Abstract
The use of combination antibiotic therapy may be beneficial against rapidly emerging resistance in Pseudomonas aeruginosa. The aim of this study was to systematically investigate in vitro bacterial killing and resistance emergence with colistin alone and in combination with imipenem against multidrug-resistant (MDR) P. aeruginosa. Time-kill studies were conducted over 48 h using 5 clinical isolates and ATCC 27853 at two inocula (∼106 and ∼108 CFU/ml); MDR, non-MDR, and colistin-heteroresistant and -resistant strains were included. Nine colistin-imipenem combinations were investigated. Microbiological response was examined by log changes at 6, 24, and 48 h. Colistin combined with imipenem at clinically relevant concentrations increased the levels of killing of MDR and colistin-heteroresistant isolates at both inocula. Substantial improvements in activity with combinations were observed across 48 h with all colistin concentrations at the low inoculum and with colistin at 4× and 16× MIC (or 4 and 32 mg/liter) at the high inoculum. Combinations were additive or synergistic against imipenem-resistant isolates (MICs, 16 and 32 mg/liter) at the 106-CFU inoculum in 9, 11, and 12 of 18 cases (i.e., 9 combinations across 2 isolates) at 6, 24, and 48 h, respectively, and against the same isolates at the 108-CFU inoculum in 11, 7, and 8 cases, respectively. Against a colistin-resistant strain (MIC, 128 mg/liter), combinations were additive or synergistic in 9 and 8 of 9 cases at 24 h at the 106- and 108-CFU inocula, respectively, and in 5 and 7 cases at 48 h. This systematic study provides important information for optimization of colistin-imipenem combinations targeting both colistin-susceptible and colistin-resistant subpopulations.
INTRODUCTION
The world is facing a growing threat from multidrug-resistant (MDR) Gram-negative “superbugs,” such as Pseudomonas aeruginosa, Acinetobacter baumannii, and Klebsiella pneumoniae (19, 30, 50). This problem is compounded by a lack of novel antimicrobial agents in the drug development pipeline for infections caused by Gram-negative bacteria (30, 31, 50), in particular those caused by P. aeruginosa (50), and novel agents with activity against this pathogen may not be available for approximately 10 years (41). This has led to the reevaluation of colistin (also known as polymyxin E), a multicomponent cationic polypeptide antibiotic that entered clinical use in 1959 but was largely replaced by aminoglycosides in the 1970s due to concerns about the potential for nephrotoxicity and neurotoxicity (12, 23, 26, 27). Owing to its significant in vitro antibacterial activity against Gram-negative “superbugs,” colistin is often the only therapeutic option available for the treatment of infections by these pathogens (1, 27, 35), and therefore, its use has increased substantially over the past 5 years, especially for critically ill patients (6, 27).
It is now evident that the concentrations of colistin achieved in the plasma of critically ill patients with the currently recommended dosage regimens are suboptimal for a significant proportion of patients (13, 43). Unfortunately, increasing the daily dose may not be an acceptable option, since nephrotoxicity is a dose-limiting adverse effect and occurs in 30 to 50% of patients (13, 16, 22). It is therefore not surprising that suboptimal concentrations cause the emergence of resistance to colistin, which seriously threatens colistin therapy (45, 48). In vivo (21, 33) and in vitro (3, 4) studies show the potential for the rapid emergence of colistin resistance with monotherapy. The phenomenon of colistin heteroresistance (the presence of colistin-resistant subpopulations in an isolate considered susceptible by MIC measurement) (56) has been reported for A. baumannii (29, 56) and K. pneumoniae (34, 44, 54) but not yet for P. aeruginosa. Heteroresistance very likely contributes to the emergence of colistin resistance. The aim of the present study was to systematically investigate the extent of in vitro bacterial killing and the emergence of colistin resistance with colistin alone and in combination with imipenem against P. aeruginosa. Key aspects of this study were the use of MDR isolates with various susceptibilities to colistin and imipenem (including colistin-heteroresistant isolates first identified in this study, as well as colistin- and imipenem-resistant strains), examination of combinations of clinically relevant drug concentrations at both low and high inocula, and monitoring of the emergence of resistance to colistin with real-time population analysis profiles (PAPs).
(Part of this study was presented at the 49th Interscience Conference on Antimicrobial Agents and Chemotherapy, San Francisco, CA, 12 to 15 September 2009.)
MATERIALS AND METHODS
Bacterial isolates.
Five clinical isolates and P. aeruginosa ATCC 27853 (American Type Culture Collection, Manassas, VA) were selected to represent a mixture of strains susceptible and resistant to colistin and imipenem, colistin-heteroresistant and nonheteroresistant strains, and multidrug-resistant (MDR) and non-MDR strains. MDR was defined as diminished susceptibility to at least two of the following five drug classes: antipseudomonal cephalosporins, antipseudomonal carbapenems, β-lactam-β-lactamase inhibitor combinations, antipseudomonal fluoroquinolones, and aminoglycosides (40). In addition, all strains were examined by PCR for the presence of genes encoding cephalosporinases and carbapenemases, i.e., IMP-, VIM-, NDM-, KPC-, CTX-M-, SHV-, and CMY-type β-lactamases (47, 58). The isolates are described in detail in Table 1. All clinical isolates were collected from patients with cystic fibrosis, had different pulsed-field gel electrophoresis patterns, and were considered unrelated according to the criteria established by Tenover et al. (53). The MICs of colistin and imipenem were determined for each isolate in four replicates in cation-adjusted Mueller-Hinton broth (CAMHB; containing 23.0 mg Ca2+/liter and 12.2 mg Mg2+/liter; Oxoid, Hampshire, England) by broth microdilution (10). Isolates were stored in tryptone soy broth (Oxoid) with 20% glycerol (Ajax Finechem, Seven Hills, NSW, Australia) at −80°C in cryovials (Simport Plastics, Beloeil, Quebec, Canada).
Table 1.
