Abstract
Background
Free radical stress leads to tissue injury and can eventually to arthritis, atherosclerosis, diabetes mellitus, neurodegenerative diseases and carcinogenesis. Several studies are ongoing worldwide to find natural antioxidants of plant origin. We assessed the in-vitro antioxidant activities and screened the phytochemical constituents of methanolic extracts of Pyrostegia venusta (Ker Gawl) Miers.
Methods
We evaluated the antioxidant potential and phytochemical constituents of P. venusta using 1,1-Diphenyl-2-picrylhydrazyl (DPPH), 2, 2'-azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) and ferric reducing antioxidant power (FRAP) assays. Gas chromatography-mass spectroscopy (GC-MS) studies were also undertaken to assess the phytochemical composition of the flower extracts.
Results
Phytochemical analyses revealed the presence of terpenoids, alkaloids, tannins, steroids, and saponins. The reducing ability of both extracts was in the range (in μm Fe(II)/g) of 112.49-3046.98 compared with butylated hydroxytoluene (BHT; 63.56 ± 2.62), catechin (972.02 ± 0.72 μm) and quercetin 3208.27 ± 31.29. A significant inhibitory effect of extracts of flowers (IC50 = 0.018 ± 0.69 mg/ml) and roots (IC50 = 0.026 ± 0.94 mg/ml) on ABTS free radicals was detected. The antioxidant activity of the extracts of flowers (95%) and roots (94%) on DPPH radicals was comparable with that of ascorbic acid (98.9%) and BHT (97.6%). GC-MS study revealed the presence of myoinositol, hexadecanoic acid, linoleic acid, palmitic acid and oleic acid in the flower extracts.
Conclusion
These data suggest that P. venusta is a natural source of antioxidants. The extracts of flowers and roots of P. venusta contain significant amounts of phytochemicals with antioxidative properties and could serve as inhibitors or scavengers of free radicals. P. venusta could be exploited as a potential source for plant-based pharmaceutical products. These results could form a sound basis for further investigation in the potential discovery of new natural bioactive compounds.
Keywords: Antioxidants, DPPH, Flavonoids, Pyrostegia venusta
Background
Oxygen is essential to many living organisms for the production of energy to fuel biological processes. However, the metabolism of oxygen generates 'free radicals' which induce oxidative damage to biomacromolecules, including DNA, proteins, membrane lipids and carbohydrates [1]. A common theme that underlies the aetiology of several degenerative disorders is free radical stress [2]. Free radicals are reported to be involved in the occurrence of numerous diseases such as cancer, diabetes mellitus, atherosclerosis, cardiovascular diseases, ageing and inflammatory diseases [3-7]. Antioxidants are vital substances because they can protect the body from the damage caused by free radicals. They exert their effect by scavenging the free radicals (i.e. reactive oxygen species (ROS) or reactive nitrogen species) universally present in biological systems [7].
There is increasing interest in the natural antioxidants (e.g. polyphenols (flavonoids and tannins)) present in plants used for medicinal and dietary purposes, which might help to prevent oxidative damage [8]. Many synthetic antioxidants (e.g. butylated hydroxyanisole (BHA)) are very effective. However, they possess certain side effects and are toxic to humans [9,10]. Hence, compounds (especially those from natural sources capable of protecting against ROS-mediated damage) may have potential applications in the prevention and/or cure of certain human diseases.
Pyrostegia venusta (Ker-Gawl) Miers (family, Bignoniaceae) is a neotropic evergreen vine widely distributed in southern Brazil. Native Brazilians use the aerial parts of P. venusta for the treatment of cough and flu. They administer its decoction orally as a general tonic and also as an infusion to treat diarrhoea, vitiligo, and jaundice [11-13]. Tonics made from the stems of this plant are useful for the treatment of diarrhoea, whereas flower preparations have been shown to attenuate vomiting [13]. Chemical investigations have shown that methanolic extracts of the roots of P. venusta contain allantoin, steroids, flavonone hesperidin (4,7-O-b-D-rutinosil-3',5-dihydroxy-4'-methoxyiflavanona) and 3-b-b-D-glicopiranosilsitosterol [11]. Similar observations regarding the isolation of n-hentriacontan (n-C31H64) 7-O-b-D-glicopiranosilacacetina), meso-inositol (myo-inositol) as well as several amino acids and sugars have been observed in the flowers [13].
After careful review of the literature, the methanolic extracts of the flowers and roots of this plant were screened for antioxidant properties. Furthermore, the flower extract was chosen for gas chromatography-mass spectroscopy (GC-MS) study to justify its prominent antioxidant activity. A considerable body of research in this area is poised to provide the pharmacological basis for the development of novel treatments based upon the unique ability to selectively eliminate free radicals. If such medicinal potential was gauged correctly, then use of this plant could justify and provide a novel pathway for the treatment of diseases such as arthritis.
Methods
All chemicals and reagents used in the present study were of analytical grade. They were purchased from Sigma Life Sciences (Mumbai, India).
