Abstract
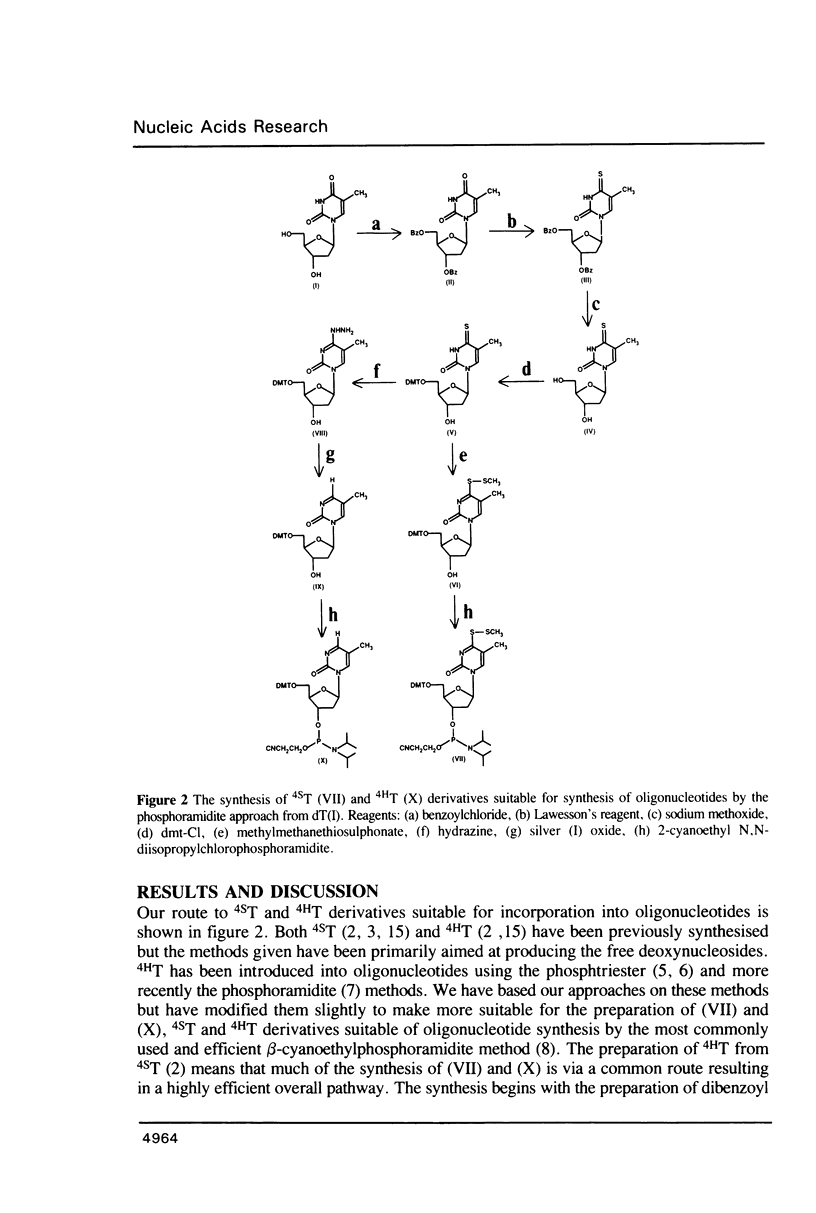
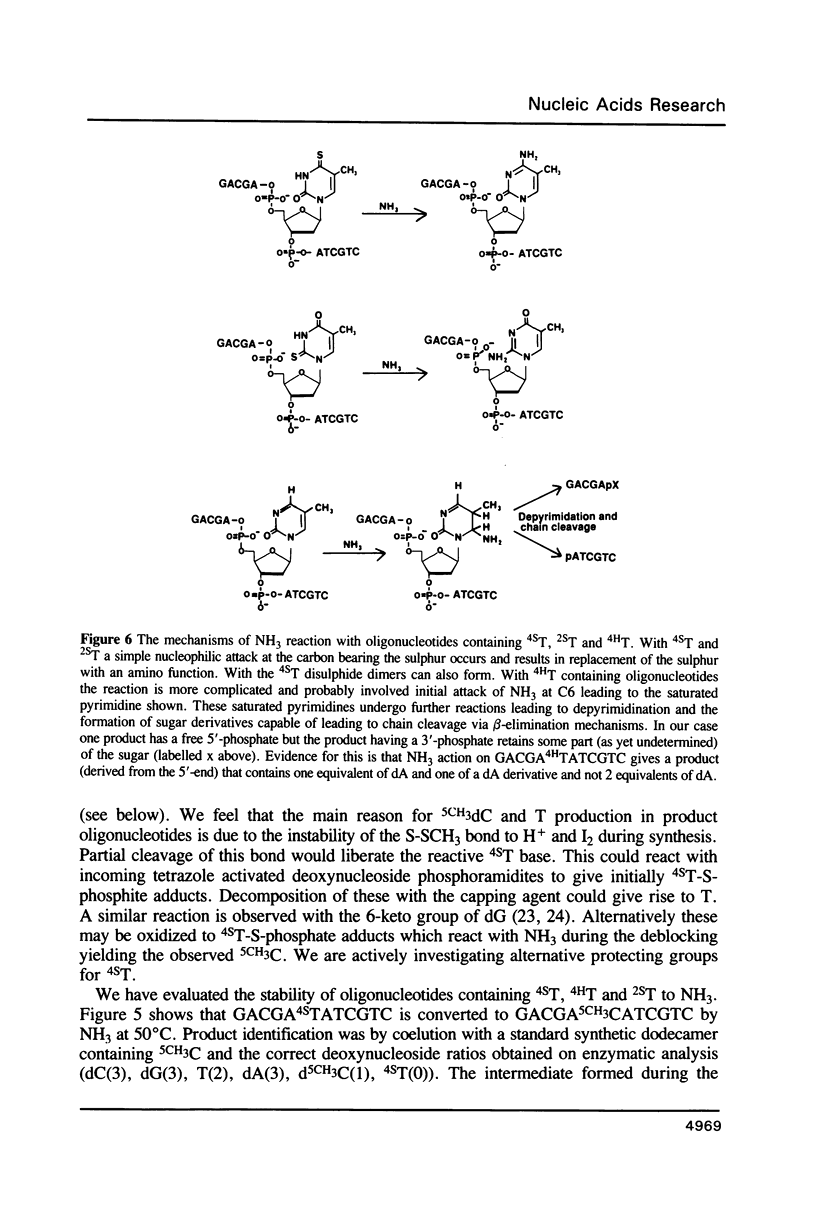
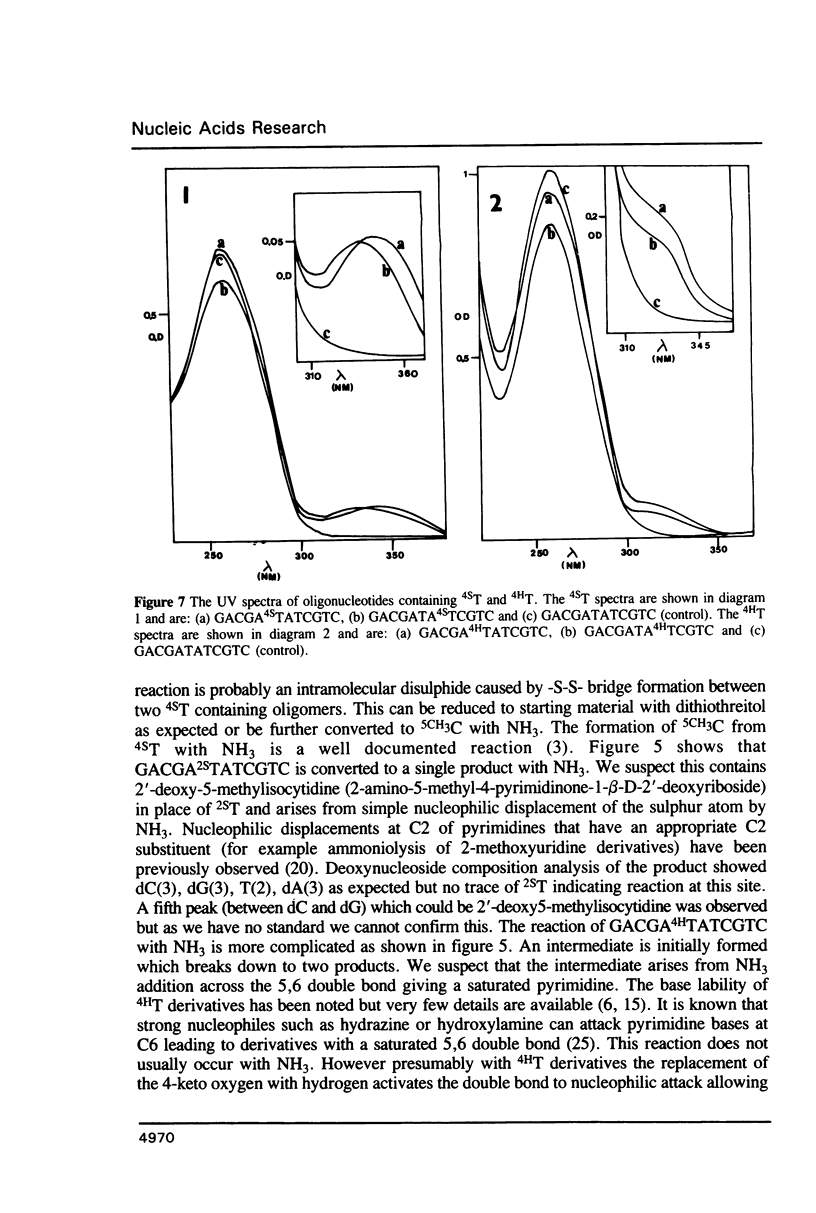
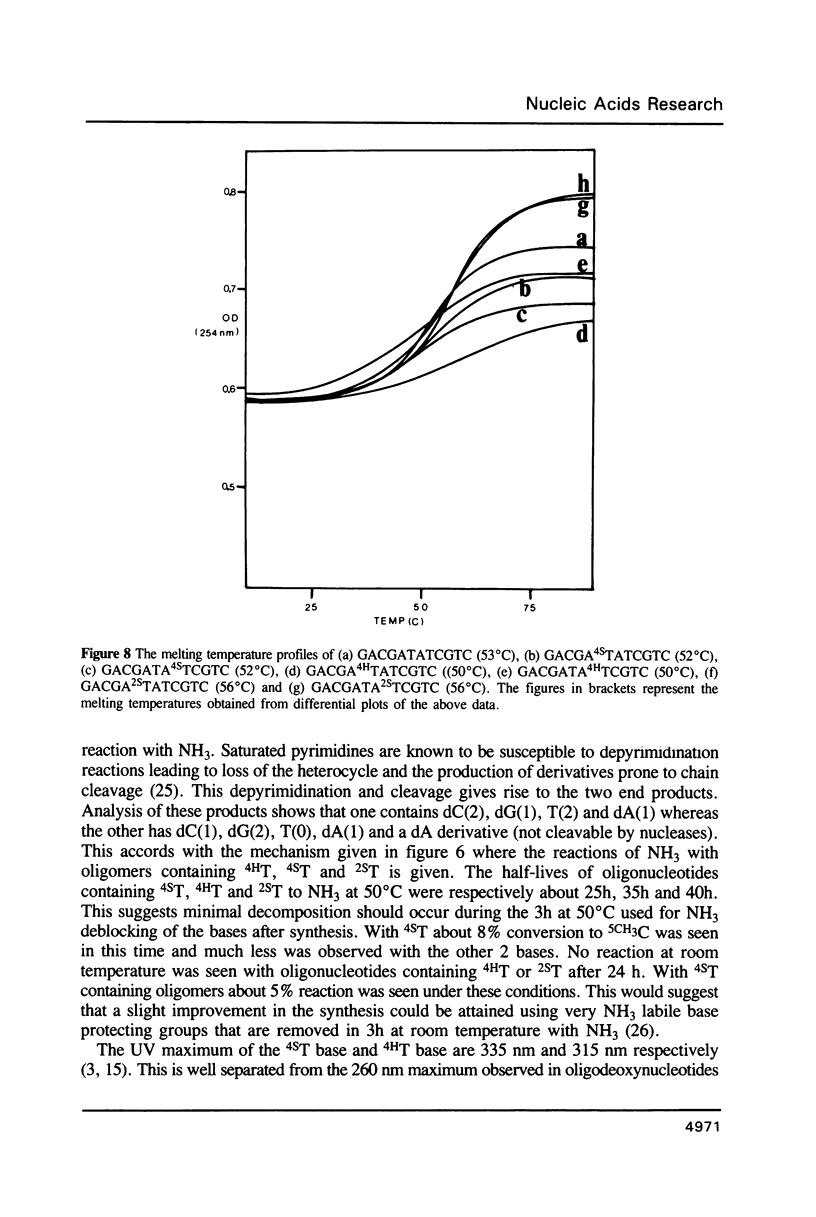
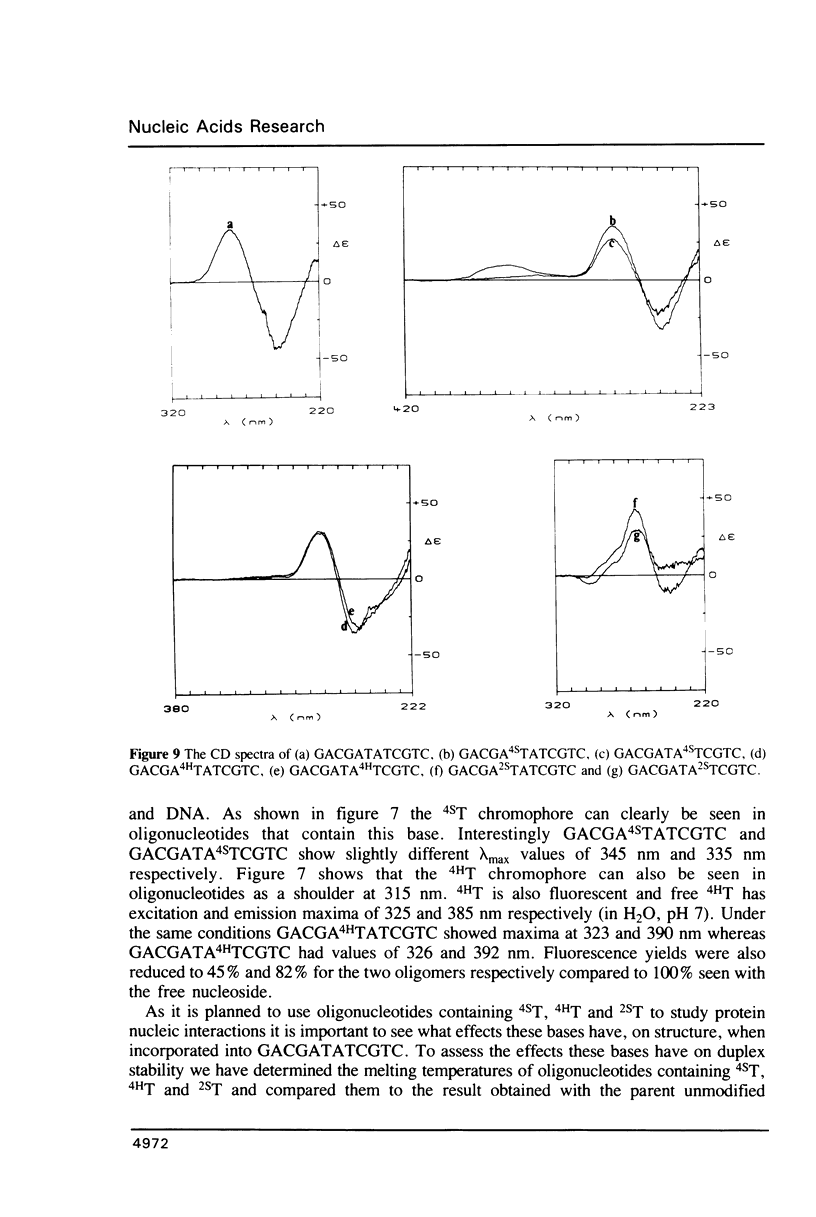
Methods are given for the synthesis of derivatives of 4-thiothymidine (4ST), 5-methyl-2-pyrimidinone-1-beta-D(2'-deoxyriboside) (4HT) and 2-thiothymidine (2ST) suitable for incorporation into oligodeoxynucleotides by the cyanoethyl phosphoramidite method. 4HT and 2ST are incorporated with no base protection but the sulphur atom in 4ST is protected with an S-sulphenylmethyl (-SCH3) function. This can be removed with dithiothreitol after synthesis. These T analogues have been incorporated into GACGATATCGTC, a self-complementary dodecamer containing the Eco RV recognition site (underlined), in place of the two T residues within this site. Although pure dodecamers are obtained in each case the syntheses are not as efficient as those seen when normal unmodified bases are used mainly due to the chemical reactivity of 4ST, 4HT and 2ST. Some of the chemical properties of oligonucleotides containing these bases (reactivity towards NH3) as well as their physical properties (melting temperatures, U.V., fluorescence and circular dichroism spectra) have been determined and are discussed.
