Abstract
Background
Comprehensive lifestyle interventions are effective in preventing diabetes and restoring glucose regulation; however, the key stimulus for change has not been identified and effects in older individuals are not established. The aim of the study was to investigate the independent and combined effects of dietary weight-loss and exercise on insulin sensitivity and restoration of normal fasting glucose in mid-aged and older women.
Design
Four-arm RCT, conducted between 2005 and 2009 and data analyzed in 2010.
Setting/participants
439 inactive, overweight/obese postmenopausal women. Interventions: Women were assigned to: dietary weight loss (n=118), exercise (n=117), exercise+diet (n=117), or control (n=87). The diet intervention was a group-based reduced-calorie program with a 10% weight-loss goal. The exercise intervention was 45 min/day, 5 days/week of moderate-to-vigorous intensity aerobic activity.
Main outcome measures
12-month change in serum insulin, C-peptide, fasting glucose, and whole body insulin resistance (HOMA-IR).
Results
A significant improvement in HOMA-IR was detected in the diet (−24%, p<0.001) and exercise+ diet (−26%, p<0.001) groups, but not in the exercise (−9%, p=0.22) group compared to controls (−2%); these effects were similar in middle-aged (50–60 years) and older women (aged 60–75 years). Among those with impaired fasting glucose (5.6–6.9 mmol/L) at baseline (n=143; 33%), the odds (95% CI) of regressing to normal fasting glucose after adjusting for weight loss and baseline levels were: 2.5 (0.8, 8.4), 2.76 (0.8, 10.0), and 3.1 (1.0, 9.9) in the diet, exercise+diet, and exercise group, respectively, compared to controls.
Conclusions
Dietary weight loss, with or without exercise, significantly improved insulin resistance. Older women derived as much benefit as did the younger postmenopausal women.
Introduction
Given the high prevalence of insulin resistance1 and its associated health burden,2–7 effective prevention strategies for high-risk individuals are a priority. Several trials have examined the efficacy of lifestyle interventions for preventing type 2 diabetes, but few have examined their effectiveness on restoring normal glucose homeostasis.
The Diabetes Prevention Program (DPP) demonstrated that combined diet and exercise therapy is effective for diabetes prevention,8 and restores normal glucose regulation more frequently than placebo.9 Moreover, the effect of lifestyle therapy was more pronounced in older compared with younger participants.10 Yet, because the DPP did not randomly assign each lifestyle component, the relative importance of the diet and exercise components could not be adequately examined. Furthermore, the DPP lifestyle intervention was resource-intensive, providing ongoing, individualized counseling.8 To our knowledge, the effectiveness of group-based adaptations of the DPP program has not been widely tested.
Since the restoration of normal glucose homeostasis is more akin to true prevention than delayed progression to overt diabetes, unraveling the relative importance of diet, exercise, and body weight in this regard has important implications for resource allocation, patient counseling, and public health.
While both obesity and diabetes risk increases with age in the U.S.,11,12 few studies have included older individuals in large enough numbers to determine whether lifestyle changes to reduce weight can improve insulin resistance, a major predictor of diabetes risk.13 Thus, the purpose of this study was to examine the independent and combined effects of 12 months of dietary weight-loss and/or aerobic exercise on insulin resistance and the restoration of normal fasting glucose among overweight postmenopausal women, and to examine whether the magnitude of these effects differed between middle-aged (50–60 years) and older women (aged 60–75 years).
The study also examined which factors, including weight loss, fat loss, or changes in cardiorespiratory fitness or physical activity, were most associated with changes in glucose homeostasis. It was hypothesized that insulin and glucose would improve in all intervention groups compared to controls, and that these changes would be mediated by the degree of weight loss. It was also hypothesized that younger women would experience greater metabolic improvements relative to older women.
Methods
Design Overview
The Nutrition and Exercise in Women (NEW) study was a 12-month RCT testing the effects of exercise and/or dietary weight loss on circulating hormones and other outcomes.14 The trial was conducted from 2005 to 2009; data were analyzed in 2010. Study procedures were reviewed and approved by the Fred Hutchinson Cancer Research Center IRB in Seattle, WA. All participants provided informed consent.
Setting and Participants
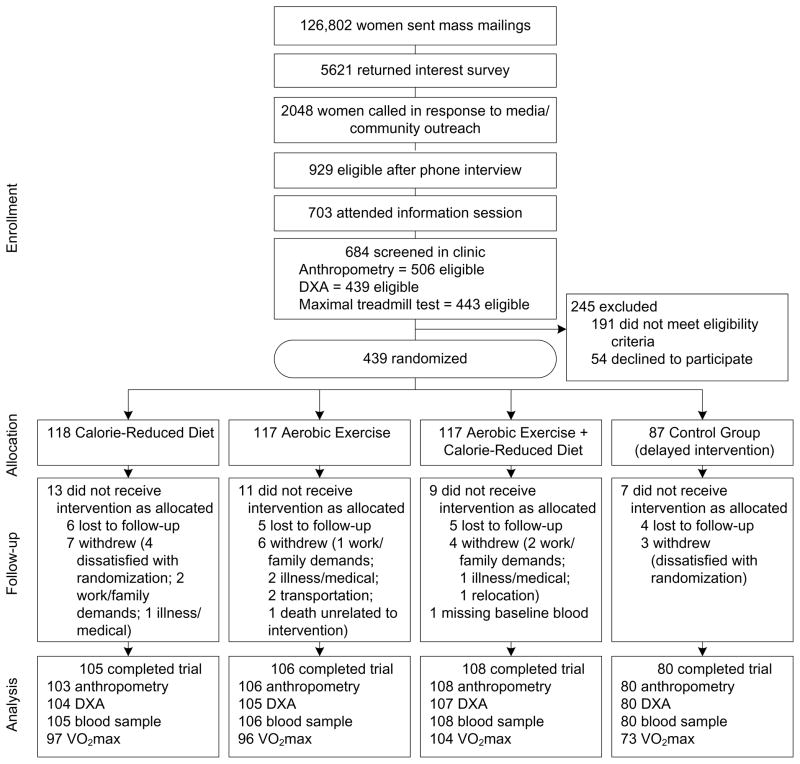
Participants were overweight or obese (BMI ≥25.0; ≥23.0 if Asian-American), postmenopausal women (aged 50–75 years) from the greater-Seattle area who were not meeting physical activity guidelines.15 Women were recruited through media and mass mailings (Figure 1). Specific exclusion criteria included: diagnosed diabetes, fasting blood glucose ≥126 mg/dL or use of diabetes medications; use of postmenopausal hormones; history of other serious medical condition(s); alcohol intake >2 drinks/day; current smoking; contraindication to the study interventions for any reason (e.g., abnormal exercise tolerance test); participation in another structured weight loss program; or use of weight loss medications.
