Abstract
Objectives
Vocal fold epithelium is exposed to reactive oxygen species from the inhaled environment and from tissue inflammation. The objective of this study was to explore the functional and structural consequences of reactive oxygen species exposure on vocal fold epithelium.
Study Design
In vitro, prospective study design.
Methods
Hydrogen peroxide (H2O2), a common reactive oxygen species, was utilized in this study. Freshly excised, viable porcine vocal fold epithelia (N = 32) were exposed to H2O2 or sham challenge for 2 hours. Electrophysiology, western blotting, and light microscopy were used to quantify the functional and structural effects of reactive oxygen species on vocal fold epithelia.
Results
Exposure to reactive oxygen species did not significantly alter transepithelial resistance. There was a small, non-significant trend for decreased concentration of epithelial junctional complex protein with reactive oxygen species challenge. Minimal changes to the gross structural appearance of vocal fold epithelia were also noted.
Conclusions
The stratified squamous epithelia of the vocal folds effectively defend against an acute reactive oxygen species challenge. The current study lays the groundwork for future investigations on the effects of reactive oxygen species on vocal fold epithelia that are compromised from phonotrauma.
Keywords: H2O2, vocal fold epithelia, reactive oxygen species
INTRODUCTION
Voice users are regularly exposed to environmental pollutants including cigarette smoke, vehicle exhaust, and industrial fumes.1 These common pollutants contain reactive oxygen species (ROS).2 Inhalation of pollutants containing ROS has the potential to adversely affect voice, through stress to the laryngeal mucosa.3 Specifically, the vocal fold epithelium as the external layer of the true vocal folds may be particularly at risk for ROS exposure from the inhaled environment during breathing. ROS can also be produced secondary to tissue inflammation from acid-peptic injury to the larynx,4 and these ROS have been implicated in inducing laryngeal hyperreactivity.5 Despite the high probability of ROS exposure, little is known regarding the direct physiologic effects of ROS on the vocal fold epithelium. Therefore, the current study sought to investigate the effects of high concentration ROS exposure on vocal fold epithelia. Specifically, we examined whether 3mM hydrogen peroxide would compromise the function and structure of native vocal fold epithelia. Hydrogen peroxide (H2O2) is a pervasive ROS found in the inhaled environment and produced systemically as a byproduct of cell metabolism6–9. The functional consequences of H2O2 exposure in monolayered epithelial cell cultures7–10 have been widely investigated, although the effects on stratified squamous epithelia of the vocal folds are not known.
The vocal fold epithelium is composed of multiple layers of squamous cells, and adjacent intercellular junctional complex proteins.11 Together, these structural features contribute to the resistance across the epithelium. High transepithelial resistance is essential for maintaining epithelial barrier function, which is critical for preventing airborne irritants and pathogens from entering the lamina propria. However, in epithelial cells of the trachea, stomach, intestines, and kidneys, ROS exposure compromises the integrity of the epithelial barrier by reducing transepithelial resistance, and this disruption to epithelial tissue has been implicated in the pathophysiology of various diseases.10 In gastric epithelia, ROS simulated by exposure to 500µM H2O2 significantly decreases transepithelial resistance.8 Similar results have been reported for intestinal and renal epithelium.7, 10 H2O2 challenge also degrades epithelial barrier integrity by lowering the concentration of proteins in the intercellular junctional complexes.8 In renal10 and airway9 epithelia, H2O2 induces disruption of Zona Occludens (ZO-1) junctional complex protein. Furthermore, ROS have the potential to induce gross structural changes. Exposure to H2O2 results in epithelial thinning and epithelial detachment in guinea pig trachea.12
At this juncture, it is unclear whether ROS such as H2O2 may affect the stratified squamous epithelium of the vocal folds in much the same way as monolayered epithelial cell cultures. Given evidence7, 9, 10 from other epithelia that ROS have the potential to adversely impact barrier integrity, it is crucial to investigate for similar changes to vocal fold epithelia. This question is of clinical and theoretical significance because environmental pollutants are known to compromise phonation1 although the physiological mechanisms underlying the adverse voice changes are unclear. Understanding the underlying mechanism is necessary to develop optimal treatments. The focus of the current study was to investigate the functional and structural consequences of ROS exposure on native, stratified squamous vocal fold epithelia. Given the critical role of the epithelium as a protective barrier, we questioned whether ROS exposure would disrupt barrier integrity by reducing transepithelial resistance and lowering junctional complex protein concentrations. We elected to study one junctional complex protein from the Zona Occludens (ZO) family. ZO-1 protein was selected because it is important for maintaining junction architecture and for interacting and connecting with other proteins that constitute the junctional complex and the cytoskeleton.13 Transepithelial resistance and ZO-1 protein concentrations were assessed using electrophysiology and western blotting techniques, respectively. To examine if changes in epithelial barrier function were accompanied by gross structural damage, light microscopy was employed. Porcine vocal folds were selected due to comparable histological properties between pig and human vocal folds.14 Data from the present study will contribute to our knowledge of the functional and structural consequences of ROS challenge to vocal fold epithelia and lay the foundation for further study into the nature of phonatory changes associated with the interaction of ROS and phonotrauma.
