are reported to be regulated in both the embryonic and adult brain (24). Such thyroid hormone-responsive genes are particularly interesting, because they are candidates to mediate the effects of thyroid hormone in both the developing and mature brain. Although the effects of thyroid hormone are spatiotemporally specific, it is quite possible that the same sets of target genes may be redeployed across the lifespan to mediate diverse developmental and neuroplastic consequences. We provide novel evidence that the major developmental morphogen Shh is a thyroid hormone-responsive gene from the very earliest stages of embryonic forebrain development into adulthood.
Perhaps the most dramatic example of the tissue morphoregulatory effects of thyroid hormone are those observed during amphibian metamorphosis, a process entirely dependent on the induction of a genetic program by thyroid hormone (53). Previous evidence indicates that Xhh is an early, direct target gene of thyroid hormone and is required to mediate the metamorphic effects of thyroid hormone in the gastrointestinal system (33). It is interesting to note that thyroid hormone-mediated induction of Xhh in tadpoles is tissue-specific with no effects observed in the brain (33). In contrast, we find that in the embryonic and adult mammalian brain, thyroid hormone perturbations induce a bidirectional regulation of the Shh signaling cascade in a region-specific manner. Although TRs are known to be expressed in the fetal brain well before the initiation of embryonic thyroid hormone synthesis (7, 9), and epidemiological evidence indicates that thyroid hormone perturbations in the first trimester influence fetal brain development (1, 54), so far, only few thyroid hormone-responsive genes within the fetus have been identified (55, 56). Our results indicate that maternal hyperthyroidism significantly up-regulates embryonic Shh expression in ventral forebrain signaling areas and the diencephalic signaling center of the Zli, and maternal hypothyroidism results in a region-specific decline of Shh within the embryonic Zli. This suggests that effective Shh signaling from ventral forebrain signaling areas is likely to be enhanced with maternal hyperthyroidism due to a robust up-regulation of the ligand, and reduced with maternal hypothyroidism, due to down-regulation of the receptor complex as well as reduced ligand from the Zli.
Our results raise the possibility that specific neurodevelopmental actions of thyroid hormone may be mediated by its control of the Shh pathway, providing a mechanistic link between thyroid hormone perturbations and early developmental effects. For example, maternal thyroid hormone deficiency is associated with impaired cortical interneuron development, in particular of parvalbumin and calretinin immunoreactive neocortical interneurons (10, 11, 57). It is particularly intriguing in this regard to note that reduced Shh signaling in the embryonic ventral forebrain alters cortical interneuron composition, reducing parvalbumin positive interneuron number (58).
Adult-onset perturbations in thyroid hormone status also significantly regulated the expression of Shh signaling components in a bidirectional fashion, suggesting a continued sensitivity of Shh expression to thyroid hormone into adulthood. T3 administration resulted in enhanced Shh mRNA levels within the adult neocortex, VDB, and lateral striatum, accompanied by significant increases in the expression of both Ptc and Smo mRNA. Excess thyroid hormone levels also appear to unmask Smo expression within the neocortex, a brain region that normally has very low levels of Smo mRNA in euthyroid controls, and this could serve to potentiate the thyroid hormone-mediated effects on the Shh signaling pathway through increased expression of both the ligand as well as the signaling receptor. The enhanced Ptc expression observed in T3-treated adult animals could be interpreted as reflective of such increases in Shh signaling, because Ptc is a transcriptional target of Shh (59, 60). T3 administration also significantly enhanced Smo mRNA within the hippocampal neurogenic niche. It is possible that enhanced Shh ligand from the VDB may be transported via the septo-hippocampal pathway resulting in increased Shh signaling within the hippocampus (61), which is supported by the evidence of enhanced Ptc expression in the DG. In this context, it is noteworthy that T3 administration is capable of increasing the postmitotic survival and neuronal differentiation of hippocampal progenitors (62), an effect that overlaps with the enhanced survival observed after treatment with Smo agonists (63).
