Abstract
We compared 68Ga-DOTA-F(ab′)2-herceptin (DOTA is 1,4,7,10-tetraazacyclododecane-N,N′,N″,N‴-tetraacetic acid [HER2 PET]) and 18F-FDG PET for imaging of tumor response to the heat shock protein 90 (Hsp90) inhibitor 17-allylamino-17-demethoxygeldanamycin (17AAG).
Methods
Mice bearing BT474 breast tumor xenografts were scanned with 18F-FDG PET and HER2 PET before and after 17AAG treatment and then biweekly for up to 3 wk.
Results
Within 24 h after treatment, a significant decrease in HER2 was measured by HER2 PET, whereas 18F-FDG PET uptake, a measure of glycolysis, was unchanged. Marked growth inhibition occurred in treated tumors but became evident only by 11 d after treatment. Thus, Her2 downregulation occurs independently of changes in glycolysis after 17AAG therapy, and Her2 reduction more accurately predicts subsequent tumor growth inhibition.
Conclusion
HER2 PET is an earlier predictor of tumor response to 17AAG therapy than 18F-FDG PET.
We recently developed a molecular imaging method for visualizing the pharmacodynamics of HER2 degradation due to heat shock protein 90 (Hsp90) inhibition. The molecular imaging technique is based on PET with 68Ga-DOTA-F(ab′)2-herceptin (DOTA is 1,4,7,10-tetraazacyclododecane-N,N′,N″,N‴-tetraacetic acid [HER2 PET]) (1).
PET is an emerging functional imaging technology, which is now broadly applied for imaging human tumors (2). 18F-FDG PET has become an important clinical tool in detecting primary and metastatic breast cancer as well as monitoring treatment response, recurrence, and general prognosis (3). The molecular basis for the success of 18F-FDG PET is the rapid reduction of tumor glucose metabolism in effective therapies (4), and this has been shown to be a very reliable early indicator of tumor response. However, 18F-FDG PET may be abnormal in benign disease as well as active inflammation. For example, response assessment can be confounded by posttreatment inflammatory change in the weeks after radiation therapy of head and neck cancers and optimal times for 18F-FDG PET to determine response can be 3–5 mo after treatment (5).
17-Allylamino-17-demethoxygeldanamycin (17AAG) is the first of a new class of drugs that downregulates several oncoproteins overexpressed by breast cancer and is currently in early clinical trials. One such target is HER2 (c-erbB2 or Her-2/neu), a protooncogene, which encodes the 185-kDa transmembrane protein human epidermal growth factor receptor 2 (6). HER2 protein overexpression is observed in 25%–30% of primary breast cancers (7). Recently, other malignancies—such as non–small cell lung cancer (8,9), ovarian cancer (9), gastric carcinomas (10), osteosarcoma (11), prostate cancer (12), renal cell carcinoma (13), pancreatic adenocarcinoma (14), salivary duct carcinoma (15), rectal cancer (16), bladder cancer (17), and biliary cancer (18)—have all been shown to have overexpression of HER2. In humans, HER2 is expressed at low levels in some normal tissues such as heart (19).
17AAG is the first of a new class of drugs that have a novel mechanism of action and targets Hsp90, a molecular chaper-one, which is responsible for the correct folding, stability, and function of a range of oncoproteins that are mutated or overexpressed in cancer (20). These include the androgen receptor and the HER family of receptor tyrosine kinases (21). HER2 is considered a clinically important molecule and routine testing is performed by many centers, especially for breast cancers. However, testing relies on invasive sampling (22). In current clinical trials with 17AAG, biopsies have been obtained in only a fraction of patients and only during the first cycle of therapy, because biopsies are invasive and a majority of patients have tumors that cannot be sampled without significant risk to the patient. The most common methods used to measure HER2 expression at the DNA and protein levels are fluorescence in situ hybridization and immunohistochemical staining (IHC). IHC is the more common method, but it is a very subjective method, which can result in low levels of agreement with regard to intensity and extent of tumor membrane staining.