METHODS
Epithelial Tissue
Fresh larynges from domestic pigs (6 months - 2 years) were obtained from the slaughterhouse and transported back to the lab in cold, isotonic saline solution. The vocal fold epithelium was excised from the underlying ligament and muscle using published procedures in our laboratory15–18 and prepared for measurements of transepithelial resistance, ZO-1 protein concentrations, and epithelial structure. These techniques are described below. Across all experiments, vocal folds were exposed to a 2 hour, 3 mM ROS challenge, simulated with H2O2. Similar concentrations of H2O2 decrease transepithelial resistance in alveolar epithelial cell monolayers within 1 hour.19 A 2 hour challenge was selected to account for the use of native, stratified true vocal folds as opposed to monolayer cell cultures.
Transepithelial Resistance (RT)
Transepithelial resistance (RT) was measured using electrophysiological techniques in an Ussing System with associated Voltage Clamp (World Precision Instruments, Sarasota, FL) in accordance with published procedures.15–18 Immediately following excision, epithelial tissues were mounted on a lucite chamber, placed in the Ussing System, and bathed with warm, oxygenated Hanks Balanced Salt Solution (HBSS) in order to maintain tissue viability. Electrodes were placed on either side of the epithelial tissue to monitor potential difference and current. Using Ohm’s Law, RT was computed by measuring the current deflection to an imposed 2mV pulse. Epithelial tissue was allowed 45–60 minutes to reach steady state (hereafter called baseline). A baseline RT of > 300Ω·cm2 was required for epithelial tissue to be included in the experiment. Once epithelial tissue reached baseline, 3mM H2O2 was added to the luminal chamber of the Ussing System to simulate acute ROS challenge (N = 10). To simulate sham challenge, HBSS was added to the luminal chamber (N = 10). RT was monitored continuously for 2 hours post-exposure to ROS or sham challenge. RT data were summarized as means ± standard error of mean. A mixed model repeated measures ANOVA was utilized to assess whether ROS altered RT as compared to sham challenge. An alpha level of < 0.05 was considered statistically significant.
