Abstract
As genetic advances become incorporated into health care delivery, disparities between developing and developed countries may become greater. By addressing genetic health care needs and specific differences of developing countries, these disparities may be mitigated. We sought to describe the attitudes and knowledge of subjects with hereditary neurological diseases in Mali before and after receiving genetic testing and counseling for the first time. A questionnaire of attitudes and knowledge items was adapted and piloted for use in Mali. We found that the majority of subjects had positive attitudes toward genetic testing and counseling, both before and afterwards. Subjects responded to approximately half of the knowledge questions regarding hereditary transmission correctly before and after genetic testing and counseling. Neither overall attitudes nor knowledge scores changed significantly from baseline. Concerns about confidentiality were expressed by the majority of subjects. These findings indicate that, despite limited knowledge of patterns of inheritance, Malians understood the sensitive nature of this information and were favorable toward receiving genetic testing and counseling for diagnostic and prognostic purposes.
Keywords: Genetic testing, Genetic counseling, Developing countries, West Africa, Attitudes, Knowledge
Introduction
Genetic testing and counseling have become standard of practice in many developed countries to confirm the diagnosis of hereditary diseases. Certain genetic conditions, such as sickle cell disease and glucose-6-phosphate dehydrogenase deficiency (G6PD), are checked regularly on all newborns in developed countries so that treatment can be initiated promptly (Carreiro-Lewandowski 2002; Bodamer et al. 2007). In Africa, many genetic conditions including sickle cell disease, G6PD, thalassemia, and porphyria, among others, affect millions of people but confirmatory genetic testing is not readily available; many are not even exposed to the concept of genetic testing. As discoveries in genetics contribute increasingly to health care delivery, the integration of genetic health care in the developing world is an important goal in the minimization of health care disparities (Modell and Darr 2002).
Various factors are important to recognize in the appropriate delivery of genetic testing and counseling. These include differences between health care systems and societies, fear of stigmatization, access to treatment, and varying attitudes toward receiving a diagnosis (Berth et al. 2002; Tan et al. 2007). Acceptance of genetic testing is also influenced by knowledge and attitudes about testing, yet this relationship between acceptance and knowledge and attitudes is poorly understood (Rose et al. 2005; Morren et al. 2007; Tan et al. 2007). This implies that, in clinical practice, genetic testing and counseling should be developed in a prudent manner (de Villiers et al. 1997; Penn et al. 2010).
Patient attitudes and knowledge regarding genetic testing have been studied in many countries. The majority of findings show that attitudes toward genetic testing are overall favorable and knowledge scores are higher among more educated individuals. However, these research results are mostly limited to highly educated Caucasian individuals in developed countries (de Villiers et al. 1997; Rew et al. 2009; Etchegary et al. 2010). Few such studies have been performed in developing countries, particularly in sub-Saharan Africa. Four South African publications and two Nigerian publications have reported results regarding attitudes and knowledge about genetic testing and counseling (Ebomoyi 1988; de Villiers et al. 1997; Moronkola and Fadairo 2006; Basson et al. 2007; Futter et al. 2009; Penn et al. 2010). Although many of the subjects in these studies were again highly educated, white individuals, some subjects were of black or mixed ancestry (Basson et al. 2007; Futter et al. 2009). Less uptake of testing in the mixed ancestry population may have been due to lower education and socioeconomic status (Futter et al. 2009). Findings from these studies showed that patients participated in hopes of reducing anxiety, planning for the future, finding a treatment or cure, or to increase potential of treatment/cure for future generations (Basson et al. 2007; Futter et al. 2009). Regardless of positive or negative results, subjects had no regrets about participating (Futter et al. 2009). Also, despite the negative psychological impact of the diseases, subjects wanted to have children (de Villiers et al. 1997). They worried about their family members with disease and about developing the disease, but, other than in the case of sickle cell disease, had low knowledge of their own disease risks (de Villiers et al. 1997; Moronkola and Fadairo 2006). Identifying the mutation causing their condition did not change their lived experience (Basson et al. 2007). Understanding the culture of the subjects was important to the appropriate delivery of genetic testing and counseling (Futter et al. 2009; Penn et al. 2010). As in other studies of genetic testing, religion was a theme as well: turning to God helped contain fear and anxiety and resulted in a positive psychological impact of strengthened religious conviction (de Villiers et al. 1997).
To gain more insight into the impact and appropriateness of delivery of genetic care in developing countries, we provided genetic testing and counseling to patients in Mali, West Africa, for hereditary neurological conditions and assessed the attitudes and knowledge of the subjects before and after the testing.
Methods
Ethics statement
We obtained approval from the National Institute of Neurological Disorders and Stroke (NINDS) and Johns Hopkins University School of Medicine Institutional Review Boards and the University of Bamako Ethics Committee for this study. Subjects provided informed consent for the collection of questionnaires and biological samples and for subsequent analyses. This study adhered to the principles expressed in the Declaration of Helsinki (2008).
