Preface
Microtubules are dynamic filamentous cytoskeletal proteins that are an important therapeutic target in tumor cells. Microtubule binding agents have been part of the pharmacopoeia of cancer for decades, and until the advent of targeted therapy microtubules were the only alternative to DNA as a therapeutic target in cancer. The screening of a variety of botanical species and marine organisms has yielded promising new antitubulin agents with novel properties. Enhanced tumor specificity, reduced neurotoxicity, and insensitivity to chemoresistance mechanisms are the three main objectives in the current search for novel microtubule binding agents.
Keywords: Animals, Antineoplastic Agents, chemistry, metabolism, therapeutic use, Humans, Microtubules, metabolism, Neoplasm Proteins, metabolism, Neoplasms, drug therapy, metabolism, Protein Binding, physiology
Introduction
Microtubules play several key roles that are important in cell proliferation, trafficking, signalling, and migration in eukaryotic cells. For this reason several microtubule binding agents have been developed with different aims, including as pesticides, antiparasitics and anticancer agents. In mammalian cells microtubules are present both in interphase cells and in dividing cells. In the latter, microtubules constituting the mitotic spindle are highly dynamic and exquisitely sensitive to therapeutic inhibitors. This explains why compounds altering microtubule function have proven to be highly active in patients with cancer. The vinca alkaloids, identified over 50 years ago 1 and the taxanes, first isolated almost 40 years ago 2,3 are currently administered in a large variety of indications including solid tumors and haematological malignancies 4–6. They are most often integrated in combination chemotherapy regimens, including in some curative regimens, for example in patients with non-Hodgkin’s lymphoma. Taxanes have become an essential component in the adjuvant and advanced setting of patients with breast cancer and are also extensively used in patients with ovarian cancer, non small cell lung cancer (NSCLC), and Kaposi’s sarcoma7,8.
A peculiarity of microtubule binding agents is their extreme structural diversity and, in many cases, structural complexity (Figure 1). It should be stressed that many agents were isolated from marine organisms or botanicals which are not cultivated, and in which they are present in minute amounts 9. Many of the most active agents such as taxanes were difficult to develop in the clinic due to scarcity of their natural sources (Pacific yew bark in the case of taxol), a problem which was in some cases later solved by partial or total synthesis of the compounds of interest, although total synthesis has not proven to be the best option for some compounds such as taxanes 10. This problem is still prevalent today for many of the novel microtubule binding agents, explaining, at least in part, the slow clinical development of many of the newer agents 11–13.
Figure 1. Chemical structures of microtubule binding agents according to binding domains.
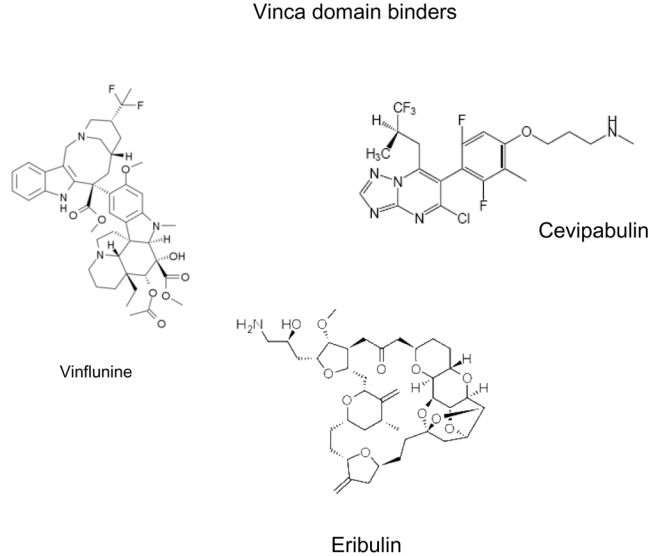
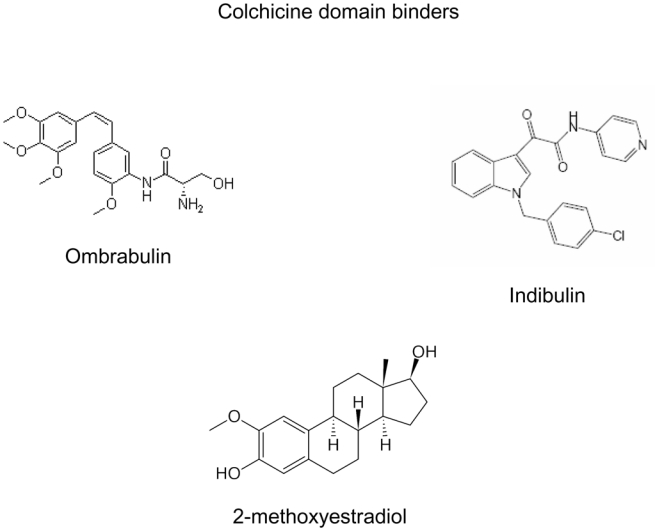
This figure shows the extreme chemical diversity as well as the complexity of many of these agents. The complex structure of certain natural compounds explains the difficulty encountered by chemists to perform total synthesis of these molecules.
In the age of small molecule targeted therapies and therapeutic monoclonal antibodies it is noteworthy that extensive resources and scores of clinical trials are still being devoted to the identification and evaluation of microtubule-targeted agents including taxanes, epothilones, vinca alkaloids, halichondrins, maytansinoids, colchicine-site binding agents, and others. This is partly due to the extremely large untapped reservoir of potential therapeutic natural compounds which influence microtubule dynamics and also to our growing understanding of the role of the microtubule cytoskeleton in cancer cells. After briefly reviewing mechanisms of action of and resistance to anticancer microtubule binding agents, we will focus on novel agents, in particular those that have recently been approved or reached the stage of clinical trials. An increasingly important issue is that of toxicity, since many of these agents cause significant neurological toxicity.
Mechanisms of action
A large number of chemically diverse substances generally originating from natural sources bind to tubulin and/or microtubules (Table 1), altering microtubule polymerization and dynamics in diverse ways. A reasonable hypothesis is that plants and animals evolved this vast number of compounds that mimic endogenous regulators of microtubule behavior in order to avoid predation. All of these compounds are antimitotic agents that inhibit cell proliferation by binding to microtubules and suppressing microtubule dynamics during the particularly vulnerable mitotic stage of the cell cycle (Figure 2). To document the suppressive effects of these agents on microtubule dynamics, most studies have used time-lapse microscopy to analyse interphase microtubules in live cells 14. Spindle microtubule dynamics are more difficult to analyse because of microtubule density but may be indirectly evaluated by the study of centromere dynamics. 15,16 These studies have confirmed that inhibition of spindle and interphase microtubule dynamics occurred at the same concentrations as those inducing mitotic arrest (Box 1).
Table 1.
Characteristics of microtubule binding agents
| Agent | Sensitivity to ABC efflux pumps | Sensitivity to β-tubulin content | references |
|---|---|---|---|
| Vincas | MDR sensitive MRP sensitive |
Sensitive to βIII-tubulin content | 44,189–191 |
| Cryptophycins | MDR insensitive | n.a. | 192,193 |
| Dolastatins | MDR sensitive | n.a. | 194 |
| Taxanes | MDR sensitive MRP2 and MRP7 sensitive |
Sensitive to βIII-tubulin content | 45,46 |
| Epothilones | MDR sensitive | No | 103,195 |
| Discodermolides | MDR sensitive MRP1 sensitive |
Sensitive to βIII-tubulin content | 196,197 |
| Cyclostreptin | MDR insensitive | n.a. | 172 |
| Laulimalides | MDR insensitive | n.a. | 198 |
| Taccalonolide | MDR insensitive | More active if high beta III content | 170 |
| Peloruside | MDR insensitive | n.a. | 169 |
| Hemiasterlin | MDR insensitive | n.a. | 168 |
| Combretastatins | MDR insensitive | Yes | 199–201 |
| 2 methoxyestradiol | MDR insensitive | Inactive against beta I | 125,202 |
MDR: multidrug resistance; ABC: ATP Binding Cassette transport pumps; n.a.: not available
Figure 2. Microtubule formation and binding sites of microtubule inhibitors.
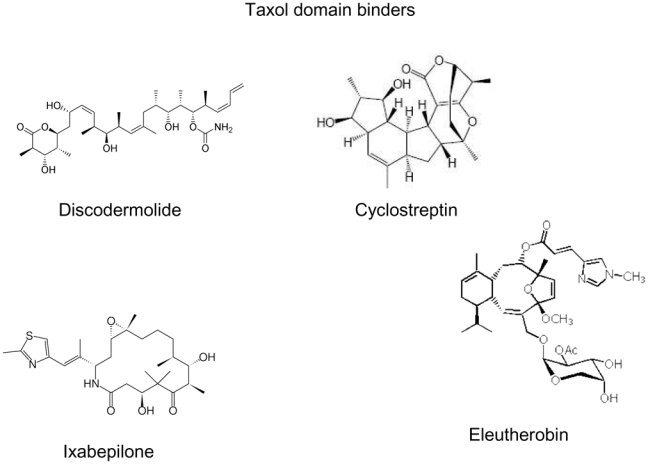
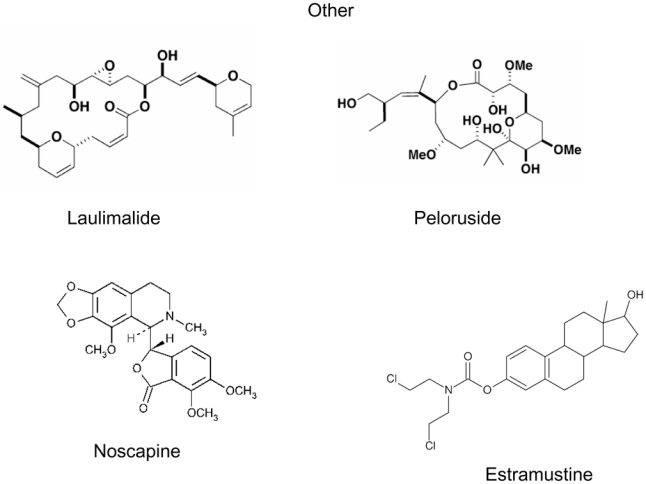
Fig 2A. Soluble tubulin dimers containing one alpha tubulin peptide and one beta tubulin peptide polymerize to form a “nucleus”. Additional dimers are added head-to-tail and the resulting microtubules are highly dynamic structures containing a (+) end characterized by an exposed β tubulin peptide and a (−) end characterized by an exposed α tubulin peptide.
Fig 2B. Binding sites of microtubule inhibitors. While vinca alkaloids bind to microtubule ends, colchicine binds to soluble dimers which can be incorporated within the microtubules. Taxanes bind along the interior surface of the microtubules.
Box 1. Microtubule dynamics.
Microtubules are dynamic structures composed of αβ-tubulin molecules that are constantly integrated or shed into the cytoplasm as the microtubules dramatically grow and shorten. Dynamics can be measured in live cells using fluorescently labelled tubulin (either labelled ex vitro and microinjected or using an expressed GFP-tag) and video-microscopy. Several parameters of dynamics can be assessed to determine the effects of microtubule targeted drugs on dynamics. These include the rates and durations of growing and shortening events and the mean frequency of rescue or catastrophe. Although these parameters are generally analysed on interphase cytoplasmic microtubules and not on spindle microtubules, systems using markers of the ends of spindle microtubules such as GFP-CENP-B have found that the suppressive effects of drugs on dynamics of interphase microtubules are very similar to their suppressive effects on mitotic microtubules.
Box 1.

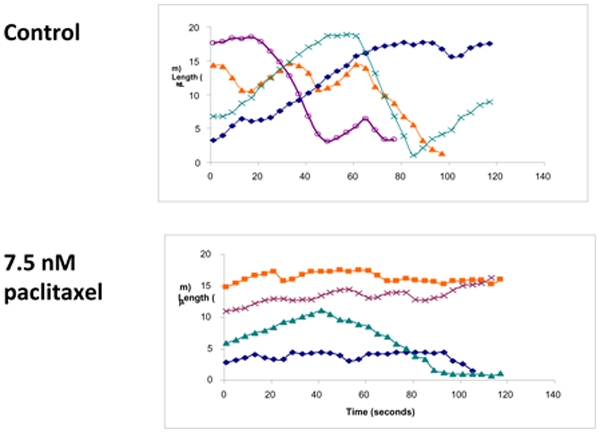
A: Time-lapse sequence analysis of microtubules, using fluorescent-labelled tubulin microinjected into human mammamy adenocarcinoma MCF7 cells
B: Reduced length changes of individual microtubules in the presence of taxol show suppression of microtubule dynamic instability by taxol
Depolymerizing vs. stabilizing agents
The microtubule-targeted antimitotic drugs are often classified into two major groups, the microtubule-destabilizing agents and the microtubule-stabilizing agents, according to their effects at high concentrations on microtubule polymer mass. The so-called “destabilizing” agents inhibit microtubule polymerization when present at high concentrations. Most of these agents bind in one of two domains on tubulin, the “vinca” domain and the “colchicine” domain (Table 1). Vinca site binders include the vinca alkaloids (vinblastine, vincristine, vinorelbine, vindesine, and vinflunine), the cryptophycins, the dolastatins, eribulin, spongistatin, rhizoxin, maytansinoids, and tasidotin. Colchicine-site binders include colchicine and its analogs, podophyllotoxin, combretastatins, CI-980, 2-methoxyestradiol, phenylahistins (diketopiperazine), steganacins, and curacins 17,18. Some of the destabilizing agents, including the hemiasterlins, estramustine, noscapine, herbicides such as carbendazim, psychoactive drugs such as phenytoin, and food components such as sulforaphane found in cruciferous vegetables 19,20, bind to novel sites on tubulin. The “microtubule-stabilizing” agents enhance microtubule polymerization at high drug concentrations and include taxol (paclitaxel, Taxol™), docetaxel (Taxotere™), the epothilones, ixabepilone (Ixempra™) and patupilone, discodermolide, eleutherobins, sarcodictyins, cyclostreptin, dictyostatin, laulimalide, rhazinilam, peloruside A, certain steroids and polyisoprenyl benzophenones. Most of the stabilizing agents bind to the same, or an overlapping, taxoid binding site on beta tubulin which is located on the inside surface of the microtubule 21. However, two of the agents, laulimalide and peloruside A, are not displaced by paclitaxel and for this reason are believed to bind to a novel site on tubulin 22,23. Overall several hundred compounds have been reported to arrest mitosis by their effects on microtubules. In all cases where it has been investigated, they do so most potently by suppressing microtubule dynamics 24,25.
