Abstract
BACKGROUND AND PURPOSE
Cyclical phosphorylation and dephosphorylation of a key residue within the C-terminal domain of the activated type 5 metabotropic glutamate (mGlu5) receptor is believed to cause the synchronous, oscillatory changes in inositol 1,4,5-trisphosphate and Ca2+ levels observed in a variety of cell types. Here, we have attempted to better define the kinase and phosphatase enzymes involved in this modulation.
EXPERIMENTAL APPROACH
Ca2+ and [3H]inositol phosphate ([3H]IPx) measurements in astrocyte preparations have been used to evaluate the effects of pharmacological inhibition of protein kinase C (PKC) and protein phosphatase activities and small interfering RNA-mediated specific PKC isoenzymic knock-down on mGlu5 receptor signalling.
KEY RESULTS
Ca2+ oscillation frequency or [3H]IPx accumulation in astrocytes stimulated by mGlu5 receptors, was concentration-dependently decreased by protein phosphatase-1/2A inhibition or by PKC activation. PKC inhibition also increased [3H]IPx accumulation two- to threefold and changed the Ca2+ response into a peak-plateau response. However, selective inhibition of conventional PKC isoenzymes or preventing changes in [Ca2+]i concentration by BAPTA-AM loading was without effect on mGlu5 receptor-stimulated [3H]IPx accumulation. Selective knock-down of PKCδ was without effect on glutamate-stimulated Ca2+ responses; however, selective PKCε knock-down in astrocytes changed Ca2+ responses from oscillatory into peak-plateau type.
CONCLUSION AND IMPLICATIONS
These data confirm the acute regulation of mGlu5 receptor signalling by protein kinases and protein phosphatases and provide novel data pinpointing the isoenzymic dependence of this regulation in the native mGlu5 receptor-expressing rat cortical astrocyte. These data also highlight a potential alternative mechanism by which mGlu5 receptor signalling might be therapeutically manipulated.
Keywords: metabotropic glutamate receptor, mGlu5, protein kinase C, protein phosphatase, Ca2+ oscillation, phosphoinositide hydrolysis, astrocyte (astroglia)
Introduction
Astroglia were originally considered to play a solely homeostatic role in the brain, providing neurones with the correct environment in which to fulfil their many roles. However, the idea that astrocytes play a crucial part in signalling within the CNS has now emerged, with evidence to show that this type of glial cell can communicate with other astrocytes and also with neurones, giving rise to the concept of the ‘tripartite synapse’ (Pasti et al., 1997; Allen and Barres, 2009). Astrocytes express an array of G protein-coupled receptors (GPCRs), including adrenoceptors and muscarinic, 5-hydroxytryptaminergic, glutamatergic, purinergic and GABAergic receptors (Porter and McCarthy, 1997; Agulhon et al., 2008).
Glutamate is the major excitatory neurotransmitter in the CNS, and its effects are mediated by activation of ionotropic and metabotropic glutamate (mGlu) receptors. Eight receptor subtypes constitute the mGlu1–8 receptor family (Conn and Pin, 1997; receptor nomenclature follows Alexander et al., 2009), with a number of studies providing evidence for mGlu3 and mGlu5 receptors being the major subtypes expressed in glial cells (Miller et al., 1995; Cai et al., 2000; Aronica et al., 2003). mGlu5 receptors couple primarily via Gq/11 proteins to stimulate increases in phospholipase C (PLC) activity leading to characteristic oscillatory changes in intracellular Ca2+ concentration ([Ca2+]i) via a mechanism dependent on protein kinase C (PKC) (Kawabata et al., 1996; Nakahara et al., 1997). The mechanism by which mGlu5 receptor activation leads to the generation of Ca2+ oscillations has been investigated, predominantly in recombinant cell lines, and is termed ‘dynamic uncoupling’ (Nash et al., 2002). The process of dynamic uncoupling involves rapid and repeated cycles of phosphorylation and dephosphorylation of Ser839 (Kim et al., 2005). Phosphorylation of this residue by PKC is thought to uncouple the receptor from its G protein terminating PLC activation, with rapid reactivation occurring through Ser839 dephosphorylation by an as yet poorly defined protein phosphatase activity (Nakahara et al., 1997). Thus, repeated cycles of phosphorylation and dephosphorylation of the mGlu5 receptor following agonist activation result in the characteristic Ca2+ oscillatory responses observed (Nash et al., 2001, 2002; Atkinson et al., 2006).
Protein kinase C isoenzymes have been classified into three subfamilies: conventional PKCs (cPKC, e.g. α, β and γ) which are dependent for activation on Ca2+ and diacylglycerol (DAG), novel PKCs (nPKC, e.g. δ, ε, η and θ) which are independent of Ca2+, but dependent on DAG, and atypical PKCs (aPKC, e.g. ζ and λ) which are independent of both Ca2+ and DAG (Parker and Murray-Rust, 2004). The precise PKC isoenzymes involved in mGlu5 receptor–Ca2+ dynamic uncoupling are not yet specified and there is evidence for the expression in astrocytes of an array of PKC isoenzymes, including representatives of the conventional, novel and atypical subfamilies (Rudkouskaya et al., 2008). Codazzi et al. (2001) have provided evidence for the involvement of conventional PKC isoenzymes in the generation of glutamate-induced Ca2+ oscillations. This group also showed that the oscillatory increases in intracellular Ca2+ and DAG led to conventional PKCs translocating from the cytoplasm to the plasma membrane. In contrast to these findings, in other studies, Ca2+-independent isoenzymes of PKC have been reported to phosphorylate the mGlu5 receptor to uncouple it from PLC activity (Uchino et al., 2004). While the mechanism(s) underlying mGlu5 receptor-induced Ca2+ oscillations has been investigated quite widely in recombinant systems without consensus, little has been carried out to elucidate this fundamental mechanism in a cell type endogenously expressing the mGlu5 receptor. In the present study we provide new pharmacological and molecular genetic evidence for the involvement of dynamic uncoupling in the generation of Ca2+ oscillations in rat cortical astrocytes and define the relative isoenzymic kinase(s) and phosphatase(s) activities involved in this cycle.
Methods
Astrocyte preparation and culture
All animal care and experimental procedures complied with the UK Animal (Scientific Procedures) Act 1986 and conformed to the Guide for Care and Use of Laboratory Animals US (NIH Publication No. 85-23, revised 1996). Cortical astrocytes were prepared as described previously (Bradley et al., 2009). In brief, Wistar rats (1–2 days of age) were decapitated and the cortices removed. Tissues were digested with trypsin [bovine pancreatic; 0.025% (w/v)] and dissociated by trituration with fire-polished glass pipettes. The cell suspension was centrifuged (170 xg; 8 min), and pellet resuspended in Dulbecco's minimum essential medium (DMEM) containing GlutaMAX-1 with sodium pyruvate, 4500 mg·L−1 glucose, 15% heat-inactivated FBS, 2.5 µg·mL−1 amphotericin B and 0.1 µg·mL−1 gentamicin. Cells were seeded into poly-d-lysine-coated cell culture flasks and incubated at 37°C in a humidified air: 5% CO2 atmosphere for 7 days, with medium being replaced after 4 days. On the 7th day in vitro (DIV 7), medium was replaced again and flasks were transferred to a shaking incubator overnight (37°C; 320 r.p.m.). On the following day (DIV 8), cells were washed twice with phosphate-buffered saline (PBS without Ca2+/Mg2+) and harvested with 0.25% (w/v) trypsin, 0.02% (w/v) EDTA. Cells were subsequently seeded onto pre-coated poly-D-lysine tissue culture plates for experiments. After 24 h (DIV 9), medium was replaced with DMEM containing GlutaMAX-1 with sodium pyruvate, 4500 mg·L−1 glucose, 2.5 µg·mL−1 amphotericin B and 0.1 µg·mL−1 gentamicin and G5 supplement. Cells were used for experiments at DIV 11–13.
Immunocytochemistry
Rat cortical astrocytes were seeded at 3 × 105 cells per well onto 22 mm borosilicate coverslips in culture medium and the following day medium was changed to medium containing G5 supplement (where necessary). After 48 h medium was removed, cell monolayers were washed with PBS (without Ca2+/Mg2+) and fixed with 4% paraformaldehyde in PBS for 10 min at room temperature. Cells were briefly washed in PBS and permeabilized in PBS-Triton X-100 (0.01%) containing 1% BSA, for 1 h at room temperature. After three 5 min washes, cells were incubated with a 1:100 dilution of an mGlu5 receptor antibody in PBS overnight at 4°C. The mGlu5 receptor antibody was washed off with PBS and cells were incubated with a 1:100 dilution of an antibody for glial fibrillary acidic protein (GFAP) in PBS at room temperature for 1 h. Following three further 5 min PBS wash steps, cells were incubated at room temperature for 1 h with a 1:400 dilution of secondary antibodies (Alexa Fluor® 488 and Alexa Fluor® 647). Coverslips were washed in PBS and mounted onto glass microscope using Slowfade® gold antifade reagent with 4′,6-diamidino-2-phenylindole (DAPI). Coverslips were maintained at 4°C in darkness, until analysis using an Olympus FV500 confocal microscope (Olympus, Tokyo, Japan).
Western blot analysis
Rat cortical astrocytes were seeded in poly-D-lysine-coated 60 mm dishes in culture medium and the following day medium was changed to medium containing G5 supplement (where necessary). After the indicated time periods, plates were placed on ice and washed twice with ice-cold PBS and cells were solubilized by addition of 150 µL of lysis buffer [20 mM Tris/HCl, pH 7.4, 1% Triton X-100, 10% (v/v) glycerol, 137 mM NaCl, 2 mM EDTA and a 1:100 dilution of a protease inhibitor cocktail]. Samples were collected and centrifuged at 21 000× g for 10 min at 4°C. Aliquots of the supernatant (80 µL) were extracted and added to 20 µL of 5× Laemmli sample buffer [300 mM Tris/HCl, pH 6.8, 15% (w/v) SDS, 50% (v/v) glycerol and 50 mM dithiothreitol (DTT)]. Samples were separated by 12.5% SDS-PAGE using the Bio-Rad mini-gel system. Electrotransfer to nitrocellulose membranes was performed using the Bio-Rad semi-dry blotting apparatus in transfer buffer (48 mM Tris, 39 mM glycine, 1.3 mM SDS, 20% methanol). Non-specific binding sites were blocked by incubation of the nitrocellulose membrane in 5% non-fat milk powder in TBST buffer (50 mM Tris, 150 mM NaCl, 0.1% Tween-20) for 1 h at room temperature. Membranes were subsequently incubated overnight at 4°C with primary antibody (dilutions specified in figure legends). Blots were then washed three times for 10 min in TBST and incubated at room temperature for 60 min with the secondary antibody conjugated to horseradish peroxidase (1:1000 dilution in 5% milk/TBST). Following this, blots were washed three times for 10 min in TBST before chemiluminescence detection using ECL reagent and exposure to HyperFilm.