Study purpose
This project was part of an ongoing study at the NINDS, where patients with various hereditary neurological diseases are seen in a neurogenetics clinic. As many neurological conditions overlap in their presentation and are difficult to diagnose by clinical examination alone, a neurologist in Mali (MT) sought collaboration with the Neurogenetics Branch at NINDS through the National Institutes of Health (NIH) intramural site in Bamako, Mali. The goal of this collaboration was to provide genetic testing to substantiate or redirect the clinical diagnoses for patients seen in the intramural site in Mali. Because this was the first time the Malian patients were exposed to genetic testing, we evaluated their attitudes and knowledge about genetic testing and counseling to better understand their needs and expectations. A questionnaire was therefore administered before and after genetic testing and counseling, if appropriate. All evaluations were provided to patients free of charge. The biological samples collected in Mali were used for clinically indicated genetic testing. The samples were stored behind locked doors in both Mali and at the NIH while the test results were pending.
Sample
To be enrolled in the study, individuals were first screened in the Neurology Department of Point G Hospital in Bamako. Eligibility criteria included those with a familial component or a phenotype characteristic of a hereditary neurological disorder. Children under 18 and individuals with cognitive deficits were excluded. Thirty-eight people from different regions of Mali who were screened at the Point G Hospital met the initial criteria and completed pre-counseling questionnaire. Malian physicians and investigators from the NIH then evaluated them and proposed a set of genetic testing to confirm the diagnoses. The post-counseling questionnaire was administered 3 to 6 months after the pre-counseling questionnaire, once genetic test results were confirmed positive. Genetic testing results were returned to them by the genetic counselor together with further genetic counseling, followed by administration of the post-questionnaire. Seven were not eligible for the post-counseling questionnaire or did not complete it.
Of the 31 adults for whom pre- and post-counseling data were obtained, 18 probands received positive genetic testing results for their familial diagnoses, which included muscular dystrophies (Duchenne, limb girdle), ataxias (ataxia telangiectasia, SCA3 and SCA7), myotonia congenita, Lafora disease, and Kearns–Sayre syndrome. Thirteen respondents received negative or inconclusive genetic testing results for phenotypes of hereditary spastic paraparesis, limb girdle muscular dystrophy, and non-specific mental retardation (please see Fig. 1 for flow of study and Table 1 for details of the total number of positive diagnoses, which includes immediate relatives of probands).
Fig. 1.
Flow diagram of study, including results of genetic testing with counseling (GT & GC). Although only one proband per family was tested, often each family had more than one affected individual, as reflected by the middle boxes. For the final sample of 31 adults who responded to the questionnaire, the relationship to the tested, affected individual in the family is shown in the box furthest to the right
Table 1.
Summary of positive diagnoses
| Diagnosis | No. affected per family | Transmission | Gene | Nucleotide change | Peptide change |
|---|---|---|---|---|---|
| Limb girdle muscular dystrophy, type 2D | 3 | AR | SGCA | Novel, homozygous 574C > T | R192X |
| Limb girdle muscular dystrophy, type 2B | 1 | AR | DYSF | 2 novel heterozygous mutations, 2643 + 1G > A, 4018G > C | Splice site, G1340R |
| Ataxia telangiectasia | 2 | AR | ATM | Novel Nt7985T > A | V2662D |
| Duchenne muscular dystrophy | 2 | X-linked | DMD | Novel 3784delG | 1262E > Frameshift KX20 |
| Duchenne muscular dystrophy | 2 | X-linked | DMD | Novel 1423G > T | 475E > X |
| Lafora disease | 2 | AR | EPM2B | Novel homozygous 560A > C | H187P |
| Myotonia congenita | 1 | AR | CLCN1 | Homozygous 501C > G, heterozygous 313C > T, heterozygous 1672C > T | F167L, R105C, P558S |
| Spinocerebellar ataxia, type 3 | 2 | AD | ATXN3 | Heterozygous CAG repeat expansion (73, 14) | Polyglutamine expansion |
| Spinocerebellar ataxia, type 7 | 3 | AD | ATXN7 | Heterozygous CAG repeat expansion (56, 12) | Polyglutamine expansion |
| Spinocerebellar ataxia, type 7 | 2 | AD | ATXN7 | Heterozygous CAG repeat expansion (59, 11) | Polyglutamine expansion |
| Kearns–Sayre syndrome | 1 | Mitoch | Mitoch DNA | 5,585 bp deletion | N/A |
AD autosomal dominant, AR autosomal recessive, Mitoch mitochondrial
Genetic counseling
The genetic counselor discussed the risks, benefits, and limitations of the proposed genetic testing. Though the counseling was tailored to the affected individual, patients were questioned about the impact of disease on the individual and on the family, along with assessing the patient’s perceived cause of the disease and what they felt was the preferred outcome of their research participation. Counseling was provided both before the genetic testing was performed and at the time of results disclosure, to assist patients in understanding and processing the psychosocial implications of the testing. The genetic counselor also included an assessment of the patient’s support system.
Genetic counseling was provided in English by a genetic counselor from the USA (ALP) and translated by a French or Malian physician into French (the official language of Mali) or Bambara (the most common primary language). Visual aids were created to assist in explaining genetics concepts such as DNA, chromosomes, and mutations to the Malian participants. Each counseling session was interactive and tailored to each patient’s condition, lasting approximately 1–2 h. Affected adults were asked if other family members could be present, and only those permitted by the affected individual were included in the counseling session.