MATERIALS AND METHODS
General
All reagents were obtained from commercial sources. 18F-FDG was purchased from Eastern Isotopes. 68Ga was obtained from a 68Ge/68Ga generator and used to label DOTA-F(ab′)2-herceptin as previously described (1). 17AAG (NSC 330507) and the egg phospholipid diluent were obtained from the Drug Synthesis and Chemistry Branch, Developmental Therapeutics Program, National Cancer Institute.
Tumor Model
Breast cancer cell line BT474 (American Type Culture Collection) was grown in a 1:1 mixture of Dulbecco’s modified Eagle medium/F-12 medium supplemented with 2 mmol/L glutamine, 50 units/mL penicillin, 50 units/mL streptomycin, and 10% heat-inactivated fetal bovine serum (Gemini Bioproducts) at a temperature of 37°C in an environment containing 5% CO2. Before use, the cells were trypsinized, counted, and suspended in Matrigel (Collaborative Biomedical Products).
Eight mice were selected for tumor size (50–200 mg) from a larger pool of mice. Tumors were allowed to develop over a period of 14 d after the subcutaneous placement (on day 20–14) of 0.72 mg/d 17β-estradiol pellets just below the left shoulder, followed by injection of 1 × 107 BT474 tumor cells combined with Matrigel on the right hip. Four- to 6-wk-old nu/nu athymic female mice were obtained from the National Cancer Institute–Frederick Cancer Center and maintained in ventilated cages. The animals were fed and given water ad lib. Experiments were performed under an Institutional Animal Care and Use Committee–approved protocol and the experiments followed institutional guidelines for the proper and humane use of animals in research. BT474 tumor cells (1 × 107) were injected together with Matrigel on their right hip. Before tumor cell inoculation, 0.72 mg/d 17β-estradiol pellets (Innovative Research of America) were placed subcutaneously just below their left shoulder. Tumors (50–200 mg) developed after a period of 14–18 d.
PET Studies
These animals were divided into 2 groups of 5 mice each. Both groups were injected with either 15 MBq 18F-FDG or 3–8 MBq 68Ga-F(ab′)2-herceptin in the tail vein. At 1 and 3 h after injection, respectively, the animals were sedated using isoflurane and imaged with a microPET camera (Concorde Microsystems Inc.). Coincident data were collected for the 511-keV γ-rays with a 250- to 750-keV window for 10 min. The images were reconstructed and regions of interest (ROIs) were drawn around the tumors and major organs. One group of animals then received a total of 150 mg/kg of 17AAG dissolved in an egg phospholipid, over 24 h in 3 doses of 50 mg/kg each (day 0). The other group of animals was not treated. The animals were then imaged with a regimen of weekly 18F-FDG or biweekly 68Ga-F(ab′)2-herceptin on alternating days for the next 3 wk, and the tumor size and body mass were monitored.
