Abstract
Hapten induced contact hypersensitivity (CHS) in the skin is a delayed type cellular immune response which can be mediated by CD8+ T cells that produce IFN-γ or IL-17. However, mechanisms for these cytokines in the elicitation of CHS remain to be fully elucidated. Here we show that adoptive transfer of CHS with hapten primed wild type CD8+ T cells is reduced in IFN-γR−/− or IL-17R−/− mice compared to wild type controls. The infiltration of granulocytes and macrophages in the hapten challenged skin of IL-17R−/− recipients is significantly reduced whereas it is less affected in IFN-γR−/− recipients although CD8+ T cell infiltration is inhibited in both recipients. In contrast, the activity of reactive oxidative species is significantly inhibited in IFN-γR−/− but is less affected in IL-17R−/− recipients. Further analysis reveals that the expression of chemokines and cytokines is differentially regulated in the hapten challenged skin of IFN-γR−/− or IL-17R−/− recipients compared to wild type controls. Interestingly, injection of recombinant IL-17 in the skin induces inflammation with a high level of leukocyte infiltration whereas injection of IFN-γ induces inflammation with a high level of reactive oxidative species. Moreover, neutralization of IL-17 in IFN-γR−/− or IFN-γ in IL-17R−/− mice further suppresses the adoptive transfer of CHS by hapten primed wild type CD8+ T cells. The study demonstrates that IFN-γ and IL-17 mediate the elicitation of CHS by different mechanisms and that both cytokines are required for optimal responses. This outcome improves understanding of pathogenesis and provides new insights into therapeutic strategies for CHS.
Keywords: Rodent, Skin, T cells, Inflammation, Cytokine
INTRODUCTION
Hapten induced contact hypersensitivity (CHS) is a T cell mediated delayed type immune responses, which used to be considered a Th1 mediated delayed type hypersensitivity in the skin. In animal models, hapten primed CD8+ T cells produce a large amount of IFN-γ and are primary effector cells (Tc1) whereas CD4+ cells develop into Th2 type cells which play regulatory roles in the responses (1–3). Recent studies have demonstrated that hapten primed CD8+ T cells are able to develop into a distinct subpopulation that produces IL-17 (TIL-17) and plays a role in the elicitation of CHS responses (4). CHS responses are suppressed in IL-17 deficient mice, which results from a defect in the activation of CD4+ T cells and reduction of IFN-γ by hapten primed T cells (5). Clinical evidence also indicates that T cells from nickel allergic patients produce both IFN-γ and IL-17 (6). However, it remains to be determined whether IFN-γ and IL-17 mediate the elicitation of CHS responses by different mechanisms and whether both cytokines are required for optimal responses.
IFN-γ has been considered as an important inflammatory cytokine which plays a key role in the type 1 immune responses (7). IFN-γ has effects on the production of cytokines, chemokines and anti-microbial substances such as reactive oxidative species (ROS) and peroxidates (8–10). It is generally accepted that IFN-γ plays important roles in CHS responses and a defect in IFN-γ signals suppresses CHS responses (11, 12). However, controversial studies have been reported to show that IFN-γ plays a minor role in CHS (13, 14). It is to note that the most of studies in the gene knockout mice included effects of IFN-γ signals on the development of effector cells as well as on the function of effector cells in the elicitation of inflammatory reactions. Therefore, it remains to be dissected whether IFN-γ mediated regulation of CHS responses is based on its effects on the development of effector cells and/or its effects on cutaneous inflammations during the elicitation of CHS responses.
IL-17 plays important roles in CHS responses and many other inflammatory diseases (4, 15, 16). A line of studies indicate that a primary effect for IL-17 is to regulate the infiltration of neutrophils in inflammatory tissues (5, 15–18). IL-17 can regulate the production of a panel of cytokines and chemokines, which may be a mechanism for IL-17 mediated regulation of leukocyte migration (19–23). In CHS responses, neutralization of IL-17 by antibodies reduces the infiltration of neutrophils and macrophages and inhibits the production of inflammatory cytokines in hapten challenge skin tissues (4). It remains to be determined whether IL-17 regulates ROS activities in the hapten challenged skin and collaborates with IFN-γ in the elicitation of CHS responses.
In the current studies, the role of IFN-γ and IL-17 signals in the elicitation of CHS responses was examined in IL-17R or IFN-γR deficient mice. We intended to examine the effect of IFN-γ and IL-17 signals on the development of CD8+ effector cells during the sensitization of hapten primed T cells and on the function of CD8+ T cells in the elicitation of CHS responses. Furthermore, mechanisms for IL-17 and IFN-γ mediated effects on inflammatory reactions in the skin were determined. The outcome of studies has demonstrated specific roles of these two cytokines in CD8+ T cell mediated CHS responses and provided new insights into mechanisms for CD8+ Tc1 and TIL-17 mediated immune responses.
MATERIALS AND METHODS
Animals and Reagents
IL-17R−/− mice on C57BL/6 background were provided by Amgen. IFN-γR−/− mice on C57BL/6 background and wild type C57BL/6 mice (6–8 weeks of age) were purchased from the Jackson Laboratory (Bar Harbor, Maine). All animal procedures were performed according to NIH guidelines and the protocols were approved by the Institute Animal Care and Use Committee of the University of Alabama at Birmingham.
Monoclonal rat anti-mouse IL-17 antibody (TC11-18H10) was purchased from Southern Biotechnology (Birmingham, AL). Hybridoma cell lines XMG1.2 (anti-IFN-γ), GK1.5 (anti-CD4), Lyt-2 (anti-CD8) and were from the ATCC (Manassas, VA). Antibodies used for flow cytometry, immunohistochemical staining and ELISA reagents were purchased from Pharmingen (San Diego, CA). Recombinant mice IL-17, IFN-γ, IL-4 and GM-CSF were from R&D Systems.
Rat anti-mouse Inducible Nitric Oxide Synthase (iNOS) antibody was purchased from Calbiochem (Darmstadt, Germany). DAB peroxidase Substrate (3, 3′-Diaminobenzine TABLET SETS), dinitrofluorobenzene (DNFB), dinitrobenzenesulfonic acid, sodium salt (DNBS), normal rat IgG, PMA, and ionomycin were from Sigma (St. Louis, MO).
Contact hypersentivity responses
Mice were sensitized with 0.5% DNFB on two consecutive days and challenged with 0.2% DNFB five days later. CHS was measured every 24 hours after challenge. Mice that were not sensitized but challenged with 0.2% DNFB served as negative controls (4). In some experiments, CD4+ or CD8+ T cells were depleted in vivo by treatment of animals with specific antibodies. Briefly, mice were treated by i.p. injection of 100μg/mouse of GK1.5, Lyt-2 or rat IgG on three consecutive days and were rested two days before sensitization (2).
To examine adoptive transfer of CHS responses by hapten primed T cells, C57Bl/6 mice were sensitized with DNFB and the draining lymph node cells were harvested 5 days later (2). CD8+ T cells were purified using MACS system according to the manufacturers’ instruction (Miltenyi Biotec Inc., Auburn, CA) and transferred intravenously into naïve wild type, IL-17R−/−, or IFN-γR−/− mice (5×106cells/mouse). The recipient mice were challenged with DNFB immediately after T cell transfer and CHS was measured 24 hours after challenge. In some experiments, the recipient mice were treated twice intraperitoneally with anti-IL-17 (200μg/mouse), anti-IFN-γ (400 μg/mouse) or rat IgG (400 μg/mouse) one day before and on the day of T cell transfer.
Induction of cutaneous inflammation by injection of recombinant cytokines
Mice were injected subcutaneously in the ear skin with 50 ng IL-17 or 50 ng IFN-γ in 20 μl PBS every other day for 5 times. Injection of PBS alone served as a negative control. The cytokine dose and injection protocol were optimized in preliminary experiments. Ear thickness was measured every other day between the injections. Ear tissues were harvested 24 hours after the last injection for analysis.