Instrument development and pilot testing
A questionnaire was developed to measure attitudes and knowledge toward genetic testing and counseling in Mali. Three items were created after reviewing the literature (Pieterse et al. 2005). In addition, 26 items were adapted from previous questionnaires, including the NIH Cancer Genetic Studies Consortium core instruments, the Hemachromatosis and Iron Overload Study, the Johns Hopkins Study of Cystic Fibrosis Carrier Testing, and the Outcomes of Education and Counseling for Hereditary Nonpolyposis Colon Cancer Testing Baseline Questionnaire (Bernhardt et al. 1996; Lerman et al. 1997; McLaren et al. 2003). Finally, questions regarding demographics, spirituality, general health, and neurological disease history were added.
To establish content validity, the questionnaire was reviewed by a group of five content scientists from the USA. The experts had over 15 years of experience each in genetic counseling and questionnaire development (Elizabeth Thomson, Donald Hadley), international health (particularly in Africa), and questionnaire development (Peter Winch, Benjamin Wilfond, and Lori Leonard). Each expert reviewed the questionnaire separately and provided suggestions. As Mali has a story-telling tradition, experts suggested adding three vignettes related to genetic testing and counseling, and place five to six attitudes questions after each vignette. Six knowledge items were written in the form of true/false/don’t know. For the 23 attitude items, Likert-style questions were developed since this format had been used previously with success in Mali (Leplege et al. 2006). The final set of 29 questions (six knowledge items plus 23 attitudes items) was then translated into French, the official language of Mali, then Bambara, and back-translated into French. Discrepancies between the three languages were then reviewed and agreed upon by one of the official translators (fluent in all three languages), two Malian physicians (GL and MS, one fluent in all three and one in French and Bambara), a Malian psychologist (SC, fluent in French and Bambara), and an American nurse (KGM, fluent in English and French). With the exception of the official translator, the questionnaire was administered by these same individuals at both time points.
The translated questionnaire was pilot tested in ten subjects from five Malian families with hereditary diseases. Proportion of correct responses to the six knowledge questions was generally low for four items, while two were answered correctly by the majority. Nevertheless, all of the items were felt to be important and hence were retained in the final questionnaire. The six knowledge items yielded a reliability (KR-20) coefficient of 0.35, which was likely influenced by both the small number of items and of pilot subjects (Waltz et al. 2004). The 23 attitude questions were completed by nine respondents and showed a Cronbach alpha = 0.87. Later, for the baseline questionnaire, four items had a low item-total correlation (<0.2). One of these was removed, but the other three were related to stigma about genetic testing, a topic that had not been systematically examined in Mali. Thus, it was determined that the stigma items were analyzed separately from the general attitudes items. Upon removing these four items from the main scale, the Cronbach’s alpha of the remaining 19-item attitudes scale increased from 0.80 to 0.86 (range of item-total correlation = 0.14–0.71). The post-questionnaire with the same 19 items also had a good Cronbach’s alpha of 0.88. The three item stigma scale had a Cronbach’s alpha of 0.30, again likely due to the small number of items (Waltz et al. 2004).
Data analysis
Descriptive statistics were calculated for data regarding demographics, spirituality, and disease history. In addition, means and standard deviations were calculated for attitudes and knowledge toward genetic testing and counseling. The 19 attitudes items and six knowledge items were scored separately. Attitude items were scored on the basis of the four-point Likert scale responses: strongly disagree = 0 to strongly agree = 3. Responses to negatively phrased items were reversed for appropriate scoring. Total attitude scores were evaluated for favorable overall attitudes toward receiving genetic testing and counseling: attitude scale scores with a sum of greater than 38 represented an average of favorable responses to each item (possible range = 0–57). In addition, percentages were calculated for each attitude item based on the binary variable of agree/disagree where the sum of 0’s + 1’s = % disagree and 2’s + 3’s = % agree. Knowledge items were rated as follows: correct = 1 and incorrect/don’t know = 0 (possible range = 0–6). For each scale (attitudes and knowledge), pre- and post-comparisons using the Wilcoxon signed rank test were conducted to distinguish any significant differences in overall scores between time points. Mann–Whitney U and Kruskal–Wallis tests were used post hoc to compare attitude and knowledge scores by gender, age, education level, spirituality, baseline knowledge, positive or negative results, certainty of test result, actual transmission, and relationship to affected individuals.
Results
Participant characteristics
Table 2 summarizes the characteristics of study participants. The largest category of respondents was affected adults, with the rest mothers, fathers, and other blood relatives comprised of siblings, aunts, and uncles. This reflected the mothers’ propensity to accompany children to the doctor in this society. Of the 31 participants, gender, age, and education level distributed evenly. The mean age was 39 years (range = 19–69). All were Muslim, with 97% praying daily, 64% attending service daily or weekly, and 30% reading spiritual texts daily. The participants were from seven different Malian ethnic groups, with the most common being Bambara (40.6%), which is representative of the general population. Twenty-nine percent were single, 58% married, 3% separated or divorced, and 10% widowed. Seventy-one percent rated the health of themselves, if affected, or their affected relative as “poor” on a scale of good to bad health, “poor” being the worst rating.