Figure 1.
Flow of participants through the Nutrition and Exercise in Women (NEW) trial
Note: DXA, dual emission X-ray absorptiometry
Randomization and Interventions
Eligible women were randomized to: (1) dietary weight loss (n=118); (2) moderate-to-vigorous intensity aerobic exercise (n=117); (3) combined diet and exercise (n=117); or (4) control (no intervention; n=87). Computerized random assignment was stratified according to BMI (≥ or <30 kg/m2) and participants’ self-reported race/ethnicity (black, white, other). A permuted blocks randomization, wherein the control assignment was randomly eliminated from each block with a probability of approximately 1 in 4, was used to achieve a proportionally smaller control group. One participant that was randomized to diet+exercise was excluded from analysis due to missing baseline blood measures.
The exercise intervention progressed to 45 minutes of moderate-to-vigorous intensity exercise at a target heart rate of 70%–85% observed maximum, 5 days/week, by the 7th week. Participants attended three supervised sessions/week at the study facility and exercised 2 days/week at home. Participants recorded exercise mode, duration, peak heart rate, and perceived exertion at each session. Activities of ≥4 METs16 were counted toward the prescribed target.
The dietary weight-loss intervention involved a modification of the DPP8 and Look AHEAD17 lifestyle behavior change programs with goals of: 1200–2000 kcal/day, <30% calories from fat, and 10% weight loss by 6 months with maintenance thereafter. Participants met individually with a dietitian at least twice, followed by weekly group meetings (5–10 women) for 6 months. Thereafter, participants attended monthly group meetings, in addition to biweekly phone or email contact. A combination of individual and group-based approaches was used to maximize the benefits of personalized recommendations with the social support and cost effectiveness of a group setting. Women completed daily food logs for at least 6 months or until they reached their 10% weight-loss goal. Food logs were collected by the dietitian and returned with feedback. Logs, weekly weigh-ins, and session attendance were tracked to promote dietary adherence. Participants who did not meet their weight loss goal by 6 months were encouraged to continue weight loss efforts and were offered additional sessions, while women who reached their goal were allowed to continue losing but were monitored to ensure that BMI did not go below 18.5. Participants who were randomized to dietary weight loss + exercise received separate sessions and were instructed not to discuss diet during supervised exercise. The control group was instructed not to change their diet or exercise behavior for 12 months. At study completion, they were offered 4 group nutrition classes and 8 weeks of exercise training.
Outcomes and Follow-Up
All study measures were obtained and analyzed by trained personnel who were blinded to the participants’ randomization status. Demographic information, medical history, dietary patterns (via 120-item self-administered food frequency questionnaire18) were collected at baseline and 12 months. At both time points, participants wore pedometers (Accusplit, Silicon Valley, CA) for 7 consecutive days to determine an average daily step count. Cardiorespiratory fitness (VO2max) was assessed using a maximal graded treadmill test according to a modified branching protocol.19 Heart rate and oxygen uptake were continuously monitored with an automated metabolic cart (MedGraphics, St. Paul, MN).
Body mass index was calculated from weight and height, measured to the nearest 0.1 kg and 0.1cm, respectively, with a balance beam scale and stadiometer. Waist circumference was measured to the nearest 0.5cm at the minimal waist. Body composition was measured on a DXA whole-body scanner (GE Lunar, Madison, WI).
Fasting venous blood samples (50 mL) were collected during clinic visits prior to randomization and at 12 months. Participants consumed no food and drank only water for 12 hours prior and were requested not to exercise for 24 hours preceding the blood draw. Blood was processed within 1 hour and samples were stored at −70°C.
Blood samples were analyzed in batches such that each participant’s samples were assayed simultaneously, the numbers of samples from each arm were approximately equal, participant randomization dates were similar, and sample order was random. All but four samples were analyzed for insulin and C-peptide in a single batch. Excluding these samples did not affect the results.
Insulin was analyzed at the University of Washington (UW) Clinical Nutrition Research Unit Laboratory, and quantified by a 48-hour, polyethylene glycol–accelerated, double antibody radioimmunoassay. The intra-assay coefficient of variation (CV) was 4.5%. C-peptide and glucose were analyzed at the UW Northwest Lipid Research Laboratories. C-peptide was analyzed using a two-site immunoenzymometric assay (Tosoh AIA 1800 auto analyzer; Tosoh Bioscience, Inc., San Francisco). Glucose was quantified using a ClinicalChemistry Autoanalyzer with the hexokinase method. The intra-assay CV for C-peptide was 4.3%. The intra- and inter-assay CVs for glucose were 1.1% and 3.5%, respectively.
The homeostasis assessment model (HOMA-IR= fasting insulin (mU/L) × fasting glucose (mmol/L)/22.5)20 was used as a surrogate measure of whole-body insulin resistance.
Statistical Analysis
In cases of missing values, no change from baseline was assumed. Repeating the main analyses using a multiple imputation method did not meaningfully affect any of the findings. Age-adjusted partial Pearson correlation coefficients were calculated between baseline anthropometric and blood measures. Generalized linear models were used to test for differences in baseline values across study arms, and a chi-square test to detect differences in the prevalence of impaired fasting glucose.
Descriptive data are presented as M (SD). Due to their non-normal distribution, blood measures were log-transformed prior to further analysis. These data are presented as geometric means (95% CIs) unless otherwise indicated. Mean changes in insulin, C-peptide, glucose, and HOMA-IR from baseline to 12 months, stratified by group, were computed; intervention effects on these variables were examined based on the assigned treatment at randomization, regardless of adherence or study retention (i.e., intent-to-treat). Mean 12-month changes in the exercise, diet, and diet+exercise groups were compared to controls using the generalized estimating equations (GEE) modification of linear regression to account for intra-individual correlation over time. The effect of age (50–60 years vs 60–75 years) was assessed by including interaction terms in these models. Analyses were also repeated after stratification by age group. Adjustment for multiple comparisons was made using Bonferroni correction (two-sided alpha=0.05/3).
