Abstract
Regionally and functionally diverse types of astrocytes exist throughout the central nervous system and participate in nearly every aspect of normal and abnormal neural function. Therefore, human astrocyte subtypes are a useful tool for understanding brain function, modulating disease processes, and promoting neural regeneration. Here we describe a protocol for directed differentiation and maintenance of functional astroglia from human pluripotent stem cells in a chemically defined system. Human stem cells are first differentiated to neuroepithelial cells with or without exogenous patterning molecules(days 0 –21). Regular dissociation of the neuroepithelial clusters in suspension, and in the presence of mitogens, permits generation of astroglial subtypes over along term expansion (days 21–90). Finally, the astroglial progenitors are either amplified for an extended time or differentiated to functional astrocytes upon removal of mitogens and the addition of CNTF ( days > 90). This method generates robust populations of functionally diversified astrocytes with high efficiency.
Introduction
Astrocytes, one class of glial cells, are the most abundant cells in the vertebrate central nervous system. Like neurons, there are many types of astrocytes based on their locations, morphologies, gene expression profiles, and their responses to environmental signals1,2. Astrocytes are vital for maintaining a homeostatic environment for the healthy brain, which ensures appropriate and precise neuron-neuron communication. Under pathological conditions, astrocytes react to prevent further damage or may contribute to the demise of the brain. Therefore, understanding how to regulate astrocyte activity will benefit the treatment of a range of neurological injuries and diseases.
Background information about astrocytes
Despite their abundance in number and importance in function, astrocytes have been less well studied compared to neuronal types. This is partly due to fewer reliable markers for the astroglial lineage and the difficulty in culturing/expanding purified astrocytes under defined conditions. Astrocytes can be identified by their typical stellate morphology and their expression of S100β and glial fibrillary acidic protein (GFAP), but markers for astroglial progenitors are relatively vague3. A2B5 can mark some astroglial progenitors, but A2B5 -expressing cells can also generate oligodendrocytes4 and in humans also neurons5. CD44 is more specific to astrocytes6, although it is expressed at a relatively late developmental stage. Astrocytes are typically cultured from rodent brain tissues during the gliogenic stage, i.e., around birth. They may be expanded briefly but quickly undergo senescence in the absence of growth factor s7,8. Undefined culture media, including sera, are generally required for the survival of cultured astrocytes. Besides, primary astrocyte cultures are often contaminated with other cell types, especially microglia, which can significantly confound functional analysis of astrocytes. For regional astrocyte subtypes, one has to obtain specific brain tissues, which is particularly difficult for astrocyte cultures of human origin. Though fetal derived neural cells from different regions have been useful for generating neural subtypes in culture 9 and are available commercially, differentiation of astroglia from induced pluripotent stem cells (iPSCs)is the most convenient option to obtain disease-specific tissue. Therefore, there is a critical need of a defined culture system for culturing and expanding purified or enriched human astrocyte subtypes from human pluripotent stem cells (PSCs).
Derivation of human astrocytes
An ideal source of human astrocytes is PSCs, including embryonic stem cells (ESCs) and iPSCs. Astrocyte differentiation has been demonstrated from human ESC s10–14 and recently from human iPSCs15,16, though high variability exists between methods and results in only a small percent out of total neural cells depending upon length of time ( Table 1). Until recently17, directed differentiation of astrocytes from human ESCs or iPSCs has not been reported. In addition, the functional identity of in vitro generated astrocytes and a protocol to direct stem cells to astrocyte subclasses with different regional and functional identities have not been reported. In the protocol described here we found that human stem cell -derived neuroepithelial cells differentiate into glial progenitors when neurogenesis is repressed through regular dissociation of neuroepithelial clusters. These glial progenitors can be expanded extensively and produce a nearly pure population of functional astroglia, measured by gene expression profiles, uptake of neurotransmitters and ions, support of neuronal maturation, and engraftment into the mouse brain. Importantly, by patterning the neuroepithelial cells, we are able to guide human stem cells to diverse classes of astrocytes that express differential sets of homeodomain factors and bear specific functional properties17.
Table 1.
