Abstract
Brain injury, genetic manipulations, and pharmacological treatments can result in alterations of motor skills in mice. Fine motor coordination and balance can be assessed by the beam walking assay. The goal of this test is for the mouse to stay upright and walk across an elevated narrow beam to a safe platform. This test takes place over 3 consecutive days: 2 days of training and 1 day of testing. Performance on the beam is quantified by measuring the time it takes for the mouse to traverse the beam and the number of paw slips that occur in the process. Here we report the protocol used in our laboratory, and representative results from a cohort of C57BL/6 mice. This task is particularly useful for detecting subtle deficits in motor skills and balance that may not be detected by other motor tests, such as the Rotarod.
Protocol
1. This Protocol is Based on Those of Southwell et. al. (2009) and Carter et. al. (2001).
The beam apparatus consists of 1 meter beams with a flat surface of 12 mm or 6 mm width) resting 50 cm above the table top on two poles. A black box is placed at the end of the beam as the finish point. (Plywood and other types of wood for the beams, poles, and box can be found at most hardware stores). Nesting material from home cages is placed in the black box to attract the mouse to the finish point. A lamp (with 60 watt light bulb) is used to shine light above the start point and serves as an aversive stimulus. The time to cross the center 80 cm is measured by two motion detectors (fabricated by Visopia): one at 0 cm that starts a timer and one at 80 cm that stops the timer. A nylon hammock (which can be obtained from a local fabric store) is stretched below the beam, ~7.5 cm above the table top, to cushion any falls. A video camera is set on a tripod to record the performance. The apparatus used in this demonstration was built on site.
A cohort of at least 10 mice for each condition is recommended for this test. Mice are housed as groups, when possible, in a 12 hour light (7am-7pm)/dark cycle and acclimated to their cages in the behavioral facility for at least 2 weeks prior to the start of the test. Approximately 10 min prior to training/testing, the mice can be transported to the room containing the beam apparatus.
On training days, each mouse crosses the 12 mm beam 3 times and then the 6 mm beam 3 times. Beams of wider widths (i.e. 28 mm) can be used for mice that are not able to cross the narrower ones or for larger mice. Mice are placed at one end of a beam and the time required to cross to the escape box at the other end (80 cm away) is measured by the motion detectors. The timer is started by the nose of the mouse entering the center 80 cm, and stopped when the animal reaches the end of the 80 cm. The mice rest for 10 min in their home cages between training sessions on the two beams. During this intertrial period, another 3-4 mice can be trained to make efficient use of time. C57BL/6 mice typically cross the beam with a minimal amount of stopping or stalling. However, when they do stall, sniff or look around without proceeding forward, the investigator should encourage the mouse to continue moving forward by prodding, poking, or pushing it from behind with gloved fingers. Once the mice are in the safe box, they are allowed some time (~15 secs) to rest there before the next trial. After each training session, the mice are returned directly to their home cages and taken back into their housing room. Of note, the mice may stall more if they are over-trained and become too familiar with the task and apparatus. On the other hand, training can be continued for additional days if the mice still do not traverse the entire beam successfully after 2 days of training. If treatments are applied to the mice, a baseline measure of performance should be obtained prior to treatment.
On the test day, times to cross each beam are recorded. Two successful trials in which the mouse did not stall on the beam are averaged. Video recordings can be used for finer analysis of slipping and other observable motor deficits. An alternative way to assess beam walking is by a neurological scoring system1,2, which rates the inability to cross the beam as a score of "1" and the ability to traverse the beam normally with both paws on the beam surface and fewer than two foot slips as a "7." A slip is defined as the foot coming off the top of the beam. In this indexing system, a score of "3" would correspond to crossing the beam by dragging limbs. An animal that scores 1 on two trials is not retested. Description of performance on the beam related to other scores can be found in Feeney et al., (1982) and Carter et al., (2001).
The beams and box are cleaned of mouse droppings and wiped with towels soaked with 70% ethanol and then water before the next beam is placed on the apparatus.
2. Representative Results:
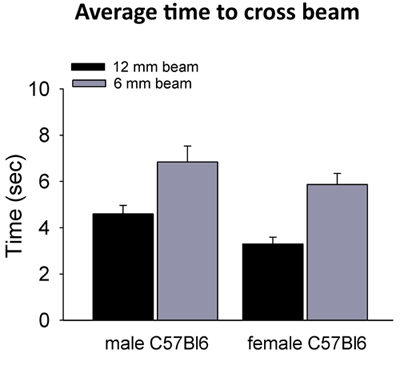 Figure 1. Typical cross times of male and female C57Bl6 on 6mm and 12mm beams. A cohort of 9 to 11 week old C57BL/6-NCRL WT mice were tested on both beams after 2 days of training. Average times for crossing the 12 mm beam were 4.6 ± 0.4 sec for males (n=14) and 3.3 ± 0.3 sec for females (n=11). Times on the 6 mm beam were 6.8 ± 0.7 sec for males (n=13) and 5.9 ± 0.5 sec for females (n=10) (errors are reported as S.E.M.).
Figure 1. Typical cross times of male and female C57Bl6 on 6mm and 12mm beams. A cohort of 9 to 11 week old C57BL/6-NCRL WT mice were tested on both beams after 2 days of training. Average times for crossing the 12 mm beam were 4.6 ± 0.4 sec for males (n=14) and 3.3 ± 0.3 sec for females (n=11). Times on the 6 mm beam were 6.8 ± 0.7 sec for males (n=13) and 5.9 ± 0.5 sec for females (n=10) (errors are reported as S.E.M.).
