Abstract
Background
Stroke is becoming more common in Germany as the population ages. Its long-term sequelae can be alleviated by early reperfusion in stroke units and by complication management and functional restoration in early-rehabilitation and rehabilitation centers.
Methods
Selective review of the literature.
Results
Successful rehabilitation depends on systematic treatment by an interdisciplinary team of experienced specialists. In the area of functional restoration, there has been major progress in our understanding of the physiology of learning, relearning, training, and neuroenhancement. There have also been advances in supportive pharmacotherapy and robot technology.
Conclusion
Well-organized acute and intermediate rehabilitation after stroke can provide patients with the best functional results attainable on the basis of our current scientific understanding. Further experimental and clinical studies will be needed to expand our knowledge and improve the efficacy of rehabilitation.
Each year in Germany, 200 000 persons sustain their first stroke, and another 60 000 sustain a stroke after one or more prior strokes; roughly one citizen in five will have a stroke at some time in his or her life (1, 4). About 80% of strokes are ischemic, and 20% are hemorrhagic (2). More than a quarter of stroke patients are under age 65 (3).
Better preventive care is needed for the vascular diseases underlying stroke, including the detection of risk factors (hypertension, smoking, lack of exercise, overweight, and other risk factors), treatment with appropriate medications, and help with the necessary accompanying changes in lifestyle. Acute stroke treatment in Germany, on the other hand, is relatively better developed.
Initial care in stroke units
At present, 45% to 50% of all patients with acute stroke in Germany are treated in stroke units in which treatments that establish reperfusion can be provided and acute complications managed (4). Ideally, the stroke unit infrastructure enables the rapid reopening of occluded cerebral vessels, with immediate restoration of brain function. Because of the accompanying circumstances, however—in particular, the latency from the onset of symptoms to the initial examination by a neurologist—only 7% to 10% of stroke patients currently undergo thrombolysis (4).
More than half of the patients admitted to a stroke unit with an acute stroke can be discharged directly home after treatment. Fewer than 5% of them die (4). About 5% are highly unlikely to recover from their stroke because of its severity, because they are very old or severely demented, or because of accompanying illnesses, such as cardiac or renal failure; for such patients, only nursing care and palliative measures are indicated (5). More than 25% of patients are markedly functionally disabled when they are ready to be discharged from acute care and therefore stand to benefit from early rehabilitation and rehabilitation (4).
Determinants of functional recovery
The site, extent, and nature of the stroke (ischemic vs. hemorrhagic) are the main determinants of functional recovery. Recovery is hindered above all by the involvement of major white-matter tracts and by damage or disconnection of the hippocampus, a structure that plays a key role in the learning and relearning of neurological functions (6).
In general, neurological function recovers less well in the elderly, though many very old persons recover surprisingly well from a stroke. The decisive factor here is the brain’s reserve capacity, i.e., the absence of pre-existing damage in the form of subclinical vascular lesions. Accordingly, persons who had led an active lifestyle till just before the stroke (7) and those with no more than a minor degree of leukoaraiosis (pre-existing white-matter damage) have been found to recover particularly well from stroke. In fact, the beneficial effect of not having leukoaraiosis is of comparable magnitude to that of thrombolysis. Thus, pre-existing brain damage is more important than age as a determinant of the chance of recovery (8).