Changes in body weight, body composition, pedometer steps/day, and VO2max were similarly calculated and used to predict the observed change in metabolic variables by linear regression. These potential mediating variables were examined continuously, then categorically as: 3 clinically relevant categories of weight loss:21, 22 <5%, ≥5%–10%, and ≥10% loss; change in pedometer steps/day corresponding to a decrease/no change (≤0 steps/day), an increase up to 2 miles/day (1–3520 steps/day), or >2 miles/day (>3520 steps/day); and tertiles of change in total body fat (kg) and VO2max (L/min). Models included age, group assignment, and the baseline value of the outcome variable as covariates.
Participants’ fasting glucose levels were classified as ‘impaired’ (5.6–6.9 mmol/L) or ‘normal’ (<5.6 mmol/L).23 Logistic regression was used to calculate the ORs for regression from impaired to normal fasting glucose within each intervention arm compared to controls, adjusting for age, weight loss, and baseline blood glucose level. Corresponding ORs were calculated for categories of weight loss (1%–5%, ≥5%–10%, ≥10%, relative to no change/gain) and approximate tertiles of change in VO2max. All statistical analyses were performed using SAS software version 9.1 (SAS Institute, Cary, NC).
Results
Participants
At 12 months, 398 of 438 participants completed physical exams and provided a blood sample, 397 underwent a DXA scan, and 371 completed a treadmill test; 39 did not complete the study (Figure 1). There were no differences in relevant variables between groups at baseline, except the percentage of daily calories consumed as fat (p=0.02) (Table 1).
Table 1.
Selected baseline characteristics of randomized women
| Variable | CONTROL | EXERCISE | DIET | DIET+EXERCISE |
|---|---|---|---|---|
| M ± SD or n (%) | M ± SD or n (%) | M ± SD or n (%) | M ± SD or n (%) | |
| n | 87 | 117 | 118 | 116 |
| Age (years) | 57.4 (4.4) | 58.1 (5.0) | 58.1 (5.9) | 58.0 (4.4) |
| Ethnicity (%) | ||||
| Non-Hispanic white | 74 (85.1) | 98 (83.8) | 101 (85.6) | 99 (85.3) |
| Non-Hispanic black | 6 (6.9) | 15 (12.8) | 9 (7.6) | 5 (4.3) |
| Hispanic | 3 (3.5) | 2 (1.7) | 2 (1.7) | 5 (4.3) |
| Other (American Indian, Asian, or Unknown) | 4 (4.6) | 2 (1.7) | 6 (5.1) | 7 (6.0) |
| College graduate | 59 (67.8) | 70 (59.9) | 76 (64.4) | 81 (69.8) |
| Full-time employment | 47 (63.5) | 53 (55.2) | 58 (55.2) | 64 (62.1) |
| Married or living with partner | 59 (73.8) | 71 (60.7) | 79 (67.0) | 69 (60.0) |
| Weight (kg) | 84.2 (12.5) | 83.7 (12.3) | 84.0 (11.8) | 82.6 (10.8) |
| BMI | 30.7 (3.9) | 30.7 (3.7) | 31.1 (3.9) | 31.0 (4.3) |
| Waist circumference (cm) | 94.8 (10.2) | 95.1 (10.1) | 94.6 (10.2) | 93.7 (9.9) |
| Body fat (%) | 47.3 (4.4) | 47.3 (4.1) | 47.0 (4.3) | 47.4 (4.5) |
| Average calories consumed (kcal/day)a | 1988 (669) | 1986 (589) | 1884 (661) | 1894 (639) |
| Relative % calories from fatb | 35.6 (6.9) | 33.6 (6.9) | 33.1 (6.3) | 35.3 (7.3) |
| VO2max (ml/kg/min) | 23.1 (4.1) | 22.5 (4.1) | 22.7 (3.8) | 23.6 (4.1) |
| Pedometer steps/day (7-day average) | 5605 (2334) | 5777 (2129) | 5539 (2257) | 5952 (2354) |
| Insulin (pmol/L) | 93.20 (44.80) | 86.95 (55.84) | 91.26 (60.56) | 87.23 (59.80) |
| C-peptide (nmol/L) | 0.92 (0.31) | 0.87 (0.33) | 0.89 (0.36) | 0.86 (0.33) |
| Glucose (mmol/L) | 5.38 (0.46) | 5.32 (0.45) | 5.37 (0.48) | 5.33 (0.43) |
| HOMA-IR | 3.22 (1.65) | 2.99 (2.19) | 3.17 (2.25) | 3.01 (2.22) |
Daily kcal values derived from FFQ were truncated <600 kcal and >4000
% calories from fat= (total calories derived from fat/total daily caloric intake)
No significant differences in baseline variables between groups, except in daily calories consumed as fat (p=0.02)
Adherence to Interventions
Intervention adherence and body composition changes in this sample have been recently reported.14 Briefly, women who were randomized to exercise alone participated in moderate-to-vigorous activity for a M (SD) of 163.3 (70.6) minutes/week, while women who were randomized to diet+exercise participated for 171.5 (62.9) minutes/week. Both groups significantly increased average pedometer steps/day (+2416 and +3471 steps/d, respectively) and VO2max (+0.17 and +0.12 L/min, respectively) compared to baseline. Women who were randomized to diet+exercise increased pedometer steps/day more than women who did exercise alone (p=0.006).
Percentage of calories from fat decreased in both the diet alone (−18%,) and diet+exercise (−20.0%) groups. Average fiber intake and daily fruit and vegetable servings also increased in the diet alone (+3.4 g/day; +1.7 servings/day, respectively; both p<0.001) and diet+exercise (+2.8 g/day; +1.4 servings/day, both p<0.0001) groups, but not in the exercise group (+0.53 g/day; 0 servings/day, both p>0.5) compared to controls (−0.2 g/day; −0.9 servings/day). In both groups, women attended an average of 27 diet counseling sessions (86%). There were no significant differences in measures of adherence between mid-aged (n=304) and older (n=134) women (data not shown).