Collection and validation of samples
The flowers and roots of P. venusta were collected from Bhopal (capital of Madhya Pradesh, India). Plants were cross-identified by their vernacular names and later validated at the Department of Botany, Sarojini Naidu Government Girls P.G. College (Bhopal, India). Voucher specimens (accession number Bot./210609 and Bot./210610) were deposited for future reference in the herbarium of Sarojini Naidu Government Girls P.G. College.
Processing of samples of roots and flowers
The withered flowers and roots (250-g each) of this plant were washed vigorously with tap water to remove soil and dust. The flowers and roots were left in the shade to dry for 15-20 days. All dried material was chopped into small fragments. They were then reduced into a fine powder with a mortar and pestle. The powder could then pass through a sieve of pore size 0.5 mm. Powdered samples were extracted at room temperature thrice with methanol for 48 h on an orbital shaker to make methanolic extracts [14,15]. Finally, the methanolic extracts were concentrated using a rota-evaporator (4001; Heidolph Instruments, Schwabach, Germany) at a reduced pressure and at < 40°C.
Phytochemical analyses
The presence of phytochemicals such as alkaloids, saponins, tannins (5% ferric chloride), terpenoids (2,4-dintrophenyl hydrazine) and steroids (Liebermann-Burchard test) were evaluated according to the methods described by Edeoga et al. [16].
Alkaloids
Dragendorff's reagent was prepared by mixing 0.4 g of bismuth subnitrate in 10 ml HCl (12 N) with 5 g of potassium iodide in 50 ml distilled water. Then, 0.5 g of the extract were stirred with 5 ml of 1% aqueous HCl on a steam bath. A few drops of Dragendorff's reagent were used to treat 1 ml of the filtrate. Orange precipitation indicated the presence of alkaloids.
Steroids
Acetic anhydride (2 ml) was added to 0.5-g methanolic extracts in 2 ml of H2SO4. The change in colour from violet to blue or green indicated the presence of steroids.
Terpenoids
The Salkowski test was undertaken to ascertain if terpenoids were present. Five millilitres of extract were mixed in 2 ml of chloroform and layered over 3 ml of concentrated H2SO4. A reddish-brown colour of the interface demonstrated the presence of terpenoids.
Tannins
About 0.5 g of the dried powdered sample was boiled in 20 ml of water and then filtered. A few drops of 0.1% ferric chloride was added to the filtrate and observed for brownish green or a blue-black colouration. Presence of tannins was further confirmed by the gelatin test. One millilitre of extract (300 mg/ml) was added to 2 ml of sodium chloride (2%), filtered and mixed with 5 ml of 1% gelatine solution. A precipitate indicated the presence of tannins.
Saponins
The frothing test was used to check for the presence of saponins. Two grammes of the methanolic extract was mixed in 20 ml of distilled water, boiled in a water bath, and filtered. Ten millilitres of the filtrate was taken aside, and an additional 5 ml of distilled water added and shaken vigorously to generate a stable, persistent froth. Froth formation indicated the presence of saponins.
In-vitro antioxidant assays
1,1-Diphenyl-2-picrylhydrazyl (DPPH) radical scavenging assay
The effect of extracts on DPPH radicals was estimated according to the method of Blois [17] with minor modifications. The methanolic extract was lyophilised and dilutions from 0.02 mg/ml to 0.1 mg/ml prepared. One millilitre (0.135 mM) of DPPH solution was mixed with 1.0 ml of extract (in methanol). The reaction mixture was vortex-mixed thoroughly and incubated at room temperature in the dark for 30 min. Reduction in the absorbance of the mixture was measured at 517 nm using ascorbic acid as a control. Scavenging of DPPH radicals by the extract was calculated using the following formula:
where Abscontrol is the absorbance of DPPH and Abssample is the absorbance of the DPPH radical + sample extract/standard. The half maximal inhibitory concentration (IC50) values denoted the concentration of sample required to scavenge 50% of DPPH free radicals.
2, 2'-azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) radical scavenging assay
The ABTS assay method was used as directed by the study by Re et al. [18]. ABTS solution (7 mM) and 2.4 mM potassium persulfate (PPS) solution were mixed in equal volume and left to react for 12 h in the dark to prepare a working solution. One millilitre of a diluted working solution of ABTS-PPS was mixed with 1 ml of plant extracts, and the absorbance read at 734 nm after 7 min. ABTS.+the scavenging capacity of the extract were compared with standard butylated hydroxytoluene (BHT). The percentage inhibition of the formation of ABTS.+ was calculated using the following formula:
where Abscontrol is the absorbance of ABTS radical + methanol and Abssample is the absorbance of the ABTS radical + sample extract/standard.