ZO-1 protein concentrations
ZO-1 protein concentrations were measured with western blotting following published procedures in our laboratory.20 Immediately following excision, vocal fold epithelial tissues were diced and incubated in 3mM H2O2 (N = 3) or sham (N = 3) for 2 hours. Next, the tissues were snap-frozen in liquid nitrogen, and stored in −80° C freezer in preparation for western blotting. To determine protein concentrations, samples were homogenized on ice in homogenization buffer (protease inhibitor cocktail, 1.0M Tris pH 7.5, 200mM EGTA, 20% Triton X-100, 10% SDS, 10mg/ml PMSF, 14.21M 2-mercaptoethanol, and DDI water). Samples were sonicated (Model 500 Sonic Dismembrator, Fisher Scientific) at duty cycle 20% and output 4–6 for 30 pulses. Following centrifugation at 12,000g at 4°C for 12 minutes, aliquots of supernatants were assayed for protein concentrations by the Bradford method. 100μg of protein was mixed with an equal volume of 2× sample buffer (0.35M Tris-Cl, 10% SDS, 30% glycerol, 0.6M DTT, and 0.012% bromophenol blue), loaded onto a gradient (7% + 12%) sodium dodecyl sulfate polyacrimide gel, electrophoresed, and then transferred to a PVDF membrane. The membrane was blocked with 5% dry milk in TBST (Tris-Buffered Saline with Tween 20) at room temperature for 1 hour and immunoblotted with primary antibody directly against ZO-1 (2µg/mL, Invitrogen) at 4°C overnight. The membrane was rinsed and stained with a horseradish peroxidase (HRP)-linked species-specific goat anti-mouse IgG antibody (1:3000) at room temperature for 1.5 hours and developed using ECL reagent. The exposure time varied from 30 seconds to several minutes depending on the signal strength. β-actin (1:1000) was used as a loading control, the corresponding secondary antibody (1:5000) for β-actin was HRP-conjugated goat anti-mouse IgG. For determination of internal standard β-actin (42 kDa), the blots were stripped with stripping buffer (Pierce), and immunoblotted with rabbit anti-mouse actin antibody. The bands were analyzed using densitometry with Image J software. The values of ZO-1 proteins were normalized to those of β-actin obtained from the same gel and the percentage of change in ZO-1 protein concentrations were compared for H2O2 and sham groups. An alpha of <0.05 was considered statistically significant.
Epithelial structure
Epithelial structure was assessed using published light microscopy procedures.15 Following excision as described above, epithelial tissues were incubated at 37°C in 3mM H2O2 (N = 3) or sham (N = 3) for 2 hours. Immediately following incubation, vocal fold samples were immersed in 10% buffered neutral formalin and allowed to fixate for 24 hours. Following fixation, samples were dehydrated using an ETOH series and embedded in paraffin for sectioning. 5µm sections were cut and stained with hematoxylin-eosin for light microscopic analysis.
A pathologist blinded to challenge type (H2O2 or sham) assessed the histological data for damage. The pathologist first confirmed that the samples were stratified squamous epithelia and then analyzed structural damage on a 0–5 scale (0=None; 1=Minimal; 2=Mild; 3=Moderate; 4=Marked, 5=Severe). This scale has been used previously by our laboratory to quantify reflux damage in vocal fold tissue.15 The pathologist used the scale to assess for the following: (1) epithelial splitting/shedding, (2) epithelial erosion/attenuation, (3) epithelial intracellular or intercellular edema, (4) basilar to sub-basilar edema/vacuolization, and (5) interstitial edema. Mann-Whitney U tests were used to compare structural damage in tissue exposed to H2O2 and sham challenge. An alpha of < 0.05 was considered statistically significant.
RESULTS
Transepithelial Resistance (RT)
H2O2 challenge did not adversely affect RT (Figure 1). There was a non-significant challenge by time interaction effect (p = 0.44) suggesting that even 2 hours of exposure to ROS did not significantly alter RT as compared to sham challenge. RT increased by 94 Ω·cm2 in epithelial tissue exposed to H2O2 and by 64 Ω·cm2 in epithelial tissue exposed to sham challenge. Epithelial tissue in both groups maintained a robust RT demonstrating high tissue viability despite exposure to challenges for 2 hours.
Figure 1.
Transepithelial resistance (RT) in response to reactive oxygen species (ROS) exposure with H2O2. Tissues in sham challenge were exposed to Hanks Balanced Salt Solution. 2 hours of ROS exposure did not significantly change RT as compared to sham challenge (p > .05). Error bars represent standard error of the mean.
ZO-1 concentrations
Following H2O2 exposure, there appeared to be a slight decrease in the band density of ZO-1 (Figure 2A). However overall, H2O2 challenge did not significantly lower the levels of ZO-1 in vocal fold epithelial tissue as compared to sham challenge (p = 0.17, Figure 2B).
Figure 2.