Full text
PDF












Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bloxham D. P., Wilton D. C. Modification of pig heart lactate dehydrogenase with methyl methanethiosulphonate to produce an enzyme with altered catalytic activity. Biochem J. 1977 Mar 1;161(3):643–651. doi: 10.1042/bj1610643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faerber P., Scheit K. H. Die chemische Synthese von 2.4-Dithio-thymin-nucleosiden. Chem Ber. 1970;103(5):1307–1311. doi: 10.1002/cber.19701030502. [DOI] [PubMed] [Google Scholar]
- Gildea B., McLaughlin L. W. The synthesis of 2-pyrimidinone nucleosides and their incorporation into oligodeoxynucleotides. Nucleic Acids Res. 1989 Mar 25;17(6):2261–2281. doi: 10.1093/nar/17.6.2261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivanov V. I., Minchenkova L. E., Schyolkina A. K., Poletayev A. I. Different conformations of double-stranded nucleic acid in solution as revealed by circular dichroism. Biopolymers. 1973;12(1):89–110. doi: 10.1002/bip.1973.360120109. [DOI] [PubMed] [Google Scholar]
- McLaughlin L. W., Leong T., Benseler F., Piel N. A new approach to the synthesis of a protected 2-aminopurine derivative and its incorporation into oligodeoxynucleotides containing the Eco RI and Bam HI recognition sites. Nucleic Acids Res. 1988 Jun 24;16(12):5631–5644. doi: 10.1093/nar/16.12.5631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pon R. T., Usman N., Damha M. J., Ogilvie K. K. Prevention of guanine modification and chain cleavage during the solid phase synthesis of oligonucleotides using phosphoramidite derivatives. Nucleic Acids Res. 1986 Aug 26;14(16):6453–6470. doi: 10.1093/nar/14.16.6453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rappaport H. P. The 6-thioguanine/5-methyl-2-pyrimidinone base pair. Nucleic Acids Res. 1988 Aug 11;16(15):7253–7267. doi: 10.1093/nar/16.15.7253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulhof J. C., Molko D., Teoule R. The final deprotection step in oligonucleotide synthesis is reduced to a mild and rapid ammonia treatment by using labile base-protecting groups. Nucleic Acids Res. 1987 Jan 26;15(2):397–416. doi: 10.1093/nar/15.2.397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seeman N. C., Rosenberg J. M., Rich A. Sequence-specific recognition of double helical nucleic acids by proteins. Proc Natl Acad Sci U S A. 1976 Mar;73(3):804–808. doi: 10.1073/pnas.73.3.804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha N. D., Biernat J., McManus J., Köster H. Polymer support oligonucleotide synthesis XVIII: use of beta-cyanoethyl-N,N-dialkylamino-/N-morpholino phosphoramidite of deoxynucleosides for the synthesis of DNA fragments simplifying deprotection and isolation of the final product. Nucleic Acids Res. 1984 Jun 11;12(11):4539–4557. doi: 10.1093/nar/12.11.4539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verheyden J. P., Moffatt J. G. Halo sugar nucleosides. I. Iodination of the primary hydroxyl groups of nucleosides with methyltriphenoxyphosphonium iodide. J Org Chem. 1970 Jul;35(7):2319–2326. doi: 10.1021/jo00832a047. [DOI] [PubMed] [Google Scholar]