Summary of reports on human astrocyte differentiation
| Reference | Cellular source | Differentiation efficiency and functional analysis |
|---|---|---|
| 5 | ESCs | GFAP+ cells rarely seen at 21–23 days. No functional studies. |
| 10 | ESCs | Few GFAP+ cells at 3 weeks but increases after 45 days. Observed post engraftment into rodent brain. |
| 11 | ESCs | 26% of total are GFAP+ cells at 10 weeks. Observed post engraftment into rodent brain. |
| 12 | ESCs | >50% of total are GFAP+ cells at 25 weeks. No functional studies. |
| 13 | ESCs | 2% of total are GFAP+ at 4 weeks. Observed post engraftment into rodent brain. |
| 14 | ESCs | GFAP+ cells appear after 9 weeks. Appearance correlated with neuronal synaptogenesis. |
| 15 | ESCs and iPSCs | GFAP+ cells appear around 12 weeks. No functional studies. |
| 16 | iPSCs | A proportion are GFAP+ at >6 weeks Observed post engraftment into rodent brain within teratoma. |
| 17 | ESCs and iPSCs | >90% of total are GFAP+ by 24 weeks. Physiologically passive, uptake glutamate, propagate calcium waves, promote synaptogenesis, observed post engraftment into rodent brain. |
| 18 | ESCs and iPSCs | Majority of cells are GFAP+ after; day 20 sorting to CD184+/CD271-/CD44-/CD24+ neural stem cells, 3 weeks of culture, sorting to CD184+/CD44+ glial progentiors, and 6 passages. Observed post engraftment into rodent brain. |
| 24 | 8 week fetal tissue | 50% of total are GFAP+ up to 24 weeks. No functional studies. |
Based on these findings, we have devised an efficient and reproducible protocol for directing human pluripotent stem cells to immature astrocytes. Compared to primary astrocyte cultures and antibody-based sorting of glial progenitors18, the present protocol is simple in operation and yields a nearly pure population of astrocytes without sorting. A large quantity of the enriched human astrocytes can be generated because of the self-renewing property of PSCs and because of the ability of the human glial progenitors to expand for several months. Unlike primary astrocyte cultures, the human stem cell-differentiated astrocytes do not contain immune cells, such as microglia, which confounds experimental settings through activation of astrocytes. In contrast to serum-containing media, our protocol allows growth and differentiation of human astrocytes under chemically defined media. More importantly, the present protocol describes for the first time the procedure for generating astrocytes with specific regional and functional characteristics. Because of the human origin, the potential disadvantage of the protocol is the length of the long-term culture for obtaining mature astrocytes as compared to primary cultures. However, this shortcoming is readily overcome by the simplicity of the culture system and the long-term expandability of the progenitors. This protocol should also allow generation of astrocytes from human iPSCs with specific pathological traits.
Experimental Design
The astrocyte differentiation procedure encompasses three major stages (Fig. 1 ). The PSCs are first converted to multipotent neuroepithelial cells with the ability to be regionally specified, otherwise known as neural stem cells, in the first 3 weeks (this is discussed in detail in references19,20, where the procedural steps are described) followed by transition of the neuroepithelia to astroglial progenitors in the next 2 months, and finally generation of immature astrocytes after long term expansion and differentiation for one week. The entire process, except the first 4 days of transition period, involves the use of a simple neural medium with different additives at specific stages. In each of the stages, cell identity is determined by morphology and expression of cell type-specific markers. Astrocyte identity may be further ascertained by their functional attributes such as glutamate uptake and effects upon co -cultured neurons17.
Figure 1. Timeline of astrocyte generation.
Cells are directed through developmental identities of human pluripotent stem cells (PSC), embryoid bodies (EB), neuroepithelial cells (NE), progenitors, and then immature astrocytes through 3 stages composed of a total of 13 steps. Cell identity may be confirmed by immunostaining after CNTF treatment of dissociated cells (quality control)at the recommended timepoints. For regional patterning, morphogens can be added from days 10–21.
In the first stage, human PSC-derived neuroepithelial cells with the ability to be regionally patterned may be generated or obtained through various means, either by differentiation of PSCs cocultured with mouse embryonic fibroblasts19–21(Fig. 2a), differentiation of PSCs in feeder-free conditions22, or from commercially available sources. Under an adherent culture mode, neuroepithelial differentiation can be readily confirmed by the appearance of columnar epithelia that organize into neural tube-like rosettes at day 10–15 of differentiation (Fig. 2c). The non-neural colonies, indicated by lack of the typical rosettes (Fig. 2d), can be easily removed, thus enriching the neural population.