MICs for the P. aeruginosa isolates used in this study
| Isolate | MIC (mg/liter)a |
Cephalosporinase and carbapenemase typing | MDRb | |
|---|---|---|---|---|
| Colistin | Imipenem | |||
| ATCC 27853c | 1 | 2 | Negative | No |
| 19147 n/m | 128 | 4 | IMP and CTX-M positived | Yes |
| 19056 muc | 0.5 | 4 | Negative | Yes |
| 20509 n/mc | 0.5 | 1 | Negative | No |
| 19271 n/mc | 2 | 32 | Negative | Yes |
| 20891 n/mc | 1 | 16 | Negative | Yes |
CLSI breakpoints for colistin were ≤2 mg/liter for susceptibility, 4 mg/liter for intermediacy, and ≥8 mg/liter for resistance. For imipenem, the breakpoints were ≤4 mg/liter for susceptibility, 8 mg/liter for intermediacy, and ≥16 mg/liter for resistance (10).
Defined as diminished susceptibility to ≥2 of the following 5 drug classes: antipseudomonal cephalosporins, antipseudomonal carbapenems, β-lactam-β-lactamase inhibitor combinations, antipseudomonal fluoroquinolones, and aminoglycosides (40).
Colistin heteroresistant. Heteroresistance to colistin was defined as the existence, in an isolate for which the colistin MIC was ≤2 mg/liter, of subpopulations able to grow in the presence of >2 mg/liter colistin (55).
Contains genes encoding an IMP-type carbapenemase and a CTX-M-type ESBL.
Antibiotics.
Colistin sulfate (lot 109K1574; 23,251 U/mg) was purchased from Sigma-Aldrich, St. Louis, MO. Colistin (sulfate) was employed in the current study because it is the active antibacterial agent formed in vivo after administration of its inactive prodrug, colistin methanesulfonate (CMS) (5). Imipenem (Primaxin; batch K5942) was purchased from Merck Sharp & Dohme, NSW, Australia. Stock solutions of each antibiotic were prepared according to the respective manufacturer's instructions immediately prior to each experiment in order to minimize loss from degradation; then the solutions were sterilized by filtration with a 0.22-μm-pore-size Millex-GP filter (Millipore, Bedford, MA).
PAPs.
The possible existence of colistin-resistant subpopulations at baseline was determined via population analysis profiles (PAPs) (inoculum, ∼108 CFU/ml). Colistin heteroresistance was defined as the existence, in a colistin-susceptible isolate (i.e., MIC, ≤2 mg/liter) of subpopulations that were able to grow in the presence of >2 mg/liter colistin in the PAPs. Samples of bacterial cell suspensions (50 μl), appropriately diluted with saline, were spirally plated onto Mueller-Hinton agar (Media Preparation Unit, The University of Melbourne, Parkville, Australia) impregnated with colistin (0, 0.5, 1, 2, 3, 4, 6, 8, or 10 mg/liter) by using an automatic spiral plater (WASP; Don Whitley Scientific, West Yorkshire, United Kingdom). Colonies were counted using a ProtoCOL colony counter (Synbiosis, Cambridge, United Kingdom) after 24 h of incubation (48 h for plates with small colonies) at 35°C; the limit of detection was 20 CFU/ml (equivalent to 1 colony per plate), and the limit of quantification (LOQ) was 400 CFU/ml (equivalent to 20 colonies per plate), as specified in the ProtoCOL manual. Real-time PAPs for colistin were also conducted at the end of time-kill studies (see below).
Time-kill studies.
To explore the antimicrobial activities of colistin and imipenem combinations, time-kill studies with each antibiotic alone or in combination were conducted on all isolates at two different starting inocula (∼106 and ∼108 CFU/ml). For monotherapy with colistin or imipenem, 2-fold multiples of the MIC (0.25× to 64× MIC) were employed for susceptible isolates. For the colistin-resistant isolate (19147 n/m; MIC, 128 mg/liter), a single colistin concentration of 32 mg/liter was employed. Imipenem concentrations of 1, 8, and 32 mg/liter were used for imipenem-resistant isolates. In combination experiments, both antibiotics were studied at concentrations of 0.5×, 4×, and 16× MIC for susceptible isolates; for resistant isolates, concentrations of 1, 4, and 32 mg/liter for colistin and 1, 8, and 32 mg/liter for imipenem were employed. In total, nine colistin-imipenem combinations were examined for each isolate at each inoculum.
Prior to each experiment, isolates were subcultured onto horse blood agar (Media Preparation Unit) and were incubated at 35°C overnight. One colony was then selected and grown overnight in 10 ml CAMHB at 37°C; from this colony, an early-log-phase culture was obtained. Each antibiotic was added alone or in combination to 20 ml of a log-phase broth culture of approximately 106 or 108 CFU/ml to yield the desired concentrations. Each 20-ml culture was placed in a sterile 50-ml polypropylene tube (Greiner Bio One) and was incubated in a shaking water bath at 37°C. Serial samples (100 μl) were collected aseptically for viable-cell counting at 0, 0.5, 1, 2, 4, 6, 24, and 48 h and for PAPs at 48 h (see above) for all experiments involving colistin (including combination arms) and for viable-cell counting only at 0, 1, 2, 4, 6, 24, and 48 h for experiments with imipenem alone. Immediately after sampling and serial dilution, 50 μl of the bacterial cell suspension was spirally plated onto nutrient agar with enumeration after 24 h of incubation (48 h for plates with small colonies) as described under “PAPs” above.
Pharmacodynamic (PD) analysis.
Microbiological responses to monotherapy and combination therapy were examined using the log change method, comparing the change in bacterial counts (log10 CFU/ml) from that at 0 h [log10(CFU0)] to that at a given time (t) (6, 24, or 48 h) [log10(CFUt)], as follows: log change = log10(CFUt) − log10(CFU0).
Single antibiotic or combination regimens causing a reduction of ≥1 log10 CFU/ml from the initial inoculum at 6, 24, or 48 h were considered active. We considered synergy to be indicated by a ≥2-log10 CFU/ml-lower bacterial count with the combination than with its most active component at the specified time (42); additivity was defined by a 1- to <2-log10 CFU/ml-lower bacterial count with the combination.
RESULTS
Microbiological response.