Tumor volumes were determined by external vernier caliper measurements along the longest axis (x-axis) and the axis perpendicular to the longest axis (y-axis). The tumor volume was then estimated by using the following formula:
RESULTS
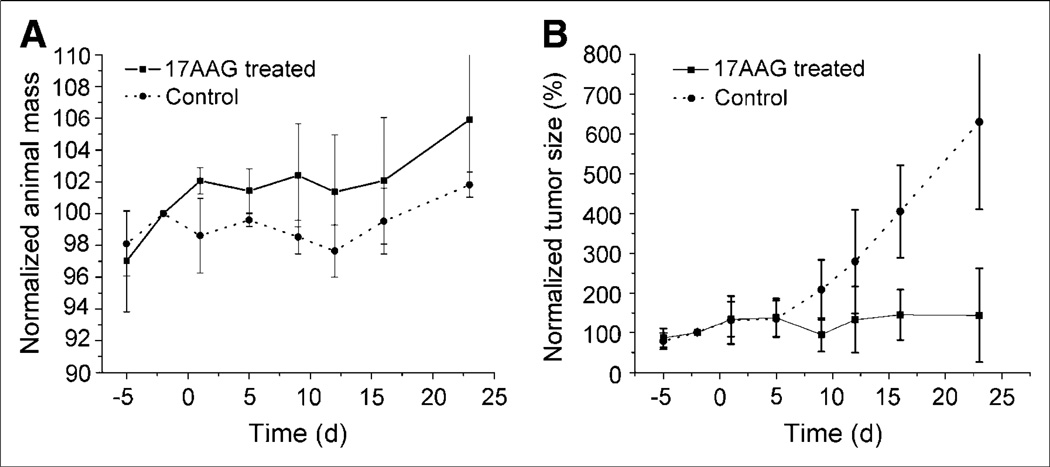
The effect of 17AAG on both the animal mass and the tumor volume is seen in Figures 1A and 1B. Throughout the study period, there were no significant differences seen in the animal mass of the control and treated animals but, overall, the 17AAG-treated animals had higher body masses (Fig. 1A). After treatment with 17AAG, there was no significant difference in the tumor volumes of the 2 groups of animals up to 5 d. However, by day 9, the tumors of the control animals had doubled in size and the 17AAG-treated animals had not significantly increased in size. The tumors in the control animals continued to grow exponentially with an apparent doubling time of 9.1 d and increased in size by a factor of 6.3 by 23 d. In comparison, the 17AAG-treated animals were unchanged in size at 9 d and were only 50% larger after 23 d.
Figure 1.
(A) Comparison of body mass of nude mice treated with 17AAG vs. control animals. Animals were treated with 3 × 50 mg/kg 17AAG over 24 h on day 0. (B) Comparison of BT474 tumor growth in nude mice treated with 17AAG vs. control animals.
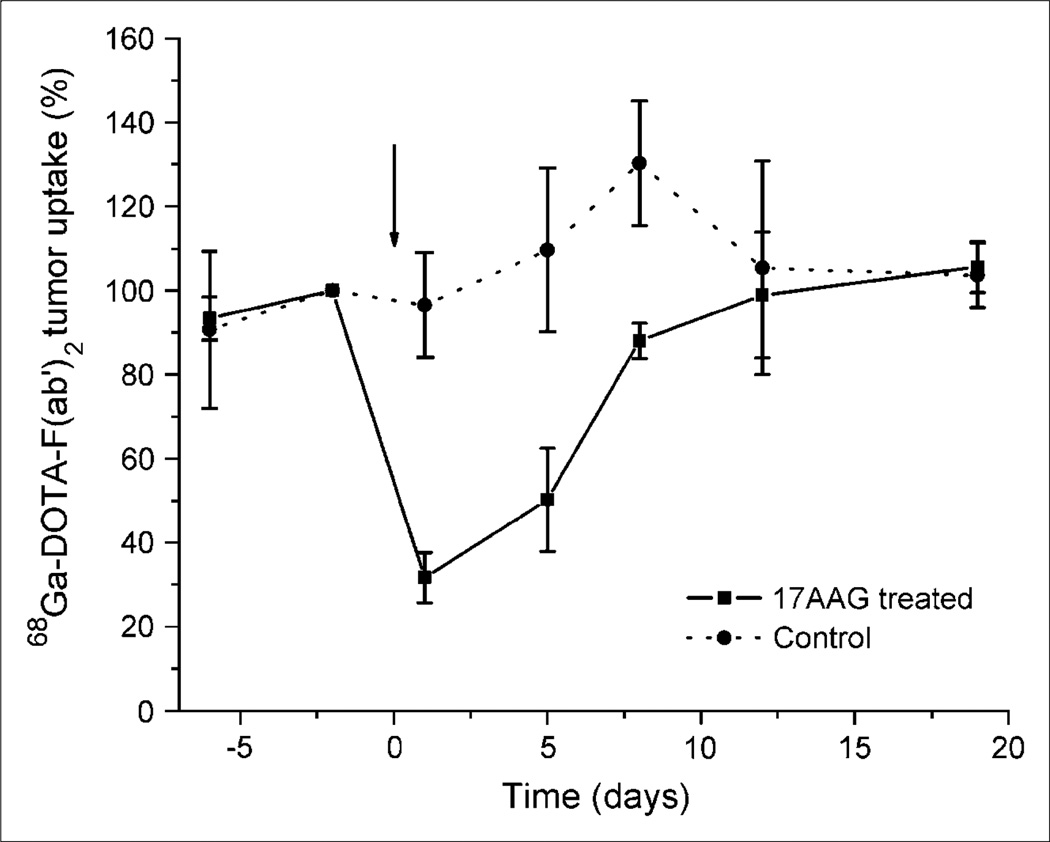
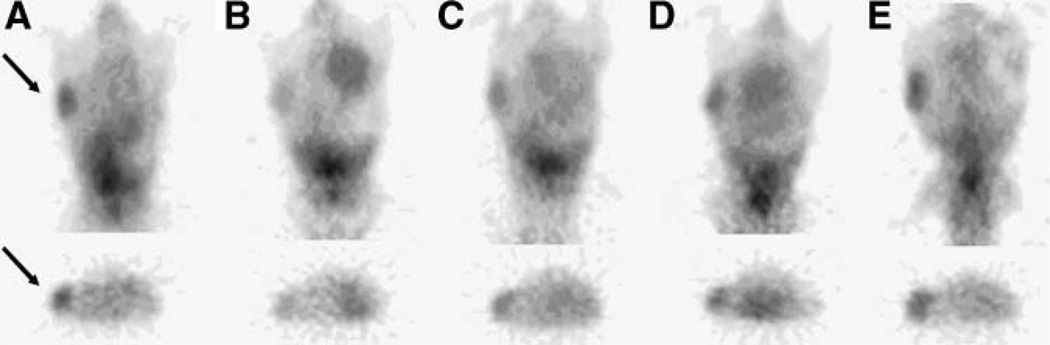
