Abstract
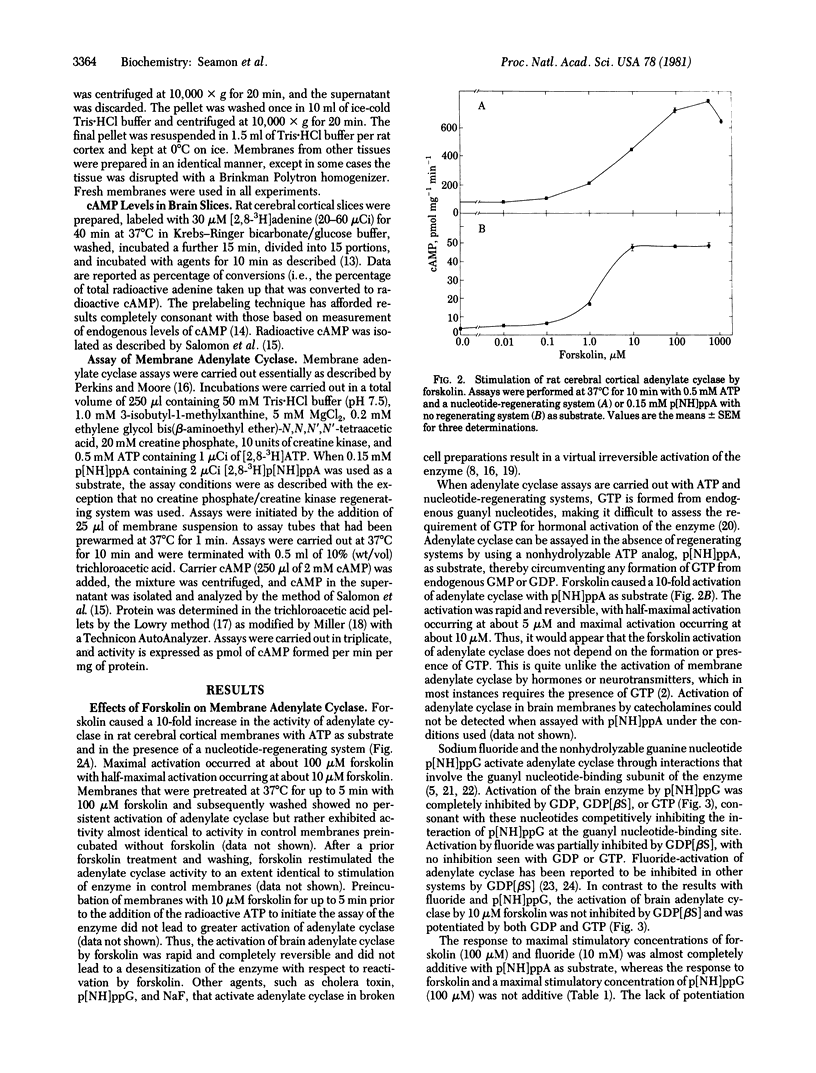
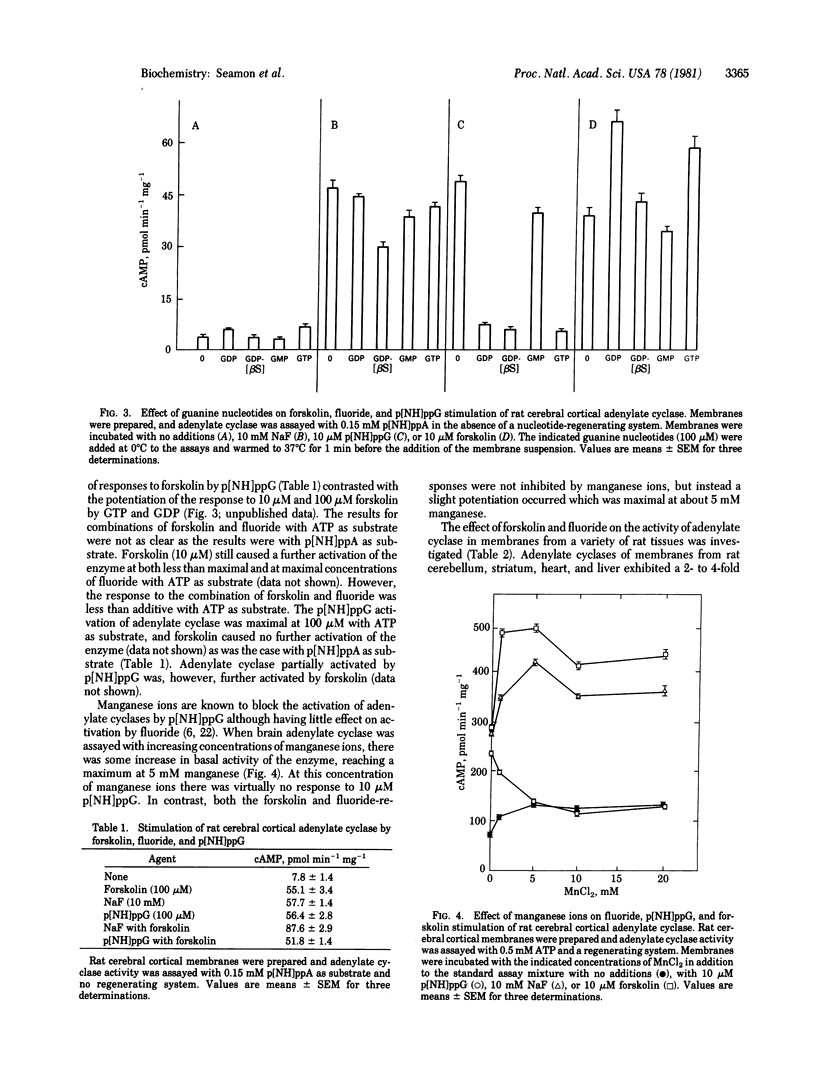
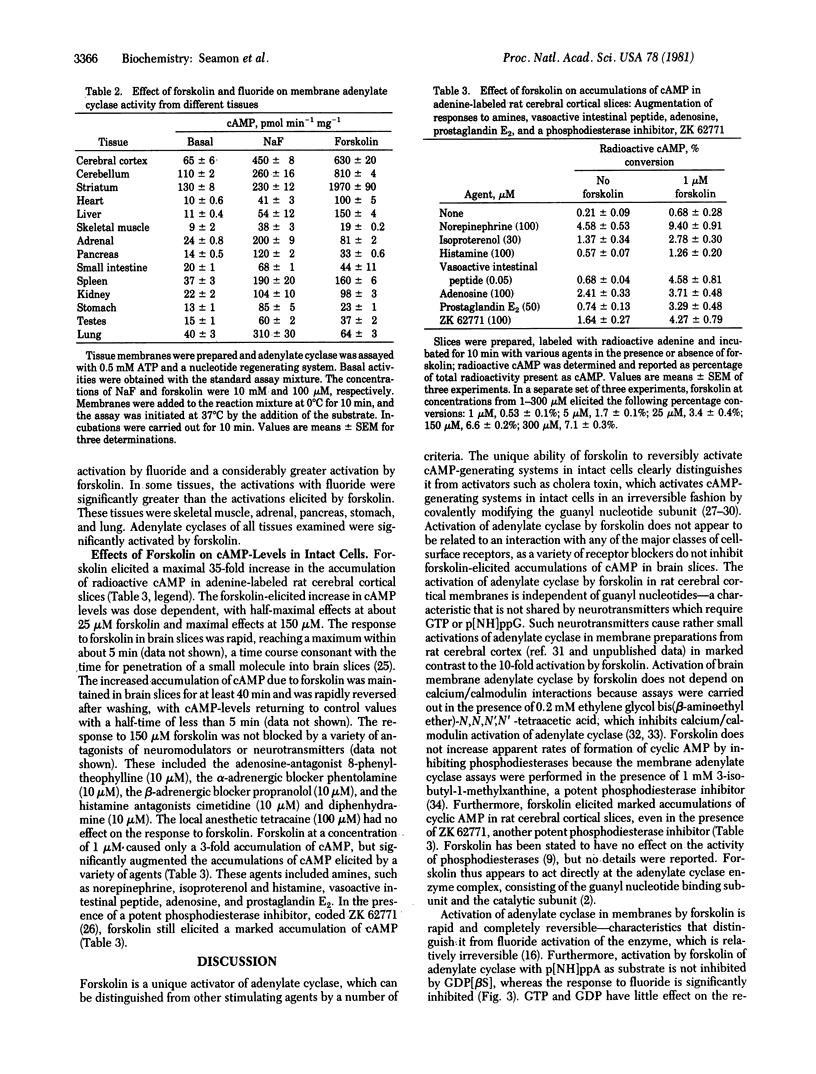
The diterpene, forskolin [half-maximal effective concentration (EC50), 5-10 microM] activates adenylate cyclase [ATP pyrophosphate-lyase (cyclizing), EC 4.6.1.1] in rat cerebral cortical membranes in a rapid and reversible manner. Activation is not dependent on exogenous guanyl nucleotides and is not inhibited by guanosine 5'-O-(2-thiodiphosphate) when assayed with adenosine 5'-[beta, gamma-imido]triphosphate as substrate. GTP and GDP potentiate responses to forskolin. The activations of adenylate cyclase by forskolin and guanosine 5'-[beta, gamma-imido]triphosphate p[NH]ppG are not additive, whereas activations by forskolin and fluoride are additive or partially additive. The responses of adenylate cyclase to forskolin or fluoride are not inhibited by manganese ions, whereas the response to p[NH]ppG is completely blocked. Activation of adenylate cyclase by forskolin is considerably greater than the activation by fluoride in membranes from rat cerebellum, striatum, heart, and liver, while being about equal or less than the activation by fluoride in other tissues. Forskolin (EC50, 25 microM) causes a rapid and readily reversible 35-fold elevation of cyclic AMP in rat cerebral cortical slices that is not blocked by a variety of neurotransmitter antagonists. Low concentrations of forskolin (1 microM) augment the response of cyclic AMP-generating systems in brain slices to norepinephrine, isoproterenol, histamine, adenosine, prostaglandin E2, and vasoactive intestinal peptide. Forskolin would appear to activate adenylate cyclase through a unique mechanism involving both direct activation of the enzyme and facilitation or potentiation of the modulation of enzyme activity by receptors or the guanyl nucleotide-binding subunit, or both.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradham L. S., Holt D. A., Sims M. The effect of Ca2+ on the adenyl cyclase of calf brain. Biochim Biophys Acta. 1970 Feb 24;201(2):250–260. doi: 10.1016/0304-4165(70)90299-0. [DOI] [PubMed] [Google Scholar]
