Abstract
Background
Although studies have reported a high prevalence of end-stage renal disease in human immunodeficiency virus (HIV)-infected individuals, little is known about moderate impairments in kidney function. Cystatin C measurement may be more sensitive than creatinine for detecting impaired kidney function in persons with HIV.
Methods
We evaluated kidney function in the Fat Redistribution and Metabolic Change in HIV Infection (FRAM) cohort, a representative sample of 1008 HIV-infected persons and 290 controls from the Coronary Artery Risk Development in Young Adults (CARDIA) study in the United States.
Results
Cystatin C level was elevated in HIV-infected individuals; the mean±SD cystatin C level was 0.92±0.22 mg/L in those infected with HIV and 0.76±0.15 mg/L in controls (P<.001). In contrast, both mean creatinine levels and estimated glomerular filtration rates appeared similar in HIV-infected individuals and controls (0.87±0.21 vs 0.85±0.19 mg/dL [to convert to micromoles per liter, multiply by 88.4] [P=.35] and 110±26 vs 106±23 mL/min/1.73 m2 [P=.06], respectively). Persons with HIV infection were more likely to have a cystatin C level greater than 1.0 mg/L (OR, 9.8; 95% confidence interval, 4.4-22.0 [P<.001]), a threshold demonstrated to be associated with increased risk for death and cardiovascular and kidney disease. Among participants with HIV, potentially modifiable risk factors for kidney disease, hypertension, and low high-density lipoprotein concentration were associated with a higher cystatin C level, as were lower CD4 lymphocyte count and coinfection with hepatitis C virus (all P<.001).
Conclusions
Individuals infected with HIV had substantially worse kidney function when measured by cystatin Clevel compared with HIV-negative controls, whereas mean creatinine levels and estimated glomerular filtration rates were similar. Cystatin C measurement could be a useful clinical tool to identify HIV-infected persons at increased risk for kidney and cardiovascular disease.
Kidney disease has been identified as an important complication of human immunodeficiency virus (HIV) infection.1 Investigators have noted the increasing prevalence of HIV in the population with end-stage renal disease, as well as the high prevalence of kidney disease in persons with HIV.2-5 This high prevalence may be owing to the longer life expectancy in the highly active antiretroviral therapy era. Whether HIV infection has a direct effect on kidney function is not known. In addition, there is substantial evidence that HIV infection is independently associated with many risk factors for kidney disease, such as adverse changes in lipid levels,6,7 increased insulin resistance,8,9 and microalbuminuria.10 Taken together, these factors illustrate the need for a population-based study of kidney function in HIV-infected individuals and healthy controls.
Cystatin C is an alternative measure of kidney function that is more sensitive in detecting moderate kidney disease than creatinine-based measures in the general population. Dharnidharka et al11 conducted a meta-analysis that found cystatin C to be superior to creatinine as a marker of glomerular filtration rate (GFR). Studies have noted cystatin C level to be a more sensitive marker of kidney function in the setting of chronic disease or aging, conditions often accompanied by a change in body composition.12-17 Creatinine-based measures of kidney function are limited in these populations, likely because of the association of creatinine with muscle mass. Although creatinine-based equations have been developed to adjust for demographic characteristics that predict muscle mass, these equations have not been validated in HIV-infected subjects.1
The present study compared the kidney function of HIV-positive individuals and HIV-negative controls using both creatinine and cystatin C measurements in the Fat Redistribution and Metabolic Change in HIV Infection (FRAM) cohort, a representative sample of HIV-infected participants with age-matched, population-based controls in the United States. In addition, we compared the respective associations of cystatin C and creatinine level with lean body mass and examined predictors of higher cystatin C level in HIV-infected participants and HIV-negative controls.
METHODS
STUDY DESIGN
This was a cross-sectional analysis of 1008 HIV-infected participants and 290 controls in the FRAM study with cystatin C or creatinine measurements. The FRAM study was designed to evaluate the prevalence and correlates of changes in fat distribution, insulin resistance, and dyslipidemia in a representative sample of HIV-positive participants and HIV-negative controls in the United States.18,19 The methods have been described in detail previously.18,19 Individuals infected with HIV were recruited from 16 HIV or infectious disease clinics or cohorts in 1999.19 Control participants were selected from the Visceral Fat and Metabolic Rate in Young Adults (VIM) study, which is an ancillary study to the Coronary Artery Risk Development in Young Adults (CARDIA) study.18 The CARDIA study includes a population-based sample of healthy 18- to 30-year-old white and African American women and men in the United States. Participants in the VIM ancillary study were from 2 centers (Oakland, California, and Birmingham, Alabama) and had previously participated in rigorous fat and metabolic tests that were part of the FRAM protocol. Participants in the VIM study were similar in age and race distribution to HIV-infected persons in the United States and had a body mass index distribution similar to the parent CARDIA study. The protocol was approved by institutional review boards at all sites.
Analyses were restricted to African Americans and whites with cystatin C or creatinine measurements. Ten participants with self-reported kidney disease were excluded. Analyses that compared HIV-infected persons with controls excluded HIV-infected individuals with recent opportunistic infections and were restricted to those between the ages of 33 and 45 years (n=519), since the control population only included participants within this age range.