Available types of rehabilitation
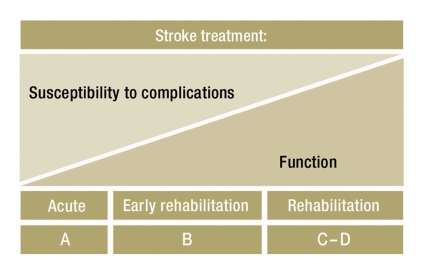
Another relevant factor for the success of rehabilitation after stroke is the available infrastructure for rehabilitative care. In Germany, a stepwise (phased) model of rehabilitation for stroke patients prevails, as recommended by the Nationwide Rehabilitation Task Force (Bundesarbeitsgemeinschaft für Rehabilitation); in this model, the available broad spectrum of rehabilitative measures can be exploited to a greater or lesser extent, partially or sequentially, depending on the patient’s individual needs (Figure 1). Emergency care in a stroke unit is called Phase A, while early neurological rehabilitation constitutes Phase B of treatment and rehabilitation, characterized by a still high demand for medical treatment (sometimes including intensive care). In Phase C of post-stroke rehabilitation, patients can actively participate in their therapy but still need medical treatment and nursing assistance. Phase D is the phase right after early mobilization; strictly speaking, this phase corresponds to the idea of rehabilitation in the narrower definition of post hospital curative treatment. Phase E consists of occupational reintegration, Phase F of continuing measures to support, maintain, or improve function.
Figure 1.
Simplified diagram of the time course of susceptibility to complications and of functional recovery during rehabilitation after stroke. The phases of rehabilitation (A–D) are indicated below.
In the early phase after stroke, the patient’s prognosis is determined mainly by potential complications due to the disturbance of elementary brain functions (9). Thanks to modern stroke unit care and neurological intensive-care medicine, 20% more patients survive their strokes today than 20 years ago, but those who survive are also more severely affected early on than their historical counterparts (10). These patients’ outcome depends nearly entirely on the prevention and management of complications, notably (Table):
Table. Complication rates of strokes that require early rehabilitation (based on [13]).
| Complication | |
| Overall | 60–90% |
| Impaired regulation of breathing | 20–60% |
| Dysphagia | 35–70% |
| Aspiration pneumonia | 10–20% |
| Urinary tract infection | 10–30% |
| Pain | 15–40% |
| Depression | 15–25% |
| Recurrent stroke | 5–30% |
| Epileptic seizures | 10% |
| Myocardial infarction | 2–6% |
| Congestive heart failure | 3–10% |
| Cardiac arrest / arrhythmia | 2–8% |
| Gastrointestinal hemorrhage | 3–5% |
| Deep vein thrombosis (lower limb) | 2–4% |
| Decubitus ulcer | 1–4% |
| Pulmonary embolism | 1% |
impaired control of breathing
dysphagia
aspiration pneumonia
recurrent stroke
urinary tract infection
sugar and electrolyte disturbances
cardiac arrhythmia
thrombosis.
The early Scandinavian experience revealed that stroke patients are more likely to make a good recovery if they are hospitalized in a stroke unit rather than on a general medical ward, even if they are not treated with thrombolysis (11). The specialization and focusing of medical teams has markedly lowered the mortality and morbidity of acute stroke (9, 12). More empirical data are needed before we can say whether the same holds true for early neurological rehabilitation.
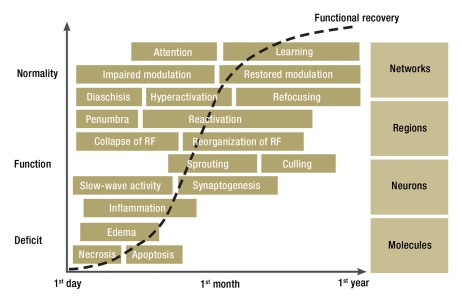
Functional recovery is based on the restitution of brain tissue and on the relearning of, and compensation for, lost functions. Brain tissue restitution involves an interlinked cascade of biological processes (Figure 2) (14, 15). It has not yet been directly observed in man; we thus do not yet know how variable it is from one patient to the next, to what extent it can be influenced by current modes of treatment, and whether it might be promoted by specific drug therapy with inflammatory modulators, growth factors, or other agents. The spontaneous restitution of brain tissue takes time and is the underlying mechanism for the recovery of wakefulness, attention, swallowing, and mobility that is often observed over weeks after a stroke.
Figure 2.
