Abstract
The uncarboxylated form of the osteoblast-specific secreted molecule osteocalcin is a hormone favoring glucose handling and increasing energy expenditure. As a result, the absence of osteocalcin leads to glucose intolerance in mice, while genetically modified mice with an increase in uncarboxylated osteocalcin are protected from type 2 diabetes and obesity. Here, we tested in the mouse the therapeutic potential of intermittent administration of osteocalcin. We found that daily injections of osteocalcin at either 3 or 30 ng/g/day significantly improved glucose tolerance and insulin sensitivity in mice fed a normal diet. This was attributable, in part, to an increase in both β-cell mass and insulin secretion. When mice were fed a high-fat diet (HFD), daily injections of osteocalcin partially restored insulin sensitivity and glucose tolerance. Moreover, mice treated with intermittent osteocalcin injections displayed additional mitochondria in their skeletal muscle, had increased energy expenditure and were protected from diet-induced obesity. Finally, the hepatic steatosis induced by the HFD was completely rescued in mice receiving osteocalcin daily. Overall, these results provide evidence that daily injections of osteocalcin can improve glucose handling and prevent the development of type 2 diabetes.
Keywords: Glucose, Insulin, Osteocalcin, High fat diet, Energy expenditure, Liver steatosis
1. INTRODUCTION
In recent years, a converging spectrum of observations has established that bone is an endocrine organ regulating, among other functions, glucose metabolism and energy expenditure [1-3]. The hormone responsible for these functions is osteocalcin that, when in its uncarboxylated form, favors β-cell proliferation, insulin expression and secretion by β-cells of the pancreas and sensitivity to insulin in peripheral tissues [4, 5]. Several clinical investigations support the notion that an association between glucose metabolism and osteocalcin exists in humans [6-13]. Most of these studies however, did not take into account uncarboxylated osteocalcin levels or the effect of vitamin K status. In the mouse, some, if not all, of the osteocalcin functions are mediated through a recently identified receptor expressed in osteocalcin target cells [14, 15]. The biological importance of this hormone is such that genetically modified mice demonstrating an increase in osteocalcin activity are protected from diet-induced obesity and type 2 diabetes [4].
Based on these observations gathered in several laboratories [4, 5, 16], and using genetically modified animals, the therapeutic relevance of osteocalcin has been tested in mice through continuous delivery of this molecule via subcutaneous osmotic pump [17]. Although the results were encouraging, the mode of administration was cumbersome. To circumvent this difficulty, we tested whether administration of osteocalcin by daily injections, a mode of administration used for other hormonal treatments [18-20], would be a viable alternative.
Here we show that once-a-day injections of osteocalcin are at least as efficient as its continuous infusion to lower blood glucose and increase β-cell mass, insulin secretion and sensitivity in mice fed a normal diet. Furthermore, we show that this treatment partially corrects the glucose intolerance caused by a high-fat diet, and fully rescued the liver steatosis caused by the same diet. These data add credence to the emerging notion that osteocalcin could be a treatment for type 2 diabetes.
2. MATERIAL AND METHODS
2.1. Recombinant Osteocalcin Purification
Purification of bacterially produced mouse uncarboxylated osteocalcin was performed as described [17]. Concentration and integrity of the recombinant osteocalcin protein was determined using an osteocalcin ELISA assay [21], previously calibrated against a commercially available osteocalcin RIA (Immutopics). Endotoxin concentration in the recombinant osteocalcin preparations was determined as being below the detection limit (0.12 EU) of the Limulus Amebocyte Lysate assay (Cambrex).
2.2. Animals and Daily Injections
C57Bl/6J mice were purchased from The Jackson Laboratory. Recombinant osteocalcin was freshly diluted in saline solution (0.9% NaCl) at a concentration of 0.3 ng/μl or 3 ng/μl and mice were injected once a day (6 p.m.) intraperitoneally (i.p.) with 10 μl/g of this solution or with saline solution (vehicle). In the study on normal diet, daily injections were initiated at 8 weeks of age. In the high-fat diet study, mice were fed a high-fat diet (58% fat, Research diet D12331) starting at 8 weeks of age and daily injections of osteocalcin or vehicle were initiated 8 weeks later.
2.3. Blood Parameters Measurement
Feeding and fasted (6 h or 16 h) blood glucose were measured in the morning using an Accu-Check glucometer (Roche). Serum insulin was measured using an ELISA assay (Mercodia). HOMA-IR was calculated using this formula [22]:
2.4. Metabolic Tests
In mice maintained on normal chow diet, glucose tolerance tests were performed following 16 h overnight fasting. A two g/kg dose of glucose was administrated by i.p. injection, and blood glucose was measured at the indicated time points. In experiments involving glucose-intolerant mice maintained on high-fat diet, animals were fasted for only 6 h and a dose of 1 g/kg of glucose was injected. Insulin tolerance tests were performed after 4 h of fasting: insulin (Humulin, Lilly; 0.5 or 1.5 units/kg) was injected i.p., and blood glucose was measured at the indicated time points. In the glucose-stimulated insulin secretion test, glucose was injected (3 g/kg) in mice after an overnight fast. Serum was then collected from tail veins at the indicated times and serum insulin was subsequently measured by ELISA (Mercodia).
2.5. Physiological Measurements
To measure food intake, mice were individually housed in metabolic cages (Nalgene) and fed ad libitum. Food consumption was determined by weighing the powdered diet before and after a 24 h period. Energy expenditure parameters were measured using a six-chamber Oxymax system (Columbus Instruments). After a 24 h acclimatation period, oxygen consumption (VO2) and carbon dioxide production (VCO2) data were collected for 24 h. Heat production was calculated by indirect calorimetry using the following formulas:
Physical activity was measured using infrared beams connected to the Oxymax system. Ambulatory activity (xamb) corresponds to a count of beam breaks during the interval. Total activity (xtot) represents fine movement (i.e. grooming) and ambulatory activity.