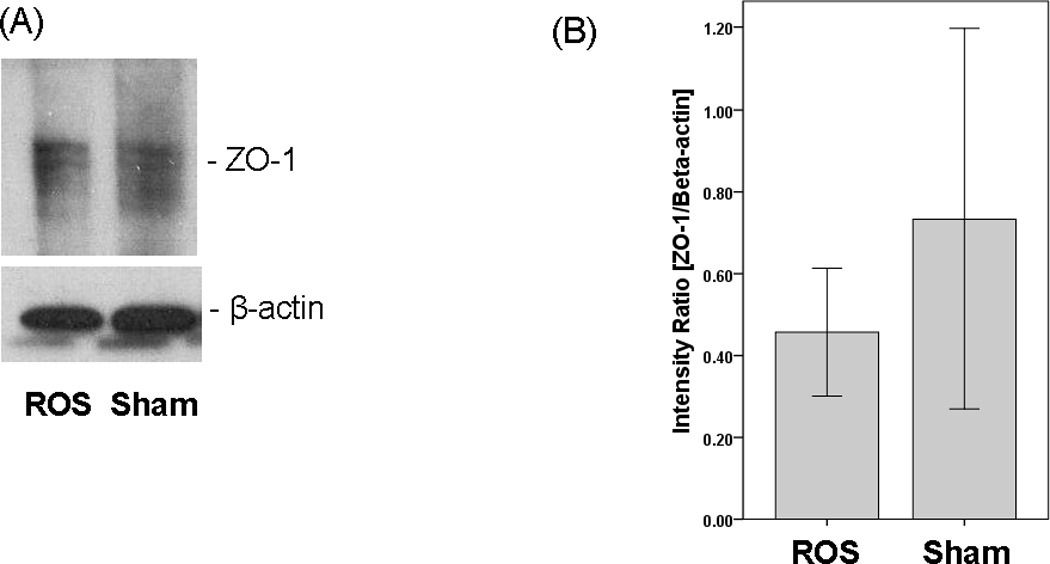
Expression of ZO-1 proteins in vocal fold epithelia following 2 hours of ROS exposure with H2O2. Tissues in sham challenge were exposed to Hanks Balanced Salt Solution. (A) Representative Western Blot. (B) ROS treatment did not significantly alter ZO-1 expression in vocal fold epithelia. Error bars represent standard deviation.
Epithelial structure
Epithelial tissue exposed to H2O2 showed signs of minimal structural damage (Figure 3). However, the structural changes associated with H2O2 exposure were not statistically significant as compared to sham challenge for any of the damage criteria: epithelial splitting/shedding (p = 0.82), epithelial erosion/attenuation (p = 1.00), epithelial intracellular or intercellular edema (p = 1.00), basilar to sub-basilar edema/vacuolization (p = 0.64) and interstitial edema (p = 0.82).
Figure 3.
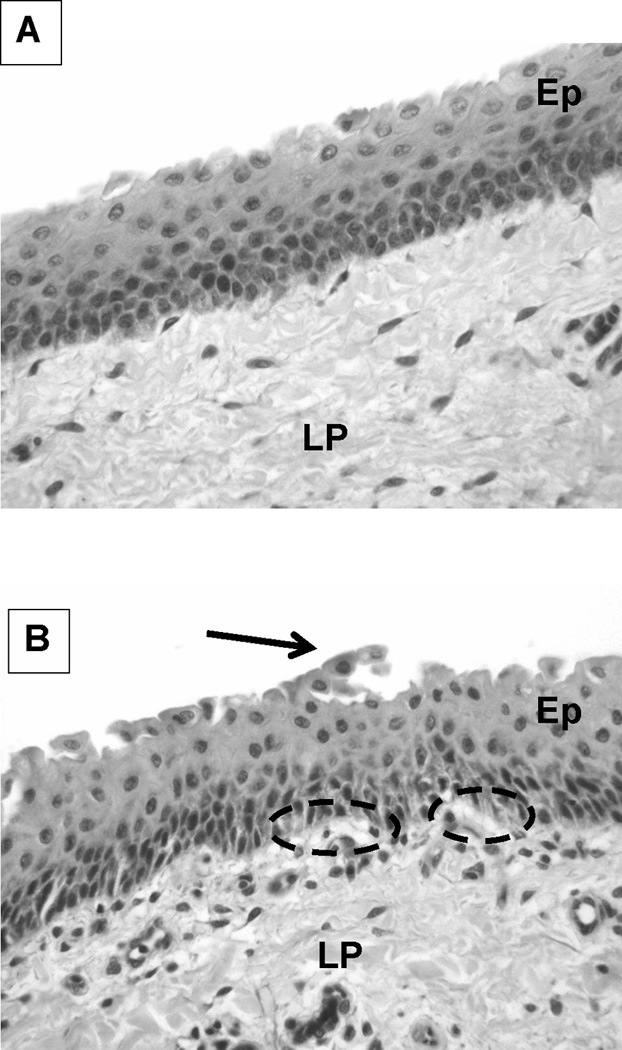
Light microphotographs (H&E stain, 40X magnification) of representative vocal fold epithelial samples obtained after 2 hours of exposure to (A) Sham and (B) ROS with H2O2. Ep = stratified squamous epithelium; LP = lamina propria; arrowhead = epithelial shedding; dashed oval = sub-basilar edema or vacuolization.
DISCUSSION
This study examined whether acute exposure to reactive oxygen species (ROS) is detrimental to vocal fold epithelial structure and function. Simulating ROS by exposing epithelial cells to H2O2 is a widely used methodology.7, 9, 21, 22 H2O2 was added to the apical surface of vocal fold epithelia in order to simulate ROS exposure from inhaled sources or that generated from inflammatory cells that migrate to the airway lumen. Here we show that continuous exposure to 3mM H2O2 did not significantly reduce the transepithelial resistance (RT). Although there was a decrease in ZO-1 protein concentrations after exposure to H2O2 as compared to sham challenge, this decrease did not reach statistical significance. Additionally, there was minimal, non-significant evidence for gross structural change after H2O2 exposure. Some of the structural changes include epithelial shedding and vacuolization as depicted in Figure 3.