Histological and immunohistochemical staining of skin tissues
To examine the histology of skin tissues, skin samples were collected at different time points following hapten challenge. Skin tissues were fixed in 10% formalin and paraffin embedded tissue sections were made. Tissue sections were stained with H&E.
To detect leukocyte infiltration in the skin, cryo-sections (5 μm) were cut and fixed with cold acetone for 10 min. After 10 min rehydration in PBS, sections were incubated with 5μg/ml anti-CD16/CD32 antibody (2.4G/2) to block non-specific binding and then stained 30 min at room temperature with 100μl, 5μg/ml biotin labeled Gr-1, CD11b antibodies. After three washes with PBS, the binding of biotin labeled Gr-1, CD11b antibodies were detected with streptavodin-Alexa488 (1:1000, 30min at room temperature). The expression of iNOS was detected by a rat anti-mouse iNOS followed by the second antibodies PE-labeled goat anti-rat IgG. Sections were counterstained with fluorescence dye DAPI and mounted. Pictures were taken microscopically with a 20x objective. Positive cells were counted in 10 fields of each group (3 mice). The average of positive cells per field was calculated and the difference between groups was analyzed statistically by Student’s t-test. The iNOS expression level, the fluorescent density per square mm, was evaluated by using Image-pro Plus 6.0 analysis software (Media Cybernetics Inc., Bethesda, MD). Average levels of iNOS expression in ten fields were calculated and the difference between groups was analyzed statistically.
To examine myeloperoxidase positive cells in the skin, unfixed cryo-sections (5 μm) were incubated with 100μl of DAB peroxidase substrate. The reaction was stopped by washing in PBS. Pictures were taken microscopically with a 20x objective. Positive cells were counted in 10 fields of each group (3 mice). Average numbers of positive cells per field were calculated and the difference between groups was analyzed statistically.
Measurement of cytokine production by hapten primed T cells
Bone Marrow Dendritic Cells (BM-DC) were generated from bone marrow of C57BL/6 as reported in our previous studies (4). BM-DC were labeled with DNBS (5mM, 1ml/107cells, 37°C, 15min) and cultured with primed T cells from DNFB sensitized wild type, IL-17R−/− or IFN-γR−/− mice (2×106/ml T cells and 2×105/ml DCs in 200μl/well). Supernatants were harvested 48 hours after cultures for measurement of cytokines by ELISA as described (4).
Since a very small portion (<1%) of IL-17 or double cytokine producing T cells can be detected in flow cytometric analysis when CD8+ T cells are stimulated with hapten labeled DC (4), stimulation with anti-CD3 antibody provides strong signals and enables us to detect a change in cytokine producing cells. Our previous studies demonstrate that the stimulation with plate bound anti-CD3 antibody induces a higher level of cytokine production by hapten primed T cells but does not change the pattern of cytokine production (2). To detect intracellular cytokines, primed T cells were stimulated with plate-bound anti-CD3 (25μg/ml, 30μl/well). Cells were harvested 48 hours after cultures and re-stimulated with PMA (50ng/ml), ionomycin (500ng/ml) in the present of Golgi stop (1μl/ml, BD Pharmingen) for 4–6 hrs. Cells were then collected and incubated with 5μg/ml anti-CD16/CD32 antibody (2.4G2) to block non-specific binding. The cells were first stained with Alex488-conjugated anti-CD4 or CD8 antibodies. The cells were then fixed and permeabilized with Cytofix/Cytoperm buffer (BD Pharmingen) and incubated with PE-conjugated anti-IL-17, APC-conjugated anti-IFN-γ (BD Pharmingen) or isotype mouse IgG controls. Samples were analyzed in a FACSCalibur flow cytometer (BD Biosciences) (4).
Real-time RT-PCR
Total RNA was isolated from homogenized skin tissues by using TRIzol Reagent (GibcoBRL) according to the instructions provided. Two micrograms of RNA were used for synthesis of cDNA with a cDNA synthesis kit from Bio-Rad. Real time PCR was performed with iQ SYBRO Green Supermix Kit in a MyiQ real time qPCR system (Bio-Rad) according to the manufacture’s instructions. The expression level of cytokines was normalized to the housekeeping gene GAPDH in each sample.
The sequences for primers were as follows: IFN-γ (221bp), forward, 5′-ACA ATC AGG CCA TCA GCA AC-3′, reverse, 5′-TCA GCA GCG ACT CCT TTT CC-3′. IL-17 (249bp), forward: 5′-TCT CAT CCA GCA AGA GAT CC -3′, reverse, 5′-GAA TCT GCC TCT GAA TCC AC -3′. CD8 (189bp), forward, 5′-AAC TCC AGC TCC AAA CTC C-3′, reverse, 5′-GAC TGA GCA GAA ATA GTA GCC-3′. IP10 (208bp), forward, 5′-CGT CAT TTT CTG CCT CAT CC-3′, reverse, 5′-CAG ACA TCT CTG CTC ATC ATT C-3′. IL-6 (202bp), forward, 5′-CCT CTC TGC AAG AGA CTT CC-3′, reverses, 5′-GCA CAA CTC TTT TCT CAT TTC C-3′. IL-1β(206bp), forward, 5′-ACA GCA GCA CAT CAA CAA GAG-3′, reverse, 5′-ATG GGA ACG TCA CAC ACC AG-3′. KC (164bp), forward, 5′-GAT TCA CCT CAA GAA CAT CCA G-3′, reverse, 5′-TGG GGA CAC CTT TTA GCA TC-3′. MCP1 (299bp), forward, 5′-AAC TCC CAT CCC AAT CAC C-3′, reverse, 5′-CCT CCA TCA ACC ACT TTT CC-3′. MIG (297bp), forward, 5′-CCG CTG TTC TTT TCC TTT TG-3′, reverse, 5′-TCC CCC TCT TTT GCT TTT TC-3′. MIP-1β(189bp): forward, 5′-TCC CAC TTC CTG CTG TTT C-3′, reverse, 5′-CAG TTC AAC TCC AAG TCA CTC-3′.
Statistical analysis
In CHS responses, each group will contained three mice and the increase of ear swelling was calculated from 6 ears and presented as mean±SEM. The differences between experimental groups were analyzed using the Student’s t-test with p<0.05 being considered statistically significant.
RESULTS
The level of CD8, IL-17 and IFN-γ signals is associated with the kinetics of CHS responses
CD8+ T cells are primary effector cells in CHS responses (24–26). It was reported that the early recruitment of CD8+ T cells in the hapten challenged skin was required for the elicitation of CHS responses (27). Although hapten primed CD8+ T cells are able to produce IFN-γ and IL-17 (4), it is yet to be examined whether the recruitment of CD8+ T cells in the hapten challenged skin is associated with the expression of IFN-γ and IL-17 signals and the kinetic of CHS responses. To examine it, C57Bl/6 wild type mice were sensitized and challenged with dinitrofluorobenzene (DNFB) as described (4). Ear skin tissues were harvested at various times following challenge and subjected to analysis. Since few CD8+ T cells could be detected in the hapten challenged skin by immunohistochemical staining with antibodies (data not shown), the CD8, IL-17 and IFN-γ mRNA levels were measured by real time RT-PCR. Results indicated that mRNA signals for CD8, IFN-γ and IL-17 were increased in the skin following hapten challenge and peaked at 48 hours. The increase of CD8 was closely correlated with the levels of IFN-γ and IL-17 signals (Fig 1a). Furthermore, the level of CD8, IL-17 and IFN-γ signals was correlated with the kinetic of CHS responses, which peaked at 48 hours following hapten challenge and then declined (Fig. 1b). The role of CD8+ T cells in CHS responses was confirmed by that the depletion of CD8+ T cells significantly reduced CHS responses whereas depletion of CD4+ T cells increased the response (Fig. 1b). These results implicate that the expression level of IFN-γ and IL-17 is closely correlated with the migration of effector CD8+ T cells in hapten challenged skin tissues and is associated with the kinetics of CHS responses. It is to note that IL-17 and IFN-γ may be expressed not only by CD8+ T cells but also by other cells in the skin during CHS responses.