At 12 months, the mean weight loss was −2.4% (p=0.03) in the exercise group, −8.5% (p<0.001) in the diet group, and −10.8% (p<0.001) in the diet+exercise group, compared to −0.8% loss among controls. Weight loss was not significantly different between middle-aged and older women within each study arm (all p >0.2). Women in all intervention groups significantly reduced waist circumference and % body fat (all p<0.01) compared to controls.14 Lean mass decreased significantly in the diet alone group (p=0.005) but was preserved in exercisers (both p>0.10).
Baseline Associations
Baseline BMI, waist circumference, and total fat mass (kg) were correlated with insulin (r=0.39, r=0.47, r=0.25), C-peptide (r=0.43, r=0.50, r=0.33), glucose (r=0.26, r=0.24, r=0.24), and HOMA-IR (r=0.40, r=0.47, r=0.27) (all p<0.01). VO2max and pedometer steps/day were also significantly correlated with each of the metabolic variables (r= −0.11 to −0.24, all p<0.05).
Intervention Effects
Compared to controls, women in the diet and diet+exercise groups experienced significant reductions in serum insulin, C-peptide, and glucose (Table 2). HOMA-IR decreased by 24% (p<0.001) in the diet alone group and by 26% (p<0.001) in the diet+exercise group, compared to 2% in controls. In contrast, no overall significant treatment effects were observed for women assigned to exercise alone (−9%, p=0.19) (Table 2). Compared to diet alone, women receiving diet+exercise did not experience greater improvements in insulin (p=0.69), C-peptide (p=0.32), glucose (p=0.52), or HOMA-IR (p=0.64), but did have significantly greater improvements than women who were randomized to exercise alone (insulin: p<0.001; C-peptide: p<0.001; glucose: p=0.03; HOMA-IR: p<0.001). No significant age interactions were detected; the magnitude of metabolic improvement was similar between middle-aged and older women in each intervention group compared to controls (Table 3). However, within the exercise alone group, mid-aged women did experience greater improvement in insulin, C-peptide, and HOMA-IR compared to older women.
Table 2.
Baseline, 12-month, and % change values in serum insulin, C-peptide, glucose, and HOMA-IR across four arms of the NEW trial
| Variable | CONTROL | EXERCISE | DIET | DIET + EXERCISE | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline Geo M (95% CI) |
12 months Geo M 95% CI |
Changea (%) |
Baseline Geo M 95% CI |
12 months Geo M 95% CI |
Change* (%) |
Pb | Baseline Geo M 95% CI |
12 months Geo M 95% CI |
Change* (%) |
pt | Baseline Geo M 95% CI |
12 months Geo M 95% CI |
Change* (%) |
pt | |
| Insulin (pmol/L) | 83.27 (75.21, 92.23) | 81.67 (73.83, 90.35) | −1.9 | 75.98 69.24, 83.34 |
70.08 64.17, 76.46 |
−7.8 | 0.22 | 76.40 68.62, 89.94 |
59.38 53.55, 65.91 |
−22.3 | <0.001 | 74.10 67.02, 92.02 |
56.32 50.91, 62.37 |
−24.0 | <0.001 |
| C-peptide (nmol/L) | 0.87 0.81, 0.94 |
0.88 0.82, 0.94 |
1.1 | 0.82 0.77, 0.87 |
0.79 0.74, 0.84 |
−3.7 | 0.14 | 0.83 0.77, 0.89 |
0.73 0.68, 0.78 |
−12.0 | <0.001 | 0.81 0.76, 0.86 |
0.69 0.65, 0.74 |
−14.8 | <0.001 |
| Glucose (mmol/L) | 5.37 5.27, 5.46 |
5.36 5.27, 5.45 |
0.2 | 5.30 5.22, 5.38 |
5.25 5.17, 5.32 |
−0.9 | 0.29 | 5.35 5.27, 5.44 |
5.22 5.14, 5.30 |
−2.4 | 0.008 | 5.31 5.23, 5.39 |
5.16 5.07, 5.22 |
−2.8 | 0.002 |
| HOMA-IR | 2.83 2.54, 3.16 |
2.78 2.49, 3.09 |
−1.8 | 2.55 2.31, 2.82 |
2.33 2.12, 2.56 |
−8.6 | 0.19 | 2.59 2.31, 2.90 |
1.96 1.76, 2.19 |
−24.3 | <0.001 | 2.50 2.24, 2.78 |
1.84 1.65, 2.05 |
−26.4 | <0.001 |
percentage change from baseline to 12 months
p-value comparing change from baseline to 12 months in each intervention group versus controls
Table 3.
12-month changes in body weight and metabolic markers in women aged 50–60 years and 60–75 years, within each intervention arm
| Control | Exercise | Diet | Diet + Exercise | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 50–60 years | 60–75 years | 50–60 years | 60–75 years | p | 50–60 years | 60–75 years | p | 50–60 years | 60–75 years | p | |
| Body weight (%) | −0.5 | −1.6 | −2.0 | −3.2 | 0.83 | −9.1 | −7.2 | 0.05 | −10.4 | −11.6 | 0.75 |
| % Body Fat | 0.1 | −1.3 | −2.8 | −2.0 | 0.57 | −9.9 | −3.1 | 0.01 | −12 | −6.1 | 0.57 |
| Insulin (%) | −1.8 | −2.4 | −13.6 | 6.1* | 0.12 | −21.8 | −23.1 | 0.75 | −23.1 | −26.0 | 0.78 |
| C-peptide (%) | 0.0 | 0.1 | −6.4 | 2.9* | 0.26 | −11.4 | −13.2 | 0.62 | −13.5 | −16.3 | 0.52 |
| Glucose (%) | 0.5 | −1.5 | −1.2 | −3.8 | 0.45 | −2 | −0.8 | 0.77 | −2.6 | −4.2 | 0.82 |
| HOMA-IR (%) | −1.1 | −3.9 | −14.6 | 5.2* | 0.11 | −23.1 | −26 | 0.80 | −25 | −29.3 | 0.82 |
p= age interaction effect in each intervention arm compared to controls
p<0.01 between age groups within intervention arm
As expected, the magnitude of weight loss was associated with metabolic improvement in insulin, C-peptide, glucose and HOMA-IR (Table 4). These associations were independent of changes in cardiorespiratory fitness (Table 4) and pedometer steps/day. Greater metabolic improvement was observed with greater magnitude of change in average pedometer steps/day; however, these associations were not significant after adjusting for weight change, except for C-peptide. The magnitude of change in VO2max (L/min) was not associated with metabolic changes.