There are different hypotheses for why the predicted, adverse effects of ROS on vocal fold epithelium were not observed in this study. H2O2was purposely selected as the ROS in this study because it is commonly found in the inhaled environment and is also produced secondary to tissue inflammation.6, 8 In cultured human bronchial epithelial cells, ROS challenge with 0.1mM H2O2 lowered RT and disrupted ZO-1 staining patterns.9 At higher concentrations, H2O2 may be detrimental to cell viability in monolayered epithelial cell culture.23 Our data demonstrating that 3mM H2O2 did not significantly reduce RT or ZO-1 protein concentrations, demonstrates that the viability of stratified squamous vocal fold epithelia were not compromised even after high concentration H2O2 exposure. These data suggest that healthy, stratified squamous epithelium may be able to tolerate acute ROS exposure without adverse consequences to barrier integrity. To explore this hypothesis further, future studies will investigate the effects of a series of ROS concentrations.
Another possibility for the non-significant results observed here could be associated with the duration of ROS exposure. Research23 shows that 16 hour ROS incubation durations are detrimental to epithelial cell viability, but only 2 hour exposure durations were used in this study. We selected a shorter duration to simulate clinically-relevant ROS exposure to the vocal folds. Within the timeline used in this study, H2O2 ROS did not induce any significant functional or structural damage to vocal fold epithelial tissue. Whether 2 hour exposures would yield different results in vivo is not clear from the current study. In vivo, the presence of antioxidants in surface fluid bathing the vocal folds may help combat oxidative stress. The antioxidant glutathione has been found in airway surface fluid24 and is likely also present in the surface fluid bathing the vocal folds.
The results of the current study are the first to demonstrate that healthy, native vocal fold epithelia can withstand acute, high concentration ROS. Previous studies4, 5 have demonstrated increased sensitivity to ROS in rodent larynges that are already inflamed secondary to acid-peptic injury. The interaction between preexisting inflammation and epithelial vulnerability to ROS and oxidative stress is an area awaiting further in vitro and in vivo investigation. While stratified squamous epithelia may provide a robust barrier to acute ROS, future studies will also examine the role of antioxidant enzymes within the vocal folds, in defending against environmental and systemic challenges.