- Brostrom C. O., Huang Y. C., Breckenridge B. M., Wolff D. J. Identification of a calcium-binding protein as a calcium-dependent regulator of brain adenylate cyclase. Proc Natl Acad Sci U S A. 1975 Jan;72(1):64–68. doi: 10.1073/pnas.72.1.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassel D., Pfeuffer T. Mechanism of cholera toxin action: covalent modification of the guanyl nucleotide-binding protein of the adenylate cyclase system. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2669–2673. doi: 10.1073/pnas.75.6.2669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassel D., Selinger Z. Mechanism of adenylate cyclase activation by cholera toxin: inhibition of GTP hydrolysis at the regulatory site. Proc Natl Acad Sci U S A. 1977 Aug;74(8):3307–3311. doi: 10.1073/pnas.74.8.3307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheung W. Y., Bradham L. S., Lynch T. J., Lin Y. M., Tallant E. A. Protein activator of cyclic 3':5'-nucleotide phosphodiesterase of bovine or rat brain also activates its adenylate cyclase. Biochem Biophys Res Commun. 1975 Oct 6;66(3):1055–1062. doi: 10.1016/0006-291x(75)90747-0. [DOI] [PubMed] [Google Scholar]
- Clark R. B. Endogenous GTP and the regulation of epinephrine stimulation of adenylate cyclase. J Cyclic Nucleotide Res. 1978 Apr;4(2):71–85. [PubMed] [Google Scholar]
- Downs R. W., Jr, Spiegel A. M., Singer M., Reen S., Aurbach G. D. Fluoride stimulation of adenylate cyclase is dependent on the guanine nucleotide regulatory protein. J Biol Chem. 1980 Feb 10;255(3):949–954. [PubMed] [Google Scholar]
- Eckstein F., Cassel D., Levkovitz H., Lowe M., Selinger Z. Guanosine 5'-O-(2-thiodiphosphate). An inhibitor of adenylate cyclase stimulation by guanine nucleotides and fluoride ions. J Biol Chem. 1979 Oct 10;254(19):9829–9834. [PubMed] [Google Scholar]