Suppression of microtubule dynamics
Both classes of drugs, those that increase and those that decrease microtubule polymerization at high concentrations, potently suppress microtubule dynamics at 10 to 100-fold lower concentrations. The sensitivity of microtubule dynamics to regulation means that both kinds of microtubuleregulating drugs can kinetically stabilize the microtubules without changing the microtubule polymer mass. At a very basic mechanistic level, these two classes of drugs act similarly to block mitosis. Supporting this common mechanism of action is the finding that taxanes and vincas or estramustine can be combined clinically in chemotherapy regimens with no apparent antagonism 26–28. In addition, combinations of taxanes with vincas, estramustine or colchicine analogs have shown synergism in vitro 29,30. At high concentrations, there are clear differences in their cellular effects on microtubule mass 31. However, to target cells as they enter mitosis in order to gain maximum therapeutic efficacy it may be important it may be more important to maintain a low drug concentration in the tumor cells or in their adjacent endothelial cells for a reasonably long duration than to achieve a brief pulse of high intracellular drug concentration 32.
Antiangiogenic and vascular-disrupting effects
The tumor vasculature is a superb therapeutic target as it is easily accessible to blood-borne drugs, and tumor cells generally die unless continually supplied with oxygen and nutrients from the blood. The two approaches to inhibit vascular function are to inhibit angiogenesis (the formation of new blood vessels), and to destroy the integrity of existing tumor vasculature using vascular-disrupting agents 33. Formation of new blood vessels involves both proliferation and migration of endothelial cells, and both of these processes appear to be extraordinarily sensitive to microtubule-targeted drugs 25,34. It has been suggested that prolonged exposure times and frequent dosing of low concentrations of microtubule-targeted drugs, the so-called “metronomic” schedules, may favor the antiangiogenic properties of these agents but clinical confirmation of such an effect will require both randomized trials and the demonstration of an antiangiogenic effect in patients 32,35.
Since the late 1990’s, the combretastatins and N-acetylcolchicinol-O-phosphate, compounds that resemble colchicine and bind in the colchicine domain on tubulin, have undergone extensive development as vascular-disrupting agents 36. When combretastatin-A-4 phosphate (CA-4-P) is added to cultures of endothelial cells, microtubules rapidly depolymerize, cells become round within minutes, bleb and detach 37. When administered to rodents, the bloodflow may drop by >95% in less than an hour, vascular permeability increases and haemorrhaging from peripheral tumor vessels occurs 38–40. These vascular-disrupting agents appear to be fairly specific for tumor vasculature although the reasons for this specificity are not known. Since the targeted endothelial cells are non-tumor cells, a potential advantage of this approach is that the cells may be less susceptible to the development of resistance to these drugs than genetically unstable tumor cells. The development of these agents has also prompted novel methods aiming to evaluate changes in tumor perfusion, such as dynamic MRI measurements of gadolinium diethylenetriaminepentaacetate uptake and washout, and positron emission tomography of 15O-labeled water or dynamic contrast enhanced magnetic resonance imaging. 40–42
Several currently-used microtubule-targeted agents, such as the vinca alkaloids, damage tumor vasculature in animal models. It is our belief that the difference between these classical anti-mitotic anti-proliferative microtubule-targeted agents and the novel agents that are undergoing clinical testing as vascular-disrupting agents may rely on the fact that the effects of novel vascular-disrupting agents are more rapidly reversible, either because of the reversibility of their binding to tubulin, or their lack of long-term retention in cells. Those agents which exert depolymerizing effects over a short period of time may act best as anti-vascular agents while those that are retained and induce a long-term mitotic arrest may work best as antiproliferative agents.
Mechanisms of resistance
Understanding mechanisms of resistance to microtubule-binding agents is a key element in the development of novel, more potent microtubule-targeted compounds. Resistance to microtubule-binding agents can occur at several levels in the pharmacodynamics of these agents, including primarily cellular efflux of the anticancer agents, ineffective interaction with the target, and deficient induction of apoptosis. In addition, resistant tumors and cell lines show a multitude of changes in protein and microRNA expression whose relationship to the actions of microtubules is not always easy to discern.
ABC proteins and drug efflux
Membrane efflux pumps of the ATP binding cassette (ABC) family represent the primary resistance mechanism developed by tumor cells when these are exposed to microtubule binding agents in vitro 43. While Pgp, the product of the mdr1 gene is responsible for the “classical multidrug resistant phenotype” (MDR) and actively effluxes both vincas and taxanes, thereby reducing their intracellular concentrations and cytotoxic activity, other transporters transport only some types of antitubulin agents. Vincas are actively transported by the MRP1 protein, taxanes are substrates for MRP2 and MRP7, and epothilone B is transported by MRP7 44–46. Given the potential importance of these efflux pumps as mechanisms of resistance to chemotherapy, newer agents which are insensitive to active efflux have been identified and further developed (Table 1). The clinical relevance of ABC pumps in patients with cancer remains controversial, with limited data to support the routine study of these proteins in patients 47. While the expression of these pumps in primary tumors often correlates with a lower response rate to therapy with microtubule-targeted agents, the presence and/or function of ABC proteins in clinical samples is not generally used to tailor therapy in individual patients due to difficulties in standardizing assays 48,49. Attempts to reverse drug resistance by combining microtubule agents with inhibitors of drug efflux proteins have been disappointing 50. Conversely, the fact that microtubule-binding agents constitute substrates for ABC efflux pumps significantly limits their diffusion inside the central nervous system, and constitutes an obstacle to their oral administration, suggesting that novel compounds which are less susceptible to transport by ABC proteins could possess original pharmacokinetic profiles 51.
Alterations in microtubules
A second level of resistance to antitubulin agents consists in alterations in the target of these agents, the tubulin/microtubule complex. Qualitative or quantitative modifications of microtubules which can influence drug binding or the effects of drug binding on tubulin conformation and/or GTPase activity are likely to influence sensitivity to microtubule binding agents. These microtubule-based mechanisms of resistance to microtubule binding agents are extremely varied, and concern either individual components of the microtubule array itself or regulatory proteins. A variety of proteins participate in tubulin protein folding, tubulin dimer sequestration, microtubule dynamics or interact with microtubules and tubulin and participate in their regulatory pathways. These include the proteins FHit, survivin, MAP2, MAP4, stathmin, STOP and survivin 24,52–57. Alterations in the levels, intracellular localizations (nuclear or cytoplasmic), post-translational modifications and function of these proteins are likely to influence sensitivity to microtubule binding agents.
Microtubules are composed of at least 13 isotypes of α- and β-tubulin. The quantitative tubulin isotype composition of microtubules has been reported to influence sensitivity to microtubule binding agents. Most notably, increased levels of beta tubulin III is associated with reduced response rates to taxanes in several tumors including lung, breast and ovarian cancers 54,58. In contrast, epothilones may be indifferent to beta III tubulin content 59. In addition to beta III tubulin, increased levels of beta V and beta II tubulins have also been associated with taxane resistance 60–62. In contrast, decreased expression of class III beta-tubulin and increased levels of MAP4 protein have been detected in vinca resistant cell lines along with increased microtubule stability in these resistant cells as identified by the high levels of polymerized tubulin 63. However, in contrast, small interfering RNA-mediated knockdown of either betaII- or betaIVb-tubulin hypersensitized lung cancer cell lines to Vinca alkaloids 64. It is worth noting that the role of beta III tubulin expression in cancer may extend beyond its role in drug resistance. Recent studies have found that beta III tubulin appears to be a “survival factor” that can increase the incidence and progression of cancer irrespective of drug treatments 65. These preclinical data have been confirmed in the clinic since high levels of beta III tubulin have been found to be associated with worse prognosis and lower response rates in a variety of tumor types 58,66.
There are several reports of mutations in tubulin genes in cell lines resistant to microtubule binding agents 67–69. However, confirmation of these observations in the clinic is currently lacking. In spite of early suggestions that mutations in the taxol binding site were found in patients with NSCLC 70, subsequent studies have found no evidence that polymorphisms in beta tubulin genes are frequent events in clinical samples 71,72.
Resistance due to deficient apoptotic signaling
A third mechanism of resistance to microtubule binding agents involves apoptotic signalling downstream of the microtubule insults to which tumor cells are exposed. Microtubules physically interact with a variety of cell organelles and various regulatory proteins (Box 2). An interesting case is that of P53 protein and sensitivity to taxol. High hopes were raised by the observation that inactivation of P53 — a common mechanism of resistance to anticancer agents — induced preferential sensitivity to taxol in normal human or murine fibroblasts 73. However, later observations suggested that P53 status had little or no impact on sensitivity to taxanes74,75. Several studies have failed to establish P53 as a predictive factor of response to taxanes in the clinic76,77. p53 may influence sensitivity to microtubule binding agents by regulating microtubule composition and dynamics thereby suggesting that p53 is not only a guardian of the genome but also of the microtubule cytoskeleton as well 57. Apoptotic regulators or effectors also influence sensitivity to taxanes, for example a small molecule inhibitor of BclXL sensitized tumor cells to paclitaxel 78.
Box 2. Interactions of microtubules with other proteins and cellular organelles.
Microtubules interact with a variety of intracellular components including mitochondria, the Golgi apparatus, the endoplasmic reticulum, and lysosomes. In the mitotic spindle microtubules allow the proper alignment of chromosomes during metaphase, followed by the equal distribution of chromatids to the two daughter cells during anaphase. This phenomenon relies on the physical interaction between microtubule (+) ends of the microtubules and the kinetochores. A number of key proteins involved in cell cycle and/or apoptosis have also been shown to physically interact with microtubules. P53 is physically associated with dynein, a microtubule motor protein. Bcl2, survivin and several other proteins that play a role in cell survival also colocalize with microtubules although it is not clear whether microtubules serve as molecular scaffolds for these proteins to exert their activity or whether the proteins are sequestered by microtubules and therefore functionally inactive.
It is also becoming clear that the balance of expression of proteins that have no currently recognized direct interactions with microtubules or tubulin can also play a role in resistance or sensitivity to microtubule-targeted drugs, possibly through a complex web of interactions with other proteins that are part of the recognized microtubule functions in transport, cell cycle, signalling, and apoptosis. Examples of these include prohibitin, glutathione-S-transferase π, α-defensins, inflammation, GTSE-1 (G(2) and S phase-expressed-1)-protein modulation of p21, and hypoxia and hypoxia-inducible factor 1 α {Bublik #249; Patel #250; Huang #251; Bauer #252; Townsend, 2003 #253}. Micro RNAs have also been found to contribute to resistance to microtubule-targeted drugs. For example miR-125b conferred resistance to paclitaxel by suppressing the pro-apoptotic BAK1 and miR-148a increased sensitivity to paclitaxel by decreasing expression of mitogen and stress-activated protein kinase MSK1 {Zhou #254; Fujita #255}.
Novel microtubule targeted agents and/or formulations
Microtubule-binding agents are unique among anticancer agents not only because of their original mechanisms of action but also because of their extreme structural diversity. In most cases natural agents with potent antitumor activity have led the way for original synthetic analogues. Surprisingly this remains true even for the vinca and taxane families, the first members of which have been in clinical use for decades (Table 2).
Table 2.