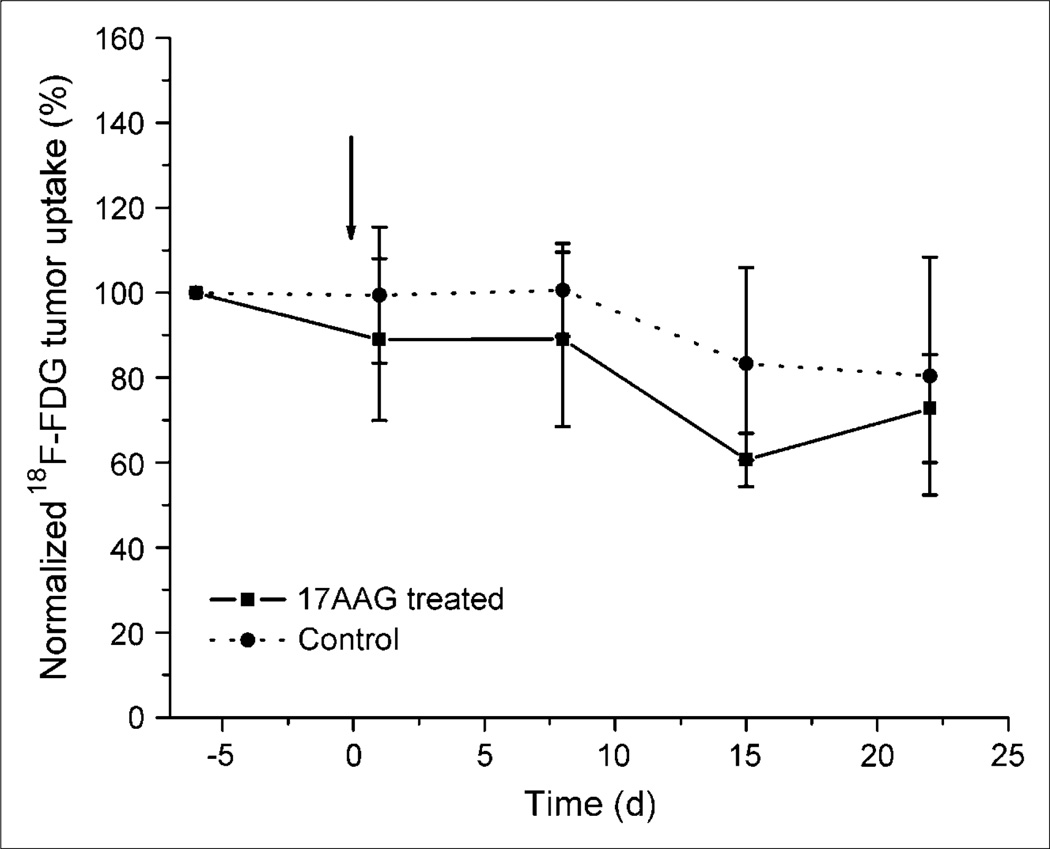
The biodistribution of radioactivity in all animals clearly show a similar uptake in all organs except the tumors. The combined microPET ROI paired analysis data of 68Ga-F(ab′)2-herceptin (Fig. 2), show that the 17AAG-treated animals had a significant (P < 0.01) 70% reduction in uptake, whereas the control animals showed a slight increase over the baseline study performed 3 d earlier. A series of sequential microPET images of the same mouse after injection with 68Ga-F(ab′)2-herceptin are shown in Figure 3. The 17AAG-treated animals had a reduced uptake of 68Ga-F(ab′)2-herceprin lasting until 5 d after treatment. HER2 levels had returned to pretreatment levels at 12 d after treatment and correlated with the regrowth of the tumors. The control animals showed no significant changes in HER2 expression. The 18F-FDG ROI data analysis of the 2 groups of animals revealed no significant differences in 18F-FDG metabolism (Fig. 4).
Figure 2.
Comparison of 68Ga-DOTA-F(ab′)2-herceptin uptake in nude mice with BT474 tumors before and after 17AAG treatment. Animals were treated with 3 × 50 mg/kg of 17AAG over 24 h on day 0 and data are normalized to day –2 uptake.
Figure 3.
microPET images (coronal slice and transverse slice through tumor) of 68Ga-DOTA-F(ab′)2-herceptin uptake in the same mouse with a BT474 tumor at –2 d (A), 1 d (B), 5 d (C), 8 d (D), and 12 d (E) after treatment with 3 × 50 mg/kg of 17AAG over 24 h on day 0. Arrows indicate tumor.