Total [3H]inositol phosphate accumulation assay
Rat cortical astrocytes were seeded at 150 × 103 cells per well in poly-d-lysine-coated 24-well plates. The following day, cells were incubated in fresh medium containing G5 supplement and 2.5 µCi·mL−1[3H]inositol for 48 h. Cell monolayers were washed twice and incubated in Krebs-Henseleit buffer (KHB; composition – 118.6 mM NaCl, 4.7 mM KCl, 4.2 mM NaHCO3, 1.3 mM CaCl2, 1.2 mM MgSO4, 1.2 mM KH2PO4, 11.7 mM glucose, 10 mM HEPES, pH 7.4) containing glutamic-pyruvic transaminase (GPT; 3 U·mL−1) and pyruvate (5 mM) for 25 min at 37°C. LiCl (10 mM) was added for a further 20 min prior to agonist incubations for 20 min. Reactions were terminated by aspiration of buffer followed by rapid addition of 500 µL of ice-cold trichloroacetic acid (0.5 M). Samples were extracted from each well and mixed with 100 µL of EDTA (10 mM, pH 7.0) and 500 µL of a 1:1 mixture of tri-n-octylamine and 1,1,2-trichlorofluoroethane. Samples were centrifuged at 21 000× g. for 2 min and 400 µL of the upper aqueous phase was transferred into fresh microfuge tubes containing 100 µL of NaHCO3 (62.5 mM). The [3H]inositol mono-, bis- and trisphosphate fraction ([3H]IPx) was recovered by anion-exchange chromatography on Dowex AG1-X8 formate columns and radioactivity was determined by liquid scintillation counting.
Single-cell intracellular [Ca2+]i assay
Rat cortical astrocytes were seeded onto 22 mm borosilicate coverslips at 3 × 105 cells per well and grown to approximately 80% confluency in medium containing G5 supplement. Cells were loaded with Fura-2 AM (4 µM) in KHB containing pluronic acid F1272 (0.36 mg·mL−1) for 45–60 min at room temperature. Coverslips were then transferred to the stage of a Nikon Diaphot inverted epifluorescence microscope, with an oil immersion objective (×40) and a SpectraMASTER II module (PerkinElmer Life Sciences). Cells were excited at wavelengths of 340 and 380 nm using a SpectraMASTER II monochromator and emission was recorded at wavelengths above 520 nm. The ratio of fluorescence intensities at these wavelengths is given as an index of [Ca2+]i. All experiments were performed at 37°C; drug additions were made via a perfusion line.
siRNA knock-down of endogenous PKCδ or PKCε levels
Expression of PKC isoenzymes was determined using standard immunoblotting protocols as described above using specific antibodies against PKCδ and PKCε. Rat cortical astrocytes (2 × 106) were transfected with 0.5 or 1.5 µg of negative-control, anti-PKCδ or anti-PKCε siRNAs. The anti-PKCδ siRNAs used for endogenous PKCδ knock-down were PKCδ siRNA 1 (#s139243; 5′-GAUUCAAGGUCUAUAACUAtt-3′) and PKCδ siRNA 2 (#s139244; 5′-GCAAGGUACUGCUUGCAGAtt-3′). Anti-PKCε siRNAs used for knock-down of endogenous PKCε were PKCε siRNA 1 (#s218083; 5′-AGCCGAGAAUUAAAACCAAtt-3′) and PKCε siRNA 3 (#s131201; 5′-CUCUAUUGCUGCUUCCAGAtt-3′). Following nucleofection, cells were plated onto poly-d-lysine-coated plates in astrocyte culture medium, which was replaced 24 h later by a similar medium containing G5 supplement. Cells were lysed 48 h later, and expression of PKCδ or PKCε was assessed by immunoblotting (see above). PKC expression was quantified using the GeneGnome image analysis system (Syngene, Cambridge, UK).
Data analysis
Concentration–response relationships were analysed by non-linear regression using GraphPad Prism 5.0 software (San Diego, CA, USA). For statistical tests, where only two datasets were being compared, an unpaired Student's t-test (two-tailed) was used, where P < 0.05 was deemed statistically significant. Where more than two datasets were compared, one-way or two-way analysis of variance (anova) tests were used with P < 0.05 being accepted as significantly different. anova tests were followed by the Bonferroni's or Dunnett's post hoc test. All statistical analyses were performed using GraphPad Prism 5.0 software.
Materials
Tissue culture reagents, G5 supplement and Fura-2 AM were from Invitrogen (Paisley, UK). Myo-[3H]inositol was from GE Healthcare (Chalfont St. Giles, UK). Glutamate, staurosporine, calyculin A, cantharidin (S)-3,5-dihydroxyphenylglycine (DHPG) and 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) were obtained from Tocris Cookson Ltd. (Bristol, UK). Phorbol 12,13-dibutyrate (PDBu) and the antibody raised against glial fibrillary acidic protein (GFAP) were from Sigma-Aldrich Chemical Co. (Poole, UK). Ro 31-8220, Gö6976 and myristoylated PKC 20-28 were obtained from Merck Biosciences (Nottingham, UK). Antibody raised against the C-terminus of mGlu5 receptor was obtained from Millipore (Durham, UK). Antibodies raised against specific PKC isoenzymes were purchased from BD Biosciences (Oxford, UK). Silencer® pre-designed small interfering (si)RNA targeted to PKCδ and PKCε and control siRNA were purchased from Ambion (TX, USA). Rat astrocyte nucleofector® kit was from Lonza (Wokingham, UK). All other chemicals and reagents were purchased from Sigma Chemical Co. (Poole, Dorset, UK).
Results
mGlu5 receptor expression and function in rat cerebrocortical astrocytes
Expression of the mGlu5 receptor has been widely reported in cortical astrocytes (Miller et al., 1995). In our experiments, expression of mGlu5 receptors was detectable but low in DIV 7 glial preparations. Following overnight shaking to dissociate non-astrocyte cell types, G5 growth factor supplement was added and mGlu5 receptor immunoreactivity increased over the subsequent 48–96 h; in contrast, mGlu1 receptor immunoreactivity was not detectable (data not shown). Immunocytochemistry showed that astrocytes have a typically stellate morphology and mGlu5 receptor expression was colocalized in astrocytic processes with GFAP (Figure 1A).
Figure 1.
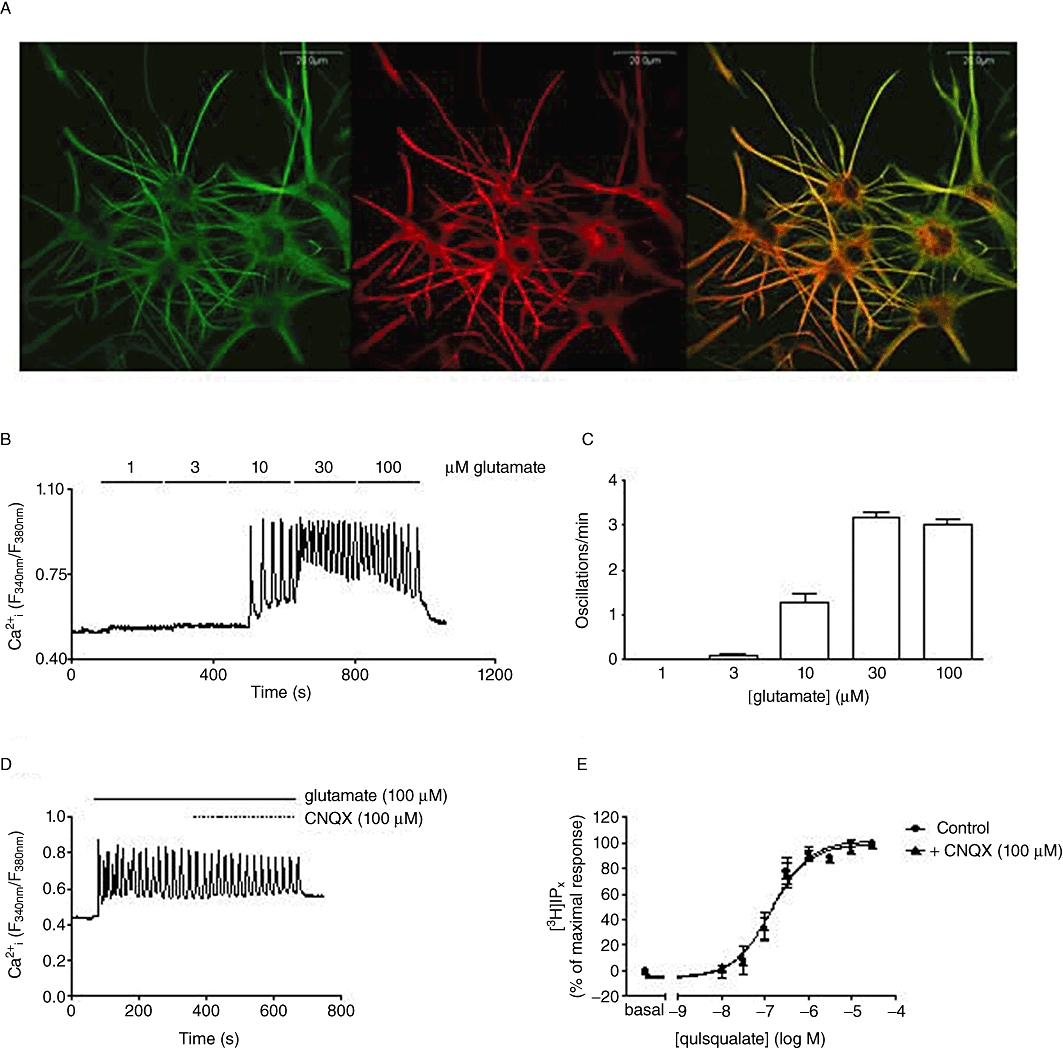
Characterization of mGlu5 receptor expression and signalling in rat cortical astrocytes. Detection of mGlu5 receptor expression in cultured rat cortical astrocytes by immunocytochemistry (A). Double immunostaining was performed, using antibodies raised against the mGlu5 receptor (left panel) or GFAP (centre panel). Image overlay is shown in the right panel. (B) Representative trace showing the effects of increasing glutamate concentration on single-cell Ca2+ oscillation frequency in rat cortical astrocytes. Mean data (±SEM for at least 30 cells over at least three separate experiments), showing the number of oscillations in a 3 min period in astrocytes stimulated incrementally with increasing concentrations of glutamate, are shown in panel C. Panels D and E show the effects of the AMPA receptor inhibitor, CNQX, on agonist-stimulated Ca2+ responses and [3H]IPx accumulations in rat cortical astrocytes. (D) Representative trace showing Ca2+ oscillations elicited by glutamate (100 µM) perfusion for 5 min followed by co-perfusion of glutamate (100 µM) with CNQX (100 µM). Data are representative of at least 50 cells over three separate experiments. (E) Concentration–response curves showing quisqualate-stimulated [3H]IPx accumulations in cells pre-incubated in the absence or presence of CNQX (100 µM; 15 min): pEC50 (M) values (mean ± SEM; three separate experiments performed in duplicate) were 6.86 ± 0.13 and 6.84 ± 0.13 in the absence and presence of CNQX respectively.
Stimulation of the mGlu5 receptor is known to induce robust oscillatory changes in [Ca2+]i in astrocytes (Nakahara et al., 1997). At low concentrations of glutamate (3–10 µM) low frequency (∼1 oscillation per minute), baseline Ca2+ oscillations were observed (Figure 1B,C), whereas at maximally effective glutamate concentrations (≥30 µM) Ca2+ oscillations on a raised baseline were observed with frequencies of approximately three oscillations per minute (Figure 1B,C). Neither single-cell Ca2+ responses to glutamate (Figure 1D), nor [3H]IPx responses to quisqualate (Figure 1E) were affected by the presence of the AMPA/kainate receptor antagonist, CNQX.
