Abstract
Chromaffin cells are considered as one of the most valuable models to study regulated exocytosis. In these cells, like in other neuroendocrine systems, an intricate cortical cytoskeleton acts as a retentive network impeding vesicle access to plasma membrane. Therefore, during stimulation this structure suffers a transient reorganization allowing active transport of vesicles toward secretory sites. Interestingly, a combination of confocal microscopy studies and mathematical modeling is showing us new aspects of the influence of cortical cytoskeleton in shaping the secretory properties of excitable cells. In this new vision the F-actin-myosin II cortical cytoskeleton is organized forming polygonal cages with the molecular machinery of exocytosis composed by SNARE proteins and voltage-dependent calcium channels associating with its border. In this way the cytoskeleton not only holds together the essential elements acting during secretion, but we proposed that could also act as a structural factor opposing to the free diffusion of the calcium signal and therefore sustains high levels of the intracellular signal triggering exocytosis.
Key words: chromaffin cells, exocytosis, F-actin cytoskeleton, intracellular calcium, SNARE proteins, fluorescence microscopy
Chromaffin cells forming part of the adrenal medulla have been widely used as a model to study the molecular mechanisms of exocytosis.1 In these cells like in other neuroendocrine systems cytoskeletal elements form a dense cortical structure that was thought to impede the access of the catecholamine containing granules to the secretory sites in the plasma membrane.2,3 The initial role of the cortical F-actin cytoskeleton as a barrier was redefined later when it was demonstrated that the same cytoskeletal elements were essential also to actively transport vesicles within the cortical region.4,5 More recently the introduction of transmitted light microscopy implemented in confocal microscopes as an alternative to visualize the dynamics changes occurring in the F-actin cortex during secretion allowed the understanding of how this structure could accomplish a dual role as a simultaneous retentive and transport system.6 In this study, high definition time-lapse images shown that secretagogues induce a Ca2+-dependent, rapid (2–5 s) and transient (40–60 s cycle) disassembling of specific areas of the cortical cytoskeleton. Interestingly, during cytoskeletal reorganization it was observed the formation of channel-alike or open structures perpendicular to the plasma membrane plane conducting vesicles to the cell limits, thus F-actin-myosin II dynamics appear to associate to transient and complex redistributions of viscous cytoskeletal filaments rather than the simple fragmentation of a cortical barrier. In addition to the complex functional role of the cortical cytoskeleton, this technique provided 3-D reconstructions of its organization (see Fig. 1A), depicting the cortex as an intricate and dynamic network presenting multiple free spaces in form of polygonal cages where the vesicles allocate in contact with internal walls.7 Having in mind the complexity of the cortical cytoskeleton it was interesting to examine its influence in different aspects of the secretory cycle starting from the generation of the intracellular calcium signal and ending in the shape of secretory kinetics.
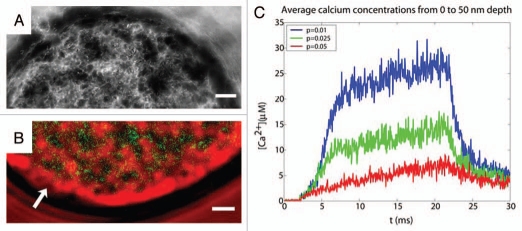
Figure 1.
(A) 3-D reconstruction of the F-actin cortex of a cultured chromaffin cell based in transmitted light images from different planes. The image shows the characteristic network and cages formed by the F-actin cortex. (B) Heterogeneity of intracellular calcium signals during chromaffin cell stimulation. Simultaneous observation of the maximum levels of Fluo-3 signals ([Ca2+]i in green) and transmitted light images of F-actin cytoskeleton (red) showing the spatial coincidence of Fluo-3 signals an empty spaces devoid of cytoskeleton (dark areas). Arrows indicate areas of the F-actin periphery experiencing cortical disruptions. (C) Modeling cytoskeleton cages as diffusion barriers enhances secretory kinetics and sustained calcium levels. Calcium signals for a region between 0 to 50 nm from the membrane surface in a two parallelepiped model. The traces depicted correspond to three increasing porosities of the cytoskeleton: 0.01, 0.025 and 0.05.
A first attempt to solve this matter was recently accomplished by combining fluorescent confocal with transmitted light microscopy to study the distribution of calcium signals, secretory machinery components and the cortical cytoskeleton.8,9 Without any doubt, the location of some of the molecular elements responsible for exocytosis in association with the borders of the cortical cytoskeletal cages was a key finding to understand how this structure could accomplish its role in secretion. In this sense, it was shown that voltage-dependent calcium channels of the L and P/Q subtypes present a preferential disposition in clusters located at the border of the open spaces of variable size formed by the F-actin cortex.9 Moreover, it was demonstrated also that microdomains of t-SNAREs also colocalized with calcium channels associating with the border of cytoskeletal cages. The importance of such a disposition was demonstrated further when it was observed that exocytosis occurs with granules that contact the walls of these cytoskeletal cages.