Although adult hyperthyroidism significantly enhanced Shh ligand expression, we did not observe any change in the ligand in adult-onset hypothyroid animals within the brain regions examined. It is possible to speculate that despite reduced circulating thyroid hormone levels, thyroid hormone availability in specific adult brain regions may not exhibit as steep a decline through the modulation of local deiodinase expression and activity. It is possible that the relative extent of unliganded TR repressor effects on Shh mRNA expression may still be minimal in the adult-onset hypothyroid brain. However, a single previous study indicates that postnatal hypothyroidism reduces Shh expression in the cerebellum, an effect suggested to contribute to the cerebellar morphological defects observed with postnatal hypothyroidism (64). In contrast to the extensive effects of adult-onset hyperthyroidism on expression of the Shh cascade, hypothyroidism in adulthood resulted in a spatially restricted and selective down-regulation of Smo expression within the DG. Adult-onset hypothyroidism is known to result in a neurogenic decline, through a reduced proliferation, survival, and neuronal differentiation of hippocampal progenitors (12, 13, 15). Shh is essential to the maintenance of the adult hippocampal stem cell niche and is known to modulate the proliferation, survival, and maintenance of adult progenitors (29, 31). We have previously demonstrated an effect of adult-onset hypothyroidism on hippocampal progenitors (13). Hypothyroidism resulted in a significant decrease in Smo mRNA levels selectively in the DG, raising the intriguing possibility that the neurogenic decline observed in hypothyroid animals may be mediated through a reduced responsivity to Shh signaling.
Subchronic T3 treatment also enhanced the number of β-galactosidase immunopositive cells within the neocortex of Shh+/LacZ reporter mice, and colocalization experiments revealed that the expression of β-galactosidase was neuronal. We did not observe β-galactosidase expression in GFAP-positive astrocytes or NG2/RIP immunopositive oligodendrocytes in the neocortex. Recent studies suggest that thyroid hormone enhances oligodendrocytic progenitor recruitment and maturation to promote recovery in animal models of demyelination (65–67), and in some cases, it is accompanied by enhanced Shh expression (65). Further, under conditions of damage, reactive astrocytes within the cortex are reported to express Shh and induce oligodendrocytic progenitor proliferation (68). Shh administration into the neocortex of normal animals is also capable of recruiting premyelinating oligodendrocytic progenitors (30, 69). Our results indicate that T3 administration in normal animals induces neuronal Shh expression, and this may serve to recruit premyelinating oligodendrocytic progenitors even in the absence of overt damage.
The rapid effects of acute T3 administration on increased Shh expression in vivo and in cortical neurons in vitro suggest that the regulation of Shh expression by thyroid hormone in the brain may involve direct mechanisms. In contrast, the enhanced Smo and Ptc expression observed in adult-onset hyperthyroid animals is more likely to be a secondary consequence of changes in expression of the ligand. In silico analysis indicated the presence of putative TREs within upstream regulatory sequences of the Shh gene. Thyroid hormone mediated transcriptional regulation is associated with recruitment of coactivators and targeted histone acetylation changes in upstream regions of thyroid hormone-responsive genes (70). Previous studies from Xenopus demonstrate that T3 bound to TRs can recruit histone acetyl transferases to its target genes (71, 72), and the expression of thyroid hormone-responsive genes in Xenopus has also been shown to correlate with the acetylation status of their promoters (73). Our results demonstrate significant and rapid in vivo increases in global histone H3 and H4 acetylation both at the putative TRE sites within upstream regions of the Shh gene, as well as close to the transcriptional start site. Although evidence of rapid increases in histone acetylation provides partial support to the notion that Shh is directly transcriptionally regulated by thyroid hormone, future experiments are required to identify promoter elements that mediate thyroid hormone responsivity of the Shh gene. Further experiments to elucidate the role of select TR isoforms in thyroid hormone-mediated Shh transcriptional regulation, to address whether TRs exert their effects as homodimers or heterodimers with RXR, RAR, or TRAP, and to identify the TREs or retinoic acid response elements that mediate TH responsivity are required to unequivocally demonstrate that Shh is a direct thyroid hormone target gene.
To the best of our knowledge, we provide the first evidence that Shh expression is robustly regulated by thyroid hormone in the brain. Recent elegant studies have demonstrated an opposite regulation, showing that Shh signaling can modulate thyroid hormone activity in neuronal cells through the regulation of deiodinases 2 and 3. Shh has been reported to reduce thyroid hormone signaling through decreased deiodinase 2 and enhanced deiodinase 3 activity in astrocytes and neurons, respectively (74). This then evokes the tantalizing but speculative possibility of a negative feedback loop wherein thyroid hormone enhances neuronal Shh signaling and Shh serves to restrict thyroid hormone-mediated effects via deiodinase activity modulation. This raises the possibility of a powerful interplay between these two systems that may then play an important role in neurodevelopment, adult plasticity, and pathophysiology.