- Gill D. M., Meren R. ADP-ribosylation of membrane proteins catalyzed by cholera toxin: basis of the activation of adenylate cyclase. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3050–3054. doi: 10.1073/pnas.75.7.3050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howlett A. C., Sternweis P. C., Macik B. A., Van Arsdale P. M., Gilman A. G. Reconstitution of catecholamine-sensitive adenylate cyclase. Association of a regulatory component of the enzyme with membranes containing the catalytic protein and beta-adrenergic receptors. J Biol Chem. 1979 Apr 10;254(7):2287–2295. [PubMed] [Google Scholar]
- Johnson G. L., Kaslow H. R., Bourne H. R. Reconstitution of cholera toxin-activated adenylate cyclase. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3113–3117. doi: 10.1073/pnas.75.7.3113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Limbird L. E., Hickey A. R., Lefkowitz R. J. Unique uncoupling of the frog erythrocyte adenylate cyclase system by manganese. Loss of hormone and guanine nucleotide-sensitive enzyme activities without loss of nucleotide-sensitive, high affinity agonist binding. J Biol Chem. 1979 Apr 25;254(8):2677–2683. [PubMed] [Google Scholar]
- Lindner E., Dohadwalla A. N., Bhattacharya B. K. Positive inotropic and blood pressure lowering activity of a diterpene derivative isolated from Coleus forskohli: Forskolin. Arzneimittelforschung. 1978;28(2):284–289. [PubMed] [Google Scholar]
- Londos C., Lad P. M., Nielsen T. B., Rodbell M. Solubilization and conversion of hepatic adenylate cyclase to a form requiring MnATP as substrate. J Supramol Struct. 1979;10(1):31–37. doi: 10.1002/jss.400100104. [DOI] [PubMed] [Google Scholar]
- Londos C., Salomon Y., Lin M. C., Harwood J. P., Schramm M., Wolff J., Rodbell M. 5'-Guanylylimidodiphosphate, a potent activator of adenylate cyclase systems in eukaryotic cells. Proc Natl Acad Sci U S A. 1974 Aug;71(8):3087–3090. doi: 10.1073/pnas.71.8.3087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss J., Vaughan M. Activation of adenylate cyclase by choleragen. Annu Rev Biochem. 1979;48:581–600. doi: 10.1146/annurev.bi.48.070179.003053. [DOI] [PubMed] [Google Scholar]
- Neer E. J. Physical and functional properties of adenylate cyclase from mature rat testis. J Biol Chem. 1978 Aug 25;253(16):5808–5812. [PubMed] [Google Scholar]