CONCLUSIONS
Vocal fold epithelia were exposed to H2O2 for 2 hours. H2O2 is a common reactive oxygen species (ROS). Exposure to H2O2 did not significantly alter the transepithelial resistance, ZO-1 protein concentrations, or the gross structural appearance of epithelial tissue, suggesting that healthy, stratified squamous epithelium may effectively withstand a 2 hour ROS challenge. These data provide basic understanding of the functional and structural consequences of 2 hour ROS challenge to vocal fold epithelia and lay the groundwork for further investigations with longer exposure durations.
ACKNOWLEDGEMENTS
We thank the Purdue University Histopathology Service Laboratory for assistance with tissue preparation for light microscopy. We also gratefully acknowledge the contributions of Dr. Paul Snyder with histology scoring.
Funding: National Institute on Deafness and other Communication Disorders (NIH), Grant #008690 and National Institute of Environmental Health Sciences (NIH), Grant#008146.
Footnotes
Experiments on excised larynges were completed per regulations at Purdue University
Presentation: Annual meeting of the American Laryngologic Association, April 27th–28th, 2011, Chicago, IL.
Conflict of Interest: None
REFERENCES
- 1.Sataloff R. Pollution and its effect on the voice. In: Sataloff R, editor. Professional Voice: The Science and Art of Clinical Care. 2nd ed. San Diego, CA: Singular; 1997. pp. 387–391. [Google Scholar]
- 2.Huang M, Lin W, Ma Y. A study of reactive oxygen species in mainstream of cigarette. Indoor Air. 2005;15:135–140. doi: 10.1111/j.1600-0668.2005.00330.x. [DOI] [PubMed] [Google Scholar]
- 3.Leonard R, Charpied G, Faddis B. Effects of chronic ozone (O3) exposure on vocal-fold mucosa in bonnet monkeys. J Voice. 1995;9:443–448. doi: 10.1016/s0892-1997(05)80208-5. [DOI] [PubMed] [Google Scholar]
- 4.Tsai T, Chang S, Ho C, Kou Y. Neural and hydroxyl radical mechanisms underlying laryngeal airway hyperreactivity induced by laryngeal acid-pepsin insult in anesthetized rats. J Appl Physiol. 2006;101:328–338. doi: 10.1152/japplphysiol.00064.2006. [DOI] [PubMed] [Google Scholar]
- 5.Tsai T, Chang S, Hi C, Kou Y. Role of ATP in the ROS-mediated laryngeal airway hyperreactivity induced by laryngeal acid-pepsin insult in anesthetized rats. J Appl Physiol. 2009;106:1584–1592. doi: 10.1152/japplphysiol.91517.2008. [DOI] [PubMed] [Google Scholar]
- 6.MacNee W. Oxidative stress and lung inflammation in airways disease. Eur J Pharmacol. 2001;429:195–207. doi: 10.1016/s0014-2999(01)01320-6. [DOI] [PubMed] [Google Scholar]
- 7.Rao R, Baker R, Baker S, Gupta A, Holycross M. Oxidant-induced disruption of intestinal epithelial barrier function: Role of protein tyrosine phosphorylation. Am J Physiol. 1997;36:812–823. doi: 10.1152/ajpgi.1997.273.4.G812. [DOI] [PubMed] [Google Scholar]
- 8.Hashimoto K, Oshima T, Tomita T, et al. Oxidative stress induces gastric epithelial permeability through claudin-3. Biochem Biophys Res Commun. 2008;376:154–157. doi: 10.1016/j.bbrc.2008.08.140. [DOI] [PubMed] [Google Scholar]
- 9.Chapman K, Waters C, Miller W. Continuous exposure of airway epithelial cells to hydrogen peroxide: Protection by KGF. J Cell Physiol. 2002;192:71–80. doi: 10.1002/jcp.10115. [DOI] [PubMed] [Google Scholar]