Ferric reducing antioxidant power (FRAP) assay
A modified method of that used by Benzie and Strain [19] was adopted for the FRAP assay. A solution of 20 mM FeCl3·6H2O, 300 mM acetate buffer (3.1 g C2H3NaO2·3H2O in 16 ml C2H4O2, pH 3.6) and 10 mM 2,4,6-tripyridyl-s-triazine (TPTZ) in 40 mM HCl) was prepared. At the time of establishing the assay, 25 ml acetate buffer, 2.5 ml TPTZ, and 2.5 ml FeCl3·6H2O was mixed to prepare the FRAP solution. Plant extract (150 μl) was mixed with 2850 μl of FRAP solution and incubated at room temperature in the dark for 30 min. Absorbance of the intense blue-coloured product (ferrous tripyridyltriazine complex) was measured at 593 nm. The observed absorbance of the sample was calculated by putting the values on a linear standard curve plotted between 200 μM to 1000 μM FeSO4. Results were expressed in μM Fe(II)/g dry mass of methanolic extracts of flowers and roots.
GC-MS analyses of methanolic extracts of P. venusta
Flower extracts of P. venusta were chosen for GC-MS studies due to their potent antioxidant activity. GC-MS analyses were carried out on an Agilent Technologies 7890A-GC system (Agilent Technologies, Santa Clara, CA, USA) coupled to XLMSD-5975C equipment operating in electrospray ionisation (EI) mode at 70 eV. A HP-5 MS column (30 m × 250 μm × 0.25 μm; Sigma-Aldrich, St Louis, MO, USA) was used. The temperature programme was 100-180°C at 15°C min-1 and 180-300°C at 5°C min-1 with a 10-min hold at 300°C. The injector temperature was 250°C. The flow rate of the carrier gas (helium) was 1 ml/min. A split ratio of 1:5 was used. Identification of each individual constituent of the volatile compound was achieved by comparing the retention times with those of authentic compounds as well as the spectral data obtained from the Wiley Library and National Institute of Standards and Technologies library.
Statistical analyses
Statistical analyses of results were undertaken using Statistical Analysis System software 9.2 (SAS, Cary, NC, USA). One-way analysis of variance (ANOVA) was determined using the Student's t- test. Results were considered significant and very significant if P values were < 0.05 and < 0.01, respectively. Observations were recorded in triplicate and represented as the mean ± SD of five separate experiments.
Results
Phytochemical analyses
Phytochemical screening of the methanol extracts of the flowers and roots of P. venusta showed the presence of terpenoids, alkaloids, tannins, steroids, and saponins (Table 1).
Table 1.
Phytochemical screening of methanol extract of flower and root of Pyrostegia venusta (Ker-Gawl.) Miers
| S.No. | Constituents | Methanol extract | |
|---|---|---|---|
| Flower | Root | ||
| 1 | Alkaloids | ||
| • Dragendorff's test | ++ | ++ | |
| 2 | Terpenes and steroids | ||
| • Salkowski test | ++ | ++ | |
| • Libarman- Burchard's test | ++ | ++ | |
| 3 | Tannins | ||
| • FeCl3 test | ++ | ++ | |
| • Gelatin test | ++ | ++ | |
| 4 | Saponins | - | ++ |
| • Frothing test | |||
Key:- = Negative (absent)
++ = Positive (present)
In-vitro antioxidant activity: inhibition of DPPH radicals
The DPPH approach is widely applied to measure the antioxidant properties of compounds. DPPH·is an organic nitrogen radical with ultraviolet-visible absorption in the range 515-520 nm, and the colour of its solution fades upon reduction [20]. The dose-response curve of DPPH radical scavenging activity of the methanolic extracts of the flowers and roots of P. venusta were compared with those of BHT and ascorbic acid (Figure 1). The flower extracts almost identical free-radical scavenging activity (95%) as those of the roots (94%) at 0.1 mg/ml. The scavenging activity of controls (ascorbic acid and BHT) was 98.9% and 97.6%, respectively. The IC50 values obtained for flowers and roots were 0.026 ± 0.41 mg/ml and 0.034 ± 0.52 mg/ml, and for ascorbic acid and BHT were 0.014 ± 0.66 mg/ml and 0.029 ± 0.35 mg/ml, respectively.
Figure 1.
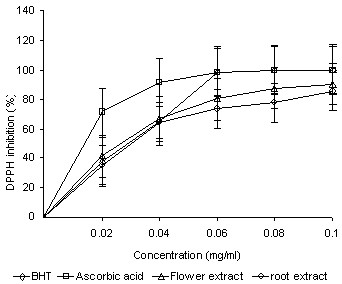
PPH scavenging activities of the methanolic extracts of the flowers and roots of P. venusta.
ABTS radical scavenging assay
ABTS oxidized with PPS (absorption maxima at 734 nm) leads to the generation of ABTS free radicals. This method is based on the ability of antioxidants to quench the ABTS·+ radical cation [18]. Methanol extracts of the flowers and roots of P. venusta were rapid and effective scavengers of the ABTS radical (Figure 2) and this activity was comparable with that of BHT. At 0.1 mg/ml, the percentage inhibition was 98% for BHT, 96% for flower extracts, and 85% for root extracts. The IC50 value for BHT, flowers, and roots were 0.012 ± 0.33 mg/ml, 0.018 ± 0.69 mg/ml and 0.026 ± 0.94 mg/ml, respectively. The relative reducing power of all the extracts was in the order: BHT > flower extracts > root extracts.
