Abstract
Associations between marine seaweeds and bacteria are widespread, with endobiotic bacterial-algal interactions being described for over 40 years. Also within the siphonous marine green alga Bryopsis, intracellular bacteria have been visualized by electron microscopy in the early ‘70s, but were up to now never molecularly analyzed. To study this partnership, we examined the presence and phylogenetic diversity of microbial communities within the cytoplasm of two Bryopsis species by combining fluorescence in situ hybridization (FISH), denaturing gradient gel electrophoresis (DGGE) and 16S rRNA gene clone libraries. Sequencing results revealed the presence of Arcobacter, Bacteroidetes, Flavobacteriaceae, Mycoplasma, Labrenzia, Phyllobacteriaceae and Xanthomonadaceae species. Although the total diversity of the endobiotic communities was unique to each Bryopsis culture, Bacteroidetes, Mycoplasma, Phyllobacteriaceae, and in particular Flavobacteriaceae bacteria, were detected in several Bryopsis samples collected hundreds of kilometres apart. This suggests that Bryopsis closely associates with well-defined endophytic bacterial communities of which some members possibly maintain an endosymbiotic relationship with the algal host.
Introduction
Marine macroalgal-bacterial associations range from beneficial, harmful or neutral, over obligate or facultative, to ecto- or endophytic interactions [1]. Elaborating the latter, endobiotic associations between marine macroalgal hosts and bacteria have been reported over the past 40 years. Besides reports of bacterial endosymbionts associated with red algal galls [2]–[4], endophytic bacteria have been microscopically observed in the vacuolar as well as cytoplasmatic regions of various bryopsidalean green algae, including Bryopsis, Penicillus, Halimeda, Udotea and Caulerpa [5]–[10]. These seaweeds are composed of a single, giant tubular cell and form an interesting biotic environment for bacterial communities. The giant cell contains millions of nuclei and chloroplasts in a thin cytoplasmic layer surrounding a large central vacuole. The cytoplasm typically exhibits vigorous streaming, enabling transport of nutrients, organelles and various biomolecules across the plant [11]. In Bryopsis ‘bacteria-like particles’ have been visualized in the cytoplasm by means of transmission electron microscopy in vegetative thalli as well as in the gametes, the latter suggesting vertical transmission of the endophytic bacteria [5]. This implies a stable and specific relationship between the algal host and its endobionts in which both partners may provide mutualistic ecological benefits. To date, the diversity of the intracellular microbial communities associated with Bryopsis remains unidentified. Up till now investigations of the bacterial endophytic diversity of siphonous macroalgae have been limited to Caulerpa species and revealed endosymbiotic Alphaproteobacteria with the potential to photosynthesize, detoxify and/or fix nitrogen [9], [10]. The endophytic bacteria in Bryopsis may similarly possess ecologically significant functions and bioactive potential since Bryopsis is a substantial source of bioactive compounds such as therapeutic kahalalides which may be of bacterial origin [12], [13].
In order to explore these algal-endophytic bacterial interactions, we previously developed a surface sterilization protocol for the complete elimination of bacterial epiphytes from the Bryopsis surface [14]. We showed that Bryopsis samples treated with a combined chemical and enzymatic approach (i.e. a mixture of cetyltrimethylammonium bromide (CTAB) lysis buffer, proteinase K and the bactericidal cleanser Umonium Master) remained intact after sterilization and showed no remaining bacterial fluorescence on their surface when stained with a DNA fluorochrome. Successful 16S rRNA gene DGGE analysis following this surface sterilization treatment showed that endophytic DNA was still present within the sterilized Bryopsis samples, allowing specific molecular processing of the endophytes [14].
In this study, we verified the presence of bacteria inside two Bryopsis species from the Mexican west coast by a combination of fluorescence in situ hybridization (FISH), denaturing gradient gel electrophoresis (DGGE) and clone libraries.
Materials and Methods
Ethics Statement
No specific permits were required for the described field studies, i.e. the collection of algal samples from the Mexican west coast, because marine algae are not included in the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES, http://www.cites.org/eng/disc/species.shtml). The authors confirm that the location is not privately-owned or protected in any way and that the field studies did not involve endangered or protected species.
Algal material
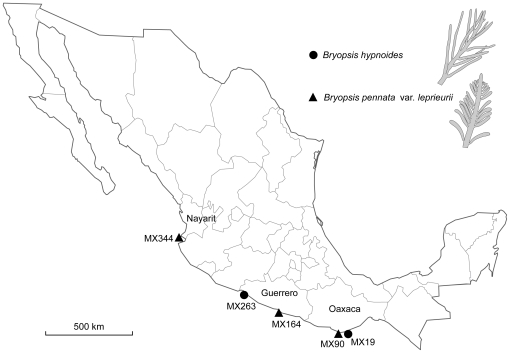
Five Bryopsis specimens were collected in February 2009 along the Pacific Mexican coast at different sites located between Mazunte Beach (Oaxaca, southwest Mexico) and Playa Careyero (Nayarit, central Mexico) (Figure 1). These five samples were classified in two different species with samples MX19 and MX263 representing Bryopsis hypnoides J.V. Lamouroux and MX90, MX164, and MX344 representing Bryopsis pennata J.V. Lamouroux var. leprieurii (Kützing) Collins and Hervey individuals. After sampling, living specimens were rinsed with sterile seawater and transferred to the laboratory in plastic vessels containing a small amount of sterile seawater. In the laboratory, clean apical fragments of the Bryopsis specimens were isolated and cultured in sterile 1 x modified Provasoli enriched seawater [15] at 23°C under 12h:12h (Light:Dark) conditions with a photon flux rate of 25–30 µE m−2s−1. This isolation procedure was repeated for several months until the Bryopsis cultures were free of eukaryotic contamination. Thus, the Bryopsis isolates were kept in culture for eight months prior to molecular analyses in October 2009. After isolation, all five unialgal Bryopsis cultures were maintained in the laboratory under the culture conditions described above.
Figure 1. Bryopsis sampling sites along the Pacific Mexican coast.
Bryopsis hypnoides (•) and Bryopsis pennata var. leprieurii (▴) samples were collected from following sites: Playa el Pantheon (MX19), Mazunte Beach (MX90), Acapulco (MX164), Playa las Gatas (MX263) and Playa Careyero (MX344).
Fluorescence in situ hybridization
Unialgal Bryopsis thalli were fixed in 4% paraformaldehyde and 0.25% glutaraldehyde in 50 mM PIPES (piperazine-N,N′-bis(2-ethanesulfonic acid)) buffer, pH 7.2 for 2 hours. After dehydration through a graded ethanol series from 30% to 80%, ethanol was subsequently replaced by LR white embedding medium (London Resin, UK). Samples were loaded in gelatine capsules and allowed to polymerize at 37°C for 3 days. Semithin sections were cut using glass knives on a Microm HM360 microtome (Microm International GmbH, Germany) and collected on Vectabond-coated (Vector Laboratories, USA) slides. In situ hybridization was performed as described by Daims et al. [16] with 200 µL formamide per mL hybridization buffer, an incubation of 90 min at 46°C, and the universal bacterial Cy3-labelled EUB338 probe mix [17]. Algal DNA and cell wall counterstaining was performed by adding a mix of 4,6-diamidino-2-phenylindole (DAPI) and calcofluor to the sections for 7 min in the dark at room temperature. Sections were mounted in AF-1 antifadent (Citifluor, UK) and viewed with an Olympus BX51 epifluorescence microscope fitted with a DAPI/FITC/TRITC triple band filter. The Bryopsis specimens were not surface-sterilized prior to hybridization due to potential morphological losses.