KIDNEY FUNCTION
Cystatin C was measured in serum specimens, previously frozen and stored at −70°C, with a BNII nephelometer (Dade Behring Inc, Deerfield, Illinois) that used a particle-enhanced immunonephelometric assay (N Latex Cystatin C).20 The coefficient of variation for between-run imprecision was less than 2% for cystatin C.20 Additional characteristics of the assay have been reported previously.21
Creatinine level was used as a comparative measurement of kidney function, assayed by the enzymatic method at Covance Laboratories, Indianapolis, Indiana. Estimated GFR (eGFR) was calculated using the abbreviated (4-variable) Modification of Diet in Renal Disease (MDRD) equation,22 as recommended by the National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative. Prior literature found an increased risk of poor kidney and cardiovascular outcomes associated with a cystatin C level of greater than 1.0 mg/L15,23; therefore, this categorization was added as a secondary outcome. Because 4% of controls had a cystatin C level greater than 1.0 mg/L, a comparable creatinine-based end point was an eGFR lower than 75 mL/min/1.73 m2, which was the fourth percentile in the control population. Chronic kidney disease (CKD) was defined as an eGFR lower than 60 mL/min/1.73 m.2,24
OTHER MEASUREMENTS
Age, sex, ethnicity, medical history, and risk factors for HIV were determined by self-report, and alcohol, tobacco, and illicit drug use were assessed by standardized questionnaire. Height, weight, and blood pressure were measured by standardized protocols, and whole body magnetic resonance imaging was performed to quantify lean body mass.25 A single laboratory measured lipid levels, glucose metabolism, and other markers from blood specimens (Covance Laboratories). A random urine sample was collected to measure albumin, creatinine, and glucose levels. Hypertension was defined as a systolic blood pressure higher than 140 mm Hg, a diastolic blood pressure higher than 90 mm Hg, or medication use, and diabetes was defined as a fasting glucose level of 126 mg/dL or greater (to convert to millimoles per liter, multiply by 0.0555) or medication use. CD4 lymphocyte count and percentage and HIV RNA levels were measured in HIV-infected participants, and research associates performed medical chart abstraction of medications and medical history at HIV clinic sites. The duration of HIV infection was measured as the time since first positive HIV diagnosis. Hepatitis C virus (HCV) RNA levels were determined using the Bayer Versant 3.0 (bDNA) assay (Bayer Diagnostics, Berkeley, California), and values of 615 IU/mL or greater were defined as HCV positive.
STATISTICAL ANALYSIS
Characteristics of HIV-infected participants and controls were compared and tested for statistical significance using an unpaired t test for continuous variables, and Fisher exact test for categorical variables. The Mann-Whitney test was used when nonnormality was found.
Multivariable linear regression models were used to examine the association of HIV infection with cystatin C, creatinine, and eGFR levels and were built using stepwise regression, with P≤.05 as the criterion for entry and retention. Sex, age, ethnicity, and HIV status were forced to be included in every model. Additional candidate variables for inclusion in the multivariable models included tobacco use; hypertension; diabetes; systolic blood pressure; glucose, high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C), triglyceride, C-reactive protein, and uric acid levels; body mass index; angiotensin-converting enzyme inhibitor or angiotensin receptor blocker use; albumin-creatinine ratio; proteinuria; and current illicit drug use. In addition, the models were adjusted for total lean body mass to account for differences in creatinine generation. Interactions between sex and other factors in the model were assessed. Confidence intervals (CIs) were determined using the bias-corrected accelerated bootstrap method because the error residuals appeared nonnormal. In addition, with the same stepwise method and candidate predictors, multivariable logistic regression models were used to determine if HIV infection was associated with elevated cystatin C level (>1.0 mg/L) or reduced eGFR (<75 mL/min/1.73 m2). Logistic regression models were assessed at each stage using the Hosmer-Lemeshow goodness of fit test.
InHIV-infected participants, the associations of cystatin Clevel, creatinine level, and eGFR with lean body mass were tested using the Spearman rank correlation test. Separate multivariable regression analyses were performed among HIV-infected participants and controls to determine which factors were predictive of cystatin C levels. Sex, age, and ethnicity were forced to be included in both HIV and control models, and HIV RNA level (log10) and CD4 lymphocyte count (log2) at the time of study visit were forced to be included in the HIV model. In addition to the aforementioned variables, candidates related to HIV infection included AIDS diagnosis by CD4 lymphocyte count or opportunistic infection, HIV duration, HCV infection (by virus detection), recent opportunistic infection status, days since last opportunistic infection, and HIV risk factors. In multivariable models controlling for the aforementioned factors that were independent predictors, we evaluated the total duration of use of each individual antiretroviral drug and antiretroviral class.
All statistical analyses were conducted using the SAS system, version 9.1 (SAS Institute Inc, Cary, NC).
RESULTS
On average, participants with HIV infection were approximately 6 months younger and more often male and smokers compared with those without HIV (Table 1). The HIV-infected participants were more likely to report a history of hypertension and diabetes, although they had lower mean systolic blood pressure and were more likely to be taking an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker. Human immunodeficiency virus infection was also associated with lower BMI and HDL-C and LDL-C levels; higher triglyceride, C-reactive protein, and uric acid levels; and a greater prevalence of an albumin-creatinine ratio greater than 30 or proteinuria.