Table 4.
Changes in serum insulin, C-peptide, glucose and HOMA-IR among NEW participants stratified according to 12-month weight loss, fat loss, change in cardiorespiratory fitness, and change in average pedometer steps/day
| Weight Loss (%)a | ||||
|---|---|---|---|---|
| <5% | ≥5%–10% | ≥10% | p trendc | |
| n | 155 | 74 | 122 | |
| Insulin (pmol/L) | −5.83 (39.38) | −17.15 (44.52) | −29.66 (34.31) | <0.001 |
| C-peptide (nmol/L) | 0.007 (0.17) | −0.10 (0.19) | −0.19 (0.18) | <0.001 |
| Glucose (mmol/L) | −0.01 (0.35) | −0.20 (0.34) | −0.21 (0.37) | <0.001 |
| HOMA-IR | −0.23 (1.63) | −0.69 (1.74) | −1.10 (1.25) | <0.001 |
|
| ||||
|
Total fat loss (kg)a*
| ||||
| Tertile 3 | Tertile 2 | Tertile 1 | P trendc | |
| n | 117 | 117 | 117 | |
| Insulin (pmol/L) | −1.88 (22.29) | −19.65 (51.60) | −27.99 (36.67) | <0.001 |
| C-peptide (nmol/L) | 0.01(0.12) | −0.11 (0.23) | −0.0.18 (0.17) | <0.001 |
| Glucose (mmol/L) | 0.008 (0.35) | −0.15 (0.36) | −0.22 (0.35) | <0.001 |
| HOMA-IR | −0.06 (0.89) | −0.78 (2.06) | −1.06 (1.40) | <0.001 |
|
| ||||
|
ΔVO2max (L/min)b**
| ||||
| Tertile 3 | Tertile 2 | Tertile 1 | p trendc | |
| n | 80 | 75 | 78 | |
| Insulin (pmol/L) | −8.75 (36.88) | −17.36 (31.46) | −17.64 (51.88) | 0.01 |
| C-peptide (nmol/L) | −0.06 (0.18) | −0.09 (0.17) | −0.09 (0.23) | 0.03 |
| Glucose (mmol/L) | −0.04 (0.32) | −0.18 (0.40) | −0.12 (0.38) | 0.08 |
| HOMA-IR | −0.31 (1.49) | −0.67 (1.18) | −0.69 (2.09) | 0.01 |
|
| ||||
|
ΔSteps/dayb***
| ||||
| <1 | 1–3520 | >3520 | p trendc | |
| n | 68 | 72 | 93 | |
| Insulin (pmol/L) | −9.38 (28.89) | −6.04 (28.34) | −24.72 (53.41) | 0.001 |
| C-peptide (nmol/L) | −0.03 (0.15) | −0.04 (0.16) | −0.15 (0.22) | <0.001 |
| Glucose (mmol/L) | −0.07 (0.34) | −0.08 (0.367) | −0.19 (0.39) | 0.01 |
| HOMA-IR | −0.35 (1.16) | −0.25 (1.08) | −0.94 (2.15) | 0.001 |
groups include women randomized to any intervention (n=351)
groups include women randomized to exercise or exercise + diet (n=233)
adjusted for age, intervention arm, and baseline value of metabolic variable of interest
additionally adjusted for 12-month change in pedometer steps/day;
additionally adjusted for 12-month % weight change
Fat loss (group M): tertile =0.06 kg; tertile 2=−4.5 kg; tertile 3=−12.0 kg
VO2max(group M): tertile 1=−0.12 L/minute; tertile 2=0.12 L/minute; tertile 3=0.44 L/minute
3520 steps is equivalent to ≈2 miles
Higher baseline fasting glucose was a strong predictor of metabolic improvement in participants randomized to interventions (results not shown). In analyses limited to women with impaired fasting glucose at baseline, the odds (95% CI) of regression to fasting glucose <5.6 mmol/L at 12 months, independent of baseline level, age, and % weight change were 3.16 (1.02, 9.75) in the exercise alone group, 1.97 (0.63, 6.16) in the diet alone group, and 2.35 (0.68, 8.18) in the diet+exercise group (Table 5). The AOR of returning to normal fasting glucose in the diet+exercise group compared to diet alone was 1.17 (0.43, 3.16). Corresponding odds associated with categories of weight loss and VO2max are also shown in Table 5. Overall, the 1-year incidence of developing impaired fasting glucose among women with normal baseline values was 18% among controls, 6% in the exercise alone group, 4% in the diet alone group, and 2% in the diet+exercise group.
Table 5.
Odds of regression to normal fasting glucose (NFG; <5.6 mmol/L) at 12 months among study participants with impaired fasting glucose (IFG; 5.6–6.9 mmol/L) at baseline.
| IFG | IFG | Incident IFG | Odds of returning to NFG (95% CI)b | ||
|---|---|---|---|---|---|
| Baseline (n) | 12 mo (n) | 12 moa | Model 1 | Model 2 | |
| Control | 30 | 23 | 10/57 | 1.00 (ref) | 1.00 |
| Diet | 43 | 23 | 3/75 | 3.19 (1.09, 9.28) | 1.97 (0.63–6.16) |
| Exercise | 35 | 17 | 5/82 | 3.64 (1.21, 11.01) | 3.16 (1.02–9.75) |
| Diet+Exercise | 35 | 14 | 2/81 | 5.17 (1.71–15.69) | 2.35 (0.68–8.18) |
| Body Weight Change (%) | |||||
| ≤0% loss (no change or gain) | 35 | 25 | 9/81 | 1.00 | 1.00 |
| <5% loss | 43 | 28 | 4/61 | 1.65 (0.60–4.51) | 1.62 (0.58–4.50)c |
| 5%–10% loss | 27 | 10 | 1/56 | 4.97 (1.63–15.11) | 5.12 (1.65–15.86)c |
| ≥10% loss | 38 | 14 | 6/88 | 6.26 (2.14–18.29) | 6.86 (2.27–20.77)c |
| ptrend=0.001 | |||||
| Change in VO2max (L/min)d | |||||
| No change or deterioration | 46 | 24 | 10/154 | 1.00 | 1.00 |
| +0.01–0.14 L/minute | 50 | 36 | 4/47 | 1.17 (0.45–2.99) | 0.78 (0.27–2.21)e |
| ≥+0.14 L/minute | 47 | 17 | 6/94 | 2.78 (1.27–6.07) | 2.48 (1.11–5.57)e |
| ptrend=0.337 | |||||
Incident IFG = new cases of IFG (100–125 mg/dL) among women with values <100mg/dL at baseline
Model 1: adjusted for age, baseline serum fasting glucose; Model 2: additionally adjusted for % body weight change. Stratified analyses were also adjusted for group randomization.
