Abstract
OBJECTIVE
Reports of increased circulating fibroblast growth factor 21 (FGF21) levels in obesity indicate that FGF21 may be implicated in body weight homeostasis. We sought to investigate the existence of FGF21 in human cerebrospinal fluid (CSF) and, if present, the relationship between CSF FGF21 with body adiposity and metabolic parameters.
RESEARCH DESIGN AND METHODS
CSF and corresponding plasma FGF21 were measured by an enzyme-linked immunosorbent assay (18 men and 20 women, aged 19–80 years, and BMI 16.2–38.1 kg/m2) and correlated to body adiposity and metabolic parameters.
RESULTS
CSF and plasma FGF21 increased in particular with rising BMI and fat mass. In CSF, FGF21 was detectable at concentrations ~40% that of plasma levels. CSF and plasma FGF21 levels were significantly positively correlated with BMI and fat mass, body weight, plasma insulin, and homeostasis model assessment of insulin resistance. Plasma FGF21 levels were significantly negatively correlated with plasma adiponectin. When subjected to multiple regression analysis, only fat mass was predictive of plasma FGF21 (β = 0.758; P = 0.004) and CSF FGF21 (β = 0.767; P = 0.007). The CSF-to-plasma FGF21 ratio was significantly negatively correlated with BMI, fat mass, and plasma FGF21. Subjects in the highest plasma FGF21 quintile had a lower CSF-to-plasma FGF21 ratio (12.7% [9.7–14.9%]) compared with those in the lowest plasma FGF21 quintile (94.7% [37.3–99.8%]) (P < 0.01).
CONCLUSIONS
Our observations have important implications with respect to the potential central actions of FGF21. Future research should seek to clarify whether FGF21 would be beneficial in the management of obesity and its metabolic complications.
The fibroblast growth factor (FGF) family has 22 members that exert a wide range of biological effects (1). In addition to its role in regulating cell growth and differentiation (2), three members of the FGF family, specifically, FGF19, FGF21, and FGF23 (members of the same FGF subfamily), have been shown to have significant metabolic functions (3). FGF19 is produced by the intestinal epithelium and has been implicated in regulating cholesterol and bile-acid synthesis (4). FGF23, on the other hand, is produced by the bone and regulates phosphate absorption and vitamin D biosynthesis via its actions on the kidneys (5). FGF21, in contrast, is predominantly produced in the liver as well as adipose tissue and has been shown to be an important factor in the homeostatic mechanisms regulating glucose and lipid metabolism (6).
FGF21 recently has been described as a metabolic regulator and a key hormonal mediator of the adaptive starvation response; FGF21 levels were positively associated with obesity and the metabolic syndrome and increased in type 2 diabetes (7–11). Of relevance, FGF21 has been reported to alleviate obesity in mice (12).
The hypothalamus is the key regulatory center for energy balance and is abundant with peptides that regulate satiety (13). Many of these peptides also are produced in peripheral sites, in the case of FGF21, as mentioned above, the liver, and adipose tissue. Recently, an elegant study by Hsuchou et al. (14) demonstrated that FGF21 crosses the blood-brain barrier (BBB) in mice and affirmed that peripheral FGF21 could reach the brain directly and thus potentially exert its central effects (14,15).
Given the above, we investigated the presence of FGF21 in human cerebrospinal fluid (CSF) and examined the potential contribution of circulating FGF21 to the brain by studying the CSF concentrations of FGF21 in relation to corresponding plasma levels, body adiposity, and metabolic parameters within the same cohort of subjects.