Figure 1.
The level of CD8, IFN-γ and IL-17 in hapten challenged skin tissues is associated with the kinetics of CHS responses. A). Wild type mice were sensitized and challenged as described. Ear skin tissues were harvested at the indicated times following challenge and the mRNA levels of CD8, IFN-γ and IL-17 were determined by real time RT-PCR. B). Mice were depleted of CD4+ (CD4del) or CD8+ (CD8del) T cells prior to hapten sensitization. CHS responses were measured following challenge. The treatment with rat IgG served as a control. Mice that were not sensitized were challenged and served as naive control (Naïve). The values are shown as means ± SEM of 3 mice per group. The difference between the control and T cell depleted groups is statistically significant at all indicated times following challenge (P<0.05).
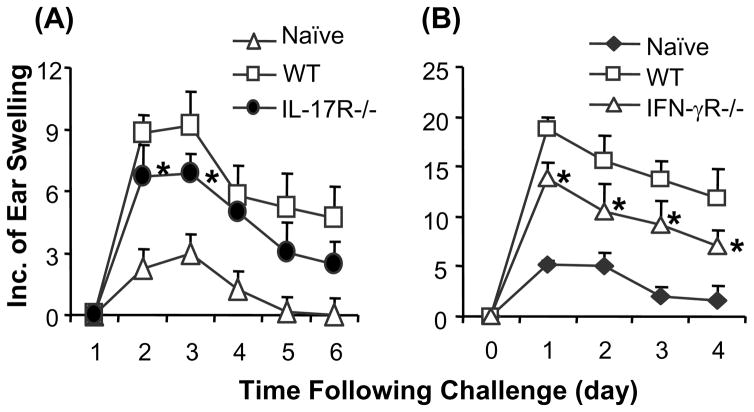
CHS responses are inhibited in IL-17R−/− or IFN-γR−/− mice
It was reported that CHS responses were reduced in IL-17 deficient mice (5). It is unknown whether a defect in IL-17R affects CHS responses although inflammatory diseases are suppressed in the IL-17R−/− mice (28, 29). Controversial results were reported about CHS responses in IFN-γR−/− mice (11–14). To determine the role of IL-17 and IFN-γ signals in CHS responses in the current studies, mice were sensitized and challenged with DNFB as described in previous studies (4). Results indicated that CHS responses were significantly reduced in IFN-γR−/− and IL-17R−/− mice compared to wild type controls (Fig. 2). This implicates that a defect in either IFN-γR or IL-17R suppresses CHS responses.
Figure 2.
CHS responses are inhibited in IFN-γR−/− or IL-17R−/− mice. Mice were sensitized with DNFB and challenged as described. CHS responses were measured. Mice that were not sensitized were challenged and served as naive control (Naïve). Compared to wild type mice, CHS responses are significantly inhibited in IL-17R−/− (A) or IFN-γR−/− (B) mice. The values are shown as means ± SEM of 3 mice per group (* P<0.05). The data are representative of 2–3 independent experiments.
The reduction of CHS responses in IFN-γR−/− and IL-17R−/− mice could result from a defect in the induction of hapten specific effector cells during sensitization or suppression of inflammatory reactions at the elicitation phase. It is known that IFN-γ is an important cytokine for the development of Th1 and Tc1 cells (30, 31). However, less is known about the role of IL-17 signals in the development of hapten specific effector cells. To examine it, mice were sensitized with DNFB and the draining lymph node cells were harvested 5 days later. The hapten primed T cells were stimulated in vitro with DNBS labeled bone marrow derived dendritic cells (BM-DC) as described previously (4). In our hands, the number of hapten primed CD4+ and CD8+ T cells which expressed a high level of activation marker CD44 was not significantly altered by the gene deficiency (Supplement figure). Our previous studies demonstrate that CD8+ T cells which expressed a high level of CD44 are effector cells for the elicitation of CHS responses (3). Studies from other groups also show that T cell growth and activation are not affected in IFN-γR−/− or IL-17R−/− mice (20, 32–34). However, hapten primed T cells from IFN-γR−/− mice produced less IFN-γ but more IL-17 than those from wild type controls (Fig. 3a). Interestingly, hapten primed T cells from IL-17R−/− mice exhibited a similar phenotype to those from IFN-γR−/− mice (Fig. 3a). Further analysis of intracellular cytokines indicated that a defect in IFN-γR or IL-17R suppressed the development of CD4+ Th1 and CD8+ Tc1 cells whereas it increased CD4+ Th17 and CD8+ TIL-17 cell differentiation (Fig. 3b). The results suggest that IFN-γ and IL-17 mediated signals may have an effect on T cell differentiation rather than activation in hapten sensitized mice and the effect may be a mechanism for their roles in CHS responses.
Figure 3.
The cytokine phenotype of primed T cells from hapten sensitized IFN-γR−/− and IL-17R−/− mice. Mice were sensitized with DNFB and the draining lymph node cells were harvested 5 days later. A). The cells were stimulated with DNBS labeled BM-DC for 2 days. Cytokine concentrations in culture supernatants were measured by ELISA. The data (mean ± SEM) are representative of 2 independent experiments. *P<0.05). B). The cells were stimulated with plate-bound anti-CD3 antibodies for 2 days. IL-17 and IFN-γ positive CD4+ and CD8+ T cells were analyzed by flow cytometry. The numbers indicate the percentage of positive cells in the gated CD4+ or CD8+ T cell population. The data are representative of 3 independent experiments.
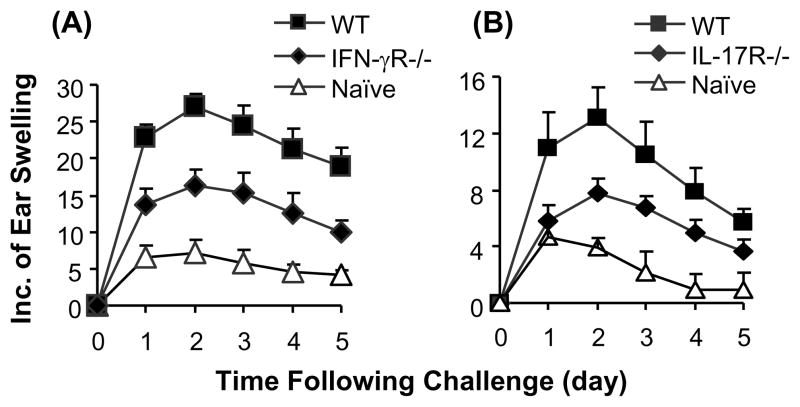
Adoptive transfer of CHS responses by hapten primed CD8+ T cells is suppressed in IFN-γR−/− or IL-17R−/− recipient mice
Since the defect in IFN-γR or IL-17R affected the development of hapten primed T cells, the results described above could not dissect the role of IFN-γ and IL-17 in the elicitation of CHS responses. To examine the role of these cytokines in the elicitation phase of CHS responses, hapten primed wild type CD8+ T cells were purified and transferred to naïve IFN-γR−/−, IL-17R−/− or wild type recipient mice. The recipient mice were then challenged and CHS responses were measured. In this adoptive transfer model, CD8+ effector cells were from hapten primed wild type mice and were able to produce and respond normally to IFN-γ and IL-17. Therefore, a change in the magnitude of CHS responses will indicate a role of IFN-γ or IL-17 in CD8+ T cell mediated elicitation of inflammatory reactions. Results showed that the transfer of CHS responses by primed wild type CD8+ T cells was significantly suppressed in IL-17R−/− or IFN-γR−/− mice compared to wild type recipients (Fig. 4). This result implicates that both IL-17 and IFN-γ signals are required for the elicitation of CHS responses.