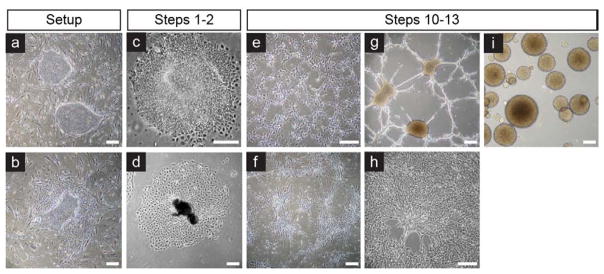
Figure 2. Stages of astroglial differentiation.
(a) PSCs should exhibit defined edges without (b) signs of differentiation. (c) Multilayered columnar cells are indicative of neuroepithelial origin, while (d) flattened or dark clusters are non neural. (e, g, i) Astroglial progenitors will display a stellate morphology when attached and eventually reform astrospheres in the presence of mitogens. (f, h) In contrast, non neural fibroblast-like cells will remain attached. Scale bars = 100 μm.
It is during the neuroepithelial differentiation period from day 8 to day 15 in which cells can be readily patterned to regional progenitors for generating diverse types of neurons. We recently discovered that it is during the same progenitor period that astrocyte subtypes are specified17. Therefore, application of morphogens at this period will allow generation of different classes of astrocytes, in the same manner as generating neuronal subtype s. For example, addition of sonic hedgehog will permit generation of astrocytes with ventral forebrain phenotypes while those generated without addition of morphogens exhibit cerebral cortical characteristics. Astrocytes generated from retinoic acid-patterned progenitors exhibit spinal cord phenotypes. This protocol is designed to allow generation of astrocytes with versatile and specific phenotypes.
In the second phase, the floating neural progenitor aggregates are expanded in suspension culture with EGF and FGF2, mitogenic factors commonly used for propagating neurosphere cultures23. Since the human neuroepithelial cells give rise to predominantly neurons in the following month before gliogenesis, the progenitor spheres are mechanically triturated weekly to reduce the neurogenic potential and to expand the gliogenic progenitors in the presence of the mitogens, a modification of a protocol for expansion of human fetal-derived neurospheres24. By about day 90, the vast majority of the progenitors can be identified by immunodetection with antibodies against S100β and CD44, both of which are expressed in astroglial progenitors and astrocytes, thus the floating aggregates are referred to as “astrospheres”. These glial progenitors may be expanded for additional months as astrospheres or as sphere-reforming monolayers (Fig. 2e, g, i).
Finally, in the third stage, the astroglial progenitors are differentiated to functional astrocytes by removal of mitogens, which will cause cells to exit the cell cycle. CNTF is added for 6 days to activate gliogenic gene expression, though CNTF is not necessary to induce GFAP expression at later time periods. The astrocyte identity is ascertained by expression of S100β and later GFAP( Fig. 3), with largely absence of neurons. If cells have been regionally patterned at the neuroepithelial stage, these will carry distinctive markers, such as Hoxb4 for posterior identity, or Nkx2.1 for ventral identity17, though expression of Nkx2.1 decreases over time. Functional properties of the in vitro produced human astrocytes may be verified by their electrophysiological properties (absent or minimal sodium currents, no action potentials, glutamate induced currents), glutamate uptake, calcium wave networks, and their ability to maintain the astrocytic phenotype after transplantation into a mouse central nervous system. In addition, astrocyte monolayers or conditioned media can support neuronal maturation.
Figure 3. Confirmations of astroglial differentiation with astrocyte markers.
The efficiency of astrocyte generation can be followed by the step wise expression of S100β and then GFAP (Hoechst nuclear stain = Ho). Eventually all cells express both markers and display a star shaped morphology. Scale bars = 50 μm.
Materials
Reagents
2-Mercaptoethanol(Sigma, M7522) Caution: 2-mercaptoethanol is combustible and very hazardous in case of contact with skin, contact with eyes, ingestion, and inhalation. Overexposure can result in death. Wear protective clothing, avoid contact, and keep away from sources of ignition.