Protein phosphatase regulation of mGlu5 receptor-mediated Ca2+ and [3H]IPx responses
We explored the effects of two protein phosphatase inhibitors, calyculin A and cantharidin, which are reported to inhibit PP1 and PP2A activities. Both calyculin A (100 nM; 5 min; Figure 2A,C) and cantharidin (100 µM; 15 min; Figure 2B,D) caused marked reductions in the Ca2+ oscillation frequency stimulated by glutamate (100 µM). Previously, we have undertaken analyses of Ca2+ signalling behaviours in CHO-lac-mGlu5a cells (Atkinson et al., 2006; Bradley et al., 2009) classifying responses into four categories [non-responders (NR); single peak (SP), oscillatory (OS), peak-and-plateau (PP)]. Here, we undertook similar analyses to assess the effects of protein phosphatase inhibition on the distribution of single-cell Ca2+ responses between these types of response. Following treatment with calyculin A and cantharidin, the number of cells that responded with an oscillatory (OS) Ca2+ pattern decreased, while the number of cells that either failed to respond (NR) or responded with only a single peak (SP) response increased (Figure 2E). In addition, Ca2+ oscillation frequency was markedly decreased in the subpopulation of astrocytes (≥80%) initially exhibiting this pattern of response (Figure 2F).
Figure 2.
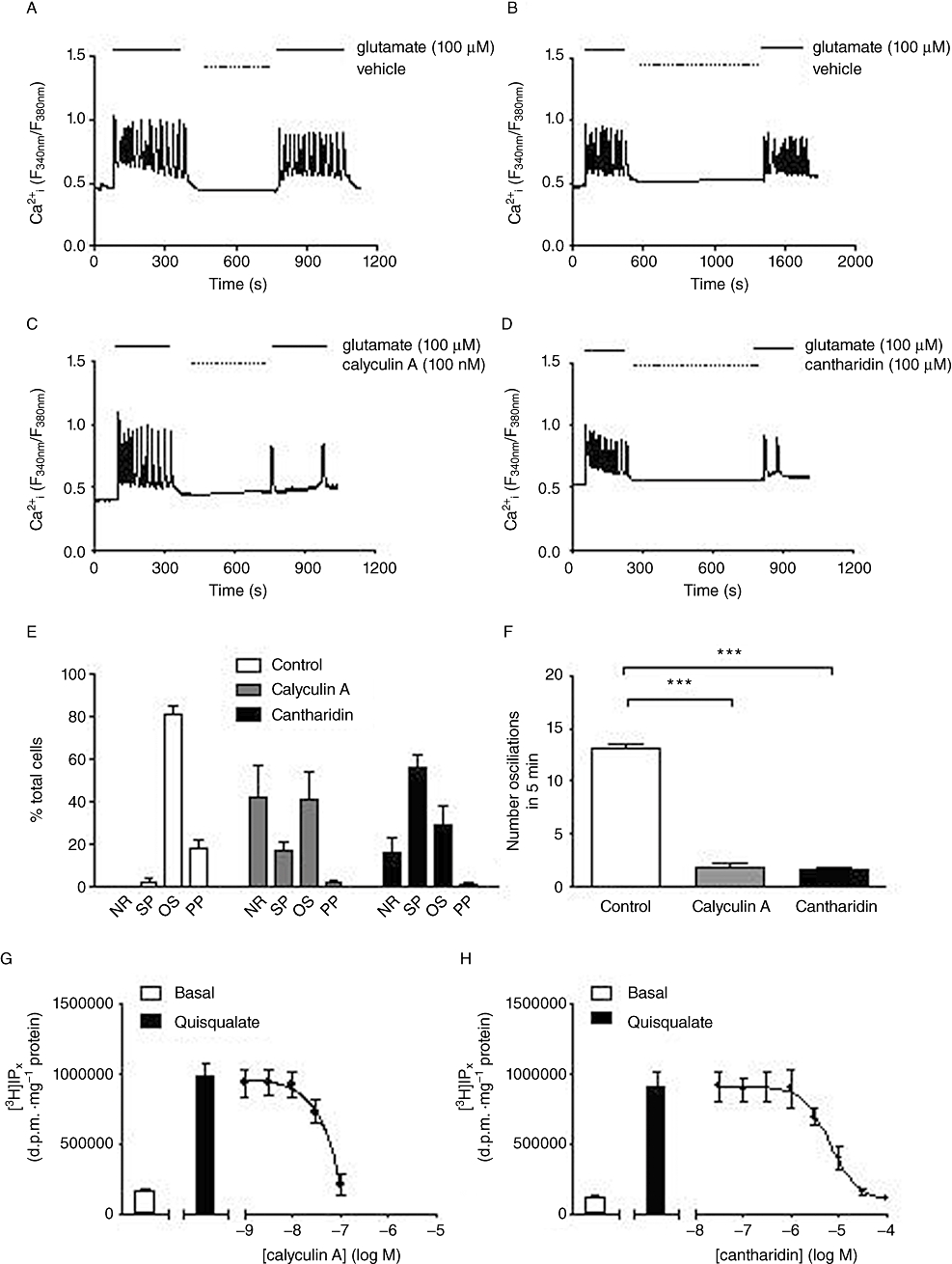
Effects of protein phosphatase inhibition on agonist-stimulated single-cell Ca2+ responses and [3H]IPx accumulations in rat cortical astrocytes. Representative traces showing the response of single astrocytes to perfusion with glutamate (100 µM) for 5 min followed immediately by the indicated incubation period with vehicle (A, B), 100 nM calyculin A (C) or 100 µM cantharidin (D), followed by a second glutamate (100 µM) perfusion period for 5 min. Data were analysed to assess the effects of each inhibitor on the distribution of glutamate-stimulated Ca2+ responses between no-response (NR), single-peak (SP), oscillatory (OS) or peak-plateau (PP) behaviours (E), or for effects on Ca2+ oscillation frequency (F). Data are shown as means ± SEM for least 20 individual cells recorded over at least three separate experiments. ***P < 0.001, significantly different as shown (unpaired Student's t-test). Panels G and H show basal and quisqualate (10 µM)-stimulated [3H]IPx accumulations in astrocytes (means ± SEM for three to four separate experiments performed in duplicate) following pre-incubation with various concentrations of calyculin A (for 5 min, G) or cantharidin (for 15 min, H). The mean pIC50 (M) value for cantharidin inhibition of the quisqualate-stimulated response was 5.15 ± 0.09; a pIC50 value was not determined for calyculin A; however, a concentration of 100 nM was sufficient to cause complete inhibition of the agonist-stimulated response.
Pre-incubation of [3H]inositol-labelled astrocytes with calyculin A (100 nM; 5 min) or cantharidin (100 µM; 15 min) caused concentration-dependent decreases in [3H]IPx accumulation stimulated by quisqualate (10 µM; Figure 2G,H). In contrast, pre-incubation with the calcineurin inhibitor, FK506 (10 µM; 30 min), had no significant effects on [3H]IPx accumulation stimulated by quisqualate (10 µM; data not shown).
Effects of altering PKC activity on mGlu5 receptor-mediated responses
Addition of increasing concentrations of the PKC activator, PDBu (0.01–0.1 µM), caused concentration-dependent decreases in single-cell glutamate-evoked Ca2+ oscillation frequency and a shift in the distribution of Ca2+ responses between the different response categories (NR, SP, OS, PP), such that the number of cells that failed to respond (NR), or to respond with a single transient (SP) were increased (Figure 3A–E). Similarly, PDBu also caused a concentration-dependent inhibition [pIC50 (M), 7.65 ± 0.08] of [3H]IPx accumulation stimulated by quisqualate (10 µM; Figure 3F).
Figure 3.
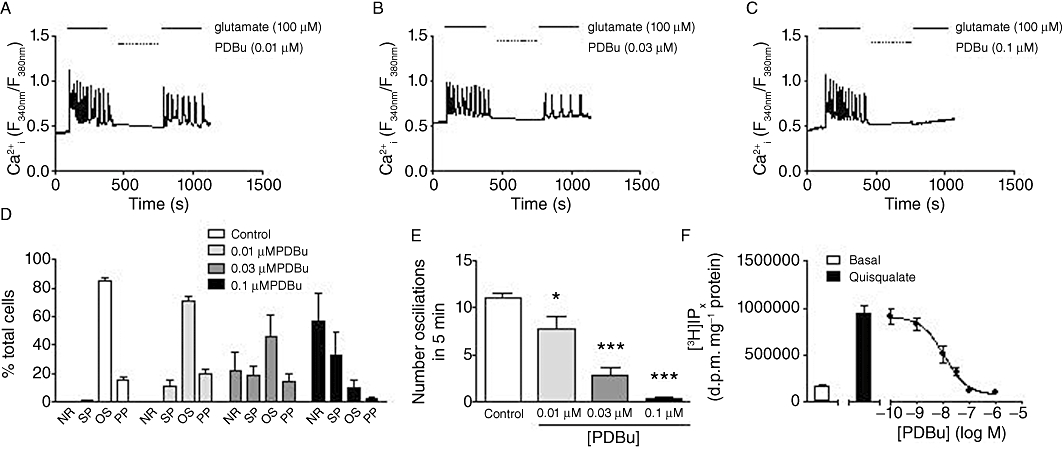
Effects of PKC stimulation on agonist-stimulated Ca2+ oscillations and [3H]IPx accumulation in rat cortical astrocytes. Representative traces showing Ca2+ oscillations elicited by glutamate (100 µM) before and after a 5 min incubation period with 10 nM (A), 30 nM (B) and 100 nM (C) PDBu. Panel D shows the distribution of glutamate-stimulated Ca2+ responses between no-response (NR), single-peak (SP), oscillatory (OS) or peak-plateau (PP) behaviours in the absence or presence of increasing PDBu concentrations. Panel E shows the mean number of oscillations elicited by glutamate (100 µM) stimulation before and after incubation with 10 nM, 30 nM or 100 nM PDBu. Data are shown as means ± SEM for at least 24 individual cells over three to four separate experiments. *P < 0.05, ***P < 0.001 significantly different from control (one-way anova; Bonferroni's multiple comparison test). Panel F shows basal and quisqualate (10 µM)-stimulated [3H]IPx accumulation in astrocytes following pre-incubation for 5 min with the indicated concentrations of PDBu [pIC50 (M) = 7.96 ± 0.21; n = 4].
Inhibition of PKC activity using the broad-spectrum PKC inhibitors, staurosporine (3 µM; 15 min) or Ro 31-8220 (10 µM; 15 min), caused ≥2-fold increases in the maximal quisqualate-stimulated [3H]IPx accumulation, without significant change in the apparent potency of quisqualate to stimulate this response [Figure 4A; pEC50 (M) values: control, 6.98 ± 0.06; +staurosporine, 6.95 ± 0.07; +Ro 31-8220, 6.89 ± 0.10]. Time-course experiments showed that quisqualate-stimulated [3H]IPx accumulations were linear over the 20 min incubation period in the absence and presence of PKC inhibitors (data not shown). The effect of staurosporine or Ro 31-8220 on the maximal [3H]IPx accumulation stimulated by quisqualate was concentration-dependent (Figure 4B), yielding pEC50 (M) values of 6.47 ± 0.09 and 5.61 ± 0.16, for staurosporine and Ro 31-8220 respectively. While activation of PKC activity by PDBu (1 µM) could almost completely suppress quisqualate-stimulated [3H]-IPx accumulation under control conditions, in the presence of staurosporine or Ro 31-8220 the inhibitory effect of acute PKC stimulation was completely lost (Figure 4C). In addition, the potentiating effect of staurosporine pre-incubation (3 µM; 15 min) on agonist-stimulated [3H]IPx accumulation appeared to be selective for the mGlu5 receptor in rat cortical astrocytes, as this inhibitor was without effect on the small, but significant, [3H]IPx accumulations stimulated by methacholine (1 mM), noradrenaline (300 µM) or ATP (300 µM) (Figure 4D).
Figure 4.