Figure 4.
Adoptive transfer of CHS responses is inhibited in IFN-γR−/− or IL-17R−/− mice. CD8+ T cells from DNFB sensitized wild type (WT) mice were purified and transferred intravenously into naïve WT, IFN-γR−/− or IL-17R−/− recipient mice. The mice were challenged and CHS responses were measured. Mice that were neither transferred with cells nor sensitized were challenged and served as naive control (Naïve). The adoptive transfer of CHS responses is significantly reduced in IFN-γR−/− (A) or IL-17R−/− (B) recipients compared to WT controls at all indicated time points (P<0.05). The values are shown as means ± SEM of 3 mice per group. The data are representative of 3 independent experiments.
Different patterns of inflammatory reactions in the hapten challenged skin of IFN-γR−/− and IL-17R−/− mice
The infiltration of neutrophils and macrophages in the skin is a characteristic of allergic contact dermatitis in patients as well as CHS responses in animal models (24, 25, 35). It was unknown whether a defect in IFN-γ or IL-17 signals affected the patterns of inflammatory reactions in CHS responses. To examine it, skin tissues from mice which were adoptively transferred with hapten primed wild type CD8+ T cells were collected 24 hours after hapten challenge. Infiltration of leukocytes and activities of inflammatory enzymes in the skin tissues were examined. Results showed that a defect in IL-17R significantly reduced the infiltration of Gr-1+ granulocytes and CD11b+ macrophages (Fig. 5). This is in accordance with our previous studies indicating that leukocyte infiltration in the skin of CHS responses is reduced by anti-IL-17 neutralizing antibodies (4). Interestingly, the infiltration of granulocytes and macrophages was less affected in IFN-γR−/− mice compared to wild type controls. It suggests that IL-17 may have primary effects whereas IFN-γ may have minor effects on the regulation of leukocyte infiltration during CHS responses.
Figure 5.
Different patterns of inflammatory reactions in the hapten challenged skin tissues of IFN-γR−/− or IL-17R−/− mice. CD8+ T cells from DNFB sensitized wild type (WT) mice were purified and transferred intravenously into naïve WT, IFN-γR−/− or IL-17R−/− recipient mice. The mice were then challenged and ear skin tissues were harvested 24 hours after challenge. Cryo-sections (5 μM) of ear tissues were stained with Gr-1 (granulocytes), CD11b (monocytes/macrophages), iNOS antibodies or DBA, a substrate for myeloperoxidase (MPO+) as described in the Materials and Methods. The number of positive cells was counted microscopically. The data represent mean ± SEM of 10 fields. The intensity of iNOS staining was evaluated with the software ImagePro Plus. * P<0.05. The data are representative of 3 independent experiments.
Reactive oxidative species (ROS) are produced during inflammations and important regulators for inflammatory reactions (36). Inducible nitric oxide synthase (iNOS) and myeloperoxidase (MPO), which are primarily produced by macrophages and granulocytes (36, 37), have been reported to play roles in cutaneous inflammations and CHS responses (38–40). IFN-γ is a stimulator for both iNOS and MPO (37, 41) whereas it is unknown whether IL-17 regulates ROS activities in inflammatory tissues. In the hapten challenged skin, iNOS and MPO activities were significantly inhibited in IFN-γR−/− mice compared to wild type mice (Fig. 5). The activity of iNOS was less affected while MPO was reduced in IL-17R−/− mice. The results indicate that IFN-γ signals are important for the regulation of ROS activities in CHS responses.
Different patterns of cytokines and chemokines in the hapten challenged skin of IFN-γR−/− or IL-17R−/− mice
The elicitation of CHS responses requires the production of cytokines and chemokines in the hapten challenged skin, which regulate the infiltration and activity of leukocytes in inflammatory reactions (42, 43). Both IFN-γ and IL-17 have been reported to regulate the expression of certain cytokines and chemokines in infectious and autoimmune diseases (21, 22, 44). In CHS responses, the induction of chemokines is considered to be a mechanism for IFN-γ mediated effects on inflammatory reactions (45–47) whereas less is known about IL-17 mediated regulation of cytokines and chemokines, especially at the elicitation phase of CHS responses. Moreover, it is unclear whether IFN-γ and IL-17 induced different patterns of cytokines and chemokines, which may provide clues to specific mechanisms for IFN-γ and IL-17 mediated inflammatory reactions. We examined whether a defect in IFN-γR or IL-17R affected the pattern of cytokines and chemokines in the skin of CHS responses that were transferred by primed wild type CD8+ T cells. In the experiments, skin tissues were harvested at 24 hours following hapten challenge and were subjected to analysis of cytokines and chemokines by real time RT-PCR.
Results showed an interesting pattern of cytokine and chemokine expression in IFN-γR−/− and IL-17R−/− mice compared to wild type controls (Fig. 6). The expression of macrophage chemotaxis protein-1 (MCP-1), monocyte inhibitory protein-1 (MIP-1α), and interferon gamma induced protein 10 (IP-10) in the skin of CHS responses was decreased in both IFN-γR−/− and IL-17R−/− mice compared to wild type controls. Similarly, CD8 signal was reduced in both gene knockout mice, suggesting that the infiltration of CD8+ T cells in hapten challenged skin might be inhibited. However, monokine induced by interferon gamma (Mig) was undetectable in IFN-γR−/− mice whereas it was hardly affected in IL-17R−/− mice. In contrast, the levels of IL-6, IL-1β and KC (a mouse homologue to human IL-8) were significantly reduced in IL-17R−/− mice whereas they were less affected in IFN-γR−/− mice. These cytokines and chemokines have been reported to play important roles in CHS responses (47–50). The differential regulation of the cytokines and chemokines suggests that IL-17 and IFN-γ may mediate CHS responses by different mechanisms.
Figure 6.
The expression of cytokines and chemokines is differentially regulated in the hapten challenged skin tissues of IFN-γR−/− or IL-17R−/− mice. CD8+ T cells from DNFB sensitized wild type (WT) mice were purified and transferred intravenously into naïve WT, IFN-γR−/− or IL-17R−/− recipient mice. The mice were then challenged and ear skin tissues were harvested 24 hours after challenge. RNA was extracted from tissue samples and the level of mRNA for cytokines and chemokines was determined by real time RT-PCR. Each sample was triplicated and values were normalized to the level of house-keeping gene GAPDH (mean ± SD). The data are representative of 2 independent experiments.
Injection of recombinant IFN-γ or IL-17 induces cutaneous inflammatory reactions
Although the data described above indicated that a defect in IFN-γR or IL-17R suppressed CD8+ T cell mediated elicitation of CHS responses, one may argue that the defect in IFN-γR or IL-17R could affect the behavior of CD8+ effector cells during CHS responses and a direct effect of IL-17 and IFN-γ on cutaneous inflammations needs to be verified. In order to confirm the effect of IFN-γ and IL-17 on cutaneous inflammations, recombinant IFN-γ or IL-17 were injected subcutaneously in the ear skin of wild type mice. Results indicated that the injection of IL-17 or IFN-γ induced ear swellings whereas controls with injection of PBS only showed a background level of responses (Fig. 7a). The injection of IL-17 or IFN-γ in IL-17R−/− or IFN-γR−/− mice, respectively, did not induce significant ear swelling (data not shown). Histological analysis showed that injection of IL-17 or IFN-γ induced swelling and leukocyte infiltration in the injected skin, a similar phenomenon observed in the skin of CHS responses (Fig. 7b). Further analysis showed that although both IL-17 and IFN-γ induced significant levels of leukocyte infiltration compared to PBS controls, injection of IL-17 induced a high level of Gr-1+ and CD11b+ cells whereas IFN-γ induced a high level of MPO positive cells in the skin (Fig. 7c).
Figure 7.