Phases and levels of brain tissue restoration and functional restitution after stroke (RF = receptive fields)
Lost functions can be compensated for or relearned after stroke because of the complexity and plasticity of the human brain. The complexity of the brain, with over one trillion specified synaptic nerve connections, is genetically determined only in its coarse structure (16). At a finer level, synapses are specified by a process of plastic adaptation that depends on interaction with the environment. Thus, for example, the part of the brain that normally becomes the visual cortex can grow to subserve language or tactile perception if visual input is lacking during development because of congenital blindness (17). While complexity enables functional compensation, the inherent adaptive plasticity of the system enables functional reorganization. Relearning and compensation are intertwined. Functional recovery benefits from stepwise training, beginning with simple and supported functions and moving onward to complex and increasingly automatic sensorimotor interactions. Large-scale randomized and controlled trials of specific modes of therapy are hard to conduct, both because of the heterogeneity of functional disturbances after stroke (site and extent of lesions, degree of pre-existing brain damage, and severity of neurological deficit) and because of the difficulty of recruiting patients; thus, no more than a few such trials have been carried out to date. On the other hand, small-scale controlled trials can point to the basic principles underlying functional recovery. Such trials have shown that the main factors affecting outcome are individual adaptation of therapy and the intensity and frequency of training (18).
The recovery of motor function
The rehabilitation of walking
In our experience, various formalized physiotherapeutic methods, such as proprioceptive neuromuscular facilitation or the methods of Bobath and Vojta, are all about equally effective (19). All are based on the transfer of learned performance from one motor task to another. The Bobath method, for example, involves intensive preparatory training for walking in the sitting and standing positions. In the rehabilitation of stance and gait, a task-specific repetitive approach is increasingly being used in addition to conventional therapeutic approaches: i.e., the motor task to be learned must be practiced by repeating it as many times as possible.
We recommend organizing the temporal course of rehabilitation in three transitional phases with different goals, corresponding to the patient’s deficit at the time of each phase:
the bedridden patient is mobilized out of bed;
the patient, having been mobilized into a wheelchair, learns to walk again;
the patient, having regained the ability to walk, learns to do so rapidly and steadily, also under the prevailing conditions of everyday life.
Very early mobilization of stroke patients has been shown to lead to significantly better functional recovery. Thus, one should try to reach the concisely stated goal “Out of bed!” within two days, as long as the patient is hemodynamically stable (20). Mobilization over the edge of the bed into a wheelchair, with repeated practicing of transfers, should be supplemented by training in how to drive a wheelchair and how to stand up at a standing desk, in order to prevent functional impairment through contraction of the hip and knee flexors. Further benefits of verticalization are:
circulatory training,
prevention of pneumonia and venous thrombosis,
stimulation of the autonomic nervous system,
sensory activation through standing.
Once the patient, having been mobilized out of the wheelchair, can sit at the edge of the bed and can tolerate verticalization for at least ten minutes, the training of ambulation can begin.
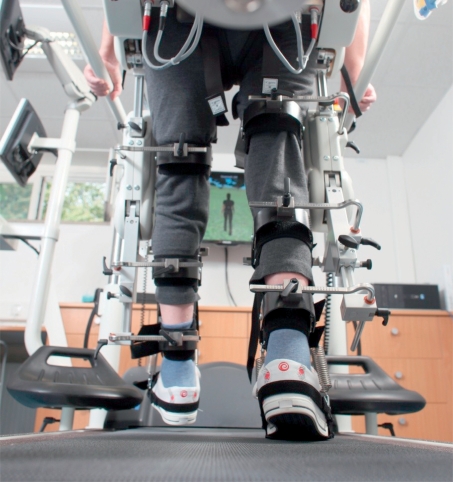
The number of footsteps practiced per training session appears to be essential, whichever particular method is used. The physical work needed to practice a large number of steps is, however, difficult for physiotherapists to carry out unaided, and thus gait-training machines are recommended for this purpose (Figure 3). The machine enables the patient, wearing a safety belt, to practice walking over and over again. It does not replace the physiotherapist, but the combination of the machine and the physiotherapist is more effective than the latter alone and can prevent one-quarter of all cases of inability to walk that would otherwise ensue (21).