Table 2.
Sample characteristics (N = 31)
| Questionnaire respondents | N (%) | |
|---|---|---|
| Variable | ||
| Gender | Male | 16 (52) |
| Female | 15 (48) | |
| Relationship to patient | Self | 12 (39) |
| Mother | 8 (26) | |
| Father | 5 (16) | |
| Other | 6 (19) | |
| Education | None | 10 (33) |
| Primary | 7 (23) | |
| High school | 6 (20) | |
| Superior | 7 (23) | |
| Religion | Muslim | 31 (100) |
| Age | 18–30 | 11 (36) |
| 31–44 | 10 (32) | |
| 45–70 | 10 (32) | |
With regard to their general health, most participants saw a physician (81%), while some saw a traditional healer only (6%), some both (6%), and some physician, traditional healer, and teacher of the Koran (6%) (numbers may not add to exactly 100% due to rounding). Forty-five percent of patients asked others’ opinion before deciding whom to see for medical advice. Of those, 54% asked their spouse, 31% their parents, 8% their child, and 8% neighbor. Of those who saw a physician, 23% saw a doctor regularly, while 77% only saw a doctor when they felt sick. Forty-eight percent of respondents had never had a regular check up, but 32% had been seen for one in the past year. Fifty-eight percent stated they had a regular clinic, while 39% stated they had someone they considered their regular doctor.
Attitudes towards genetic testing and counseling
Overall, participants had positive attitudes towards receiving genetic testing and counseling (score > 38), both before and afterwards. The baseline mean score was 44.8 (range 29–57, SD 8.0) and the post-attitudes mean score was 42.6 (range 34–57, SD 7.0). No significant difference in pre- and post-attitudes mean scores was observed using a Wilcoxon Signed Rank test. No significant differences in pre- and post-attitudes were noted by gender, age, education level, baseline knowledge, positive or negative results, certainty of test result, actual transmission, or level of spirituality, which was captured by the proxy question “how often do you read spiritual texts”.
Percentages of subjects who agreed with individual attitudes items are presented in Fig. 2 on a descriptive basis. Before and after genetic testing and counseling, almost all participants favored general aspects of genetic testing and counseling and items focusing on family and children. Fewer agreed with items regarding disclosing results to a spouse, the information helping to decide to have future children, or genetics being the cause of the disease. A few respondents thought this information would not help because “the disorder is God’s will” or “one cannot change what one has,” but these numbers declined after genetic testing and counseling.
Fig. 2.
Percentages of subjects who agreed with individual items on the attitudes scale
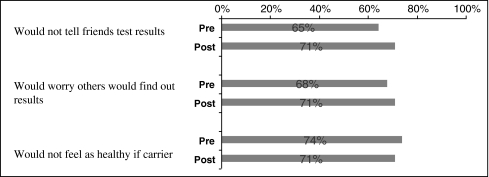
Three questions regarding stigma are described separately due to their low item to total correlation with the overall attitudes scale (Fig. 3). The majority of subjects expressed concerns regarding confidentiality both before and after genetic testing and counseling: the majority of subjects would not tell friends results and were worried others would find out their results. Both of these percentages increased after genetic testing. In contrast, the affirmative responses to the third stigma question, “I would not feel as healthy if I were a carrier,” decreased after genetic testing and counseling.
Fig. 3.
Percentages of subjects’ attitudes regarding stigma
However, no significant pre- and post-differences were found in any attitudes or stigma items when correcting for multiple testing with the Bonferroni adjustment.
Knowledge
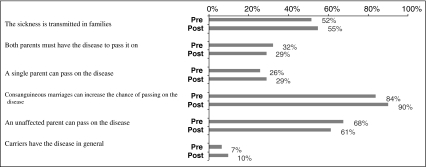
The overall pre-knowledge mean was 2.68 (±1.19), with a range from 0 to 6. Post-knowledge results were similar, with a mean of 2.74 (±1.24) and the same range. Overall knowledge scores were below 50% correct before (mean 2.68/6.0 or 45%) and after (mean 2.74/6.0 or 46%) genetic testing and counseling (please see Fig. 4 for percentages of correct responses to knowledge items). However, when the item including the concept of carrier was removed, the baseline knowledge mean was 2.61 (±1.12), with a range from 0 to 5, and the post-knowledge results were 2.65 (±1.14), range 0–5. This represents a correct response rate of 52% (pre) and 53% (post). The knowledge data reflected an increase in scores for patients with low pre-knowledge scores (<3). However, those who initially scored higher (>3) maintained high scores.
Fig. 4.
Percentage of genetic knowledge items answered correctly
When the impact of genetic testing and counseling was evaluated, no significant difference in overall pre- and post-knowledge mean scores was observed whether evaluating all six knowledge items or only five of six, less the carrier item. Using the Mann–Whitney U and Kruskal–Wallis tests, no differences were found in pre- or post-knowledge levels by gender, age, education level, or level of spirituality. Furthermore, no differences in post-knowledge were found by the type of genetic transmission, the certainty of test results, or whether the results were positive or negative. At baseline, aunts and uncles differed from affected individuals, mothers, and fathers in that the former responded to fewer items correctly (p = 047). This was no longer the case after genetic testing and counseling.