Thirty-eight Caucasian subjects participated in the study (18 men and 20 women, aged 19–80 years, and BMI 16.2–38.1 kg/m2). Fourteen participants had normal body weight, 14 subjects were overweight (BMI 25 to <30 kg/m2), and 10 subjects were obese (BMI ≥30 kg/m2). Exclusion criteria included a history of diabetes, congestive heart failure, liver or kidney disease, or malignancy or signs of inflammation, pregnancy, and any drugs influencing body weight such as corticosteroids or contraceptives. During the study, three subjects (two men and one woman) were diagnosed with type 2 diabetes (fasting plasma glucose levels >7 mmol/L), as per the American Diabetes Association criteria. After an overnight fast, subjects undertook simultaneous sampling of blood and CSF (1 mL) via a lumbar puncture after local anesthesia (2 mL mepivacaine–HCL 1%). Blood samples immediately were centrifuged. Plasma and CSF samples were prepared within 1 h and stored at −80°C until assayed. All patients underwent anthropometric measurements. Body composition was measured by standard bioelectrical impedance analysis (BIA 2000-M, Data Input, Hofheim, Germany). Frequencies of 1, 5, 50, and 100 Hz were used; also, Eurobody software (Data Input) was used to analyze body fat mass. The study was approved by the local research ethics committee, and written informed consent was obtained from all participants, in accordance with the guidelines in the Declaration of Helsinki 2000.
Biochemical and hormonal analysis.
Plasma and CSF glucose (Beckman Glucose Analyzer II; Beckman Instruments, Munich, Germany) and insulin (commercial competitive double-antibody radioimmunoassay, Pharmacia Insulin RIA 100; Pharmacia Diagnostics, Uppsala, Sweden) with an assay limit of sensitivity of 1.8 pmol/L and an intra-assay coefficient of variation of <4.5% were