Surface sterilization, DNA extraction and PCR
To identify the endophytic bacterial diversity, approximately 2 grams (ww) of each unialgal Bryopsis sample was surface-sterilized as previously described [14] prior to a total DNA extraction using a CTAB protocol modified from Doyle and Doyle [18]. These extracts, containing both algal and bacterial DNA, were subjected to rbcL and 16S rRNA gene PCR amplifications following protocols outlined in Hanyuda et al. [19] and Lane [20] with, respectively, primer pairs 7F/R1391 and 27F/1492R. All obtained PCR amplicons were purified using a Nucleofast 96 PCR clean up membrane system (Machery-Nagel, Germany) according to the manufacturer's instructions.
Cloning and DGGE
To determine the bacterial diversity, purified 16S rRNA gene amplicons from the algal extracts were cloned using the pGEM®-T Vector System (Promega Benelux, The Netherlands). For each Bryopsis sample a clone library of 150 clones was prepared, the diversity of which was examined via short fragment sequencing (see below). For dereplication, clones' short sequences were grouped into the same operational taxonomic unit (OTU) when having ≥97% similarity. From each OTU, representative clones were selected for full length (±1450 bp) 16S rRNA gene sequencing (see below). Clone libraries' coverage was verified by DGGE analysis of each Bryopsis DNA extract and its representative clones. A V3 PCR with primers F357-GC/R518 and subsequent DGGE analysis were carried out as described previously [14], with a denaturing gradient of 45–65%. DGGE banding patterns were normalized using the BioNumerics 5.1 software (Applied Maths, Belgium). DGGE bands from the algal extracts which showed no correspondence with OTU band positions were excised from the polyacrylamide gel following Van Hoorde et al. [21] and sequenced (±150 bp) as described below.
Sequencing
RbcL genes, DGGE bands as well as short and full length 16S rRNA genes were sequenced on an ABI PRISM 3130xl Genetic Analyzer (Applied Biosystems, USA) by means of the BigDye® xTerminator™ v.3.1 Cycle Sequencing and Purification Kit (Applied Biosystems, USA) according the protocol of the supplier. Primers used were, respectively, 7F/R1391 [19], F357/R518 [21], BKL1 [22] and T7/SP6 (Promega Benelux, The Netherlands). Sequences obtained were assembled in BioNumerics, compared with nucleotide databases via BLAST and chimera-checked using Bellerophon [23]. The bacterial 16S rRNA gene and Bryopsis chloroplast 16S rRNA gene and rbcL sequences were submitted to GenBank under accession numbers JF521593-JF521615 (see also Table 1).
Table 1. Taxonomic affiliation of the clones representing the bacterial OTUs, sorted per Bryopsis sample.
| Host | 16S rRNA gene sequence analysis of bacterial clones | |||||||
| Bryopsis sample | Chloroplast 16S rRNA gene1 and rbcL accession no. | OTU no.2 | OTU representative clone name | Accession no. | OTU library %/sample3 | Higher taxonomic ranks | Three closest NCBI matches | Accession no. (Query coverage/Maximum identity) |
| MX19 | JF521612JF521594 | OTU-3 | MX19.8 | JF521598 | 0.8% | Bacteroidetes; unclassified Bacteroidetes | Uncultured bacterium clone Dstr_N15 | GU118164 (99/94) |
| Uncultured bacterium clone SGUS845 | FJ202831 (100/92) | |||||||
| Endosymbiont of Acanthamoeba sp. KA/E21 | EF140637 (100/91) | |||||||
| OTU-2 | MX19.9 | JF521606 | 14.2% | Tenericutes, Mollicutes, Mycoplasmatales, Mycoplasmataceae | Uncultured bacterium clone GB96 | GU070687 (100/97) | ||
| Uncultured bacterium clone frc89 | HQ393440 (100/93) | |||||||
| Uncultured bacterium isolate SRODG064 | FM995178 (100/90) | |||||||
| OTU-4 | MX19.12 | JF521607 | 3% | Proteobacteria; Alphaproteobacteria; Rhizobiales; Phyllobacteriaceae | Uncultured Rhizobiales bacterium clone PRTBB8661 | HM799061 (99/99) | ||
| Uncultured Rhizobiaceae bacterium clone TDNP_Wbc97_42_3_189 | FJ517108 (100/97) | |||||||
| Uncultured alpha proteobacterium clone D2F10 | EU753666 (100/97) | |||||||
| OTU-1 | MX19.14 | JF521603 | 2.3% | Bacteroidetes; Flavobacteria; Flavobacteriales | Uncultured bacterium clone SHFH601 | FJ203530 (99/96) | ||
| Uncultured Bacteroidetes bacterium clone CN77 | AM259925 (100/94) | |||||||
| Uncultured bacterium clone SINP825 | HM127741 (99/89) | |||||||
| MX90 | JF521615JF521597 | OTU-1 | MX90.40 | JF521602 | 6.5% | Bacteroidetes; Flavobacteria; Flavobacteriales | Uncultured bacterium clone SHFH601 | FJ203530 (99/96) |
| Uncultured Bacteroidetes bacterium clone CN77 | AM259925 (100/94) | |||||||
| Uncultured bacterium clone SINP825 | HM127741 (99/88) | |||||||
| MX164 | JF521611JF521593 | OTU-5 | MX164.9 | JF521609 | 63.6% | Proteobacteria; Gammaproteobacteria; Xanthomonadales; Xanthomonadaceae | Gamma proteobacterium strain OS-28 | EF612351 (100/94) |
| Uncultured Luteibacter sp. clone SMa210 | AM930508 (100/94) | |||||||
| “Luteibacter jiangsuensis” JW-64-1 | FJ848571 (100/93) | |||||||
| OTU-1 | MX164.14 | JF521600 | 7.1% | Bacteroidetes; Flavobacteria; Flavobacteriales | Uncultured bacterium clone SHFH601 | FJ203530 (99/96) | ||
| Uncultured Bacteroidetes bacterium clone CN77 | AM259925 (100/94) | |||||||
| Uncultured bacterium clone SINP825 | HM127741 (99/89) | |||||||
| OTU-6 | MX164.20 | JF521610 | 3.6% | Proteobacteria; Epsilonproteobacteria; Campylobacterales; Campylobacteraceae | Arcobacter marinus type strain CL-S1T | EU512920 (96/93) | ||
| “Arcobacter molluscorum” type strain CECT7696T | FR675874 (94/94) | |||||||
| Uncultured Arcobacter sp. clone bo13C09 | AY862492 (96/93) | |||||||
| OTU-4 | MX164.59 | JF521608 | 5% | Proteobacteria; Alphaproteobacteria; Rhizobiales; Phyllobacteriaceae | Phylobacteriaceae bacterium strain DG943 | AY258089 (97/99) | ||
| Uncultured bacterium clone Apal_F11 | GU118131 (99/98) | |||||||
| Uncultured bacterium clone MSB-2G6 | EF125460 (100/97) | |||||||
| MX263 | JF521613JF521595 | OTU-2 | MX263.1 | JF521605 | 22.6% | Tenericutes, Mollicutes, Mycoplasmatales, Mycoplasmataceae | Uncultured bacterium clone GB96 | GU070687 (100/97) |
| Uncultured bacterium clone frc89 | HQ393440 (100/93) | |||||||
| Uncultured bacterium isolate SRODG064 | FM995178 (100/90) | |||||||
| OTU-1 | MX263.61 | JF521604 | 4% | Bacteroidetes; Flavobacteria; Flavobacteriales | Uncultured bacterium clone SHFH601 | FJ203530 (99/96) | ||
| Uncultured Bacteroidetes bacterium clone CN77 | AM259925 (100/94) | |||||||
| Uncultured bacterium clone SINP825 | HM127741 (99/89) | |||||||
| OTU-3 | MX263.73 | JF521599 | 1.4% | Bacteroidetes; unclassified Bacteroidetes | Uncultured bacterium clone Dstr_N15 | GU118164 (99/94) | ||
| Uncultured bacterium clone SGUS845 | FJ202831 (100/92) | |||||||
| Endosymbiont of Acanthamoeba sp. KA/E21 | EF140637 (100/91) | |||||||
| MX344 | JF521614JF521596 | OTU-1 | MX344.2 | JF521601 | 2.2% | Bacteroidetes; Flavobacteria; Flavobacteriales | Uncultured bacterium clone SHFH601 | FJ203530 (99/96) |
| Uncultured Bacteroidetes bacterium clone CN77 | AM259925 (100/94) | |||||||
| Uncultured bacterium clone SINP825 | HM127741 (99/89) | |||||||
Chloroplast 16S rRNA gene sequences were derived from clones MX19.1, MX90.9, MX164.1, MX263.48 and MX344.10 with an OTU library percentage of, respectively, 79.7, 93.5, 20.7, 68 and 97.8 percent per sample.