Discussion
Although attitudes and knowledge towards genetic testing have been studied in many countries, only a handful of such studies have been performed in sub-Saharan Africa. Yet as genetic studies increase in number and informativity in Africa due to the greater genetic variation in African populations, the perspectives of African people themselves become increasingly important to capture.
Consistent with findings in South African and Nigerian studies, our study reveals that people in Mali are favorable towards getting tested and receiving a genetic diagnosis (de Villiers et al. 1997; Basson et al. 2007; Futter et al. 2009). As in these previous studies, patients agreed the information would be helpful to plan for the future, know their diagnosis for a potential treatment, and help their children (Basson et al. 2007; Futter et al. 2009). Also, receiving positive or negative results did not impact attitudes (Futter et al. 2009). Studies in Muslim populations likewise found favorability towards genetic testing (Yumuk et al. 2004; Raz and Schicktanz 2009). Whereas Raz and Schicktanz (2009) and Yumuk et al. (2004) administered questionnaires to a wide range of subjects, including patients, caregivers, nurses, teachers, and students, our study focused on affected individuals and their family members.
Our findings about attitudes are consistent with studies in other populations as well. Li et al. (2007) did a pre- and post-analysis of US parents’ attitudes toward genetic testing for childhood hearing loss. The pre- and post-questionnaires were not paired as in our case, but the results showed that parents of children with bilateral sensorineural hearing loss who responded to the questionnaire after genetic testing had higher testing intention, believed more strongly in the benefits of testing, were less likely to consider the deafness to be caused by non-genetic reasons, and were less likely to defer the decision to test. Affected parents were less likely than unaffected parents to consider that the deafness was caused by non-genetic reasons. On a descriptive basis, we likewise found that subjects continued to strongly favor genetic testing and counseling, that willingness to give blood, a proxy for intention to test, remained at 100%, and that fewer patients attributed causality to stress and bad luck after genetic testing and counseling. More people did attribute causality to infection after genetic testing and counseling. This may be because, for some patients in our study, test results were negative or non-conclusive. In fact, an infectious cause of disease could not be completely ruled out for spastic paraparesis, although testing for HTLV1, a virus known to be involved in spastic paraparesis, was done on three families and none had a positive serology. Thus, the favorability judged by the response to the question, “this information did not help because the disease in the family is due to an infection similar to malaria or HIV” may have been misrepresented.
The high rate of overall favorability in our sample may be a true reflection of Malians’ receptivity to genetic testing and counseling. Another possible explanation for the finding of favorable attitudes is the lack of health insurance discrimination, with the overall lack of health insurance in Mali. Health insurance discrimination has been reported as a potential reason explaining why clinical genetic testing is negatively perceived and rejected in the West (Armstrong et al. 2000; Peterson et al. 2002). Fewer Malians, albeit still the majority, were interested in receiving genetic testing and counseling for the sake of making decisions about whether or not to have future children or knowing the likelihood of transmission to future children. Studies of religious cohorts (97% of participants in this study prayed daily and 64% attended service daily or weekly) seem to indicate that religious people tend to perceive genetic testing as a tool to better prepare families rather than use that information to decide not to have children or to terminate a pregnancy (Thomas et al. 2007). However, it is important not to stereotype counselees who may be religious since many factors affect a counselee’s decision-making process (Ahmed et al. 2006).
The level of knowledge about genetic testing and counseling in the Malian sample was moderate to low, with most respondents answering slightly less than half of the knowledge questions correctly before and after testing and counseling. Genetic knowledge has been found to be low in other samples with subjects who, as in our cohort, have a lower education level (Morren et al. 2007). In particular, the term “carrier” was poorly understood, with 90% responding incorrectly to “carriers have the disease in general”. When this item was removed, subjects responded to slightly greater than half of the questions correctly both before and afterwards. Our finding is not surprising in that many of our subjects had not likely been exposed to genetics concepts before our study. Thus, the amount of information provided was new to most of our participants, and they were exposed to these new concepts in only two sessions. We found that affected individuals scored higher than the more distantly related relatives in the study. A similar trend has been reported. For example, in a study comparing genetic knowledge between subjects with Parkinson’s disease in the USA and Singapore, US patients consistently scored higher than patients from Singapore, but caregivers in both countries scored lower than patients (Tan et al. 2007). Bedouin subjects with known genetic disease in their families likewise responded to knowledge questions more accurately than those without known genetic risk (Raz and Schicktanz 2009). In our study, the knowledge difference between affected individuals and more distantly related relatives did not remain significant after genetic testing and counseling, suggesting that more distantly related relatives may have improved in knowledge.