Selected microtubule-binding agents which have been approved or have undergone clinical evaluation
| Binding domain | Family | Agent | Approved Indications * | Clinical trials | Comments |
|---|---|---|---|---|---|
| Vinca | Vincas | Vincristine | ALL, lymphomas Various solid tumors |
Various tumor types | Natural compound Generic Parenteral administration |
| Vinblastine | Lymphomas Various solid tumors |
Various tumor types | Natural compound Generic Parenteral administration |
||
| Vinorelbine | Breast, NSCLC | Various tumor types | Semi-synthetic Generic Oral and parenteral administration |
||
| Vindesine | ALL, lymphoma Lung cancer | Various tumor types | Semi-synthetic Generic Parenteral administration |
||
| Vinflunine (Javlor®, Pierre Fabre) | Bladder | Breast in combination with Herceptin | Semi-synthetic Parenteral administration |
||
| Liposomal vincristine | - | leukemia melanoma, myeloma, sarcoma | Prolonged and regular delivery | ||
| Dolastatins | soblidotin (TZT-1027) | - | Phase I in advanced solid tumors No ongoing trials |
Responses in NSCLC and esophageal cancer | |
| romidepsin Istodax® Gloucester Pharmaceuticals |
Cutaneous T cell lymphoma | Myeloma, lymphoma, solid tumors | Dolastatin 15 analog | ||
| brentuximab vedotin (SGN 35) | - | Phase III trial recruiting in Hodgkin’s disease | Antibody-vectorized agent directed against CD30 positive malignancies | ||
| Cryptophycins | Cryptophycin 52 LY355703 | - | Phase II NSCLC Terminated | Caused significant neurological toxicity | |
| Halichondrin | Eribulin (E7389, NSC 707389) | - | Phase III in advanced breast cancer | Improved OS when compared to treatment of physician’s choice | |
| Hemiasterlin | E-7974 | - | Phase I | Hematological MTD | |
| Maytansinoids | Mertansine immunoconjugates (BT-062, IMGN388, BIIB015) | - | Head and neck, oesophagus, advance HER2 positive breast cancer, myeloma | Phase II and III underway | |
| Folate vectorized vinca alkaloid | EC-145 | Ovarian, endometrial, lung cancer | Folate-targeted vinca alkaloid conjugate | ||
| Taxane | Taxanes | Paclitaxel Taxol® |
Ovarian, breast, NSCLC | Various solid tumor types | May induce hypersensitivity reactions |
| Docetaxel Taxotere® |
Breast, NSCLC, prostate, stomach, head and neck | Various solid tumor types | May induce hypersensitivity reactions | ||
| cabazitaxel (XRP6258) Jevtana® |
Metastatic hormone-resistant prostate | Approved June 2010 | |||
| Milataxel (MAC-321, TL-139) | - | Phase II mesothelioma | Active in preclinical models of resistance to taxanes 203,204 | ||
| Larotaxel (XRP9881) | - | Phase III pancreatic | Active in preclinical models of resistance to taxanes, poor MDR substrate 205–207. | ||
| Ortataxel IDN-5109 BAY 59-8862 |
- | Phase II taxane-resistant tumors | Active in Pgp-expressing models 208 | ||
| Tesetaxel | - | Phase II gastric | Oral administration | ||
| DJ-927 | Phase II colorectal Phase II melanoma |
Is not transported by Pgp209,210 | |||
| BMS 275183 | - | Phase II NSCLC Terminated | Oral administration Unpredictable pharmacokinetics |
||
| TPI 287 (ARC-100) | - | Phase II prostate cancer Phase I pediatric CNS cancers |
Investigated in neurological tumors in combination with temozolomide | ||
| Nab-paclitaxel (ABI-007) Abraxane® Abraxis Bioscience Nab-docetaxel (ABI-008) |
Breast cancer | Various solid tumors Prostate cancer |
Shorter infusion times than paclitaxel Does not require premedication |
||
| NKTR-105 | Phase I | PEGylated formulation of docetaxel; pre-treatment with corticosteroids not required | |||
| Epothilones | Ixabepilone Ixempra® Bristol Myers Squibb |
Breast cancer | Solid tumors | Several ongoing trials in solid tumors Is not a substrate for Pgp |
|
| Patupilone (epothilone B) | - | Brain metastases in breast cancer, ovarian, melanoma, other solid tumors | Penetrates in the CNS Is not a substrate for Pgp Possesses radiosensitizing properties | ||
| Sagopilone | - | Glioblastoma, prostate, lung cancers | First fully synthetic epothilone Penetrates in the CNS | ||
| KOS 1584 (epothilone D) | - | NSCLC Phase II | Investigated in breast and prostate cancer | ||
| Discodermolide | - | - | Phase I Terminated | Pulmonary toxicity | |
| Colchicine | CI-980 | - | - | Phase II trials Terminated | No responses observed in sarcoma or colorectal cancer211,212 |
| 2 methoxy-estradiol (ME2) Panzem® EntreMed |
- | - | Phase II in prostate, myeloma, glioblastoma | Endogenous metabolite of estradiol with no affinity for estrogen receptor Side effects: DVT and increased transaminases |
|
| 1069C85 | - | - | Phase I Terminated | Oral administration 213 | |
| ABT 751 E7010 |
- | - | Phase II in various solid tumors No ongoing trials |
Orally bioavailable sulfonamide Neurotoxicity 214 |
|
| Indibulin | - | - | Phase I/II in metastatic breast cancer | Discriminates between neuronal and non-neuronal tubulin 148,215 | |
| Combretastatins | Fosbretabulin (CA4 phosphate) | - | Phase II in lung and thyroid cancer, glioma | Vascular disrupting agent | |
| Verubulin | Phase II glioblastoma | Vascular disrupting agent | |||
| Crinobulin | - | Phase I | Vascular disrupting agent | ||
| Plinabulin | Phase I | Vascular disrupting agent | |||
| Ombrabulin | - | Phase III in sarcoma | Vascular disrupting agent | ||
| Other | Noscapinoids | Noscapine | - | Phase II multiple myeloma | Oral opium alkaloid used as antitussive |
| Estramustine | - | Prostate cancer | Combination with taxanes, vincas, ixabepilone in prostate cancer | Generic Binds to microtubule associated protein |
ALL: acute lymphoblastic leukemia; CNS: central nervous system; DVT: deep vein thrombosis; MTD: maximal tolerated dose; NSCLC: non small cell lung cancer; OS: overall survival; Pgp: P glycoprotein
Data in this table have been obtained from clinicaltrials.gov, Pubmed, ASCO, company sites and the Thomson Pharma Partnering database.
Vinca domain binding agents
Vinca alkaloids (vincristine, vinblastine, vindesine and vinorelbine), originally isolated from the periwinkle plant Catharanthus rosea, represent the oldest and to this day most diversified family (in terms of number of approved compounds within a given family) of microtubule targeted agents. Vinflunine (Javlor™), a novel fluorinated compound which was obtained by superacid transformation of vinorelbine in the presence of fluorhydric acid, has recently been approved for the second-line treatment of bladder cancer 86. Also a liposomal formulation of the off-patent agent vincristine, which allows a prolonged and regular delivery of this active compound, is currently the object of clinical trials.
The dolastatin family, originally identified by isolation of marine peptides from the ocean shell-less mollusk Dolabella auricularia, includes dolastatin 10, cemadotin, tasidotin (ILX651), soblidotin, and malevamide E 87. While dolastatin 10 itself was not active in patients with various tumors including advanced breast cancer or pancreaticobiliary cancers, its analog soblidotin jnduced minor responses in patients with NSCLC and a partial response in a patient with advanced esophageal cancer in a phase I trial but was not further evaluated in a phase II trial 88,89. Romidepsin, a dolastatin 15 analog which also possesses activity as an HDAC inhibitor, was recently found to be active in cutaneous T cell lymphoma, with a 34% objective response rate.90
Eribulin mesylate, a synthetic halichondrin derivative, was found to be active in patients with metastatic breast cancer relapsing after anthracyclines and taxanes. In a randomized phase III trial patients receiving single agent eribulin mesylate benefited from significant improvement in overall survival when compared to patients treated according to physician’s choice 91.
Taxol domain binding agents
Besides paclitaxel (Taxol™) and docetaxel (Taxotere™), cabazitaxel (Jevtana™, XRP 6258, RPR116258, Sanofi-Aventis) has displayed promising results in patients with breast and prostate cancer and has recently been approved by the FDA for the treatment of hormone-resistant metastatic prostate cancer after failure of docetaxel 92,93. Issues with currently available taxanes include their mode of administration, currently limited to the intravenous route, their poor water solubility, requiring the use of surfactants such as Cremophor and ethanol for intravenous administration, with an associated risk of hypersensitivity reactions 94,95, and the nearly universal recurrence of disease when patients are treated in the advanced setting. Some of the novel taxanes are poor substrates for ABC transport pumps and may in some cases be administered orally or pass through the blood-brain barrier, a particularly important property for the treatment of CNS metastases.
Conversely a phase II trial evaluating BMS 275183 given orally twice weekly in patients with relapsing NSCLC was terminated because of highly variable pharmacokinetics. Unpredictable individual pharmacokinetics is a major limitation in the development and use of orally administered anticancer agents.
Novel taxane formulations are being developed with the intent of reducing issues associated with poor solubility or hypersensitivity. In a phase III trial comparing nanoparticle albumin-bound paclitaxel (Abraxane™, nab-paclitaxel) and conventional docetaxel for the therapy of patients with metastatic breast cancer, nab-paclitaxel was associated with better outcome as well as with a lower rate of severe neutropenia and a similar rate of reversible sensory neuropathy 96. Nab-paclitaxel has also demonstrated activity in other settings including melanoma, gynaecological tumors and prostate cancer 97–99. Several novel generic formulations of paclitaxel and docetaxel aim to eliminate surfactants from current formulations, which may eventually lead to reduced hypersensitivity reactions 100.
Epothilones were originally isolated from the myxobacterium Sorangium cellulosum. They represent a promising novel family of agents for cancer treatment as they may retain activity against taxane-resistant tumors 101,102. Epothilones are easier to produce than taxanes, display good water solubility and do not appear to be substrates for the Pgp efflux pump 103, allowing passage through the blood brain barrier 104. Besides ixabepilone (Ixempra™), a semisynthetic analog of epothilone B, which is currently approved for the treatment of advanced taxane-resistant breast cancer in the United States, several other epothilones are currently being studied in clinical trials. These include patupilone 105, sagopilone 106–108 and KOS-862 (epothilone D) 109,110 which are being evaluated in various solid tumor types.
Colchicine domain binding agents
Combretastatins represent an exciting family of microtubule targeted agents as they are lead compounds of the vascular targeting or vascular disrupting agents, compounds which produce rapid disruption of tumor blood flow, probably by their effects on the microtubule cytoskeleton of endothelial cells. In phase I trials combretastatin A4 (CA4), isolated from the Combretum caffrum tree, induced unusual toxicities including tumor pain, ataxia and cardiovascular modifications, including prolonged QTc interval and ECG modifications consistent with acute coronary syndrome 111–113. Fosbretabulin (CA4 phosphate) is currently being evaluated in combination trials in patients with anaplastic thyroid cancer and with chemotherapy naive lung cancer 114. Other antivascular agents that have undergone clinical evaluation include ZD6126 115, OXI4503 116, ombrabulin (AVE8062A)117, crinobulin (EPC2407)118 as well as auristatin PE (TZT-1027, a dolastatin derivative) 119 which binds in the Vinca domain. A key issue for the approval of this family of agents will be the lack of significant toxicity on normal vasculature, as well as the mode of administration in combination with other agents.
Additional agents binding at or near the colchicine binding site of tubulin such as CI-980 and 1069C85 have been discontinued while ABT-751, and indibulin are currently in phase I 120. 2-methoxyestradiol (ME2), displayed limited activity in patients with hormone-refractory prostate cancer 121, breast cancer 122 and multiple myeloma 123 leading to improved formulations consisting of nanocrystal colloidal solutions 124. The lack of myelosuppression by ME2 has been attributed to the resistance of the hematopoietic-specific beta tubulin to this agent 125.
Other agents
Several other agents with original properties have undergone clinical evaluation. Cevipabulin (TTI-237) is an unusual agent which appears to bind the vinca site but promotes microtubule polymerization 126 Noscapine, which has the ability to cross the blood-brain barrier 127 is currently being evaluated in a phase I/II trial in patients with multiple myeloma (NCT00912899). A number of analogs with increased potency are under investigation.
Toxicity of microtubule targeted agents
The evaluation of some microtubule binding agents has been discontinued because of significant toxicity. This is exemplified by the discodermolides which are highly potent natural polyketide products isolated from the Caribbean sponge Discodermia dissolute, which appear to be synergistic with taxol 128,129. A phase I trial of this compound (Novartis) initiated in 2004 was interrupted because of significant pulmonary toxicity. Dictyostatin is a structurally related compound for which the total synthesis has recently been obtained 130. Cryptophycins were obtained from cyanobacteria or were prepared by total synthesis. While some disease stabilisation was observed in patients receiving cryptophycin 52 (LY355703), there were no responses in patients treated for advanced NSCLC in spite of significant neurological toxicity 131,132.
Neurological toxicity
A major limitation in the use of microtubule-targeted agents is the high rate of neuropathy induced by these compounds 133. This potentially severe and dose-limiting side effect, which is dose-cumulative and more frequent in patients with preexisting neuropathy, be it due to chronic alcoholism or diabetes mellitus, usually manifests itself as a painful and debilitating peripheral axonal neuropathy for which there is currently no effective symptomatic treatment 134. This has prompted the search for predictive factors such as neurologic function tests or biological markers such as myelin basic protein and gliofibrillar acid protein 135–137 Other manifestations include constipation or intestinal paralysis due to neurological toxicity against the autonomic nervous system. While symptoms tend to disappear a few months after the end of treatment, some patients retain significant sequelae several years after therapy. The preferential toxicity of these agents for the nervous system is not understood at a mechanistic level but can be partially explained both by the relative abundance of tubulin in neurons, and the importance of an intact, functional microtubule cytoskeleton for adequate nerve conduction.
Peripheral neuropathy has been a limiting factor in the development of several agents, leading, as in the case of cryptophycins, to termination of their development. In contrast, there have been few reports of central nervous system (CNS) toxicity with the currently administered agents, partly due to the fact that they are Pgp efflux pump substrates and thus do not cross the blood brain barrier. The development of newer agents which are not substrates of Pgp might be associated with CNS toxicity, or with activity against tumors within the CNS 104. The question of neuropathy is particularly important when considering the combination of these agents with other potentially neurotoxic agents. Among the classical agents, the platinum compounds, which induce peripheral neurotoxicity to various degrees, are commonly used in combination with taxanes and vincas, in particular in patients with NSCLC or with germ cell tumors, in the latter case with a large proportion of long term survivors 138. Among the more recently approved agents, several compounds, such as bortezomib or thalidomide, can also induce high grade peripheral neuropathy in a significant proportion of patients. The mechanisms of neurotoxicity have not been precisely determined for all of these compounds and may or may not be related to microtubules 139,140. The combination of these agents with microtubule-targeted agents may therefore prove to be difficult and assays, quite likely based on genetic polymorphisms, predicting high grade sensory neuropathy in individual patients would be of great use.
A major difficulty in the screening of novel agents is the lack of adequate preclinical models of druginduced peripheral neuropathy. Glial cell cultures are extensively used to analyse this type of toxicity in vitro, but animal models that reliably correlate with or predict neurotoxicity in patients remain imperfect 141–145. The development of reliable predictive models would be of great use for the future development of novel agents and of neuroprotective compounds. Alternatively the identification of differences between the microtubule cytoskeleton in peripheral nerves and tumor cells could serve as a basis to design or select novel agents with reduced neuropathy. Eribulin induced no significant reduction in nerve conduction velocity or amplitude in caudal and digital nerves when administered to mice at the maximal tolerated dose 146. Phase I and II clinical trials of eribulin demonstrated significant activity with only a low incidence of neuropathy and no grade 4 neuropathy 147. Indibulin (ZIO-301/D-24851) has been reported to distinguish between mature neuronal tubulin and non-neuronal tubulin and has entered clinical evaluation as an oral formulation 148,149. In a phase I study, ispinesib (SB-715992), a kinesin inhibitor was found to induce myelosuppression but no neurotoxicity 150. Phase II trials evaluating ispinesib as a single agent have not yet demonstrated significant activity 151,152.