All bacterial OTUs containing clones derived from different Bryopsis strains had minimal intra-OTU sequence similarities of ≥97% ranging from exactly 97% in OTU-4, over 99.3% and 99.7% in, respectively, OTU-2 and OTU-1, to no less than 99.9% pairwise similarity in OTU-3.
Especially noteworthy is the abundance of OTU-5 in the MX164 sample's clone library. While the bacterial OTUs 1, 3, 4 and 6 have a low occurrence of 0.8–7.1% and OTU-2 a considerable presence of 14.2–22.6% in their respective clone libraries, OTU-5 amounts to a substantial percentage (63.6%) of the MX164 sample's clones. In addition, only Bryopsis sample MX263 comprised chimeric Flavobacteriaceae-Bryopsis chloroplast 16S rRNA gene sequences which made up 4% of the sample's clone library.
Phylogenetic analyses
Two sets of alignments, made using MUSCLE [24], were considered for phylogenetic analyses. The first one, consisting of a concatenated chloroplast 16S rRNA gene and rbcL dataset, was used for the creation of a Bryopsis phylogram. A second set of alignments was assembled to assess 16S rRNA gene phylogenetic relationships between the Bryopsis-associated bacterial endophytes and known bacterial species, including BLAST hits and algae-associated bacteria described in the literature. The most suitable model for phylogenetic analysis was selected using the AIC criterion in jModelTest [25]. Subsequently, the Bryopsis host and bacterial datasets were analyzed by means of the maximum likelihood (ML) algorithm in PhyML v3.0 [26] under a HKY + G4 model via the University of Oslo Bioportal website (http://www.bioportal.uio.no//). Reliability of ML trees was evaluated based on 100 bootstrap replicates. Output ML trees were subsequently visualized in Mega 4.0 [27] and edited with Adobe® Illustrator® CS5.
Results
Fluorescence in situ hybridization
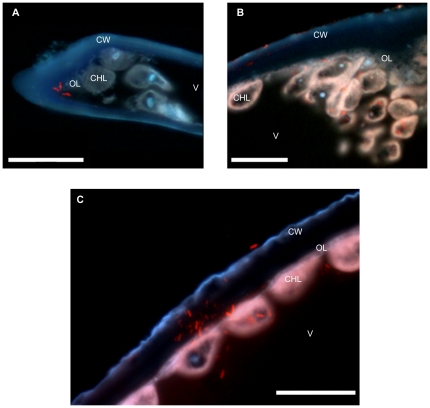
To confirm the observation of endogenous bacteria in Bryopsis made by Burr and West [5], Bryopsis sections were hybridized with the universal bacterial EUB338 probe mix labelled with Cy3. Figures 2A–C depict clear binding of the red fluorescent probe mix to bacterial rRNA present throughout the cytoplasm; both in the outer layer next to the cell wall, which contains most of the organelles except the chloroplasts (Figures 2A–C), as well as in the inner chloroplast layer immediately adjacent to the vacuole (Figures 2B–C). These hybridization results demonstrate the presence of metabolically active bacteria within the Bryopsis cytoplasm. Since the Bryopsis thalli were not surface sterilized before fixation, the EUB338 probe mix also hybridized with epiphytic bacterial rRNA on the cell wall (Figures 2B–C).
Figure 2. Epifluorescence microscopy images of Bryopsis sections hybridized with the universal bacterial Cy3-EUB338 probe mix (red).
DAPI (light blue) and calcofluor (dark blue) were used as counter stains to visualize algal DNA in nuclei and chloroplasts and the algal cell wall, respectively. Metabolically active bacteria (red) are present throughout the Bryopsis cytoplasm: in the outer layer (OL) next to the cell wall (CW) which contains most of the organelles like mitochondria, endoplasmic reticulum, and nuclei (A–C), and in the inner chloroplast layer (CHL) immediately adjacent to the vacuole (V) (B–C). Since the Bryopsis thalli were not surface sterilized before fixation, the red probe also hybridized with epiphytic bacteria on the calcofluor stained cell wall (B–C). The scale bar on all images is 20 µm.
Bacterial diversity within Bryopsis algae: Cloning
Five clone libraries were created using the amplified 16S rRNA gene fragments from samples MX19, MX90, MX164, MX263 and MX344. After clone dereplication, 16S rRNA gene sequences from all five clone libraries covered no more than seven unique OTUs. By far the most common OTU, representing 72% of the total clones screened, showed ≥96% sequence similarity with the B. hypnoides chloroplast 16S ribosomal RNA gene (AY221722). The six remaining OTUs, on the other hand, contained bacterial sequences belonging to the phyla Bacteroidetes, Proteobacteria or Tenericutes (Table 1). OTU-1 was detected in all five Bryopsis cultures and had 96% sequence similarity with an uncultured Flavobacteriales bacterium (FJ203530) associated with the coral Montastraea faveolata. OTU-2 and 3 were only present in the B. hypnoides samples. OTU-2 is related to uncultured Mycoplasmataceae bacteria isolated from the intestine of the small abalone Haliotis diversicolor (GU070687, HQ393440). OTU-3 is allied to unclassified Bacteroidetes bacteria associated with corals (GU118164, FJ202831) or Acanthamoeba species (EF140637). OTU-4 sequences were detected in cultures MX19 and MX164, and showed high similarity (≥97%) with Phyllobacteriaceae bacteria isolated from seawater (HM799061, FJ517108), dinoflagellates (AY258089), stromatolites (EU75366) or corals (GU118131). OTU-5 and 6 were only present in B. pennata var. leprieurii sample MX164 and are distantly related (93–94%) to, respectively, Luteibacter sp. (Xanthomonadaceae) present in soil (EF612351, AM930508, FJ848571) and Arcobacter strains (Campylobacteraceae) recovered from mussels (FR675874) and seawater surrounding seaweeds and starfish (EU512920).