No significant difference in overall pre- and post-knowledge mean scores was observed in the sample, although there was a trend toward increased knowledge among those who scored lowest at baseline. Often, knowledge scores increase with genetic testing and counseling, but the context must be taken into account. For example, studies done in the USA regarding genetic testing and counseling for breast cancer show increased knowledge but often include samples of highly educated Caucasian women (Green et al. 2004; Wang et al. 2005). In the majority of our sample, the level of education was primary school or below. Therefore, many subjects in our study likely received information on genetic transmission patterns for the first time. Interestingly, the question regarding consanguineous marriages was answered correctly by over 80% of the subjects, and this percentage increased after genetic testing and counseling. Since consanguineous marriages are common in some Malian ethnic groups, this suggests that genetics knowledge improves more among Malians with increased exposure and familiarity to genetic concepts, as with their Western counterparts. The idea of increased exposure to genetics concepts resulting in improved knowledge retention is further supported by studies in Africa and the Middle East (Moronkola and Fadairo 2006; Raz and Schicktanz 2009). For example, sickle cell disease is common in Nigeria, with a prevalence of 20 per 1,000 births and a carrier rate of 24% (Sickle Cell Anemia 2006). Moronkola and Fadairo (2006) reported students had a high knowledge of sickle cell disease and of their own sickle cell genotype. This may have been due to factors including the common nature of the disease and the level of the students’ education.
We found that patients and families in Mali, as elsewhere, value specific diagnostic information even if they do not have a complete understanding of the type of genetic transmission or risk (de Villiers et al. 1997; Basson et al. 2007; Etchegary et al. 2010). Regardless of knowledge of the exact transmission pattern, the patients often left with a specific diagnosis and a more general understanding of their disorder. This is an important role of genetic counseling, as the patients wanted to know how this would affect their family, and they received helpful information regarding what to anticipate (prognosis). Thus, although the questionnaire indicated limited knowledge of specific transmission patterns and what it means to be a carrier, which were new concepts for the patients, the majority understood that the condition was in general passed down in families and wanted as much information as possible. This is similar to the situation for many North American patients who may not have a complete understanding of transmission or carrier status, but who find genetic testing useful for diagnosis and prognosis (Etchegary et al. 2010).
Concerns about confidentiality were expressed by the majority of our subjects, revealing that patients likely understood the sensitive nature of a genetic diagnosis. Studies among African American women show that, although women had positive attitudes about testing, they were concerned about risks and limitations of testing (Hughes et al. 2003; Kessler et al. 2005). Similarly, although patients in the USA and Singapore had positive attitudes toward testing, they expressed concerns about discrimination (Tan et al. 2007).
A qualitative study would complement our study and elucidate some of our findings. For instance, it would be helpful to understand why more distant relatives showed more positive attitudes. Such a study might also support our anecdotal findings: in spending time with several subjects, we found, similarly to findings in a study of hereditary non-polyposis colorectal cancer in the Navajo, some participants completely accepted a genetic explanation for their disease, while others believed in more traditional concepts such as curses (Lynch et al. 1996). Due to the novelty of the information for many of our subjects, they may have had a difficult time separating an ultimate origin of the disease, such as God or fate, from a more immediate biological cause. Other anecdotal benefits identified that require further research included (1) relief for some that the disease was not a curse or a punishment in response to a bad action and (2) elimination of the need to spend the family’s money on unnecessary therapies after receiving a genetic diagnosis.
Several limitations are noted. Before data collection, we expected a more homogenous group of subjects with hereditary neurological diseases of mainly autosomal recessive inheritance. The large variability of conditions and thus transmission patterns (recessive, dominant, X-linked, mitochondrial, and unknown), however, presented a challenge for the genetic counselor to cover the same material for each family and for the knowledge questions to broadly represent an understanding of heredity. The genetic counselor also noted being less able to rely on nonverbal cues during counseling due to the language difference. Time between original questionnaires and post-questionnaires also varied depending on the length of time needed for molecular testing results. Due to the small sample size, it is difficult to discern whether there was truly no difference in pre- and post-attitudes and knowledge or whether the difference was too small to detect. The recruited patients were known patients of MT rather than randomly selected individuals; this selection bias may have contributed to the positive attitudes toward genetic testing and counseling as the patients in the sample consisted of people who may favor medical interventions. Potential response bias might be due to the fact that Malians felt they had to respond affirmatively when a physician was in the room based on the long-term relationship between the patient and the physician over the course of the disease. Finally, these findings are not generalizable to subjects with other conditions or in other populations.
Despite these limitations, our study contributes important findings to the literature on genetic testing and counseling in developing countries, particularly Mali. We discovered that Malians of all education levels had overall positive attitudes toward genetic testing and counseling, both before and afterwards. Overall attitudes and knowledge did not change significantly before and after the process of genetic testing and counseling. The majority of subjects also expressed a desire to protect their confidentiality, which also did not change. Further research, including a larger sample size and specific hereditary disorders, especially for the knowledge questions, would be needed to further substantiate these findings.
The genetic testing performed in this study was primarily for diagnostic purposes. We believe that people in Mali deserve access to testing if it is available. Moreover, having a genetic diagnosis may help them cope with the stigma of their disease by providing an explanation other than bad fate. The ultimate goal is to lessen health care disparities between developed and developing countries, and our work is an important step in this direction.
Acknowledgments
The contents of this publication are solely the responsibility of the authors and do not necessarily represent the official view of the National Institutes of Health. We thank the patients and families for their participation and patience and express our gratitude to the many experts who contributed invaluable insight, including Dr. Gail Geller, Dr. Peter Winch, Dr. Marie Diener-West, Dr. Elizabeth Thomson, Dr. Lori Leonard, Mr. Donald Hadley, Dr. Benjamin Wilfond, and Dr. Kevin Flanigan. We also thank the Medical School and the Hospital of Point “G”, Bamako, Mali. The work was supported by funds from the NINDS and NINR intramural programs.
