Abstract
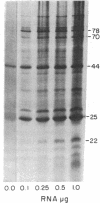
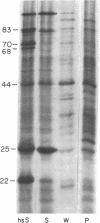
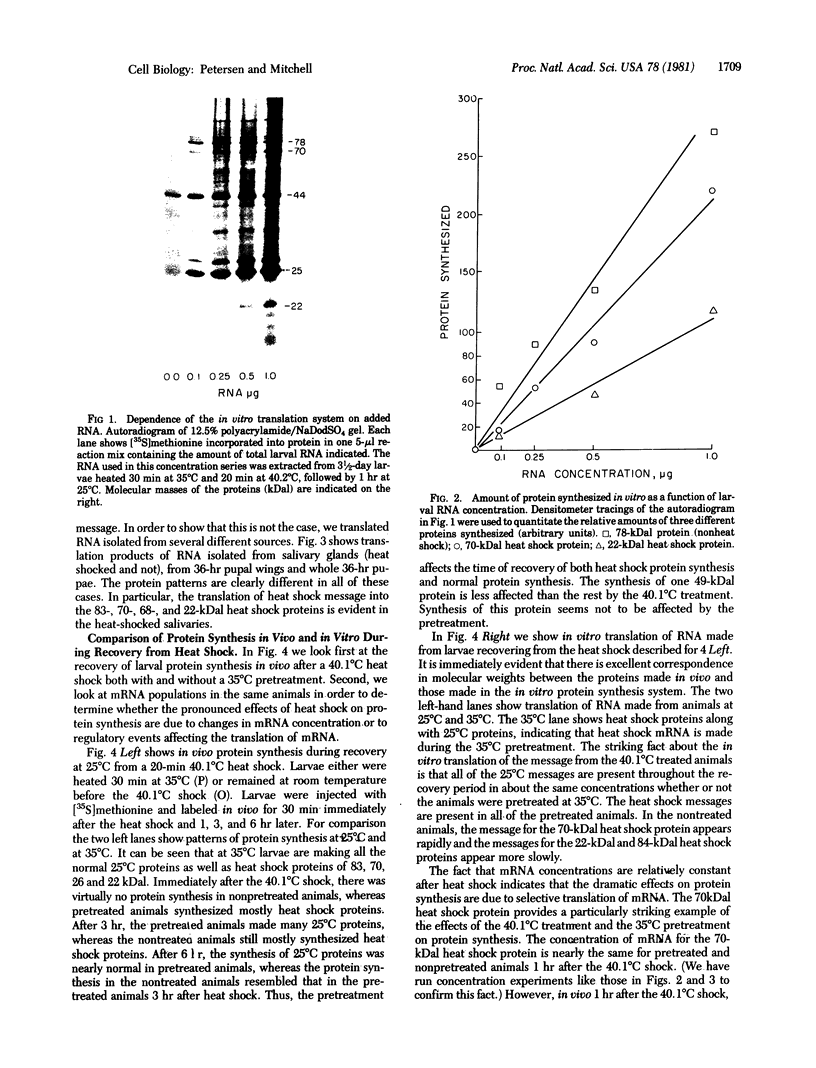
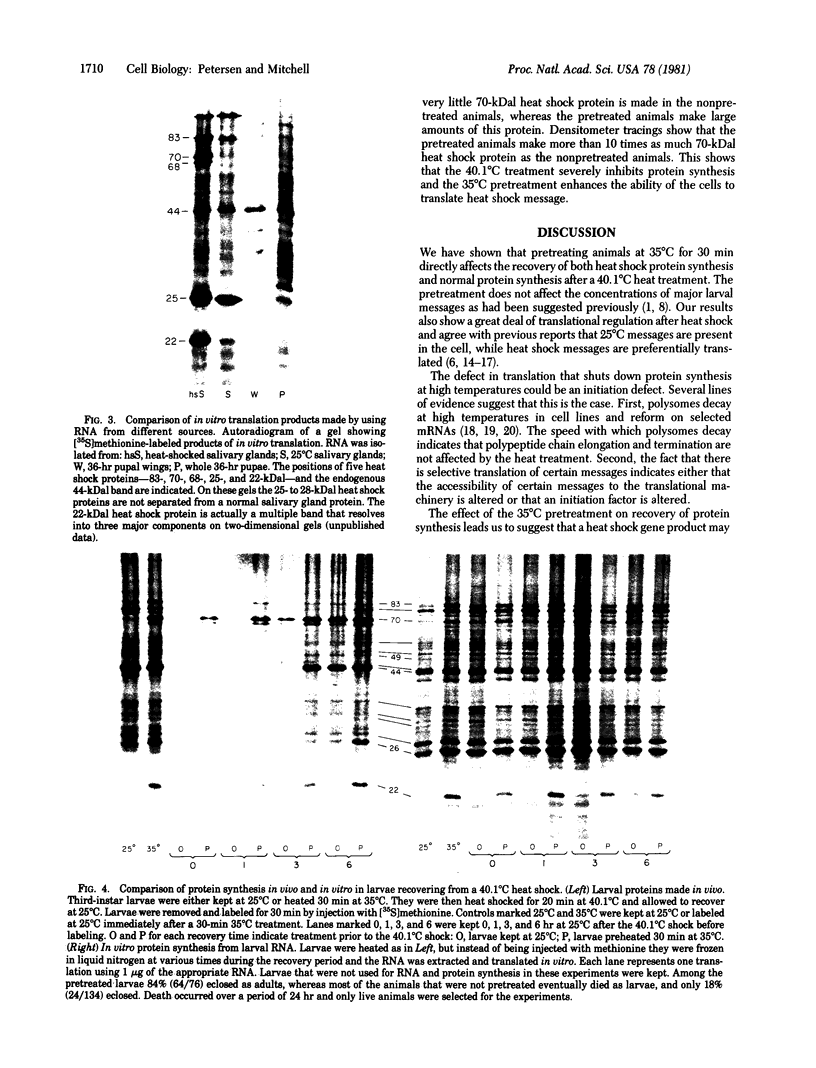
A mild heat shock at 35 degrees C, which induces heat shock gene expression, greatly enhances survival and the recovery of protein synthesis in Drosophila cells after a higher temperature heat shock. The 35 degrees C treatment is also effective in preventing heat-induced developmental defects in pupae. We show here that the major larval mRNAs are present in approximately normal (25 degrees C) concentrations after a 40.1 degrees C heat shock whether or not the animals receive a pretreatment. This indicates that the pretreatment affects translation directly rather than messenger concentration. We also observe selective translation of heat shock messages and some 25 degrees C messages during recovery from heat shock.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashburner M., Bonner J. J. The induction of gene activity in drosophilia by heat shock. Cell. 1979 Jun;17(2):241–254. doi: 10.1016/0092-8674(79)90150-8. [DOI] [PubMed] [Google Scholar]
- Ashburner M. Patterns of puffing activity in the salivary gland chromosomes of Drosophila. V. Responses to environmental treatments. Chromosoma. 1970;31(3):356–376. doi: 10.1007/BF00321231. [DOI] [PubMed] [Google Scholar]
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerner E. W., Schneider M. J. Induced thermal resistance in HeLa cells. Nature. 1975 Aug 7;256(5517):500–502. doi: 10.1038/256500a0. [DOI] [PubMed] [Google Scholar]
- Henle K. J., Karamuz J. E., Leeper D. B. Induction of thermotolerance in Chinese hamster ovary cells by high (45 degrees) or low (40 degrees) hyperthermia. Cancer Res. 1978 Mar;38(3):570–574. [PubMed] [Google Scholar]