Bacterial diversity within Bryopsis algae: DGGE
Coverage of the clone libraries was verified by comparing DGGE community profiles of the different Bryopsis DNA extracts with the banding pattern of clones from their respective OTUs, including representative clones with 16S rRNA gene chloroplast and chimeric sequences. As shown in Figure 3 the OTUs DGGE bands overlap well with the individual bands of the MX extracts' DGGE profiles, indicating adequate clone library coverage. MX samples 19, 164 and 344, however, all showed one band in their DGGE profile not represented by an OTU band. Consequently, these three DGGE bands (A, B and C, respectively) were excised and sequenced. The sequence of DGGE band A showed 100% similarity with the chimeric sequences detected in MX sample 263, not unexpected given its corresponding band position with clone MX263.66. DGGE band B was identified as forming part of the OTU-2 cluster with 100% sequence similarity with clone MX19.9, whereas DGGE band C showed no correspondence with any bacterial OTU detected. Hence, the latter DGGE band was assigned to a new OTU, i.e. OTU-7. BLAST searches revealed that this OTU-7 is closely related to Labrenzia species isolated from the green seaweed Ulva rigida (FN811315), crustose coralline red algae (HM178529) and the dinoflagellate Karlodinium micrum (HM584720).
Figure 3. Normalized DGGE profiles of MX DNA extracts and their representative OTUs.
DGGE bands marked with letters A, B and C, which did not match any of the individual OTU bands, were excised from the polyacrylamide gel and sequenced. The first and last lanes contain a known molecular marker used for normalization.
Figure 4 depicts the endophytic diversity results from the clone libraries and DGGE analyses plotted on a phylogram representing the relations between the five Bryopsis samples. From Figure 4 we can deduce that Flavobacteriaceae (OTU-1), Mycoplasma (OTU-2), Bacteroidetes (OTU-3) and Phyllobacteriaceae (OTU-4) species were present in more than one Bryopsis sample examined. Even though the endobiotic community members were to a certain extent similar, the total diversity of the endophytic community was unique to each Bryopsis sample. None of the Bryopsis samples harbored the same number or range of bacterial endophytes.
Figure 4. Endophytic diversity results (right) plotted against the Bryopsis host phylogeny (left).
The OTU diversity (1–7) displayed on the right summarizes the diversity results from the clone libraries and DGGE analyses. The concatenated chloroplast 16S rRNA gene - rbcL maximum likelihood tree on the left classifies the Bryopsis MX samples in two distinct species clades with 100% bootstrap support. The scale bar indicates 0.002 nucleotide changes per nucleotide position.
Bacterial diversity within Bryopsis algae: Phylogenetic analysis
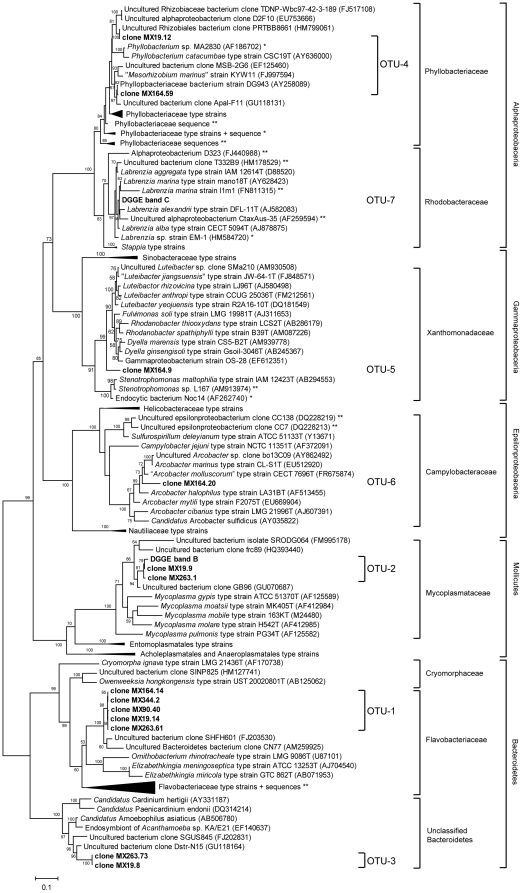
A wide-range phylogenetic tree (Figure 5, tree without compressed branches see Figure S1) was created, which includes bacterial OTUs determined in this study (clones and DGGE bands), significant BLAST hits (Table 1), type strains from the Bacteroidetes, Proteobacteria and Tenericutes division, and algae-associated bacteria described in the literature (Table S1). As could be predicted from the BLAST maximum identity scores (Table 1), none of the endobiotic bacterial sequences clustered tightly with cultivated bacterial type strains. Consequently, all endophytic bacterial OTUs derived from Bryopsis represent new species or genera which in some cases match previously sequenced unclassified bacteria. These OTU sequences, however, all showed at least 93% sequence similarity with their best BLAST hit which generally resulted in phylogenetic placements with good bootstrap support. Accordingly, all OTU-1 sequences formed a distinct and well-supported (98%) clade within the Flavobacteriaceae family and most likely represent a new genus given their low sequence similarities (87% at most) with Flavobacteriaceae type strains. The similarity among the five OTU-1 sequences, however, was 99.7%, suggesting all sequences belong to the same new Flavobacteriaceae genus even though they were derived from different Bryopsis samples collected several hundred kilometres apart. Likewise, the Bacteroidetes OTU-3 clones were virtually identical displaying 99.9% pairwise similarity. These OTU-3 clones, found in B. hypnoides samples MX19 and MX263, belong to a single clade (100% bootstrap support) of unclassified Bacteroidetes, but are distantly related to other unclassified Bacteroidetes symbionts. The OTU-2 clade, consisting of clones MX19.9 and MX263.1 and DGGE band B, fell into the genus Mycoplasma with 100% bootstrap support although these clones showed low levels of similarity (≤90%) with Mycoplasma type strains. All OTU-2 sequences presumably belong to one and the same new Mycoplasma species (99.7 intra-OTU sequence similarity). The majority of the endophytic bacterial OTUs, however, were affiliated with the Proteobacteria phylum and belonged to the Alpha-, Gamma- and Epsilonproteobacteria. Particularly, OTU-5 and 6, both consisting of clones exclusively obtained from B. pennata var. leprieurii sample MX164, most probably represent a new genus of Xanthomonadaceae and a new Arcobacter species, respectively. OTU-4 and 7 are robustly affiliated (100% bootstrap support) with the Alphaproteobacteria class and belong to the Rhizobiales and Rhodobacterales, respectively. Despite the high sequence similarity of OTU-7 with algal-associated Labrenzia species, relatedness of DGGE band C with the Labrenzia alexandrii type strain (AJ582083) and an uncultured Labrenzia bacterium isolated from Caulerpa taxifolia (AF259594) lacks bootstrap support. The shortness of the DGGE band C sequence (±150 bp) and, consequently, the poor resolution within this clade, made it difficult to conclude whether OTU-7 represents a new Labrenzia species. Finally, OTU-4 is the only OTU containing clones derived from different Bryopsis samples in which the representative clones, i.e. clone MX19.12 and MX164.59, did not cluster together. This is in agreement with the 97% intra-OTU sequence similarity. Hence, both clones belong to the Phyllobacteriaceae clade with good bootstrap support (80%), but most likely represent two different new species or genera because of their low sequence similarities (96% at most) with Phyllobacteriaceae type strains.