Accutase (Innovative Cell Technology, cat. no. AT104)
Dispase (Gibco, cat. no. 17105-041)
D-MEM/F-12 (1X), liquid, 1:1 (Gibco, cat. no. 11330)
Dimethyl sulfoxide (DMSO, Sigma, cat. no. D2650)
Dulbecco’s Phosphate-Buffered Saline (D-PBS) (1X) liquid (Gibco, cat. no. 14190)
Epidermal Growth Factor human (EGF, Sigma, cat. no. E9644)
Fetal Bovine Serum, Certified, Heat-Inactivated (FBS, Gibco, 10082147)
GlutaMAX-I (100X), liquid (Gibco, 35050)
Heparin sodium salt from porcine intestinal mucosa (Sigma, cat. no. H3149)
Knockout Serum Replacement (KOSR, Gibco, cat. no. 10828) Critical: Stable up to 4 weeks at 4°Cafter thawing.
MEM Non-Essential Amino Acids Solution 10 mM (100X), liquid (NEAA, Gibco, cat. no. 11140)
N-2 Supplement (100X), liquid (Gibco, cat. no. 17502-048)
Natural Mouse Laminin, 1 mg/ml (Laminin, Invitrogen cat. no. 23017-015)
Paraformaldehyde (PFA, Sigma-Aldrich, cat. no. P6148) Caution: PFA is very hazardous in case of skin contact, eye contact, ingestion, and inhalation leading to irritation. Over exposure can lead to death. Wear protective clothing, avoid contact, and use in fume hood when working with the dry form.
Poly-L-ornithine hydrobromide(PLO, Sigma, cat. no. P3655)
Recombinant Human FGF basic (146 aa)(FGF2, R and D Systems, cat. no. 233 -FB)
Recombinant Human CNTF (CNTF, R and D Systems, cat. no. 257 -NT)
Recombinant Human Sonic Hedgehog (C24II) N-Terminus, CF(SHH, R and D Systems, cat. no. 1845-SH-025/CF)
Retinoic Acid (RA, Sigma, cat. no. R2625) Critical: Extremely sensitive to light, air, and oxidizing agents. Prepare immediately after opening ampule.
Trypan Blue Solution (Sigma, cat. no. T1854)
Antibodies
CD44 (BD Biosciences, cat. no. 550538)
GFAP (Dako, cat. no. Z0334, Chemicon, cat. no. MAB360)
S100 (Abcam, cat. no. ab868) Note: Antibody detects both A and B protein isoforms
S100β ( Abcam, cat. no. ab11178)
Biological material
Neuroepithelial cells (NE, see Reagent Setup for preparation)
Equipment
6 well and 24 well plates (Nunc, cat. no. 140675 and 142475)
9″ Disposable Borosilicate Glass Pasteur Pipets (Fisher Scientific, cat. no. 13-678-20D)
15-ml Centrifuge Tubes(RPI, cat. no. 163224)
CL2 Benchtop Centrifuge(Thermo, cat. no. 004260F)
Cryogenic vials (Corning, cat. no. 430488)
Freezing container (Nalgene, cat. no. 5100-0001)
Hausser Bright-Line counting chamber (Fisher Scientific, cat. no. 02-671-10)
HERAcell 150 CO2 Incubator(Thermo Scientific, cat. no. 50077952)
Phase Contrast Inverted Microscope (Nikon, Eclipse TS100)
Polystyrene Disposable Serological Pipets, 5 and 10 ml (Fisher Scientific, cat. no. 13-678-11D and 13-678-11E)
RD German Coverslip, 12mm, #1 (Bellco, cat. no. 1943-10012)
Steriflip (Millipore, cat. no. SCGP00525)
T25 flasks (TPP, cat. no. 90026)
Reagent setup
Ciliary neurotrophic factor (CNTF, 10 μg/ml)
Dissolve 10 μg into 1 ml of sterile D-PBS + 0.1% human or bovine serum albumin. Aliquot and store at −80°C.
Dispase (1 U/ml)
Dissolve 50 units into 50 ml of D-MEM/F-12. Warm at 37°C for 10 min to dissolve and filter with a 50 ml Steri-flip. Store at 4°C for up to 2 weeks.
Epidermal growth factor (EGF, 100 μg/ml)
Dissolve 200 μg into 2 ml of sterile D -PBS. Aliquot and store at −80°C.
Fibroblast growth factor 2 (FGF2, 100 μg/ml)
Dissolve 25 μg into 250 μl of sterile D-PBS. Aliquot and store at −80°C.
Freezing medium (1 ml)
Combine 800 μl of NM, 100 μl of FBS, and 100 μl of DMSO. Use immediately or alternatively prepare aliquots and store at −20°C.
Heparin (20 mg/ml)
Dissolve 20 mg into 1 ml of D-MEM/F-12. Aliquot and store at −80°C.