measured. The estimate of insulin resistance by homeostasis model assessment of insulin resistance (HOMA-IR) score was calculated as previously described (16). Adiponectin levels in plasma and CSF were determined using a commercially available radioimmunoassay kit (Linco Research, St. Charles, MO) according to the manufacturer’s protocol, with an intra-assay coefficient of variation of 6.2%. FGF21 levels in plasma and CSF were measured using a commercially available ELISA kit (BioVendor, Oxford, U.K.), according to the manufacturer’s protocol, with an intra-assay coefficient of variation of 3.6%.
Statistics.
Because plasma and CSF FGF21 were not distributed normally, as checked by histograms and the Kolmogorov-Smirnoff test, nonparametric tests were used. Data were analyzed by the Mann-Whitney U test or the Kruskal-Wallis ANOVA (post hoc analysis: Dunn test), according to the number of groups compared. Data are medians (interquartile range). Spearman rank correlation was used for the calculation of associations between variables. Subsequently, if individual bivariate correlations achieved statistical significance, variables were entered into a linear regression model and multiple regression analysis with either plasma FGF21, CSF FGF21, or CSF/plasma FGF21 as a dependent variable was performed to test the joint effect of different parameters on plasma FGF21, CSF FGF212, and CSF/plasma FGF21, respectively. All statistical analyses were performed using SPSS version 18.0 (SPSS, Chicago, IL). P < 0.05 was considered significant.
RESULTS
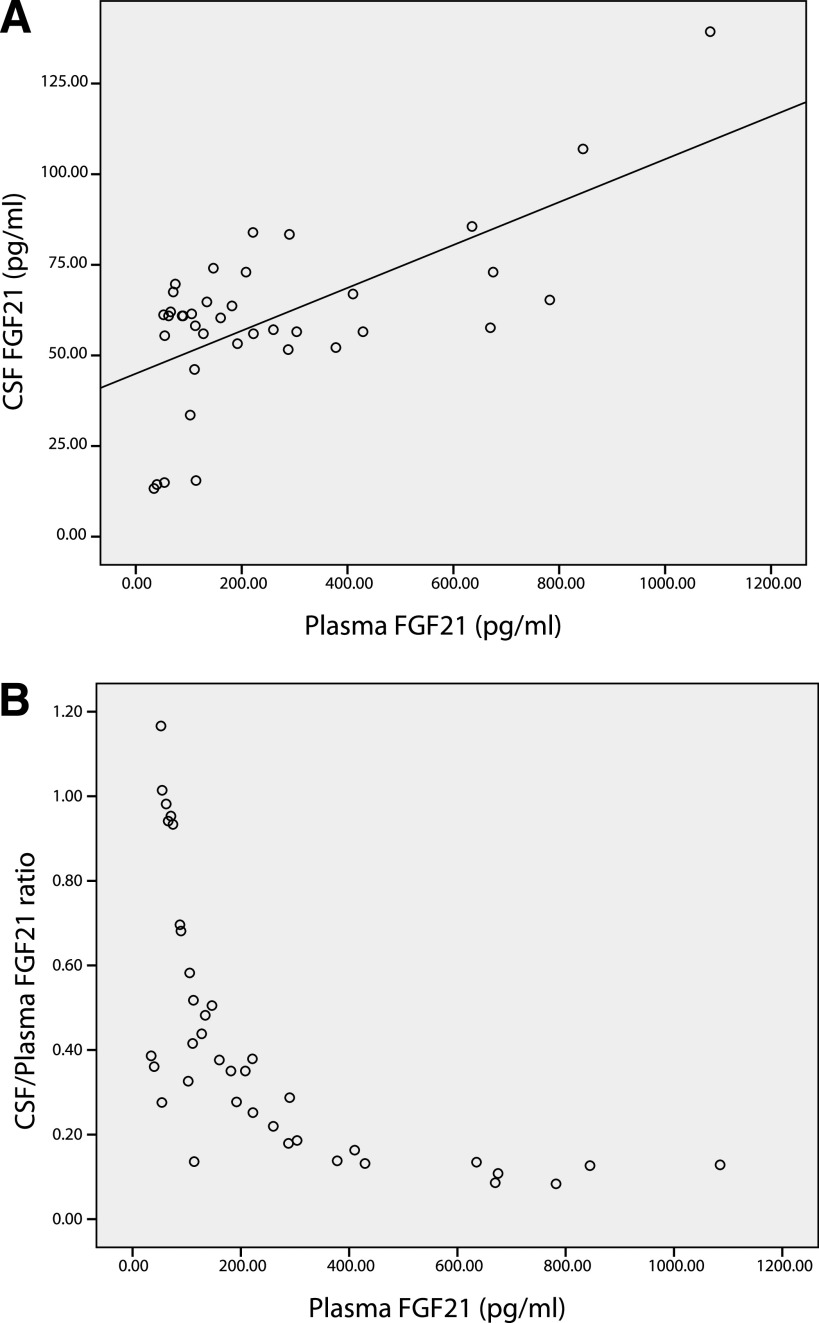
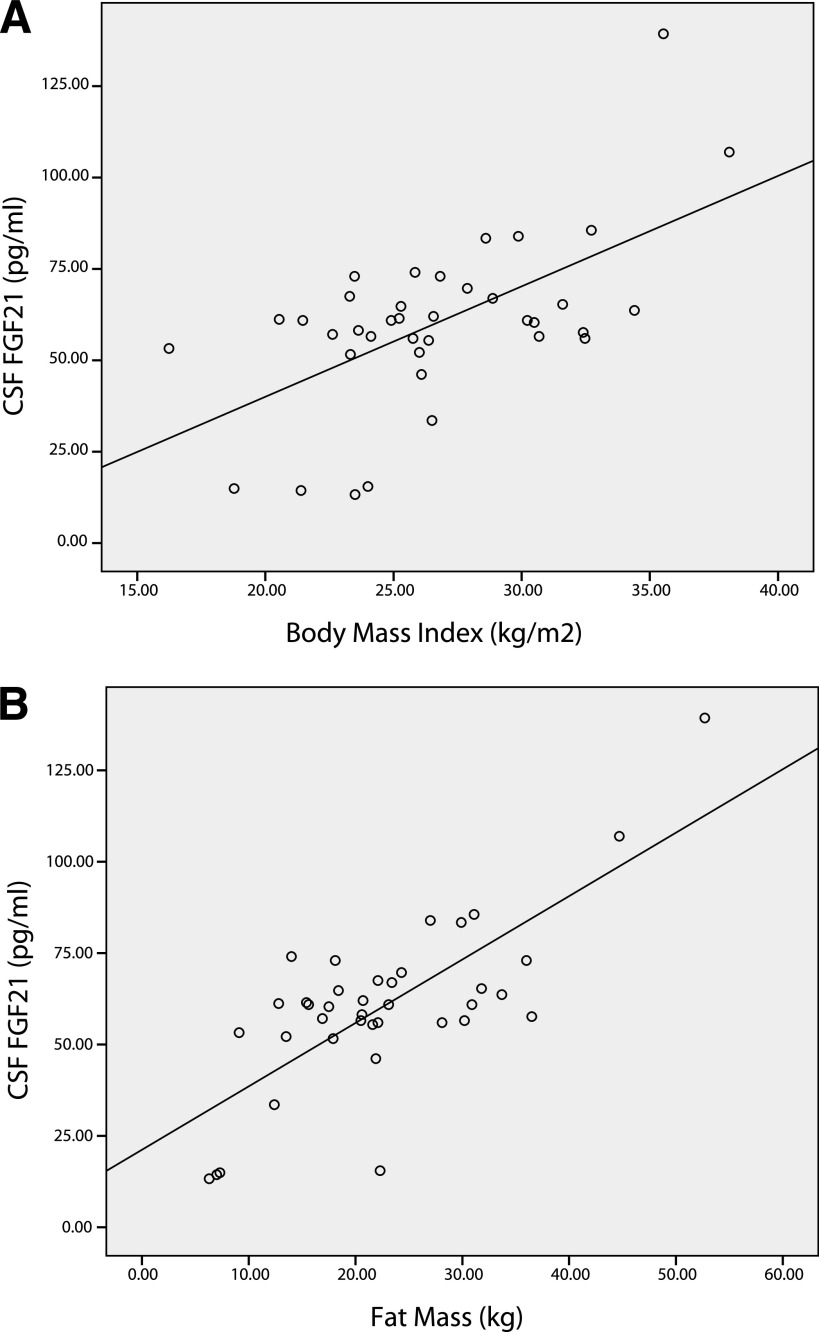