Injection of recombinant IFN-γ and IL-17 induces inflammatory reactions in the skin. Recombinant IFN-γ, IL-17 (50 ng/mouse) or PBS was injected subcutaneously in ear skin every other day. A). Ear swelling was measured every other day between the injection days. Injection of IFN-γ or IL-17 induces significant levels of ear swelling compared to PBS treated controls. *P<0.05, **P<0.01. B). Ear skin tissues were collected at 24 hours after the last injection (day 10) and subjected to histology analysis. C). Frozen sections of skin tissues (same as figure B) were stained and the number of positive cells per field was counted microscopically. MPO: myeloperoxidase positive cells. The data show means ± SEM of 10 fields and are representative of two independent experiments. **P<0.01.
IFN-γ and IL-17 signals are coordinated in the elicitation of CHS responses
Having shown that IL-17 and IFN-γ are required for the elicitation of CHS responses and they regulate cutaneous inflammatory reactions by different mechanisms, the next question was whether IL-17 and IFN-γ signals are coordinated in the response. To test it, primed CD8+ T cells from hapten sensitized wild type mice were transferred into naïve IFN-γR−/− mice that were pretreated with an anti-IL-17 antibody or IL-17R−/− mice that were pretreated with an anti-IFN-γ antibody. The treatment with normal rat IgG served as a control. CHS responses in the recipient mice were measured following hapten challenge. Results indicated that CHS responses were further suppressed in the gene knockout mice that were pretreated with the anti-cytokine antibodies compared to those that were pretreated with control rat IgG (Fig. 8). The results implicate that IL-17 and IFN-γ are coordinated in the elicitation of CHS responses and both signals are required for an optimal response elicited by hapten primed CD8+ T cells.
Figure 8.
Both IFN-γ and IL-17 signals are required for optimal CHS responses. CD8+ T cells from DNFB sensitized wild type (WT) mice were purified and transferred intravenously into naïve WT, IFN-γR−/− or IL-17R−/− recipient mice that were pre-treated with antibodies. CHS responses were measured following challenge. Mice that were neither transferred with T cells nor sensitized were challenged and served as naive control (Naïve). (A). IL-17R−/− recipient mice were pre-treated with an anti-IFN-γ antibody. (B). IFN-γR−/− recipient mice were pre-treated with an anti-IL-17 antibody. The treatment with rat IgG served as a control. The treatment of mice with anti-IL-17 or anti-IFN-γ antibodies significantly reduces CHS responses compared to the control treatment with rat IgG (P<0.05 at the indicated times). The values are shown as means ± SEM of 3 mice per group. The data are representative of 2 independent experiments.
DISCUSSION
CHS responses represent a delayed type cellular immune response, which is mediated by CD8+ T cells that produce IFN-γ (Tc1) or IL-17 (TIL-17) (4, 12, 51). In the current studies, effector mechanisms for IL-17 and IFN-γ signals in the elicitation of CHS responses are dissected by adoptive transfer of CHS responses with primed wild type CD8+ T cells in IL-17R−/− or IFN-γR−/− mice. The data demonstrate that IL-17 and IFN-γ mediate the elicitation of CHS responses by different mechanisms and that both cytokines are required for optimal CHS responses.
CHS is a unique model of delayed type hypersensitivity responses, which is primarily mediated by CD8+ T cells (1–3). The data from the current studies show that the infiltration of CD8+ T cells is correlated with the level of IL-17 and IFN-γ signals in the hapten challenged skin tissues during CHS responses and that these are associated with the kinetics of CHS responses. Depletion of CD8+ T cells significantly diminishes CHS responses whereas adoptive transfer of hapten primed CD8+ T cells can elicit CHS responses in naïve recipient mice. Moreover, the adoptive transfer of CHS responses by wild type primed CD8+ T cells is suppressed in IFN-γR−/− or IL-17R−/− recipient mice, implicating that both IFN-γ and IL-17 signals are required for the elicitation of CHS responses and that both CD8+ Tc1 and TIL-17 cells are effector cells. This is further supported by that blockade of both cytokine signals will lead to significantly deeper suppression of CHS responses than a defect in single cytokine.
The role of IFN-γ in CHS responses is controversial in previous studies (11–14). An open question was whether IFN-γ is an effector cytokine required for the elicitation of CHS responses or regulates primarily the development of effector cells. Our studies demonstrate that IFN-γ is an indispensable effector cytokine in the elicitation of CHS responses. 1. CHS responses are reduced in IFN-γR−/− mice even though both CD4+ and CD8+ IL-17 producing T cell populations are increased; 2. The adoptive transfer of CHS responses with wild type primed CD8+ T cells is suppressed in IFN-γR−/− recipient; and 3. Injection of IFN-γ induces cutaneous inflammations.
A line of recent studies have demonstrated that a defect in Th1 or IFN-γ enhances the development of Th17 and exaggerates inflammatory reactions in some autoimmune diseases (52). In IL-17 defect mice, T cell growth is not affected whereas the production of IFN-γ is inhibited (5). Our data show that a defect in either IFN-γ or IL-17 signals inhibits IFN-γ procuring T cells but increases the development of hapten specific CD4+ and CD8+ T cells which produce IL-17 (Fig. 3). One explanation is that the decrease in IFN-γ producing T cells may at least in part contribute to the increase of IL-17 producing T cells. Further experiments are required for investigating mechanisms of IL-17 mediated effects on the differentiation of T cells and physiological relevance to CHS responses. Nonetheless, the effect of IFN-γ and IL-17 on the development of hapten primed T cells complicates the study in the effect of these cytokines on the elicitation of CHS responses. Adoptive transfer of CHS with hapten primed wild type effector cells can dissect mechanisms for IFN-γ and IL-17 mediated elicitation of CHS responses. The reduction of CHS responses in IL-17R−/− or IFN-γR−/− mice which are sensitized and challenged (Fig. 2) is not as dramatic as in the gene knockout mice which are transferred by primed wild type CD8+ T cells (Fig. 4). It suggests that the effect of the gene deficiency on the sensitization of T cells may compensate for the magnitude of CHS responses at the elicitation phase. It is to note that the current studies are focused on the role of IL-17 and IFN-γ signals in CD8+ T cell mediated elicitation of CHS responses. It does not exclude that CD4+ T cells which can produce both IL-17 and IFN-γ may be involved. Since CD4+ T cells are minor effector cells for the elicitation of CHS responses in animal models (1–3), different approaches have to be taken to address it in future studies. Moreover, CTL activity has been reported to be a mechanism for CD8+ T cell mediated CHS responses (53). Although both IFN-γ and IL-17 have been reported to regulate CTL activity (54, 55), it remains to be determined whether regulation of CTL activity is a mechanism for IL-17 and IFN-γ mediated elicitation of CHS responses.
Mechanisms for IFN-γ and IL-17 in the elicitation of CHS responses are incompletely understood. The current studies demonstrate that IFN-γ and IL-17 regulate the elicitation of CHS responses by different mechanisms. IL-17 signals are primarily required for the infiltration of leukocytes whereas IFN-γ signals are more important for the regulation of ROS activities in inflammatory tissues. The infiltration of CD11b+ macrophages and Gr-1+ granulocytes is significantly reduced in the skin of IL-17R−/− but are less affected in IFN-γR−/− mice (Fig. 5). This result is further supported by injection of IL-17 cytokine in the skin, which induces a higher level of leukocyte infiltration than injection of IFN-γ (Fig. 7c). It is to note that injection of IFN-γ does lead to leukocyte infiltration in the skin whereas the infiltration of leukocytes in the skin of IFN-γR−/− mice is little affected. It is possible that in the IFN-γR−/− mice, the lack of IFN-γ mediated signals may affect the production of other cytokines, such as up-regulation of IL-17. This may compensate for the regulation of leukocyte infiltration.
