Abstract
The immediate-early 1 (IE1) and IE2 proteins encoded by the major immediate-early (MIE) transcription unit of cytomegaloviruses are thought to play key roles in the switch between latent- and lytic-cycle infection. Whilst IE2 is essential for triggering the lytic cycle, the exact roles of IE1 have not been resolved. An MIE–exon 4-deleted rat cytomegalovirus (ΔIE1) failed to synthesize the IE1 protein and did not disperse promyelocytic leukaemia bodies early post-infection, but was still capable of normal replication in fibroblast cell culture. However, ΔIE1 had a diminished ability to infect salivary glands persistently in vivo and to reactivate from spleen explant cultures ex vivo. Quantification of viral genomes in spleens of infected animals revealed a reduced amount of ΔIE1 virus produced during acute infection, suggesting a role for IE1 as a regulator in establishing a chronic or persistent infection, rather than in influencing the latency or reactivation processes more directly.
The cytomegaloviruses (CMVs) are a large family of β-herpesviruses that are extremely species-specific. Whilst the initial infection is usually asymptomatic, the virus can become latent in multiple organs and reactivate during periods of immunosuppression. Like other herpesviruses, the CMVs display an ordered cascade of gene expression during productive infection (Mocarski et al., 2007). The immediate-early (IE) genes are expressed shortly after virus entry into the cell, and the major immediate-early (MIE) mRNA is expressed in the presence of cycloheximide in vitro. The IE proteins appear to regulate the expression of many downstream viral genes and are believed to be critical for reactivation processes following the transcriptional activation of the CMV MIE region (Stinski & Isomura, 2008).
The structural organization of the MIE regions of human CMV (HCMV), African green monkey CMV, murine CMV (MCMV) and rat CMV (RCMV) are very similar to each other (Meier & Stinski, 2006; Sandford et al., 1993). An enhancer region lies upstream of two spliced genes that share an untranslated leader sequence (exon 1), exon 2, which contains the first ATG, and exon 3. Two proteins are expressed from the MIE locus. In HCMV and RCMV, these are IE1 and IE2, and in MCMV they are IE1 and IE3. Thus, exon 4 is spliced to exon 2/3 to form IE1, whilst alternate splicing of exon 5 to exon 2/3 forms IE2 (IE3 in MCMV; see Fig. 1a) (Keil et al., 1987; Messerle et al., 1992; Sandford et al., 1993; Stinski et al., 1983). No viable IE2/IE3 deletion mutants of any CMV have been isolated, suggesting that IE2/IE3 is absolutely essential for virus replication even in cell culture (Angulo et al., 2000; Marchini et al., 2001; White et al., 2004), but IE1 is generally considered non-essential in cell culture, especially at high m.o.i.
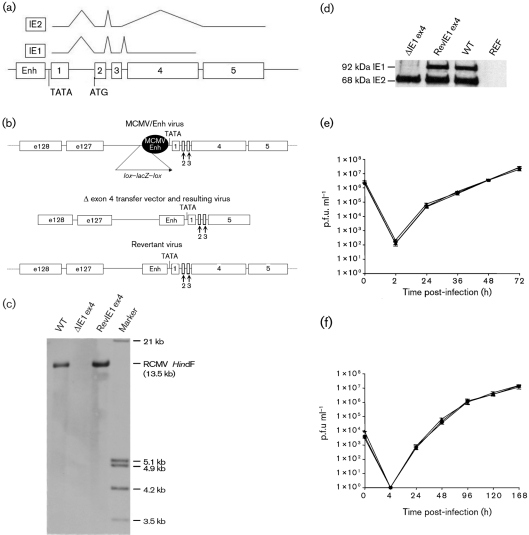
Fig. 1.
Construction and characterization of recombinant viruses. (a) Structural organization of the MIE region of RCMV. The MIE enhancer (Enh) drives the two main IE transcripts, IE1 and IE2, by alternative splicing. (b) Isolation of mutant viruses following homologous recombination in REF/IE1 cells. (c, d) Recombinant viruses were analysed by HindIII digest, Southern blotting [using a probe directed against exon 4 (c)] and sequencing. The deletion of the IE1 protein was confirmed by Western blot analysis using a polyclonal antiserum directed against a peptide within exon 3 of the RCMV MIE region, which therefore detects both the IE1 and IE2 proteins (d). (e, f) For comparison of growth capacities, one-step [m.o.i. of 0.01 (e)] and multi-step [m.o.i. of 5 (f)] growth curves were done with WT RCMV (▴), ΔIE1ex4 (▪) and RevIE1ex4 (▴).