Table 1.
Characteristics of HIV-Infected and Control Participantsa
| Variable | HIV (n=519) |
Control (n=290) |
P Value |
|---|---|---|---|
| Demographic/clinical characteristic | |||
| Age, y | 39.5±3.6 | 40.1±3.5 | .02 |
| African American | 225 (43) | 139 (48) | .21 |
| Female sex | 154 (30) | 138 (48) | <.001 |
| Current smoker | 242 (49) | 48 (17) | <.001 |
| Diabetes | 32 (6) | 9 (3) | .07 |
| Hypertension | 115 (23) | 45 (16) | .02 |
| Systolic blood pressure, mm Hg | 115±14 | 118±16 | .002 |
| ACE/ARB use | 33 (6) | 4 (1) | <.001 |
| BMI | 25.5±5.0 | 27.9±5.6 | <.001 |
| Glucose | |||
| <100 mg/dL | 405±78 | 235±81 | .14 |
| 100-125 mg/dL | 88±17 | 48 ± 17 | |
| >125 mg/dL | 25±5 | 6±2 | |
| HDL-C, mg/dL | 44±16 | 52±15 | <.001 |
| LDL-C, mg/dL | 107±42 | 123±34 | <.001 |
| Triglycerides, mg/dL | 217±224 | 120±122 | <.001b |
| CRP, μg/mL | 4.0±8.0 | 3.1±6.0 | .001b |
| Uric acid, mg/dL | 5.9±1.7 | 5.3±1.5 | <.001 |
| Proteinuria or ACR >30 | 70 (17) | 13 (6) | <.001 |
| Lean body mass, L | 28.2±5.3 | 28.4±5.8 | .67 |
| HIV-associated variable | |||
| Current CD4 lymphocyte count, cells/uL |
405 (257) | NA |
NA |
| HIV RNA, ×103/mL | 33.5±97.9 | NA | NA |
| Duration HIV, y | 8.6±3.9 | NA | NA |
| Hepatitis C virus | 98 (19) | 5 (2) | <.001 |
| Heroin use | 10 (2) | NA | NA |
Abbreviations: ACE, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index (calculated as weight in kilograms divided by height in meters squared); CRP, C-reactive protein; HDL-C, high-density lipoprotein cholesterol; HIV, human immunodeficiency virus; LDL-C, low-density lipoprotein cholesterol; NA, not applicable.
SI conversion factors: To convert to millimoles per liter, multiply by 0.0555 for glucose, by 0.0259 for cholesterol, and by 0.0113 for triglycerides; to convert CRP to nanomoles per liter, multiply by 9.524; to convert uric acid to micromoles per liter, multiply by 59.48.
Data are given as number (percentage of patients) or mean±SD value unless otherwise specified.
P values were calculated using the Mann-Whitney test; a t test was used for all other continuous variables.
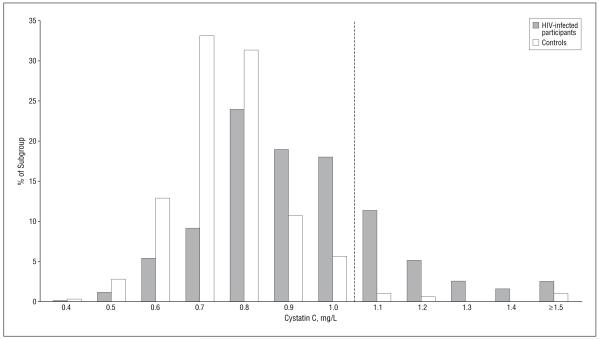
Cystatin C level was higher in HIV-infected individuals; the mean±SD cystatin C level was 0.92±0.22 mg/L in those infected with HIV and 0.76±0.15 mg/L in controls (P<.001) (Table 2). The distribution of cystatin C levels in the HIV-infected vs control participants is shown in Figure 1. In multivariable models, adjustment for demographic and clinical characteristics attenuated the difference by about 30%, but it remained significant (Table 2). In contrast, mean creatinine levels were similar in HIV-infected and control participants in both unadjusted and adjusted models (Table 2). No significant interactions were observed between race or sex and any factor in the models.
Table 2.
A Comparison of Cystatin C Level and Creatinine Level as Measures of Kidney Function in HIV-Infected and Control Participants
| Measure of Kidney Function | HIV-Infected Individuals (n=519) |
Controls (n=290) |
P Value |
|---|---|---|---|
| Cystatin C, mean±SD, mg/L | 0.92±0.22 | 0.76±0.15 | <.001 |
| Unadjusted difference (95% CI)a | 0.16 (0.14 to 0.19) | … | <.001 |
| Adjusted difference (95% CI)a,b | 0.11 (0.08 to 0.14) | … | <.001 |
| Cystatin C >1.0 mg/L, No. (%) | 156 (31) | 11 (4) | <.001 |
| Unadjusted OR (95% CI)a | 10.8 (5.7 to 20.2) | … | <.001 |
| Adjusted OR (95% CI)a,b | 9.8 (4.4 to 22.0) | … | <.001 |
| Creatinine, mean±SD, mg/dL | 0.87±0.21 | 0.85±0.19 | .35 |
| Unadjusted difference (95% CI)a | 0.02 (−0.01 to 0.05) | … | .19 |
| Adjusted difference (95% CI)a,b | 0.00 (−0.03 to 0.03) | … | .78 |
| eGFR, mean±SD, mL/min/1.73 m2 | 110±26 | 106±23 | .06 |
| Unadjusted difference (95% CI)a | 4 (0 to 7) | … | .04 |
| Adjusted difference (95% CI)a,b | 2 (−2 to 7) | … | .35 |
| eGFR <75 mL/min/1.73 m2, No. (%) | 28 (5) | 13 (4) | .62 |
| Unadjusted OR (95% CI)a | 1.22 (0.62 to 2.39) | … | .57 |
| Adjusted OR (95% CI)a,b | 1.28 (0.56 to 2.92) | … | .55 |
Abbreviations: CI, confidence interval; eGFR, estimated glomerular filtration rate; HIV, human immunodeficiency virus; OR, odds ratio.