An important question arises from these observations: what is the benefit of organizing the secretory machinery components in association with a network of cytoskeletal cavities? To answer this question, we proposed mathematical models of secretory behavior based in Monte Carlo algorithms,10 with the inclusion of active zone geometry as cylindrical domains where calcium channel clusters and secretory sites locate either in the walls or in the center of the structure modelled. A quantification of which of the two geometries will lead to a faster secretion resulted in [Ca2+]i maps with most robust initial [Ca2+]i rise in the proximity of the active site when this originates near the border of the simulated spherical cage. In consequence, the predicted [Ca2+]i distributions resulted in a faster exocytotic response when the active site and calcium channels are forming clusters associated with the border of a cytoskeletal cage.9
An important aspect behind our model approach is to study the influence of cytoskeleton walls in the propagation of calcium and, in other words, if the F-actin cytoskeleton is a factor contributing to the heterogeneity of cytosolic [Ca2+]i. Again, we approached this question combining fluorescent confocal determination of [Ca2+]i signals (Fluo-3 levels in green in Fig. 1B) with transmitted light microscopy visualization of cytoskeletal cages (red colored structures in Fig. 1B). This experimental approach certainly revealed that [Ca2+]i signals are higher in the cytosol spaces devoid of cytoskeletal structures (dark spaces in Fig. 1B), but this was observed also with other fluorescent dyes insensitive to calcium.9 Therefore, it seems plausible to conclude that the F-actin structures are a barrier for the free diffusion of relatively low molecular weight compounds such as fluorescent dyes. This does not mean that substances cannot spread along the entire cytoplasm, since the cytoskeletal cages are communicated among them,6 but clearly establishes a new vista of the presence of gel or sol states of the cytoplasm as a factor opposing to the homogeneous distribution of intracellular messengers and metabolites.
If the F-actin cytoskeleton is relatively impermeable to [Ca2+]i, the cytoskeletal porosity might be an important factor to consider when modeling calcium distributions and secretory kinetics. We simplify the modeling approach assuming two parallelepiped cytoskeleton cages with a P/Q calcium channel cluster located in the center. In this model the porosity is determined by the probability of a given ion of crossing cages when encountering one of the five relevant walls and they display the same porosity for incoming and outgoing particles. Figure 1C shows [Ca2+]i time courses from 0 to 50 nm of the membrane surface for three different porosities of 0.01, 0.025 and 0.05. As expected a 2–5-fold change in porosity indicating different barrier properties produces very different levels of [Ca2+]i, therefore suggesting that the F-actin dense walls of cytoskeletal cages could be acting as factors determining the persistence and heterogeneity of calcium signals in chromaffin cells and in consequence determining fast and sustained secretory kinetics.
In conclusion, a multidisciplinary approach combining different confocal microscopy techniques with mathematical modeling of secretory active zone geometry is revealing that the influence of the cortical F-actin cytoskeleton is a key factor to hold together the molecular constituents of the secretory machinery, to determine the precise site for granule exocytosis and to shape the secretory responses in neuroendocrine cells.
Acknowledgments
This work was supported by grants from the Spanish Ministry of Science and Innovation (MICINN, BFU200800731) and the Generalitat Valenciana (ACOMP2009/044) to L.M.G., and Fundación BBVA and I-MATH project C3-0136 to A.G.
References
- 1.Bader MF, Holz RW, Kumakura K, Vitale N. Exocytosis: the chromaffin cell as a model system. Ann NY Acad Sci. 2002;971:178–183. doi: 10.1111/j.1749-6632.2002.tb04461.x. [DOI] [PubMed] [Google Scholar]
- 2.Trifaro JM, Bader MF, Doucet JP. Chromaffin cell cytoskeleton: its possible role in secretion. Can J Biochem Cell Biol. 1985;63:661–679. doi: 10.1139/o85-084. [DOI] [PubMed] [Google Scholar]
- 3.Aunis D, Bader MF. The cytoskeleton as a barrier to exocytosis in secretory cells. J Exp Biol. 1988;139:253–266. doi: 10.1242/jeb.139.1.253. [DOI] [PubMed] [Google Scholar]
- 4.Lang T, Wacker I, Wunderlich I, Rohrbach A, Giese G, Soldati T, et al. Role of actin cortex in the subplasmalemmal transport of secretory granules in PC-12 cells. Biophys J. 2000;78:2863–2877. doi: 10.1016/S0006-3495(00)76828-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Oheim M, Stuhmer W. Tracking chromaffin granules on their way through the actin cortex. Eur Biophys J. 2000;29:67–89. doi: 10.1007/s002490050253. [DOI] [PubMed] [Google Scholar]
- 6.Giner D, Neco P, Frances MM, Lopez I, Viniegra S, Gutierrez LM. Real-time dynamics of the F-actin cytoskeleton during secretion from chromaffin cells. J Cell Sci. 2005;118:2871–2880. doi: 10.1242/jcs.02419. [DOI] [PubMed] [Google Scholar]
- 7.Giner D, Lopez I, Villanueva J, Torres V, Viniegra S, Gutierrez LM. Vesicle movements are governed by the size and dynamics of F-actin cytoskeletal structures in bovine chromaffin cells. Neuroscience. 2007;146:659–669. doi: 10.1016/j.neuroscience.2007.02.039. [DOI] [PubMed] [Google Scholar]
- 8.Villanueva J, Torregrosa-Hetland CJ, Gil A, Gonzalez-Velez V, Segura J, et al. The organization of the secretory machinery in chromaffin cells as a major factor in modeling exocytosis. HFSP J. 2010;4:85–92. doi: 10.2976/1.3338707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Torregrosa-Hetland CJ, Villanueva J, Giner D, Lopez-Font I, Nadal A, Quesada I, et al. The F-actin cortical network is a major factor influencing the organization of the secretory machinery in chromaffin cells. J Cell Sci. 2011;124:727–734. doi: 10.1242/jcs.078600. [DOI] [PubMed] [Google Scholar]
- 10.Segura J, Gil A, Soria B. Modeling study of exocytosis in neuroendocrine cells: influence of the geometrical parameters. Biophys J. 2000;79:1771–1786. doi: 10.1016/S0006-3495(00)76429-0. [DOI] [PMC free article] [PubMed] [Google Scholar]