Figure 3.
Robot-assisted gait training in a virtual environment. The system enables early verticalization of the patient and an early start of gait training, with a continuous transition from mainly robot-initiated to increasingly independent movement. (Photograph by Roland Magunia)
Severe arthrosis in the lower limbs, congestive heart failure, or a markedly abnormal spastic posture of the joints requires either specific treatment or else reduction of the intensity of physiotherapy. If the patient suffers from spastic pes equinus, with a twisted food and claw toes, timely treatment with a splint or intramuscular injection of botulinum toxin is recommended. Specialized centers offer the further option of surgical myotendolysis, which enables wider opening of a joint once contractures have formed. If the patient can walk a short distance independently, using a mechanical aid, then the steadiness, speed, and maximal duration of gait can all be improved. These are the important characteristics of gait for everyday life.
The rehabilitation of arm and hand function
Some 80% of stroke patients suffer from upper limb paresis. Those who are less severely affected can begin to move their fingers again in the first four to six weeks after the stroke and rapidly regain the ability to extend the wrist and fingers actively. This is a strong predictor of the restoration of hand function (22). Physiotherapists try to train the patient’s returning arm and hand function with repetitive practice, paying special attention to strength, coordination, and speed, and to integrate hand function into the patient’s everyday activities (23). Learned non-use of the limb (because the other hand is much faster and more accurate) should be watched for. If it should arise, so-called constrained induced movement therapy is a treatment option for the chronic phase that has been found effective in multiple scientific studies. It involves binding the unaffected hand and intensively practicing the use of the affected one in small group sessions (24).
It is much harder to rehabilitate an upper limb that is so severely effected that the hand—in particular, the wrist and finger extensors—remain plegic four to six weeks after the stroke, so that the patient can carry out no more than synergistic movements of the shoulder and elbow. In such cases, the limb should be positioned and passively mobilized so as to prevent shoulder pain (never lift the arm by simply picking up the hand!) and spastic flexor posture. The benefit of physiotherapy for restoring arm and hand function is debated. The probability of restoring useful hand function for everyday life six months after the stroke is less than 5% (25). If the decision is made to pursue physiotherapy, it should be begun early. If weakness is so severe that active therapy is not possible, then passive or assistive therapy should be provided by an experienced therapist, with the use of facilitation techniques. Isolated movements are repetitively practiced. Alternatively, robots and mechanical apparatus can be used (e1, e2). Two types of therapy are currently established for use in the chronic phase:
constrained induced movement therapy, described above, for patients who can voluntarily extend their wrist and finger joints;
botulinum toxin injections to treat spastic flexion, with the aim of reducing muscle tone, facilitating hand care, and alleviating pain.
The recovery of cognitive function
Neurocognitive functional recovery involves consciousness, attention, language, memory, and planning; these are complex, adaptive functions with correspondingly complex and dynamic neural substrates. Here, too, the relevant factors for recovery are time and stepwise training, which we shall illustrate through the example of language training. One-third of all stroke patients suffer from aphasia, and two-thirds of these have a permanent language deficit of greater or lesser severity (e3). Five or more hours of speech therapy per week are associated with significantly better functional recovery than the spontaneous course after stroke (e4). Speech therapy can be conducted at the level of words, sentences, or conversations. Functional improvement is usually limited to the particular area practiced and can be lost if maintenance training is not kept up. If the patient’s condition allows more complex pragmatic communication situations to be trained, this may contribute, by way of improved strategies, to an indirect generalization of the effect of training.