- Partington C. R., Daly J. W. Effect of gangliosides on adenylate cyclase activity in rat cerebral cortical membranes. Mol Pharmacol. 1979 May;15(3):484–491. [PubMed] [Google Scholar]
- Perkins J. P. Adenyl cyclase. Adv Cyclic Nucleotide Res. 1973;3:1–64. [PubMed] [Google Scholar]
- Perkins J. P., Moore M. M. Adenyl cyclase of rat cerebral cortex. Activation of sodium fluoride and detergents. J Biol Chem. 1971 Jan 10;246(1):62–68. [PubMed] [Google Scholar]
- Rodbell M. The role of hormone receptors and GTP-regulatory proteins in membrane transduction. Nature. 1980 Mar 6;284(5751):17–22. doi: 10.1038/284017a0. [DOI] [PubMed] [Google Scholar]
- Salomon Y., Londos C., Rodbell M. A highly sensitive adenylate cyclase assay. Anal Biochem. 1974 Apr;58(2):541–548. doi: 10.1016/0003-2697(74)90222-x. [DOI] [PubMed] [Google Scholar]
- Schramm M., Rodbell M. A persistent active state of the adenylate cyclase system produced by the combined actions of isoproterenol and guanylyl imidodiphosphate in frog erythrocyte membranes. J Biol Chem. 1975 Mar 25;250(6):2232–2237. [PubMed] [Google Scholar]
- Schultz J., Daly J. W. Cyclic adenosine 3',5'-monophosphate in guinea pig cerebral cortical slices. I. Formation of cyclic adenosine 3',5'-monophosphate from endogenous adenosine triphosphate and from radioactive adenosine triphosphate formed during a prior incubation with radioactive adenine. J Biol Chem. 1973 Feb 10;248(3):843–852. [PubMed] [Google Scholar]
- Schwabe U., Miyake M., Ohga Y., Daly J. W. 4-(3-Cyclopentyloxy-4-methoxyphenyl)-2-pyrrolidone (ZK 62711): a potent inhibitor of adenosine cyclic 3',5'-monophosphate phosphodiesterases in homogenates and tissue slices from rat brain. Mol Pharmacol. 1976 Nov;12(6):900–910. [PubMed] [Google Scholar]
- Smellie F. W., Davis C. W., Daly J. W., Wells J. N. Alkylxanthines: inhibition of adenosine-elicited accumulation of cyclic AMP in brain slices and of brain phosphodiesterase activity. Life Sci. 1979 Jun 25;24(26):2475–2482. doi: 10.1016/0024-3205(79)90458-2. [DOI] [PubMed] [Google Scholar]