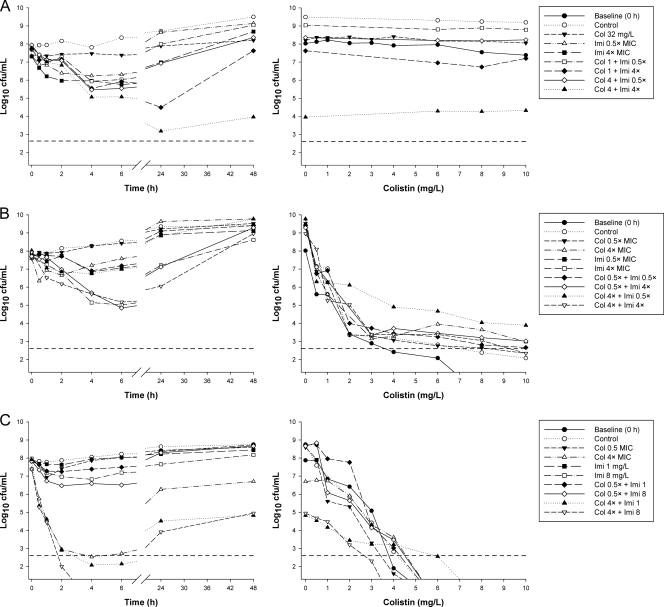
The various susceptibilities of the isolates to colistin are evident in the PAPs obtained prior to colistin treatment (Fig. 1). Representative time-kill profiles for colistin and imipenem monotherapy and combination therapy are shown in Fig. 2 (inoculum, ∼106 CFU/ml) and 3 (inoculum, ∼108 CFU/ml). Log changes in viable cell counts at each inoculum with clinically relevant colistin concentrations are presented in Tables 2 and 3. Additional time-kill and log change data are presented in the supplemental material. At the 106-CFU/ml inoculum, regrowth was observed to various extents for all susceptible isolates at 48 h with colistin monotherapy with the majority of concentrations. Regrowth with imipenem monotherapy at concentrations of ≥4× or 8× MIC was more variable and substantially less for susceptible isolates at 48 h, even when extended-spectrum β-lactamases (ESBLs) were present. An inoculum effect with colistin monotherapy was generally observed (Fig. 2 and 3, left). Killing by imipenem was generally slightly slower at the high inoculum than at the low inoculum, although the extents of reduction in bacterial counts (log10 CFU/ml) were comparable at the two inocula (Fig. 2 and 3).
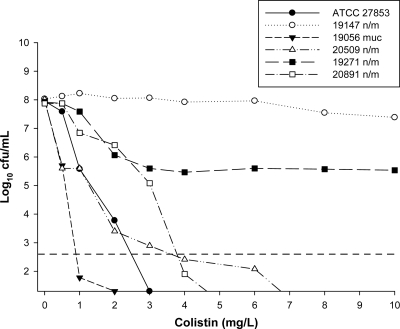
Fig. 1.
Baseline PAPs of the reference strain and all clinical isolates at an initial inoculum of ∼108 CFU/ml. The y axis starts from the limit of detection, and the LOQ is indicated by the dashed horizontal line.
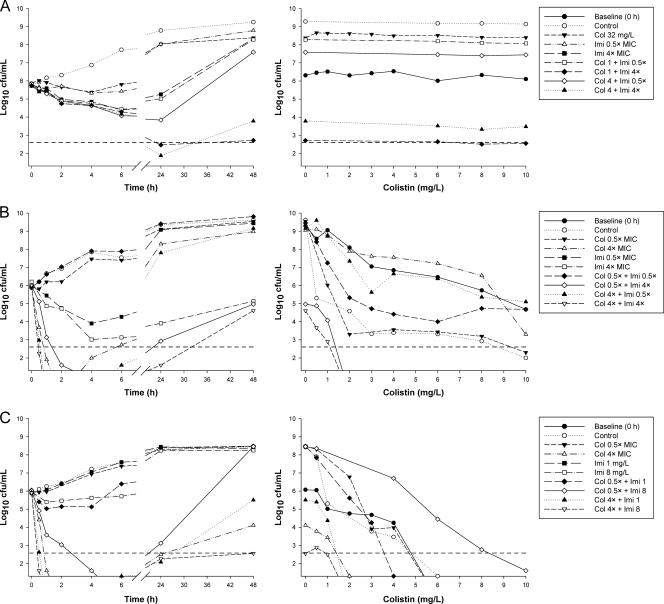
Fig. 2.
(Left) Representative time-kill curves with various clinically relevant concentrations of colistin (Col) and imipenem (Imi) alone and in combination at an inoculum of ∼106 CFU/ml. (Right) PAPs at baseline (0 h) and after 48 h of exposure to colistin monotherapy, colistin-imipenem combination therapy, or neither antibiotic (control). (A) 19147 n/m (colistin resistant, imipenem susceptible, MDR); (B) 20509 n/m (susceptible to colistin and imipenem, non-MDR); (C) 20891 n/m (colistin susceptible, imipenem resistant, MDR). The y axis starts from the limit of detection, and the LOQ is indicated by the dashed horizontal line.
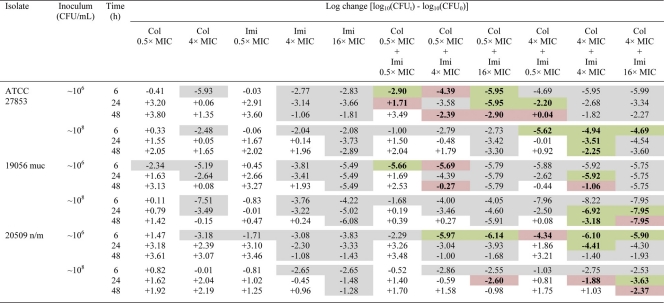
Table 2.
Log changes in viable cell counts at 6, 24, and 48 h with various clinically relevant concentrations of colistin and imipenem against three P. aeruginosa isolates susceptible to both antibioticsa
Col, colistin; Imi, imipenem. A gray background indicates activity (a reduction of ≥1 log10 CFU/ml below the initial inoculum); a green background indicates synergy (a ≥2-log10 decrease in the number of CFU/ml with the combination from that with its most active component); and a red background indicates additivity (a 1.0- to <2-log10 decrease in the number of CFU/ml with the combination from that with its most active component).