The goal of all rehabilitative treatments is to achieve the greatest possible autonomy and, ideally, complete psychosocial reintegration for the patient. Some 40% of stroke patients who undergo rehabilitation can re-enter the working world afterward (e5). Motor deficits play only a moderate role in determining whether the patient can return to work, while cognitive deficits, particularly with respect to attention and memory, often limit the patient’s autonomy, even if the patient does not perceive this (e6). Neuromodulation, which will be discussed in the next section, can be used in such situations alongside targeted neurocognitive training.
Neuromodulation
Neuroenhancement is the use of drugs or electrical stimulation to improve wakefulness, mood, and learning ability to further enhance functional recovery after stroke. The underlying principle is that the processing of incoming stimuli by the central nervous system is normally modulated according to their significance for the organism. Significant contexts are those that are linked to attention, emotion, novelty, or reward. When incoming stimuli are monotonous, the activity level of the ascending brainstem pathways declines. Because these pathways function, in part, through the use of typical neurotransmitters such as noradrenaline and dopamine, their modulating effects on learning can be reproduced by the exogenous administration of these transmitters or their precursors. In this way, brainstem activation that is slowly declining, or that has been impaired by brain damage, can be compensated for or even driven to supranormal levels. Initial randomized clinical trials have yielded promising results (e7– e9), which, however, still need to be confirmed in further studies (e10). Multiple clinical trials of transcranial direct-current (DC) stimulation to facilitate functional recovery after stroke are now in progress (e11).
Special considerations for elderly patients
Stroke is also a classic disease of old age. The more functionally relevant comorbidities the patient has, the clearer the indication for geriatric rehabilitation. Nearly all patients aged 80 and above undergo geriatric rehabilitation.
After acute treatment for stroke, elderly patients can undergo either a complex early rehabilitative geriatric treatment in an acute hospital or else a course of geriatric rehabilitation, which, unlike specialized neurological rehabilitation for younger stroke patients, is not divided into phases. The multimodal and multidisciplinary treatment plan for the patient’s functional deficits is established on the basis of the geriatric assessment. Secondary stroke prevention, particularly through increased exercise, is not merely indicated, but actually especially effective in the elderly, who have a higher incidence of stroke than younger patients do. Nonetheless, treatment recommendations for elderly patients are still based on extrapolation from the findings of trials carried out on younger patients (e12). Relevant scientific data are particularly scarce for patients in advanced old age.
Brain plasticity basically remains intact into old age. Problems arise, however, through comorbidities such as congestive heart failure and diabetes, and through the patient’s functional limitations—physical, mental, emotional, and psychosocial. Stroke complications are more common in old age, and confer a worse prognosis (Box). Precise data are unavailable, as these effects depend both on the severity of the stroke and on the extent of pre-existing damage.
Box. Common complications of stroke in elderly patients*.
Falls (particularly common after discharge from acute hospital care)
Incontinence (urinary, fecal)
Delirium / development of dementia
Depression / anxiety disorders
Dysphagia / aspiration pneumonia
Pulmonary embolism / thrombosis
Psychosocial problems
Drug side effects and interactions
*that are observed in clinical practice
Because the structure of ambulatory care has improved in recent years, early hospital discharge programs for geriatric stroke patients are now being tested (“hospital at home,” outpatient geriatric rehabilitation).
The prevention of falls in elderly patients often receives too little attention. Most falls, and most fractures, occur after the patient has been discharged home. This fact underscores the importance of training not just strength and endurance, but also balance (e13). Osteoporosis prophylaxis with calcium and vitamin D supplementation is often given as further supportive therapy.
Future prospects
In Germany, rehabilitation (including early rehabilitation) after stroke are now mostly performed in private institutions that are tightly organized and that frequently carry out assessments of the care that they provide, as they must do to remain competitive. In this way, the quality of neurological and geriatric early rehabilitation and rehabilitation after stroke can steadily improve, but only within the framework of established knowledge. Acute stroke care in Germany has been markedly improved through the combined influence of quality management and scientific studies, both experimental and clinical/interventional. The same might be achievable for neurological rehabilitation (including early rehabilitation) after stroke. So far, however, the opportunities in this area have not yet been adequately explored. This is so both for structural reasons—narrow scientific infrastructure, strong economic orientation—and for reasons of content: a greater variety of factors can affect the outcome of stroke rehabilitation than can affect the early outcome of acute stroke.