Figure 2.
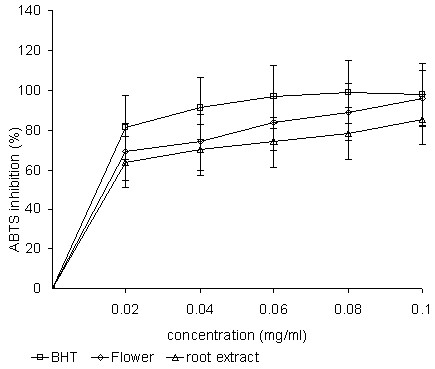
ABTS activities of the methanolic extracts of the flowers and roots of P. venusta.
FRAP assay
The FRAP assay can be used to assess the antioxidant potential in the extracts of flowers and roots of P. venusta by showing their ability to reduce the TPTZ-Fe(III) complex to TPTZ-Fe(II). The reducing ability of root extracts (3046.98 ± 60.87 μm Fe(II)/g) were close to that of quercetin (3208.27 ± 31.29 μm Fe(II)/g), which is the most researched type of flavonoid. The reducing ability of flower extracts was 112.49 ± 37.11 μm Fe(II)/g (Table 2).
Table 2.
Total antioxidant activity of the methanolic extracts of the flowers and roots of Pyrostegia venusta (Ker-Gawl.) Miers
| Extracts | FRAP |
|---|---|
| Flowers | 112.49 ± 37.11 |
| Root | 3046.98 ± 60.87 |
| Ascorbic acid | 1632.1 ± 16.71 |
| BHT | 63.56 ± 2.62 |
| Catechin | 972.02 ± 0.72 |
| Quercetin | 3208.27 ± 31.29 |
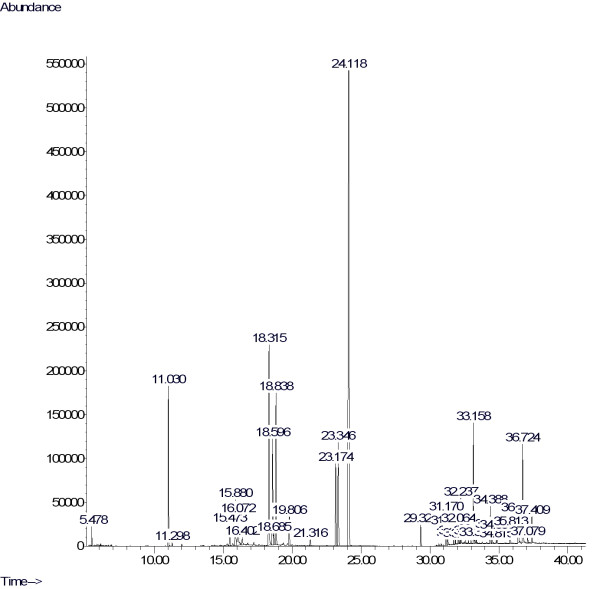
GC-MS study
The GC-MS study indicated that the phytochemicals myoinositol, hexadecanoic acid, linoleic acid, oleic acid, stigmasteryl tosylate, diazoprogesterone, arabipyranose, propanoic acid, pentamethyldisilanyl ester, acetophenone, trans-3-Hexenedioic acid, and 9-Octadecenoic acid (Z)-methyl ester (Table 3 and Figure 3) were in the flower extracts.
Table 3.
Phytocomponents identified in the methanolic extract of flowers of Pyrostegia venusta by GC-MS
| RT | Name of Compound | Molecular | MW | Peak Area (%) |
|---|---|---|---|---|
| 5.478 | Acetophenone | C8H8O | 120.058 | 0.325 |
| 11.032 | .alpha.-l-Mannopyranoside, methyl 6-deoxy-2,3,4-tris-O-(trimethylsilyl)- | C30H70O9Si6 | 394.203 | 4.952 |
| 11.295 | 3H-3a,7-Methanoazulene, 2,4,5,6,7,8-hexahydro-1,4,9,9-tetramethyl-, [3aR-(3a.alpha.,4.beta.,7.alpha.)]- (Synonym Cyperene) | C15H24 | 204.188 | 0.101 |
| 15.473 | trans-3-Hexenedioic acid, bis(trimethylsilyl) ester | C12H24O4Si2 | 288.121 | 0.914 |
| 15.878 | .beta.-DL-Arabinopyranose, 1,2,3,4-tetrakis- O-(trimethylsilyl)- (Synonym- B Arabipyranos | C17H42O5Si4 | 438.211 | 2.498 |
| 16.072 | Ethylmalonate, ethyltrimethylsilyl ester | C8H16O4Si | 232.113 | 2.311 |
| 16.402 | Propionic acid, pentamethyldidilanyl ester | C8H20O2Si2 | 204.1 | 0.519 |
| 18.315 | Glycoside, .alpha.-methyl-trtrakis-O-(trimethylsilyl)- | 482.237 | 11.713 | |
| 18.596 | Hexadecanoic acid, methyl ester (Synonym-Palmitic Acid) | C17H34O2 | 274.196 | 5.394 |
| 18.688 | D-Xylose, tetrakis(trimethylsilyl)- | C18H45NO5Si4 | 438.211 | 0.618 |
| 18.838 | Glycoside,.alpha.-methyl-trtrakis-O-(trimethylsilyl)- | C19H46O6Si4 | 482.237 | 7.364 |
| 19.806 | Gluconic acid, 2-methoxime, tetra(trimethylsilyl)-, trimethylsilyl ester | 583.267 | 1.503 | |
| 23.174 | 9,12-Octadecadienoic acid, methyl ester (Synonym Linoleic acid) | C19H34O2 | 294.256 | 4.225 |
| 23.346 | 9-Octadecenoic acid (Z)-, methyl ester (Synonym OleicAcid) | C19H36O2 | 296.272 | 5.606 |
| 24.118 | Myo-Inositol, 1,2,3,4,5,6-hexakis-O-(trimethylsilyl)- | C24H60O6Si6 | 612.301 | 33.033 |
| 31.17 | Docosanoic acid, methyl ester (Synonym Hysterene) | C23H46O2 | 354.35 | 0.709 |
| 31.291 | 1,2-Benzenedicarboxylic acid, mono(2-ethylhexyl) ester (Synonym Pthalic Acid) | C16H22O4 | 278.152 | 0.454 |