SI conversion factor: To convert creatinine to micromoles per liter, multiply by 88.4.
Participants with HIV vs controls.
Sex, age, and ethnicity were included in all models; other candidate variables were selected by a stepwise procedure.
Figure 1.
Distribution of cystatin C levels in human immunodeficiency virus (HIV)-infected vs control participants. The dotted line represents the cut point for preclinical kidney disease (>1.0 mg/L).
The prevalence of cystatin C level greater than 1.0 mg/L was 31% in the HIV-infected participants compared with only 4% in controls (P<.001). Human immunodeficiency virus infection was associated with a greater than 10-fold odds of having a cystatin C level greater than 1.0 mg/L (OR, 10.8; 95% CI, 5.7 to 20.2 [P<.001]), which remained high after multivariable adjustment (odds ratio, 9.8; 95% CI, 4.4 to 22.0 [P<.001]).
Only 28 of the 519 HIV-infected participants (5%) had an eGFR lower than 75 mL/min/1.73 m2 compared with 13 of the 290 controls (4%) (P=.62). Human immunodeficiency virus infection was associated with a slightly increased odds for lower eGFR in unadjusted and adjusted models, but this effect did not reach statistical significance (Table 2). Individuals infected with HIV had a similar prevalence of an eGFR lower than 60 mL/min/1.73 m2 (CKD) compared with controls, although the overall prevalence was low (2% [10 of 519] in HIV-infected participants compared with 1% [4 of 290] of controls [P=.78]). Contrary to expectation, the mean eGFR was slightly higher in HIV-infected participants compared with controls, indicating better kidney function, but this comparison was not significant in the adjusted model.
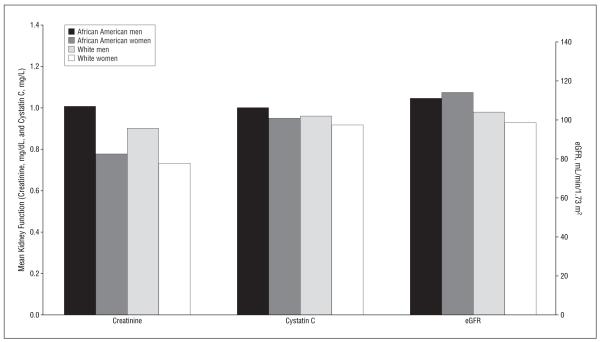
In an unadjusted model of HIV-infected participants, cystatin C level had little correlation with lean body mass (r=−0.01; 95% CI, −0.08 to 0.06 [P=.87]), whereas creatinine level was positively correlated (r=0.34; 95% CI, 0.28 to 0.40 [P<.001]). The use of the creatinine-based MDRD equation removed this association; the correlation of eGFR with lean body mass was r=−0.04 (95% CI, −0.11 to 0.03) (P=.27). In HIV-infected participants, there was substantially less variation in mean cystatin C levels across ethnicity and sex strata compared with creatinine levels, although use of the MDRD equation partially attenuated the variation (Figure 2).
Figure 2.
Cystatin C level, creatinine level (to convert to micromoles per liter, multiply by 88.4), and estimated glomerular filtration rate (eGFR) by ethnicity and sex strata in human immunodeficiency virus–infected participants.
We next used multivariable models to examine factors associated with higher cystatin C level, separately among HIV-infected individuals and controls (Table 3). In HIV-infected participants, risk factors for kidney disease associated with higher cystatin C level included lower HDL-C level, higher uric acid level, albumin-creatinine ratio greater than 30 or proteinuria, hypertension, and higher C-reactive protein level. Unexpectedly, a glucose level greater than 125 mg/dL was associated with lower cystatin C level compared with a glucose level lower than 100 mg/dL, although the association of a glucose level between 100 and 125 mg/dL with cystatin C was similar to a glucose level lower than 100 mg/dL. Lower HDL-C level, higher uric acid level, and albumin-creatinine ratio greater than 30 or proteinuria were also associated with a higher cystatin C level in controls, as was current smoking. Similar trends were seen when hypertension, C-reactive protein level, and glucose level greater than 125 mg/dL were forced into the control model and smoking was forced into the HIV model. Factors associated with HIV that were associated with a higher cystatin C level included decreased current CD4 lymphocyte count, coinfection with HCV, current heroin use, and longer duration of efavirenz and indinavir sulfate use. Participants with HIV and HCV coinfection had substantially higher prevalence of cystatin C levels greater than 1.0 mg/L compared with those with HIV alone (54% vs 31%) (P<.001).