Key Messages.
In Germany, one stroke patient in four undergoes early rehabilitation or rehabilitation.
The goals of rehabilitation, including early rehabilitation, are complication management and restitution of function.
The pillars of functional restitution are early activation and intense practice.
Further supportive techniques that may be used include training robots and neuromodulation.
Quality management enables the optimal implentation of already established knowledge, but post-stroke rehabilitation now needs to be made more scientific so that our knowledge in this area can be extended.
Acknowledgments
Translated from the original German by Ethan Taub, M.D.
Footnotes
Conflict of interest statement
Prof. Hesse is a partner in Reha-Technologies GmbH (Bolzano, Italy), a company that manufactures and distributes robots and other devices for use in rehabilitation after stroke. He also leads courses in the use of botulinum toxin A for the treatment of spasticity after stroke on behalf of the Merz Pharmaceuticals company.
The other authors state that they have no conflict of interest.
References
- 1.Seshadri S, Beiser A, Kelly-Hayes M, et al. The Lifetime risk of Stroke: Estimates From the Framingham Study. Stroke. 2006;37:345–350. doi: 10.1161/01.STR.0000199613.38911.b2. [DOI] [PubMed] [Google Scholar]
- 2.Runchey S, McGee S. Does this patient have a hemorrhagic stroke?: clinical findings distinguishing hemorrhagic stroke from ischemic stroke. JAMA. 2010;303:2280–2286. doi: 10.1001/jama.2010.754. [DOI] [PubMed] [Google Scholar]
- 3.Rothwell PM, Coull AJ, Silver LE, et al. Population-based study of event-rate, incidence, case fatality, and mortality for all acute vascular events in all arterial territories (Oxford Vascular Study) Lancet. 2005;366:1773–1783. doi: 10.1016/S0140-6736(05)67702-1. [DOI] [PubMed] [Google Scholar]
- 4.Heuschmann P, Busse O, Wagner, et al. für das Kompetenznetz Schlaganfall dDSGsdSDS-H: Schlaganfallhäufigkeit und Versorgung von Schlaganfallpatienten in Deutschland. Akt Neurol. 2010;37:333–340. [Google Scholar]
- 5.Burton CR, Payne S, Addington-Hall J, Jones A. The palliative care needs of acute stroke patients: a prospective study of hospital admissions. Age Ageing. 2010;39:554–559. doi: 10.1093/ageing/afq077. [DOI] [PubMed] [Google Scholar]
- 6.Meinzer M, Mohammadi S, Floel A, et al. Integrity of the hippocampus and surrounding white matter is correlated with language training success in aphasia. Neuroimage. 2010 doi: 10.1016/j.neuroimage.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 7.Krarup LH, Truelsen T, Gluud C, et al. Prestroke physical activity is associated with severity and long-term outcome from first-ever stroke. Neurology. 2008;71:1313–1318. doi: 10.1212/01.wnl.0000327667.48013.9f. [DOI] [PubMed] [Google Scholar]
- 8.Arsava EM, Rahman R, Rosand J, et al. Severity of leukoaraiosis correlates with clinical outcome after ischemic stroke. Neurology. 2009;72:1403–1410. doi: 10.1212/WNL.0b013e3181a18823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stroke Unit trialists collaboration Organised inpatient (stroke unit) care for stroke. Cochrane Database Syst Rev. 2007. CD000197. [DOI] [PubMed] [Google Scholar]