Figure 5. A wide-range maximum likelihood tree showing the phylogenetic positions of endophytic clones and DGGE bands.
Phylogenies were inferred from 16S rRNA gene sequences determined in this study (in bold), BLAST hits (see Table 1), Bacteroidetes, Proteobacteria and Mollicutes type strains, and algae-associated bacteria described in the literature (see Table S1). The tree was generated in PhyML according the HKY + G4 algorithmic model. Bootstrap values above 50% are indicated at the branch nodes and the scale bar shows 10 nucleotide substitutions per 100 nucleotides. Asterisks denote sequences previously isolated from micro * - and macroalgae**. The same phylogenetic tree without compressed branches is depicted in Figure S1.
Discussion
Forty years after Burr and West [5] observed endogenous ‘bacteria-like particles’ in Bryopsis hypnoides, this is the first study to verify the presence of metabolically active endophytic bacteria inside the Bryopsis cytoplasm by means of the FISH technique. Mainly due to the intense background autofluorescence of algal cells, reports of successful FISH applications on macroalgae are limited to analyses of macroalgal surface-associated bacteria [28] and algal gall endosymbionts [4]. The use in this study of semithin algal sections and a triple band filter, however, made it possible to discriminate bacterial FISH signals from autofluorescence of algal pigments using standard FISH protocols in combination with epifluorescence microscopy. Even though Bryopsis samples were not surface-sterilized prior to hybridization to avoid potential morphological losses, the solid embedding at the start of the FISH protocol proved successful in immobilizing the epiphytes on the Bryopsis surface (data not shown). This prevented the detachment and potential spread of surface bacteria during sectioning. Consequently, our FISH results strongly suggest the presence of bacteria within Bryopsis cells.
In this study, the first insights are provided into the identity and phylogenetic diversity of endobiotic bacterial communities within Bryopsis. Despite the limited number of samples studied, our results indicate that Bryopsis harbors endophytic bacterial communities which are not very complex (i.e. only 7 bacterial OTUs detected), but taxonomically diverse including Arcobacter, Bacteroidetes, Flavobacteriaceae, Mycoplasma, Labrenzia, Phyllobacteriaceae and Xanthomonadaceae species. Although the composition of the total endophytic community seems unique to each Bryopsis culture, Bacteroidetes, Flavobacteriaceae, Mycoplasma and Phyllobacteriaceae species were detected in two or more Bryopsis samples. In particular OTU-1 Flavobacteriaceae species are present in all five Bryopsis cultures, which were collected from diverse sites along the Mexican west coast. Delbridge and colleagues [10] made similar observations when comparing the endosymbiotic communities within four different Caulerpa species. While the endosymbiotic communities seemed unique to each Caulerpa individual, all community members were photosynthetic Alphaproteobacteria.
Also within Bryopsis, Alphaproteobacteria appear well represented. This is not unexpected, since Alphaproteobacteria are frequently associated with macroalgae [1], [29], [30], an alliance which may be linked to dimethylsulfoniopropionate (DMSP) exchange [31]. Particularly OTU-7, belonging to the marine phototrophic and CO-oxidizing Labrenzia genus [32], [33], is closely related to an uncultured bacterium reported by Meusnier et al. [29] in their study on the total bacterial community associated with Caulerpa taxifolia. Although Labrenzia species have not been reported as endophytes, the presence of Rhizobiales-specific proteins in L. aggregata [34] may hint at potential endosymbiotic features. The Rhizobiales order contains various well-known nitrogen fixing plant symbionts, mainly in terrestrial habitats but also in marine environments [35]. Moreover, Rhizobiales bacteria are common epiphytes on green [31], [36], brown [37], [38] and red [36] macroalgae; and a Rhodopseudomonas species with the potential to fix nitrogen was isolated from the inside of C. taxifolia [9]. Also within Bryopsis, Rhizobiales species seem to be well established as clones MX19.12 and MX164.59 (OTU-4) likely represent two different new Phyllobacteriaceae species or genera clustering together with, respectively, a free-living marine Phyllobacteriaceae bacterium [39] and a dinoflagellate-associated anoxygenic photosynthetic bacterial strain [40]. In addition, we amplified a Phyllobacteriaceae nitrogenase-like light-independent protochlorophyllide reductase gene (submitted to GenBank under accession number JN048464) from Bryopsis sample MX164 by the nifH protocol described by De Meyer et al. [41], supporting the above suggested relatedness of OTU-4 to photosynthetic bacteria.
Besides the presence of Alphaproteobacteria in three of the five Bryopsis cultures studied, endophytes from the Gamma- and Epsilonproteobacteria order seem restricted to a single Bryopsis sample. The latter endophytes (OTU-6) most likely belong to a new Arcobacter species within the Campylobacteraceae family. Arcobacter species are mainly known as potential human and animal pathogens, but have also been isolated from diverse marine environments including seawater surrounding seaweeds [42], [43]. Despite their ecologically significant functions like nitrogen fixation, denitrification, sulfide oxidation and manganese reduction [42], [44], they are not frequently reported as endobionts [45], [46]. On the other hand, members of the Xanthomonadaceae family to which OTU-5 belongs, are well-known plant endophytes [47] and have previously been isolated from marine algae [38], [48]. Since many Xanthomonadaceae species cause plant diseases, the high number of Xanthomonadaceae endophytes within Bryopsis MX164 could be a sign of infection. The alga, however, showed no visible disease symptoms (e.g. bleaching), indicating a neutral or beneficial relationship.
In the Bacteroidetes group, we found two distinct clusters (i.e. OTU-1 and OTU-3) of endophytic bacteria, one within the Flavobacteriaceae family and one belonging to unclassified Bacteroidetes. The Flavobacteriaceae endophytes (OTU-1) show an especially strong association with Bryopsis as evidenced by their occurrence in all five samples. The phylum Bacteroidetes, and in particular the family Flavobacteriaceae, forms one of the major components of marine bacterioplankton and mediates a substantial proportion of the carbon flow and nutrient turnover in the sea during and following algal blooms [49]. Moreover, many novel Bacteroidetes members, some of which were characterized as morphogenesis inducers [50], have been isolated from the surfaces of marine macroalgae [1]. Whereas Bacteroidetes bacteria are obviously common epiphytes on macroalgae, Meusnier and co-workers [29] suggested the existence of an endophytic Cytophaga-Flavobacteria-Bacteroidetes (CFB) bacterium within Caulerpa taxifolia. In addition, Bacteroidetes bacteria are well-known endosymbionts of amoebae, plant-parasitic nematodes and insects [51]–[53]. Phylogenetic analysis, however, revealed that the Bacteroidetes endophytes of Bryopsis are more closely related to bacteria tightly associated with corals and sponges [54]–[56] than to CFB sequences isolated from green [29], [50], brown [38] and red [57], [58] macroalgae.