2.6. Pancreas Histomorphometry
Pancreata were fixed in 10% neutral formalin, embedded in paraffin and sectioned at 5 μm. Immunohistochemistry was performed using rabbit anti-insulin (SataCruz, 1:100) and ABC Elite kits and conterstained with Mayer's hematoxylin. To evaluate islets size and numbers, 3 sections (each 100 μm apart) were analyzed using the Osteomeasure software. β-cell area represents the surface positive for insulin immunostaining divided by the total pancreatic surface. β-cell mass was calculated as the β-cell area multiplied by pancreatic weight. Islet number corresponds to the number of islets divided by the area of pancreas in mm2.
2.7. Muscle Mitochondria Histomorphometry
Superficial gastrocnemius muscles were fixed in 4% PFA/2% glutaraldehyde/0.1 M sodium cacodylate pH 7.3, post-fixed in 1% osmium tetraoxide and embedded in epoxy resin (Epon). Ultrathin sections (80 nm) were stained with aqueous uranyl acetate and lead citrate and examined with a JEOL 2000FX transmission electron microscope. Sixteen electron micrographs per mouse were digitized and the area and number of clearly distinguishable mitochondria were analyzed using the Osteomeasure software.
2.8. Liver Histomorphometry
Liver samples (5×5×5 mm) were fixed in paraformaldehyde 4% PFA/1X PBS, washed 3 times with 1X PBS and equilibrated in sucrose 20%/1X PBS, before being embedded in OCT compound (Tissue-Tek). Samples were next sectioned at 10 μm using a cryostat. The sections were air-dried, post fixed in formalin, rinsed with 60% isopropanol, stained with Oil Red O (in 60% isopropanol) and counterstained with hematoxylin. The Oil Red O positive area over total area was quantified using the Image J software.
2.9. Gene Expression Analysis
Real-time PCR was performed on DNAseI-treated total liver RNA converted to cDNA by MMLV reverse transcriptase using Taq SYBR Green Supermix (Biorad) with ROX on an MX3000 instrument; β-actin amplification was used as an internal reference. The primers used were Tnfa forward (5’-TATGGCTCAGGGTCCAACTC-3’) and Tnfa reverse (5’-CTCCCTTTGCAGAACTCAGG-3’), and β-actin forward (5’-GACCTCTATGCCAACACAGT-3’) and reverse (5’-AGTACTTGCGCTCAGGAGGA-3’).
2.10. Statistical Analysis
Results are given as means±standard error of the mean (SEM). Statistical analyses were performed using unpaired, two-tailed Student's t test.
3. RESULTS
3.1. Daily injections of osteocalcin improve glucose tolerance and insulin sensitivity in wild-type mice
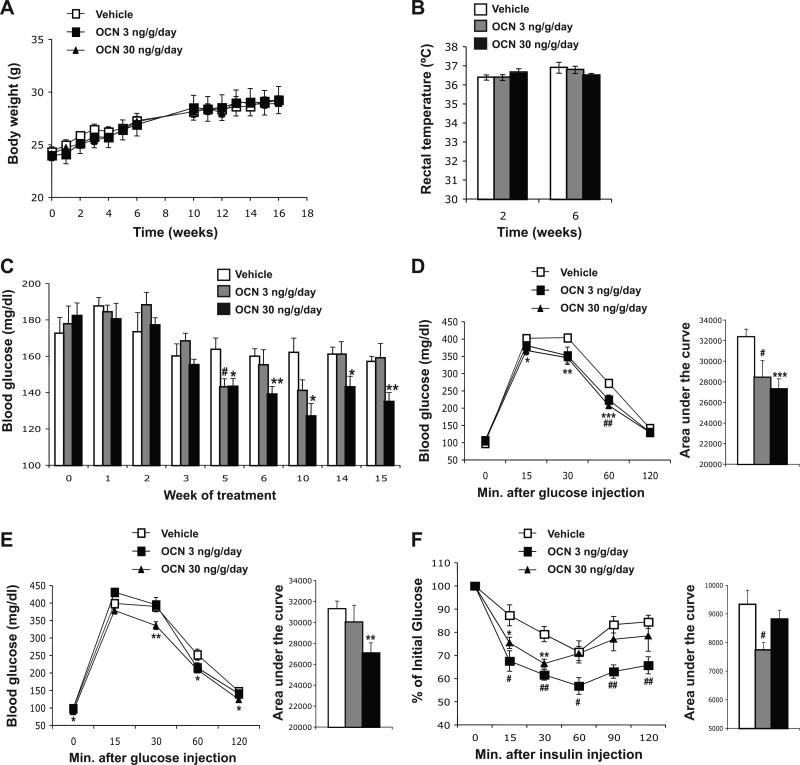
To determine if intermittent administration of osteocalcin can affect glucose metabolism in mice, wild-type animals fed a normal chow diet were injected daily with either saline or osteocalcin (3 or 30 ng/g/day). We used these two doses based on a previous study showing that continuous infusion of 3 or 30 ng/hr osteocalcin have optimal effect on insulin secretion and sensitivity [17]. We first verified that daily injections of osteocalcin had no significant effect on body weight or body temperature (Figure 1A and B). We also measured feeding blood glucose in all treated mice once a week. As shown in Figure 1C, after 5 weeks of injections blood glucose was significantly reduced in mice treated with 30 ng/g/day of osteocalcin. This decrease lasted until the end of the treatment. Blood glucose was also decreased when mice were injected daily with 3 ng/g of osteocalcin although this effect was transient (Figure 1C). Glucose tolerance tests performed after 4 and 8 weeks of treatment revealed that daily injection of osteocalcin improved blood glucose clearance in a dose dependent manner (Figure 1D and E). Finally, both doses of osteocalcin were able to improve insulin sensitivity in wild-type mice as determined by an insulin tolerance test (Figure 1F).