Other toxicities
Myeloid toxicity is frequently observed with microtubule-targeted agents, with subtle differences between compounds within the same family 153. Neutropenia is often the most frequent and/or severe side-effect observed in combination regimens including these agents 70,86,154. In several recent phase II studies neutropenia was one of the dose-limiting toxicities 88,155–158. This toxicity, which is often added to similar toxicities of other agents used in combination regimens, is usually manageable. In contrast, some toxicities are relatively compound specific, such as fluid retention observed in patients receiving docetaxel or diarrhoea after patupilone therapy 159–161.
An intriguing issue concerns the possible mutagenic properties of microtubule binding agents and henceforth the risk that they may increase the risk of secondary tumors. Given the fact that cells exposed to these compounds can develop aneuploidy due to missegregation, there is a theoretical risk that these agents might increase the risk of iatrogenic leukemias and/or solid tumors. Chromosomal instability and an aneuploid-prone phenotype have been described to be correlated with response to taxanes 162,163. Administration of paclitaxel to nude mice and to rhesus monkeys has caused prolonged aneugenicity and abnormal mitoses, respectively, but clinical confirmation of such an effect has yet to be demonstrated 163,164. As these agents have been widely used in combination with alkylating agents, and the initial indications mostly concerned patients whose life expectancy was short, it has been difficult to establish whether these agents are potentially carcinogenic per se. As a result of the widespread use of these agents in the adjuvant setting, in patients whose prognosis may be globally favorable, the question of whether microtubule-targeted agents increase the risk of secondary neoplasms has become clinically relevant.
Improving therapy with microtubule-targeted agents
Microtubules represent a highly-validated target in cancer therapy, explaining the abundance of efforts to develop novel agents directed against this target. All of the currently approved compounds bind directly to tubulin, either to soluble tubulin or to tubulin that is polymerized into microtubules, although the binding occurs at different sites on the tubulin molecule or to different regions of the microtubule. Novel approaches aim to improve upon existing compounds either by selecting agents that are insensitive to resistance mechanisms, that increase tumor selectivity, that reduce side effects such as peripheral neuropathy or by targeting the numerous other components of the tubulin/microtubule complex.
Several promising agents have been reported in preclinical models. These include eleutherobin165, laulimalide166,167, hemiasterlins168, peloruside A22,169, taccalonolide170, coumarins171 and cyclostreptin172. Most of the novel agents have been selected because of their activity in models that show resistance to taxanes. Several of these novel agents are not substrates of efflux pumps such as Pgp or other ATP-Binding Cassette proteins. In some cases these agents are also insensitive to the presence of mutations in beta tubulin and/or to overexpression of specific tubulin isotypes, in particular tubulin βIII. This has led some investigators to identify either βIII-indifferent agents, or βIIItargeted agents 170,173. The demonstration that tumor aggressivity and in some cases of sensitivity to chemotherapy is influenced by the content of βIII tubulin isotype suggests that the development of agents targeting this isotype would be of particular interest in patients with high risk disease due to high expression of this isotype. Such a strategy is corroborated by the reports that inhibition of tubulin III by oligonucleotides and by silencing RNA induced sensitization of tumor cells to various anticancer agents 62,64. In this regard, secotaxoids, which are predicted to bind well to beta III tubulin isotype and retain activity in paclitaxel resistant preclinical models appeared to be particularly promising but have not been further evaluated in the scope of recent clinical trials174. Another attractive approach involves vectorisation of microtubule binding agents to the tumor cell using a monoclonal antibody. Maytansine conjugates are being studied in various indications, in particular in haematological diseases and breast cancer 175–177. A recent trial of trastuzumab-DM1, a maytansinoid conjugated to the anti-HER2 therapeutic antibody trastuzumab, showed good efficacy in metastatic breast cancer and the CD-56 targeting antibody-maytansine conjugate, lorvotuzumabmertansine, has shown promising results in solid and liquid tumors that express CD56 178,179.
It is now clear that alterations in microtubule dynamics are the main mechanism of action of microtubule binding agents 24,180. Given the multiple roles of microtubules, several proteins other than tubulin itself are likely to constitute therapeutic targets in cancer cells. These potential targets include proteins involved in the lifecycle of tubulin peptides and dimers as well as proteins involved in microtubule nucleation, dynamics, and interaction with chromosomes or cellular organelles. Of particular interest are the motor proteins such as kinesin Eg5 (for which the first inhibitors such as AZD4877are currently being evaluated 181) and tau protein182, a key microtubule-associated protein which has been correlated with outcome in patients with breast cancer. Another potential target is survivin183, a protein that is intimately involved in spindle microtubule behaviour as well as apoptosis. Other potential targets include MCAK, a mitotic centromere-associated protein that regulates microtubule dynamics184, and stathmin185, an important regulator of the soluble tubulin dimer pool as well as dynamics.
Another important avenue for the optimization of microtubule binding agents is the identification of patient subsets most susceptible to respond to therapy or to develop significant toxicity, using tumor-related parameters or patient characteristics 186. This approach is of particular interest in diseases such as lung cancer, in which there are several therapeutic alternatives, none of which has clearly proven to be superior 187. A randomized trial is currently analyzing the potential benefit of ixabepilone in patients with βIII tubulin-positive lung cancer (NCT00723957). Analyses of targeted polymorphisms in patients receiving microtubule-binding agents has not yet allowed the identification of patients with the highest chance of response or the highest risk of developing dose-limiting side effects of chemotherapy 188. High throughput analyses of large patient cohorts and validation series will help establish personalized therapy with microtubule-binding agents.
Concluding thoughts
In light of the development of microtubule-targeted agents over the past decades, the recent approvals of a novel vinca alkaloid, a novel taxane and the first epothilone, and the recent advances in the understanding of the role of the microtubule cytoskeleton in cancer cells, the stakes are high that this family of anticancer compounds not only will still be in use years from now, but will also will be considerably enriched with less toxic and highly active molecules. The tremendous diversity of naturally occurring compounds interacting with mammalian microtubules represents a largely untapped source for future anticancer agents. A major aim in this very dynamic field will be to purify, screen and ultimately offer to the cancer patient the best of nature’s gems.
Acknowledgments
This work was supported in part by the Association pour la Recherche contre le Cancer and the Ligue contre le Cancer
Glossary
- Adjuvant therapy
a treatment which is administered to patients with minimal or no detectable sign of disease, in order to prevent disease recurrence
- Catastrophe
the switch of a growing or stable microtubule end to rapid shortening
- Dynamics
the nonequilibrium dynamic behaviors of microtubules in cells which are crucial to their functions. The two kinds consist of “dynamic instability” in which the ends of individual microtubules randomly switch between phases of growth and shortening and “treadmilling” which consists of net growth at one microtubule end and a balanced net shortening at the opposite end resulting in a flow of tubulin subunits through the microtubules. Microtubule dynamics are much faster during mitosis than in interphase and are crucial to cell division, making mitotic cells highly susceptible to microtubule-targeted drugs. They are also important in the trafficking of elements within the cell and for cell migration; their suppression is thought to impair cell metastasis
- Kinetochore
the complex assemblage of proteins at the chromosome centromere to which dynamic mitotic spindle microtubules attach, ultimately producing equal segregation of chromosomes to the daughter cells
- Microtubule-associated proteins (MAPs)
a number of proteins bind very tightly to microtubules and can be purified along with the microtubules. The most famous of these are tau and Microtubule-associated proteins 2 and 4. In addition, many proteins can bind less tightly to microtubules in cells and regulate their behaviour
- Microtubule binding agents
drugs and endogenous regulators of microtubule dynamics can bind selectively to several sites on a microtubule. They can bind preferentially to one or both microtubule ends (vincas, eribulin, cryptophycins, maytansinoids and others) or to the sides of the microtubule (taxanes, epothilones). They may also copolymerize into the microtubule with the tubulin (colchicines)
- Tubulin dimer
the heterodimeric protein subunit that polymerizes into microtubules. Each subunit is composed of one α-tubulin and one β-tubulin molecule
- Tubulin isotype
there are at least 13 different isotypes of α- and β-tubulin. The tubulin isotype composition of cells varies between cell types within the same tissue and between tissues. For example, brain cells contain high amounts of βIII-tubulin, but non-neuronal cells generally contain only low amounts of this isotype. Isotype content also differs between tumor cells and the nontumor cells of the same tissue. The complement of tubulin isotypes can be induced to change in response to treatment by many drugs
- Rescue
the switch of a shortening microtubule end to growth or to a state of stable microtubule length
Footnotes
Further information
Initiation of a phase I/II study of oral indibulin in breast cancer patients by ZIOPHARM http://ir.ziopharm.com/releasedetail.cfm?ReleaseID=457504
Conflicts of interest: CD has received research funding from Pierre Fabre, Sanofi-Aventis and has worked as a consultant for Sanofi-Aventis and Bristol Myers Squibb
MAJ has received research support from Bristol Myers Squibb, Eisai Pharmaceuticals, and Immunogen
Contributor Information
Charles Dumontet, Oncogénèse et progression tumorale INSERM : U590, Université Claude Bernard - Lyon I, CRLCC Léon Bérard, 28, Rue Laennec 69373 Lyon Cedex 08,FR.
Mary Ann Jordan, Dept. Mol., Cell., Devel. Biology Neuroscience Res. Inst. University of California Santa Barbara, US.
References
- 1.Noble RL, Beer CT, Cutts JH. Role of chance observations in chemotherapy: Vinca rosea. Ann N Y Acad Sci. 1958;76:882–94. doi: 10.1111/j.1749-6632.1958.tb54906.x. [DOI] [PubMed] [Google Scholar]
- 2.Schiff PB, Fant J, Horwitz SB. Promotion of microtubule assembly in vitro by taxol. Nature. 1979;277:665–7. doi: 10.1038/277665a0. Initial description that paclitaxel induces tubulin polymerization. [DOI] [PubMed] [Google Scholar]
- 3.Wani MC, Taylor HL, Wall ME, Coggon P, McPhail AT. Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc. 1971;93:2325–7. doi: 10.1021/ja00738a045. [DOI] [PubMed] [Google Scholar]
- 4.Pajk B, et al. Anti-tumor activity of capecitabine and vinorelbine in patients with anthracycline- and taxane-pretreated metastatic breast cancer: findings from the EORTC 10001 randomized phase II trial. Breast. 2008;17:180–5. doi: 10.1016/j.breast.2007.09.002. [DOI] [PubMed] [Google Scholar]
- 5.Norris B, et al. Phase III comparative study of vinorelbine combined with doxorubicin versus doxorubicin alone in disseminated metastatic/recurrent breast cancer: National Cancer Institute of Canada Clinical Trials Group Study MA8. J Clin Oncol. 2000;18:2385–94. doi: 10.1200/JCO.2000.18.12.2385. [DOI] [PubMed] [Google Scholar]
- 6.Dimitroulis J, Stathopoulos GP. Evolution of non-small cell lung cancer chemotherapy (Review) Oncol Rep. 2005;13:923–30. [PubMed] [Google Scholar]