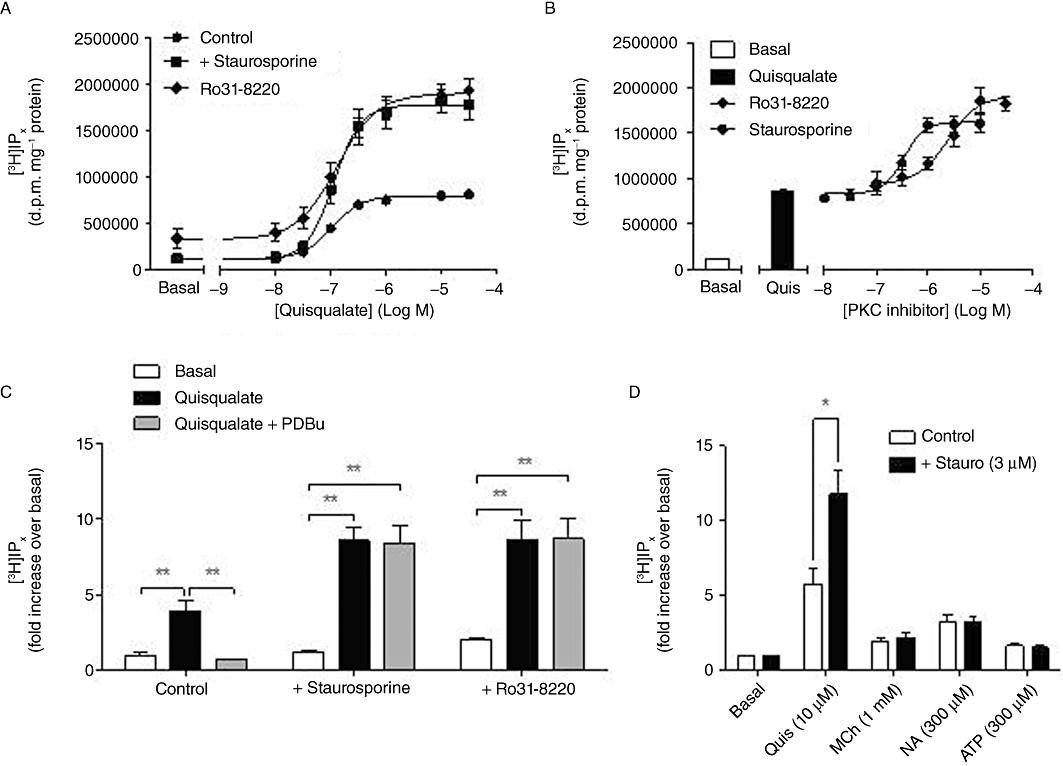
Effects of PKC inhibition on agonist-stimulated [3H]IPx accumulation in rat cortical astrocytes. Concentration–response curves (A) for quisqualate-stimulated [3H]IPx accumulation in cells pretreated with vehicle (control), staurosporine (3 µM; 15 min) or Ro 31-8220 (10 µM; 15 min): mean (±SEM) pEC50 (M) values were 6.97 ± 0.05, 6.93 ± 0.05 and 6.89 ± 0.13 respectively. Concentration-dependence (B) of [3H]IPx accumulation for staurosporine (0.01–10 µM) or Ro 31-8220 (0.1–30 µM) pretreated astrocytes stimulated with quisqualate (10 µM): mean pEC50 (M) values for potentiation of the quisqualate response by staurosporine or Ro 31-8220 were 6.38 ± 0.10 and 5.33 ± 0.05 respectively. Panel C shows the effects of acute PDBu (1 µM; 5 min pre-incubation) treatment on [3H]IPx accumulations stimulated by 10 µM quisqualate in the absence or presence of staurosporine (3 µM) or Ro 31-8220 (10 µM): data are shown as means ± SEM for three to four separate experiments performed in duplicate. **P < 0.005 significantly different as shown (one-way anova followed by Bonferroni's multiple comparison test). Panel D shows [3H]IPx accumulations stimulated by quisqualate (10 µM), methacholine (MCh) (1 mM), noradrenaline (NA) (300 µM) or ATP (300 µM) in the absence or presence of the broad spectrum PKC inhibitor, staurosporine (3 µM; 15 min pre-incubation). Data shown are means ± SEM for three separate experiments performed in duplicate. Additions of quisqualate, MCh, NA or ATP significantly increased [3H]IPx accumulations over basal, but only in the case of quisqualate was there a significant effect (*P < 0.05; paired Student's t-test), of staurosporine.
At the single-cell level, glutamate (100 µM) perfusion initiated typical Ca2+ oscillatory patterns in 80% of astrocytes analysed; however, following a brief incubation period with staurosporine (3 µM; 15 min), subsequent glutamate addition produced a peak-plateau Ca2+ response in 100% of cells (Figure 5A,B).
Figure 5.
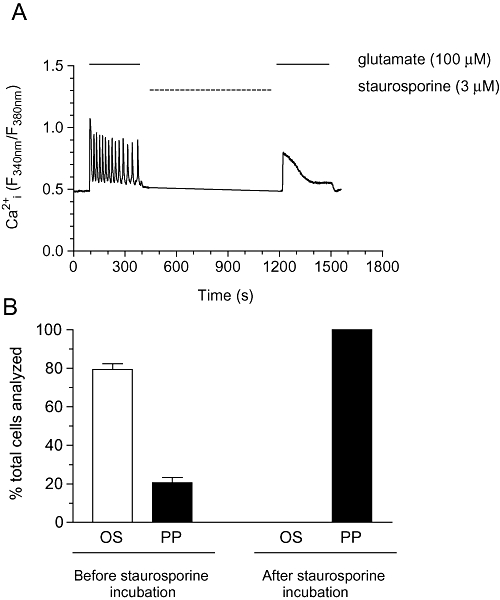
Effects of staurosporine on glutamate-stimulated Ca2+ oscillations in rat cortical astrocytes. (A) Representative trace showing Ca2+ oscillations elicited by glutamate (100 µM) before and after a 15 min incubation period with staurosporine (3 µM). Panel B shows % of cells displaying oscillatory (OS) or peak-plateau (PP) behaviours to glutamate (100 µM) stimulation, with and without staurosporine addition. Data are shown as means ± SEM from 51 individual cells over three separate experiments.
PKC isoenzymes involved in dynamic uncoupling are independent of [Ca2+]i
To investigate the Ca2+ dependence of PKC isoenzymes involved in receptor phosphorylation and hence the generation of mGlu5 receptor-mediated Ca2+ oscillations, we initially used a pharmacological approach to inhibit conventional PKC isoenzymes, that is, those that are activated by Ca2+ and DAG binding. Pre-incubation with Gö6976 (10 µM; 20 min) or myristoylated PKC 20-28 (10 µM; 30 min) had no effect on the Ca2+ oscillation frequency stimulated by glutamate (100 µM) addition (Figure 6A–C), with ≥80% of cells displaying robust Ca2+ oscillations following glutamate perfusion in both control and inhibitor-treated cells (data not shown). In addition, pre-incubation with either Gö6976 or myristoylated PKC 20-28 had no significant effect on [3H]IPx accumulations stimulated by quisqualate (10 µM) addition (Figure 6D). To further explore the dependence of the PKC isoenzyme(s) involved in mGlu5 receptor phosphorylation, we loaded astrocytes with BAPTA-AM (to chelate and prevent changes in [Ca2+]i) and assessed the effect of this manipulation on agonist-stimulated [3H]IPx accumulation (Figure 6E). BAPTA-AM (100 µM; 30 min) had no significant effect on quisqualate-stimulated [3H]IPx accumulation [quisqualate concentration–response curve pEC50 (M) values: control, 7.02 ± 0.06; +BAPTA-AM, 6.92 ± 0.08]. To demonstrate that [Ca2+]i is effectively clamped by the BAPTA-AM-loading protocol Ca2+ responses to glutamate (100 µM) control and BAPTA-AM-loaded (100 µM; 30 min) cells are also shown (Figure 6E).
Figure 6.
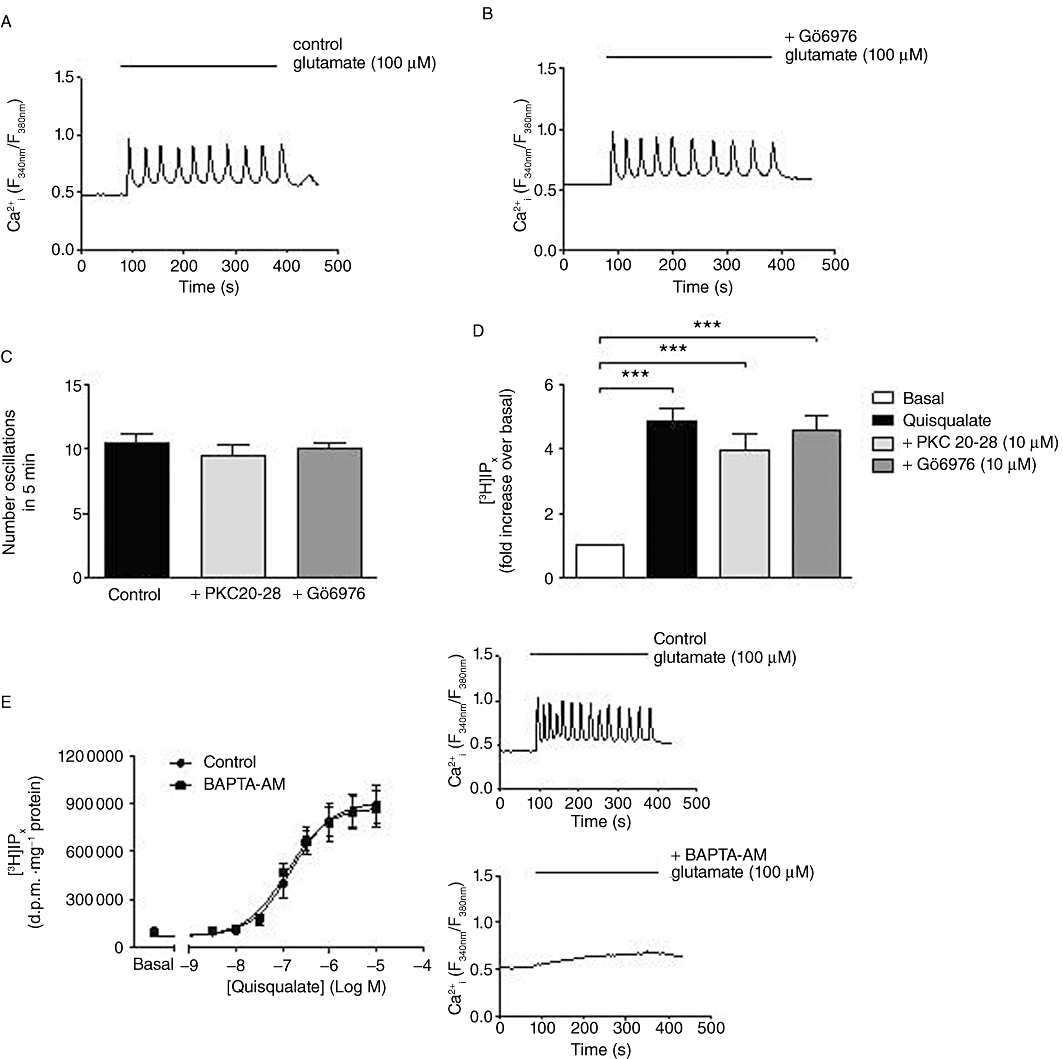
Assessing the Ca2+ dependence of the PKC isoenzyme(s) involved in mGlu5 receptor regulation. Representative traces showing Ca2+ oscillations elicited by glutamate (100 µM) in control (A) and Gö6976 (10 µM; 20 min)-pretreated (B) cells. Panel C shows the number of oscillations elicited by glutamate (100 µM) stimulation in control, PKC 20-28- and Gö6976-pre-incubated cells: data are shown as means ± SEM from at least 30 individual cells over three separate experiments. Panel D shows [3H]IPx accumulations in response to stimulation with 10 µM quisqualate when cells were pre-incubated with PKC 20-28 (10 µM; 30 min) or Gö6976 (10 µM; 20 min): data shown are means ± SEM for six separate experiments performed in duplicate. ***P < 0.001 significantly different as shown (one-way anova followed by Bonferroni's multiple comparison test). Panel E shows concentration–response curves for [3H]IPx accumulations after 20 min stimulation with various concentrations of quisqualate in control and BAPTA-AM (100 µM; 30 min)-loaded cells. Mean (±SEM) pEC50 (M) values for quisqualate-stimulated responses were 7.02 ± 0.06 in control and 6.92 ± 0.08 in BAPTA-loaded cells. Insets to panel E show representative single-cell Ca2+ responses to 5 min perfusion with glutamate (100 µM) in control (upper) and BAPTA-loaded (lower) cells.