Table 3.
Independent Predictors of Cystatin C Level in HIV-Infected and Control Participants
| Variable | HIV-Infected Participants, Coefficient (95% CI)a (n=1008) |
P Value | Controls, Coefficient (95% CI)a (n=290) |
P Value |
|---|---|---|---|---|
| Risk factors for kidney disease (per mg/L higher cystatin C level) | ||||
| Log HDL-C (mg/dL) | −0.13 (−0.27 to −0.06) | <.001 | −0.15 (−0.23 to 0.08) | <.001 |
| Uric acid (mg/dL) | 0.05 (0.04 to 0.06) | <.001 | 0.05 (0.03 to 0.08) | <.001 |
| ACR >30 or proteinuria | 0.07 (0.01 to 0.13) | .02 | 0.21 (0.06 to 0.48) | .006 |
| Hypertension | 0.08 (0.04 to 0.13) | <.001 | … | … |
| Log CRP (μg/mL) | 0.02 (0.00 to 0.03) | .007 | … | … |
| Glucose | ||||
| 100-125 vs <100 mg/dL | −0.01 (−0.05 to 0.03) | .55 | … | … |
| >125 vs <100 mg/dL | −0.14 (−0.21 to −0.06) | .001 | … | … |
| Current smoker | … | … | 0.07 (0.02 to 0.14) | .001 |
| HIV-associated factors | ||||
| Log current CD4 lymphocyte count | −0.03 (−0.06 to −0.02) | <.001 | … | … |
| Hepatitis C virus | 0.12 (0.07 to 0.16) | <.001 | … | … |
| Current heroin use | 0.17 (0.02 to 0.55) | .02 | … | … |
| Total duration of efavirenz use | 0.03 (0.01 to 0.07) | .007 | … | … |
| Total duration of indinavir sulfate use | 0.01 (0.00 to 0.02) | .03 | … | … |
Abbreviations: ACR, albumin-creatinine ratio; CI, confidence interval; CRP, C-reactive protein; HDL-C, high-density lipoprotein cholesterol; HIV, human immunodeficiency virus; ellipses, variables with P values >.05 were not retained in the model.
SI conversion factor: To convert glucose to millimoles per liter, multiply by 0.0555.
Coefficients are reported per 1 mg/L higher cystatin C level. The model includes adjustment for age, sex, ethnicity, and current HIV viral load (in HIV-infected participants).
COMMENT
Individuals infected with HIV had substantially worse kidney function, as measured by cystatin C level, compared with HIV-negative controls, even after adjustment for demographic characteristics and clinical factors. After multivariable adjustment, the HIV-infected participants had more than a 9-fold increased odds of having a cystatin C level greater than 1.0 mg/L, a level that was demonstrated to be associated with an increased risk for death and cardiovascular and kidney disease in an elderly cohort.23 In contrast, there was little difference in creatinine levels between HIV-positive individuals and controls. Creatinine level may not adequately capture kidney function because of its strong associations with ethnicity, sex, and body composition. While eGFR equations are often used to adjust for these factors, eGFRs were also similar in HIV infection and in controls. In participants with HIV, hypertension and low HDL-C level—potentially modifiable risk factors for kidney disease—were associated with worse kidney function, as were lower CD4 lymphocyte count and coinfection with HCV.
Several studies have noted an increased prevalence of kidney disease in the HIV-infected population, as well as an increased prevalence of HIV-infection in the population with end-stage renal disease.1-5 The majority of studies of HIV and kidney disease have focused on end-stage renal disease or HIV-associated nephropathy, a condition marked by proteinuria and glomerulopathy, but few have compared kidney function in HIV-infected individuals and noninfected controls. One prior study from the FRAM cohort found approximately a 5-fold odds for microalbuminuria in HIV-infected individuals compared with controls, and predictors of microalbuminuria included both risk factors for kidney disease and markers of HIV severity.10 Our findings expand on a small study of HIV-infected individuals (n=77) and HIV-negative volunteers (n=18) in Poland. The investigators found that cystatin C levels were elevated in HIV infection and were correlated with viral load; however, there were no significant differences in eGFR levels26
A recent study described elevated cystatin C level (>1.0 mg/L) in the setting of normal eGFR as “preclinical kidney disease,” and this condition has been associated with increased risk of progression to CKD, cardiovascular disease, and death.23 Participants without CKD who had cystatin C concentrations greater than 1.0 mg/L had a 4-fold risk for progressing to CKD after 4 years of follow-up compared with participants without elevated cystatin C level. Using this cystatin C-based definition, we found that approximately one-third of persons with HIV have pre-clinical kidney disease, a condition that was undetected by creatinine-based measures. If truly reflective of underlying kidney dysfunction, elevated cystatin C concentrations may identify persons with a greater risk for kidney toxic effects from medication use, worsening kidney disease, cardiovascular disease, or death. Since only 2% had an eGFR lower than 60 mL/min/1.73 m2, this potential risk would be missed using traditional measures of kidney function.