Prevention of virus latency or reactivation could provide potential attractive targets for therapeutic intervention, but at present very little is known about these processes in CMV. Partly because of the high levels of CpG suppression of IE1 exon 4, but not of the IE2 exon 5, coding regions compared with the rest of the viral genome (Honess et al., 1989), and because of its association with metaphase chromosomes, which resembles a feature of Epstein–Barr virus EBNA1 (Lafemina et al., 1989), the HCMV IE1 protein has at various times been proposed to be involved in the establishment of or reactivation from latency. To study the influence of IE1 in this context, several deletion mutants have been constructed and analysed. Both partial and complete IE1 exon 4 deletion mutants of HCMV have been described (Ahn et al., 1998; Greaves & Mocarski, 1998; Huh et al., 2008; Lee et al., 2004; Mocarski et al., 1996). They are characterized by defective replication at a low m.o.i. in primary human fibroblasts, a slower lytic replication cycle especially in promyelocytic leukaemia body (PML)-overexpressing cells, greatly reduced ability to form or spread as plaques after DNA transfection, increased apoptosis and a greater susceptibility to inhibition by beta interferon, but none of these have or could be assessed in vivo in human hosts or animal models.
Rodent CMVs have long provided well-established animal models to study latency and reactivation in vivo (Reddehase et al., 2002, 2008). By analogy to HCMV, an MCMV IE1 deletion mutant has been constructed that replicates similarly to wild-type (WT) virus at both high and low m.o.i. in vitro, but is attenuated in vivo (Ghazal et al., 2005). Here, we investigated the influence of IE1 on RCMV (English isolate) replication in vitro and in vivo, as well as its ability to reactivate ex vivo.
Despite the similarity in overall MIE region structural organization, there is little similarity (just 15 % identity) at the predicted amino acid level between human, Old World primate and rodent CMV IE1 proteins. The major conserved feature in exon 4 is a glutamate-rich domain found toward the C terminus in all versions. Therefore, they could play very different roles or act through very different mechanisms. To examine the role of IE1 during RCMV replication, we constructed an IE1 mutant virus with a completely deleted exon 4 (ΔIE1ex4). Virion DNA of a previously described recombinant RCMV with the endogenous RCMV enhancer replaced by the MCMV enhancer preceded by a lox–lacZ–lox cassette (Sandford et al., 2001) was co-transfected with a transfer vector lacking exon 4 (Fig. 1b) into rat embryo fibroblast (REF) cells, providing IE1 complementation in trans. Homologous recombination in REF cells resulted in the loss of the lacZ expression cassette and thus in white plaques, and also in reconstitution of the endogenous RCMV enhancer (Fig. 1b). A revertant virus (RevIE1ex4) was also constructed as a control for the absence of other inadvertent genetic defects by co-transfection of virion DNA with the revertant transfer vector as above, except that screening was for β-galactosidase-positive virus. RevIE1ex4 was passaged through a REF cell line expressing the Cre protein, which resulted in the loss of the lacZ cassette, with one loxP site remaining in the virus. Both recombinant viruses were purified by limiting dilution and analysed by Southern blotting (Fig. 1c) and DNA sequencing of the MIE region. Relative total virion DNA : p.f.u. ratios were measured to show that ΔIE1ex4 does not have many more non-infectious particles than WT and that adjacent exons are unaffected (see Supplementary Figs S1 and S2, available in JGV Online).
To verify that the lack of exon 4 resulted in the lack of IE1 protein expression in infected cells at low m.o.i., we performed Western blot analysis using a polyclonal rabbit antiserum that recognizes a domain within exon 3 of both IE1 and IE2. Thus, the 68 kDa IE2 protein could be detected at normal levels but, as expected, ΔIE1ex4 failed to express any of the 92 kDa IE1 protein (Fig. 1d). Next, we compared growth characteristics of ΔIE1ex4 with those of WT RCMV and RevIE1ex4 in fibroblast tissue culture. As has been shown for MCMV (Ghazal et al., 2005), ΔIE1ex4 replicated with almost-identical efficiency to WT and RevIE1ex4 viruses at both low and high m.o.i. in this highly permissive cell type (Fig. 1e, f).