| 31.712 | Methyl 10-methyl-undecanoate | C13H26O2 | 214.193 | 0.12 |
| 31.873 | [1,2,4]Triazolo[1,5-a]pyrimidine-6-carboxylic acid, 4,7-dihydro-7-imino-, ethyl ester | 207.076 | 0.124 | |
| 32.064 | Dotriacontane | C32H66 | 450.516 | 0.402 |
| 32.179 | Silicic acid, diethyl bis(trimethylsilyl) ester | C10H28O4Si3 | 296.13 | 0.074 |
| 32.237 | Tetracosanoic acid, methyl ester | C25H50O2 | 382.381 | 1.12 |
| 32.59 | Di-n-decylsulfone | C20H42O2S | 346.291 | 0.101 |
| 32.984 | Dodecahydropyrido[1,2-b]isoquinolin-6-one | C13H21N | 207.162 | 0.163 |
| 33.159 | Heptacosane | C27H56 | 380.438 | 3.202 |
| 33.264 | Tetrasiloxane, decamethyl- | C10H30O3Si4 | 310.127 | 0.252 |
| 33.367 | Tetradecanoic acid, 12-methyl-, methyl ester | C16H32O2 | 256.24 | 0.18 |
| 34.388 | Stigmasteryl tosylate | C29H48O | 566.379 | 1.493 |
| 34.857 | 2-p-Nitrophenyl-oxadiazol-1,3,4-one-5 | C8H5N3O4 | 207.028 | 0.408 |
| 35.813 | 2-Methyl-6-(5-methyl-2-thiazolin-2-ylamino)pyridine | C10H13N3S | 208.146 | 0.623 |
| 36.418 | Diazoprogesterone | C21H30N4 | 338.247 | 1.511 |
| 36.724 | 1,6-Dibromo-2-cyclohexylpentane | C11H20Br2 | 309.993 | 4.622 |
| 37.079 | Cyclotrisiloxane, hexamethyl- | C6H18Si3 | 222.056 | 0.261 |
| 37.409 | cis-2-Hexen-1-ol, trimethylsilyl ether | C6H12O | 172.128 | 1.308 |
Figure 3.
Chromatogram obtained from GC-MS with the methanolic extracts of the flowers of P. venusta.
Discussion
Interest in finding naturally occurring antioxidants for use in foods or medicinal materials to prevent free-radical imbalance has increased considerably over the past few years [21]. Use of synthetic antioxidants such as butylated hydroxyanisole (BHA) and BHT is restricted due to their carcinogenicity [21]. Therefore, the need for identifying alternate, natural and safe sources of antioxidants (especially of plant origin) has increased in recent years [22]. The therapeutic benefits of secondary metabolites of plant origin have been researched in several recent studies [23]. The past decade has seen considerable changes in the opinion regarding the applications of ethnopharmacological therapeutics.
In the present study, P. venusta was assessed for its antioxidant potential using DPPH, ABTS and FRAP assays. This is the first time that this has been carried out. Phytochemical analyses of P. venusta extracts revealed the presence of terpenoids, alkaloids, tannins, steroids, and saponins. All of these compounds have been shown to be potent antioxidants [24,25].
Terpenoids have been reported to have anti-inflammatory, antioxidant and neuroprotective activities [26]. Saponins and tannins are known to have analgesic and anti-inflammatory properties [27]. Tannins and saponins appear to have considerable cancer-prevention properties [28]. Alkaloid-containing plants have been used by humans for centuries for therapeutic and recreational purposes. They are known for their antimalarial, antimicrobial and cytotoxic activities [29]. Cytotoxic compounds are potentially interesting on their own or as lead compounds for the development of new anti-cancer drugs as well as drugs against parasites and viral infections. P. venusta containing these compounds may serve as a potential source of bioactive compounds in the prevention or cure of free radical-based disorders.