Figure 1. Daily injections of osteocalcin improve glucose tolerance in wild-type mice.
Analyses were performed in mice injected daily with the indicated doses of osteocalcin (OCN) or with vehicle for the indicated time. (A) Body weight variation during the 16 weeks of treatment. (B) Rectal temperature after 2 and 6 weeks of daily injections of osteocalcin; 6 to 7 mice per group were analyzed. (C) Blood glucose measured after feeding. Glucose tolerance test performed after 4 weeks (D) and 8 weeks (E) of treatment. (F) Insulin tolerance test performed after 4 weeks of treatment (Insulin injection: 0.5 U/kg). For D-F the calculated area under the curve is represented in histograms on the right. #: p<0.05, ##: p<0.01 when comparing Vehicle and OCN 3 ng/g/day groups. *: p<0.05, **: p<0.01, ***: p<0.001 when comparing Vehicle and OCN 30 ng/g/day groups. Eight to 15 mice per group were analyzed.
3.2. Daily injections of osteocalcin increase insulin secretion and β-cell mass in wild-type mice
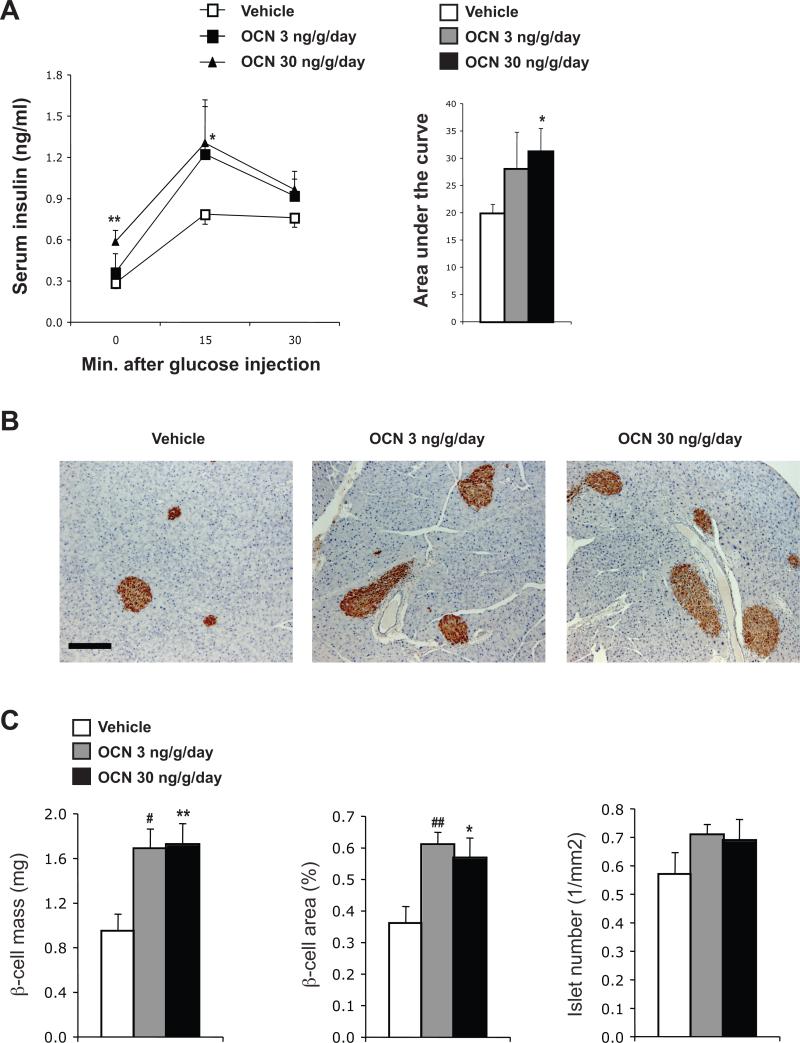
We next tested whether daily injections of osteocalcin in wild-type mice could increase insulin secretion. After 16 weeks of treatment, a glucose-stimulated insulin secretion test demonstrated that insulin secretion was dose-dependently increased by intermittent injections of osteocalcin (Figure 2A). This increased insulin secretion was accompanied by a significant expansion of the β-cell mass and of the area covered by β-cells in the pancreas (Figure 2B and C). Taken together, the results obtained in wild-type mice suggest that intermittent injections of osteocalcin significantly improve glucose tolerance, insulin secretion and insulin sensitivity. Since 30 ng/g/day injections of osteocalcin were more efficient in affecting both insulin sensitivity and insulin secretion, we selected this dose for all subsequent experiments.
Figure 2. Daily injections of osteocalcin increase insulin secretion and β-cell mass in wild-type mice.
Analyses were performed in mice injected daily with the indicated doses of osteocalcin (OCN) or vehicle for 16 weeks. (A) Glucose-stimulated insulin secretion test. The calculated area under the curve is represented in the histogram on the right. (B) Insulin immunostaining (counterstained with hematoxylin) showing larger islets in the pancreas of mice injected daily with osteocalcin (magnification 100X, scale bar: 200 μm). (C) Histomorphometric comparisons of β-cell mass, β-cell area and islet number. #: p<0.05, ##: p<0.01 when comparing Vehicle and OCN 3 ng/g/day groups. *: p<0.05, **: p<0.01 when comparing Vehicle and OCN 30 ng/g/day groups. Six to 15 mice per group were analyzed.