The dispersion of PML nuclear bodies is thought to increase viral transcription efficiency. Both the HCMV and MCMV IE1 proteins are responsible for the disruption of PML-associated nuclear bodies very early after infection (Ahn & Hayward, 1997, 2000; Ahn et al., 1998; Ghazal et al., 2005; Nevels et al., 2004; Tang & Maul, 2003; Tavalai et al., 2006; Wilhelmi et al., 2008; Wilkinson et al., 1998; Xu et al., 2001). To reveal a possible role for RCMV IE1 in PML disruption, REF cells were either mock-infected or infected with WT RCMV, ΔIE1ex4 or RevIE1ex4. PML bodies were detected by a polyclonal antiserum (Ahn et al., 1998) 8 h post-infection and visualized by fluorescence microscopy (Fig. 2a). Whereas both WT and RevIE1ex4 dispersed PML bodies normally, ΔIE1ex4 did not. Both immunofluorescence analysis (IFA) and Western blotting confirmed that there was no change in the pattern or levels of expression of the IE2 protein in typical punctate pre-replication compartment (pre-RC) structures from the mutant virus. Therefore, as ΔIE1ex4 replicated efficiently in fibroblast cell culture, it seems that the dispersion of PML is not a prerequisite for RCMV to replicate in vitro.
Fig. 2.
Analysis of IE1 function in fibroblast cell culture and in vivo. (a) IE1 is needed for disruption of PML bodies 8 h after infection of REF cells. REF cells were either mock-infected or infected with WT RCMV, ΔIE1ex4 or RevIE1ex4 at an m.o.i. of 3 and subjected to double-label IFA with polyclonal antisera against IE1/IE2 exon3 (fluorescein isothiocyanate, green) and PML (rhodamine, red). Nuclei were stained with DAPI (blue). IE2 was detected as punctate pre-replication compartment (pre-RC) domains in all three viruses. (b) Quantitative PCR of DNA extracted from latently infected rat spleens 120 days after infection with WT RCMV, ΔIE1ex4 and RevIE1ex4.
In the rat animal model, WT virus becomes detectable in the salivary gland (by direct virus culture) by 2 weeks post-infection, where it persists for at least 12 weeks thereafter, but is cleared from the spleen by 1 week post-infection before latency is established (not directly culturable). To investigate IE1 biological function in vivo, we compared infections of rats with WT, ΔIE1ex4 and RevIE1ex4, using 1×107 p.f.u. administered intraperitoneally. We monitored acute infection by harvesting spleens at 3 days and salivary glands at 16 days post-infection. In spleens, all viruses could be detected in the infected animals by a direct plaque assay (Table 1). However, virus could be detected in all salivary glands of animals infected with either WT or RevIE1ex4, whereas only five of eight animals infected with ΔIE1ex4 were positive, with a 10-fold reduction in the number of viral plaques. Thus, ΔIE1ex4 can replicate normally in the spleen during acute infection and can spread to the salivary gland, but appears to have reduced replication efficiency. Next, we investigated virus persistence in spleen and salivary gland explants. As expected, neither virus could be detected by direct plaque assay at 120 days post-infection in the spleen. In salivary glands, both infectious WT and RevIE1ex4 virus were detectable in all 12 animals, but directly infectious ΔIE1ex4 virus was only detected in four of 12 animals.
Table 1.
Detection of WT RCMV, ΔIE1ex4 and RevIE1ex4 in Sprague–Dawley rats at different time points after infection
Acute infection was monitored in spleens 3 days post-infection (six animals each) and in salivary glands 16 days post-infection (eight animals each). WT virus and RevIE1ex4 could be detected in all spleens and salivary glands, whereas ΔIE1ex4 was only detectable in five of eight salivary glands with an approx. 10-fold lower amount of virus present. ΔIE1ex4 was also found in only four of 12 animals in salivary glands 120 days post-infection. In contrast to WT virus and RevIE1ex4, ΔIE1ex4 could only be reactivated from one of 12 spleens ex vivo.
| Virus detected in: | WT | RevIE1ex4 | ΔIE1ex4 |
|---|---|---|---|
| Acute infection | |||
| Spleen day 3 | 6/6 | 6/6 | 6/6 |
| Salivary gland day 16 | 8/8 | 8/8 | 5/8* |
| Persistent virus 120 days p.i. | |||
| Spleen | 0/12 | 0/12 | 0/12 |
| Salivary gland | 12/12 | 12/12 | 4/12 |
| Reactivated virus 120 days p.i. | |||
| Spleen | 12/12 | 12/12 | 1/12 |
*Approx. 10-fold less virus than WT.