- 7.Gridelli C, et al. Treatment of advanced non-small-cell lung cancer in the elderly: results of an international expert panel. J Clin Oncol. 2005;23:3125–37. doi: 10.1200/JCO.2005.00.224. [DOI] [PubMed] [Google Scholar]
- 8.Markman M. Antineoplastic agents in the management of ovarian cancer: current status and emerging therapeutic strategies. Trends Pharmacol Sci. 2008;29:515–9. doi: 10.1016/j.tips.2008.07.007. [DOI] [PubMed] [Google Scholar]
- 9.Amador ML, Jimeno J, Paz-Ares L, Cortes-Funes H, Hidalgo M. Progress in the development and acquisition of anticancer agents from marine sources. Ann Oncol. 2003;14:1607–15. doi: 10.1093/annonc/mdg443. [DOI] [PubMed] [Google Scholar]
- 10.Nicolaou KC, et al. Total synthesis of taxol. Nature. 1994;367:630–4. doi: 10.1038/367630a0. [DOI] [PubMed] [Google Scholar]
- 11.de Lemos E, et al. Total synthesis of discodermolide: optimization of the effective synthetic route. Chemistry. 2008;14:11092–112. doi: 10.1002/chem.200801478. [DOI] [PubMed] [Google Scholar]
- 12.Busch T, Kirschning A. Recent advances in the total synthesis of pharmaceutically relevant diterpenes. Nat Prod Rep. 2008;25:318–41. doi: 10.1039/b705652b. [DOI] [PubMed] [Google Scholar]
- 13.Wender PA, Hegde SG, Hubbard RD, Zhang L. Total synthesis of (−)-laulimalide. J Am Chem Soc. 2002;124:4956–7. doi: 10.1021/ja0258428. [DOI] [PubMed] [Google Scholar]
- 14.Sammak PJ, Borisy GG. Direct observation of microtubule dynamics in living cells. Nature. 1988;332:724–6. doi: 10.1038/332724a0. [DOI] [PubMed] [Google Scholar]
- 15.Kelling J, Sullivan K, Wilson L, Jordan MA. Suppression of centromere dynamics by Taxol in living osteosarcoma cells. Cancer Res. 2003;63:2794–801. [PubMed] [Google Scholar]
- 16.Okouneva T, Azarenko O, Wilson L, Littlefield BA, Jordan MA. Inhibition of centromere dynamics by eribulin (E7389) during mitotic metaphase. Mol Cancer Ther. 2008;7:2003–11. doi: 10.1158/1535-7163.MCT-08-0095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hamel E, Covell DG. Antimitotic peptides and depsipeptides. Curr Med Chem Anticancer Agents. 2002;2:19–53. doi: 10.2174/1568011023354263. [DOI] [PubMed] [Google Scholar]
- 18.Lacey E, Gill JH. Biochemistry of benzimidazole resistance. Acta Trop. 1994;56:245–62. doi: 10.1016/0001-706x(94)90066-3. [DOI] [PubMed] [Google Scholar]
- 19.Azarenko O, Okouneva T, Singletary KW, Jordan MA, Wilson L. Suppression of microtubule dynamic instability and turnover in MCF7 breast cancer cells by sulforaphane. Carcinogenesis. 2008;29:2360–8. doi: 10.1093/carcin/bgn241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lobert S, Ingram JW, Correia JJ. Additivity of dilantin and vinblastine inhibitory effects on microtubule assembly. Cancer Res. 1999;59:4816–22. [PubMed] [Google Scholar]
- 21.Buey RM, et al. Microtubule interactions with chemically diverse stabilizing agents: thermodynamics of binding to the paclitaxel site predicts cytotoxicity. Chem Biol. 2005;12:1269–79. doi: 10.1016/j.chembiol.2005.09.010. [DOI] [PubMed] [Google Scholar]
- 22.Hamel E, et al. Synergistic effects of peloruside A and laulimalide with taxoid site drugs, but not with each other, on tubulin assembly. Mol Pharmacol. 2006;70:1555–64. doi: 10.1124/mol.106.027847. [DOI] [PubMed] [Google Scholar]
- 23.Huzil JT, et al. A unique mode of microtubule stabilization induced by peloruside A. J Mol Biol. 2008;378:1016–30. doi: 10.1016/j.jmb.2008.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jordan MA, Kamath K. How do microtubule-targeted drugs work? An overview. Curr Cancer Drug Targets. 2007;7:730–42. doi: 10.2174/156800907783220417. [DOI] [PubMed] [Google Scholar]
- 25.Zhou J, Giannakakou P. Targeting microtubules for cancer chemotherapy. Curr Med Chem Anticancer Agents. 2005;5:65–71. doi: 10.2174/1568011053352569. [DOI] [PubMed] [Google Scholar]
- 26.Infante JR, et al. Phase II trial of weekly docetaxel, vinorelbine, and trastuzumab in the first-line treatment of patients with HER2-positive metastatic breast cancer. Clin Breast Cancer. 2009;9:23–8. doi: 10.3816/CBC.2009.n.004. [DOI] [PubMed] [Google Scholar]
- 27.William WN, Jr, et al. Phase II Study of Vinorelbine and Docetaxel in the Treatment of Advanced Non-Small-Cell Lung Cancer as Frontline and Second-Line Therapy. Am J Clin Oncol. 2009 doi: 10.1097/COC.0b013e318199fb99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hudes GR, et al. Phase II study of estramustine and vinblastine, two microtubule inhibitors, in hormone-refractory prostate cancer. J Clin Oncol. 1992;10:1754–61. doi: 10.1200/JCO.1992.10.11.1754. [DOI] [PubMed] [Google Scholar]
- 29.Giannakakou P, Villalba L, Li H, Poruchynsky M, Fojo T. Combinations of paclitaxel and vinblastine and their effects on tubulin polymerization and cellular cytotoxicity: characterization of a synergistic schedule. Int J Cancer. 1998;75:57–63. doi: 10.1002/(sici)1097-0215(19980105)75:1<57::aid-ijc10>3.0.co;2-a. Preclinical study analysing cytotoxicity on cell lines showing that under certain conditions a vinca alkaloid and a taxane can be synergistic. [DOI] [PubMed] [Google Scholar]
- 30.Photiou A, Shah P, Leong LK, Moss J, Retsas S. In vitro synergy of paclitaxel (Taxol) and vinorelbine (navelbine) against human melanoma cell lines. Eur J Cancer. 1997;33:463–70. doi: 10.1016/s0959-8049(97)89023-5. [DOI] [PubMed] [Google Scholar]
- 31.Jordan MA, Toso RJ, Thrower D, Wilson L. Mechanism of mitotic block and inhibition of cell proliferation by taxol at low concentrations. Proc Natl Acad Sci U S A. 1993;90:9552–6. doi: 10.1073/pnas.90.20.9552. Demonstration that taxol modifies microtubule dynamics at concentrations that do not affect microtubule mass and shares a common antiproliferative mechanism with vinblastine. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ng SS, et al. Influence of formulation vehicle on metronomic taxane chemotherapy: albumin-bound versus cremophor EL-based paclitaxel. Clin Cancer Res. 2006;12:4331–8. doi: 10.1158/1078-0432.CCR-05-2762. [DOI] [PubMed] [Google Scholar]
- 33.Tozer GM, Kanthou C, Baguley BC. Disrupting tumour blood vessels. Nat Rev Cancer. 2005;5:423–35. doi: 10.1038/nrc1628. [DOI] [PubMed] [Google Scholar]
- 34.Lippert JW., 3rd Vascular disrupting agents. Bioorg Med Chem. 2007;15:605–15. doi: 10.1016/j.bmc.2006.10.020. [DOI] [PubMed] [Google Scholar]
- 35.Dark GG, et al. Combretastatin A-4, an agent that displays potent and selective toxicity toward tumor vasculature. Cancer Res. 1997;57:1829–34. [PubMed] [Google Scholar]
- 36.Griggs J, Metcalfe JC, Hesketh R. Targeting tumour vasculature: the development of combretastatin A4. Lancet Oncol. 2001;2:82–7. doi: 10.1016/S1470-2045(00)00224-2. [DOI] [PubMed] [Google Scholar]
- 37.Kanthou C, Tozer GM. The tumor vascular targeting agent combretastatin A-4-phosphate induces reorganization of the actin cytoskeleton and early membrane blebbing in human endothelial cells. Blood. 2002;99:2060–9. doi: 10.1182/blood.v99.6.2060. [DOI] [PubMed] [Google Scholar]
- 38.Tozer GM, et al. Mechanisms associated with tumor vascular shut-down induced by combretastatin A-4 phosphate: intravital microscopy and measurement of vascular permeability. Cancer Res. 2001;61:6413–22. These authors describe the rapid and reversible in vivo effect of the vascular disrupting agent combretastatin in a tumor implanted in a rat model. [PubMed] [Google Scholar]
- 39.Hori K, Saito S, Kubota K. A novel combretastatin A-4 derivative, AC7700, strongly stanches tumour blood flow and inhibits growth of tumours developing in various tissues and organs. Br J Cancer. 2002;86:1604–14. doi: 10.1038/sj.bjc.6600296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Anderson HL, et al. Assessment of pharmacodynamic vascular response in a phase I trial of combretastatin A4 phosphate. J Clin Oncol. 2003;21:2823–30. doi: 10.1200/JCO.2003.05.186. [DOI] [PubMed] [Google Scholar]
- 41.Beauregard DA, et al. Magnetic resonance imaging and spectroscopy of combretastatin A4 prodrug-induced disruption of tumour perfusion and energetic status. Br J Cancer. 1998;77:1761–7. doi: 10.1038/bjc.1998.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Galbraith SM, et al. Combretastatin A4 phosphate has tumor antivascular activity in rat and man as demonstrated by dynamic magnetic resonance imaging. J Clin Oncol. 2003;21:2831–42. doi: 10.1200/JCO.2003.05.187. [DOI] [PubMed] [Google Scholar]
- 43.Fojo AT, Menefee M. Microtubule targeting agents: basic mechanisms of multidrug resistance (MDR) Semin Oncol. 2005;32:S3–8. doi: 10.1053/j.seminoncol.2005.09.010. [DOI] [PubMed] [Google Scholar]
- 44.Breuninger LM, et al. Expression of multidrug resistance-associated protein in NIH/3T3 cells confers multidrug resistance associated with increased drug efflux and altered intracellular drug distribution. Cancer Res. 1995;55:5342–7. [PubMed] [Google Scholar]
- 45.Huisman MT, Chhatta AA, van Tellingen O, Beijnen JH, Schinkel AH. MRP2 (ABCC2) transports taxanes and confers paclitaxel resistance and both processes are stimulated by probenecid. Int J Cancer. 2005;116:824–9. doi: 10.1002/ijc.21013. [DOI] [PubMed] [Google Scholar]
- 46.Hopper-Borge E, Chen ZS, Shchaveleva I, Belinsky MG, Kruh GD. Analysis of the drug resistance profile of multidrug resistance protein 7 (ABCC10): resistance to docetaxel. Cancer Res. 2004;64:4927–30. doi: 10.1158/0008-5472.CAN-03-3111. [DOI] [PubMed] [Google Scholar]
- 47.Kuttesch JF, et al. P-glycoprotein expression at diagnosis may not be a primary mechanism of therapeutic failure in childhood rhabdomyosarcoma. J Clin Oncol. 1996;14:886–900. doi: 10.1200/JCO.1996.14.3.886. [DOI] [PubMed] [Google Scholar]
- 48.Beck WT, et al. Methods to detect P-glycoprotein-associated multidrug resistance in patients’ tumors: consensus recommendations. Cancer Res. 1996;56:3010–20. [PubMed] [Google Scholar]
- 49.Meisel C, Roots I, Cascorbi I, Brinkmann U, Brockmoller J. How to manage individualized drug therapy: application of pharmacogenetic knowledge of drug metabolism and transport. Clin Chem Lab Med. 2000;38:869–76. doi: 10.1515/CCLM.2000.126. [DOI] [PubMed] [Google Scholar]
- 50.Lhomme C, et al. Phase III study of valspodar (PSC 833) combined with paclitaxel and carboplatin compared with paclitaxel and carboplatin alone in patients with stage IV or suboptimally debulked stage III epithelial ovarian cancer or primary peritoneal cancer. J Clin Oncol. 2008;26:2674–82. doi: 10.1200/JCO.2007.14.9807. [DOI] [PubMed] [Google Scholar]
- 51.Fromm MF. P-glycoprotein: a defense mechanism limiting oral bioavailability and CNS accumulation of drugs. Int J Clin Pharmacol Ther. 2000;38:69–74. doi: 10.5414/cpp38069. [DOI] [PubMed] [Google Scholar]
- 52.Chaudhuri AR, et al. The tumor suppressor protein Fhit. A novel interaction with tubulin. J Biol Chem. 1999;274:24378–82. doi: 10.1074/jbc.274.34.24378. [DOI] [PubMed] [Google Scholar]
- 53.Cheung CH, et al. Survivin counteracts the therapeutic effect of microtubule de-stabilizers by stabilizing tubulin polymers. Mol Cancer. 2009;8:43. doi: 10.1186/1476-4598-8-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Don S, et al. Neuronal-associated microtubule proteins class III beta-tubulin and MAP2c in neuroblastoma: role in resistance to microtubule-targeted drugs. Mol Cancer Ther. 2004;3:1137–46. [PubMed] [Google Scholar]
- 55.Tian G, et al. Pathway leading to correctly folded beta-tubulin. Cell. 1996;86:287–96. doi: 10.1016/s0092-8674(00)80100-2. These authors report the nature and interactions between the tubulin binding cofactors, key proteins involved in the proper folding of α and b tubulin peptides. [DOI] [PubMed] [Google Scholar]
- 56.Alli E, Bash-Babula J, Yang JM, Hait WN. Effect of stathmin on the sensitivity to antimicrotubule drugs in human breast cancer. Cancer Res. 2002;62:6864–9. [PubMed] [Google Scholar]
- 57.Galmarini CM, et al. Drug resistance associated with loss of p53 involves extensive alterations in microtubule composition and dynamics. Br J Cancer. 2003;88:1793–9. doi: 10.1038/sj.bjc.6600960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Seve P, Dumontet C. Is class III beta-tubulin a predictive factor in patients receiving tubulin-binding agents? Lancet Oncol. 2008;9:168–75. doi: 10.1016/S1470-2045(08)70029-9. [DOI] [PubMed] [Google Scholar]
- 59.Dumontet C, Jordan MA, Lee FF. Ixabepilone: targeting betaIII-tubulin expression in taxane-resistant malignancies. Mol Cancer Ther. 2009;8:17–25. doi: 10.1158/1535-7163.MCT-08-0986. [DOI] [PubMed] [Google Scholar]
- 60.Bhattacharya R, Cabral F. Molecular basis for class V beta-tubulin effects on microtubule assembly and paclitaxel resistance. J Biol Chem. 2009;284:13023–32. doi: 10.1074/jbc.M900167200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Haber M, et al. Altered expression of M beta 2, the class II beta-tubulin isotype, in a murine J774.2 cell line with a high level of taxol resistance. J Biol Chem. 1995;270:31269–75. doi: 10.1074/jbc.270.52.31269. [DOI] [PubMed] [Google Scholar]