Figure 4.
Comparison of 18F-FDG uptake in nude mice with BT474 tumors before and after 17AAG treatment. Animals were treated with 3 × 50 mg/kg of 17AAG over 24 h on day 0 and data are normalized to day –6 uptake.
Thus, Her2 downregulation occurs independently of changes in tumor glycolysis and is an early predictor of subsequent growth inhibition in 17AAG-treated breast xenografts.
DISCUSSION
18F-FDG is a Food and Drug Administration–approved radiopharmaceutical used clinically to stage and monitor tumor response. 18F-FDG is taken up by cells in competition with other sugars and, once inside the cells, it is phosphorylated by hexokinase. The phosphorylation product is polar and cannot diffuse out of the cell. It can be dephosphorylated back to 18F-FDG by glucose-6-phosphatase, but this dephosphorylation occurs slowly. The net result is that malignant cells, which generally have an accelerated glycolysis rate and decreased ability to make energy aerobically, generally have elevated uptakes and retention of 18F-FDG. We used 68Ga in this study primarily because it is a PET isotope that allows quantification of tumor uptake, but also because it has a short half-life that allows repeated imaging with no background activity from earlier studies. In this study, the 17AAG treatment had no immediate effect on 18F-FDG uptake, whereas 68Ga-F(ab′)2-herceptin uptake was markedly reduced after treatment.