- Kelley P. M., Schlesinger M. J. The effect of amino acid analogues and heat shock on gene expression in chicken embryo fibroblasts. Cell. 1978 Dec;15(4):1277–1286. doi: 10.1016/0092-8674(78)90053-3. [DOI] [PubMed] [Google Scholar]
- Li G. C., Hahn G. M. A proposed operational model of thermotolerance based on effects of nutrients and the initial treatment temperature. Cancer Res. 1980 Dec;40(12):4501–4508. [PubMed] [Google Scholar]
- Lindquist S. Varying patterns of protein synthesis in Drosophila during heat shock: implications for regulation. Dev Biol. 1980 Jun 15;77(2):463–479. doi: 10.1016/0012-1606(80)90488-1. [DOI] [PubMed] [Google Scholar]
- McAlister L., Finkelstein D. B. Heat shock proteins and thermal resistance in yeast. Biochem Biophys Res Commun. 1980 Apr 14;93(3):819–824. doi: 10.1016/0006-291x(80)91150-x. [DOI] [PubMed] [Google Scholar]
- McCormick W., Penman S. Regulation of protein synthesis in HeLa cells: translation at elevated temperatures. J Mol Biol. 1969 Jan;39(2):315–333. doi: 10.1016/0022-2836(69)90320-9. [DOI] [PubMed] [Google Scholar]
- McKenzie S. L., Henikoff S., Meselson M. Localization of RNA from heat-induced polysomes at puff sites in Drosophila melanogaster. Proc Natl Acad Sci U S A. 1975 Mar;72(3):1117–1121. doi: 10.1073/pnas.72.3.1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller M. J., Xuong N. H., Geiduschek E. P. A response of protein synthesis to temperature shift in the yeast Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5222–5225. doi: 10.1073/pnas.76.10.5222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirault M. E., Goldschmidt-Clermont M., Moran L., Arrigo A. P., Tissières A. The effect of heat shock on gene expression in Drosophila melanogaster. Cold Spring Harb Symp Quant Biol. 1978;42(Pt 2):819–827. doi: 10.1101/sqb.1978.042.01.082. [DOI] [PubMed] [Google Scholar]