- 10.Suarez JI. Outcome in neurocritical care: advances in monitoring and treatment and effect of a specialized neurocritical care team. Crit Care Med. 2006;34:232–238. doi: 10.1097/01.CCM.0000231881.29040.25. [DOI] [PubMed] [Google Scholar]
- 11.Langhorne P, Pollock A. What are the components of effective stroke unit care? Age Ageing. 2002;31:365–371. doi: 10.1093/ageing/31.5.365. [DOI] [PubMed] [Google Scholar]
- 12.Langhorne P, Lewsey JD, Jhund PS, et al. Estimating the impact of stroke unit care in a whole population: an epidemiological study using routine data. J Neurol Neurosurg Psychiatry. 2010 doi: 10.1136/jnnp.2009.195131. [DOI] [PubMed] [Google Scholar]
- 13.Kumar S, Selim MH, Caplan LR. Medical complications after stroke. Lancet Neurol. 2010;9:105–118. doi: 10.1016/S1474-4422(09)70266-2. [DOI] [PubMed] [Google Scholar]
- 14.Cramer SC, Riley JD. Neuroplasticity and brain repair after stroke. Curr Opin Neurol. 2008;21:76–82. doi: 10.1097/WCO.0b013e3282f36cb6. [DOI] [PubMed] [Google Scholar]
- 15.Carmichael ST. Themes and Strategies for Studying the Biology of Stroke Recovery in the Poststroke Epoch. Stroke. 2008;39:1380–1388. doi: 10.1161/STROKEAHA.107.499962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Knecht S, Henningsen H, Elbert T, Flor H, Höhling C, Pantev C, Taub E. Reorganization and perceptual changes after amputation. Brain. 1996;119:1213–1219. doi: 10.1093/brain/119.4.1213. [DOI] [PubMed] [Google Scholar]
- 17.Amedi A, Floel A, Knecht S, Zohary E, Cohen LG. Transcranial magnetic stimulation of the occipital pole interferes with verbal processing in blind subjects. Nat Neurosci. 2004;7:1266–1270. doi: 10.1038/nn1328. [DOI] [PubMed] [Google Scholar]
- 18.Langhorne P, Coupar F, Pollock A. Motor recovery after stroke: a systematic review. Lancet Neurol. 2009;8:741–754. doi: 10.1016/S1474-4422(09)70150-4. [DOI] [PubMed] [Google Scholar]
- 19.Hummelsheim H, Mauritz KH. The neurophysiological basis of exercise physical therapy in patients with central hemiparesis. Fortschr Neurol Psychiatr. 1993;61:208–216. doi: 10.1055/s-2007-999089. [DOI] [PubMed] [Google Scholar]
- 20.Cumming TB, Thrift AG, Collier JM, et al. Very early mobilization after stroke fast-tracks return to walking: further results from the phase II AVERT randomized controlled trial. Stroke. 2011;42:153–158. doi: 10.1161/STROKEAHA.110.594598. [DOI] [PubMed] [Google Scholar]
- 21.Mehrholz J, Werner C, Kugler J, Pohl M. Electromechanical-assisted training for walking after stroke. Cochrane Database Syst Rev. 2007 doi: 10.1002/14651858.CD006185.pub2. CD006185. [DOI] [PubMed] [Google Scholar]
- 22.Smania N, Paolucci S, Tinazzi M, et al. Active finger extension: a simple movement predicting recovery of arm function in patients with acute stroke. Stroke. 2007;38:1088–1090. doi: 10.1161/01.STR.0000258077.88064.a3. [DOI] [PubMed] [Google Scholar]
- 23.Platz T. Evidence-based arm rehabilitation—a systematic review of the literature. Nervenarzt. 2003;74:841–849. doi: 10.1007/s00115-003-1549-7. [DOI] [PubMed] [Google Scholar]
- 24.Wolf SL, Winstein CJ, Miller JP, et al. Effect of constraint-induced movement therapy on upper extremity function 3 to 9 months after stroke: the EXCITE randomized clinical trial. JAMA. 2006;296:2095–2104. doi: 10.1001/jama.296.17.2095. [DOI] [PubMed] [Google Scholar]