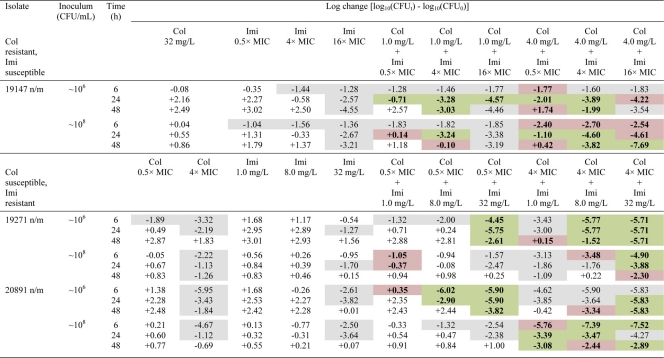
Table 3.
Log changes in viable cell counts at 6, 24, and 48 h with various clinically relevant concentrations of colistin and imipenem against one colistin-resistant, imipenem-susceptible isolate and two colistin-susceptible, imipenem-resistant isolates of P. aeruginosaa
Col, colistin; Imi, imipenem. A gray background indicates activity (a reduction of ≥1 log10 CFU/ml below the initial inoculum); a green background indicates synergy (a ≥2-log10 decrease in the number of CFU/ml with the combination from that with its most active component); and a red background indicates additivity (a 1.0- to <2-log10 decrease in the number of CFU/ml with the combination from that with its most active component). For colistin-resistant isolate 19147 n/m, synergy or additivity was compared with imipenem monotherapy only.
Fig. 3.
(Left) Representative time-kill curves with various clinically relevant concentrations of colistin (Col) and imipenem (Imi) alone and in combination at an inoculum of ∼108 CFU/ml. (Right) PAPs at baseline (0 h) and after 48 h of exposure to colistin monotherapy, colistin-imipenem combination therapy, or neither antibiotic (control). (A) 19147 n/m (colistin resistant, imipenem susceptible, MDR); (B) 20509 n/m (susceptible to colistin and imipenem, non-MDR); (C) 20891 n/m (colistin susceptible, imipenem resistant, MDR). The y axis starts from the limit of detection, and the LOQ is indicated by the dashed horizontal line.
Isolates susceptible to both colistin and imipenem.
At the 106-CFU/ml inoculum, the addition of colistin at 0.5× MIC to imipenem (all concentrations) resulted in additivity or synergy at 6 h in 7 of 9 cases (i.e., 3 combinations against 3 isolates), achieving a ∼2- to 3-log10 greater kill than that with the most active equivalent monotherapy, and undetectable bacterial counts in many cases (Table 2 and Fig. 2). By 24 or 48 h, improvements in activity with combination therapy over that with the most active monotherapy (usually imipenem) were modest, particularly when only clinically relevant concentrations of colistin (0.5× or 4× MIC) were considered. Of the 27 cases (i.e., 9 combinations against 3 isolates), 7 at 24 h and 8 at 48 h showed additivity or synergy, although only 1 case resulted in activity (i.e., ≥1-log10 kill) if equivalent monotherapy with either drug was inactive. A similar pattern of activity was observed at the 108-CFU/ml inoculum. For ATCC 27853, combinations containing colistin at 4× MIC provided an additional ∼2-log10 kill over that with active monotherapy at 6 h. Against all three isolates, there were 10 and 9 cases of additivity/synergy at 24 and 48 h, respectively, mostly involving colistin at 4× or 16× MIC (Table 2).
Imipenem-resistant isolates.
For the two imipenem-resistant isolates (19271 n/m and 20891 n/m), there was no evidence of carbapenemase activity; most likely, an alternative resistance mechanism, such as the loss of major outer membrane proteins, was present. At the low inoculum, combination therapy resulted in substantial improvements in bacterial kill with all colistin concentrations across 48 h. At 6 h, additivity/synergy occurred in 9 of 18 cases (i.e., 9 combinations across 2 isolates), predominantly against isolate 19271 n/m; additivity/synergy occurred with combinations containing colistin at all concentrations and produced additional reductions of ∼2- to 6-log10 CFU/ml over that with usually active colistin monotherapy (Table 3 and Fig. 2). In 5 of 6 cases involving colistin at 4× or 16× MIC against 19271 n/m, bacterial counts were reduced to below the limit of detection (i.e., 20 CFU/ml). Substantial improvements in activity against both isolates were also observed at 24 and 48 h at all colistin concentrations. Additivity/synergy occurred in 11 and 12 of 18 cases at 24 and 48 h, respectively, resulting in an additional ∼1- to 4-log10 kill at 24 h and >2.5-log10 kill at 48 h over that with monotherapy (Table 3 and Fig. 2). Interestingly, the combinations of colistin at 0.5×, 4×, or 16× MIC with imipenem at 32 mg/liter each reduced the bacterial loads of both isolates to below the limit of detection at 24 h; the maximum reduction in bacterial counts (log10 CFU/ml) at 24 h with colistin monotherapy at 16× MIC was ∼4.5. Improvements in activity with combination therapy at the high inoculum also occurred at all time points but were essentially restricted to combinations containing colistin at 4× or 16× MIC. Ten of 12 cases at 6 h containing colistin at 4× MIC (Table 3) or 16× MIC (data not shown) showed additivity or synergy. At 24 and 48 h, the addition of imipenem at all concentrations to colistin at 4× or 16× MIC produced additivity/synergy in more than half of all cases and substantially improved the activity over that with each antibiotic alone (by as much as ∼4-log10 kill).
Colistin-resistant isolate.