Table 1 shows the anthropometric, biochemical, and hormonal parameters in all subjects and respective associations with plasma FGF21, CSF FGF21, and the CSF-to-plasma FGF21 ratio (Fig. 1). Table 2 presents data on plasma and CSF FGF21 with respect to body fat, body weight status, and sex. Plasma and CSF FGF21 increased in particular with rising BMI and fat mass (Table 2); no sex differences were found in agreement with recent studies (8–11). In CSF, FGF21 was detectable at concentrations ~40% that of plasma levels (Table 1).
TABLE 1.
Demographics and correlations with plasma and CSF FGF21 and the CSF-to-plasma FGF21 (n = 38)
| Variable | Median (interquartile range) | Plasma FGF21 | CSF FGF21 | CSF-to-plasma FGF21 ratio |
|---|---|---|---|---|
| Age (years) | 53.0 (42.0–64.0) | 0.100 | 0.174 | 0.067 |
| BMI (kg/m2) | 26.1 (23.5–30.2) | 0.576* | 0.479* | −0.385† |
| Body weight (kg) | 75.2 (66.3–88.3) | 0.449* | 0.389† | −0.272 |
| Fat mass (kg) | 21.8 (15.6–29.9) | 0.601* | 0.574* | −0.389† |
| Glucose | ||||
| Plasma (mmol/L) | 4.9 (4.6–5.4) | 0.076 | 0.032 | −0.002 |
| CSF (mmol/L) | 3.2 (3.1–3.5) | 0.201 | −0.038 | −0.232 |
| CSF-to-plasma ratio | 0.65 (0.61–0.70) | 0.130 | −0.145 | −0.251 |
| Insulin | ||||
| Plasma (pmol/L) | 64.7 (43.4–110.9) | 0.374† | 0.457* | −0.210 |
| CSF (pmol/L) | 1.8 (1.8–2.3) | 0.142 | −0.065 | −0.157 |
| CSF-to-plasma ratio | 0.029 (0.023–0.045) | −0.205 | −0.417* | 0.066 |
| HOMA-IR | 2.5 (1.6–4.2) | 0.385† | 0.413* | −0.234 |
| Adiponectin | ||||
| Plasma (µg/mL) | 10.6 (8.9–16.0) | −0.341† | 0.132 | 0.371† |
| CSF (ng/mL) | 4.1 (3.0–7.2) | −0.064 | −0.015 | 0.028 |
| CSF-to-plasma ratio | 0.00038 (0.00029–0.00058) | 0.130 | −0.150 | −0.184 |
| FGF21 | ||||
| Plasma (pg/mL) | 153.5 (87.5–290.2) | — | — | — |
| CSF (pg/mL) | 60.9 (55.4–66.9) | 0.430* | — | — |
| CSF-to-plasma ratio | 0.35 (0.16–0.52) | −0.830* | −0.033 | — |
Spearman rank correlation was used for the calculation of associations between variables. P < 0.05 was considered significant.