In our infected adult rat model, we define latency as resolution of productive infection at the organismal level, such that virus cannot be detected by direct plaque-assay culture, but can still be detected as stable low levels of DNA and by explant co-cultivation; however, latency in any organ can coexist with productive infection in the salivary glands, as shown in the mouse model (Simon et al., 2006). To examine whether IE1 plays a role in reactivation from latency in explants at 120 days post-infection, spleens were excised and small pieces were co-cultured on REF cells. Whereas spleens from all 12 WT and RevIE1ex4 virus-infected animals consistently reactivated after 2 weeks culture, only one of 12 spleens from ΔIE1ex4-infected animals reactivated 3 weeks after explant. Nevertheless, six of the six spleens tested contained mutant viral DNA, as detected by PCR (see Supplementary Fig. S3, available in JGV Online). Therefore, ΔIE1ex4 can become latent in the spleen, but demonstrates a reduced ability to reactivate. This points to a substantial, but nevertheless non-essential role for IE1 in reactivation (as virus could be recovered from one spleen) from latency ex vivo, as has recently been reported for an MCMV IE1 deletion mutant (Busche et al., 2009).
Mock-infected animals were negative for infectious virus at all time points. In addition, DNA was isolated from infected spleens harvested 120 days post-infection and measured for latent DNA levels with an RCMV IE1-specific probe by quantitative PCR methods described previously (Voigt et al., 2007). Each sample was measured in duplicate and absolute quantities were calculated by using a calibration curve of serial dilutions of subcloned plasmids containing IE1 or c-myc DNA fragments. Total viral DNA levels were normalized by calculating the IE1 : 100 copies c-myc ratio. For both the WT and RevIE1ex4 viruses, ratios ranged between 0.03 and 6.20 for WT-infected and between 0.08 and 6.70 for RevIE1ex4-infected animals. In contrast, ratios for animals infected with ΔIE1ex4 ranged from 0.005 to 0.040 (Fig. 2b). Therefore, ΔIE1ex4 can evidently become latent in the spleen, but its ability to reactivate is severely compromised. Whereas the overall effect of restricted reactivation must be caused by the deletion in IE1, it is not necessarily related to any direct qualitative deficiency of the mutant viral genomes present to become latent or to reactivate, but may rather simply reflect the lower quantity of mutant virus present. Less mutant virus was detectable in the spleen, and therefore the chance of it being reactivated is likely to be small (Reddehase et al., 1994). The IE1 deletion also results in an acute replication deficit, which is in accordance with a lower titre in the salivary gland at day 16 post-infection. However, because ΔIE1ex4 is attenuated in vivo, the infection conditions probably did not provide equal levels of multiplication of the viruses, and thus did not produce an equivalent viral genome load during latency.
To exclude contamination with WT or RevIE1ex4 virus in this one reactivated animal and to see whether the reduced acute replication in vivo plus reduced reactivation of ex vivo ΔIE1ex4 was related to the lack of IE1 protein expression, viruses were isolated from salivary glands that had been persistently infected, as well as from spleens from which virus had been reactivated ex vivo. Recovered viruses were grown on REF cells and lysates were analysed by Western blot assays. Again, only 68 kDa IE2 but not 92 kDa IE1 protein was detected (see Supplementary Fig. S4, available in JGV Online).
Overall, our results indicate greatly reduced levels of acute replication by RCMV lacking the IE1 gene in the in vivo model in the salivary gland, which evidently lead to much lower levels of stably maintained latency and of reactivation in the spleen. The results are consistent with the findings of reduced virulence and lower virus titres for MCMV lacking IE1 in adult BALB/c and SCID mice (Ghazal et al., 2005), although these authors did not address latency or reactivation. Similar findings were also recently described in a newborn mouse model (Busche et al., 2009). However, by using 10- to 100-fold higher compensatory levels of MCMV lacking IE1 than of WT controls to generate equal levels of established latency, the latter authors measured stable latency and used three distinct criteria for reactivation (including lung explants), but found no evidence for a role of IE1 in these two latter aspects of MCMV biology. Therefore, we can only conclude that there is a strong positive effect of IE1 on acute infection levels in vivo, whereas it is quite plausible that RCMV ΔIE1ex4 (like MCMV) has no defect in the ability to maintain or reactivate from latency.
Supplementary Material
Acknowledgments
We thank Franziska Bührdel for technical assistance.
Footnotes
Four supplementary figures are available with the online version of this paper.