Bacterial killing at the 106-CFU/ml inoculum was substantially enhanced at 24 h: all combinations tested were additive or synergistic, and only one combination (colistin at 1 mg/liter plus imipenem at 0.5× MIC) was inactive (Table 3). The addition of all colistin concentrations to imipenem at 4× or 16× MIC produced ∼3.5- to 4.5-log10 kill at 24 h, substantially higher than that with equivalent imipenem monotherapy. At 48 h, all colistin concentrations in combination with imipenem at 4× MIC were synergistic (∼2- to 4-log10 kill) and substantially improved activity over that with equivalent monotherapy. The addition of colistin at 32 mg/liter to imipenem (all concentrations) was additive or synergistic at a substantially earlier time (6 h), with ∼1- to 2-log10 greater kill than that with equivalent imipenem monotherapy (overall kill, ∼3 log10 CFU/ml). At the high inoculum, additivity was achieved at 6 h with all combinations containing colistin at 4 mg/liter (Table 3) and 32 mg/liter (data not shown), and activity was enhanced by ∼1-log10 kill over that with imipenem monotherapy. Eight of 9 combinations at 24 h and 7 of 9 combinations at 48 h were additive or synergistic, encompassing all colistin concentrations and in many cases resulting in additional reductions of ∼1 log10 to 4 log10 CFU/ml over that with the most active monotherapy (imipenem at 16× MIC). This enhancement of activity was particularly evident with combinations containing colistin at 4 or 32 mg/liter, and on two occasions, when colistin was combined with imipenem at 16× MIC, no viable bacteria were detected at 48 h.
Emergence of colistin resistance.
For the 4 colistin-heteroresistant isolates (Table 1), the proportion of resistant subpopulations at 108 CFU/ml ranged from 2.2 × 10−7 to 4.7 × 10−3 (Fig. 1). With colistin monotherapy against isolates susceptible to both colistin and imipenem, real-time PAPs performed at 48 h in the time-kill studies demonstrated increases in colistin-resistant subpopulations at both the low and high inocula with clinically relevant colistin concentrations (examples are shown in Fig. 2 and 3; see also the supplemental material); no such increase was observed with isolate 19056 muc at the high inoculum. Against imipenem-resistant isolate 19271 n/m, colistin concentrations of 0.25× to 64× MIC at the low inoculum and 1× to 64× MIC at the high inoculum resulted in nearly 100% of the remaining cells at 48 h growing in the presence of 10 mg/liter colistin. In contrast, no increase in colistin-resistant subpopulations was observed for the imipenem-resistant isolate 20891 n/m at either inoculum. Combination therapy against colistin-susceptible isolates generally had little effect on the proportion of colistin-resistant subpopulations at 48 h at either inoculum; the shapes of the PAPs were very similar to those obtained with equivalent colistin monotherapy (Fig. 2 and 3).
DISCUSSION
Although colistin has been commercially available for more than 50 years (27), reliable PK/PD data have emerged only recently. Population PK studies have shown that plasma colistin concentrations achieved with currently recommended CMS dosage regimens are likely to be suboptimal for many patients, typically generating average steady-state plasma colistin concentrations of ∼2 to 3 mg/liter, with some patients achieving concentrations as high as ∼10 mg/liter (13, 18, 24, 28, 32, 43). Increasing the daily dose of CMS for such patients may not be an option, since nephrotoxicity, which occurs in ∼30 to 50% of patients (16, 22), is a dose-limiting adverse effect. Given these circumstances and the current last-line status of colistin therapy, we chose to examine not only synergy but also additivity, since even a relatively small increase in activity with combination therapy may be beneficial for patient care. Because colistin is almost entirely unbound in CAMHB (3), colistin concentrations of 0.5× and 4× MIC for isolates with MICs of ≤1 mg/liter and 16× MIC for isolates with MICs of ≤0.5 mg/liter (1 and 4 mg/liter for colistin-resistant isolates) used in our study are clinically relevant, even assuming that binding of colistin by plasma in patients is similar to that in animals (i.e., ∼50% bound) (25). Considering the effect of protein binding, all the imipenem concentrations employed are readily achieved in plasma (49).
Because some data show that the activities of both colistin (8) and imipenem (36) are attenuated at high inocula compared to those at low inocula, experiments were conducted at inocula of both ∼106 and ∼108 CFU/ml. An inoculum effect was generally observed for colistin monotherapy, whereas no obvious inoculum effect was present for imipenem (Fig. 2 and 3). Regrowth of all isolates was observed with colistin monotherapy, even at colistin concentrations well above those that can safely be achieved clinically. Similar regrowth with colistin (or polymyxin B) monotherapy has been observed for colistin-susceptible P. aeruginosa both in vitro (4, 8, 15, 51) and in vivo (21). For A. baumannii and K. pneumoniae, regrowth following colistin monotherapy has been attributed to the amplification of colistin-resistant subpopulations (11, 44, 52), with colistin heteroresistance reported in both species (17, 29, 44, 54). We have reported here, for the first time, colistin heteroresistance in P. aeruginosa. The emergence of colistin resistance following colistin monotherapy has been reported previously for P. aeruginosa at both low and high inocula (4, 8), and a similar phenomenon was observed in the present study for all isolates except 20981 n/m. While P. aeruginosa can undergo adaptive resistance to polymyxins (14), the presence of colistin heteroresistance at baseline and the changes in PAPs after treatment suggest that regrowth following colistin monotherapy may be due to amplification of preexisting colistin-resistant subpopulations. This possibility suggests that care is required with colistin monotherapy against P. aeruginosa, even where isolates appear susceptible on the basis of MICs.
The addition of imipenem to colistin at both inocula generally resulted in substantial improvements in bacterial killing over that with equivalent monotherapy against MDR P. aeruginosa isolates resistant to either antibiotic, even when ESBLs were present. The improvements in activity against these isolates were observed across the 48-h duration, with all colistin concentrations at the low inoculum and with colistin at 4× and 16× MIC (or 4 and 32 mg/liter) at the high inoculum. Notably, the total reductions in bacterial counts (log10 CFU/ml) achieved with combinations containing lower colistin concentrations (0.5× and 4× MIC or 1 and 4 mg/liter) were on many occasions similar in magnitude to the reductions achieved with combinations containing colistin at 16× MIC, particularly at the 106-CFU/ml inoculum (Table 3). This suggests that combinations of colistin and imipenem containing clinically relevant colistin concentrations may be as effective as combinations containing higher concentrations against MDR isolates when resistance to either drug is present. This is an important result, given that colistin-induced nephrotoxicity is a dose-limiting adverse effect.