Analyses additionally adjusted for change in VO2max;
Groups represent approximate tertiles of change in cardiorespiratory fitness
Analyses additionally adjusted for change in % body weight change
Discussion
A 12-month group-based modification of the DPP/LookAHEAD weight loss interventions8,17 resulted in significant weight loss and improved insulin resistance. However, adding a prescribed 225 minutes/week of aerobic exercise did not yield significant added benefits in these outcomes up to 12 months. Furthermore, the effect of these lifestyle interventions was similar in middle-aged and older women. Although no overall improvement in HOMA-IR was detected in women randomized to exercise alone compared to controls, 12 months of exercise training was associated with improved odds of regressing from impaired to normal fasting glucose (<5.6 mmol/L), independent of weight loss. These findings support previous reports that modest weight loss (5%–10% body weight) is associated with improved insulin sensitivity and glucose tolerance.8,9,24–26 They are also consistent with reports that regular exercise can improve insulin sensitivity, even in the absence of substantial weight reduction;27,28 however, in the current study, this effect was limited to women with impaired fasting glucose at baseline. The mechanisms postulated to account for these favorable changes include a preferential reduction in visceral fat and/or enhanced metabolic efficiency of muscle.29,30
In the DPP, increasing age was associated with more physical activity, greater weight loss, and a lower incidence of diabetes, but not with reported caloric intake or reversion to normal fasting glucose.10 Other large type 2 diabetes prevention trials have not included older adults or have been unable to examine age-related differences in the response to lifestyle interventions.31–33 To our knowledge, no previous randomized trial has examined differences in the response to weight loss and exercise interventions in mid-aged and older women. In this study, there were no significant differences in any measure of intervention adherence between middle-aged and older women, and the magnitude of the difference between middle-aged and older women was not significantly different from controls in any of the intervention arms. Due to limited sample size, it was not possible to examine the reversion to normal fasting glucose by age group.
Although women assigned to exercise alone failed to achieve significant weight loss compared to controls, they experienced significant reductions in waist circumference and body fat. Larson-Meyer et al.34 recently demonstrated that under conditions of equal energy deficits, overweight men and women who were randomized to caloric restriction alone or in combination with exercise showed similar reductions in body weight, total body fat and visceral fat. Yet, those who performed exercise had significantly greater improvements in insulin sensitivity and other markers of cardiovascular risk. Enhanced parasympathetic tone and endothelial function have been proposed as pathways through which exercise may influence cardiometabolic health beyond ‘traditional’ risk factors.35
A limitation of the current study is that the sample was predominantly Caucasian and therefore it was not possible to examine the consistency of the current findings across racial/ethnic groups. In addition, fasting glucose was used rather than glucose tolerance testing or HgA1C, and potential differences in insulin secretion versus insulin action could not be compared between treatment arms. However, impaired fasting glucose is an important clinical marker in itself.23 Additional strengths of the present study include its relatively large size and adequate statistical power to examine differences in insulin sensitivity between middle-aged and older women assigned to multiple interventions. Furthermore, the group-based lifestyle interventions used in this study were less staff-time intensive than those used in the DPP8 and many other large trials,36 yet yielded substantial weight loss and improvements in glucose homeostasis. This suggests that group-based diet and exercise programs may be as effective as one-on-one counseling in eliciting favorable improvements. Future studies to determine the comparative effectiveness of these programs with larger and more diverse groups, and in other settings would provide an important contribution to the current literature.
This study provides evidence that clinically meaningful improvements in insulin sensitivity can be achieved through caloric restriction alone or with the addition of exercise. Furthermore, a prescribed exercise dose of 225 minutes/week resulted in regression to normal glucose levels for more than half of women with impaired fasting glucose at baseline. In the DPP, individuals who were randomized to lifestyle intervention who did not achieve the desired goal of 7% reduction in body weight but attained the physical activity goal of >150 minutes/week experienced a 44% reduction in the risk of progression to diabetes.9 The optimal exercise dose for ‘treatment’ of impaired fasting glucose remains unknown but clearly warrants further investigation.
Weight loss among older people remains controversial, primarily due to concern over the potentially deleterious loss of muscle and bone mass. The exercise program tested in this study did not include resistance training; however resistance exercise has been shown to have favorable effects on glycemic control37 and to help preserve lean mass during weight loss.25 Davidson et al.27 have previously shown that the combination of resistance and aerobic exercise was the optimal exercise strategy for simultaneous reduction in insulin resistance and functional limitation in obese older adults. The current findings and those of others therefore suggest that weight loss combined with exercise is likely to be the safest and most effective approach to glycemic control among older adults.
In the U.S., approximately 57 million adults have impaired fasting glucose, and up to 70% will progress to overt type 2 diabetes in their lifetime without effective intervention.38 True disease prevention should be counted as the maintenance or restoration of normal glucose regulation rather than a prolonged prediabetic state, with which there remains significant health risk.2 The American Diabetes Association currently recommends that people with prediabetes be referred to an effective program promoting 5%–10% body weight loss and at least 150 minutes/week of moderate physical activity.39 While maximum benefit will be achieved with successful weight loss, ongoing effort to promote regular physical activity has the potential for health benefits among women with impaired fasting glucose, and to help prevent the potentially deleterious loss of lean mass in older individuals undergoing weight loss.
Acknowledgments
Funding/Support: This work was supported by the National Cancer Institute at the NIH (grant number: R01 CA102504, U54-CA116847, 5KL2RR025015-03 to K.F.S, R25 CA94880 and 2R25CA057699-16 to A.K.); and the Canadian Institutes of Health Research (Fellowship to K.L.C & C.M). None of the funding agencies were involved in the trial design or conduct. While working on the trial, Dr. Alfano was employed at The Ohio State University, and located to NCI following completion of her effort on the NEW trial.