References
- Ahn, J. H. & Hayward, G. S. (1997). The major immediate-early proteins IE1 and IE2 of human cytomegalovirus colocalize with and disrupt PML-associated nuclear bodies at very early times in infected permissive cells. J Virol 71, 4599–4613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahn, J. H. & Hayward, G. S. (2000). Disruption of PML-associated nuclear bodies by IE1 correlates with efficient early stages of viral gene expression and DNA replication in human cytomegalovirus infection. Virology 274, 39–55. [DOI] [PubMed] [Google Scholar]
- Ahn, J. H., Brignole, E. J., III & Hayward, G. S. (1998). Disruption of PML subnuclear domains by the acidic IE1 protein of human cytomegalovirus is mediated through interaction with PML and may modulate a RING finger-dependent cryptic transactivator function of PML. Mol Cell Biol 18, 4899–4913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angulo, A., Ghazal, P. & Messerle, M. (2000). The major immediate-early gene IE3 of mouse cytomegalovirus is essential for viral growth. J Virol 74, 11129–11136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busche, A., Marquardt, A., Bleich, A., Ghazal, P., Angulo, A. & Messerle, M. (2009). The mouse cytomegalovirus immediate-early 1 gene is not required for establishment of latency or for reactivation in the lungs. J Virol 83, 4030–4038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghazal, P., Visser, A. E., Gustems, M., Garcia, R., Borst, E. M., Sullivan, K., Messerle, M. & Angulo, A. (2005). Elimination of IE1 significantly attenuates murine cytomegalovirus virulence but does not alter replicative capacity in cell culture. J Virol 79, 7182–7194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greaves, R. F. & Mocarski, E. S. (1998). Defective growth correlates with reduced accumulation of a viral DNA replication protein after low-multiplicity infection by a human cytomegalovirus IE1 mutant. J Virol 72, 366–379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honess, R. W., Gompels, U. A., Barrell, B. G., Craxton, M., Cameron, K. R., Staden, R., Chang, Y. N. & Hayward, G. S. (1989). Deviations from expected frequencies of CpG dinucleotides in herpesvirus DNAs may be diagnostic of differences in the states of their latent genomes. J Gen Virol 70, 837–855. [DOI] [PubMed] [Google Scholar]
- Huh, Y. H., Kim, Y. E., Kim, E. T., Park, J. J., Song, M. J., Zhu, H., Hayward, G. S. & Ahn, J. H. (2008). Binding STAT2 by the acidic domain of human cytomegalovirus IE1 promotes viral growth and is negatively regulated by SUMO. J Virol 82, 10444–10454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keil, G. M., Ebeling-Keil, A. & Koszinowski, U. H. (1987). Sequence and structural organization of murine cytomegalovirus immediate-early gene 1. J Virol 61, 1901–1908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lafemina, R. L., Pizzorno, M. C., Mosca, J. D. & Hayward, G. S. (1989). Expression of the acidic nuclear immediate-early protein (IE1) of human cytomegalovirus in stable cell lines and its preferential association with metaphase chromosomes. Virology 172, 584–600. [DOI] [PubMed] [Google Scholar]
- Lee, H. R., Kim, D. J., Lee, J. M., Choi, C. Y., Ahn, B. Y., Hayward, G. S. & Ahn, J. H. (2004). Ability of the human cytomegalovirus IE1 protein to modulate sumoylation of PML correlates with its functional activities in transcriptional regulation and infectivity in cultured fibroblast cells. J Virol 78, 6527–6542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchini, A., Liu, H. & Zhu, H. (2001). Human cytomegalovirus with IE-2 (UL122) deleted fails to express early lytic genes. J Virol 75, 1870–1878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meier, J. L. & Stinski, M. F. (2006). Major immediate-early enhancer and its gene products. In Cytomegaloviruses: Molecular Biology and Immunology, pp. 151–166. Edited by M. J. Reddehase. Wymondham, UK: Caister Academic Press.
- Messerle, M., Buhler, B., Keil, G. M. & Koszinowski, U. H. (1992). Structural organization, expression, and functional characterization of the murine cytomegalovirus immediate-early gene 3. J Virol 66, 27–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mocarski, E. S., Kemble, G. W., Lyle, J. M. & Greaves, R. F. (1996). A deletion mutant in the human cytomegalovirus gene encoding IE1(491aa) is replication defective due to a failure in autoregulation. Proc Natl Acad Sci U S A 93, 11321–11326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mocarski, E. S., Shenk, T. & Pass, R. F. (2007). Cytomegaloviruses. In Fields Virology, 5th edn, pp. 2701–2772. Edited by D. M. Knipe & P. M. Howley. Philadelphia, PA: Lippincott Williams & Wilkins.