3.3. Daily injections of osteocalcin improve glucose tolerance and insulin sensitivity in mice fed a high-fat diet
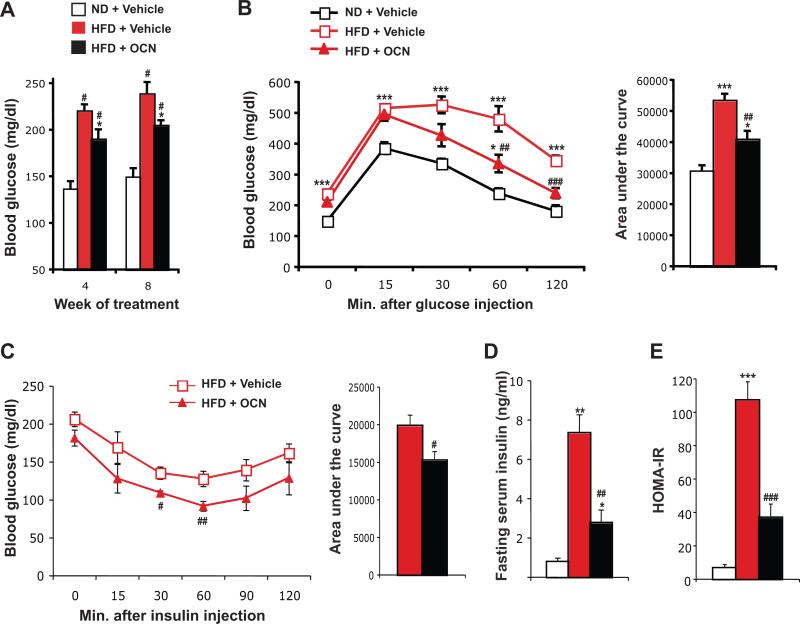
Given the effectiveness of the intermittent injections of osteocalcin in improving glucose tolerance in wild-type mice, we next asked what would be the therapeutic potential of this way of administering osteocalcin in a model of diet-induced type 2 diabetes. In this experiment, 8 week-old mice were fed either a normal diet or a high-fat diet to induce obesity and insulin resistance [23]. Eight weeks later, the mice fed the high-fat diet were randomly separated into two groups, one injected daily with 30 ng/g of osteocalcin and one injected with vehicle alone, while the mice on normal chow diet were injected daily with vehicle. After 4 to 8 weeks of treatment, mice fed the high-fat diet and injected with vehicle displayed elevated fasting blood glucose and serum insulin, glucose intolerance and insulin resistance compared to mice fed a normal diet (Figure 3A-E). Mice fed the high-fat diet and treated with daily injection of osteocalcin displayed reduced fasting glucose and a partial normalization of their glucose tolerance (Figure 3A-B). This improvement in glucose tolerance could be explained, at least in part, by an improvement in insulin sensitivity, as demonstrated by a lowered insulin tolerance test, a 70% reduction in fasting serum insulin and a similar decrease in HOMA-IR (Figure 3C-E). Thus, in this model of type 2 diabetes, daily injections of osteocalcin improve glucose tolerance mainly by favoring insulin sensitivity.
Figure 3. Daily injections of osteocalcin improve glucose tolerance and insulin sensitivity in mice fed a high-fat diet.
Analyses were performed in mice fed a normal diet (ND) or a high-fat diet (HFD) for 8 weeks and then injected daily with 30 ng/g/day of osteocalcin (OCN) or with vehicle for the indicated time. (A) Blood glucose after 6 h fasting. (B) Glucose tolerance test performed after 8 weeks of treatment. (C) Insulin tolerance test performed after 8 weeks of treatment (Insulin injection: 1.5 U/kg). For B and C the calculated area under the curve is represented in histograms on the right. (D) Fasting serum insulin level. (E) HOMA-IR (see Material and Methods). #: p<0.05, ##: p<0.01, ###: p<0.001 when comparing HFD + Vehicle and HFD + OCN groups. *: p<0.05, **: p<0.01, ***: p<0.001 when comparing to ND + Vehicle group. Five to 8 mice per group were analyzed.
3.4. Daily injections of osteocalcin increase energy expenditure but not physical activity and prevent obesity in mice fed a high-fat diet
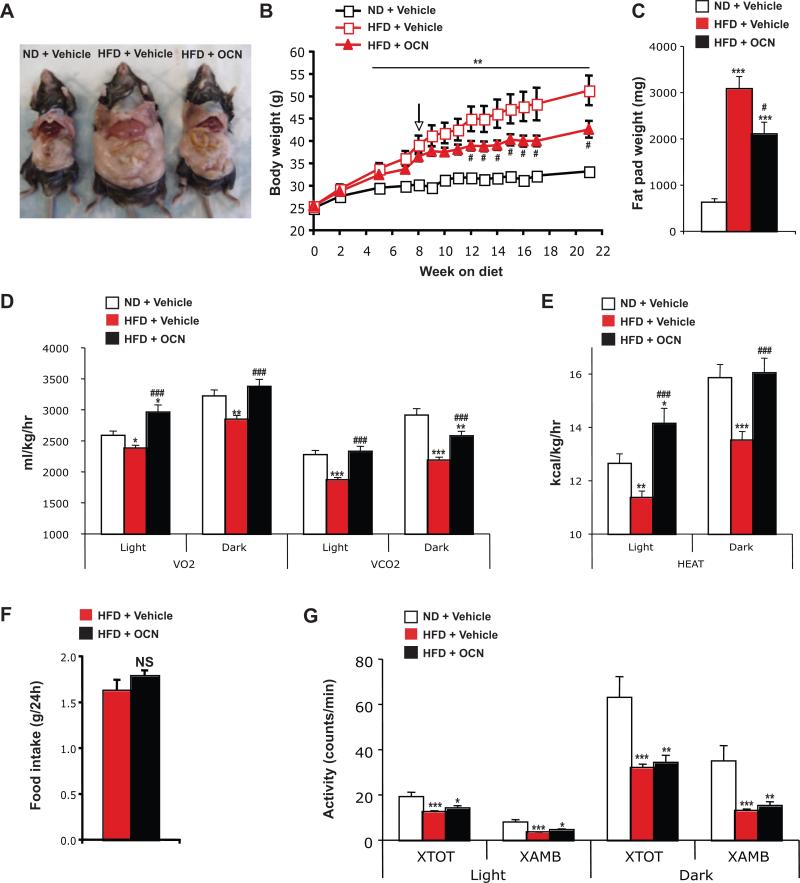
While conducting the aforementioned study, we noticed that the mice fed the high-fat diet and injected daily with osteocalcin were gaining significantly less weight and were leaner than mice fed the same diet but injected with vehicle (Figure 4A and B). This decrease in body weight was associated with a significant decrease in the weight of fat deposits, in particular the epididymal fat pads (Figure 4A and C). To understand how osteocalcin daily injections could prevent body weight gain, we measured oxygen consumption (VO2), carbon dioxide production (VCO2), heat production, food intake and physical activity in these mice. Mice fed the high-fat diet and injected with vehicle presented a reduction in VO2, VCO2 and heat production compared to the mice maintained on a normal chow diet (Figure 4D and E). In contrast, we found that in mice maintained on high-fat diet, daily injection of osteocalcin increased VO2 and VCO2 as well as heat production to a level comparable, or even superior in the case of the light phase, to the one observed in mice fed a normal diet (Figure 4D and E). Importantly, food intake and physical activity were unaffected by intermittent delivery of osteocalcin (Figure 4F and G).