The effect of 17AAG on HER2-positive tumors has been published (23). In athymic mice, with CWRSA6 prostate cancer xenografts, a single dose of 50 mg/kg 17AAG suppressed HER2 expression at 4 h after injection, but HER2 levels had returned to normal by 48 h after injection. In the current experimental setting, a higher dose of 17AAG lowered expression of HER2 until 5 d after treatment and had a stronger long-term effect on the overall growth of the tumors, which was apparent at 9 d after treatment. However, the 68Ga-F(ab′)2-herceptin imaging does rapidly show the desired effect of 17AAG on HER2-positive tumors.
Over the past year, several groups have published phase I clinical trials on 17AAG therapy (24–27). 17AAG binds to and inactivates Hsp90, but direct measurement of the biomarker in the target tissue is problematic and investigators have used indirect measurements, such as the upregulation of Hsp70 or downregulation of such client proteins as LCK, Raf-1, CDK4, and so forth. Goetz et al. (24) used peripheral blood mononuclear cells (PBMCs) to study the effect of 17AAG on biomarkers and saw no significant change in Hsp90 levels, but they reported an induction of Hsp70 levels at all doses studied (15–431 mg/m2/wk, up to 4 wk). Grem et al. (25) used a more frequent dosing scheme (10–56 mg/m2/d, 5 d/wk every 3 wk) and monitored protein levels in PBMCs. They found an increase in Hsp70 at doses of >14 mg/m2 (15/16 patients) and decreases in Raf-1 at doses of >28 mg/m2 (5/8 patients). Ramanathan et al. (26) also used PBMCs and Hsp90/Hsp70 levels to monitor 17AAG effects (10–395 mg/m2/wk) but concluded that they were uninformative pharmacodynamic markers. Banerji et al. (27) looked at the effects of 17AAG (10–450 mg/m2/wk) on peripheral blood leukocytes (PBLs) as well as tumor biopsies. Induction of Hsp70 levels in PBLs was seen at low doses of 17AAG (80 mg/m2/wk). At the highest dose level (450 mg/m2/wk), PBLs showed cRaf-1, LCK, and CDK4 depletions and Hsp70 inductions in 3 of 3, 0 of 4, 0 of 4, and 3 of 4 patients, respectively. At the higher dose level (320–450 mg/m2/wk), tumor biopsies revealed LCK and CDK4 depletions and Hsp70 inductions in 4 of 6, 8 of 9, and 8 of 9 patients. However, these authors noted a lack of correlation between biomarkers found in PBLs and tumor biopsies. These reports show the need for a noninvasive method for determining the effects of 17AAG on Hsp90 levels, and clinical trials with 68Ga-F(ab′)2-herceptin are planned.
CONCLUSION
68Ga-F(ab′)2-herceptin PET is an earlier predictor of tumor response to 17AAG therapy than 18F-FDG PET.
ACKNOWLEDGMENT
Financial support was provided by NIH grants 5-P50-CA86438-04, 1-P01CA94060-02, and 5-R24CA83084-05.