In conclusion, we find that the Shh pathway displays a striking and selective regulation within the adult brain and in distinct signaling areas of the early embryonic forebrain under conditions of adult-onset, and maternal, hyper- and hypothyroidism. We show that Shh expression is rapidly regulated both in vivo and in vitro in response to thyroid hormone, accompanied by significant epigenetic histone modifications within upstream regions of the Shh gene. Both thyroid hormone and Shh have been independently demonstrated to regulate diverse aspects of central nervous system development, including effects on proliferation and maturation, and to retain a profound influence on the adult brain, modulating recruitment, survival, and differentiation of neuronal and oligodendrocytic progenitors (13, 14, 29, 31). Our results suggest that specific developmental and plasticity associated actions of thyroid hormone may be mediated by its control of the Shh pathway and motivate future experiments to determine the mechanistic contribution of Shh to the effects of thyroid hormone in both the developing and mature nervous system.
Supplementary Material
Acknowledgments
We thank S. Agashe and S. Banerjee for technical assistance.
This work was supported by intramural funds from Tata Institute of Fundamental Research and by Wellcome Trust Senior Overseas Fellowships in Biomedical Sciences 04082003114133 (to V.A.V.) and 056684/Z/99/Z (to S.T.).
Disclosure Summary: The authors have nothing to disclose.
Footnotes
- AcH3
- Acetylated H3
- AcH4
- acetylated H4
- ChIP
- chromatin immunoprecipitation
- CT
- threshold cycle
- DG
- dentate gyrus
- e
- embryonic day
- GFAP
- glial fibrillary acidic protein
- H3
- histone 3
- H4
- histone 4
- HPRT
- hypoxanthine phosphoribosyl transferase
- Hyp
- hypothalamus
- MAP
- microtubule-associated protein
- MMI
- 2-mercapto-1-methylimidazole
- NeuN
- neuronal nuclei
- Ptc
- patched
- PTU
- 6-n-propyl-2-thiouracil
- qPCR
- quantitative PCR
- Shh
- sonic hedgehog
- Smo
- smoothened
- SVZ
- subventricular zone
- TR
- thyroid hormone receptor
- TRE
- thyroid hormone response element
- VDB
- vertical limb of the diagonal band
- VT
- ventral telecephalon
- Xhh
- Xenopus hedgehog
- Zli
- zona limitans intrathalamica.
References
- 1. Porterfield SP, Hendrich CE. 1993. The role of thyroid hormones in prenatal and neonatal neurological development—current perspectives. Endocr Rev 14:94–106 [DOI] [PubMed] [Google Scholar]
- 2. Anderson GW, Schoonover CM, Jones SA. 2003. Control of thyroid hormone action in the developing rat brain. Thyroid 13:1039–1056 [DOI] [PubMed] [Google Scholar]
- 3. Anderson GW. 2001. Thyroid hormones and the brain. Front Neuroendocrinol 22:1–17 [DOI] [PubMed] [Google Scholar]
- 4. Akaike M, Kato N, Ohno H, Kobayashi T. 1991. Hyperactivity and spatial maze learning impairment of adult rats with temporary neonatal hypothyroidism. Neurotoxicol Teratol 13:317–322 [DOI] [PubMed] [Google Scholar]
- 5. Williams GR. 2008. Neurodevelopmental and neurophysiological actions of thyroid hormone. J Neuroendocrinol 20:784–794 [DOI] [PubMed] [Google Scholar]
- 6. Landers KA, McKinnon BD, Li H, Subramaniam VN, Mortimer RH, Richard K. 2009. Carrier-mediated thyroid hormone transport into placenta by placental transthyretin. J Clin Endocrinol Metab 94:2610–2616 [DOI] [PubMed] [Google Scholar]
- 7. Iskaros J, Pickard M, Evans I, Sinha A, Hardiman P, Ekins R. 2000. Thyroid hormone receptor gene expression in first trimester human fetal brain. J Clin Endocrinol Metab 85:2620–2623 [DOI] [PubMed] [Google Scholar]
- 8. Morreale de Escobar G, Obregon MJ, Escobar del Rey F. 2004. Role of thyroid hormone during early brain development. Eur J Endocrinol 151(Suppl 3):U25–U37 [DOI] [PubMed] [Google Scholar]