The DPPH test provided information about the reactivity of the tested compounds with a stable free radical. Because of its extra electron, the DPPH radical gives a strong absorption band at 517 nm under visible spectroscopy (a deep purple colour), which vanishes in the presence of a free-radical scavenger. DPPH·is usually employed as a reagent to evaluate the free-radical scavenging activity of antioxidants [30]. In the DPPH·assay, antioxidants could reduce the stable radical DPPH·to the yellow-coloured DPPH. This suggests that the plant extracts contained compounds capable of donating hydrogen to a free radical to remove the extra electron (which is responsible for the activity of free radicals).
Proton-radical scavenging is an important attribute of antioxidants [31]. The protonated radical ABTS has characteristic absorbance maxima at 734 nm, which decreases with the scavenging of proton radicals [31]. The scavenging activity of the ABTS radical by the plant extracts was found to be appreciable. This implies that the plant extracts may be useful for treating free radical-related pathological damage (especially at a higher concentration).
The FRAP assay measures the reducing ability of antioxidants against the oxidative effects of ROS. The reducing potentials of the methanolic extracts of the flowers and roots of P. venusta were estimated from their ability to reduce the TPTZ-Fe(III) complex to the TPTZ-Fe(II) complex.
Assays such as ABTS, FRAP and DPPH have shown that plant extracts may be useful for treating free radical-related pathological damage [21].
In the present study, the flowers of P. venusta were subjected to phytochemical evaluation and GC-MS analyses but the compounds responsible for the antioxidant activity need to be explored. GC-MS analyses revealed the presence of myoinositol, hexadecanoic acid, linoleic acid, oleic acid, stigmasteryl tosylate, diazoprogesterone, arabipyranose, propanoic acid, pentamethyldisilanyl ester, acetophenone, trans-3-Hexenedioic acid, and 9-octadecenoic acid (Z)-methyl ester. These phytochemicals have been shown to possess antimicrobial, anti-cancer, hypercholesterolaemic and anti-ulcerogenic activities (Table 4) [32,33]. The current pioneering study suggests that this extract is a potent therapeutic agent. It paves the way for the development of several treatment regimens based on this extract. In addition, research is continuing to identify and purify the active compounds responsible for antioxidant activity.
Table 4.
Phyto-components and its biological activities obtained through the GC/MS Study of Pyrostegia venusta
| RT | Name of compound | Active biological activity** |
|---|---|---|
| 5.478 | Acetophenone | Antibacterial, fingicide,pesticide, hypnotic, perfimery,sporofic |
| 11.295 | 3H-3a,7-Methanoazulene, 2,4,5,6,7,8-hexahydro-1,4,9,9-tetramethyl-, [3aR-(3a.alpha.,4.beta., 7.alpha.)]-(Cyperene) | Antimalarial and Antiplasmodial |
| 15.473 | Hexadecanoic acid, methyl ester (Synonym-Palmitic Acid) | Antioxidant, hypocholesterolemic nematicide, pesticide, anti-androgenic flavor, hemolytic, 5- Alpha reductase inhibitor |
| 23.174 | 9,12-Octadecadienoic acid, methyl ester (Synonym-Linoleic acid) | Antiinflammatory, hypocholesterolemic cancer preventive, hepatoprotective, nematicide, insectifuge, antihistaminic antieczemic, antiacne, 5-Alpha reductase inhibitor, antiandrogenic, antiarthritic, anticoronary, insectifuge |
| 31.291 | 1,2-Benzenedicarboxylic acid, mono (2-ethylhexyl) ester (Synonym- Pthalic Acid) |
Used in preparation of perfumes and cosmetics, Used as plasticized vinyl seats on furniture and in cars, and clothing including jackets, raincoats and boots. Used in textiles, as dyestuffs, cosmetics and glass making. |
| 24.118 | Myo-Inositol, 1,2,3,4,5,6-hexakis-O-(trimethylsilyl)- | Antidepression, Liver problems, panic disorders and diabetes |
| 23.346 | 9-Octadecenoic acid (Z)-, methyl ester | 5-Alpha-Reductase-Inhibitor, Allergenic, Alpha- Reductase-Inhibitor, Anemiagenic, Antialopecic, Antiandrogenic, Antiinflammatory, Antileukotriene-D4 (Anti-platelet activating factor), Dermatitigenic Insectifuge Perfumery, Propecic Cancer- Preventive, Choleretic, Flavor, Hypocholesterolemic, Irritant, Percutaneostimulant |
| 34.388 | Stigmasteryl tosylate | Antihepatotoxic, Antiinflammmatory, Antiophidic,Antioxidant, Artemecide, Extrogenic, Sedative |
**Activity source: Dr. Duke's Phytochemical and Ethnobotanical Database.
Conclusion
The present study confirmed the in-vitro antioxidant potential of P. venusta, with results comparable with those of standard compounds such as ascorbic acid and BHT. These data further support the view that the flowers and roots of P. venusta are promising sources of natural antioxidants, and could be seen as potential sources of useful drugs. Nonetheless, further in-vivo studies and purification of the compounds responsible for antioxidant activity are needed.