REFERENCES
- 1.Smith-Jones PM, Solit DB, Akhurst T, Afroze F, Rosen N, Larson SM. Imaging the pharmacodynamics of HER2 degradation in response to Hsp90 inhibitors. Nat Biotechnol. 2004;22:701–706. doi: 10.1038/nbt968. [DOI] [PubMed] [Google Scholar]
- 2.Czernin J. Clinical applications of FDG-PET in oncology. Acta Med Austriaca. 2002;29:162–170. doi: 10.1046/j.1563-2571.2002.02042.x. [DOI] [PubMed] [Google Scholar]
- 3.Kumar R, Alavi A. Fluorodeoxyglucose-PET in the management of breast cancer. Radiol Clin North Am. 2004;42:1113–1122. doi: 10.1016/j.rcl.2004.08.005. [DOI] [PubMed] [Google Scholar]
- 4.Avril NE, Weber WA. Monitoring response to treatment in patients utilizing PET. Radiol Clin North Am. 2005;43:189–204. doi: 10.1016/j.rcl.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 5.Yao M, Graham MM, Smith RB, et al. Value of FDG PET in assessment of treatment response and surveillance in head-and-neck cancer patients after intensity modulated radiation treatment: a preliminary report. Int J Radiat Oncol Biol Phys. 2004;60:1410–1418. doi: 10.1016/j.ijrobp.2004.05.058. [DOI] [PubMed] [Google Scholar]
- 6.Coussens L, Yang-Feng TL, Liao YC, et al. Tyrosine kinase receptor with extensive homology to EGF receptor shares chromosomal location with neu oncogene. Science. 1985;230:1132–1139. doi: 10.1126/science.2999974. [DOI] [PubMed] [Google Scholar]
- 7.Press MF, Pike MC, Chazin VR, et al. Her-2/neu expression in node-negative breast cancer: direct tissue quantitation by computerized image analysis and association of overexpression with increased risk of recurrent disease. Cancer Res. 1993;53:4960–4970. [PubMed] [Google Scholar]
- 8.Zinner RG, Kim J, Herbst RS. Non-small cell lung cancer clinical trials with trastuzumab: their foundation and preliminary results. Lung Cancer. 2002;37:17–27. doi: 10.1016/s0169-5002(02)00035-1. [DOI] [PubMed] [Google Scholar]
- 9.Li D, Williams JI, Pietras RJ. Squalamine and cisplatin block angiogenesis and growth of human ovarian cancer cells with or without HER-2 gene over-expression. Oncogene. 2002;21:2805–2814. doi: 10.1038/sj.onc.1205410. [DOI] [PubMed] [Google Scholar]
- 10.Takehana T, Kunitomo K, Kono K, et al. Status of c-erbB-2 in gastric adenocarcinoma: a comparative study of immunohistochemistry, fluorescence in situ hybridization and enzyme-linked immuno-sorbent assay. Int J Cancer. 2002;98:833–837. doi: 10.1002/ijc.10257. [DOI] [PubMed] [Google Scholar]
- 11.Akatsuka T, Wada T, Kokai Y, et al. ErbB2 expression is correlated with increased survival of patients with osteosarcoma. Cancer. 2002;94:1397–1404. doi: 10.1002/cncr.10360. [DOI] [PubMed] [Google Scholar]
- 12.Morris MJ, Reuter VE, Kelly WK, et al. HER-2 profiling and targeting in prostate carcinoma. Cancer. 2002;94:980–986. [PubMed] [Google Scholar]
- 13.Latif Z, Watters AD, Bartlett JM, Underwood MA, Aitchison M. Gene amplification and overexpression of HER2 in renal cell carcinoma. BJU Int. 2002;89:5–9. [PubMed] [Google Scholar]
- 14.Safran H, Steinhoff M, Mangray S, et al. Overexpression of the HER-2/neu oncogene in pancreatic adenocarcinoma. Am J Clin Oncol. 2001;24:496–499. doi: 10.1097/00000421-200110000-00016. [DOI] [PubMed] [Google Scholar]