- 9. Bradley DJ, Towle HC, Young WS., 3rd 1992. Spatial and temporal expression of α- and β-thyroid hormone receptor mRNAs, including the β2-subtype, in the developing mammalian nervous system. J Neurosci 12:2288–2302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Cuevas E, Ausó E, Telefont M, Morreale de Escobar G, Sotelo C, Berbel P. 2005. Transient maternal hypothyroxinemia at onset of corticogenesis alters tangential migration of medial ganglionic eminence-derived neurons. Eur J Neurosci 22:541–551 [DOI] [PubMed] [Google Scholar]
- 11. Ausó E, Lavado-Autric R, Cuevas E, Del Rey FE, Morreale De Escobar G, Berbel P. 2004. A moderate and transient deficiency of maternal thyroid function at the beginning of fetal neocorticogenesis alters neuronal migration. Endocrinology 145:4037–4047 [DOI] [PubMed] [Google Scholar]
- 12. Ambrogini P, Cuppini R, Ferri P, Mancini C, Ciaroni S, Voci A, Gerdoni E, Gallo G. 2005. Thyroid hormones affect neurogenesis in the dentate gyrus of adult rat. Neuroendocrinology 81:244–253 [DOI] [PubMed] [Google Scholar]
- 13. Desouza LA, Ladiwala U, Daniel SM, Agashe S, Vaidya RA, Vaidya VA. 2005. Thyroid hormone regulates hippocampal neurogenesis in the adult rat brain. Mol Cell Neurosci 29:414–426 [DOI] [PubMed] [Google Scholar]
- 14. Fernandez M, Pirondi S, Manservigi M, Giardino L, Calzà L. 2004. Thyroid hormone participates in the regulation of neural stem cells and oligodendrocyte precursor cells in the central nervous system of adult rat. Eur J Neurosci 20:2059–2070 [DOI] [PubMed] [Google Scholar]
- 15. Montero-Pedrazuela A, Venero C, Lavado-Autric R, Fernández-Lamo I, García-Verdugo JM, Bernal J, Guadaño-Ferraz A. 2006. Modulation of adult hippocampal neurogenesis by thyroid hormones: implications in depressive-like behavior. Mol Psychiatry 11:361–371 [DOI] [PubMed] [Google Scholar]
- 16. Billon N, Jolicoeur C, Tokumoto Y, Vennström B, Raff M. 2002. Normal timing of oligodendrocyte development depends on thyroid hormone receptor α1 (TRα1). EMBO J 21:6452–6460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Rodríguez-Peña A. 1999. Oligodendrocyte development and thyroid hormone. J Neurobiol 40:497–512 [DOI] [PubMed] [Google Scholar]
- 18. Jones SA, Jolson DM, Cuta KK, Mariash CN, Anderson GW. 2003. Triiodothyronine is a survival factor for developing oligodendrocytes. Mol Cell Endocrinol 199:49–60 [DOI] [PubMed] [Google Scholar]
- 19. Rivas M, Naranjo JR. 2007. Thyroid hormones, learning and memory. Genes Brain Behav 6(Suppl 1):40–44 [DOI] [PubMed] [Google Scholar]
- 20. Yen PM. 2001. Physiological and molecular basis of thyroid hormone action. Physiol Rev 81:1097–1142 [DOI] [PubMed] [Google Scholar]
- 21. Farwell AP, Dubord-Tomasetti SA. 1999. Thyroid hormone regulates the expression of laminin in the developing rat cerebellum. Endocrinology 140:4221–4227 [DOI] [PubMed] [Google Scholar]
- 22. Alvarez-Dolado M, Ruiz M, Del Río JA, Alcántara S, Burgaya F, Sheldon M, Nakajima K, Bernal J, Howell BW, Curran T, Soriano E, Muñoz A. 1999. Thyroid hormone regulates reelin and dab1 expression during brain development. J Neurosci 19:6979–6993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Alvarez-Dolado M, Figueroa A, Kozlov S, Sonderegger P, Furley AJ, Muñoz A. 2001. Thyroid hormone regulates TAG-1 expression in the developing rat brain. Eur J Neurosci 14:1209–1218 [DOI] [PubMed] [Google Scholar]
- 24. Dowling AL, Martz GU, Leonard JL, Zoeller RT. 2000. Acute changes in maternal thyroid hormone induce rapid and transient changes in gene expression in fetal rat brain. J Neurosci 20:2255–2265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Royland JE, Parker JS, Gilbert ME. 2008. A genomic analysis of subclinical hypothyroidism in hippocampus and neocortex of the developing rat brain. J Neuroendocrinol 20:1319–1338 [DOI] [PubMed] [Google Scholar]