Finally, three Bryopsis samples (i.e. MX19, 164 and 263) contained Mycoplasma sequences (OTU-2). Mycoplasmas are well-known human and animal parasites, but are also common members of the intestinal bacterial flora of fishes and abalones where they may provide nutrients to their hosts [46], [59], [60]. Moreover, the close affiliation of Mycoplasma sequences isolated from Bryopsis and abalone species is perhaps not at all surprising as the latter generally feeds on a broad selection of algae [61]. Also Huang and colleagues [59] postulated that the presence of Mycoplasma species in the intestinal microflora of the abalone Haliotis diversicolor could be algal-food related. Additionally, this bacterial link between Bryopsis and abalone species might be extrapolated to other marine gastropod mollusks, supporting the hypothesis of Rao et al. [13] that the production of therapeutic kahalalides by the sea slug Elysia rufescens as well as by its Bryopsis food could actually be performed through an associated microorganism. Indeed, it has been shown that several metabolites initially assigned to eukaryotes are in fact of microbial origin [1].
In summary, molecular analysis revealed, for the first time, that Bryopsis harbors relatively restricted but taxonomically diverse communities of endophytic bacteria. The presence of Phyllobacteriaceae, Bacteroidetes, Mycoplasma, and in particular Flavobacteriaceae endophytes in several Bryopsis samples collected hundreds of kilometres apart indicates a close association between these endophytes and Bryopsis plants. Even though these endophytic bacterial communities within Bryopsis cultures might not fully represent those that are present within the alga in its natural environment, the bacteria identified in this study are at least part of the natural Bryopsis endobiotic flora. Future investigations of Bryopsis algae in natural environments, however, are necessary to complete the Bryopsis-bacterial endobiosis picture.
Supporting Information
Bacterial 16S rRNA gene sequences isolated from algae (excluding BLAST hits) included in the phylogenetic analysis.
(DOCX)
A wide-range maximum likelihood tree showing the phylogenetic positions of endophytic clones and DGGE bands. Phylogenies were inferred from 16S rRNA gene sequences determined in this study (in bold), BLAST hits (see Table 1), Bacteroidetes, Proteobacteria and Mollicutes type strains, and algae-associated bacteria described in the literature (see Supplementary Table S2). The tree was generated in PhyML according the HKY + G4 algorithmic model. Bootstrap values above 50% are indicated at the branch nodes and the scale bar shows 10 nucleotide substitutions per 100 nucleotides. Asterisks denote sequences previously isolated from micro * - and macroalgae**.
(TIF)
Acknowledgments
Lana Vandersarren is gratefully acknowledged for carrying out parts of the laboratory work. We sincerely thank Heroen Verbruggen and Lennert Tyberghein for collecting the specimens.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: This research was funded by ‘Fonds Wetenschappelijk Onderzoek’ FWO-Flanders project G.0045.08. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Goecke F, Labes A, Wiese J, Imhoff JF. Chemical interactions between marine macroalgae and bacteria. Mar Ecol-Prog Ser. 2010;409:267–299. [Google Scholar]
- 2.McBride DL, Kugrens P, West JA. Light and electron microscopic observations on red algal galls. Protoplasma. 1974;79:249–264. doi: 10.1007/BF01276605. [DOI] [PubMed] [Google Scholar]
- 3.Correa JA, Flores V, Sánchez P. Deformative disease in Iridaea laminarioides (Rhodophyta): Gall development associated with an endophytic cyanobacterium. J Phycol. 1993;29:853–860. [Google Scholar]
- 4.Ashen JB, Goff LJ. Molecular and ecological evidence for species specificity and coevolution in a group of marine algal-bacterial symbioses. Appl Environ Microbiol. 2000;66:3024–3030. doi: 10.1128/aem.66.7.3024-3030.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Burr FA, West JA. Light and electron microscope observations on the vegetative and reproductive structures of Bryopsis hypnoides. Phycologia. 1970;9:17–37. [Google Scholar]
- 6.Turner JB, Friedmann EI. Fine structure of capitular filaments in the coenocytic green alga Penicillus. J Phycol. 1974;10:125–134. [Google Scholar]
- 7.Dawes CJ, Lohr CA. Cytoplasmic organization and endosymbiotic bacteria in the growing points of Caulerpa prolifera . Rev Algol. 1978;13:309–314. [Google Scholar]
- 8.Colombo PM. Occurrence of endophytic bacteria in siphonous algae. Phycologia. 1978;17:148–151. [Google Scholar]
- 9.Chisholm JRM, Dauga C, Ageron E, Grimont PAD, Jaubert JM. 'Roots' in mixotrophic algae. Nature. 1996;381:382–382. [Google Scholar]
- 10.Delbridge L, Coulburn J, Fagerberg W, Tisa LS. Community profiles of bacterial endosymbionts in four species of Caulerpa. Symbiosis. 2004;37:335–344. [Google Scholar]
- 11.Cocquyt E, Verbruggen H, Leliaert F, De Clerck O. Evolution and cytological diversification of the green seaweeds (Ulvophyceae). Mol Biol Evol. 2010;27:2052–2061. doi: 10.1093/molbev/msq091. [DOI] [PubMed] [Google Scholar]
- 12.Kan Y, Fujita T, Sakamoto B, Hokama Y, Nagai H. Kahalalide K: A new cyclic depsipeptide from the Hawaiian green alga Bryopsis species. J Nat Prod. 1999;62:1169–1172. doi: 10.1021/np990053y. [DOI] [PubMed] [Google Scholar]
- 13.Rao KV, Na M, Cook JC, Peng J, Matsumoto R, et al. Kahalalides V–Y isolated from a Hawaiian collection of the sacoglossan mollusk Elysia rufescens. J Nat Prod. 2008;71:772–778. doi: 10.1021/np070508g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hollants J, Leliaert F, De Clerck O, Willems A. How endo- is endo-? Surface sterilization of delicate samples: a Bryopsis (Bryopsidales, Chlorophyta) case study. Symbiosis. 2010;51:131–138. [Google Scholar]
- 15.West JA, McBride DL. Long-term and diurnal carpospore discharge patterns in the Ceramiaceae, Rhodomelaceae and Delesseriaceae (Rhodophyta). Hydrobiologia. 1999;398-399:101–114. [Google Scholar]