The benefits of the addition of imipenem to colistin for overall antibacterial activity were less pronounced against the three isolates susceptible to both antibiotics and were generally restricted to improvements in initial kill, i.e., up to 6 h (Table 2). Because a proportion of patients will achieve only low plasma colistin concentrations with the currently recommended dosage regimens (13, 43), the combination of colistin and imipenem at the commencement of therapy may help to quickly reduce bacterial levels so as to facilitate clearance by the immune system.
Previous time-kill studies have examined colistin in combination with carbapenems against P. aeruginosa (2, 9, 38, 39, 46). These studies examined colistin with imipenem, meropenem, or doripenem at a single inoculum (∼106 or 107 CFU/ml), though the emergence of colistin resistance was not examined (e.g., by use of PAPs). The present study is the first to investigate the emergence of colistin resistance with colistin combination therapy. In the present investigations, in cases where the combination led to extensive killing at 48 h, meaningful interpretation of the PAPs was not possible (e.g., Fig. 2B, colistin at 4× MIC as monotherapy and in combination with imipenem at 4× MIC). When bacterial numbers at 48 h were comparable, changes in PAPs with combination therapy generally mirrored those observed with equivalent exposure to colistin as monotherapy. However, in both the present study and previously reported studies (2, 9, 38, 39, 46), static concentrations and the instability of carbapenems in aqueous media may have contributed to regrowth and the emergence of colistin resistance at 48 h (20). Thus, it will be important to further assess the utility of these combinations against a range of isolates with various susceptibilities (including heteroresistant strains) in dynamic in vitro models and in vivo.
Two possible reasons for the enhanced pharmacodynamic effect observed with the combination of colistin and imipenem are subpopulation synergy and mechanistic synergy, as proposed previously (7). Subpopulation synergy involves one drug killing the subpopulation(s) resistant to the other drug, and vice versa. Four of the six isolates in the present study were colistin heteroresistant (Table 1), indicating the existence of colistin-resistant subpopulations prior to therapy. In addition, the four imipenem-susceptible isolates were imipenem heteroresistant (while the MIC was ≤4 mg/liter, subpopulations grew in the presence of >4 mg/liter imipenem [data not shown]). Another possibility is mechanistic synergy, whereby colistin and imipenem, acting on different cellular pathways, each increase the rate or extent of killing of the other drug. In Gram-negative bacteria, carbapenems must first gain entry into the periplasmic space in order to bind to critical penicillin-binding proteins located on the cytoplasmic membrane (37, 55). A number of resistance mechanisms may operate to limit the concentration of carbapenems in the periplasm, including the presence of carbapenem-hydrolyzing enzymes and the loss of outer membrane proteins (55). Polymyxins cause considerable permeabilization of the outer membrane (57). It is possible that the effect of colistin on membrane permeability results in substantially increased concentrations of imipenem in the periplasm and improved bactericidal activity. Subpopulation and mechanistic synergies are not mutually exclusive; both may operate simultaneously. Further studies, including mechanism-based mathematical modeling, to investigate the mechanism(s) underpinning the enhanced pharmacodynamic activity observed are ongoing.
In the battle against rapidly emerging bacterial resistance in Gram-negative “superbugs,” rational approaches to the use of combinations of existing antibiotics may be very beneficial. To the best of our knowledge, this is the first systematic study on the PD of colistin in combination with imipenem against P. aeruginosa, including MDR and colistin-heteroresistant strains, at both low and high inocula. Clinically relevant concentrations of colistin in combination with imipenem substantially increased bacterial killing against MDR P. aeruginosa isolates at both inocula when isolates were resistant to either antibiotic. Further investigations in in vitro pharmacodynamic systems, animal infection models, and clinical studies are warranted to optimize colistin-imipenem combinations targeting both colistin-susceptible and colistin-resistant subpopulations.
Supplementary Material
ACKNOWLEDGMENTS
The project reported here was supported by award R01AI079330 from the National Institute of Allergy and Infectious Diseases. J.L. is an Australian National Health and Medical Research Council Senior Research Fellow. D.L.P. has previously received honoraria from Merck for invited lectures and participation in advisory boards.
The content of this paper is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health.
Footnotes
Supplemental material for this article may be found at http://aac.asm.org/.
Published ahead of print on 29 August 2011.
REFERENCES
- 1. Antoniadou A., et al. 2007. Colistin-resistant isolates of Klebsiella pneumoniae emerging in intensive care unit patients: first report of a multiclonal cluster. J. Antimicrob. Chemother. 59:786–790 [DOI] [PubMed] [Google Scholar]
- 2. Aoki N., et al. 2009. Efficacy of colistin combination therapy in a mouse model of pneumonia caused by multidrug-resistant Pseudomonas aeruginosa. J. Antimicrob. Chemother. 63:534–542 [DOI] [PubMed] [Google Scholar]
- 3. Bergen P. J., et al. 2010. Pharmacokinetic/pharmacodynamic investigation of colistin against Pseudomonas aeruginosa using an in vitro model. Antimicrob. Agents Chemother. 54:3783–3789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Bergen P. J., et al. 2008. Comparison of once-, twice- and thrice-daily dosing of colistin on antibacterial effect and emergence of resistance: studies with Pseudomonas aeruginosa in an in vitro pharmacodynamic model. J. Antimicrob. Chemother. 61:636–642 [DOI] [PubMed] [Google Scholar]