- 62.Kavallaris M, Burkhart CA, Horwitz SB. Antisense oligonucleotides to class III beta-tubulin sensitize drug-resistant cells to Taxol. Br J Cancer. 1999;80:1020–5. doi: 10.1038/sj.bjc.6690507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kavallaris M, et al. Multiple microtubule alterations are associated with Vinca alkaloid resistance in human leukemia cells. Cancer Res. 2001;61:5803–9. [PubMed] [Google Scholar]
- 64.Gan PP, Pasquier E, Kavallaris M. Class III beta-tubulin mediates sensitivity to chemotherapeutic drugs in non small cell lung cancer. Cancer Res. 2007;67:9356–63. doi: 10.1158/0008-5472.CAN-07-0509. [DOI] [PubMed] [Google Scholar]
- 65.McCarroll JA, Gan PP, Liu M, Kavallaris M. Beta III-Tubulin Is a Multifunctional Protein Involved in Drug Sensitivity and Tumorigenesis in Non-Small Cell Lung Cancer. Cancer Res. 2010;70:4995–5003. doi: 10.1158/0008-5472.CAN-09-4487. Study showing that tubulin III peptide is involved not only in resistance to therapy, an observation confirmed in clinical trials by several authors, but in tumorigenesis as well. [DOI] [PubMed] [Google Scholar]
- 66.Ferrandina G, et al. Class III beta-tubulin overexpression is a marker of poor clinical outcome in advanced ovarian cancer patients. Clin Cancer Res. 2006;12:2774–9. doi: 10.1158/1078-0432.CCR-05-2715. [DOI] [PubMed] [Google Scholar]
- 67.Giannakakou P, et al. Paclitaxel-resistant human ovarian cancer cells have mutant beta-tubulins that exhibit impaired paclitaxel-driven polymerization. J Biol Chem. 1997;272:17118–25. doi: 10.1074/jbc.272.27.17118. [DOI] [PubMed] [Google Scholar]
- 68.Gokmen-Polar Y, et al. beta-Tubulin mutations are associated with resistance to 2-methoxyestradiol in MDA-MB-435 cancer cells. Cancer Res. 2005;65:9406–14. doi: 10.1158/0008-5472.CAN-05-0088. [DOI] [PubMed] [Google Scholar]
- 69.Hari M, et al. Paclitaxel-resistant cells have a mutation in the paclitaxel-binding region of beta-tubulin (Asp26Glu) and less stable microtubules. Mol Cancer Ther. 2006;5:270–8. doi: 10.1158/1535-7163.MCT-05-0190. [DOI] [PubMed] [Google Scholar]
- 70.Monzo M, et al. Paclitaxel resistance in non-small-cell lung cancer associated with beta-tubulin gene mutations. J Clin Oncol. 1999;17:1786–93. doi: 10.1200/JCO.1999.17.6.1786. This study linking mutations of tubulin genes to taxane resistance was later found to be mistaken due to sequencing of tubulin pseudogenes. [DOI] [PubMed] [Google Scholar]
- 71.Sale S, et al. Conservation of the class I beta-tubulin gene in human populations and lack of mutations in lung cancers and paclitaxel-resistant ovarian cancers. Mol Cancer Ther. 2002;1:215–25. [PubMed] [Google Scholar]
- 72.Sale S, Oefner PJ, Sikic BI. Genetic analysis of the beta-tubulin gene, TUBB, in non-small-cell lung cancer. J Natl Cancer Inst. 2002;94:776–7. doi: 10.1093/jnci/94.10.776. [DOI] [PubMed] [Google Scholar]
- 73.Wahl AF, et al. Loss of normal p53 function confers sensitization to Taxol by increasing G2/M arrest and apoptosis. Nat Med. 1996;2:72–9. doi: 10.1038/nm0196-72. These data suggested that paclitaxel could be more active in cells which had lost normal P53 function, a common occurrence in tumor cells. [DOI] [PubMed] [Google Scholar]
- 74.Fan S, Cherney B, Reinhold W, Rucker K, O’Connor PM. Disruption of p53 function in immortalized human cells does not affect survival or apoptosis after taxol or vincristine treatment. Clin Cancer Res. 1998;4:1047–54. [PubMed] [Google Scholar]
- 75.Debernardis D, et al. p53 status does not affect sensitivity of human ovarian cancer cell lines to paclitaxel. Cancer Res. 1997;57:870–4. [PubMed] [Google Scholar]
- 76.King TC, et al. p53 mutations do not predict response to paclitaxel in metastatic nonsmall cell lung carcinoma. Cancer. 2000;89:769–73. doi: 10.1002/1097-0142(20000815)89:4<769::aid-cncr8>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 77.Malamou-Mitsi V, et al. Evaluation of the prognostic and predictive value of p53 and Bcl-2 in breast cancer patients participating in a randomized study with dose-dense sequential adjuvant chemotherapy. Ann Oncol. 2006;17:1504–11. doi: 10.1093/annonc/mdl147. [DOI] [PubMed] [Google Scholar]
- 78.Shoemaker AR, et al. A small-molecule inhibitor of Bcl-XL potentiates the activity of cytotoxic drugs in vitro and in vivo. Cancer Res. 2006;66:8731–9. doi: 10.1158/0008-5472.CAN-06-0367. [DOI] [PubMed] [Google Scholar]
- 79.Bublik DR, Scolz M, Triolo G, Monte M, Schneider C. Human GTSE-1 regulates p21(CIP1/WAF1) stability conferring resistance to paclitaxel treatment. J Biol Chem. 2010;285:5274–81. doi: 10.1074/jbc.M109.045948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Patel N, et al. Rescue of paclitaxel sensitivity by repression of Prohibitin1 in drugresistant cancer cells. Proc Natl Acad Sci U S A. 107:2503–8. doi: 10.1073/pnas.0910649107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Huang L, et al. Hypoxia induced paclitaxel resistance in human ovarian cancers via hypoxia-inducible factor 1alpha. J Cancer Res Clin Oncol. 136:447–56. doi: 10.1007/s00432-009-0675-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bauer JA, et al. Identification of markers of taxane sensitivity using proteomic and genomic analyses of breast tumors from patients receiving neoadjuvant paclitaxel and radiation. Clin Cancer Res. 2010;16:681–90. doi: 10.1158/1078-0432.CCR-09-1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Townsend DM, Tew KD. The role of glutathione-S-transferase in anti-cancer drug resistance. Oncogene. 2003;22:7369–75. doi: 10.1038/sj.onc.1206940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zhou M, et al. MicroRNA-125b confers the resistance of breast cancer cells to paclitaxel through suppression of pro-apoptotic Bcl-2 antagonist killer 1 (Bak1) J Biol Chem. 2010;28:21496–507. doi: 10.1074/jbc.M109.083337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Fujita Y, et al. MiR-148a attenuates paclitaxel resistance of hormone-refractory, drug-resistant prostate cancer PC3 cells by regulating MSK1 expression. J Biol Chem. 2010;285:19076–84. doi: 10.1074/jbc.M109.079525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Bellmunt J, et al. Phase III trial of vinflunine plus best supportive care compared with best supportive care alone after a platinum-containing regimen in patients with advanced transitional cell carcinoma of the urothelial tract. J Clin Oncol. 2009;27:4454–61. doi: 10.1200/JCO.2008.20.5534. [DOI] [PubMed] [Google Scholar]
- 87.Cormier A, Marchand M, Ravelli RB, Knossow M, Gigant B. Structural insight into the inhibition of tubulin by vinca domain peptide ligands. EMBO Rep. 2008;9:1101–6. doi: 10.1038/embor.2008.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Horti J, et al. Phase I study of TZT-1027, a novel synthetic dolastatin 10 derivative, for the treatment of patients with non-small cell lung cancer. Cancer Chemother Pharmacol. 2008;62:173–80. doi: 10.1007/s00280-007-0665-7. [DOI] [PubMed] [Google Scholar]
- 89.Tamura K, et al. Phase I study of TZT-1027, a novel synthetic dolastatin 10 derivative and inhibitor of tubulin polymerization, which was administered to patients with advanced solid tumors on days 1 and 8 in 3-week courses. Cancer Chemother Pharmacol. 2007;60:285–93. doi: 10.1007/s00280-006-0382-7. [DOI] [PubMed] [Google Scholar]
- 90.Piekarz RL, et al. Phase II multi-institutional trial of the histone deacetylase inhibitor romidepsin as monotherapy for patients with cutaneous T-cell lymphoma. J Clin Oncol. 2009;27:5410–7. doi: 10.1200/JCO.2008.21.6150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Twelves C, et al. A phase III study (EMBRACE) of eribulin mesylate versus treatment of physician’s choice in patients with locally recurrent or metastatic breast cancer previously treated with an anthracycline and a taxane. J Clin Oncol. 2010;28:7S. Abst CRA1004. [Google Scholar]
- 92.Besse-Hammer T, et al. A dose-escalating study of XRP6258 in combination with capecitabine, in patients (pts) with metastatic breast cancer (MBC) progressing after anthracycline and taxane therapy: Preliminary results. - ASCO. J Clin Oncol. 2009;27 abstr 1053. [Google Scholar]
- 93.Sampath D, et al. MAC-321, a novel taxane with greater efficacy than paclitaxel and docetaxel in vitro and in vivo. Mol Cancer Ther. 2003;2:873–84. [PubMed] [Google Scholar]
- 94.Terwogt JM, Nuijen B, Huinink WW, Beijnen JH. Alternative formulations of paclitaxel. Cancer Treat Rev. 1997;23:87–95. doi: 10.1016/s0305-7372(97)90022-0. [DOI] [PubMed] [Google Scholar]
- 95.Gelderblom H, Verweij J, Nooter K, Sparreboom A. Cremophor EL: the drawbacks and advantages of vehicle selection for drug formulation. Eur J Cancer. 2001;37:1590–8. doi: 10.1016/s0959-8049(01)00171-x. [DOI] [PubMed] [Google Scholar]
- 96.Gradishar WJ, et al. Significantly longer progression-free survival with nabpaclitaxel compared with docetaxel as first-line therapy for metastatic breast cancer. J Clin Oncol. 2009;27:3611–9. doi: 10.1200/JCO.2008.18.5397. [DOI] [PubMed] [Google Scholar]
- 97.Shepard DR, et al. Phase II trial of neoadjuvant nab-paclitaxel in high risk patients with prostate cancer undergoing radical prostatectomy. J Urol. 2009;181:1672–7. doi: 10.1016/j.juro.2008.11.121. [DOI] [PubMed] [Google Scholar]
- 98.Stinchcombe TE, et al. Phase I and pharmacokinetic trial of carboplatin and albumin-bound paclitaxel, ABI-007 (Abraxane) on three treatment schedules in patients with solid tumors. Cancer Chemother Pharmacol. 2007;60:759–66. doi: 10.1007/s00280-007-0423-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Teneriello MG, et al. Phase II evaluation of nanoparticle albumin-bound paclitaxel in platinum-sensitive patients with recurrent ovarian, peritoneal, or fallopian tube cancer. J Clin Oncol. 2009;27:1426–31. doi: 10.1200/JCO.2008.18.9548. [DOI] [PubMed] [Google Scholar]
- 100.Kim TY, et al. Phase I and pharmacokinetic study of Genexol-PM, a cremophor-free, polymeric micelle-formulated paclitaxel, in patients with advanced malignancies. Clin Cancer Res. 2004;10:3708–16. doi: 10.1158/1078-0432.CCR-03-0655. [DOI] [PubMed] [Google Scholar]
- 101.Chou TC, et al. Desoxyepothilone B is curative against human tumor xenografts that are refractory to paclitaxel. Proc Natl Acad Sci U S A. 1998;95:15798–802. doi: 10.1073/pnas.95.26.15798. Demonstration of the preclinical activity of epothilones in paclitaxel-resistant models. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.De Geest K, et al. Phase II Clinical Trial of Ixabepilone in Patients With Recurrent or Persistent Platinum- and Taxane-Resistant Ovarian or Primary Peritoneal Cancer: A Gynecologic Oncology Group Study. J Clin Oncol. 2010;28:149–53. doi: 10.1200/JCO.2009.24.1455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Lee JJ, Swain SM. Development of novel chemotherapeutic agents to evade the mechanisms of multidrug resistance (MDR) Semin Oncol. 2005;32:S22–6. doi: 10.1053/j.seminoncol.2005.09.013. [DOI] [PubMed] [Google Scholar]
- 104.Hoffmann J, et al. Sagopilone crosses the blood-brain barrier in vivo to inhibit brain tumor growth and metastases. Neuro Oncol. 2009;11:158–66. doi: 10.1215/15228517-2008-072). Preclinical study showing the diffusion of sagopilone through the blood-brain barrier, raising the possibility that this agent may be active for the treatment of brain metastases. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lee D. Activity of epothilone B analogues ixabepilone and patupilone in hormone-refractory prostate cancer. Clin Prostate Cancer. 2004;3:80–2. doi: 10.1016/s1540-0352(11)70066-x. [DOI] [PubMed] [Google Scholar]
- 106.Arnold D, et al. Weekly administration of sagopilone (ZK-EPO), a fully synthetic epothilone, in patients with refractory solid tumours: results of a phase I trial. Br J Cancer. 2009;101:1241–7. doi: 10.1038/sj.bjc.6605327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Galmarini CM. Sagopilone, a microtubule stabilizer for the potential treatment of cancer. Curr Opin Investig Drugs. 2009;10:1359–71. [PubMed] [Google Scholar]
- 108.Silvani A, et al. Systemic sagopilone (ZK-EPO) treatment of patients with recurrent malignant gliomas. J Neurooncol. 2009;95:61–4. doi: 10.1007/s11060-009-9890-8. [DOI] [PubMed] [Google Scholar]
- 109.Beer TM, et al. Phase II study of KOS-862 in patients with metastatic androgen independent prostate cancer previously treated with docetaxel. Invest New Drugs. 2007;25:565–70. doi: 10.1007/s10637-007-9068-1. [DOI] [PubMed] [Google Scholar]