Although the present study did not include a direct measure of kidney function, there is substantial evidence that a higher cystatin C level reflects worse kidney function.11,14,27 Several studies have reported the superiority of cystatin C over creatinine measurement for detecting early changes in kidney function, especially in the setting of chronic disease or aging.13,14,23,28 One cross-sectional study compared HIV-positive patients using antiretroviral regimens with or without tenofovir and found that tenofovir use was associated with reduced kidney function using measurements of cystatin C level or 24-hour creatinine clearance but found no significant difference in kidney function when measured by the MDRD formula.29 The authors concluded that the mild nephrotoxic effects may have been missed by the simple formula-based estimation of kidney function. The insensitivity of creatinine measurement for capturing small changes in kidney function may explain why few studies have reported an association of HIV infection with impaired kidney function.
Several risk factors for kidney disease were more prevalent in persons with HIV infection. Hypertension, hypertriglyceridemia, inflammation, diabetes, hyperuricemia, and microalbuminuria are all well-described risk factors for reduced kidney function that were associated with a higher cystatin C level in HIV-infected participants.30-35 Adjustment for risk factors moderately reduced the magnitude of the association between HIV and cystatin C level in the linear models, suggesting that some of the elevated renal risk observed in HIV infection may be due to the increased levels of these factors. If these findings are confirmed, then future studies should evaluate whether therapies that target modifiable risk factors can delay the progression of kidney disease in HIV-infected persons.
Risk factors associated with HIV were also strongly and independently associated with higher cystatin C level, including lower CD4 lymphocyte count, which is indicative of immune dysfunction, and HCV. This suggests that in addition to the adverse metabolic effects of HIV, severity of HIV infection and HCV coinfection may exacerbate the diminished kidney function observed in these participants. Consistent with other literature, heroin use was associated with worse kidney function, although the prevalence of heroin use was low in this population and thus would not explain much of the HIV effect.36
Total duration of 2 antiretroviral therapies, efavirenz and indinavir, were also modestly associated with worse kidney function; this association may be due to direct pathogenic effects of the medications or may be a surrogate for longer or more severe HIV infection. It is important to note that our study population was recruited before the widespread use of tenofovir, the most widely cited therapy with potential nephrotoxic effects.
An alternative explanation for elevated cystatin C level may be altered production or secretion of cystatin C in HIV infection. Without a gold standard for kidney function, we are unable to conclude definitively that the observed elevations in cystatin C concentrations are indicative of diminished kidney function in HIV infection. Finally, there may have been incomplete or inadequate control for factors that confound the association of HIV infection and cystatin C level.
In conclusion, HIV infection appears to be associated with substantially worse kidney function when measured by cystatin C level, whereas creatinine levels and eGFRs were similar in HIV-infected individuals and controls. Future studies that use a direct measure of kidney function could help to define the optimal role of cystatin C for detecting reduced kidney function in HIV or other chronic infection. Early identification of kidney dysfunction may permit actions that prevent or reduce the risk of toxic effects from medication use, further renal complications, or cardiovascular outcomes. Longitudinal studies may better elucidate the HIV-associated physiologic processes that accompany declines in kidney function and thus facilitate the development of preventive therapies.
Acknowledgments
Funding/Support: The FRAM Study is supported by grants from the National Institutes of Health (R01-DK-57508, R01-HL-74814, and R01-HL-53359) including funds from the National Institute on Drug Abuse. National Institutes of Health support for this project is also provided through grants to General Clinical Research Centers (M01-RR00036, M01-RR00051, M01-RR00052, M01-RR00054, M01-RR00083, M01-RR00636, and M01-RR00865). Dr Szczech’s work is supported by grant DK02724-01A1 from the National Institutes of Health. Dr Shlipak is supported by grants R01 HL073208-01 and R01 DK066488-01, the American Federation for Aging Research and National Institute on Aging (Paul Beeson Scholars Program), and the Robert Wood Johnson Foundation (Generalist Faculty Scholars Program).
Role of the Sponsor: None of the funding agencies had any role in the design of the study, analysis of data, or preparation of the manuscript.
Footnotes
Author Contributions: Study concept and design: Odden, Grunfeld, and Shlipak. Acquisition of data: Sidney, Grunfeld, and Shlipak. Analysis and interpretation of data: Odden, Scherzer, Bacchetti, Szczech, Grunfeld, and Shlipak. Drafting of the manuscript: Odden and Scherzer. Critical revision of the manuscript for important intellectual content: Odden, Scherzer, Bacchetti, Szczech, Sidney, Grunfeld, and Shlipak. Statistical analysis: Scherzer and Bacchetti. Obtained funding: Grunfeld. Administrative, technical, and material support: Odden. Study supervision: Odden, Grunfeld, and Shlipak.
Financial Disclosure: None reported.