- 26. Horn S, Heuer H. 2010. Thyroid hormone action during brain development: more questions than answers. Mol Cell Endocrinol 315:19–26 [DOI] [PubMed] [Google Scholar]
- 27. Diez D, Grijota-Martinez C, Agretti P, De Marco G, Tonacchera M, Pinchera A, de Escobar GM, Bernal J, Morte B. 2008. Thyroid hormone action in the adult brain: gene expression profiling of the effects of single and multiple doses of triiodo-L-thyronine in the rat striatum. Endocrinology 149:3989–4000 [DOI] [PubMed] [Google Scholar]
- 28. Charytoniuk D, Porcel B, Rodríguez Gomez J, Faure H, Ruat M, Traiffort E. 2002. Sonic Hedgehog signalling in the developing and adult brain. J Physiol Paris 96:9–16 [DOI] [PubMed] [Google Scholar]
- 29. Lai K, Kaspar BK, Gage FH, Schaffer DV. 2003. Sonic hedgehog regulates adult neural progenitor proliferation in vitro and in vivo. Nat Neurosci 6:21–27 [DOI] [PubMed] [Google Scholar]
- 30. Nery S, Wichterle H, Fishell G. 2001. Sonic hedgehog contributes to oligodendrocyte specification in the mammalian forebrain. Development 128:527–540 [DOI] [PubMed] [Google Scholar]
- 31. Palma V, Lim DA, Dahmane N, Sánchez P, Brionne TC, Herzberg CD, Gitton Y, Carleton A, Alvarez-Buylla A, Ruiz i Altaba A. 2005. Sonic hedgehog controls stem cell behavior in the postnatal and adult brain. Development 132:335–344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ingham PW, McMahon AP. 2001. Hedgehog signaling in animal development: paradigms and principles. Genes Dev 15:3059–3087 [DOI] [PubMed] [Google Scholar]
- 33. Stolow MA, Shi YB. 1995. Xenopus sonic hedgehog as a potential morphogen during embryogenesis and thyroid hormone-dependent metamorphosis. Nucleic Acids Res 23:2555–2562 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Kolterud A, Grosse AS, Zacharias WJ, Walton KD, Kretovich KE, Madison BB, Waghray M, Ferris JE, Hu C, Merchant JL, Dlugosz AA, Kottmann AH, Gumucio DL. 2009. Paracrine Hedgehog signaling in stomach and intestine: new roles for hedgehog in gastrointestinal patterning. Gastroenterology 137:618–628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Lewis PM, Gritli-Linde A, Smeyne R, Kottmann A, McMahon AP. 2004. Sonic hedgehog signaling is required for expansion of granule neuron precursors and patterning of the mouse cerebellum. Dev Biol 270:393–410 [DOI] [PubMed] [Google Scholar]
- 36. Joseph LJ PO, Bhatkar SV, Nabar SJ, Samuel AM. 1996. Effect of ferrous sulfate on thyroxine replacement therapy in hypothyroid rats Med Sci Res 24:123–125 [Google Scholar]
- 37. Benekareddy M, Goodfellow NM, Lambe EK, Vaidya VA. 2010. Enhanced function of prefrontal serotonin 5-HT(2) receptors in a rat model of psychiatric vulnerability. J Neurosci 30:12138–12150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Bookout AL, Mangelsdorf DJ. 2003. Quantitative real-time PCR protocol for analysis of nuclear receptor signaling pathways. Nucl Recept Signal 1:e012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Rajendran R, Jha S, Fernandes KA, Banerjee SB, Mohammad F, Dias BG, Vaidya VA. 2009. Monoaminergic regulation of Sonic hedgehog signaling cascade expression in the adult rat hippocampus. Neurosci Lett 453:190–194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Banerjee SB, Rajendran R, Dias BG, Ladiwala U, Tole S, Vaidya VA. 2005. Recruitment of the Sonic hedgehog signalling cascade in electroconvulsive seizure-mediated regulation of adult rat hippocampal neurogenesis. Eur J Neurosci 22:1570–1580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Wilkinson MB, Xiao G, Kumar A, LaPlant Q, Renthal W, Sikder D, Kodadek TJ, Nestler EJ. 2009. Imipramine treatment and resiliency exhibit similar chromatin regulation in the mouse nucleus accumbens in depression models. J Neurosci 29:7820–7832 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Ishibashi M, McMahon AP. 2002. A sonic hedgehog-dependent signaling relay regulates growth of diencephalic and mesencephalic primordia in the early mouse embryo. Development 129:4807–4819 [DOI] [PubMed] [Google Scholar]