Conflicts of interest The authors declare that they have no conflict of interest.
References
- Ahmed S, Green JM, Hewison J. Attitudes towards prenatal diagnosis and termination of pregnancy for thalassaemia in pregnant Pakistani women in the north of England. Prenat Diagn. 2006;26(3):248–257. doi: 10.1002/pd.1391. [DOI] [PubMed] [Google Scholar]
- Armstrong K, Calzone K, Stopfer J, Fitzgerald G, Coyne J, Weber B. Factors associated with decisions about clinical BRCA1/2 testing. Cancer Epidemiol Biomarkers Prev. 2000;9(11):1251–1254. [PubMed] [Google Scholar]
- Basson F, Futter MJ, Greenberg J. Qualitative research methodology in the exploration of patients’ perceptions of participating in a genetic research program. Ophthalmic Genet. 2007;28(3):143–149. doi: 10.1080/13816810701356627. [DOI] [PubMed] [Google Scholar]
- Bernhardt BA, Chase GA, Faden RR, Geller G, Hofman KJ, Tambor ES, et al. Educating patients about cystic fibrosis carrier screening in a primary care setting. Arch Fam Med. 1996;5(6):336–340. doi: 10.1001/archfami.5.6.336. [DOI] [PubMed] [Google Scholar]
- Berth H, Balck F, Dinkel A. Attitudes toward genetic testing in patients at risk for HNPCC/FAP and the German population. Genet Test. 2002;6(4):273–280. doi: 10.1089/10906570260471804. [DOI] [PubMed] [Google Scholar]
- Bodamer OA, Hoffmann GF, Lindner M. Expanded newborn screening in Europe 2007. J Inherit Metab Dis. 2007;30(4):439–444. doi: 10.1007/s10545-007-0666-z. [DOI] [PubMed] [Google Scholar]
- Carreiro-Lewandowski E. Newborn screening: an overview. Clin Lab Sci. 2002;15(4):229–238. [PubMed] [Google Scholar]
- Villiers C, Weskamp K, Bryer A. The sword of Damocles: the psychosocial impact of familial spinocerebellar ataxia in South Africa. Am J Med Genet. 1997;74(3):270–274. doi: 10.1002/(SICI)1096-8628(19970531)74:3<270::AID-AJMG6>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- Ebomoyi E. Knowledge and attitude towards genetic screening for sickle cell disease. Hygie. 1988;7(1):33–38. [PubMed] [Google Scholar]
- Etchegary H, Cappelli M, Potter B, Vloet M, Graham I, Walker M, et al. Attitude and knowledge about genetics and genetic testing. Public Health Genomics. 2010;13(2):80–88. doi: 10.1159/000220034. [DOI] [PubMed] [Google Scholar]
- Futter MJ, Heckmann JM, Greenberg LJ. Predictive testing for Huntington disease in a developing country. Clin Genet. 2009;75(1):92–97. doi: 10.1111/j.1399-0004.2008.01044.x. [DOI] [PubMed] [Google Scholar]
- Green MJ, Peterson SK, Baker MW, Harper GR, Friedman LC, Rubinstein WS, et al. Effect of a computer-based decision aid on knowledge, perceptions, and intentions about genetic testing for breast cancer susceptibility: a randomized controlled trial. JAMA, J Am Med Assoc. 2004;292(4):442–452. doi: 10.1001/jama.292.4.442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes C, Fasaye GA, LaSalle VH, Finch C. Sociocultural influences on participation in genetic risk assessment and testing among African American women. Patient Educ Couns. 2003;51(2):107–114. doi: 10.1016/S0738-3991(02)00179-9. [DOI] [PubMed] [Google Scholar]
- Kessler L, Collier A, Brewster K, Smith C, Weathers B, Wileyto EP, et al. Attitudes about genetic testing and genetic testing intentions in African American women at increased risk for hereditary breast cancer. Genet Med. 2005;7(4):230–238. doi: 10.1097/01.GIM.0000159901.98315.FE. [DOI] [PubMed] [Google Scholar]
- Leplege A, Schemann JF, Diakite B, Toure O, Ecosse E, Jaffre Y, et al. A new condition specific quality of life measure for the blind and the partially sighted in sub-Saharan Africa, the IOTAQOL: methodological aspects of the development procedure. Qual Life Res. 2006;15(8):1373–1382. doi: 10.1007/s11136-006-0023-y. [DOI] [PubMed] [Google Scholar]
- Lerman C, Biesecker B, Benkendorf JL, Kerner J, Gomez-Caminero A, Hughes C, et al. Controlled trial of pretest education approaches to enhance informed decision-making for BRCA1 gene testing. J Natl Cancer Inst. 1997;89(2):148–157. doi: 10.1093/jnci/89.2.148. [DOI] [PubMed] [Google Scholar]
- Li Y, Steinberg AG, Bain L, Yaeger D, Bieler A, Ewing R, et al. Assessing parental attitudes toward genetic testing for childhood hearing loss: before and after genetic consultation. Am J Med Genet A. 2007;143A(14):1546–1553. doi: 10.1002/ajmg.a.31730. [DOI] [PubMed] [Google Scholar]