Elucidating the PKC isoenzyme(s) involved in mGlu5 receptor-mediated Ca2+ oscillations
We assessed the expression of different PKC isoenzymes in astrocyte cultures following differentiation with G5 supplement, using various antisera specific to PKCα, PKCβ, PKCγ, PKCδ, PKCε, PKCθ, PKCι and PKCζ (Figure 7). Of the conventional PKC isoenzymes, PKCα and PKCβ, but not PKCγ, were expressed in our astrocyte cultures. All novel PKC isoenzymes investigated, PKCδ, PKCε and PKCθ, could be detected in our cultures. In contrast, the atypical PKC isoenzymes, PKCι, but not PKCζ, immunoreactivity could be detected.
Figure 7.

Analysis of PKC isoenzyme expression in rat cortical astrocytes. Rat brain tissue and astrocyte cell lysates were prepared and resolved by 12.5% SDS-PAGE as described in Methods. Protein levels were detected using antisera specific to PKCα, PKCβ, PKCγ, PKCδ, PKCε, PKCθ, PKCι and PKCζ. Representative blots from three independent experiments are shown for each PKC isoenzyme.
In the light of our evidence for the involvement of Ca2+-independent PKC isoenzymes, we extended our study next by focusing on the novel PKCs. We transfected astrocytes with siRNAs targeting PKCδ or PKCε to deplete endogenous expression levels: optimal depletion of the relevant target was achieved 72 h after siRNA nucleofection, and this period also allowed sufficient time for differentiation of astrocytes in the G5 supplement. An isoenzyme-specific effect for each anti-PKC siRNA was supported by the lack of effect of anti-PKCε siRNA on PKCδ expression (Figure 8A) and vice versa (Figure 8B). A negative-control siRNA had no effect on either PKCδ or PKCε expression (Figure 8A,B). Quantification of the knock-down achieved by each siRNA was assessed densitometrically. The two anti-PKCδ siRNAs tested caused knock-down of their target by approximately 90% and 80%, respectively, and both anti-PKCε siRNAs achieved ≥80% depletion of PKCε (Figure 8A,B).
Figure 8.
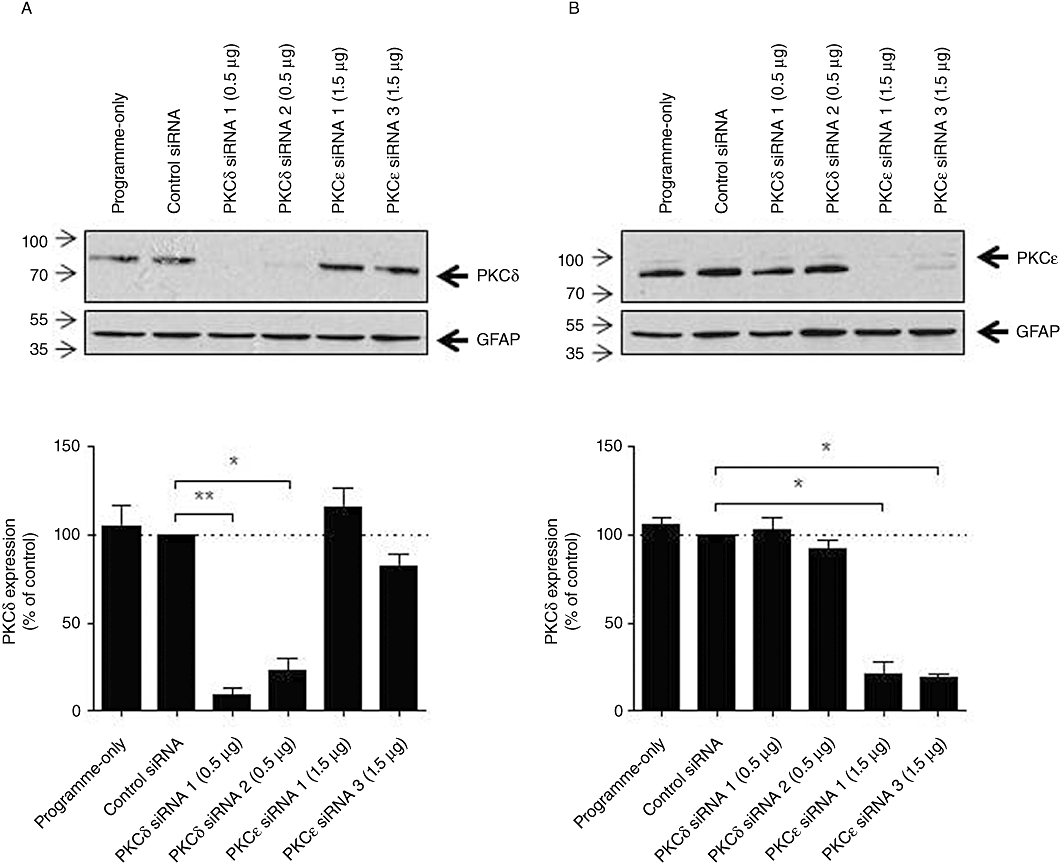
Isoenzyme-specific anti-PKC siRNAs can deplete PKC protein expression in astrocytes. Astrocytes were transfected with control, anti-PKCδ or anti-PKCε siRNAs (0.5 or 1.5 µg) by nucleofection as described in Methods. After 72 h, cells were lysed and protein levels were detected using antisera specific to PKCδ (A) and PKCε (B). Representative immunoblots from at least three separate experiments are shown with lower panels showing GFAP expression. Densitometric analyses of PKCδ and PKCε immunoreactivity for each immunoblot normalized to GFAP expression are shown in the lower panels, means ± SEM; *P < 0.05; **P < 0.005 significantly different as shown (one-way anova followed by Dunnett's multiple comparison test).
To examine the functional consequences of isoenzyme-specific depletion of PKCδ or PKCε, we utilized Ca2+ imaging to assess the effects of knock-down at a single-cell level. Astrocytes were transfected with programme-only (no siRNA present), negative-control siRNA (0.5 µg) or anti-PKCδ (0.5 µg) siRNAs; after 72 h cells were stimulated with glutamate (100 µM, 5 min; Figure 9A–D). Ca2+ responses here were subclassified as oscillatory (OS), oscillatory driving into a sustained peak (OS > PP) or a peak-plateau (PP) Ca2+ responses (see Figure 9E right-hand panels for examples of each). In astrocytes nucleofected using programme-only, or with control siRNA, an oscillatory Ca2+ response was seen in the majority (≥70%) of cells analysed. Following PKCδ knock-down, the distribution of Ca2+ responses between OS, OS > PP and PP on glutamate addition was not altered, with ∼70% of all cells analysed displaying an oscillatory Ca2+ response (Figure 9E), suggesting that mGlu5 receptor phosphorylation/dephosphorylation is unaffected by a marked (≥80%) loss of cellular PKCδ activity. Quantification of glutamate-stimulated Ca2+ oscillation frequencies showed no significant differences between vector-control and anti-PKCδ siRNA-transfected astrocytes (data not shown).
Figure 9.
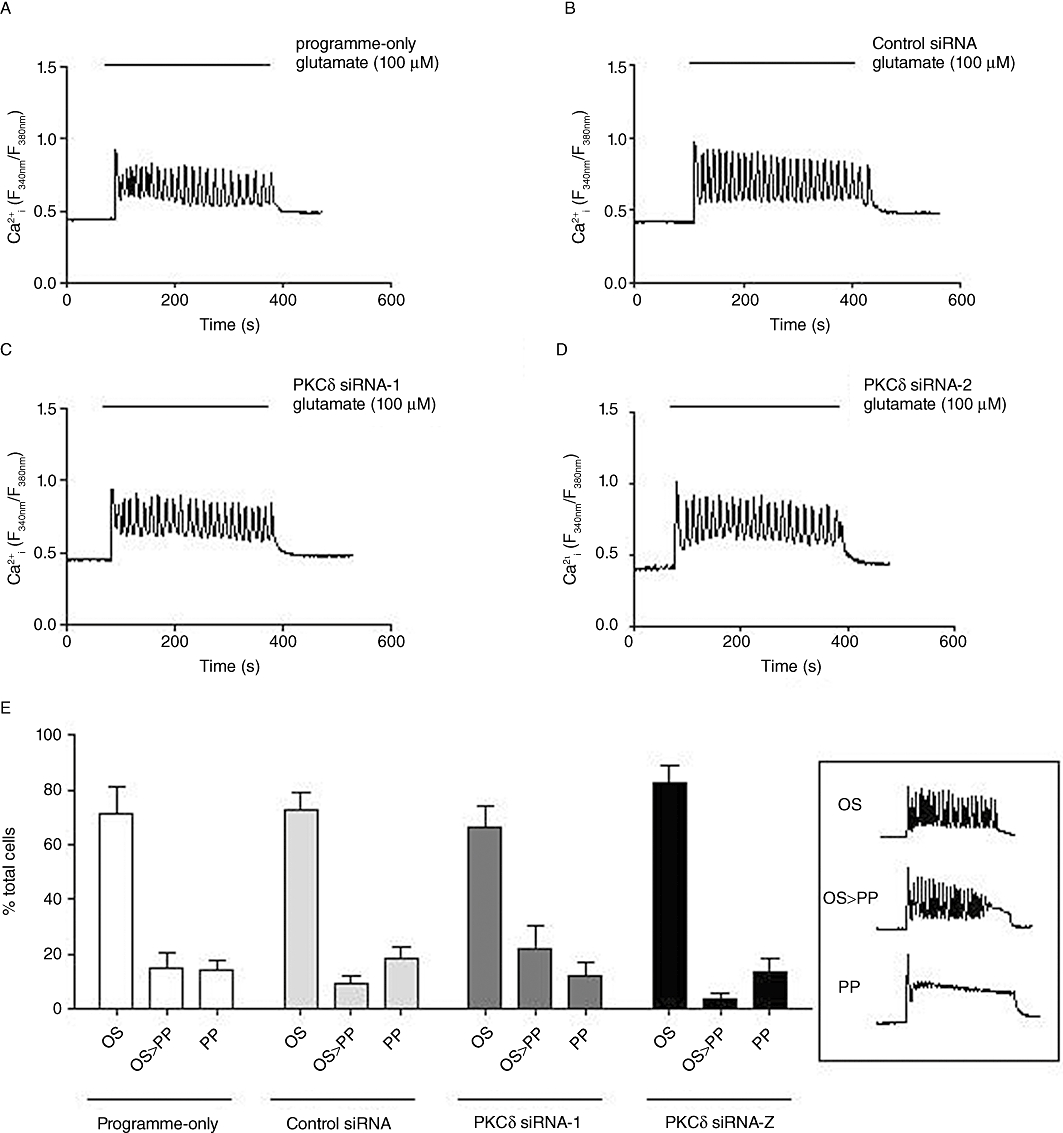
Effects of PKCδ siRNA knock-down on glutamate-stimulated single-cell Ca2+ responses in astrocytes. Representative traces showing Ca2+ responses elicited by glutamate (100 µM) stimulation in astrocytes nucleofected with no siRNA (programme-only; A), control siRNA (B), PKCδ siRNA-1 (0.5 µg) (C) or PKCδ siRNA-2 (0.5 µg) (D). Mean ± SEM data from at least 100 cells over at least four separate experiments are shown in panel E for % of the total number of cells analysed that gave an oscillatory (OS), oscillatory driving into sustained plateau (OS > PP) or a peak-plateau (PP) response when stimulated with glutamate (100 µM). Inset graphs (right) present typical Ca2+ responses defining each category.