- 10.Meyer T, Schwesinger C, Ye J, Denker B, Nigam S. Reassembly of the tight junction after oxidative stress depends on tyrosine kinase activity. J Biol Chem. 2001;276:22048–22055. doi: 10.1074/jbc.M011477200. [DOI] [PubMed] [Google Scholar]
- 11.Gray S. Cellular physiology of the vocal folds. Otolaryngol Clin North Am. 2000;33:679–697. doi: 10.1016/s0030-6665(05)70237-1. [DOI] [PubMed] [Google Scholar]
- 12.Jeppson A, Sundler F, Luts A, Waldeck B, Widmark E. Hydrogen peroxide-induced epithelial damage increases terbutaline transport in guinea pig tracheal wall: Implications for drug delivery. Pulmon Pharmac. 1991;4:73–79. doi: 10.1016/0952-0600(91)90055-8. [DOI] [PubMed] [Google Scholar]
- 13.Citi S. The cytoplasmic plaque proteins of the tight junction. In: Cereijido M, Anderson J, editors. Tight Junctions. Boca Raton, FL: CRC Press; 2001. pp. 231–264. [Google Scholar]
- 14.Gill G, Buda A, Moorghen M, Dettmar P, Pignatelli M. Characterisation of adherens and tight junctional molecules in normal animal larynx: Determining a suitable model for studying molecular abnormalities in human laryngopharyngeal reflux. J Clin Pathol. 2005;58:1265–1270. doi: 10.1136/jcp.2004.016972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Erickson E, Sivasankar M. Simulated reflux decreases vocal fold epithelial barrier resistance. Laryngoscope. 2010;120:1569–1575. doi: 10.1002/lary.20983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sivasankar M, Blazer-Yost B. Effects of long-acting beta adrenergic agonists on vocal fold ion transport. Laryngoscope. 2009;119:602–607. doi: 10.1002/lary.20091. [DOI] [PubMed] [Google Scholar]
- 17.Sivasankar M, Erickson E, Rosenblat M, Branski R. Hypertonic challenge to the vocal folds: Effects on barrier function. Otolaryng Head Neck. 2010;142:79–84. doi: 10.1016/j.otohns.2009.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sivasankar M, Nofziger C, Blazer-Yost B. cAMP regulation of ion transport in porcine vocal fold mucosa. Laryngoscope. 2008;118:1511–1517. doi: 10.1097/MLG.0b013e3181772d63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim K, Suh D. Asymmetric effects of H2O2 on alveolar epithelial barrier properties. Am J Physiology Lung Cell Mol Physiol. 1993;264:L308–L315. doi: 10.1152/ajplung.1993.264.3.L308. [DOI] [PubMed] [Google Scholar]
- 20.Shi L, Zheng W. Early lead exposure increases the leakage of the blood-cerebrospinal fluid barrier, in vitro. Human Exp Toxicol. 2007;26:159–167. doi: 10.1177/0960327107070560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bailey T, Kanuga N, Romero I, Greenwood J, Luthert P, Cheetham M. Oxidative stress affects the junctional integrity of retinal pigment epithelial cells. Invest Opthal Vis Sc. 2004;45:675–684. doi: 10.1167/iovs.03-0351. [DOI] [PubMed] [Google Scholar]
- 22.Lee H, Namkoong K, Kim D, et al. Hydrogen peroxide-induced alterations of tight junction proteins in bovine brain microvascular endothelial cells. Microvasc Res. 2004;2004:231–238. doi: 10.1016/j.mvr.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 23.Ohguro N, Fukuda M, Sasabe T, Tano Y. Concentration dependant effects of hydrogen peroxide on lens epithelial cells. Br J Ophthalmol. 1999;83:1064–1068. doi: 10.1136/bjo.83.9.1064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cantin A, North S, Hubbard R, Crystal R. Normal alveolar epithelial lining fluid contains high levels of glutathione. J Appl Physiol. 1987;63:152–157. doi: 10.1152/jappl.1987.63.1.152. [DOI] [PubMed] [Google Scholar]