REFERENCES
- 1.Gupta SK, Eustace JA, Winston JA, et al. Guidelines for the management of chronic kidney disease in HIV-infected patients: recommendations of the HIV Medicine Association of the Infectious Diseases Society of America. Clin Infect Dis. 2005;40(11):1559–1585. doi: 10.1086/430257. [DOI] [PubMed] [Google Scholar]
- 2.Eggers PW, Kimmel PL. Is there an epidemic of HIV infection in the US ESRD program? J Am Soc Nephrol. 2004;15(9):2477–2485. doi: 10.1097/01.ASN.0000138546.53152.A7. [DOI] [PubMed] [Google Scholar]
- 3.Gardner LI, Holmberg SD, Williamson JM, et al. HIV Epidemiology Research Study Group Development of proteinuria or elevated serum creatinine and mortality in HIV-infected women. J Acquir Immune Defic Syndr. 2003;32(2):203–209. doi: 10.1097/00126334-200302010-00013. [DOI] [PubMed] [Google Scholar]
- 4.Gupta SK, Mamlin BW, Johnson CS, Dollins MD, Topf JM, Dube MP. Prevalence of proteinuria and the development of chronic kidney disease in HIV-infected patients. Clin Nephrol. 2004;61(1):1–6. doi: 10.5414/cnp61001. [DOI] [PubMed] [Google Scholar]
- 5.Szczech LA, Gange SJ, van der Horst C, et al. Predictors of proteinuria and renal failure among women with HIV infection. Kidney Int. 2002;61(1):195–202. doi: 10.1046/j.1523-1755.2002.00094.x. [DOI] [PubMed] [Google Scholar]
- 6.Grunfeld C, Kotler DP, Hamadeh R, Tierney A, Wang J, Pierson RN. Hypertriglyceridemia in the acquired immunodeficiency syndrome. Am J Med. 1989;86(1):27–31. doi: 10.1016/0002-9343(89)90225-8. [DOI] [PubMed] [Google Scholar]
- 7.Grunfeld C, Pang M, Doerrler W, Shigenaga JK, Jensen P, Feingold KR. Lipids, lipoproteins, triglyceride clearance, and cytokines in human immunodeficiency virus infection and the acquired immunodeficiency syndrome. J Clin Endocrinol Metab. 1992;74(5):1045–1052. doi: 10.1210/jcem.74.5.1373735. [DOI] [PubMed] [Google Scholar]
- 8.El-Sadr WM, Mullin CM, Carr A, et al. Effects of HIV disease on lipid, glucose and insulin levels: results from a large antiretroviral-naive cohort. HIV Med. 2005;6(2):114–121. doi: 10.1111/j.1468-1293.2005.00273.x. [DOI] [PubMed] [Google Scholar]
- 9.Hadigan C, Meigs JB, Corcoran C, et al. Metabolic abnormalities and cardiovascular disease risk factors in adults with human immunodeficiency virus infection and lipodystrophy. Clin Infect Dis. 2001;32(1):130–139. doi: 10.1086/317541. [DOI] [PubMed] [Google Scholar]
- 10.Szczech LA, Grunfeld C, Scherzer R, et al. Microalbuminuria in HIV infection: the FRAM Study. AIDS. 2007;21(8):1003–1009. doi: 10.1097/QAD.0b013e3280d3587f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dharnidharka VR, Kwon C, Stevens G. Serum cystatin C is superior to serum creatinine as a marker of kidney function: a meta-analysis. Am J Kidney Dis. 2002;40(2):221–226. doi: 10.1053/ajkd.2002.34487. [DOI] [PubMed] [Google Scholar]
- 12.Odden MC, Chertow GM, Fried LF, et al. the HABC Study Cystatin C and measures of physical function in elderly adults. Am J Epidemiol. 2006;164(12):1180–1189. doi: 10.1093/aje/kwj333. [DOI] [PubMed] [Google Scholar]
- 13.Orlando R, Mussap M, Plebani M, et al. Diagnostic value of plasma cystatin C as a glomerular filtration marker in decompensated liver cirrhosis. Clin Chem. 2002;48(6, pt 1):850–858. [PubMed] [Google Scholar]
- 14.Perkins BA, Nelson RG, Ostrander BE, et al. Detection of renal function decline in patients with diabetes and normal or elevated GFR by serial measurements of serum cystatin C concentration: results of a 4-year follow-up study. J Am Soc Nephrol. 2005;16(5):1404–1412. doi: 10.1681/ASN.2004100854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shlipak MG, Sarnak MJ, Katz R, et al. Cystatin C and the risk of death and cardiovascular events among elderly persons. N Engl J Med. 2005;352(20):2049–2060. doi: 10.1056/NEJMoa043161. [DOI] [PubMed] [Google Scholar]
- 16.Poge U, Gerhardt T, Stoffel-Wagner B, Klehr HU, Sauerbruch T, Woitas RP. Calculation of glomerular filtration rate based on cystatin C in cirrhotic patients. Nephrol Dial Transplant. 2006;21(3):660–664. doi: 10.1093/ndt/gfi305. [DOI] [PubMed] [Google Scholar]
- 17.Randers E, Ivarsen P, Erlandsen EJ, et al. Plasma cystatin C as a marker of renal function in patients with liver cirrhosis. Scand J Clin Lab Invest. 2002;62(2):129–134. doi: 10.1080/003655102753611753. [DOI] [PubMed] [Google Scholar]
- 18.Friedman GD, Cutter GR, Donahue RP, et al. CARDIA: study design, recruitment, and some characteristics of the examined subjects. J Clin Epidemiol. 1988;41(11):1105–1116. doi: 10.1016/0895-4356(88)90080-7. [DOI] [PubMed] [Google Scholar]