- Mitchell H. K., Lipps L. S. Heat shock and phenocopy induction in Drosophila. Cell. 1978 Nov;15(3):907–918. doi: 10.1016/0092-8674(78)90275-1. [DOI] [PubMed] [Google Scholar]
- Palmiter R. D. Ovalbumin messenger ribonucleic acid translation. Comparable rates of polypeptide initiation and elongation on ovalbumin and globin messenger ribonucleic acid in a rabbit reticulocyte lysate. J Biol Chem. 1973 Mar 25;248(6):2095–2106. [PubMed] [Google Scholar]
- Sapareto S. A., Hopwood L. E., Dewey W. C., Raju M. R., Gray J. W. Effects of hyperthermia on survival and progression of Chinese hamster ovary cells. Cancer Res. 1978 Feb;38(2):393–400. [PubMed] [Google Scholar]
- Scott M. P., Storti R. V., Pardue M. L., Rich A. Cell-free protein synthesis in lysates of Drosophila melanogaster cells. Biochemistry. 1979 Apr 17;18(8):1588–1594. doi: 10.1021/bi00575a032. [DOI] [PubMed] [Google Scholar]
- Spradling A., Pardue M. L., Penman S. Messenger RNA in heat-shocked Drosophila cells. J Mol Biol. 1977 Feb 5;109(4):559–587. doi: 10.1016/s0022-2836(77)80091-0. [DOI] [PubMed] [Google Scholar]
- Tissières A., Mitchell H. K., Tracy U. M. Protein synthesis in salivary glands of Drosophila melanogaster: relation to chromosome puffs. J Mol Biol. 1974 Apr 15;84(3):389–398. doi: 10.1016/0022-2836(74)90447-1. [DOI] [PubMed] [Google Scholar]
- Walsh C. Appearance of heat shock proteins during the induction of multiple flagella in Naegleria gruberi. J Biol Chem. 1980 Apr 10;255(7):2629–2632. [PubMed] [Google Scholar]
- Weidner M., Combrink G. Phenotypical temperature adaptation of protein synthesis in wheat seedlings: time curves for readaptation. Plant Physiol. 1979 Jul;64(1):144–149. doi: 10.1104/pp.64.1.144. [DOI] [PMC free article] [PubMed] [Google Scholar]