- 5. Bergen P. J., Li J., Rayner C. R., Nation R. L. 2006. Colistin methanesulfonate is an inactive prodrug of colistin against Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 50:1953–1958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Boucher H. W., et al. 2009. Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin. Infect. Dis. 48:1–12 [DOI] [PubMed] [Google Scholar]
- 7. Bulitta J. B., et al. 2009. Quantifying synergy of colistin combinations against MDR Gram-negatives by mechanism-based models, abstr. A1-573, p. 41. Abstr. 49th Annu. Intersci. Conf. Antimicrob. Agents Chemother., San Francisco, CA, 12 to 15 September 2009 American Society for Microbiology, Washington, DC [Google Scholar]
- 8. Bulitta J. B., et al. 2010. Attenuation of colistin bactericidal activity by high inoculum of Pseudomonas aeruginosa characterized by a new mechanism-based population pharmacodynamic model. Antimicrob. Agents Chemother. 54:2051–2062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Cirioni O., et al. 2007. Efficacy of tachyplesin III, colistin, and imipenem against a multiresistant Pseudomonas aeruginosa strain. Antimicrob. Agents Chemother. 51:2005–2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Clinical Laboratory Standards Institute, 2010. Performance standards for antimicrobial susceptibility testing; twentieth informational supplement (M100-S20). Clinical and Laboratory Standards Institute, Wayne, PA [Google Scholar]
- 11. Dudhani R. V., Turnidge J. D., Nation R. L., Li J. 2010. fAUC/MIC is the most predictive pharmacokinetic/pharmacodynamic index of colistin against Acinetobacter baumannii in murine thigh and lung infection models. J. Antimicrob. Chemother. 65:1984–1990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Falagas M. E., Kasiakou S. K. 2005. Colistin: the revival of polymyxins for the management of multidrug-resistant gram-negative bacterial infections. Clin. Infect. Dis. 40:1333–1341 [DOI] [PubMed] [Google Scholar]
- 13. Garonzik S. M., et al. 2011. Population pharmacokinetics of colistin methanesulfonate and formed colistin in critically ill patients from a multicenter study provide dosing suggestions for various categories of patients. Antimicrob. Agents Chemother. 55:3284–3294 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Gilleland H. E., Jr., Champlin F. R., Conrad R. S. 1984. Chemical alterations in cell envelopes of Pseudomonas aeruginosa upon exposure to polymyxin: a possible mechanism to explain adaptive resistance to polymyxin. Can. J. Microbiol. 30:869–873 [DOI] [PubMed] [Google Scholar]
- 15. Gunderson B. W., et al. 2003. Synergistic activity of colistin and ceftazidime against multiantibiotic-resistant Pseudomonas aeruginosa in an in vitro pharmacodynamic model. Antimicrob. Agents Chemother. 47:905–909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hartzell J. D., et al. 2009. Nephrotoxicity associated with intravenous colistin (colistimethate sodium) treatment at a tertiary care medical center. Clin. Infect. Dis. 48:1724–1728 [DOI] [PubMed] [Google Scholar]
- 17. Hawley J. S., Murray C. K., Jorgensen J. H. 2008. Colistin heteroresistance in Acinetobacter and its association with previous colistin therapy. Antimicrob. Agents Chemother. 52:351–352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Imberti R., et al. 2010. Steady-state pharmacokinetics and BAL concentration of colistin in critically Ill patients after IV colistin methanesulfonate administration. Chest 138:1333–1339 [DOI] [PubMed] [Google Scholar]
- 19. Jones R. N. 2001. Resistance patterns among nosocomial pathogens: trends over the past few years. Chest 119:397S–404S [DOI] [PubMed] [Google Scholar]
- 20. Keel R. A., Sutherland C. A., Crandon J. L., Nicolau D. P. 2011. Stability of doripenem, imipenem and meropenem at elevated room temperatures. Int. J. Antimicrob. Agents 37:184–185 [DOI] [PubMed] [Google Scholar]
- 21. Ketthireddy S., et al. 2007. In vivo pharmacodynamics of colistin against Pseudomonas aeruginosa in thighs of neutropenic mice, abstr. A-4, p. 1. Abstr. 47th Intersci. Conf. Antimicrob. Agents Chemother., Chicago, IL, 17 to 20 September 2007 American Society for Microbiology, Washington, DC [Google Scholar]
- 22. Kwon J. A., et al. 2010. Predictors of acute kidney injury associated with intravenous colistin treatment. Int. J. Antimicrob. Agents 35:473–477 [DOI] [PubMed] [Google Scholar]
- 23. Landman D., Georgescu C., Martin D. A., Quale J. 2008. Polymyxins revisited. Clin. Microbiol. Rev. 21:449–465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Li J., et al. 2003. Steady-state pharmacokinetics of intravenous colistin methanesulphonate in patients with cystic fibrosis. J. Antimicrob. Chemother. 52:987–992 [DOI] [PubMed] [Google Scholar]
- 25. Li J., et al. 2003. Use of high-performance liquid chromatography to study the pharmacokinetics of colistin sulfate in rats following intravenous administration. Antimicrob. Agents Chemother. 47:1766–1770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Li J., Nation R. L., Milne R. W., Turnidge J. D., Coulthard K. 2005. Evaluation of colistin as an agent against multi-resistant Gram-negative bacteria. Int. J. Antimicrob. Agents 25:11–25 [DOI] [PubMed] [Google Scholar]
- 27. Li J., et al. 2006. Colistin: the re-emerging antibiotic for multidrug-resistant Gram-negative bacterial infections. Lancet Infect. Dis. 6:589–601 [DOI] [PubMed] [Google Scholar]
- 28. Li J., et al. 2005. Pharmacokinetics of colistin methanesulfonate and colistin in a critically ill patient receiving continuous venovenous hemodiafiltration. Antimicrob. Agents Chemother. 49:4814–4815 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Li J., et al. 2006. Heteroresistance to colistin in multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 50:2946–2950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Livermore D. M. 2004. The need for new antibiotics. Clin. Microbiol. Infect. 10(Suppl. 4):1–9 [DOI] [PubMed] [Google Scholar]
- 31. Livermore D. M. 2003. The threat from the pink corner. Ann. Med. 35:226–234 [DOI] [PubMed] [Google Scholar]
- 32. Markou N., et al. 2008. Colistin serum concentrations after intravenous administration in critically ill patients with serious multidrug-resistant, gram-negative bacilli infections: a prospective, open-label, uncontrolled study. Clin. Ther. 30:143–151 [DOI] [PubMed] [Google Scholar]