- 16.Daims H, Stoecker K, Wagner M. Fluorescence in situ hybridization for the detection of prokaryotes. In: Osborn AM, Smith CJ, editors. Advanced methods in molecular microbial ecology. Abingdon, UK: Bios-Garland; 2005. pp. 213–239. [Google Scholar]
- 17.Daims H, Brühl A, Amann R, Schleifer KH, Wagner M. The domain-specific probe EUB338 is insufficient for the detection of all Bacteria: Development and evaluation of a more comprehensive probe set. Syst Appl Microbiol. 1999;22:434–444. doi: 10.1016/S0723-2020(99)80053-8. [DOI] [PubMed] [Google Scholar]
- 18.Doyle JL, Doyle JJ. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull. 1987;19:11–15. [Google Scholar]
- 19.Hanyuda T, Arai S, Ueda K. Variability in the rbcL introns of Caulerpalean algae (Chlorophyta, Ulvophyceae). J Plant Res. 2000;113:403–413. [Google Scholar]
- 20.Lane DJ. 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M, editors. Nucleic acid techniques in bacterial systematics. New York, NY: John Wiley and Sons; 1991. pp. 115–175. [Google Scholar]
- 21.Van Hoorde K, Verstraete T, Vandamme P, Huys G. Diversity of lactic acid bacteria in two Flemish artisan raw milk Gouda-type cheeses. Food Microbiol. 2008;25:929–935. doi: 10.1016/j.fm.2008.06.006. [DOI] [PubMed] [Google Scholar]
- 22.De Vuyst L, Camu N, De Winter T, Vandemeulebroecke K, Van de Perre V, et al. Validation of the (GTG)5-rep-PCR fingerprinting technique for rapid classification and identification of acetic acid bacteria, with a focus on isolates from Ghanaian fermented cocoa beans. Int J Food Microbiol. 2008;125:79–90. doi: 10.1016/j.ijfoodmicro.2007.02.030. [DOI] [PubMed] [Google Scholar]
- 23.Huber T, Faulkner G, Hugenholtz P. Bellerophon: a program to detect chimeric sequences in multiple sequence alignments. Bioinformatics. 2004;20:2317–2319. doi: 10.1093/bioinformatics/bth226. [DOI] [PubMed] [Google Scholar]
- 24.Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004;32:1792–1797. doi: 10.1093/nar/gkh340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Posada D. jModelTest: phylogenetic model averaging. Mol Biol Evol. 2008;25:1253–1256. doi: 10.1093/molbev/msn083. [DOI] [PubMed] [Google Scholar]
- 26.Guindon S, Gascuel O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol. 2003;52:696–704. doi: 10.1080/10635150390235520. [DOI] [PubMed] [Google Scholar]
- 27.Tamura K, Dudley J, Nei M, Kumar S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol. 2007;24:1596–1599. doi: 10.1093/molbev/msm092. [DOI] [PubMed] [Google Scholar]
- 28.Tujula NA, Holmstrom C, Mussmann M, Amann R, Kjelleberg S, et al. A CARD-FISH protocol for the identification and enumeration of epiphytic bacteria on marine algae. J Microbiol Methods. 2006;65:604–607. doi: 10.1016/j.mimet.2005.09.006. [DOI] [PubMed] [Google Scholar]
- 29.Meusnier I, Olsen JL, Stam WT, Destombe C, Valero M. Phylogenetic analyses of Caulerpa taxifolia (Chlorophyta) and of its associated bacterial microflora provide clues to the origin of the Mediterranean introduction. Mol Ecol. 2001;10:931–946. doi: 10.1046/j.1365-294x.2001.01245.x. [DOI] [PubMed] [Google Scholar]
- 30.Hempel M, Blume M, Blindow I, Gross EM. Epiphytic bacterial community composition on two common submerged macrophytes in brackish water and freshwater. BMC Microbiol. 2008;8 doi: 10.1186/1471-2180-8-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tujula NA, Crocetti GR, Burke C, Thomas T, Holmstrom C, et al. Variability and abundance of the epiphytic bacterial community associated with a green marine Ulvacean alga. ISME J. 2010;4:301–311. doi: 10.1038/ismej.2009.107. [DOI] [PubMed] [Google Scholar]
- 32.Weber CF, King GM. Physiological, ecological, and phylogenetic characterization of Stappia, a marine CO-oxidizing bacterial genus. Appl Environ Microbiol. 2007;73:1266–1276. doi: 10.1128/AEM.01724-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Biebl H, Pukall R, Lünsdorf H, Schulz S, Allgaier M, et al. Description of Labrenzia alexandrii gen. nov., sp. nov., a novel alphaproteobacterium containing bacteriochlorophyll a, and a proposal for reclassification of Stappia aggregata as Labrenzia aggregata comb. nov., of Stappia marina as Labrenzia marina comb. nov. and of Stappia alba as Labrenzia alba comb. nov., and emended descriptions of the genera Pannonibacter, Stappia and Roseibium, and of the species Roseibium denhamense and Roseibium hamelinense. Int J Syst Evol Microbiol. 2007;57:1095–1107. doi: 10.1099/ijs.0.64821-0. [DOI] [PubMed] [Google Scholar]
- 34.Gupta R, Mok A. Phylogenomics and signature proteins for the alpha Proteobacteria and its main groups. BMC Microbiol. 2007;7:106. doi: 10.1186/1471-2180-7-106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kuykendall LD. Order VI. Rhizobiales ord. nov. In: Brenner DJ, Krieg NR, Staley JT, Garrity GM, editors. Bergey's Manual of Systematic Bacteriology (The Proteobacteria), part C (The Alpha-, Beta-, Delta-, and Epsilonproteobacteria) New York: Springer; 2005. 324 [Google Scholar]
- 36.Lachnit T, Meske D, Wahl M, Harder T, Schmitz R. Epibacterial community patterns on marine macroalgae are host-specific but temporally variable. Environ Microbiol. 2011;13:655–665. doi: 10.1111/j.1462-2920.2010.02371.x. [DOI] [PubMed] [Google Scholar]
- 37.Staufenberger T, Thiel V, Wiese J, Imhoff JF. Phylogenetic analysis of bacteria associated with Laminaria saccharina. FEMS Microbiol Ecol. 2008;64:65–77. doi: 10.1111/j.1574-6941.2008.00445.x. [DOI] [PubMed] [Google Scholar]
- 38.Wiese J, Thiel V, Nagel K, Staufenberger T, Imhoff JF. Diversity of antibiotic-active bacteria associated with the brown alga Laminaria saccharina from the Baltic Sea. Mar Biotechnol. 2009;11:287–300. doi: 10.1007/s10126-008-9143-4. [DOI] [PubMed] [Google Scholar]
- 39.Eloe EA, Shulse CN, Fadrosh DW, Williamson SJ, Allen EE, et al. Compositional differences in particle-associated and free-living microbial assemblages from an extreme deep-ocean environment. Environ Microbiol Reports. 2011;3:449–458. doi: 10.1111/j.1758-2229.2010.00223.x. [DOI] [PubMed] [Google Scholar]
- 40.Green DH, Llewellyn LE, Negri AP, Blackburn SI, Bolch CJS. Phylogenetic and functional diversity of the cultivable bacterial community associated with the paralytic shellfish poisoning dinoflagellate Gymnodinium catenatum. FEMS Microbiol Ecol. 2004;47:345–357. doi: 10.1016/S0168-6496(03)00298-8. [DOI] [PubMed] [Google Scholar]