- 25.Kwakkel G, Kollen BJ, van der Grond J, Prevo AJ. Probability of regaining dexterity in the flaccid upper limb: impact of severity of paresis and time since onset in acute stroke. Stroke. 2003;34:2181–2186. doi: 10.1161/01.STR.0000087172.16305.CD. [DOI] [PubMed] [Google Scholar]
- e1.Lo AC, Guarino PD, Richards LG, et al. Robot-assisted therapy for long-term upper-limb impairment after stroke. N Engl J Med. 2010;362:1772–1783. doi: 10.1056/NEJMoa0911341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e2.Buschfort R, Brocke J, Hess A, Werner C, Waldner A, Hesse S. Arm studio to intensify the upper limb rehabilitation after stroke: concept, acceptance, utilization and preliminary clinical results. J Rehabil Med. 2010;42:310–314. doi: 10.2340/16501977-0517. [DOI] [PubMed] [Google Scholar]
- e3.Ferro JM, Mariano G, Madureira S. Recovery from aphasia and neglect. Cerebrovasc Dis. 1999;9(Suppl 5):6–22. doi: 10.1159/000047571. [DOI] [PubMed] [Google Scholar]
- e4.Bhogal SK, Teasell R, Speechley M. Intensity of aphasia therapy, impact on recovery. Stroke. 2003;34:987–993. doi: 10.1161/01.STR.0000062343.64383.D0. [DOI] [PubMed] [Google Scholar]
- e5.Vestling M, Tufvesson B, Iwarsson S. Indicators for return to work after stroke and the importance of work for subjective well-being and life satisfaction. J Rehabil Med. 2003;35:127–131. doi: 10.1080/16501970310010475. [DOI] [PubMed] [Google Scholar]
- e6.Krause M, Polnitzky-Meissner P, Helbig P, et al. Rehabilitation treatment after stroke. An assessment of current status. Nervenarzt. 1999;70:322–329. doi: 10.1007/s001150050443. [DOI] [PubMed] [Google Scholar]
- e7.Walker-Batson D, Curtis S, Natarajan R, et al. A double-blind, placebo-controlled study of the use of amphetamine in the treatment of aphasia. Stroke. 2001;32:2093–2098. doi: 10.1161/hs0901.095720. [DOI] [PubMed] [Google Scholar]
- e8.Scheidtmann K, Fries W, Muller F, Koenig E. Effect of levodopa in combination with physiotherapy on functional motor recovery after stroke: a prospective, randomised, double-blind study. Lancet. 2001;358:787–790. doi: 10.1016/S0140-6736(01)05966-9. [DOI] [PubMed] [Google Scholar]
- e9.Knecht S. Optionen der medikamentösen Behandlung kognitiver Störungen. In: Karnath H-O, Hartje W, Ziegler W, editors. Kognitive Neurologie. Stuttgart: Georg Thieme Verlag; 2006. pp. 230–234. [Google Scholar]
- e10.Clinical Trials levodopa. 2011. Dopaminergic enhancement of learning and memory in aphasia. [Google Scholar]
- e11.Clinical Trials listing tDCS. 2011. [Google Scholar]
- e12.Bachmann S, Finger C, Huss A, Egger M, Stuck AE, Clough-Gorr KM. Inpatient rehabilitation specifically designed for geriatric patients: systematic review and meta-analysis of randomised controlled trials. BMJ. 2010;340 doi: 10.1136/bmj.c1718. c1718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e13.Batchelor F, Hill K, Mackintosh S, Said C. What works in falls prevention after stroke?: a systematic review and meta-analysis. Stroke. 2010;41:1715–1722. doi: 10.1161/STROKEAHA.109.570390. [DOI] [PubMed] [Google Scholar]