Figure 4. Daily injections of osteocalcin increase energy expenditure and prevent obesity in mice fed a high-fat diet.
Analyses were performed in mice fed a normal diet (ND) or a high-fat diet (HFD) for 8 weeks and then injected daily with 30 ng/g/day of osteocalcin (OCN) or with vehicle for the indicated time. (A) Physical appearance of the mice after 14 weeks of treatment. (B) Body weight curves. The arrow indicates the beginning of the daily injection treatment. (C) Fat pad weight after 14 weeks of treatment. Metabolic rates (D) and heat production (E) after 12 weeks of treatment during light and dark 12h phases. (F) Food intake. (G) Physical activity. VO2, volume of O2 consumed. VCO2, volume of CO2 produced. #: p<0.05, ###: p<0.001 when comparing HFD + Vehicle and HFD + OCN groups. *: p<0.05, **: p<0.01, ***: p<0.001 when comparing to ND + Vehicle group. Five to 11 mice per group were analyzed.
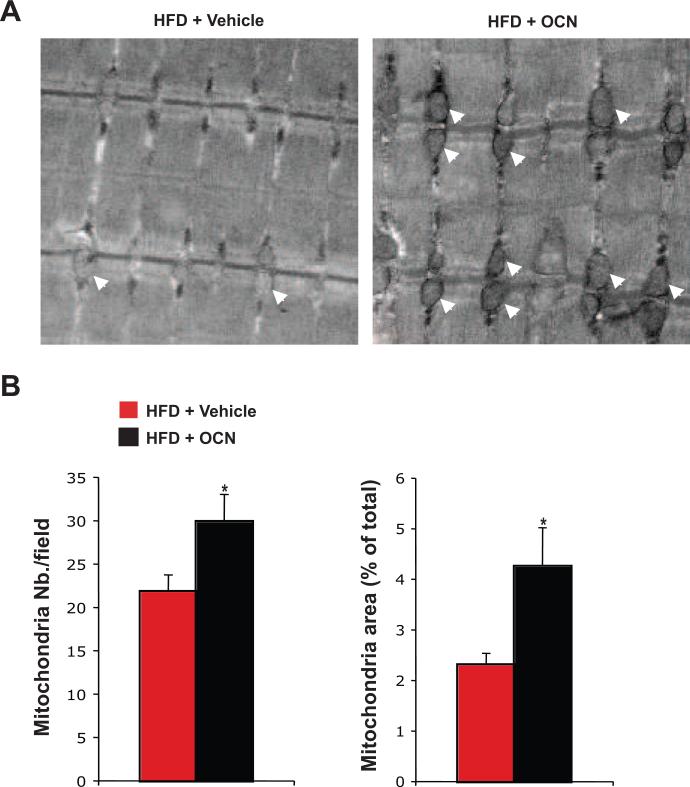
3.5. Daily injections of osteocalcin increase the number of mitochondria in mice fed a high-fat diet
The results obtained in mice fed a high-fat diet suggest that intermittent injections of osteocalcin prevent body weight gain by increasing energy expenditure. In mice, one organ responsible for heat production (i.e. energy expenditure) is brown adipose tissue (BAT). However, we did not observed any significant effect of daily osteocalcin injections on either body temperature or on the expression of genes implicated in BAT thermogenic function such as Ucp1 [24] and Pgc1α [25] (Figure 1B and data not shown), suggesting that BAT was not the mediator of the effect of intermittent osteocalcin injection on energy expenditure.
In both mice and humans, skeletal muscle also possesses mitochondrial capacity for energy expenditure [26]. Morphological analysis of skeletal muscle by transmission electron microscopy revealed larger mitochondria in mice injected daily with osteocalcin as compared to vehicle-injected animals (Figure 5A). Histomorphometric quantification also demonstrated that both the number and the area of mitochondria were significantly increased by intermittent osteocalcin injections (Figure 5B). These results suggest that daily injections of osteocalcin increased energy expenditure in mice maintained on a high-fat diet mainly by increasing the number and size of mitochondria in skeletal muscle.
Figure 5. Daily injections of osteocalcin increase the number of mitochondria in mice fed a high-fat diet.