Footnotes
No financial disclosures were reported by the authors of this paper.
Trial Registration #: www.clinicaltrials.gov Identifier NCT00470119
Author Contributions: Drs Mason and McTiernan had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the analysis. Study concept and design: Mason, Foster-Schubert, Alfano, Blackburn, McTiernan; Acquisition of data: Kong, Bain, Duggan, Campbell, Blackburn, McTiernan; Analysis and interpretation of data: Mason, Xiao, Imayama, McTiernan; Drafting of the manuscript: Mason, Foster-Schubert. Critical revision of the manuscript for important intellectual content: Foster-Schubert, Imayama, Duggan, Kong, Campbell, Blackburn, Alfano, Ulrich, McTiernan; Statistical analysis: Mason, Xiao, Imayama; Obtained funding: McTiernan; Administrative, technical, or material support: Bain, Xiao; Study supervision: McTiernan.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.International Diabetes Federation. Diabetes Atlas. 3. Brussels: International Diabetes Federation; 2006. [Google Scholar]
- 2.Reaven GM. Pathophysiology of insulin resistance in human disease. Physiol Rev. 1995;75(3):473–86. doi: 10.1152/physrev.1995.75.3.473. [DOI] [PubMed] [Google Scholar]
- 3.Coutinho M, Gerstein HC, Wang Y, Yusuf S. The relationship between glucose and incident cardiovascular events. A metaregression analysis of published data from 20 studies of 95,783 individuals followed for 12. 4 years. Diabetes Care. 1999;22(2):233–40. doi: 10.2337/diacare.22.2.233. [DOI] [PubMed] [Google Scholar]
- 4.Folsom AR, Rasmussen ML, Chambless LE, Howard G, Cooper LS, Schmidt MI, et al. Prospective associations of fasting insulin, body fat distribution, and diabetes with risk of ischemic stroke. The Atherosclerosis Risk in Communities (ARIC) Study Investigators. Diabetes Care. 1999;22(7):1077–83. doi: 10.2337/diacare.22.7.1077. [DOI] [PubMed] [Google Scholar]
- 5.Levitzky YS, Pencina MJ, D’Agostino RB, Meigs JB, Murabito JM, Vasan RS, et al. Impact of impaired fasting glucose on cardiovascular disease: the Framingham Heart Study. J Am Coll Cardiol. 2008;51(3):264–70. doi: 10.1016/j.jacc.2007.09.038. [DOI] [PubMed] [Google Scholar]
- 6.Pisani P. Hyper-insulinaemia and cancer, meta-analyses of epidemiological studies. Arch Physiol Biochem. 2008;114(1):63–70. doi: 10.1080/13813450801954451. [DOI] [PubMed] [Google Scholar]
- 7.Rapp K, Schroeder J, Klenk J, Ulmer H, Concin H, Diem G, et al. Fasting blood glucose and cancer risk in a cohort of more than 140,000 adults in Austria. Diabetologia. 2006;49(5):945–52. doi: 10.1007/s00125-006-0207-6. [DOI] [PubMed] [Google Scholar]
- 8.Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393–403. doi: 10.1056/NEJMoa012512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Perreault L, Kahn SE, Christophi CA, Knowler WC, Hamman RF. Regression from pre-diabetes to normal glucose regulation in the diabetes prevention program. Diabetes Care. 2009;32(9):1583–8. doi: 10.2337/dc09-0523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Crandall J, Schade D, Ma Y, Fujimoto WY, Barrett-Connor E, Fowler S, et al. The influence of age on the effects of lifestyle modification and metformin in prevention of diabetes. J Gerontol A Biol Sci Med Sci. 2006;61(10):1075–81. doi: 10.1093/gerona/61.10.1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mokdad AH, Bowman BA, Ford ES, Vinicor F, Marks JS, Koplan JP. The continuing epidemics of obesity and diabetes in the U. S Jama. 2001;286(10):1195–200. doi: 10.1001/jama.286.10.1195. [DOI] [PubMed] [Google Scholar]
- 12.Houston DK, Nicklas BJ, Zizza CA. Weighty concerns: the growing prevalence of obesity among older adults. J Am Diet Assoc. 2009;109(11):1886–95. doi: 10.1016/j.jada.2009.08.014. [DOI] [PubMed] [Google Scholar]
- 13.Witham MD, Avenell A. Interventions to achieve long-term weight loss in obese older people: a systematic review and meta-analysis. Age Ageing. 2010;39(2):176–84. doi: 10.1093/ageing/afp251. [DOI] [PubMed] [Google Scholar]
- 14.Foster-Schubert KE, Alfano CM, Duggan C, Xiao L, Campbell KL, Kong A, et al. Effect of exercise and diet, alone or combined, on weight and body composition in overweight-to-obese post-menopausal women. [Accepted, January, 2011];Obesity. 2011 doi: 10.1038/oby.2011.76. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.2008 Physical Activity Guidelines for Americans. DHHS; Accessed at: www.health.gov/paguidelines. [Google Scholar]
- 16.Ainsworth BE, Haskell WL, Whitt MC, Irwin ML, Swartz AM, Strath SJ, et al. Compendium of physical activities: an update of activity codes and MET intensities. Med Sci Sports Exerc. 2000;32(9 Suppl):S498–504. doi: 10.1097/00005768-200009001-00009. [DOI] [PubMed] [Google Scholar]
- 17.Ryan DH, Espeland MA, Foster GD, Haffner SM, Hubbard VS, Johnson KC, et al. Look AHEAD (Action for Health in Diabetes): design and methods for a clinical trial of weight loss for the prevention of cardiovascular disease in type 2 diabetes. Control Clin Trials. 2003;24(5):610–28. doi: 10.1016/s0197-2456(03)00064-3. [DOI] [PubMed] [Google Scholar]
- 18.Taylor HL, Jacobs DR, Jr, Schucker B, Knudsen J, Leon AS, Debacker G. A questionnaire for the assessment of leisure time physical activities. J Chronic Dis. 1978;31(12):741–55. doi: 10.1016/0021-9681(78)90058-9. [DOI] [PubMed] [Google Scholar]