- 110.Kolman A. Epothilone D (Kosan/Roche) Curr Opin Investig Drugs. 2004;5:657–67. [PubMed] [Google Scholar]
- 111.Rustin GJ, et al. Phase I clinical trial of weekly combretastatin A4 phosphate: clinical and pharmacokinetic results. J Clin Oncol. 2003;21:2815–22. doi: 10.1200/JCO.2003.05.185. [DOI] [PubMed] [Google Scholar]
- 112.Stevenson JP, et al. Phase I trial of the antivascular agent combretastatin A4 phosphate on a 5-day schedule to patients with cancer: magnetic resonance imaging evidence for altered tumor blood flow. J Clin Oncol. 2003;21:4428–38. doi: 10.1200/JCO.2003.12.986. In vivo imaging of reduced vascularization in patients receiving a vasculardisrupting agent. [DOI] [PubMed] [Google Scholar]
- 113.Cooney MM, et al. Cardiovascular safety profile of combretastatin a4 phosphate in a single-dose phase I study in patients with advanced cancer. Clin Cancer Res. 2004;10:96–100. doi: 10.1158/1078-0432.ccr-0364-3. [DOI] [PubMed] [Google Scholar]
- 114.Mooney CJ, et al. A phase II trial of fosbretabulin in advanced anaplastic thyroid carcinoma and correlation of baseline serum-soluble intracellular adhesion molecule-1 with outcome. Thyroid. 2009;19:233–40. doi: 10.1089/thy.2008.0321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.LoRusso PM, et al. Phase I clinical evaluation of ZD6126, a novel vascular-targeting agent, in patients with solid tumors. Invest New Drugs. 2008;26:159–67. doi: 10.1007/s10637-008-9112-9. [DOI] [PubMed] [Google Scholar]
- 116.Hua J, et al. Oxi4503, a novel vascular targeting agent: effects on blood flow and antitumor activity in comparison to combretastatin A-4 phosphate. Anticancer Res. 2003;23:1433–40. [PubMed] [Google Scholar]
- 117.Delmonte A, Sessa C. AVE8062: a new combretastatin derivative vascular disrupting agent. Expert Opin Investig Drugs. 2009;18:1541–8. doi: 10.1517/13543780903213697. [DOI] [PubMed] [Google Scholar]
- 118.Anthony SP, et al. Initial results of a first-in-man phase I study of EPC2407, a novel small molecule microtubule inhibitor anticancer agent with tumor vascular endothelial disrupting activity. J Clin Oncol. 2008;26S Abstract 2531. [Google Scholar]
- 119.Shnyder SD, Cooper PA, Millington NJ, Pettit GR, Bibby MC. Auristatin PYE, a novel synthetic derivative of dolastatin 10, is highly effective in human colon tumour models. Int J Oncol. 2007;31:353–60. [PubMed] [Google Scholar]
- 120.Michels J, et al. A phase IB study of ABT-751 in combination with docetaxel in patients with advanced castration-resistant prostate cancer. Ann Oncol. 2010;21:305–11. doi: 10.1093/annonc/mdp311. [DOI] [PubMed] [Google Scholar]
- 121.Sweeney C, et al. A phase II multicenter, randomized, double-blind, safety trial assessing the pharmacokinetics, pharmacodynamics, and efficacy of oral 2-methoxyestradiol capsules in hormone-refractory prostate cancer. Clin Cancer Res. 2005;11:6625–33. doi: 10.1158/1078-0432.CCR-05-0440. [DOI] [PubMed] [Google Scholar]
- 122.James J, et al. Phase I safety, pharmacokinetic and pharmacodynamic studies of 2-methoxyestradiol alone or in combination with docetaxel in patients with locally recurrent or metastatic breast cancer. Invest New Drugs. 2007;25:41–8. doi: 10.1007/s10637-006-9008-5. [DOI] [PubMed] [Google Scholar]
- 123.Rajkumar SV, et al. Novel therapy with 2-methoxyestradiol for the treatment of relapsed and plateau phase multiple myeloma. Clin Cancer Res. 2007;13:6162–7. doi: 10.1158/1078-0432.CCR-07-0807. [DOI] [PubMed] [Google Scholar]
- 124.Tevaarwerk AJ, et al. Phase I trial of 2-methoxyestradiol NanoCrystal dispersion in advanced solid malignancies. Clin Cancer Res. 2009;15:1460–5. doi: 10.1158/1078-0432.CCR-08-1599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Escuin D, et al. The hematopoietic-specific beta1-tubulin is naturally resistant to 2-methoxyestradiol and protects patients from drug-induced myelosuppression. Cell Cycle. 2009;8:3914–24. doi: 10.4161/cc.8.23.10105. [DOI] [PubMed] [Google Scholar]
- 126.Ayral-Kaloustian S, Zhang N, Beyer C. Cevipabulin (TTI-237): Preclinical and clinical results for a novel antimicrotubule agent. Methods Find Exp Clin Pharmacol. 2009;31:443–7. doi: 10.1358/mf.2009.31.7.1410793. [DOI] [PubMed] [Google Scholar]
- 127.Landen JW, et al. Noscapine crosses the blood-brain barrier and inhibits glioblastoma growth. Clin Cancer Res. 2004;10:5187–201. doi: 10.1158/1078-0432.CCR-04-0360. [DOI] [PubMed] [Google Scholar]
- 128.Honore S, et al. Synergistic suppression of microtubule dynamics by discodermolide and paclitaxel in non-small cell lung carcinoma cells. Cancer Res. 2004;64:4957–64. doi: 10.1158/0008-5472.CAN-04-0693. [DOI] [PubMed] [Google Scholar]
- 129.Martello LA, et al. Taxol and discodermolide represent a synergistic drug combination in human carcinoma cell lines. Clin Cancer Res. 2000;6:1978–87. [PubMed] [Google Scholar]
- 130.Paterson I, Gardner NM, Guzman E, Wright AE. Total synthesis and biological evaluation of potent analogues of dictyostatin: modification of the C2–C6 dienoate region. Bioorg Med Chem Lett. 2008;18:6268–72. doi: 10.1016/j.bmcl.2008.09.109. [DOI] [PubMed] [Google Scholar]
- 131.D’Agostino G, et al. A multicenter phase II study of the cryptophycin analog LY355703 in patients with platinum-resistant ovarian cancer. Int J Gynecol Cancer. 2006;16:71–6. doi: 10.1111/j.1525-1438.2006.00276.x. [DOI] [PubMed] [Google Scholar]
- 132.Edelman MJ, et al. Phase 2 study of cryptophycin 52 (LY355703) in patients previously treated with platinum based chemotherapy for advanced non-small cell lung cancer. Lung Cancer. 2003;39:197–9. doi: 10.1016/s0169-5002(02)00511-1. [DOI] [PubMed] [Google Scholar]
- 133.Canta A, Chiorazzi A, Cavaletti G. Tubulin: a target for antineoplastic drugs into the cancer cells but also in the peripheral nervous system. Curr Med Chem. 2009;16:1315–24. doi: 10.2174/092986709787846488. [DOI] [PubMed] [Google Scholar]
- 134.Argyriou AA, Koltzenburg M, Polychronopoulos P, Papapetropoulos S, Kalofonos HP. Peripheral nerve damage associated with administration of taxanes in patients with cancer. Crit Rev Oncol Hematol. 2008;66:218–28. doi: 10.1016/j.critrevonc.2008.01.008. [DOI] [PubMed] [Google Scholar]
- 135.Kuroi K, Shimozuma K. Neurotoxicity of taxanes: symptoms and quality of life assessment. Breast Cancer. 2004;11:92–9. doi: 10.1007/BF02968010. [DOI] [PubMed] [Google Scholar]
- 136.Lee JJ, et al. Changes in neurologic function tests may predict neurotoxicity caused by ixabepilone. J Clin Oncol. 2006;24:2084–91. doi: 10.1200/JCO.2005.04.2820. [DOI] [PubMed] [Google Scholar]
- 137.Lyubimova NV, Toms MG, Shakirova IN, Gurina OI, Kushlinskii NE. Biochemical parameters in the diagnosis and monitoring of neurotoxicity of antitumor cytostatics. Bull Exp Biol Med. 2001;132:1093–5. doi: 10.1023/a:1017928927399. [DOI] [PubMed] [Google Scholar]
- 138.Bokemeyer C, Berger CC, Kuczyk MA, Schmoll HJ. Evaluation of long-term toxicity after chemotherapy for testicular cancer. J Clin Oncol. 1996;14:2923–32. doi: 10.1200/JCO.1996.14.11.2923. In this group of young patients treated with cisplatinum, bleomycin and etoposide, 21% developed ototoxicity and 17% peripheral neuropathy. [DOI] [PubMed] [Google Scholar]
- 139.Schiff D, Wen PY, van den Bent MJ. Neurological adverse effects caused by cytotoxic and targeted therapies. Nat Rev Clin Oncol. 2009;6:596–603. doi: 10.1038/nrclinonc.2009.128. [DOI] [PubMed] [Google Scholar]
- 140.Poruchynsky MS, et al. Proteasome inhibitors increase tubulin polymerization and stabilization in tissue culture cells: a possible mechanism contributing to peripheral neuropathy and cellular toxicity following proteasome inhibition. Cell Cycle. 2008;7:940–9. doi: 10.4161/cc.7.7.5625. These authors present provocative data suggesting that proteasome inhibitors could alter tubulin polymerization, thereby explaining at least in part the neurotoxicity observed with these agents. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Anderl JL, Redpath S, Ball AJ. A neuronal and astrocyte co-culture assay for high content analysis of neurotoxicity. J Vis Exp. 2009;27 doi: 10.3791/1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Authier N, et al. Animal models of chemotherapy-evoked painful peripheral neuropathies. Neurotherapeutics. 2009;6:620–9. doi: 10.1016/j.nurt.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Davis RE, Schlumpf BE, Klinger PD. Comparative neurotoxicity of tubulin-binding drugs: inhibition of goldfish optic nerve regeneration. Toxicol Appl Pharmacol. 1985;80:308–15. doi: 10.1016/0041-008x(85)90088-2. [DOI] [PubMed] [Google Scholar]
- 144.Fan CY, Cowden J, Simmons SO, Padilla S, Ramabhadran R. Gene expression changes in developing zebrafish as potential markers for rapid developmental neurotoxicity screening. Neurotoxicol Teratol. 2009 doi: 10.1016/j.ntt.2009.04.065. [DOI] [PubMed] [Google Scholar]
- 145.Kiburg B, Moorer-van Delft C, Heimans JJ, Huijgens PC, Boer HH. In vivo modulation of vincristine-induced neurotoxicity in Lymnaea stagnalis, by the ACTH(4–9) analogue Org 2766. J Neurooncol. 1996;30:173–80. doi: 10.1007/BF00177268. [DOI] [PubMed] [Google Scholar]
- 146.Wozniak KM, et al. AACR. 2010 Abs 4438. [Google Scholar]
- 147.Vahdat LT, et al. Phase II study of eribulin mesylate, a halichondrin B analog, in patients with metastatic breast cancer previously treated with an anthracycline and a taxane. J Clin Oncol. 2009;27:2954–61. doi: 10.1200/JCO.2008.17.7618. [DOI] [PubMed] [Google Scholar]
- 148.Wienecke A, Bacher G. Indibulin, a novel microtubule inhibitor, discriminates between mature neuronal and nonneuronal tubulin. Cancer Res. 2009;69:171–7. doi: 10.1158/0008-5472.CAN-08-1342. [DOI] [PubMed] [Google Scholar]
- 149.Oostendorp RL, et al. Dose-finding and pharmacokinetic study of orally administered indibulin (D-24851) to patients with advanced solid tumors. Invest New Drugs. 2010;28:163–70. doi: 10.1007/s10637-009-9244-6. [DOI] [PubMed] [Google Scholar]
- 150.Blagden SP, et al. A phase I trial of ispinesib, a kinesin spindle protein inhibitor, with docetaxel in patients with advanced solid tumours. Br J Cancer. 2008;98:894–9. doi: 10.1038/sj.bjc.6604264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Tang PA, et al. Phase II study of ispinesib in recurrent or metastatic squamous cell carcinoma of the head and neck. Invest New Drugs. 2008;26:257–64. doi: 10.1007/s10637-007-9098-8. [DOI] [PubMed] [Google Scholar]
- 152.Knox JJ, et al. A phase II and pharmacokinetic study of SB-715992, in patients with metastatic hepatocellular carcinoma: a study of the National Cancer Institute of Canada Clinical Trials Group (NCIC CTG IND.168) Invest New Drugs. 2008;26:265–72. doi: 10.1007/s10637-007-9103-2. [DOI] [PubMed] [Google Scholar]
- 153.Vats T, et al. A study of toxicity and comparative therapeutic efficacy of vindesine-prednisone vs. vincristine-prednisone in children with acute lymphoblastic leukemia in relapse. A Pediatric Oncology Group study. Invest New Drugs. 1992;10:231–4. doi: 10.1007/BF00877252. [DOI] [PubMed] [Google Scholar]
- 154.Lysitsas DN, et al. Antirestenotic effects of a novel polymer-coated d-24851 eluting stent. Experimental data in a rabbit iliac artery model. Cardiovasc Intervent Radiol. 2007;30:1192–200. doi: 10.1007/s00270-007-9027-4. [DOI] [PubMed] [Google Scholar]
- 155.Aghajanian C, et al. Phase I study of the novel epothilone analog ixabepilone (BMS-247550) in patients with advanced solid tumors and lymphomas. J Clin Oncol. 2007;25:1082–8. doi: 10.1200/JCO.2006.08.7304. [DOI] [PubMed] [Google Scholar]
- 156.Bissett D, et al. Phase I and pharmacokinetic study of rhizoxin. Cancer Res. 1992;52:2894–8. [PubMed] [Google Scholar]
- 157.Cunningham C, et al. Phase I and pharmacokinetic study of the dolastatin-15 analogue tasidotin (ILX651) administered intravenously on days 1, 3, and 5 every 3 weeks in patients with advanced solid tumors. Clin Cancer Res. 2005;11:7825–33. doi: 10.1158/1078-0432.CCR-05-0058. [DOI] [PubMed] [Google Scholar]
- 158.Zatloukal P, et al. Randomized multicenter phase II study of larotaxel (XRP9881) in combination with cisplatin or gemcitabine as first-line chemotherapy in non-irradiable stage IIIB or stage IV non-small cell lung cancer. J Thorac Oncol. 2008;3:894–901. doi: 10.1097/JTO.0b013e31817e6669. [DOI] [PubMed] [Google Scholar]
- 159.Larkin JM, Kaye SB. Epothilones in the treatment of cancer. Expert Opin Investig Drugs. 2006;15:691–702. doi: 10.1517/13543784.15.6.691. [DOI] [PubMed] [Google Scholar]
- 160.Greystoke A, et al. A phase I study of intravenous TZT-1027 administered on day 1 and day 8 of a three-weekly cycle in combination with carboplatin given on day 1 alone in patients with advanced solid tumours. Ann Oncol. 2006;17:1313–9. doi: 10.1093/annonc/mdl097. [DOI] [PubMed] [Google Scholar]