Inducible nitric oxide synthase (iNOS) and myeloperoxidase (MPO), which are primarily produced by macrophages and granulocytes (36, 37), play roles in cutaneous inflammations and CHS responses (38–40). Our data suggest that stimulation of ROS activities may be a primary mechanism for IFN-γ signals in CHS responses. In support of this, injection of IFN-γ induces a higher number of MPO positive cells than injection of IL-17 in the skin (Fig. 7c). The reduction of MOP activity in the skin of IL-17R−/− mice (Fig. 5) likely results from the reduced infiltration of leukocytes which are main source of MPO (36, 37).
Inflammatory cytokines and chemokines, which are produced in the hapten challenged skin, play important roles in the elicitation of CHS responses (25, 42, 43). We have found that the expression pattern of a panel of cytokines and chemokines is different in the hapten challenged skin of IFN-γR−/− and IL-17R−/− mice (Fig. 6). KC, an important chemokine for the regulation of neutrophil infiltration in CHS responses (48), is greatly reduced in IL-17R−/− animals. This accompanied with a significant decrease in the infiltration of Gr-1+ and CD11b+ leukocytes in the hapten challenged skin of IL-17R−/− mice compared to wild type controls. In contrast, KC expression in IFN-γR−/− mice is hardly affected. It may explain at least in part why the infiltration of CD11b+ and Gr-1+ leukocytes in the skin of IFN-γR−/− mice is less affected. Similarly, IL-1 and IL-6, which play important roles in CHS (47, 49, 50), are preferentially reduced in IL-17R−/− compared to IFN-γR−/− and wild type control mice. IL-1 and IL-6 are primarily produced by leukocytes. Therefore, the reduction of leukocytes infiltration may be attributed to the decrease of the cytokines in the hapten challenged skin of IL-17R−/− mice. However, Mig, which is a chemokine induced by IFN-γ and plays a role in CHS (42), is unaffected in IL-17R−/− compared to wild type controls whereas it is undetectable in IFN-γR−/− mice. Chemokines MCP-1, MIP-1β and IP-10, which can regulate the infiltration of CD8+ T cells in immune responses (47, 48), are down regulated in both IL-17R−/− and IFN-γR−/− mice. It may contribute to the reduction of CD8+ T cell infiltration in the hapten challenged skin and the suppression of CHS responses in the gene knockout mice. It is a common feature that suppression of CHS is associated with a reduction of CD8+ T cell infiltration in the hapten challenged skin, which is found in mice that are deficient in CTL activity or chemotaxis of neutrophils (48, 53). A likely explanation is that suppression of inflammation will decrease the production of chemokines for recruitment of CD8+ T cells in the skin. The different pattern of cytokines and chemokines in the hapten challenged skin may be related to specific mechanisms for the elicitation of CHS responses. Certainly further studies are required to determine the role of a specific cytokine or chemokine in IFN-γ and IL-17 mediated inflammations in CHS responses. Although IL-17R and IFN-γR are widely expressed by many types of cells (56, 57), it is not excluded that certain types of cells may express either of them, and therefore, respond to a specific cytokine. We will examine the issue in future studies.
In summary, our studies have demonstrated that different mechanisms are employed for IFN-γ and IL-17 mediated elicitation of CHS responses which are induced by CD8+ T cells. Blockade of both IFN-γ and IL-17 will get the maximal suppression of the immune responses. The outcome not only advances our understanding of pathogenesis of the disease but also provides information for development of new therapeutic strategies.
Supplementary Material
Abbreviations
- CHS
Contact Hypersensitivity
- DNFB
Dinitrofluorobenzene
- DNBS
dinitrobenzenesulfonic acid, sodium salt
- PE
Phycoerythrin
- APC
Allophycocyanin
- BM-DC
Bone marrow derived dendritic cells
Footnotes
This publication was made possible by AR46256 from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and AI071041 from the National Institute of Allergy and Infectious Diseases (H.X). This investigation was conducted in a facility constructed with support from Research Facilities Improvement Program Grant No. C06 RR 15490 from the National Center for Research Resources, National Institutes of Health.
DISCLOSURES
All authors concur with the submission and have no financial conflict of interest.
References
- 1.Gocinski BL, Tigelaar RE. Roles of CD4+ and CD8+ T cells in murine contact sensitivity revealed by in vivo monoclonal antibody depletion. J Immunol. 1990;144:4121–4128. [PubMed] [Google Scholar]
- 2.Xu H, Dilulio NA, Fairchild RL. T cell populations primed by hapten sensitization in contact sensitivity are distinguished by polarized patterns of cytokine production: Interferon γ-producing (Tc1) effector CD8+ T cells and interleukin 4/IL-10-producing (Th2) negative regulatory CD4+ T cells. J Exp Med. 1996;183:1001–1012. doi: 10.1084/jem.183.3.1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xu H, Banerjee A, Dilulio NA, Fairchild RL. Development of effector CD8+ T cells in contact hypersensitivity occurs independently of CD4+ T cells. J Immunol. 1997;158:4721–4728. [PubMed] [Google Scholar]
- 4.He D, Wu L, Kim HK, Li H, Elmets CA, Xu H. CD8+ IL-17-Producing T Cells Are Important in Effector Functions for the Elicitation of Contact Hypersensitivity Responses. J Immunol. 2006;177:6852–6858. doi: 10.4049/jimmunol.177.10.6852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nakae S, Komiyama Y, Nambu A, Sudo K, Iwase M, Homma I, Sekikawa K, Asano M, Iwakura Y. Antigen-specific T cell sensitization is impaired in IL-17-deficient mice, causing suppression of allergic cellular and humoral responses. Immunity. 2002;17:375–387. doi: 10.1016/s1074-7613(02)00391-6. [DOI] [PubMed] [Google Scholar]
- 6.Albanesi C, Cavani A, Girolomoni G. IL-17 Is Produced by Nickel-Specific T Lymphocytes and Regulates ICAM-1 Expression and Chemokine Production in Human Keratinocytes: Synergistic or Antagonist Effects with IFN-{gamma} and TNF-{alpha} J Immunol. 1999;162:494–502. [PubMed] [Google Scholar]
- 7.Schroder K, Hertzog PJ, Ravasi T, Hume DA. Interferon-{gamma}: an overview of signals, mechanisms and functions. J Leukoc Biol. 2004;75:163–189. doi: 10.1189/jlb.0603252. [DOI] [PubMed] [Google Scholar]
- 8.Lacroix-Lamande S, Mancassola R, Naciri M, Laurent F. Role of Gamma Interferon in Chemokine Expression in the Ileum of Mice and in a Murine Intestinal Epithelial Cell Line after Cryptosporidium parvum Infection. Infect Immun. 2002;70:2090–2099. doi: 10.1128/IAI.70.4.2090-2099.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hausler KG, Prinz M, Nolte C, Weber JR, Schumann RR, Kettenmann H, Hanisch UK. Interferon-gamma differentially modulates the release of cytokines and chemokines in lipopolysaccharide- and pneumococcal cell wall-stimulated mouse microglia and macrophages. European Journal of Neuroscience. 2002;16:2113–2122. doi: 10.1046/j.1460-9568.2002.02287.x. [DOI] [PubMed] [Google Scholar]