Discussion
Performance on the balance beam is a useful measure of fine coordination and balance and has been validated by previous work. The beam test can detect motor deficits due to age, central nervous syst` lesions, and genetic and pharmacological manipulations in young and older rodents2,3,4. Mice with cortical impact lesions often exhibit contralateral slipping on the beam5,6. The beam test has also been used to differentiate the motor skills of wild-type (WT) and Huntington disease (HD) mouse models over time and to assess improvements in HD mice with treatment7,8. Another genetic mouse model where motor deficits have been detected on the balance beam include a mouse model of Sandhoff disease, a lysosomal storage disorder9. In detecting motor coordination deficits induced by benzodiazepines, the beam test was more sensitive than the Rotarod. Stanley (2005) showed that only a 30% GABA(A) receptor occupancy by diazepam or lorazepam was needed in order to observe motor deficits on the beam compared to over 70% receptor occupancy for deficits on the Rotarod10. Beams of narrower widths can be used to detect even more subtle differences.
In this study, we tested a cohort of 9 to 11 week old C57BL/6-NCRL WT mice on the 12 mm and 6 mm beams. Average times for crossing the 12 mm beam were 4.6 ± 0.4 sec for males (n=14) and 3.3 ± 0.3 sec for females (n=11). Times on the 6 mm beam were 6.8 ± 0.7 sec for males (n=13) and 5.9 ± 0.5 sec for females (n=10) (errorsare reported as S.E.M.). There were few slips and no falls by these mice on these beams. In genetically or pharmacologically manipulated animals, the slips and falls may become more frequent and can be quantified. Impaired mice may cling onto the side of the beam, which increases their time to cross. The level of activity may differ depending on the age and strain of the mice. C57BL/6 mice are typically active animals6.
Modifications of this test that may encourage the mice to move forward include using an inclined beam rather than a horizontal one2,4. The mice can also be placed in the safe platform for a few seconds to increase willingness to reach the goal2. Confounding factors for assessment of motor coordination using the balance beam include motivation, and mice that are overtrained may become bored and uncooperative. If motor deficits are found, other tests including footprint analysis of locomotor gait, mesh climbing, swimming, pole climbing, and the staircase test can be used to further assess the deficits2,4.
Disclosures
Experiments on animals were performed in accordance with the guidelines and regulations set forth by the Caltech Institutional Animal Care and Use Committee (IACUC).
Acknowledgments
Amber Southwell set up the apparatus and trained the authors in its use. Funding was provided by a grant from the NINDS to Paul Patterson.
References
- Feeney DM, Gonzalez A, Law WA. Amphetamine, haloperidol, and experience interact to affect rate of recovery after motor cortex injury. Science. 1982;217:855–857. doi: 10.1126/science.7100929. [DOI] [PubMed] [Google Scholar]
- Carter RJ, Morton J, Dunnett SB. Motor coordination and balance in rodents. Curr Protoc Neurosci. 2001;Chapter 8 doi: 10.1002/0471142301.ns0812s15. [DOI] [PubMed] [Google Scholar]
- Wallace JE, Krauter EE, Campbell BA. Motor and reflexive behavior in the aging rat. J Gerontol. 1980;35:364–370. doi: 10.1093/geronj/35.3.364. [DOI] [PubMed] [Google Scholar]
- Brooks SP, Dunnett SB. Tests to assess motor phenotype in mice: a user's guide. Nat Rev Neurosci. 2009;10:519–529. doi: 10.1038/nrn2652. [DOI] [PubMed] [Google Scholar]
- Fox GB, Fan L, Levasseur RA, Faden AI. Sustained sensory/motor and cognitive deficits with neuronal apoptosis following controlled cortical impact brain injury in the mouse. J Neurotrauma. 1998;15:599–614. doi: 10.1089/neu.1998.15.599. [DOI] [PubMed] [Google Scholar]
- Buccafusco JJ. Methods of behavioral analysis in neuroscience. 2nd edn. CRC Press; 2009. [PubMed] [Google Scholar]
- Southwell AL, Ko J, Patterson PH. Intrabody gene therapy ameliorates motor, cognitive, and neuropathological symptoms in multiple mouse models of Huntington's disease. J Neurosci. 2009;29:13589–13602. doi: 10.1523/JNEUROSCI.4286-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter RJ. Characterization of progressive motor deficits in mice transgenic for the human Huntington's disease mutation. J Neurosci. 1999;19:3248–3257. doi: 10.1523/JNEUROSCI.19-08-03248.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gulinello M, Chen F, Dobrenis K. Early deficits in motor coordination and cognitive dysfunction in a mouse model of the neurodegenerative lysosomal storage disorder, Sandhoff disease. Behav Brain Res. 2008;193:315–319. doi: 10.1016/j.bbr.2008.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanley JL. The mouse beam walking assay offers improved sensitivity over the mouse rotarod in determining motor coordination deficits induced by benzodiazepines. J Psychopharmacol. 2005;19:221–227. doi: 10.1177/0269881105051524. [DOI] [PubMed] [Google Scholar]