- 19.Tien PC, Benson C, Zolopa AR, Sidney S, Osmond D, Grunfeld C. The study of fat redistribution and metabolic change in HIV infection (FRAM): methods, design, and sample characteristics. Am J Epidemiol. 2006;163(9):860–869. doi: 10.1093/aje/kwj111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Erlandsen EJ, Randers E, Kristensen JH. Evaluation of the Dade Behring N Latex Cystatin C assay on the Dade Behring Nephelometer II System. Scand J Clin Lab Invest. 1999;59(1):1–8. doi: 10.1080/00365519950185940. [DOI] [PubMed] [Google Scholar]
- 21.Shlipak MG, Fyr CL, Chertow GM, et al. Cystatin C and mortality risk in the elderly: the health, aging, and body composition study. J Am Soc Nephrol. 2006;17(1):254–261. doi: 10.1681/ASN.2005050545. [DOI] [PubMed] [Google Scholar]
- 22.Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D, Modification of Diet in Renal Disease Study Group A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Ann Intern Med. 1999;130(6):461–470. doi: 10.7326/0003-4819-130-6-199903160-00002. [DOI] [PubMed] [Google Scholar]
- 23.Shlipak MG, Katz R, Sarnak MJ, et al. Cystatin C and prognosis for cardiovascular and kidney outcomes in elderly persons without chronic kidney disease. Ann Intern Med. 2006;145(4):237–246. doi: 10.7326/0003-4819-145-4-200608150-00003. [DOI] [PubMed] [Google Scholar]
- 24.Levey AS, Eckardt KU, Tsukamoto Y, et al. Definition and classification of chronic kidney disease: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO) Kidney Int. 2005;67(6):2089–2100. doi: 10.1111/j.1523-1755.2005.00365.x. [DOI] [PubMed] [Google Scholar]
- 25.Gallagher D, Belmonte D, Deurenberg P, et al. Organ-tissue mass measurement allows modeling of REE and metabolically active tissue mass. Am J Physiol. 1998;275(2, pt 1):E249–E258. doi: 10.1152/ajpendo.1998.275.2.E249. [DOI] [PubMed] [Google Scholar]
- 26.Jaroszewicz J, Wiercinska-Drapalo A, Lapinski TW, Prokopowicz D, Rogalska M, Parfieniuk A. Does HAART improve renal function? an association between serum cystatin C concentration, HIV viral load and HAART duration. Antivir Ther. 2006;11(5):641–645. [PubMed] [Google Scholar]
- 27.Herget-Rosenthal S, Trabold S, Pietruck F, Holtmann M, Philipp T, Kribben A. Cystatin C: efficacy as screening test for reduced glomerular filtration rate. Am J Nephrol. 2000;20(2):97–102. doi: 10.1159/000013564. [DOI] [PubMed] [Google Scholar]
- 28.Odden MC, Chertow GM, Fried LF, et al. the HABC Study Cystatin C and measures of physical function in elderly adults: the Health, Aging, and Body Composition (HABC) Study. Am J Epidemiol. 2006;164(12):1180–1189. doi: 10.1093/aje/kwj333. [DOI] [PubMed] [Google Scholar]
- 29.Mauss S, Berger F, Schmutz G. Antiretroviral therapy with tenofovir is associated with mild renal dysfunction. AIDS. 2005;19(1):93–95. doi: 10.1097/00002030-200501030-00012. [DOI] [PubMed] [Google Scholar]
- 30.Cases A, Coll E. Dyslipidemia and the progression of renal disease in chronic renal failure patients. Kidney Int Suppl. 2005;(99):S87–S93. doi: 10.1111/j.1523-1755.2005.09916.x. [DOI] [PubMed] [Google Scholar]
- 31.Fried L, Solomon C, Shlipak M, et al. Inflammatory and prothrombotic markers and the progression of renal disease in elderly individuals. J Am Soc Nephrol. 2004;15(12):3184–3191. doi: 10.1097/01.ASN.0000146422.45434.35. [DOI] [PubMed] [Google Scholar]
- 32.Jamerson KA. Preventing chronic kidney disease in special populations. Am J Hypertens. 2005;18(4, pt 2):106S–111S. doi: 10.1016/j.amjhyper.2004.11.018. [DOI] [PubMed] [Google Scholar]
- 33.Lea J, Greene T, Hebert L, et al. The relationship between magnitude of proteinuria reduction and risk of end-stage renal disease: results of the African American study of kidney disease and hypertension. Arch Intern Med. 2005;165(8):947–953. doi: 10.1001/archinte.165.8.947. [DOI] [PubMed] [Google Scholar]
- 34.Ritz E, Orth SR. Nephropathy in patients with type 2 diabetes mellitus. N Engl J Med. 1999;341(15):1127–1133. doi: 10.1056/NEJM199910073411506. [DOI] [PubMed] [Google Scholar]
- 35.Johnson RJ, Segal MS, Srinivas T, et al. Essential hypertension, progressive renal disease, and uric acid: a pathogenetic link? J Am Soc Nephrol. 2005;16(7):1909–1919. doi: 10.1681/ASN.2005010063. [DOI] [PubMed] [Google Scholar]
- 36.Dettmeyer RB, Preuss J, Wollersen H, Madea B. Heroin-associated nephropathy. Expert Opin Drug Saf. 2005;4(1):19–28. doi: 10.1517/14740338.4.1.19. [DOI] [PubMed] [Google Scholar]