Astrocytes transfected with anti-PKCε siRNAs displayed a range of Ca2+ responses with a clear shift in the distribution of (OS, OS > PP and PP) Ca2+ responses from oscillatory towards peak-plateau (Figure 10A–G). Summary data (Figure 10G) show that the number of cells displaying an oscillatory Ca2+ response was significantly reduced in PKCε-depleted cells (to ≤40% of cells analysed). Co-nucleofection with anti-PKCδ and anti-PKCε siRNAs also reduced the proportion of astrocytes exhibiting peak-plateau responses to glutamate, but the shift from OS to PP was no greater than was seen with anti-PKCε siRNA nucleofection alone (data not shown). Ca2+ oscillation frequencies stimulated by glutamate were not significantly different between control siRNA and anti-PKCε siRNA-transfected cells in the respective subpopulations of astrocytes showing this response pattern. These data suggest that the PKCε, but not the PKCδ, isoenzyme was involved in the acute regulation of mGlu5 receptor-mediated Ca2+ oscillations in astrocytes.
Figure 10.
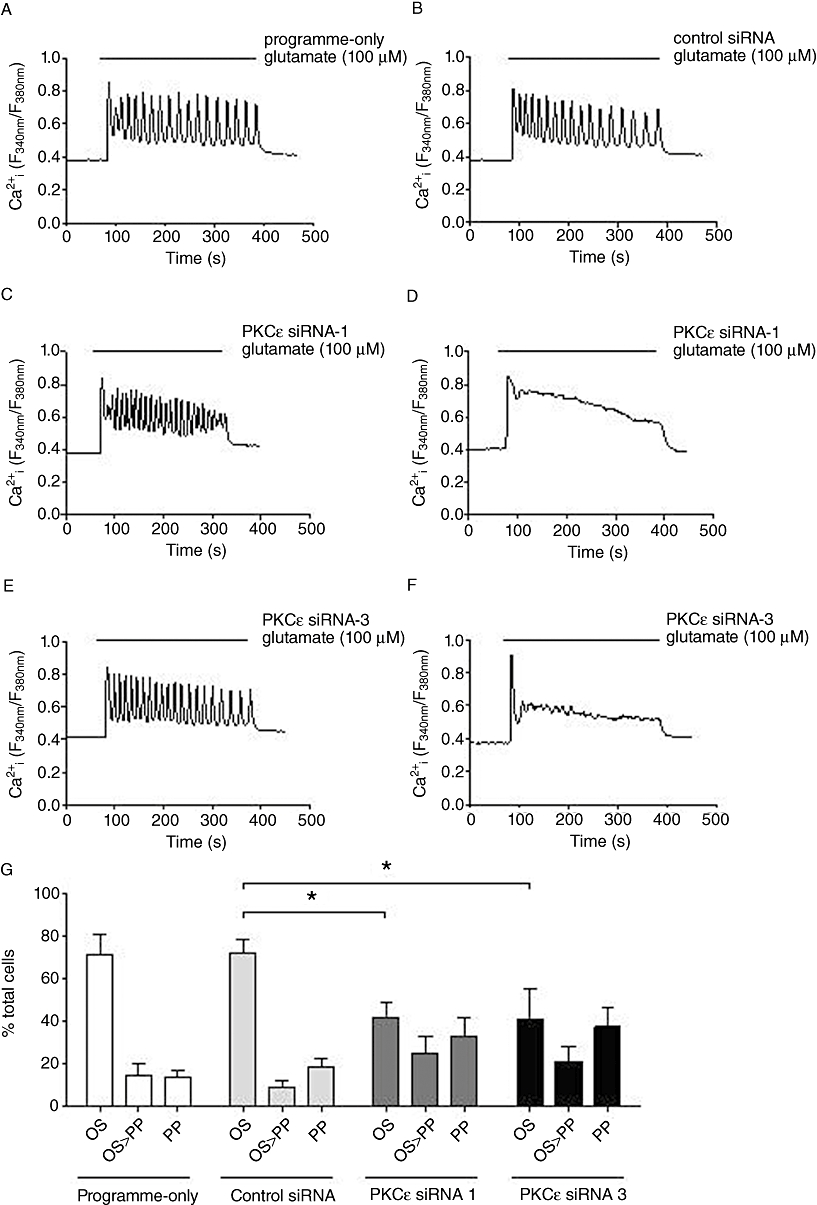
Effects of PKCε siRNA knock-down on glutamate-stimulated single-cell Ca2+ responses in astrocytes. Representative traces showing Ca2+ responses elicited by glutamate (100 µM) stimulation in astrocytes nucleofected with no siRNA (programme-only; A), control siRNA (B) PKCε siRNA-1 (1.5 µg) (C, D) or PKCε siRNA-3 (1.5 µg) (E, F). Mean ± SEM data from at least 100 cells over at least four separate experiments are shown in panel G for % of the total number of cells analysed that gave an oscillatory (OS), oscillatory driving into sustained plateau (OS > PP) or a peak-plateau (PP) response when stimulated with glutamate (100 µM). *P < 0.05 significantly different as shown (two-way anova followed by Bonferroni's multiple comparison test).
Discussion
This study was undertaken to elucidate further the mechanisms that underlie mGlu5 receptor-stimulated Ca2+ oscillations in rat cerebrocortical astrocytes, initially using a pharmacological approach, and then extending our investigation to include molecular genetic methods. Our new data both confirm and extend previous studies, performed mostly in recombinant cell systems, to uncover the specific protein phosphatase and PKC isoenzymes involved in the regulation of astrocytic mGlu5 receptor–Ca2+ signalling.
Although mGlu1 and mGlu5 receptors are structurally related and preferentially couple via Gq/11 to stimulate PLC activity, these receptors elicit contrasting Ca2+ responses at the single-cell level. In contrast to stimulation of mGlu1 receptors, which has, in the main, been reported to cause peak-plateau Ca2+ responses (Hermans et al., 1998; Kawabata et al., 1996, 1998; Atkinson et al., 2006; but see Dale et al., 2001), stimulation of the mGlu5 receptor initiates rapid and robust Ca2+ oscillations in mGlu5 receptor-expressing recombinant cell lines (Kawabata et al., 1996, 1998; Nash et al., 2002; Atkinson et al., 2006), as well as in native cells, such as astrocytes and neurones (Nakahara et al., 1997; Flint et al., 1999; Codazzi et al., 2001). mGlu5 receptor-driven Ca2+ oscillations occur in synchrony with cellular oscillations in IP3 concentration, indicating that these oscillatory responses involve a cyclical receptor-mediated on/off switching of PLC activity (Nash et al., 2001, 2002). Substantial evidence has now accumulated to support the notion that these IP3/Ca2+ oscillations are brought about by a process termed ‘dynamic uncoupling’ (Nash et al., 2002), where mGlu5 receptor-driven activation of PKC leads to receptor phosphorylation, specifically at Ser839, to uncouple the receptor from Gq/11 and an as yet poorly defined protein phosphatase activity rapidly dephosphorylates the receptor to allow restoration of mGlu5 receptor–Gq/11 coupling (Kawabata et al., 1996; Nakahara et al., 1997; Nash et al., 2002; Kim et al., 2005; but see Dale et al., 2001).
Stimulation of the astrocyte mGlu5 receptor initiates robust Ca2+ oscillations that can reach a frequency of approximately three oscillations per minute. Application of calyculin A or cantharidin, relatively selective inhibitors of the serine/threonine protein phosphatases PP1/PP2A (Ishihara et al., 1989; Honkanen, 1993), caused significant reductions in glutamate-evoked Ca2+ oscillation frequency, in agreement with previous findings (Nakahara et al., 1997; Nash et al., 2002). These data support the involvement of specific protein phosphatase isoenzyme(s) in the cyclical phosphorylation/dephosphorylation of mGlu5 receptors in astrocytes. Pharmacological inhibition of PP1/PP2A also caused concentration-dependent reductions in quisqualate-stimulated [3H]IPx accumulation, providing corroborative evidence for the involvement of protein phosphatases in the cyclical restoration of signalling initiated by mGlu5 receptors. The close proximity of GPCRs and protein phosphatases, as well as scaffolded protein kinase/phosphatase complexes, is now well documented (Pitcher et al., 1995; Bauman and Scott, 2002; Dodge-Kafka et al., 2010). Indeed, the existence of a complex of the mGlu5 receptor with PP2A has been demonstrated in neurones in vivo (Mao et al., 2005), while mGlu receptor–protein phosphatase interactions have also been reported for other mGlu receptor subtypes (Flajolet et al., 2003).
The mGlu5 receptor has also been reported to exist in a signalling complex with the protein phosphatase, calcineurin (PP2B). The C-terminal of mGlu5 possesses two binding sites for this Ca2+/calmodulin-dependent protein phosphatase that facilitate receptor dephosphorylation and recovery from PKC-mediated desensitization (Minakami et al., 1997; Alagarsamy et al., 2005). However, we found that preventing changes in [Ca2+]i (by BAPTA-AM loading) in astrocytes had no effect on mGlu5 receptor-stimulated [3H]IPx accumulation, suggesting that a Ca2+-dependent protein phosphatase was unlikely to be involved in the acute cyclical phosphorylation/dephosphorylation of the mGlu5 receptor. This finding is also in agreement with the previously reported lack of effect of FK506 on (1S,3R)-1-aminocyclopentane-1,3-dicarboxylate-induced Ca2+ oscillations in astrocytes (Nakahara et al., 1997).
Our study has also focused on the role of PKC(s) in the generation of mGlu5 receptor-mediated Ca2+ oscillations, initially through pharmacological manipulation of cellular PKC activities. Phorbol ester stimulation of PKC activity caused a concentration-dependent decrease in mGlu5 receptor-stimulated [3H]IPx accumulation and Ca2+ oscillation frequency. It has previously been reported that the PKC inhibitor, staurosporine, can completely abolish mGlu5 receptor-stimulated Ca2+ oscillations in recombinant cell systems and astrocytes (Kawabata et al., 1996; Nakahara et al., 1997). Here, we have not only investigated the effects of PKC inhibitors on single-cell Ca2+ responses evoked by mGlu5 receptors, but also looked more directly at the effects of PKC inhibition on PLC activity. In cortical astrocytes PKC inhibition converted mGlu5 receptor-driven Ca2+ oscillatory responses into sustained peak-plateau responses. The broad-spectrum PKC inhibitors, staurosporine and Ro 31-8220, each caused an approximately twofold increase in the maximum [3H]IPx accumulation stimulated by quisqualate. These data are consistent with the mGlu5 receptor existing for longer periods in a dephosphorylated, Gq/11-coupled and PLC stimulatory state in the presence of the PKC inhibitor. A similar increase in quisqualate-stimulated [3H]IPx accumulation could be observed in CHO-lac-mGlu5a cells treated overnight with PDBu to down-regulate conventional and novel PKC isoenzymes; however, G5-differentiated cortical astrocytes did not tolerate chronic exposure to phorbol ester treatment, preventing us providing such complementary data in the native mGlu5 receptor-expressing system (data not shown).