*P < 0.01.
†P < 0.05.
FIG. 1.
Relationships between CSF FGF21 (A) and the plasma FGF21 CSF-to-plasma FGF21 ratio (B) and plasma FGF21 in all subjects (n = 38). Spearman correlation coefficients were as follows: R = 0.430, P < 0.01 (A), and R = −0.830, P < 0.01 (B).
TABLE 2.
Plasma and CSF FGF21 with respect to body fat, body weight status, and sex
| Plasma FGF21 (pg/mL) | P* | CSF FGF21 (pg/mL) | P* | |
|---|---|---|---|---|
| Body fat quartile | ||||
| 1 | 58.4 (52.5–102.8) | <0.001 | 39.8 (14.9–52.2) | <0.001 |
| 2 | 153.5 (112.4–260.0) | 0.007 | 59.3 (56.5–61.5) | <0.001 |
| 3 | 174.6 (89.4–290.2) | 0.005 | 62.3 (56.5–66.9) | 0.002 |
| 4 | 652.6 (303.9–782.1) | 78.7 (73.0–85.6) | ||
| Body weight status | ||||
| Normal weight | 124.0 (54.1–208.3) | 0.007 | 56.8 (15.5–60.9) | 0.001 |
| Overweight | 120.1 (74.6–222.2) | 0.021 | 56.3 (53.3–63.7) | 0.003 |
| Obese | 532.3 (181.7–782.1) | 73.5 (65.3–85.6) | ||
| Sex | ||||
| Male | 129.5 (87.5–222.2) | 60.9 (53.3–64.8) | ||
| Female | 184.3 (82.0–419.8) | 60.6 (56.0–71.3) |
Data are median (interquartile range). n = 38: 18 men and 20 women; 14 normal-weight subjects, 14 overweight subjects (BMI 25 to <30 kg/m2), and 10 obese subjects (BMI ≥30 kg/m2).
*For comparisons with body fat quartile 4 and obesity, respectively. Group comparison by Mann-Whitney U test or Kruskal-Wallis ANOVA (post hoc analysis: Dunn test). P < 0.05 was considered significant. Differences between men and women were not significant.
Spearman rank analyses demonstrated that plasma FGF21 levels were significantly positively correlated with BMI and fat mass, both markers of body adiposity, and also body weight, plasma insulin, HOMA-IR, and CSF FGF21 (Table 1). Plasma FGF21 levels were significantly negatively correlated with plasma adiponectin and the CSF-to-plasma FGF21 ratio (Table 1). When subjected to multiple regression analysis, fat mass (β = 0.758; P = 0.004) was predictive of plasma FGF21.
CSF FGF21 levels were significantly positively correlated with BMI and fat mass (Fig. 2), as well as body weight, plasma insulin, HOMA-IR, and plasma FGF21 (Table 1). CSF FGF21 levels were significantly negatively correlated with CSF/plasma insulin (Table 1). When subjected to multiple regression analysis, fat mass (β = 0.767; P = 0.007) was predictive of CSF FGF21.
FIG. 2.