- 10.Aliberti JCS, Souto JT, Marino APMP, Lannes-Vieira J, Teixeira MM, Farber J, Gazzinelli RT, Silva JS. Modulation of Chemokine Production and Inflammatory Responses in Interferon-{{gamma}}- and Tumor Necrosis Factor-R1-Deficient Mice during Trypanosoma cruzi Infection. Am J Pathol. 2001;158:1433–1440. doi: 10.1016/s0002-9440(10)64094-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wakabayashi T, Hu DL, Tagawa YI, Sekikawa K, Iwakura Y, Hanada K, Nakane A. IFN-gamma and TNF-alpha are involved in urushiol-induced contact hypersensitivity in mice. Immunology and Cell Biology. 2005;83:18–24. doi: 10.1111/j.1440-1711.2005.01310.x. [DOI] [PubMed] [Google Scholar]
- 12.Lu B, Ebensperger C, Dembic Z, Wang Y, Kvatyuk M, Lu T, Coffman RL, Pestka S, Rothman PB. Targeted disruption of the interferon-gamma receptor 2 gene results in severe immune defects in mice. PNAS. 1998;95:8233–8238. doi: 10.1073/pnas.95.14.8233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Saulnier M, Huang S, Aguet M, Ryffel B. Role of interferon-gamma in contact hypersensitivity assessed in interferon-gamma receptor-deficient mice. Toxicology. 1995;102:301–312. doi: 10.1016/0300-483x(95)03101-k. [DOI] [PubMed] [Google Scholar]
- 14.Reeve VE, Bosnic M, Nishimura N. Interferon- is Involved in Photoimmunoprotection by UVA (320–400 nm) Radiation in Mice. J Invest Dermatol. 1999;112:945–950. doi: 10.1046/j.1523-1747.1999.00594.x. [DOI] [PubMed] [Google Scholar]
- 15.Aggarwal S, Gurney AL. IL-17: prototype member of an emerging cytokine family. J Leukoc Biol. 2002;71:1–8. [PubMed] [Google Scholar]
- 16.Kolls JK, Linden A. Interleukin-17 family members and inflammation. Immunity. 2004;21:467–476. doi: 10.1016/j.immuni.2004.08.018. [DOI] [PubMed] [Google Scholar]
- 17.Yao Z, Fanslow WC, Seldin MF, Rousseau AM, Painter SL, Comeau MR, Cohen JI, Spriggs MK. Herpesvirus Saimiri encodes a new cytokine, IL-17, which binds to a novel cytokine receptor. Immunity. 1995;3:811–821. doi: 10.1016/1074-7613(95)90070-5. [DOI] [PubMed] [Google Scholar]
- 18.Langrish CL, Chen Y, Blumenschein WM, Mattson J, Basham B, Sedgwick JD, McClanahan T, Kastelein RA, Cua DJ. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J Exp Med. 2005;201:233–240. doi: 10.1084/jem.20041257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Numasaki M, Watanabe M, Suzuki T, Takahashi H, Nakamura A, McAllister F, Hishinuma T, Goto J, Lotze MT, Kolls JK, Sasaki H. IL-17 Enhances the Net Angiogenic Activity and In Vivo Growth of Human Non-Small Cell Lung Cancer in SCID Mice through Promoting CXCR-2-Dependent Angiogenesis. J Immunol. 2005;175:6177–6189. doi: 10.4049/jimmunol.175.9.6177. [DOI] [PubMed] [Google Scholar]
- 20.Kelly MN, Kolls JK, Happel K, Schwartzman JD, Schwarzenberger P, Combe C, Moretto M, Khan IA. Interleukin-17/interleukin-17 receptor-mediated signaling is important for generation of an optimal polymorphonuclear response against Toxoplasma gondii infection. Infect Immun. 2005;73:617–621. doi: 10.1128/IAI.73.1.617-621.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ye P, Rodriguez FH, Kanaly S, Stocking KL, Schurr J, Schwarzenberger P, Oliver P, Huang W, Zhang P, Zhang J, Shellito JE, Bagby GJ, Nelson S, Charrier K, Peschon JJ, Kolls JK. Requirement of interleukin 17 receptor signaling for lung CXC chemokine and granulocyte colony-stimulating factor expression, neutrophil recruitment, and host defense. J Exp Med. 2001;194:519–527. doi: 10.1084/jem.194.4.519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kolls JK, Linden A. Interleukin-17 Family Members and Inflammation. Immunity. 2004;21:467. doi: 10.1016/j.immuni.2004.08.018. [DOI] [PubMed] [Google Scholar]
- 23.Koenders MI, Kolls JK, Oppers-Walgreen B, van den Bersselaar L, Joosten LA, Schurr JR, Schwarzenberger P, van den Berg WB, Lubberts E. Interleukin-17 receptor deficiency results in impaired synovial expression of interleukin-1 and matrix metalloproteinases 3, 9, and 13 and prevents cartilage destruction during chronic reactivated streptococcal cell wall-induced arthritis. Arthritis Rheum. 2005;52:3239–3247. doi: 10.1002/art.21342. [DOI] [PubMed] [Google Scholar]
- 24.Grabbe S, Schwarz T. Immunregulatory mechanisms involved in elicitation of allergic contact hypersensitivity. Immunology Today. 1998;19:37–44. doi: 10.1016/s0167-5699(97)01186-9. [DOI] [PubMed] [Google Scholar]
- 25.Xu H, Bjarnason B, Elmets CA. Sensitization versus elicitation in allergic contact dermatitis: potential differences at cellular and molecular levels. Am J Contact Dermat. 2000;11:228–234. doi: 10.1053/ajcd.2000.8009. [DOI] [PubMed] [Google Scholar]
- 26.Vocanson M, Hennino A, Chavagnac C, Saint-Mezard P, Dubois B, Kaiserlian D, Nicolas JF. Contribution of CD4+ and CD8+ T-cells in contact hypersensitivity and allergic contact dermatitis. Expert Review of Clinical Immunology. 2005;1:75–86. doi: 10.1586/1744666X.1.1.75. [DOI] [PubMed] [Google Scholar]
- 27.Akiba H, Kehren J, Ducluzeau MT, Krasteva M, Horand F, Kaiserlian D, Kaneko F, Nicolas JF. Skin Inflammation During Contact Hypersensitivity Is Mediated by Early Recruitment of CD8+ T Cytotoxic 1 Cells Inducing Keratinocyte Apoptosis. J Immunol. 2002;168:3079–3087. doi: 10.4049/jimmunol.168.6.3079. [DOI] [PubMed] [Google Scholar]
- 28.Kelly MN, Kolls JK, Happel K, Schwartzman JD, Schwarzenberger P, Combe C, Moretto M, Khan IA. Interleukin-17/Interleukin-17 Receptor-Mediated Signaling Is Important for Generation of an Optimal Polymorphonuclear Response against Toxoplasma gondii Infection. Infect Immun. 2005;73:617–621. doi: 10.1128/IAI.73.1.617-621.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ye P, Rodriguez FH, Kanaly S, Stocking KL, Schurr J, Schwarzenberger P, Oliver P, Huang W, Zhang P, Zhang J, Shellito JE, Bagby GJ, Nelson S, Charrier K, Peschon JJ, Kolls JK. Requirement of interleukin 17 receptor signaling for lung CXC chemokine and granulocyte colony-stimulating factor expression, neutrophil recruitment, and host defense. Journal of Experimental Medicine. 2001;194:519. doi: 10.1084/jem.194.4.519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mosmann TR, Coffman RL. TH1 and TH2 Cells: Different Patterns of Lymphokine Secretion Lead to Different Functional Properties. Annual Review of Immunology. 1989;7:145–173. doi: 10.1146/annurev.iy.07.040189.001045. [DOI] [PubMed] [Google Scholar]
- 31.Constant SL, Bottomly K. Induction of Th1 and Th2 CD4+ T cell responses: The alternative approaches. The Annual Review of Immunology. 1997;15:297–322. doi: 10.1146/annurev.immunol.15.1.297. [DOI] [PubMed] [Google Scholar]