- Nevels, M., Brune, W. & Shenk, T. (2004). SUMOylation of the human cytomegalovirus 72-kilodalton IE1 protein facilitates expression of the 86-kilodalton IE2 protein and promotes viral replication. J Virol 78, 7803–7812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddehase, M. J., Balthesen, M., Rapp, M., Jonjic, S., Pavic, I. & Koszinowski, U. H. (1994). The conditions of primary infection define the load of latent viral genome in organs and the risk of recurrent cytomegalovirus disease. J Exp Med 179, 185–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddehase, M. J., Podlech, J. & Grzimek, N. K. (2002). Mouse models of cytomegalovirus latency: overview. J Clin Virol 25, (Suppl. 2), S23–S36. [DOI] [PubMed] [Google Scholar]
- Reddehase, M. J., Simon, C. O., Seckert, C. K., Lemmermann, N. & Grzimek, N. K. (2008). Murine model of cytomegalovirus latency and reactivation. Curr Top Microbiol Immunol 325, 315–331. [DOI] [PubMed] [Google Scholar]
- Sandford, G. R., Ho, K. & Burns, W. H. (1993). Characterization of the major locus of immediate-early genes of rat cytomegalovirus. J Virol 67, 4093–4103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandford, G. R., Brock, L. E., Voigt, S., Forester, C. M. & Burns, W. H. (2001). Rat cytomegalovirus major immediate-early enhancer switching results in altered growth characteristics. J Virol 75, 5076–5083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon, C. O., Seckert, C., Grzimek, N. K. & Reddehase, M. J. (2006). Murine model of cytomegalovirus latency and reactivation: the silencing/desilencing and immune sensing hypothesis. In Cytomegaloviruses: Molecular Biology and Immunology, pp. 483–500. Edited by M. J. Reddehase. Wymondham, UK: Caister Academic Press.
- Stinski, M. F. & Isomura, H. (2008). Role of the cytomegalovirus major immediate early enhancer in acute infection and reactivation from latency. Med Microbiol Immunol 197, 223–231. [DOI] [PubMed] [Google Scholar]
- Stinski, M. F., Thomsen, D. R., Stenberg, R. M. & Goldstein, L. C. (1983). Organization and expression of the immediate early genes of human cytomegalovirus. J Virol 46, 1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang, Q. & Maul, G. G. (2003). Mouse cytomegalovirus immediate-early protein 1 binds with host cell repressors to relieve suppressive effects on viral transcription and replication during lytic infection. J Virol 77, 1357–1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tavalai, N., Papior, P., Rechter, S., Leis, M. & Stamminger, T. (2006). Evidence for a role of the cellular ND10 protein PML in mediating intrinsic immunity against human cytomegalovirus infections. J Virol 80, 8006–8018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voigt, S., Mesci, A., Ettinger, J., Fine, J. H., Chen, P., Chou, W. & Carlyle, J. R. (2007). Cytomegalovirus evasion of innate immunity by subversion of the NKR-P1B : Clr-b missing-self axis. Immunity 26, 617–627. [DOI] [PubMed] [Google Scholar]
- White, E. A., Clark, C. L., Sanchez, V. & Spector, D. H. (2004). Small internal deletions in the human cytomegalovirus IE2 gene result in nonviable recombinant viruses with differential defects in viral gene expression. J Virol 78, 1817–1830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilhelmi, V., Simon, C. O., Podlech, J., Bohm, V., Daubner, T., Emde, S., Strand, D., Renzaho, A., Lemmermann, N. A. & other authors (2008). Transactivation of cellular genes involved in nucleotide metabolism by the regulatory IE1 protein of murine cytomegalovirus is not critical for viral replicative fitness in quiescent cells and host tissues. J Virol 82, 9900–9916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkinson, G. W., Kelly, C., Sinclair, J. H. & Rickards, C. (1998). Disruption of PML-associated nuclear bodies mediated by the human cytomegalovirus major immediate early gene product. J Gen Virol 79, 1233–1245. [DOI] [PubMed] [Google Scholar]
- Xu, Y., Ahn, J. H., Cheng, M., apRhys, C. M., Chiou, C. J., Zong, J., Matunis, M. J. & Hayward, G. S. (2001). Proteasome-independent disruption of PML oncogenic domains (PODs), but not covalent modification by SUMO-1, is required for human cytomegalovirus immediate-early protein IE1 to inhibit PML-mediated transcriptional repression. J Virol 75, 10683–10695. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.