Previous attempts to define the specific PKC isoenzyme(s) involved in mGlu5 receptor dynamic uncoupling have failed to produce a consensus. Stimulation of mGlu5 receptor in HEK cells has been shown to lead to the translocation of PKCγ and δ isoenzymes from the cytoplasm to the membrane, although only PKCδ was reported to phosphorylate the mGlu5 receptor (Uchino et al., 2004). In contrast, Codazzi et al. (2001) reported in hippocampal astrocytes that mGlu5 receptor-stimulated PKC activation is dependent on repetitive DAG and Ca2+ spikes and hypothesized a role for conventional PKCs in acute receptor regulation. In contrast to the latter finding, the work presented here showed that Gö6976 and PKC 20-28, inhibitors of conventional PKCs, had no effect on mGlu5 receptor-stimulated responses in cortical astrocytes, at concentrations previously demonstrated to inhibit fully conventional (Ca2+- and DAG-dependent) PKCs (Eichholtz et al., 1993; Martiny-Baron et al., 1993). Supportive evidence for the mGlu5 receptor-regulating PKC being a Ca2+-independent isoenzyme comes from the lack of effect that ‘clamping’ of astrocyte [Ca2+]i (by BAPTA-AM loading) has on quisqualate-stimulated [3H]IPx accumulation.
To take our study further, we decided that molecular genetic approaches would be needed to more precisely pinpoint the PKC isoenzyme(s) involved in mGlu5 receptor regulation and decided to attempt isoenzyme-specific knock-down using RNAi. G5-differentiated cortical astrocyte cultures express an array of PKC isoenzymes including members of the conventional, novel and atypical subfamilies. Consistent with a previous study, we report the lack of expression of PKCγ in astrocytes, but show the expression of PKCα, β, δ, ε, θ and ι using isoenzyme-specific antibodies (Slepko et al., 1999). However, in contrast to the findings of Slepko et al. (1999), we did not detect PKCζ, consistent with the findings of Asotra and Macklin (1994). Based on our findings suggesting the involvement of a Ca2+-independent, but DAG (or at least phorbol ester)-regulated PKC activity in acute mGlu5 receptor regulation, we focused on the isoenzyme-specific knock-down of novel PKCs using validated siRNAs. The most effective PKCδ siRNA utilized here caused >90% depletion of this isoenzyme 72 h after nucleofection into astrocytes, but had no effect on glutamate-stimulated single-cell Ca2+ responses; in contrast the most effective PKCε siRNA caused ∼80% knock-down and resulted in a significant shift in the distribution of Ca2+ responses from oscillatory towards peak-plateau (see Figure 10). That glutamate-stimulated Ca2+ oscillations were abolished only in a proportion of astrocytes may have been a consequence of the incomplete knock-down of PKCε achieved. It is possible (and indeed likely) that cellular PKCε levels normally exceed those necessary to mediate mGlu5 receptor phosphorylation and therefore a large depletion is needed before an effect is seen. It also remains a possibility that PKC isoenzymes additional to PKCε are involved in mGlu5 receptor dynamic uncoupling.
Thus, our data provide new evidence to implicate PKCε, but not PKCδ in acute mGlu5 receptor phosphorylation. However, more definitive evidence will require the effects of PKCε knock-down on the phosphorylation status of Ser839 within the mGlu5 receptor C-terminal domain to be investigated. While these two closely related and similarly regulated novel PKCs often fulfil redundant, interchangeable roles, there are also examples of PKCε and PKCδ fulfilling distinct cellular functions (Pears et al., 2008; Frangioudakis et al., 2009; Sivaraman et al., 2009). It is also noteworthy that PKCε is the most abundantly expressed novel PKC in the brain, where it is thought to fulfil numerous neuronal functions, including the regulation of ion channels and neurotransmitter release (Shirai et al., 2008). Important roles for PKCε have also been highlighted in glial cells, including the differentiation of astrocytes from neural precursor cells (Steinhart et al., 2007). An earlier study investigating effects of staurosporine on astrocytic phenotype and PKC isoenzymic expression showed that this inhibitor caused membrane-associated PKCε to be translocated to the cytoplasm, without altering the distribution of the other PKC isoenzymes studied (Kronfeld et al., 1995). Such a staurosporine-induced redistribution of PKCε away from the plasma membrane may help to explain the effectiveness of this agent to ablate completely mGlu5 receptor-stimulated Ca2+ oscillations. In addition, agonist stimulation of another family C GPCR, the calcium-sensing (CaS) receptor, can also generate robust Ca2+ oscillations, most likely through the cyclical phosphorylation/dephosphorylation of a key C-terminal residue (Thr888) (Miedlich et al., 2002; Young et al., 2002; Davies et al., 2007; McCormick et al., 2010). It has also been reported that expression to constitutively active forms of PKCβ or PKCε can reduce or abolish Ca2+ oscillations, suggesting that PKCε (and perhaps other PKC isoenzymes) is an important regulator of the CaS receptor (Young et al., 2002).
In conclusion, our data provide additional, supportive evidence for the involvement of specific PKCs and protein phosphatases in the rapid cyclical phosphorylation and dephosphorylation of the mGlu5 receptor that define Ca2+ oscillation frequency generated by agonist stimulation in rat cerebrocortical astrocytes; in particular, we highlight the roles of specific Ca2+-independent PKC and protein phosphatase isoenzymes in shaping receptor-driven Ca2+ oscillatory behaviours that may in turn determine an array of glial functions, including the release of gliotransmitters (Agulhon et al., 2008; Fiacco et al., 2009) and the programming of transcriptional events (Gu and Spitzer, 1995; De Koninck and Schulman, 1998; Dolmetsch et al., 1998).
Acknowledgments
We thank the Biotechnology and Biological Sciences Research Council and GlaxoSmithKline for the award of a CASE PhD studentship to S.J.B. We also thank Dr Jeannette M. Watson (GlaxoSmithKline Neurosciences CEDD, Harlow, UK) for her support in the initial stages of this project.
Glossary
Abbreviations
- CNQX
6-cyano-7-nitroquinoxaline-2,3-dione
- DAPI
4′,6-diamidino-2-phenylindole
- DHPG
(S)-3,5-dihydroxyphenylglycine
- GFAP
glial fibrillary acidic protein
- [3H]IPx
[3H]inositol mono-, bis- and trisphosphate fraction
- mGlu
metabotropic glutamate
- PDBu
phorbol 12,13-dibutyrate
- PP1/PP2A
protein phosphatase-1/2A
- PP2B
protein phosphatase 2B
Conflict of interest
None.
References
- Agulhon C, Petravicz J, McMullen AB, Sweger EJ, Minton SK, Taves SR, et al. What is the role of astrocyte calcium in neurophysiology? Neuron. 2008;59:932–946. doi: 10.1016/j.neuron.2008.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alagarsamy S, Saugstad J, Warren L, Mansuy IM, Gereau RW, 4th, Conn PJ. NMDA-induced potentiation of mGluR5 is mediated by activation of protein phosphatase 2B/calcineurin. Neuropharmacology. 2005;49(Suppl. 1):135–145. doi: 10.1016/j.neuropharm.2005.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander SPH, Mathie A, Peters JA. Guide to receptors and channels (GRAC). 4th edn. Br J Pharmacol. 2009;158(Suppl. 1):S1–S254. doi: 10.1111/j.1476-5381.2009.00499.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen NJ, Barres BA. Neuroscience: glia – more than just brain glue. Nature. 2009;457:675–677. doi: 10.1038/457675a. [DOI] [PubMed] [Google Scholar]
- Aronica E, Gorter JA, Ijlst-Keizers H, Rozemuller AJ, Yankaya B, Leenstra S, et al. Expression and functional role of mGluR3 and mGluR5 in human astrocytes and glioma cells: opposite regulation of glutamate transporter proteins. Eur J Neurosci. 2003;17:2106–2118. doi: 10.1046/j.1460-9568.2003.02657.x. [DOI] [PubMed] [Google Scholar]
- Asotra K, Macklin WB. Developmental expression of protein kinase C isozymes in oligodendrocytes and their differential modulation by 4 β-phorbol-12,13-dibutyrate. J Neurosci Res. 1994;39:273–289. doi: 10.1002/jnr.490390305. [DOI] [PubMed] [Google Scholar]
- Atkinson PJ, Young KW, Ennion SJ, Kew JN, Nahorski SR, Challiss RA. Altered expression of protein shapes mGlu1 and mGlu5 receptor-mediated single cell inositol 1,4,5-trisphosphate and Ca(2+) signaling. Mol Pharmacol. 2006;69:174–184. doi: 10.1124/mol.105.014258. [DOI] [PubMed] [Google Scholar]
- Bauman AL, Scott JD. Kinase- and phosphatase-anchoring proteins: harnessing the dynamic duo. Nat Cell Biol. 2002;4:E203–E206. doi: 10.1038/ncb0802-e203. [DOI] [PubMed] [Google Scholar]
- Bradley SJ, Watson JM, Challiss RA. Effects of positive allosteric modulators on single-cell oscillatory Ca2+ signaling initiated by the type 5 metabotropic glutamate receptor. Mol Pharmacol. 2009;76:1302–1313. doi: 10.1124/mol.109.059170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Z, Schools GP, Kimelberg HK. Metabotropic glutamate receptors in acutely isolated hippocampal astrocytes: developmental changes of mGluR5 mRNA and functional expression. Glia. 2000;29:70–80. doi: 10.1002/(sici)1098-1136(20000101)29:1<70::aid-glia7>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- Codazzi F, Teruel MN, Meyer T. Control of astrocyte Ca(2+) oscillations and waves by oscillating translocation and activation of protein kinase C. Curr Biol. 2001;11:1089–1097. doi: 10.1016/s0960-9822(01)00326-8. [DOI] [PubMed] [Google Scholar]
- Conn PJ, Pin JP. Pharmacology and functions of metabotropic glutamate receptors. Annu Rev Pharmacol Toxicol. 1997;37:205–237. doi: 10.1146/annurev.pharmtox.37.1.205. [DOI] [PubMed] [Google Scholar]
- Dale LB, Babwah AV, Bhattacharya M, Kelvin DJ, Ferguson SS. Spatial-temporal patterning of metabotropic glutamate receptor-mediated inositol 1,4,5-trisphosphate, calcium, and protein kinase C oscillations: protein kinase C-dependent receptor phosphorylation is not required. J Biol Chem. 2001;276:35900–35908. doi: 10.1074/jbc.M103847200. [DOI] [PubMed] [Google Scholar]