- 32.Huang S, Hendriks W, Althage A, Hemmi S, Bluethmann H, Kamijo R, Vilcek J, Zinkernagel RM, Aguet M. Immune response in mice that lack the interferon-gamma receptor. Science. 1993;259:1742–1745. doi: 10.1126/science.8456301. [DOI] [PubMed] [Google Scholar]
- 33.Hsu H-C, Yang P, Wang J, Wu Q, Myers R, Chen J, Yi J, Guentert T, Tousson A, Stanus AL, Le TvL, Lorenz RG, Xu H, Kolls JK, Carter RH, Chaplin DD, Williams RW, Mountz JD. Interleukin 17-producing T helper cells and interleukin 17 orchestrate autoreactive germinal center development in autoimmune BXD2 mice. Nat Immunol. 2008;9:166. doi: 10.1038/ni1552. [DOI] [PubMed] [Google Scholar]
- 34.Lubberts E, Schwarzenberger P, Huang W, Schurr JR, Peschon JJ, van den Berg WB, Kolls JK. Requirement of IL-17 receptor signaling in radiation-resistant cells in the joint for full progression of destructive synovitis. J Immunol. 2005;175:3360–3368. doi: 10.4049/jimmunol.175.5.3360. [DOI] [PubMed] [Google Scholar]
- 35.Gaspari AA. Mechanisms of resolution of allergic contact dermatitis. American Journal of Contact Dermatitis. 1996;7:212–219. [PubMed] [Google Scholar]
- 36.Pacher P, Beckman JS, Liaudet L. Nitric Oxide and Peroxynitrite in Health and Disease. Physiol Rev. 2007;87:315–424. doi: 10.1152/physrev.00029.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wei X-q, Charles IG, Smith A, Ure J, Feng G-j, Huang F-p, Xu D, Werner Mullers, Moncada S, Liew FY. Altered immune responses in mice lacking inducible nitric oxide synthase. Nature. 1995;375:408. doi: 10.1038/375408a0. [DOI] [PubMed] [Google Scholar]
- 38.Reiners JJ, Jr, Singh KP. Susceptibility of 129/SvEv mice in two-stage carcinogenesis protocols to 12-O-tetradecanoylphorbol-13-acetate promotion. Carcinogenesis. 1997;18:593–597. doi: 10.1093/carcin/18.3.593. [DOI] [PubMed] [Google Scholar]
- 39.Ross R, Gillitzer C, Kleinz R, Schwing J, Kleinert H, Forstermann U, Reske-Kunz AB. Involvement of NO in contact hypersensitivity. Int Immunol. 1998;10:61–69. doi: 10.1093/intimm/10.1.61. [DOI] [PubMed] [Google Scholar]
- 40.Ross R, Reske-Kunz AB. The role of NO in contact hypersensitivity. International Immunopharmacology. 2001;1:1469. doi: 10.1016/s1567-5769(01)00091-1. [DOI] [PubMed] [Google Scholar]
- 41.Adachi Y, Kindzelskii AL, Petty AR, Huang JB, Maeda N, Yotsumoto S, Aratani Y, Ohno N, Petty HR. IFN-{gamma} Primes RAW264 Macrophages and Human Monocytes for Enhanced Oxidant Production in Response to CpG DNA via Metabolic Signaling: Roles of TLR9 and Myeloperoxidase Trafficking. J Immunol. 2006;176:5033–5040. doi: 10.4049/jimmunol.176.8.5033. [DOI] [PubMed] [Google Scholar]
- 42.Goebeler M, Trautmann A, Voss A, Brocker EV, Toksoy A, Gillitzer R. Differential and sequential expression of multiple chemokines during elicitation of allergic contact hypersensitivity. Am J Pathol. 2001;158:431–440. doi: 10.1016/s0002-9440(10)63986-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sebastiani S, Albanesi C, De PO, Puddu P, Cavani A, Girolomoni G. The role of chemokines in allergic contact dermatitis. Arch Dermatol Res. 2002;293:552–559. doi: 10.1007/s00403-001-0276-9. [DOI] [PubMed] [Google Scholar]
- 44.Weaver CT, Hatton RD, Mangan PR, Harrington LE. IL-17 Family Cytokines and the Expanding Diversity of Effector T Cell Lineages. Annual Review of Immunology. 2007;25:821–852. doi: 10.1146/annurev.immunol.25.022106.141557. [DOI] [PubMed] [Google Scholar]
- 45.Bellinghausen I, Brand U, Enk AH, Knop J, Saloga J. Signals involved in the early TH1/TH2 polarization of an immune response depending on the type of antigen. J Allergy Clin Immunol. 1999;103:298–306. doi: 10.1016/s0091-6749(99)70505-1. [DOI] [PubMed] [Google Scholar]
- 46.Lack G, Bradley KL, Hamelmann E, Renz H, Loader J, Leung DY, Larsen G, Gelfand EW. Nebulized IFN-gamma inhibits the development of secondary allergic responses in mice. J Immunol. 1996;157:1432–1439. [PubMed] [Google Scholar]
- 47.Nakae S, Komiyama Y, Narumi S, Sudo K, Horai R, Tagawa Y, Sekikawa K, Matsushima K, Asano M, Iwakura Y. IL-1-induced tumor necrosis factor-alpha elicits inflammatory cell infiltration in the skin by inducing IFN-gamma-inducible protein 10 in the elicitation phase of the contact hypersensitivity response. Int Immunol. 2003;15:251–260. doi: 10.1093/intimm/dxg028. [DOI] [PubMed] [Google Scholar]
- 48.Engeman T, Gorbachev AV, Kish DD, Fairchild RL. The intensity of neutrophil infiltration controls the number of antigen-primed CD8 T cells recruited into cutaneous antigen challenge sites. J Leukoc Biol. 2004;76:941–949. doi: 10.1189/jlb.0304193. [DOI] [PubMed] [Google Scholar]
- 49.Nishimura N, Tohyama C, Satoh M, Nishimura H, Reeve VE. Defective immune response and severe skin damage following UVB irradiation in interleukin-6-deficient mice. Immunology. 1999;97:77–83. doi: 10.1046/j.1365-2567.1999.00733.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kondo S, Pastore S, Fujisawa H, Shivji GM, McKenzie RC, Dinarello CA, Sauder DN. Interleukin-1 receptor antagonist suppresses contact hypersensitivity. J Invest Dermatol. 1995;105:334–338. doi: 10.1111/1523-1747.ep12320329. [DOI] [PubMed] [Google Scholar]
- 51.Ishizaki K, Yamada A, Yoh K, Nakano T, Shimohata H, Maeda A, Fujioka Y, Morito N, Kawachi Y, Shibuya K, Otsuka F, Shibuya A, Takahashi S. Th1 and Type 1 Cytotoxic T Cells Dominate Responses in T-bet Overexpression Transgenic Mice That Develop Contact Dermatitis. J Immunol. 2007;178:605–612. doi: 10.4049/jimmunol.178.1.605. [DOI] [PubMed] [Google Scholar]
- 52.Bettelli E, Oukka M, Kuchroo VK. TH-17 cells in the circle of immunity and autoimmunity. Nat Immunol. 2007;8:345. doi: 10.1038/ni0407-345. [DOI] [PubMed] [Google Scholar]
- 53.Kehren J, Desvignes C, Krasteva M, Ducluzeau MT, Assossou O, Horand F, Hahne M, Kagi D, Kaiserlian D, Nicolas JF. Cytotoxicity is mandatory for CD8(+) T cell-mediated contact hypersensitivity. J Exp Med. 1999;189:779–786. doi: 10.1084/jem.189.5.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Dunn GP, Koebel CM, Schreiber RD. Interferons, immunity and cancer immunoediting. Nat Rev Immunol. 2006;6:836. doi: 10.1038/nri1961. [DOI] [PubMed] [Google Scholar]
- 55.Hou W, Kang HS, Kim BS. Th17 cells enhance viral persistence and inhibit T cell cytotoxicity in a model of chronic virus infection. J Exp Med. 2009;206:313–328. doi: 10.1084/jem.20082030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gaffen SL. An overview of IL-17 function and signaling. Cytokine. 2008;43:402. doi: 10.1016/j.cyto.2008.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Schroder K, Hertzog PJ, Ravasi T, Hume DA. Interferon-{gamma}: an overview of signals, mechanisms and functions. J Leukoc Biol. 2004;75:163–189. doi: 10.1189/jlb.0603252. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.