- 15.Skalova A, Starek I, Kucerova V, Szepe P, Plank L. Salivary duct carcinoma: a highly aggressive salivary gland tumor with HER-2/neu oncoprotein overexpression. Pathol Res Pract. 2001;197:621–626. doi: 10.1078/0344-0338-00136. [DOI] [PubMed] [Google Scholar]
- 16.Speer G, Cseh K, Winkler G, et al. Oestrogen and vitamin D receptor (VDR) genotypes and the expression of ErbB-2 and EGF receptor in human rectal cancers. Eur J Cancer. 2001;37:1463–1468. doi: 10.1016/s0959-8049(01)00139-3. [DOI] [PubMed] [Google Scholar]
- 17.Jimenez RE, Hussain M, Bianco FJ, Jr, et al. Her-2/neu overexpression in muscleinvasive urothelial carcinoma of the bladder: prognostic significance and comparative analysis in primary and metastatic tumors. Clin Cancer Res. 2001;7:2440–2447. [PubMed] [Google Scholar]
- 18.Su WC, Shiesh SC, Liu HS, Chen CY, Chow NH, Lin XZ. Expression of oncogene products HER2/Neu and Ras and fibrosis-related growth factors bFGF, TGF-beta, and PDGF in bile from biliary malignancies and inflammatory disorders. Dig Dis Sci. 2001;46:1387–1392. doi: 10.1023/a:1010619316436. [DOI] [PubMed] [Google Scholar]
- 19.Garratt AN, Ozcelik C, Birchmeier C. ErbB2 pathways in heart and neural diseases. Trends Cardiovasc Med. 2003;13:80–86. doi: 10.1016/s1050-1738(02)00231-1. [DOI] [PubMed] [Google Scholar]
- 20.Neckers L. Heat shock protein 90 inhibition by 17-allylamino-17- demethoxygeldanamycin: a novel therapeutic approach for treating hormone-refractory prostate cancer. Clin Cancer Res. 2002;8:962–966. [PubMed] [Google Scholar]
- 21.Neckers L. Hsp90 inhibitors as novel cancer chemotherapeutic agents. Trends Mol Med. 2002;8(4 suppl):S55–S61. doi: 10.1016/s1471-4914(02)02316-x. [DOI] [PubMed] [Google Scholar]
- 22.Hanna W. Testing for HER2 status. Oncology. 2001;61 suppl 2:22–30. doi: 10.1159/000055398. [DOI] [PubMed] [Google Scholar]
- 23.Solit DB, Zheng FF, Drobnjak M, et al. 17-Allylamino-17-demethoxygeldana-mycin induces the degradation of androgen receptor and HER-2/neu and inhibits the growth of prostate cancer xenografts. Clin Cancer Res. 2002;8:986–993. [PubMed] [Google Scholar]
- 24.Goetz MP, Toft D, Reid J, et al. Phase I trial of 17-allylamino-17-demethoxygeldanamycin in patients with advanced cancer. J Clin Oncol. 2005;23:1078–1087. doi: 10.1200/JCO.2005.09.119. [DOI] [PubMed] [Google Scholar]
- 25.Grem JL, Morrison G, Guo XD, et al. Phase I and pharmacologic study of 17-(allylamino)-17-demethoxygeldanamycin in adult patients with solid tumors. J Clin Oncol. 2005;23:1885–1893. doi: 10.1200/JCO.2005.12.085. [DOI] [PubMed] [Google Scholar]
- 26.Ramanathan RK, Trump DL, Eiseman JL, et al. Phase I pharmacokinetic-pharmacodynamic study of 17-(allylamino)-17-demethoxygeldanamycin (17AAG, NSC 330507), a novel inhibitor of heat shock protein 90, in patients with refractory advanced cancers. Clin Cancer Res. 2005;11:3385–3391. doi: 10.1158/1078-0432.CCR-04-2322. [DOI] [PubMed] [Google Scholar]
- 27.Banerji U, Walton M, Raynaud F, et al. Pharmacokinetic-pharmacodynamic relationships for the heat shock protein 90 molecular chaperone inhibitor 17-allylamino, 17-demethoxygeldanamycin in human ovarian cancer xenograft models. Clin Cancer Res. 2005;11:7023–7032. doi: 10.1158/1078-0432.CCR-05-0518. [DOI] [PubMed] [Google Scholar]