Analyses were performed in mice fed a normal diet (ND) or a high fat diet (HFD) for 8 weeks and then injected daily with 30 ng/g/day of osteocalcin (OCN) or with vehicle for 14 weeks. (A) Transmission electronic micrograph of the superficial gastrocnemius muscle (magnification 20,000X). Arrowheads indicate the position of individual mitochondria. (B) Histomorphometric quantification of the number of mitochondria and of the mitochondria area. Quantification was performed on 16 different fields per animal and a total of 3 animals per group were analyzed. *: p<0.05 when comparing HFD + Vehicle and HFD + OCN groups.
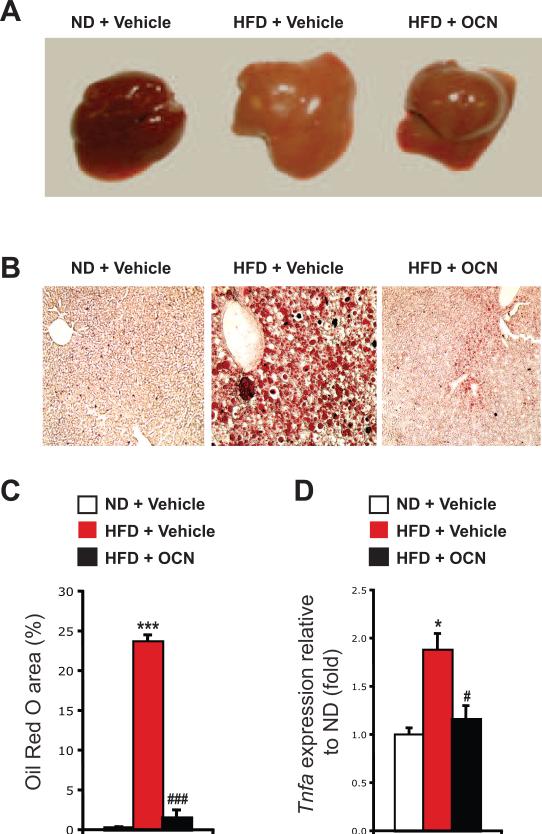
3.6. Daily injections of osteocalcin prevent diet-induced liver steatosis
Mice fed a high-fat diet for several weeks accumulate lipids in their liver and eventually develop liver steatosis [27]. We noticed that in mice injected daily with osteocalcin the liver had a normal appearance (i.e. red), while the liver of the mice injected with vehicle was steatotic (i.e. pale) and enlarged compared with the liver of mice fed a normal diet (Figure 4A and 6A). Remarkably, in liver sections stained with Oil Red O to reveal lipid content, we failed to detect any lipid accumulation in osteocalcin-treated mice fed the high-fat diet (Figure 6B and C). Moreover, expression of the gene encoding TNFα, Tnfa, was increased in the liver of high-fat diet mice, but was normalized by the daily injections of osteocalcin (Figure 6D), indicating that the inflammation which is typically associated with liver steatosis was absent in the mice treated with intermittent osteocalcin injections [28].
Figure 6. Daily injections of osteocalcin prevent liver steatosis in mice fed a high-fat diet.
Analyses were performed in mice fed a normal diet (ND) or a high-fat diet (HFD) for 8 weeks and then injected daily with 30 ng/g/day of osteocalcin (OCN) or with vehicle for 14 weeks. (A) Appearance of the dissected liver after 14 weeks of treatment. (B) Oil Red O staining of liver sections (magnification 100X). (C) Histomorphometric quantification of the Oil Red O positive area in the liver. Quantification was performed on 3 different fields per animals and a total of 4 animals per group were analyzed. (D) Relative expression of Tnfa in liver normalized to β-actin (real time PCR). Four animals per group were analyzed. #: p<0.05, ###: p<0.001 when comparing HFD + Vehicle and HFD + OCN groups. *: p<0.05, ***: p<0.001 when comparing to ND + Vehicle.
4. DISCUSSION
This study demonstrates that daily injections of osteocalcin can significantly affect glucose metabolism in wild-type mice, can improve insulin sensitivity and can prevent obesity in a mouse model of insulin resistance.
4.1. Comparison between intermittent and continuous deliveries of osteocalcin
Importantly, our results show that intermittent and continuous deliveries of osteocalcin have similar effects on insulin secretion and insulin sensitivity [17]. As such, they extend our knowledge of osteocalcin biology by suggesting a more convenient method for administrating this hormone. We observed previously that infusion of high doses of osteocalcin (3-30 ng/h) reduced fat mass and improved insulin sensitivity in wild-type mice without affecting insulin secretion, while low doses (0.3-3 ng/h) were increasing β-cell proliferation and insulin secretion with minimal effect on fat mass [17]. The doses tested here in daily injections (3 and 30 ng/g) can affect both insulin sensitivity and secretion in mice on a normal diet, without affecting body weight and fat mass (Figure 1A, and data not shown). Although 30 ng/g/day injections were clearly more efficient in increasing insulin secretion and glucose tolerance in wild-type mice, the 3 ng/g/day regimen was slightly better in improving insulin sensitivity. It is possible that this decrease in insulin sensitivity in the mice treated with 30 ng/g/day could be secondary to the increased insulin secretion in these animals with the end result of preventing hypoglycemia.
In contrast to what was observed in mice on normal diet, when mice were fed a high-fat diet, osteocalcin daily injections (30 ng/g) could efficiently reduce fat mass and improve insulin sensitivity. The observation that intermittent injections of osteocalcin have a more profound effect in mice with already altered insulin sensitivity suggests that endogenous osteocalcin levels might be related to the development of insulin resistance [6, 29, 30].
4.2. Effect on β-cell mass
We have previously demonstrated that β-cell proliferation was increased in wild-type mice after 4 weeks of continuous infusion of osteocalcin [17]. Here we show that 16 weeks of daily injections of osteocalcin can increase β-cell mass and insulin secretion in wild-type mice. These results raise the testable hypothesis that long-term osteocalcin administration could be a valuable treatment for type I diabetes.