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
VS designed the work. PR, SA and AK were responsible for preparation of the extracts, phytochemical study and in-vitro antioxidation methods. All the authors approved the final manuscript.
Pre-publication history
The pre-publication history for this paper can be accessed here:
Contributor Information
Purabi Roy, Email: purviroy@gmail.com.
Sarika Amdekar, Email: ananyasarika@gmail.com.
Avnish Kumar, Email: avnishkumar81@gmail.com.
Vinod Singh, Email: vsingh3@rediffmail.com.
Acknowledgements
The authors are grateful to the Research Fund of Barkatullah University (Bhopal, India) for laboratory support.
References
- Aiyegoro OA, Okoh AI. Preliminary phytochemical screening and In vitro antioxidant activities of the aqueous extract of Helichrysum longifolium DC. BMC Complementary and Alternative Medicine. 2010;10:21–28. doi: 10.1186/1472-6882-10-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halliwell B, Gutteridge JMC, Cross CE. Free radicals, antioxidants and human disease. Lab Clin Med. 1992;119:598–620. [PubMed] [Google Scholar]
- Di Matteo V, Esposito E. Biochemical and therapeutic effects of antioxidants in the treatment of Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis. Curr Drug target CNS Neurological Disorders. 2003;2:95–107. doi: 10.2174/1568007033482959. [DOI] [PubMed] [Google Scholar]
- Geber M, Boutron-Ruault MC, Hercberg S, Riboli E, Scalbert A, Siess MH. Food and cancer: state of the art about the protective effect of fruits and vegetables. Bull Cancer. 2002;89:293–312. [PubMed] [Google Scholar]
- Kris-Etherton PM, Hecker KD, Bonanome A, Coval SM, Binkosi A, Hilpert KF. Bioactive compounds in foods: their role in the prevention of cardiovascular disease and cancer. Amer J Med. 2002;113:71S–88S. doi: 10.1016/S0002-9343(01)00995-0. [DOI] [PubMed] [Google Scholar]
- Serafini M, Bellocco R, Wolk A, Ekstrom AM. Total antioxidant potential of fruit and vegetables and risk of gastric cancer. Gastroenterol. 2002;123:985–991. doi: 10.1053/gast.2002.35957. [DOI] [PubMed] [Google Scholar]
- Wilson RL. Biochemical mechanisms of liver injury. Academy Press, New York; 1988. Free radicals and tissue damage, mechanistic evidence from radiation studies; p. 123. [Google Scholar]
- Ames BN, Gold LS, Willet WC. The causes and prevention of cancer. Proc Natl Acad Sci USA. 1995;92:5258–5265. doi: 10.1073/pnas.92.12.5258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gülçin I, Bursa E, Şehitoğlu MH, Bilsel M, Gören AC. Polyphenol contents and antioxidant activity of lyophilized aqueous extract of propolis from Erzurum, Turkey. Food and Chemical Technology. 2010;48:2227–2238. doi: 10.1016/j.fct.2010.05.053. [DOI] [PubMed] [Google Scholar]
- Gülçin I, Topal F, Çakmakçı R, Bilsel M, Gören AC, Erdogan U. Pomological Features, Nutritional Quality, Polyphenol Content Analysis, and Antioxidant Properties of Domesticated and 3 Wild Ecotype Forms of Raspberries (Rubus idaeus L.) Journal of Food Sciences. 2011;76:585–593. doi: 10.1111/j.1750-3841.2011.02142.x. [DOI] [PubMed] [Google Scholar]
- Ferreira DT Alvares PS Houghton PJ Braz-Filho R Chemical constituents from roots of Pyrostegia venusta and considerations about its medicinal importance Química Nova 20002342–46.21874234 [Google Scholar]
- Scalon SP, Vieira MC, Lima AA, Souza CM, Mussury RM. Pregerminative treatments and incubation temperatures on the germination of "cipó-de-São-João" [Pyrostegia venusta (Ker Gawl.) Miers]-Bignoniaceae. Revista Brasileirade Plantas Medicinais. 2008;10:37–42. [Google Scholar]
- Velosoa CC, Bitencourta AD, Cabral LD, Franqui LS, Dias DF, dos Santos MH, Soncini R, Giusti-Paiva A. Pyrostegia venusta attenuate the sickness behavior induced by lipopolysaccharide in mice. Journal of Ethnopharmacology. 2010;132:355–358. doi: 10.1016/j.jep.2010.07.053. [DOI] [PubMed] [Google Scholar]
- Gülçin I, Kireçci E, Akkemik E, Topal F, Hisar O. Antioxidant and antimicrobial activities of an aquatic plant: Duckweed (Lemna minor L.) Turkish Journal of Biology. 2010;34:175–188. [Google Scholar]