- 161.Markman M. Managing taxane toxicities. Support Care Cancer. 2003;11:144–7. doi: 10.1007/s00520-002-0405-9. [DOI] [PubMed] [Google Scholar]
- 162.Swanton C, et al. Chromosomal instability determines taxane response. Proc Natl Acad Sci U S A. 2009;106:8671–6. doi: 10.1073/pnas.0811835106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Bouchet BP, et al. Paclitaxel resistance in untransformed human mammary epithelial cells is associated with an aneuploidy-prone phenotype. Br J Cancer. 2007;97:1218–24. doi: 10.1038/sj.bjc.6603936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Rao VK, et al. The extent of chromosomal aberrations induced by chemotherapy in non-human primates depends on the schedule of administration. Mutat Res. 2005;583:105–19. doi: 10.1016/j.mrgentox.2005.01.013. Demonstration in a primate model that paclitaxel can cause chromosomal aberrations after bolus injections. [DOI] [PubMed] [Google Scholar]
- 165.Hamel E, Sackett DL, Vourloumis D, Nicolaou KC. The coral-derived natural products eleutherobin and sarcodictyins A and B: effects on the assembly of purified tubulin with and without microtubule-associated proteins and binding at the polymer taxoid site. Biochemistry. 1999;38:5490–8. doi: 10.1021/bi983023n. [DOI] [PubMed] [Google Scholar]
- 166.Mooberry SL, Tien G, Hernandez AH, Plubrukarn A, Davidson BS. Laulimalide and isolaulimalide, new paclitaxel-like microtubule-stabilizing agents. Cancer Res. 1999;59:653–60. [PubMed] [Google Scholar]
- 167.Gapud EJ, Bai R, Ghosh AK, Hamel E. Laulimalide and paclitaxel: a comparison of their effects on tubulin assembly and their synergistic action when present simultaneously. Mol Pharmacol. 2004;66:113–21. doi: 10.1124/mol.66.1.113. [DOI] [PubMed] [Google Scholar]
- 168.Loganzo F, et al. Cells resistant to HTI-286 do not overexpress P-glycoprotein but have reduced drug accumulation and a point mutation in alpha-tubulin. Mol Cancer Ther. 2004;3:1319–27. [PubMed] [Google Scholar]
- 169.Gaitanos TN, et al. Peloruside A does not bind to the taxoid site on beta-tubulin and retains its activity in multidrug-resistant cell lines. Cancer Res. 2004;64:5063–7. doi: 10.1158/0008-5472.CAN-04-0771. [DOI] [PubMed] [Google Scholar]
- 170.Risinger AL, et al. The taccalonolides: microtubule stabilizers that circumvent clinically relevant taxane resistance mechanisms. Cancer Res. 2008;68:8881–8. doi: 10.1158/0008-5472.CAN-08-2037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Bailly C, et al. Synthesis and biological evaluation of 4-arylcoumarin analogues of combretastatins. J Med Chem. 2003;46:5437–44. doi: 10.1021/jm030903d. [DOI] [PubMed] [Google Scholar]
- 172.Buey RM, et al. Cyclostreptin binds covalently to microtubule pores and lumenal taxoid binding sites. Nat Chem Biol. 2007;3:117–25. doi: 10.1038/nchembio853. [DOI] [PubMed] [Google Scholar]
- 173.Mozzetti S, et al. Molecular mechanisms of patupilone resistance. Cancer Res. 2008;68:10197–204. doi: 10.1158/0008-5472.CAN-08-2091. [DOI] [PubMed] [Google Scholar]
- 174.Ferlini C, et al. The seco-taxane IDN5390 is able to target class III beta-tubulin and to overcome paclitaxel resistance. Cancer Res. 2005;65:2397–405. doi: 10.1158/0008-5472.CAN-04-3065. [DOI] [PubMed] [Google Scholar]
- 175.Lewis Phillips GD, et al. Targeting HER2-positive breast cancer with trastuzumab-DM1, an antibody-cytotoxic drug conjugate. Cancer Res. 2008;68:9280–90. doi: 10.1158/0008-5472.CAN-08-1776. [DOI] [PubMed] [Google Scholar]
- 176.Al-Katib AM, Aboukameel A, Mohammad R, Bissery MC, Zuany-Amorim C. Superior antitumor activity of SAR3419 to rituximab in xenograft models for non-Hodgkin’s lymphoma. Clin Cancer Res. 2009;15:4038–45. doi: 10.1158/1078-0432.CCR-08-2808. [DOI] [PubMed] [Google Scholar]
- 177.Beeram M, et al. A phase I study of trastuzumab-MCC-DM1 (T-DM1), a first-in-class HER2 antibody-drug conjugate (ADC), in patients (pts) with HER2+ metastatic breast cancer (BC) Journal of Clinical Oncology. 2007;25S doi: 10.1200/JCO.2009.26.2071. Abst 1042. [DOI] [PubMed] [Google Scholar]
- 178.Krop IE, et al. Phase I study of trastuzumab-DM1, an HER2 antibody-drug conjugate, given every 3 weeks to patients with HER2-positive metastatic breast cancer. J Clin Oncol. 28:2698–704. doi: 10.1200/JCO.2009.26.2071. [DOI] [PubMed] [Google Scholar]
- 179.Smith SV. Technology evaluation: huN901-DM1, ImmunoGen. Curr Opin Mol Ther. 2005;7:394–401. [PubMed] [Google Scholar]
- 180.Jordan MA, Wilson L. Microtubules as a target for anticancer drugs. Nat Rev Cancer. 2004;4:253–65. doi: 10.1038/nrc1317. [DOI] [PubMed] [Google Scholar]
- 181.Stephenson JJ, et al. Phase I multicenter study to assess the safety, tolerability, and pharmacokinetics of AZD4877 administered twice weekly in adult patients with advanced solid malignancies. J Clin Oncol. 2008;26S abstr 2516. [Google Scholar]
- 182.Pusztai L, et al. Evaluation of microtubule-associated protein-Tau expression as a prognostic and predictive marker in the NSABP-B 28 randomized clinical trial. J Clin Oncol. 2009;27:4287–92. doi: 10.1200/JCO.2008.21.6887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Altieri DC. The case for survivin as a regulator of microtubule dynamics and cell-death decisions. Curr Opin Cell Biol. 2006;18:609–15. doi: 10.1016/j.ceb.2006.08.015. [DOI] [PubMed] [Google Scholar]
- 184.Cooper JR, Wagenbach M, Asbury CL, Wordeman L. Catalysis of the microtubule on-rate is the major parameter regulating the depolymerase activity of MCAK. Nat Struct Mol Biol. 2010;17:77–82. doi: 10.1038/nsmb.1728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Rana S, Maples PB, Senzer N, Nemunaitis J. Stathmin 1: a novel therapeutic target for anticancer activity. Expert Rev Anticancer Ther. 2008;8:1461–70. doi: 10.1586/14737140.8.9.1461. [DOI] [PubMed] [Google Scholar]
- 186.Pusztai L. Markers predicting clinical benefit in breast cancer from microtubule-targeting agents. Ann Oncol. 2007;18(Suppl 12):xii15–20. doi: 10.1093/annonc/mdm534. [DOI] [PubMed] [Google Scholar]
- 187.Seve P, Dumontet C. Chemoresistance in non-small cell lung cancer. Curr Med Chem Anticancer Agents. 2005;5:73–88. doi: 10.2174/1568011053352604. [DOI] [PubMed] [Google Scholar]
- 188.Marsh S, et al. Pharmacogenetic assessment of toxicity and outcome after platinum plus taxane chemotherapy in ovarian cancer: the Scottish Randomised Trial in Ovarian Cancer. J Clin Oncol. 2007;25:4528–35. doi: 10.1200/JCO.2006.10.4752. [DOI] [PubMed] [Google Scholar]
- 189.Bruggemann EP, Currier SJ, Gottesman MM, Pastan I. Characterization of the azidopine and vinblastine binding site of P-glycoprotein. J Biol Chem. 1992;267:21020–6. [PubMed] [Google Scholar]
- 190.Chen GK, Duran GE, Mangili A, Beketic-Oreskovic L, Sikic BI. MDR 1 activation is the predominant resistance mechanism selected by vinblastine in MES-SA cells. Br J Cancer. 2000;83:892–8. doi: 10.1054/bjoc.2000.1371. Overexpression of the Pgp efflux pump is frequently observed in cell lines exposed to vinca alkaloids in vitro. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Cole SP, et al. Pharmacological characterization of multidrug resistant MRP-transfected human tumor cells. Cancer Res. 1994;54:5902–10. [PubMed] [Google Scholar]
- 192.Hua XH, et al. Biochemical genetic analysis of indanocine resistance in human leukemia. Cancer Res. 2001;61:7248–54. [PubMed] [Google Scholar]
- 193.Wagner MM, et al. In vitro pharmacology of cryptophycin 52 (LY355703) in human tumor cell lines. Cancer Chemother Pharmacol. 1999;43:115–25. doi: 10.1007/s002800050871. [DOI] [PubMed] [Google Scholar]
- 194.Toppmeyer DL, Slapak CA, Croop J, Kufe DW. Role of P-glycoprotein in dolastatin 10 resistance. Biochem Pharmacol. 1994;48:609–12. doi: 10.1016/0006-2952(94)90292-5. [DOI] [PubMed] [Google Scholar]
- 195.Chou TC, et al. Therapeutic effect against human xenograft tumors in nude mice by the third generation microtubule stabilizing epothilones. Proc Natl Acad Sci U S A. 2008;105:13157–62. doi: 10.1073/pnas.0804773105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Xiao JJ, et al. Efflux of depsipeptide FK228 (FR901228, NSC-630176) is mediated by P-glycoprotein and multidrug resistance-associated protein 1. J Pharmacol Exp Ther. 2005;313:268–76. doi: 10.1124/jpet.104.072033. [DOI] [PubMed] [Google Scholar]
- 197.Gertsch J, et al. Making epothilones fluoresce: design, synthesis, and biological characterization of a fluorescent n12-aza-epothilone (azathilone) Chembiochem. 2009;10:2513–21. doi: 10.1002/cbic.200900376. [DOI] [PubMed] [Google Scholar]
- 198.Akashi Y, et al. The novel microtubule-interfering agent TZT-1027 enhances the anticancer effect of radiation in vitro and in vivo. Br J Cancer. 2007;96:1532–9. doi: 10.1038/sj.bjc.6603769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Simoni D, et al. Heterocyclic and phenyl double-bond-locked combretastatin analogues possessing potent apoptosis-inducing activity in HL60 and in MDR cell lines. J Med Chem. 2005;48:723–36. doi: 10.1021/jm049622b. [DOI] [PubMed] [Google Scholar]
- 200.Bayes M, Rabasseda X. Gateways to clinical trials. Methods Find Exp Clin Pharmacol. 2008;30:67–99. [PubMed] [Google Scholar]
- 201.Wehbe H, Kearney CM, Pinney KG. Combretastatin A-4 resistance in H460 human lung carcinoma demonstrates distinctive alterations in beta-tubulin isotype expression. Anticancer Res. 2005;25:3865–70. [PubMed] [Google Scholar]
- 202.Schumacher G, et al. Antineoplastic activity of 2-methoxyestradiol in human pancreatic and gastric cancer cells with different multidrug-resistant phenotypes. J Gastroenterol Hepatol. 2007;22:1469–73. doi: 10.1111/j.1440-1746.2007.05033.x. [DOI] [PubMed] [Google Scholar]
- 203.Lockhart AC, et al. Phase I trial of oral MAC-321 in subjects with advanced malignant solid tumors. Cancer Chemother Pharmacol. 2007;60:203–9. doi: 10.1007/s00280-006-0362-y. [DOI] [PubMed] [Google Scholar]
- 204.Ramanathan RK, et al. A phase II study of milataxel: a novel taxane analogue in previously treated patients with advanced colorectal cancer. Cancer Chemother Pharmacol. 2008;61:453–8. doi: 10.1007/s00280-007-0489-5. [DOI] [PubMed] [Google Scholar]
- 205.Yamamoto N, Boku N, Minami H. Phase I study of larotaxel administered as a 1-h intravenous infusion every 3 weeks to Japanese patients with advanced solid tumours. Cancer Chemother Pharmacol. 2009;65:129–36. doi: 10.1007/s00280-009-1014-9. [DOI] [PubMed] [Google Scholar]
- 206.Dieras V, et al. Phase II multicenter study of larotaxel (XRP9881), a novel taxoid, in patients with metastatic breast cancer who previously received taxane-based therapy. Ann Oncol. 2008;19:1255–60. doi: 10.1093/annonc/mdn060. [DOI] [PubMed] [Google Scholar]
- 207.Metzger-Filho O, Moulin C, de Azambuja E, Ahmad A. Larotaxel: broadening the road with new taxanes. Expert Opin Investig Drugs. 2009;18:1183–9. doi: 10.1517/13543780903119167. [DOI] [PubMed] [Google Scholar]
- 208.Beer M, Lenaz L, Amadori D. Phase II study of ortataxel in taxane-resistant breast cancer. J Clin Oncol. 2008;26S abstr 1066. [Google Scholar]
- 209.Rhee J, Lee F, Saif M. Phase II trial of DJ-927 as a second-line treatment for colorectal cancer demonstrates objective responses. J Clin Oncol. 2005;284S:A3654. [Google Scholar]
- 210.Baas P, et al. Phase I/II study of a 3 weekly oral taxane (DJ-927) in patients with recurrent, advanced non-small cell lung cancer. J Thorac Oncol. 2008;3:745–50. doi: 10.1097/JTO.0b013e31817c73ff. [DOI] [PubMed] [Google Scholar]
- 211.Patel SR, et al. Phase II study of CI-980 (NSC 635370) in patients with previously treated advanced soft-tissue sarcomas. Invest New Drugs. 1998;16:87–92. doi: 10.1023/a:1006078930550. [DOI] [PubMed] [Google Scholar]
- 212.Pazdur R, et al. Phase II trial of intravenous CI-980 (NSC 370147) in patients with metastatic colorectal carcinoma. Model for prospective evaluation of neurotoxicity. Am J Clin Oncol. 1997;20:573–6. doi: 10.1097/00000421-199712000-00008. [DOI] [PubMed] [Google Scholar]
- 213.Judson I, et al. Phase I trial and pharmacokinetics of the tubulin inhibitor 1069C85--a synthetic agent binding at the colchicine site designed to overcome multidrug resistance. Br J Cancer. 1997;75:608–13. doi: 10.1038/bjc.1997.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Yamamoto K, et al. Phase I study of E7010. Cancer Chemother Pharmacol. 1998;42:127–34. doi: 10.1007/s002800050795. [DOI] [PubMed] [Google Scholar]
- 215.Kuppens IE, et al. Phase I dose-finding and pharmacokinetic trial of orally administered indibulin (D-24851) to patients with solid tumors. Invest New Drugs. 2007;25:227–35. doi: 10.1007/s10637-006-9027-2. [DOI] [PubMed] [Google Scholar]