- Davies SL, Ozawa A, McCormick WD, Dvorak MM, Ward DT. Protein kinase C-mediated phosphorylation of the calcium-sensing receptor is stimulated by receptor activation and attenuated by calyculin-sensitive phosphatase activity. J Biol Chem. 2007;282:15048–15056. doi: 10.1074/jbc.M607469200. [DOI] [PubMed] [Google Scholar]
- De Koninck P, Schulman H. Sensitivity of CaM kinase II to the frequency of Ca2+ oscillations. Science. 1998;279:227–230. doi: 10.1126/science.279.5348.227. [DOI] [PubMed] [Google Scholar]
- Dodge-Kafka KL, Bauman A, Mayer N, Henson E, Heredia L, Ahn J, et al. cAMP-stimulated protein phosphatase 2A activity associated with muscle A kinase-anchoring protein (mAKAP) signaling complexes inhibits the phosphorylation and activity of the cAMP-specific phosphodiesterase PDE4D3. J Biol Chem. 2010;285:11078–11086. doi: 10.1074/jbc.M109.034868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolmetsch RE, Xu K, Lewis RS. Calcium oscillations increase the efficiency and specificity of gene expression. Nature. 1998;392:933–936. doi: 10.1038/31960. [DOI] [PubMed] [Google Scholar]
- Eichholtz T, de Bont DB, de Widt J, Liskamp RM, Ploegh HL. A myristoylated pseudosubstrate peptide, a novel protein kinase C inhibitor. J Biol Chem. 1993;268:1982–1986. [PubMed] [Google Scholar]
- Fiacco TA, Agulhon C, McCarthy KD. Sorting out astrocyte physiology from pharmacology. Annu Rev Pharmacol Toxicol. 2009;49:151–174. doi: 10.1146/annurev.pharmtox.011008.145602. [DOI] [PubMed] [Google Scholar]
- Flajolet M, Rakhilin S, Wang H, Starkova N, Nuangchamnong N, Nairn AC, et al. Protein phosphatase 2C binds selectively to and dephosphorylates metabotropic glutamate receptor 3. Proc Natl Acad Sci USA. 2003;100:16006–16011. doi: 10.1073/pnas.2136600100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flint AC, Dammerman RS, Kriegstein AR. Endogenous activation of metabotropic glutamate receptors in neocortical development causes neuronal calcium oscillations. Proc Natl Acad Sci USA. 1999;96:12144–12149. doi: 10.1073/pnas.96.21.12144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frangioudakis G, Burchfield JG, Narasimhan S, Cooney GJ, Leitges M, Biden TJ, et al. Diverse roles for protein kinase C δ and protein kinase C ε in the generation of high-fat-diet-induced glucose intolerance in mice: regulation of lipogenesis by protein kinase C δ. Diabetologia. 2009;52:2616–2620. doi: 10.1007/s00125-009-1543-0. [DOI] [PubMed] [Google Scholar]
- Gu X, Spitzer NC. Distinct aspects of neuronal differentiation encoded by frequency of spontaneous Ca2+ transients. Nature. 1995;375:784–787. doi: 10.1038/375784a0. [DOI] [PubMed] [Google Scholar]
- Hermans E, Young KW, Challiss RA, Nahorski SR. Effects of human type 1alpha metabotropic glutamate receptor expression level on phosphoinositide and Ca2+ signalling in an inducible cell expression system. J Neurochem. 1998;70:1772–1775. doi: 10.1046/j.1471-4159.1998.70041772.x. [DOI] [PubMed] [Google Scholar]
- Honkanen RE. Cantharidin, another natural toxin that inhibits the activity of serine/threonine protein phosphatases types 1 and 2A. FEBS Lett. 1993;330:283–286. doi: 10.1016/0014-5793(93)80889-3. [DOI] [PubMed] [Google Scholar]
- Ishihara H, Martin BL, Brautigan DL, Karaki H, Ozaki H, Kato Y, et al. Calyculin A and okadaic acid: inhibitors of protein phosphatase activity. Biochem Biophys Res Commun. 1989;159:871–877. doi: 10.1016/0006-291x(89)92189-x. [DOI] [PubMed] [Google Scholar]
- Kawabata S, Kohara A, Tsutsumi R, Itahana H, Hayashibe S, Yamaguchi T, et al. Diversity of calcium signaling by metabotropic glutamate receptors. J Biol Chem. 1998;273:17381–17385. doi: 10.1074/jbc.273.28.17381. [DOI] [PubMed] [Google Scholar]
- Kawabata S, Tsutsumi R, Kohara A, Yamaguchi T, Nakanishi S, Okada M. Control of calcium oscillations by phosphorylation of metabotropic glutamate receptors. Nature. 1996;383:89–92. doi: 10.1038/383089a0. [DOI] [PubMed] [Google Scholar]
- Kim CH, Braud S, Isaac JT, Roche KW. Protein kinase C phosphorylation of the metabotropic glutamate receptor mGluR5 on Serine 839 regulates Ca2+ oscillations. J Biol Chem. 2005;280:25409–25415. doi: 10.1074/jbc.M502644200. [DOI] [PubMed] [Google Scholar]
- Kronfeld I, Zsukerman A, Kazimirsky G, Brodie C. Staurosporine induces astrocytic phenotypes and differential expression of specific PKC isoforms in C6 glial cells. J Neurochem. 1995;65:1505–1514. doi: 10.1046/j.1471-4159.1995.65041505.x. [DOI] [PubMed] [Google Scholar]
- Mao L, Yang L, Arora A, Choe ES, Zhang G, Liu Z, et al. Role of protein phosphatase 2A in mGluR5-regulated MEK/ERK phosphorylation in neurons. J Biol Chem. 2005;280:12602–12610. doi: 10.1074/jbc.M411709200. [DOI] [PubMed] [Google Scholar]
- Martiny-Baron G, Kazanietz MG, Mischak H, Blumberg PM, Kochs G, Hug H, et al. Selective inhibition of protein kinase C isozymes by the indolocarbazole Gö 6976. J Biol Chem. 1993;268:9194–9197. [PubMed] [Google Scholar]
- McCormick WD, Atkinson-Dell R, Campion KL, Mun HC, Conigrave AD, Ward DT. Increased receptor stimulation elicits differential calcium-sensing receptor(T888) dephosphorylation. J Biol Chem. 2010;285:14170–14177. doi: 10.1074/jbc.M109.071084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miedlich S, Gama L, Breitwieser GE. Calcium sensing receptor activation by a calcimimetic suggests a link between cooperativity and intracellular calcium oscillations. J Biol Chem. 2002;277:49691–49699. doi: 10.1074/jbc.M205578200. [DOI] [PubMed] [Google Scholar]
- Miller S, Romano C, Cotman CW. Growth factor upregulation of a phosphoinositide-coupled metabotropic glutamate receptor in cortical astrocytes. J Neurosci. 1995;15:6103–6109. doi: 10.1523/JNEUROSCI.15-09-06103.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minakami R, Jinnai N, Sugiyama H. Phosphorylation and calmodulin binding of the metabotropic glutamate receptor subtype 5 (mGluR5) are antagonistic in vitro. J Biol Chem. 1997;272:20291–20298. doi: 10.1074/jbc.272.32.20291. [DOI] [PubMed] [Google Scholar]
- Nakahara K, Okada M, Nakanishi S. The metabotropic glutamate receptor mGluR5 induces calcium oscillations in cultured astrocytes via protein kinase C phosphorylation. J Neurochem. 1997;69:1467–1475. doi: 10.1046/j.1471-4159.1997.69041467.x. [DOI] [PubMed] [Google Scholar]
- Nash MS, Saunders R, Young KW, Challiss RA, Nahorski SR. Reassessment of the Ca2+ sensing property of a type I metabotropic glutamate receptor by simultaneous measurement of inositol 1,4,5-trisphosphate and Ca2+ in single cells. J Biol Chem. 2001;276:19286–19293. doi: 10.1074/jbc.M007600200. [DOI] [PubMed] [Google Scholar]
- Nash MS, Schell MJ, Atkinson PJ, Johnston NR, Nahorski SR, Challiss RA. Determinants of metabotropic glutamate receptor-5-mediated Ca2+ and inositol 1,4,5-trisphosphate oscillation frequency. Receptor density versus agonist concentration. J Biol Chem. 2002;277:35947–35960. doi: 10.1074/jbc.M205622200. [DOI] [PubMed] [Google Scholar]
- Parker PJ, Murray-Rust J. PKC at a glance. J Cell Sci. 2004;117:131–132. doi: 10.1242/jcs.00982. [DOI] [PubMed] [Google Scholar]
- Pasti L, Volterra A, Pozzan T, Carmignoto G. Intracellular calcium oscillations in astrocytes: a highly plastic, bidirectional form of communication between neurons and astrocytes in situ. J Neurosci. 1997;17:7817–7830. doi: 10.1523/JNEUROSCI.17-20-07817.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pears CJ, Thornber K, Auger JM, Hughes CE, Grygielska B, Protty MB, et al. Differential roles of the PKC novel isoforms, PKCδ and PKCε, in mouse and human platelets. PLoS ONE. 2008;3:e3793. doi: 10.1371/journal.pone.0003793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitcher JA, Payne ES, Csortos C, DePaoli-Roach AA, Lefkowitz RJ. The G-protein-coupled receptor phosphatase: a protein phosphatase type 2A with a distinct subcellular distribution and substrate specificity. Proc Natl Acad Sci USA. 1995;92:8343–8347. doi: 10.1073/pnas.92.18.8343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porter JT, McCarthy KD. Astrocytic neurotransmitter receptors in situ and in vivo. Prog Neurobiol. 1997;51:439–455. doi: 10.1016/s0301-0082(96)00068-8. [DOI] [PubMed] [Google Scholar]
- Rudkouskaya A, Chernoguz A, Haskew-Layton RE, Mongin AA. Two conventional protein kinase C isoforms, α and βI, are involved in the ATP-induced activation of volume-regulated anion channel and glutamate release in cultured astrocytes. J Neurochem. 2008;105:2260–2270. doi: 10.1111/j.1471-4159.2008.05312.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirai Y, Adachi N, Saito N. Protein kinase Cε: function in neurons. FEBS J. 2008;275:3988–3994. doi: 10.1111/j.1742-4658.2008.06556.x. [DOI] [PubMed] [Google Scholar]
- Sivaraman V, Hausenloy DJ, Kolvekar S, Hayward M, Yap J, Lawrence D, et al. The divergent roles of protein kinase Cε and δ in simulated ischaemia-reperfusion injury in human myocardium. J Mol Cell Cardiol. 2009;46:758–764. doi: 10.1016/j.yjmcc.2009.02.013. [DOI] [PubMed] [Google Scholar]
- Slepko N, Patrizio M, Levi G. Expression and translocation of protein kinase C isoforms in rat microglial and astroglial cultures. J Neurosci Res. 1999;57:33–38. doi: 10.1002/(SICI)1097-4547(19990701)57:1<33::AID-JNR4>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- Steinhart R, Kazimirsky G, Okhrimenko H, Ben-Hur T, Brodie C. PKCε induces astrocytic differentiation of multipotential neural precursor cells. Glia. 2007;55:224–232. doi: 10.1002/glia.20454. [DOI] [PubMed] [Google Scholar]
- Uchino M, Sakai N, Kashiwagi K, Shirai Y, Shinohara Y, Hirose K, et al. Isoform-specific phosphorylation of metabotropic glutamate receptor 5 by protein kinase C (PKC) blocks Ca2+ oscillation and oscillatory translocation of Ca2+-dependent PKC. J Biol Chem. 2004;279:2254–2261. doi: 10.1074/jbc.M309894200. [DOI] [PubMed] [Google Scholar]
- Young SH, Wu SV, Rozengurt E. Ca2+-stimulated Ca2+ oscillations produced by the Ca2+-sensing receptor require negative feedback by protein kinase C. J Biol Chem. 2002;277:46871–46876. doi: 10.1074/jbc.M207083200. [DOI] [PubMed] [Google Scholar]