- 19.Pate R, Blair S, Durstine J. Guidelines for Exercise Testing and Prescription. Philadelphia, Pa: Lea & Febinger; 1991. pp. 70–72. [Google Scholar]
- 20.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–9. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 21.Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults--The Evidence Report. NIH. Obes Res. 1998;6(Suppl 2):51S–209S. [PubMed] [Google Scholar]
- 22.Christian JG, Tsai AG, Bessesen DH. Interpreting weight losses from lifestyle modification trials: using categorical data. Int J Obes. 2010;34(1):207–9. doi: 10.1038/ijo.2009.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2003;26 (Suppl 1):S5–20. doi: 10.2337/diacare.26.2007.s5. [DOI] [PubMed] [Google Scholar]
- 24.Weiss EP, Racette SB, Villareal DT, Fontana L, Steger-May K, Schechtman KB, et al. Improvements in glucose tolerance and insulin action induced by increasing energy expenditure or decreasing energy intake: a randomized controlled trial. Am J Clin Nutr. 2006;84(5):1033–42. doi: 10.1093/ajcn/84.5.1033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ross R, Dagnone D, Jones PJ, Smith H, Paddags A, Hudson R, et al. Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men. A randomized, controlled trial. Ann Intern Med. 2000;133(2):92–103. doi: 10.7326/0003-4819-133-2-200007180-00008. [DOI] [PubMed] [Google Scholar]
- 26.Tuomilehto J, Lindstrom J, Eriksson JG, Valle TT, Hamalainen H, Ilanne-Parikka P, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med. 2001;344(18):1343–50. doi: 10.1056/NEJM200105033441801. [DOI] [PubMed] [Google Scholar]
- 27.Davidson LE, Hudson R, Kilpatrick K, Kuk JL, McMillan K, Janiszewski PM, et al. Effects of exercise modality on insulin resistance and functional limitation in older adults: a randomized controlled trial. Arch Intern Med. 2009;169(2):122–31. doi: 10.1001/archinternmed.2008.558. [DOI] [PubMed] [Google Scholar]
- 28.Houmard JA, Tanner CJ, Slentz CA, Duscha BD, McCartney JS, Kraus WE. Effect of the volume and intensity of exercise training on insulin sensitivity. J Appl Physiol. 2004;96(1):101–6. doi: 10.1152/japplphysiol.00707.2003. [DOI] [PubMed] [Google Scholar]
- 29.Menshikova EV, Ritov VB, Toledo FG, Ferrell RE, Goodpaster BH, Kelley DE. Effects of weight loss and physical activity on skeletal muscle mitochondrial function in obesity. Am J Physiol Endocrinol Metab. 2005;288(4):E818–25. doi: 10.1152/ajpendo.00322.2004. [DOI] [PubMed] [Google Scholar]
- 30.Hughes VA, Fiatarone MA, Fielding RA, Kahn BB, Ferrara CM, Shepherd P, et al. Exercise increases muscle GLUT-4 levels and insulin action in subjects with impaired glucose tolerance. Am J Physiol. 1993;264(6 Pt 1):E855–62. doi: 10.1152/ajpendo.1993.264.6.E855. [DOI] [PubMed] [Google Scholar]
- 31.Li G, Zhang P, Wang J, Gregg EW, Yang W, Gong Q, et al. The long-term effect of lifestyle interventions to prevent diabetes in the China Da Qing Diabetes Prevention Study: a 20-year follow-up study. Lancet. 2008;371(9626):1783–9. doi: 10.1016/S0140-6736(08)60766-7. [DOI] [PubMed] [Google Scholar]
- 32.Saaristo T, Moilanen L, Korpi-Hyovalti E, Vanhala M, Saltevo J, Niskanen L, et al. Lifestyle intervention for prevention of type 2 diabetes in primary health care: one-year follow-up of the Finnish National Diabetes Prevention Program (FIN-D2D) Diabetes Care. 2010;33(10):2146–51. doi: 10.2337/dc10-0410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ramachandran A, Snehalatha C, Mary S, Mukesh B, Bhaskar AD, Vijay V. The Indian Diabetes Prevention Programme shows that lifestyle modification and metformin prevent type 2 diabetes in Asian Indian subjects with impaired glucose tolerance (IDPP-1) Diabetologia. 2006;49(2):289–97. doi: 10.1007/s00125-005-0097-z. [DOI] [PubMed] [Google Scholar]
- 34.Larson-Meyer DE, Redman L, Heilbronn LK, Martin CK, Ravussin E. Caloric restriction with or without exercise: the fitness versus fatness debate. Med Sci Sports Exerc. 2009;42(1):152–9. doi: 10.1249/MSS.0b013e3181ad7f17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Joyner MJ, Green DJ. Exercise protects the cardiovascular system: effects beyond traditional risk factors. J Physiol. 2009;587(Pt 23):5551–8. doi: 10.1113/jphysiol.2009.179432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Walker KZ, O’Dea K, Gomez M, Girgis S, Colagiuri R. Diet and exercise in the prevention of diabetes. J Hum Nutr Diet. 2010;23(4):344–52. doi: 10.1111/j.1365-277X.2010.01061.x. [DOI] [PubMed] [Google Scholar]
- 37.Sigal RJ, Kenny GP, Boule NG, Wells GA, Prud’homme D, Fortier M, et al. Effects of aerobic training, resistance training, or both on glycemic control in type 2 diabetes: a randomized trial. Ann Intern Med. 2007;147(6):357–69. doi: 10.7326/0003-4819-147-6-200709180-00005. [DOI] [PubMed] [Google Scholar]
- 38.Cowie CC, Rust KF, Byrd-Holt DD, Eberhardt MS, Flegal KM, Engelgau MM, et al. Prevalence of diabetes and impaired fasting glucose in adults in the U.S. population: National Health And Nutrition Examination Survey 1999–2002. Diabetes Care. 2006;29(6):1263–8. doi: 10.2337/dc06-0062. [DOI] [PubMed] [Google Scholar]
- 39.American Diabetes Association. Executive summary: Standards of medical care in diabetes--2010. Diabetes Care. 2010;33 (Suppl 1):S4–10. doi: 10.2337/dc10-S004. [DOI] [PMC free article] [PubMed] [Google Scholar]