- 33. Matthaiou D. K., et al. 2008. Risk factors associated with the isolation of colistin-resistant gram-negative bacteria: a matched case-control study. Crit. Care Med. 36:807–811 [DOI] [PubMed] [Google Scholar]
- 34. Meletis G., Tzampaz E., Sianou E., Tzavaras I., Sofianou D. 2011. Colistin heteroresistance in carbapenemase-producing Klebsiella pneumoniae. J. Antimicrob. Chemother. 66:946–947 [DOI] [PubMed] [Google Scholar]
- 35. Michalopoulos A. S., Karatza D. C. 2010. Multidrug-resistant Gram-negative infections: the use of colistin. Expert Rev. Anti Infect. Ther. 8:1009–1017 [DOI] [PubMed] [Google Scholar]
- 36. Mizunaga S., Kamiyama T., Fukuda Y., Takahata M., Mitsuyama J. 2005. Influence of inoculum size of Staphylococcus aureus and Pseudomonas aeruginosa on in vitro activities and in vivo efficacy of fluoroquinolones and carbapenems. J. Antimicrob. Chemother. 56:91–96 [DOI] [PubMed] [Google Scholar]
- 37. Nicolau D. P. 2008. Carbapenems: a potent class of antibiotics. Expert Opin. Pharmacother 9:23–37 [DOI] [PubMed] [Google Scholar]
- 38. Pankuch G. A., Lin G., Seifert H., Appelbaum P. C. 2008. Activity of meropenem with and without ciprofloxacin and colistin against Pseudomonas aeruginosa and Acinetobacter baumannii. Antimicrob. Agents Chemother. 52:333–336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Pankuch G. A., Seifert H., Appelbaum P. C. 2010. Activity of doripenem with and without levofloxacin, amikacin, and colistin against Pseudomonas aeruginosa and Acinetobacter baumannii. Diagn. Microbiol. Infect. Dis. 67:191–197 [DOI] [PubMed] [Google Scholar]
- 40. Paterson D. L. 2006. The epidemiological profile of infections with multidrug-resistant Pseudomonas aeruginosa and Acinetobacter species. Clin. Infect. Dis. 43(Suppl. 2):S43–S48 [DOI] [PubMed] [Google Scholar]
- 41. Payne D. J., Gwynn M. N., Holmes D. J., Pompliano D. L. 2007. Drugs for bad bugs: confronting the challenges of antibacterial discovery. Nat. Rev. Drug Discov. 6:29–40 [DOI] [PubMed] [Google Scholar]
- 42. Pillai S. K., Moellering R. C., Eliopoulos G. M. 2005. Antimicrobial combinations, p. 365–400 In Lorian V. (ed.), Antibiotics in laboratory medicine, 5th ed. Lippincott Williams & Wilkins, Philadelphia, PA [Google Scholar]
- 43. Plachouras D., et al. 2009. Population pharmacokinetic analysis of colistin methanesulfonate and colistin after intravenous administration in critically ill patients with infections caused by gram-negative bacteria. Antimicrob. Agents Chemother. 53:3430–3436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Poudyal A., et al. 2008. In vitro pharmacodynamics of colistin against multidrug-resistant Klebsiella pneumoniae. J. Antimicrob. Chemother. 62:1311–1318 [DOI] [PubMed] [Google Scholar]
- 45. Richards M. J., Edwards J. R., Culver D. H., Gaynes R. P. 1999. Nosocomial infections in medical intensive care units in the United States. National Nosocomial Infections Surveillance System. Crit. Care Med. 27:887–892 [DOI] [PubMed] [Google Scholar]
- 46. Rynn C., Wootton M., Bowker K. E., Alan Holt H., Reeves D. S. 1999. In vitro assessment of colistin's antipseudomonal antimicrobial interactions with other antibiotics. Clin. Microbiol. Infect. 5:32–36 [DOI] [PubMed] [Google Scholar]
- 47. Sidjabat H., et al. 2011. Carbapenem resistance in Klebsiella pneumoniae due to the New Delhi metallo-beta-lactamase. Clin. Infect. Dis. 52:481–484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Spencer R. C. 1996. Predominant pathogens found in the European Prevalence of Infection in Intensive Care Study. Eur. J. Clin. Microbiol. Infect. Dis. 15:281–285 [DOI] [PubMed] [Google Scholar]
- 49. Standiford H. C., et al. 1986. Imipenem coadministered with cilastatin compared with moxalactam: integration of serum pharmacokinetics and microbiologic activity following single-dose administration to normal volunteers. Antimicrob. Agents Chemother. 29:412–417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Talbot G. H., et al. 2006. Bad bugs need drugs: an update on the development pipeline from the Antimicrobial Availability Task Force of the Infectious Diseases Society of America. Clin. Infect. Dis. 42:657–668 [DOI] [PubMed] [Google Scholar]
- 51. Tam V. H., et al. 2005. Pharmacodynamics of polymyxin B against Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 49:3624–3630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Tan C. H., Li J., Nation R. L. 2007. Activity of colistin against heteroresistant Acinetobacter baumannii and emergence of resistance in an in vitro pharmacokinetic/pharmacodynamic model. Antimicrob. Agents Chemother. 51:3413–3415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Tenover F. C., et al. 1995. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J. Clin. Microbiol. 33:2233–2239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Turnidge J. D., Bell J. M., Jones R. N. 2007. Emergence of colistin-resistant Klebsiella spp. and Enterobacter spp. in the Asia-Pacific region: a SENTRY antimicrobial surveillance program report, abstr. C2-2054, p. 148. Abstr. 47th Intersci. Conf. Antimicrob. Agents Chemother., Chicago, IL, 17 to 20 September 2007 American Society for Microbiology, Washington, DC [Google Scholar]
- 55. Yang Y., Bhachech N., Bush K. 1995. Biochemical comparison of imipenem, meropenem and biapenem: permeability, binding to penicillin-binding proteins, and stability to hydrolysis by beta-lactamases. J. Antimicrob. Chemother. 35:75–84 [DOI] [PubMed] [Google Scholar]
- 56. Yau W., et al. 2009. Colistin hetero-resistance in multidrug-resistant Acinetobacter baumannii clinical isolates from the Western Pacific region in the SENTRY antimicrobial surveillance programme. J. Infect. 58:138–144 [DOI] [PubMed] [Google Scholar]
- 57. Zhang L., Dhillon P., Yan H., Farmer S., Hancock R. E. 2000. Interactions of bacterial cationic peptide antibiotics with outer and cytoplasmic membranes of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 44:3317–3321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Zhao W. H., Hu Z. Q. 2010. Beta-lactamases identified in clinical isolates of Pseudomonas aeruginosa. Crit. Rev. Microbiol. 36:245–258 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.