- 43. Bertrand N, Dahmane N. 2006. Sonic hedgehog signaling in forebrain development and its interactions with pathways that modify its effects. Trends Cell Biol 16:597–605 [DOI] [PubMed] [Google Scholar]
- 44. Rash BG, Grove EA. 2007. Patterning the dorsal telencephalon: a role for sonic hedgehog? J Neurosci 27:11595–11603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Blackshaw S, Scholpp S, Placzek M, Ingraham H, Simerly R, Shimogori T. 2010. Molecular pathways controlling development of thalamus and hypothalamus: from neural specification to circuit formation. J Neurosci 30:14925–14930 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Vieira C, Garda AL, Shimamura K, Martinez S. 2005. Thalamic development induced by Shh in the chick embryo. Dev Biol 284:351–363 [DOI] [PubMed] [Google Scholar]
- 47. Larsen CW, Zeltser LM, Lumsden A. 2001. Boundary formation and compartition in the avian diencephalon. J Neurosci 21:4699–4711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Wilson SW, Houart C. 2004. Early steps in the development of the forebrain. Dev Cell 6:167–181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Traiffort E, Charytoniuk DA, Faure H, Ruat M. 1998. Regional distribution of sonic hedgehog, patched, and smoothened mRNA in the adult rat brain. J Neurochem 70:1327–1330 [DOI] [PubMed] [Google Scholar]
- 50. Bernal J. 2002. Action of thyroid hormone in brain. J Endocrinol Invest 25:268–288 [DOI] [PubMed] [Google Scholar]
- 51. Nunez J, Celi FS, Ng L, Forrest D. 2008. Multigenic control of thyroid hormone functions in the nervous system. Mol Cell Endocrinol 287:1–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Chantoux F, Francon J. 2002. Thyroid hormone regulates the expression of NeuroD/BHF1 during the development of rat cerebellum. Mol Cell Endocrinol 194:157–163 [DOI] [PubMed] [Google Scholar]
- 53. Shi YB, Wong J, Puzianowska-Kuznicka M, Stolow MA. 1996. Tadpole competence and tissue-specific temporal regulation of amphibian metamorphosis: roles of thyroid hormone and its receptors. Bioessays 18:391–399 [DOI] [PubMed] [Google Scholar]
- 54. Oppenheimer JH, Schwartz HL. 1997. Molecular basis of thyroid hormone-dependent brain development. Endocr Rev 18:462–475 [DOI] [PubMed] [Google Scholar]
- 55. Sinha RA, Pathak A, Mohan V, Bandyopadhyay S, Rastogi L, Godbole MM. 2008. Maternal thyroid hormone: a strong repressor of neuronal nitric oxide synthase in rat embryonic neocortex. Endocrinology 149:4396–4401 [DOI] [PubMed] [Google Scholar]
- 56. Dowling AL, Iannacone EA, Zoeller RT. 2001. Maternal hypothyroidism selectively affects the expression of neuroendocrine-specific protein A messenger ribonucleic acid in the proliferative zone of the fetal rat brain cortex. Endocrinology 142:390–399 [DOI] [PubMed] [Google Scholar]
- 57. Berbel P, Marco P, Cerezo JR, DeFelipe J. 1996. Distribution of parvalbumin immunoreactivity in the neocortex of hypothyroid adult rats. Neurosci Lett 204:65–68 [DOI] [PubMed] [Google Scholar]
- 58. Xu Q, Guo L, Moore H, Waclaw RR, Campbell K, Anderson SA. 2010. Sonic hedgehog signaling confers ventral telencephalic progenitors with distinct cortical interneuron fates. Neuron 65:328–340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Marigo V, Tabin CJ. 1996. Regulation of patched by sonic hedgehog in the developing neural tube. Proc Natl Acad Sci USA 93:9346–9351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Charytoniuk D, Traiffort E, Hantraye P, Hermel JM, Galdes A, Ruat M. 2002. Intrastriatal sonic hedgehog injection increases Patched transcript levels in the adult rat subventricular zone. Eur J Neurosci 16:2351–2357 [DOI] [PubMed] [Google Scholar]