- Lynch HT, Drouhard T, Vasen HF, Cavalieri J, Lynch J, Nord S, et al. Genetic counseling in a Navajo hereditary nonpolyposis colorectal cancer kindred. Cancer. 1996;77(1):30–35. doi: 10.1002/(SICI)1097-0142(19960101)77:1<30::AID-CNCR7>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- McLaren CE, Barton JC, Adams PC, Harris EL, Acton RT, Press N, et al. Hemochromatosis and iron overload screening (HEIRS) study design for an evaluation of 100,000 primary care-based adults. Am J Med Sci. 2003;325(2):53–62. doi: 10.1097/00000441-200302000-00001. [DOI] [PubMed] [Google Scholar]
- Modell B, Darr A. Science and society: genetic counseling and customary consanguineous marriage. Nat Rev Genet. 2002;3(3):225–229. doi: 10.1038/nrg754. [DOI] [PubMed] [Google Scholar]
- Moronkola OA, Fadairo RA. University students in Nigeria: knowledge, attitude toward sickle cell disease, and genetic counseling before marriage. Int Q Community Health Educ. 2006;26(1):85–93. doi: 10.2190/JN25-4353-75PK-3733. [DOI] [PubMed] [Google Scholar]
- Morren M, Rijken M, Baanders AN, Bensing J. Perceived genetic knowledge, attitudes towards genetic testing, and the relationship between these among patients with a chronic disease. Patient Educ Couns. 2007;65(2):197–204. doi: 10.1016/j.pec.2006.07.005. [DOI] [PubMed] [Google Scholar]
- Penn C, Watermeyer J, MacDonald C, Moabelo C. Grandmothers as gems of genetic wisdom: exploring South African traditional beliefs about the causes of childhood genetic disorders. J Genet Couns. 2010;19(1):9–21. doi: 10.1007/s10897-009-9252-x. [DOI] [PubMed] [Google Scholar]
- Peterson EA, Milliron KJ, Lewis KE, Goold SD, Merajver SD. Health insurance and discrimination concerns and BRCA1/2 testing in a clinic population. Cancer Epidemiol Biomarkers Prev. 2002;11(1):79–87. [PubMed] [Google Scholar]
- Pieterse A, Dulmen S, Ausems M, Schoemaker A, Beemer F, Bensing J. QUOTE-gene(ca): development of a counselee-centered instrument to measure needs and preferences in genetic counseling for hereditary cancer. Psychooncology. 2005;14(5):361–375. doi: 10.1002/pon.853. [DOI] [PubMed] [Google Scholar]
- Raz AE, Schicktanz S. Diversity and uniformity in genetic responsibility: moral attitudes of patients, relatives and lay people in Germany and Israel. Med Health Care Philos. 2009;12(4):433–442. doi: 10.1007/s11019-009-9215-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rew L, Mackert M, Bonevac D. A systematic review of literature about the genetic testing of adolescents. J Spec Pediatr Nurs. 2009;14(4):284–294. doi: 10.1111/j.1744-6155.2009.00210.x. [DOI] [PubMed] [Google Scholar]
- Rose A, Peters N, Shea JA, Armstrong K. The association between knowledge and attitudes about genetic testing for cancer risk in the United States. J Health Commun. 2005;10(4):309–321. doi: 10.1080/10810730590950039. [DOI] [PubMed] [Google Scholar]
- Sickle Cell Anaemia. 2006. Report by the Secretariat. World Health Organization. 59th World Health Assembly.
- Tan EK, Lee J, Hunter C, Shinawi L, Fook-Chong S, Jankovic J. Comparing knowledge and attitudes towards genetic testing in Parkinson’s disease in an American and Asian population. J Neurol Sci. 2007;252(2):113–120. doi: 10.1016/j.jns.2006.10.016. [DOI] [PubMed] [Google Scholar]
- Thomas S, Herbert D, Street A, Barnes C, Boal J, Komesaroff P. Attitudes towards and beliefs about genetic testing in the haemophilia community: a qualitative study. Haemophilia. 2007;13(5):633–641. doi: 10.1111/j.1365-2516.2007.01454.x. [DOI] [PubMed] [Google Scholar]
- Waltz CF, Strickland OL, Lenz ER (eds) (2004) Measurement in nursing and health research, 3rd edn. Springer, New York
- Wang C, Gonzalez R, Milliron KJ, Strecher VJ, Merajver SD. Genetic counseling for BRCA1/2: a randomized controlled trial of two strategies to facilitate the education and counseling process. Am J Med Genet A. 2005;134A(1):66–73. doi: 10.1002/ajmg.a.30577. [DOI] [PubMed] [Google Scholar]
- Yumuk PF, Gumus M, Ekenel M, Basaran G, Aliustaoglu M, Topal A, et al. Attitude toward genetic testing for cancer risk in Istanbul. Genet Test. 2004;8(2):169–173. doi: 10.1089/gte.2004.8.169. [DOI] [PubMed] [Google Scholar]