Relationships between CSF FGF21 (A) and BMI CSF FGF21 (B) and fat mass in all subjects (n = 38). Spearman correlation coefficients were as follows: R = 0.479, P < 0.01 (A) and R = 0.574, P < 0.01 (B).
The CSF-to-plasma FGF21 ratio was significantly negatively correlated with BMI, fat mass, and plasma FGF21 (Table 1). The CSF-to-plasma FGF21 ratio was significantly positively correlated with plasma adiponectin (Table 1). When subjected to multiple regression analysis, none of the variables were predictive of the CSF-to-plasma FGF21 ratio.
As mentioned above, CSF FGF21 was significantly positively correlated with plasma FGF21 (R = 0.430; P < 0.01) (Table 2). There was a significant linear relation between CSF and plasma FGF21 in lean subjects (BMI <25 kg/m2) (R = 0.657; P = 0.011) and in overweight/obese subjects (BMI ≥25 kg/m2) (R = 0.753; P < 0.01).
Furthermore, we also found that subjects in the highest plasma FGF21 quintile had a median (interquartile range) CSF-to-plasma FGF21 ratio of 12.7% (9.7–14.9%) compared with 94.7% (37.3–99.8%) in those in the lowest plasma FGF21 quintile (P < 0.01), corresponding BMI (32.0 kg/m2 [27.9–34.1] vs. 22.4 kg/m2 [21.0–23.4]; P < 0.01), and fat mass (33.9 kg [27.3–40.6] vs. 14.2 kg [7.2–21.9]; P < 0.01).
DISCUSSION
Our study demonstrates, for the first time, the existence of FGF21 in human CSF and the relationship between CSF FGF21 and corresponding plasma FGF21 levels, body adiposity, and hormonal and metabolic parameters. We report significantly higher circulating FGF21 levels in obese compared with lean subjects, in agreement with previous studies (12,17). Furthermore, we found significantly higher CSF FGF21 levels in obese compared with lean subjects. In addition, we found that both plasma and CSF FGF21 were significantly positively correlated with BMI and fat mass, both markers of body adiposity as well as body weight, plasma insulin, and HOMA-IR. Importantly, fat mass was the only determining factor of both plasma and CSF FGF21 levels. Given the recently reported central nervous system actions of FGF21 (15), we postulate that the higher FGF21 levels in the CSF of obese individuals may be associated with dysfunctional FGF21 signaling in the brain. This concept seems plausible because dysfunctional central nervous system signaling of other growth factors and adipokines, such as insulin and leptin, respectively, has been associated with obesity (18). Moreover, it has been reported that obesity is an FGF21-resistant state (19).
We also found a significant negative correlation between plasma FGF21 and plasma adiponectin consistent with previous reports (7,8,20,21). Moreover, we detected adiponectin in human CSF, albeit in relatively low levels to plasma adiponectin, in keeping with a previous study (22). Of note, although it has been suggested that adiponectin may play an important role in the cross-talk between peripheral tissues and the central nervous system, in particular, with respect to energy homeostasis, this idea in humans remains contentious (23).
Recently, a landmark study by Sarruf et al. (15) showed that continuous intracerebroventricular infusion of FGF21 enhanced appetite and energy production in rodents by increasing insulin sensitivity via increased insulin-induced suppression of hepatic gluconeogenesis; they suggested that FGF21 interacts with FGF receptor-1 (predominantly expressed in the arcuate and ventromedial nuclei of the hypothalamus, areas of the hypothalamus that also mediate the central metabolic effects of insulin, leptin, oleate, and glucose) and regulates gluconeogenesis.