Neural medium (NM, 500 ml)
Combine 490 ml of D-MEM/F-12, 5 ml of NEAA, 5 ml of N-2 supplement, and 50 μl of heparin. Store at 4°Cfor up to 2 weeks.
Neuroepithelial cells (NE)
Follow steps 1–27 of reference 19 to differentiate human PSCs to neuroepithelial cells19. At step 27 (day 10), there should be columnar epithelial cells in each colony. Critical: Start with stem cell colonies that have no signs of differentiation (Fig. 2a, b). TROUBLESHOOTING
Paraformaldehyde (PFA, 4% wt/vol)
Dissolve 220 mg sodium phosphate monobasic and 1.22 g sodium phosphate dibasic into 50 ml water. In a fume hood, add 4 g PFA in 50 ml of 60°Cwater while stirring. Add 2–3 drops of 2N NaOH to dissolve. Combine the two solutions and cool to 22°C Adjust pH to 7.4 with HCl, filter with a Steriflip, and store at 4°Cup to two weeks. Caution: PFA is very hazardous in case of skin contact, eye contact, ingestion, and inhalation leading to irritation. Over exposure can lead to death.
Polyornithine (PLO, 1 mg/ml)
Dissolve 100 mg into 100 ml of boric acid buffer, pH 8.4. Filter with a Steriflip and store at −20°C.
PSC medium (PSCM, 500 ml)
Combine 390 ml of D-MEM/F-12, 100 ml of KOSR, 5 ml of NEAA, 5 ml of 100X GlutaMAX, and 3.6 μl of 2-mercaptoethanol. Store at 4°C up to 2 weeks. Caution: 2-mercaptoethanol is combustible and very hazardous in case of contact with skin, contact with eyes, ingestion, and inhalation. Over-exposure can result in death. Wear protective clothing, avoid contact, and keep away from sources of ignition.
Retinoic acid (RA, 100 mM, 1 mM)
Dissolve 30 mg into 1 ml of DMSO for a 100 mM solution. Aliquot and store at − 20°C. Before use, dilute 50 μl into 5 ml ethanol for a 1 mM solution and store at −20° up to 2 weeks. Critical: Keep RA protected from light as RA is light sensitive.
Sonic hedgehog (SHH, 100 μg/ml)
Dissolve 25 μg into 250 μl of sterile D-PBS for a 100 μg/ml stock. Aliquot and store at −80°C.
Equipment setup
Cell culture incubation conditions
Set the incubator at 37° and connect to a CO2 tank for a final concentration of 5%. Add water for humid conditions.
PLO and laminin coated coverslips
Acid etch the coverslips by overnight shaking in concentrated nitric acid, followed by 8 washes with distilled water over a period of 24 hours. Insert acid etched coverslips into each well of a 24 well plate in sterile conditions, add 0.5 ml of ethanol to each well to sterilize, and after the ethanol evaporates wash 2 times with sterile water.. Dilute the PLO stock solution 1:10 into sterile water. Add 75 μl of diluted PLO onto each coverslip and store in an incubator overnight. Remove PLO and dry 30 minutes, followed by 3 water washes. Dry again and store at −20°Cup to one month. 1 hour before use, dilute 25 μl of laminin solution into 1.25 ml of NM and add 50 μl onto each of the PLO treated coverslips. Do not let the solution spread off the coverslip onto the edge of the well. Store in an incubator for at least 1 hour prior to plating cells.
Procedure
Regional specificaton of neuroepithelial cells (optional) and formation of free floating aggregates. Timing 11 days. Days 10–21
-
1
Observe the neural differentiation culture under a phase contrast scope. The attached neuroepithelial cell colonies should form a uniform multilayer of cells (Fig. 2c). If cells become single layer or dark clusters of dead cells appear (Fig. 2d), the clusters should be mechanically removed and dispensed. Feed the culture with fresh media every 2–3 days with exchange of half of the media.
-
2
Without the addition of morphogens, human PSC derived neural cells will exhibit dorsal telencephalic characteristics25. If astrocytes of different regional identities are needed, add morphogens starting at the early stage of neuroepithelial cell differentiation (day 10 from the beginning of PSC differentiation). RA (1:2,000 dilution for 0.5 μM) or SHH (1:1000-1:200 dilution for 100–500 ng/ml) may be added to NM to caudalize or ventralize the progenitor cells, respectively, or a combination of both will generate caudal/ventral cells. These molecules should be continuously present in the NM until day 21 for complete patterning.