4.3. Effect on energy expenditure and obesity
We are presenting here evidence that intermittent osteocalcin injections efficiently increase energy expenditure and slow down body weight gain in obese mice. Of note, the effect of daily osteocalcin injections on body weight is independent of caloric intake. Instead, this effect appears to result from an increased mitochondrial mass in the skeletal muscles. This result is in line with the observation that Esp-/- mice, which have increased serum levels of undercarboxylated active osteocalcin, also present an increase in muscle mitochondrial area [4]. Interestingly, we found that both continuous and intermittent administration of osteocalcin increase energy expenditure, although they do it through different mechanisms. While osteocalcin infusion significantly increases Ucp1 and Pgc1α in BAT [17], we observed here that daily osteocalcin injections favor energy expenditure mainly by increasing mitochondrial number in the skeletal muscle. The exact mechanism by which osteocalcin stimulates mitochondrial mass in muscle remains to be determined, but the recent identification of a receptor for osteocalcin should allow us to address this question [14].
4.4. Prevention of liver steatosis
Liver steatosis (fatty liver) is often associated with obesity and insulin resistance [28]. We show here that daily injection of osteocalcin can prevent the appearance of liver steatosis in mice fed a high-fat diet. This protective effect of osteocalcin might be linked to the ability of this hormone to decrease serum triglycerides levels and fat accumulation in wild-type and obese mice [17]. It could also be related to the positive action of osteocalcin on insulin sensitivity in muscle, white adipose tissue or other insulin target tissues [4, 17]. Another possibility is that osteocalcin acts directly on hepatocytes to block the accumulation of lipids in the liver. Again, the recent identification of a receptor modulating the action of osteocalcin in testis [14] should help to determine the precise target cells implicated in the metabolic response to this hormone.
In conclusion, this study shows that intermittent injections of osteocalcin have several beneficial effects on energy metabolism in mice: they improve insulin sensitivity, prevent obesity and protect against liver steatosis. Daily injections of osteocalcin thus appear to be an interesting option for the treatment of type 2 diabetes.
ACKNOWLEDGEMENTS
We thank Dr. F. Oury for assistance with the daily injections, and Ms. L. Malynowsky for sectioning and imaging by electron microscopy the muscle samples. This work was supported by grants from the NIH (G.K.), JDRF (P.D.), Canadian Institutes of Health Research (M.D.M) and Fond de la recherche en santé du Québec (M.F.).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Fukumoto S, Martin TJ. Bone as an endocrine organ. Trends Endocrinol Metab. 2009;20(5):230–6. doi: 10.1016/j.tem.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 2.de Paula FJ, Horowitz MC, Rosen CJ. Novel insights into the relationship between diabetes and osteoporosis. Diabetes Metab Res Rev. 2010;26(8):622–30. doi: 10.1002/dmrr.1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Razzaque MS. Osteocalcin: a pivotal mediator or an innocent bystander in energy metabolism? Nephrol Dial Transplant. 2011;26(1):42–5. doi: 10.1093/ndt/gfq721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lee NK, Sowa H, Hinoi E, Ferron M, Ahn JD, Confavreux C, Dacquin R, Mee PJ, McKee MD, Jung DY, Zhang Z, Kim JK, Mauvais-Jarvis F, Ducy P, Karsenty G. Endocrine regulation of energy metabolism by the skeleton. Cell. 2007;130(3):456–69. doi: 10.1016/j.cell.2007.05.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ferron M, Wei J, Yoshizawa T, Del Fattore A, DePinho RA, Teti A, Ducy P, Karsenty G. Insulin signaling in osteoblasts integrates bone remodeling and energy metabolism. Cell. 2010;142(2):296–308. doi: 10.1016/j.cell.2010.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kanazawa I, Yamaguchi T, Yamauchi M, Yamamoto M, Kurioka S, Yano S, Sugimoto T. Serum undercarboxylated osteocalcin was inversely associated with plasma glucose level and fat mass in type 2 diabetes mellitus. Osteoporos Int. 2011;22(1):187–94. doi: 10.1007/s00198-010-1184-7. [DOI] [PubMed] [Google Scholar]
- 7.Kanazawa I, Yamaguchi T, Yamamoto M, Yamauchi M, Kurioka S, Yano S, Sugimoto T. Serum osteocalcin level is associated with glucose metabolism and atherosclerosis parameters in type 2 diabetes mellitus. J Clin Endocrinol Metab. 2009;94(1):45–9. doi: 10.1210/jc.2008-1455. [DOI] [PubMed] [Google Scholar]
- 8.Yeap BB, Chubb SA, Flicker L, McCaul KA, Ebeling PR, Beilby JP, Norman PE. Reduced serum total osteocalcin is associated with metabolic syndrome in older men via waist circumference, hyperglycemia, and triglyceride levels. Eur J Endocrinol. 2010;163(2):265–72. doi: 10.1530/EJE-10-0414. [DOI] [PubMed] [Google Scholar]
- 9.Yamauchi M, Yamaguchi T, Nawata K, Takaoka S, Sugimoto T. Relationships between undercarboxylated osteocalcin and vitamin K intakes, bone turnover, and bone mineral density in healthy women. Clin Nutr. 2010;29(6):761–5. doi: 10.1016/j.clnu.2010.02.010. [DOI] [PubMed] [Google Scholar]