In order for FGF21 to exert its central effects, and given that FGF21 is mainly produced in the liver, circulating FGF21 should traverse the BBB and/or blood-CSF barriers. Of relevance, Hsuchou et al. (14) surmised that FGF21 crosses the BBB by simple diffusion in mice. Our novel finding of a significant positive correlation between plasma FGF21 and CSF FGF21 supports this notion. In addition, we found significant negative correlations between the CSF-to-plasma FGF21 ratio and BMI and fat mass. Also, we found that subjects in the highest plasma FGF21 quintile had a median CSF-to-plasma FGF21 ratio of 12.7%, compared with 94.7% in those in the lowest plasma FGF21 quintile. This observation suggests that CSF FGF21 may be derived from the central neural tissue and that a possible discrepancy in the production of FGF21 by the central nervous system may explain the differences in the CSF-to-plasma FGF21 ratio between the obese and lean subjects in our study. Of relevance, Yamashita et al. (24) reported that FGF23 is produced in the brain, in particular, the ventrolateral thalamic nuclei. It is therefore plausible that FGF21 also may be produced in the brain. Moreover, it is probable that FGF21 has protein binding and that differences in protein binding in obese and lean subjects also may explain our findings. Furthermore, it is possible that the efficiency of FGF21 uptake into the CSF is reduced in obese individuals, possibly secondary to saturation of transporters. When making this point, we are conscious of the good evidence by Hsuchou et al. (14), who showed that FGF21 crosses the BBB nonsaturably. The coexistence of nonsaturable and saturable mechanisms is plausible given that other adipokines (e.g., leptin) have been shown to cross the BBB through both nonsaturable and saturable mechanisms (25). Taken together, given that FGF21 crosses the BBB via a relatively slow passive diffusion and exhibits considerable degradation in the central nervous system (i.e., within 20 min) (14), production of FGF21 within the brain may be the main explanation for our findings. Additional studies are needed to clarify these points.
A limitation of this study is that we have not assessed the presence of other members of the FGF family in human CSF. In addition, we have not measured the energy status in our study subjects. Given that intracerebroventricular infusion of FGF21 enhanced appetite and energy production in rodents, it would be interesting to establish the relationship between the levels of other members of the FGF family in human CSF with FGF21 and to correlate this with the energy status of our study subjects. Future studies are needed to elucidate these points.
In conclusion, our novel observations have important implications with respect to the potential central actions of FGF21. Future research also should seek to clarify whether FGF21 would be beneficial in the management of obesity and its metabolic complications.
ACKNOWLEDGMENTS
The General Charities of the City of Coventry funded this study.
No potential conflicts of interest relevant to this article were reported.
B.K.T. researched data, contributed to discussion, and wrote, reviewed, and edited the manuscript. M.H. researched data, contributed to discussion, and reviewed and edited the manuscript. R.A. researched data and reviewed and edited the manuscript. W.K., H.L., and H.S.R. contributed to discussion and reviewed and edited the manuscript.
H.S.R. acknowledges S. Waheguru, University of Warwick, for his continual support.
REFERENCES
- 1.Ornitz DM, Itoh N. Fibroblast growth factors. Genome Biol 2001;2:REVIEWS3005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gospodarowicz D. Localisation of a fibroblast growth factor and its effect alone and with hydrocortisone on 3T3 cell growth. Nature 1974;249:123–127 [DOI] [PubMed] [Google Scholar]
- 3.Beenken A, Mohammadi M. The FGF family: biology, pathophysiology and therapy. Nat Rev Drug Discov 2009;8:235–253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Inagaki T, Choi M, Moschetta A, et al. Fibroblast growth factor 15 functions as an enterohepatic signal to regulate bile acid homeostasis. Cell Metab 2005;2:217–225 [DOI] [PubMed] [Google Scholar]