- 61. Traiffort E, Moya KL, Faure H, Hässig R, Ruat M. 2001. High expression and anterograde axonal transport of aminoterminal sonic hedgehog in the adult hamster brain. Eur J Neurosci 14:839–850 [DOI] [PubMed] [Google Scholar]
- 62. Kapoor R, van Hogerlinden M, Wallis K, Ghosh H, Nordstrom K, Vennstrom B, Vaidya VA. 2010. Unliganded thyroid hormone receptor α1 impairs adult hippocampal neurogenesis. FASEB J 24:4793–4805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Bragina O, Sergejeva S, Serg M, Zarkovsky T, Maloverjan A, Kogerman P, Zarkovsky A. 2010. Smoothened agonist augments proliferation and survival of neural cells. Neurosci Lett 482:81–85 [DOI] [PubMed] [Google Scholar]
- 64. Hasebe M, Ohta E, Imagawa T, Uehara M. 2008. Expression of sonic hedgehog regulates morphological changes of rat developing cerebellum in hypothyroidism. J Toxicol Sci 33:473–477 [DOI] [PubMed] [Google Scholar]
- 65. Harsan LA, Steibel J, Zaremba A, Agin A, Sapin R, Poulet P, Guignard B, Parizel N, Grucker D, Boehm N, Miller RH, Ghandour MS. 2008. Recovery from chronic demyelination by thyroid hormone therapy: myelinogenesis induction and assessment by diffusion tensor magnetic resonance imaging. J Neurosci 28:14189–14201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Calza L, Fernandez M, Giuliani A, Aloe L, Giardino L. 2002. Thyroid hormone activates oligodendrocyte precursors and increases a myelin-forming protein and NGF content in the spinal cord during experimental allergic encephalomyelitis. Proc Natl Acad Sci USA 99:3258–3263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Fernandez M, Giuliani A, Pirondi S, D'Intino G, Giardino L, Aloe L, Levi-Montalcini R, Calzà L. 2004. Thyroid hormone administration enhances remyelination in chronic demyelinating inflammatory disease. Proc Natl Acad Sci USA 101:16363–16368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Amankulor NM, Hambardzuman D, Pyonteck SM, Becher OJ, Joyce JA, Holland EC. 2009. Sonic hedgehog pathway activation is induced by acute brain injury and regulated by injury-related inflammation. J Neurosci 29:10299–10308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Loulier K, Ruat M, Traiffort E. 2006. Increase of proliferating oligodendroglial progenitors in the adult mouse brain upon sonic hedgehog delivery in the lateral ventricle. J Neurochem 98:530–542 [DOI] [PubMed] [Google Scholar]
- 70. Lee KC, Li J, Cole PA, Wong J, Kraus WL. 2003. Transcriptional activation by thyroid hormone receptor-β involves chromatin remodeling, histone acetylation, and synergistic stimulation by p300 and steroid receptor coactivators. Mol Endocrinol 17:908–922 [DOI] [PubMed] [Google Scholar]
- 71. Chen JD, Li H. 1998. Coactivation and corepression in transcriptional regulation by steroid/nuclear hormone receptors. Crit Rev Eukaryot Gene Expr 8:169–190 [DOI] [PubMed] [Google Scholar]
- 72. Xu L, Glass CK, Rosenfeld MG. 1999. Coactivator and corepressor complexes in nuclear receptor function. Curr Opin Genet Dev 9:140–147 [DOI] [PubMed] [Google Scholar]
- 73. Sachs LM, Shi YB. 2000. Targeted chromatin binding and histone acetylation in vivo by thyroid hormone receptor during amphibian development. Proc Natl Acad Sci USA 97:13138–13143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Freitas BC, Gereben B, Castillo M, Kalló I, Zeöld A, Egri P, Liposits Z, Zavaki AM, Maciel RM, Jo S, Singru P, Sanchez E, Lechan RM, Bianco AC. 2010. Paracrine signaling by glial cell-derived triiodothyronine activates neuronal gene expression in the rodent brain and human cells. J Clin Invest 120:2206–2217 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.