- Tohma HS, Gülçin I. Antioxidant and radical scavenging activity of aerial parts and roots of Turkish liquorice (Glycyrrhiza glabra L.) International Journal of Food Properties. 2010;13:657–671. doi: 10.1080/10942911003773916. [DOI] [Google Scholar]
- Edeogal HO, Okwu DE, Mbaebie BO. Phytochemical constituents of some Nigerian medicinal plants. African Journal of Biotechnology. 2005;7:685–688. [Google Scholar]
- Blois MS. Antioxidant determination by the use of a stable free radical. Nature. 1958;181:1199–1200. doi: 10.1038/1811199a0. [DOI] [Google Scholar]
- Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans CA. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Bio Med. 1999;26:1231–1237. doi: 10.1016/S0891-5849(98)00315-3. [DOI] [PubMed] [Google Scholar]
- Benzie IFF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": the FRAP assay. Anal Biochem. 1996;239:70–76. doi: 10.1006/abio.1996.0292. [DOI] [PubMed] [Google Scholar]
- Gülçin I. Antioxidant properties of resveratrol: A structure-activity insight. Innovative Food Science and Emerging Technologies. 2010;11:210–218. doi: 10.1016/j.ifset.2009.07.002. [DOI] [Google Scholar]
- Mahdavi DL, Salunkhe DK. In: Food Antioxidants. Mahdavi DL, Deshpande SS, Salunkhe DK, editor. New York: Marcel Dekker; 1995. Toxicological aspects of food antioxidant; pp. 267–293. [Google Scholar]
- Zainol MK, Abd-Hamid A, Yusof S, Muse R. Antioxidant activity and total phenolic compounds of leaf, root and petiole of four accessions of Centella asiatica (L.) Urban. Food Chem. 2003;81:575–81. doi: 10.1016/S0308-8146(02)00498-3. [DOI] [Google Scholar]
- Nayak BS, Lexley MPP. Catharanthus roseus flower extract has wound healing activity in Sprague Dawley rats. BMC Complementary and Alternative Medicine. 2010;10:21–28. doi: 10.1186/1472-6882-10-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hafidh RR, Abdulamir A, Bakar A, Abas F, Jahanshiri F, Sekawi Z. Antioxidant Research in Asia in the Period from 2000-2008. American Journal of Pharmacology and Toxicology. 2009;4(3):48–66. [Google Scholar]
- Anchana Chanwitheesuk, Aphiwat Teerawutgulrag, Nuansri Rakariyatham. Screening of antioxidant activity and antioxidant compounds of some edible plants of Thailand. Food Chemistry. 2005;92:491–497. doi: 10.1016/j.foodchem.2004.07.035. [DOI] [Google Scholar]
- Lingli Mu, Junping Kou, Danni Zhu, Boyang Yu. Comparison of Neuroprotective Effects of Flavonoids, Terpenoids, and Their Combinations from Ginkgo bilobaon Ischemia-Reperfusion-Injured Mice. Pharmaceutical Biology. 2007;45:728–733. doi: 10.1080/13880200701575486. [DOI] [Google Scholar]
- Lai FR, Wen QB, Li L, Wu H, Li XF. Antioxidant activities of water-soluble polysaccharide extracted from mung bean (Vigna radiata L.) hull with ultrasonic assisted treatment. Carbohyd Polym. 2010;81(2):323–329. doi: 10.1016/j.carbpol.2010.02.011. [DOI] [Google Scholar]
- Sur P, Chaudhuri T, Vedasiromoni JR, Gomes A, Ganguly DK. Antiinflamatory and antioxidant property of saponins of tea [Camellia sinensis(L)O. Kuntze] root extract. Phytotherapy Reserach. 2001;15:174–176. doi: 10.1002/ptr.696. [DOI] [PubMed] [Google Scholar]
- Oloyede KG, Oke MJ, Raji Y, Olugbade AT. Antioxidant and Anticonvulsant Alkaloids in Crinum ornatum bulb Extract. World journal of chemistry. 2010;5(1):26–31. [Google Scholar]
- Krishnaraju AV, Rao CV, Rao VN, Reddy KN, Trimurtulu G. In vitro and In vivo Antioxidant Activity of Aphanamixis polystachya Bark. Americal Journal of Infectious Diseases. 2009;2:60–67. [Google Scholar]
- Thaiponga K, Boonprakoba U, Crosbyb K, Cisneros-Zevallosc L. Byrnec DH. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. Journal of Food Composition and Analysis. 2006;19:669–675. doi: 10.1016/j.jfca.2006.01.003. [DOI] [Google Scholar]
- Sakthivel K, Palani S, Santhosh Kalash, Devi K, Senthil Kumar B. Phytoconstituents analysis by GC-MS, cardioprotective and antioxidant activity of Buchanania axillaris against Doxorubicin-Induced cardio toxicity in albino rats. International Journal of Pharmaceutical Studies and Research. 2010;1:34–48. [Google Scholar]
- Valko M, Leibfritz D, Moncola J, Cronin MD, Mazura M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Review Int J Biochem Cell Biol. 2007;39:44–84. doi: 10.1016/j.biocel.2006.07.001. [DOI] [PubMed] [Google Scholar]