- 10.Winhofer Y, Handisurya A, Tura A, Bittighofer C, Klein K, Schneider B, Bieglmayer C, Wagner OF, Pacini G, Luger A, Kautzky-Willer A. Osteocalcin is related to enhanced insulin secretion in gestational diabetes mellitus. Diabetes Care. 2010;33(1):139–43. doi: 10.2337/dc09-1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kim SH, Lee JW, Im JA, Hwang HJ. Serum osteocalcin is related to abdominal obesity in Korean obese and overweight men. Clin Chim Acta. 2010;411(23-24):2054–7. doi: 10.1016/j.cca.2010.08.046. [DOI] [PubMed] [Google Scholar]
- 12.Diamanti-Kandarakis E, Livadas S, Katsikis I, Piperi C, Aimilia M, Papavassiliou AG, Panidis D. Serum concentrations of carboxylated osteocalcin are increased and associated with several components of the polycystic ovarian syndrome. J Bone Miner Metab. 2010 doi: 10.1007/s00774-010-0211-2. [DOI] [PubMed] [Google Scholar]
- 13.Zhou M, Ma X, Li H, Pan X, Tang J, Gao YC, Hou X, Lu H, Bao Y, Jia W. Serum osteocalcin concentrations in relation to glucose and lipid metabolism in Chinese individuals. Eur J Endocrinol. 2009 doi: 10.1530/EJE-09-0585. [DOI] [PubMed] [Google Scholar]
- 14.Oury F, Sumara G, Sumara O, Ferron M, Chang H, Smith CE, Hermo L, Suarez S, Roth BL, Ducy P, Karsenty G. Endocrine Regulation of Male Fertility by the Skeleton. Cell. 2011;144(5):796–809. doi: 10.1016/j.cell.2011.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pi M, Wu Y, Quarles LD. GPRC6A Mediates Responses to Osteocalcin in beta-Cells In Vitro and Pancreas In Vivo. J Bone Miner Res. 2011 doi: 10.1002/jbmr.390. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fulzele K, Riddle RC, DiGirolamo DJ, Cao X, Wan C, Chen D, Faugere MC, Aja S, Hussain MA, Bruning JC, Clemens TL. Insulin receptor signaling in osteoblasts regulates postnatal bone acquisition and body composition. Cell. 2010;142(2):309–19. doi: 10.1016/j.cell.2010.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ferron M, Hinoi E, Karsenty G, Ducy P. Osteocalcin differentially regulates beta cell and adipocyte gene expression and affects the development of metabolic diseases in wild-type mice. Proc Natl Acad Sci U S A. 2008;105(13):5266–70. doi: 10.1073/pnas.0711119105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Piters KM, Kumar D, Pei E, Bessman AN. Comparison of continuous and intermittent intravenous insulin therapies for diabetic ketoacidosis. Diabetologia. 1977;13(4):317–21. doi: 10.1007/BF01223272. [DOI] [PubMed] [Google Scholar]
- 19.Pertzelan A, Kauli R, Assa S, Greenberg D, Laron Z. Intermittent treatment with human growth hormone (GH) in isolated GH deficiency and in multiple pituitary hormone deficiencies. Clin Endocrinol (Oxf) 1976;5(1):15–24. [PubMed] [Google Scholar]
- 20.Kaji T, Tanaka H, Holst JJ, Redstone H, Wallace L, de Heuval E, Sigalet DL. The effects of variations in dose and method of administration on glucagon like peptide-2 activity in the rat. Eur J Pharmacol. 2008;596(1-3):138–45. doi: 10.1016/j.ejphar.2008.07.070. [DOI] [PubMed] [Google Scholar]
- 21.Ferron M, Wei J, Yoshizawa T, Ducy P, Karsenty G. An ELISA-based method to quantify osteocalcin carboxylation in mice. Biochem Biophys Res Commun. 2010;397(4):691–6. doi: 10.1016/j.bbrc.2010.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Turner RC, Holman RR, Matthews D, Hockaday TD, Peto J. Insulin deficiency and insulin resistance interaction in diabetes: estimation of their relative contribution by feedback analysis from basal plasma insulin and glucose concentrations. Metabolism. 1979;28(11):1086–96. doi: 10.1016/0026-0495(79)90146-x. [DOI] [PubMed] [Google Scholar]
- 23.Rossmeisl M, Rim JS, Koza RA, Kozak LP. Variation in type 2 diabetes--related traits in mouse strains susceptible to diet-induced obesity. Diabetes. 2003;52(8):1958–66. doi: 10.2337/diabetes.52.8.1958. [DOI] [PubMed] [Google Scholar]
- 24.Ricquier D. Respiration uncoupling and metabolism in the control of energy expenditure. Proc Nutr Soc. 2005;64(1):47–52. doi: 10.1079/pns2004408. [DOI] [PubMed] [Google Scholar]
- 25.Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell. 1998;92(6):829–39. doi: 10.1016/s0092-8674(00)81410-5. [DOI] [PubMed] [Google Scholar]
- 26.Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, Messadeq N, Milne J, Lambert P, Elliott P, Geny B, Laakso M, Puigserver P, Auwerx J. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell. 2006;127(6):1109–22. doi: 10.1016/j.cell.2006.11.013. [DOI] [PubMed] [Google Scholar]
- 27.Anstee QM, Goldin RD. Mouse models in non-alcoholic fatty liver disease and steatohepatitis research. Int J Exp Pathol. 2006;87(1):1–16. doi: 10.1111/j.0959-9673.2006.00465.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Browning JD, Horton JD. Molecular mediators of hepatic steatosis and liver injury. J Clin Invest. 2004;114(2):147–52. doi: 10.1172/JCI22422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pittas AG, Harris SS, Eliades M, Stark P, Dawson-Hughes B. Association between serum osteocalcin and markers of metabolic phenotype. J Clin Endocrinol Metab. 2009;94(3):827–32. doi: 10.1210/jc.2008-1422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kindblom JM, Ohlsson C, Ljunggren O, Karlsson MK, Tivesten A, Smith U, Mellstrom D. Plasma osteocalcin is inversely related to fat mass and plasma glucose in elderly Swedish men. J Bone Miner Res. 2009;24(5):785–91. doi: 10.1359/jbmr.081234. [DOI] [PubMed] [Google Scholar]