- 5.ADHR Consortium. Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23. Nat Genet 2000;26:345–348 [DOI] [PubMed] [Google Scholar]
- 6.Kharitonenkov A, Shiyanova TL, Koester A, et al. FGF-21 as a novel metabolic regulator. J Clin Invest 2005;115:1627–1635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhang X, Yeung DC, Karpisek M, et al. Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes 2008;57:1246–1253 [DOI] [PubMed] [Google Scholar]
- 8.Gälman C, Lundåsen T, Kharitonenkov A, et al. The circulating metabolic regulator FGF21 is induced by prolonged fasting and PPARalpha activation in man. Cell Metab 2008;8:169–174 [DOI] [PubMed] [Google Scholar]
- 9.Li L, Yang G, Ning H, Yang M, Liu H, Chen W. Plasma FGF-21 levels in type 2 diabetic patients with ketosis. Diabetes Res Clin Pract 2008;82:209–213 [DOI] [PubMed] [Google Scholar]
- 10.Li K, Li L, Yang M, Zong H, Liu H, Yang G. Effects of rosiglitazone on fasting plasma fibroblast growth factor-21 levels in patients with type 2 diabetes mellitus. Eur J Endocrinol 2009;161:391–395 [DOI] [PubMed] [Google Scholar]
- 11.Lin Z, Zhou Z, Liu Y, et al. Circulating FGF21 levels are progressively increased from the early to end stages of chronic kidney diseases and are associated with renal function in Chinese. PLoS ONE 2011;6:e18398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Coskun T, Bina HA, Schneider MA, et al. Fibroblast growth factor 21 corrects obesity in mice. Endocrinology 2008;149:6018–6027 [DOI] [PubMed] [Google Scholar]
- 13.Enriori PJ, Evans AE, Sinnayah P, et al. Diet-induced obesity causes severe but reversible leptin resistance in arcuate melanocortin neurons. Cell Metab 2007;5:181–194 [DOI] [PubMed] [Google Scholar]
- 14.Hsuchou H, Pan W, Kastin AJ. The fasting polypeptide FGF21 can enter brain from blood. Peptides 2007;28:2382–2386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sarruf DA, Thaler JP, Morton GJ, et al. Fibroblast growth factor 21 action in the brain increases energy expenditure and insulin sensitivity in obese rats. Diabetes 2010;59:1817–1824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412–419 [DOI] [PubMed] [Google Scholar]
- 17.Mraz M, Bartlova M, Lacinova Z, et al. Serum concentrations and tissue expression of a novel endocrine regulator fibroblast growth factor-21 in patients with type 2 diabetes and obesity. Clin Endocrinol (Oxf) 2009;71:369–375 [DOI] [PubMed] [Google Scholar]
- 18.Morton GJ, Cummings DE, Baskin DG, Barsh GS, Schwartz MW. Central nervous system control of food intake and body weight. Nature 2006;443:289–295 [DOI] [PubMed] [Google Scholar]
- 19.Fisher FM, Chui PC, Antonellis PJ, et al. Obesity is a fibroblast growth factor 21 (FGF21)-resistant state. Diabetes 2010;59:2781–2789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dostálová I, Kaválková P, Haluzíková D, et al. Plasma concentrations of fibroblast growth factors 19 and 21 in patients with anorexia nervosa. J Clin Endocrinol Metab 2008;93:3627–3632 [DOI] [PubMed] [Google Scholar]
- 21.Matuszek B, Lenart-Lipińska M, Duma D, Solski J, Nowakowski A. Evaluation of concentrations of FGF-21—a new adipocytokine in type 2 diabetes. Endokrynol Pol 2010;61:50–54 [PubMed] [Google Scholar]
- 22.Kos K, Harte AL, da Silva NF, et al. Adiponectin and resistin in human cerebrospinal fluid and expression of adiponectin receptors in the human hypothalamus. J Clin Endocrinol Metab 2007;92:1129–1136 [DOI] [PubMed] [Google Scholar]
- 23.Dridi S, Taouis M. Adiponectin and energy homeostasis: consensus and controversy. J Nutr Biochem 2009;20:831–839 [DOI] [PubMed] [Google Scholar]
- 24.Yamashita T, Yoshioka M, Itoh N. Identification of a novel fibroblast growth factor, FGF-23, preferentially expressed in the ventrolateral thalamic nucleus of the brain. Biochem Biophys Res Commun 2000;277:494–498 [DOI] [PubMed] [Google Scholar]
- 25.Nam SY, Kratzsch J, Kim KW, Kim KR, Lim SK, Marcus C. Cerebrospinal fluid and plasma concentrations of leptin, NPY, and alpha-MSH in obese women and their relationship to negative energy balance. J Clin Endocrinol Metab 2001;86:4849–4853 [DOI] [PubMed] [Google Scholar]