- 41.De Meyer SE, Van Hoorde K, Vekeman B, Braeckman T, Willems A. Genetic diversity of rhizobia associated with indigenous legumes in different regions of Flanders (Belgium). Soil Biol Biochem. 2011;43:2384–2396. [Google Scholar]
- 42.Wirsen CO, Sievert SM, Cavanaugh CM, Molyneaux SJ, Ahmad A, et al. Characterization of an autotrophic sulfide-oxidizing marine Arcobacter sp. that produces filamentous sulfur. Appl Environ Microbiol. 2002;68:316–325. doi: 10.1128/AEM.68.1.316-325.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kim HM, Hwang CY, Cho BC. Arcobacter marinus sp. nov. Int J Syst Evol Microbiol. 2010;60:531–536. doi: 10.1099/ijs.0.007740-0. [DOI] [PubMed] [Google Scholar]
- 44.Thamdrup B, Rossello-Mora R, Amann R. Microbial manganese and sulfate reduction in Black Sea shelf sediments. Appl Environ Microbiol. 2000;66:2888–2897. doi: 10.1128/aem.66.7.2888-2897.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Romero J, García-Varela M, Laclette JP, Espejo RT. Bacterial 16S rRNA gene analysis revealed that bacteria related to Arcobacter spp. constitute an abundant and common component of the oyster microbiota (Tiostrea chilensis). Microb Ecol. 2002;44:365–371. doi: 10.1007/s00248-002-1063-7. [DOI] [PubMed] [Google Scholar]
- 46.Tanaka R, Ootsubo M, Sawabe T, Ezura Y, Tajima K. Biodiversity and in situ abundance of gut microflora of abalone (Haliotis discus hannai) determined by culture-independent techniques. Aquaculture. 2004;241:453–463. [Google Scholar]
- 47.Saddler GS, Bradbury JF. Order III. Xanthomonadales ord. nov. In: Brenner DJ, Krieg NR, Staley JT, Garrity GM, editors. Bergey's Manual of Systematic Bacteriology (The Proteobacteria), part B (The Gammaproteobacteria) New York: Springer; 2005. 63 [Google Scholar]
- 48.Seibold A, Wichels A, Schütt C. Diversity of endocytic bacteria in the dinoflagellate Noctiluca scintillans. Aquat Microb Ecol. 2001;25:229–235. [Google Scholar]
- 49.Pinhassi J, Bowman JP, Nedashkovskaya OI, Lekunberri I, Gomez-Consarnau L, et al. Leeuwenhoekiella blandensis sp. nov., a genome-sequenced marine member of the family Flavobacteriaceae. Int J Syst Evol Microbiol. 2006;56:1489–1493. doi: 10.1099/ijs.0.64232-0. [DOI] [PubMed] [Google Scholar]
- 50.Matsuo Y, Suzuki M, Kasai H, Shizuri Y, Harayama S. Isolation and phylogenetic characterization of bacteria capable of inducing differentiation in the green alga Monostroma oxyspermum. Environ Microbiol. 2003;5:25–35. doi: 10.1046/j.1462-2920.2003.00382.x. [DOI] [PubMed] [Google Scholar]
- 51.Horn M, Wagner M. Bacterial endosymbionts of free-living amoebae. J Eukaryot Microbiol. 2004;51:509–514. doi: 10.1111/j.1550-7408.2004.tb00278.x. [DOI] [PubMed] [Google Scholar]
- 52.Noel GR, Atibalentja N. ‘Candidatus Paenicardinium endonii’, an endosymbiont of the plant-parasitic nematode Heterodera glycines (Nemata: Tylenchida), affiliated to the phylum Bacteroidetes. Int J Syst Evol Microbiol. 2006;56:1697–1702. doi: 10.1099/ijs.0.64234-0. [DOI] [PubMed] [Google Scholar]
- 53.Kikuchi Y. Endosymbiotic bacteria in insects: Their diversity and culturability. Microbes Environ. 2009;24:195–204. doi: 10.1264/jsme2.me09140s. [DOI] [PubMed] [Google Scholar]
- 54.Thiel V, Leininger S, Schmaljohann R, Brümmer F, Imhoff J. Sponge-specific bacterial associations of the Mediterranean sponge Chondrilla nucula (Demospongiae, Tetractinomorpha). Microb Ecol. 2007;54:101–111. doi: 10.1007/s00248-006-9177-y. [DOI] [PubMed] [Google Scholar]
- 55.Sunagawa S, DeSantis TZ, Piceno YM, Brodie EL, DeSalvo MK, et al. Bacterial diversity and White Plague Disease-associated community changes in the Caribbean coral Montastraea faveolata. ISME J. 2009;3:512–521. doi: 10.1038/ismej.2008.131. [DOI] [PubMed] [Google Scholar]
- 56.Sunagawa S, Woodley CM, Medina M. Threatened corals provide underexplored microbial habitats. PLoS ONE. 2010;5:e9554. doi: 10.1371/journal.pone.0009554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Nylund GM, Cervin G, Persson F, Hermansson M, Steinberg PD, et al. Seaweed defence against bacteria: a poly-brominated 2-heptanone from the red alga Bonnemaisonia hamifera inhibits bacterial colonisation. Mar Ecol Prog Ser. 2008;369:39–50. [Google Scholar]
- 58.Penesyan A, Marshall-Jones Z, Holmstrom C, Kjelleberg S, Egan S. Antimicrobial activity observed among cultured marine epiphytic bacteria reflects their potential as a source of new drugs. FEMS Microbiol Ecol. 2009;69:113–124. doi: 10.1111/j.1574-6941.2009.00688.x. [DOI] [PubMed] [Google Scholar]
- 59.Huang ZB, Guo F, Zhao J, Li WD, Ke CH. Molecular analysis of the intestinal bacterial flora in cage-cultured adult small abalone, Haliotis diversicolor. Aquac Res. 2010;41:e760–e769. [Google Scholar]
- 60.Bano N, DeRae Smith A, Bennett W, Vasquez L, Hollibaugh JT. Dominance of Mycoplasma in the guts of the Long-Jawed Mudsucker, Gillichthys mirabilis, from five California salt marshes. Environ Microbiol. 2007;9:2636–2641. doi: 10.1111/j.1462-2920.2007.01381.x. [DOI] [PubMed] [Google Scholar]
- 61.Naidoo K, Maneveldt G, Ruck K, Bolton J. A comparison of various seaweed-based diets and formulated feed on growth rate of abalone in a land-based aquaculture system. J Appl Phycol. 2006;18:437–443. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Bacterial 16S rRNA gene sequences isolated from algae (excluding BLAST hits) included in the phylogenetic analysis.
(DOCX)
A wide-range maximum likelihood tree showing the phylogenetic positions of endophytic clones and DGGE bands. Phylogenies were inferred from 16S rRNA gene sequences determined in this study (in bold), BLAST hits (see Table 1), Bacteroidetes, Proteobacteria and Mollicutes type strains, and algae-associated bacteria described in the literature (see Supplementary Table S2). The tree was generated in PhyML according the HKY + G4 algorithmic model. Bootstrap values above 50% are indicated at the branch nodes and the scale bar shows 10 nucleotide substitutions per 100 nucleotides. Asterisks denote sequences previously isolated from micro * - and macroalgae**.
(TIF)