-
3
At day 15, a time point w hen multiple rosette formations are present throughout the NE culture, lift attached clusters. Typically, 3–10 clearly distinguishable rosettes are observed within one NE colony and the numbers vary between clusters. Note that non-neural colonies with absence of rosette formation should be mechanically removed before lifting the neural colonies to avoid contamination with non-neural cells. To lift, exert a force upon the clusters with a 1 ml pipette until they detach, without breaking excessively into single cells. Flat, non neural cells that migrate on the outer edge of the colonies should remain attached to the plate because of their higher degree of attachment. Critical: Minimize breaking up the neural colonies at this stage for maximum cell survival.
-
4
Transfer floating aggregates to a 15-ml conical tube and centrifuge for 2 minutes at 100g, a speed used for all subsequent steps. Remove old media (supernatant) by vacuum and resuspend the aggregates with prewarmed NM. Typically, from one 6-well plate of neuroepithelial cells, resuspend into 24 ml of NM. If morphogens are to be added to the culture system, dilute into NM before use.
-
5
Using a pipette, transfer 8 ml of resuspended aggregates into each of three T25 flasks. Different sized flasks may be used if desired. Critical: The neuroepithelial aggregates tend to stick on the inside of the pipette which results in a decreased yield. Therefore, rinse the pipette with media before transferring the aggregates to avoid losing spheres.
-
6
Every 2–3 days, replace media with fresh prewarmed NM +/− morphogens by tilting the flask to allow floating aggregates to settle to the bottom. Remove 75% of old media with a sterilized glass Pasteur pipette and replace with fresh NM media+/− morphogens. Critical: Clusters attached lightly to the flask should be lifted during addition of new media, simply by swirling the flask. If clusters are firmly attached or cells form a monolayer, only transfer the floating aggregates to a new flask and discard the attached cells. TROUBLESHOOTING
Induction of gliogenesis. Timing 69 days. Days 21–90
-
7
On day 21, replace media with NM + EGF and FGF2 (10 ng/ml each) to expand the spheres. Morphogens are no longer necessary at this late stage for regional patterning. As described above, 75% of media is replaced every 2–3 days and spheres tightly attached to the flask should be discarded during exchange of new flasks.
-
8
During days 21–90, break any large spheres into multiple smaller clusters. To break spheres, take large spheres into a flamed, curved Pasteur pipette that has been rinsed with media at least three times to avoid attachment of spheres to the wall of pipette. Then force the spheres out once or twice. A detailed procedure for making these pipettes has been described elsewhere21. Critical: Care should be taken to flame polish the Pasteur pipette to smooth edges and an appropriate aperture (0.2–0.5 mm in diameter), as a more narrow opening will cause excessive damage and cell death.
-
9
Between days 21–90, the EGF/FGF2 expanded and regularly triturated progenitor clusters will switch from neurogenic to gliogenic, and these aggregates are referred to as astrospheres. At day 90, verify the efficiency of gliogenesis by immunostaining. Expected outcome at this time period is that ~80% of cells are S100β+, ~80% are CD44+, and~10% are GFAP+ (Fig. 3). We regard these cells as astroglial progenitors/immature astrocytes17. At this stage, in addition to proceeding to step 10, there are the following optional steps: attaching monolayers of astroglial progenitors for maturation and experimentation (option A), immunostaining for cell type confirmation (option B), or freeze/thawing the cells to and from storage ( Box 1). Commonly, most cells are further expanded to generate large quantities of astrocytes (Step 10) whilst a small proportion is fixed for immunocytochemistry to track the differentiation process.
Box 1. Freezing and thawing astroglial progenitor cells. Timing 1 hour.
At any time point along the astroglial differentiation, cells can be frozen in liquid nitrogen for future use as dissociated cells or as astrospheres. Transfer cells to be frozen from one flask to a 15-ml tube and centrifuge at 100g.
Remove the supernatant and resuspend by slowly adding freezing media.
Transfer the cell suspension to cryogenic vials and store overnight at −80°C within a freezing container. The next day, store vials in liquid nitrogen.
For thawing cells, remove vials from liquid nitrogen and immediately place in a 37°C water bath.
Transfer cell suspension to a 15-ml tube and wash by slowly adding 10 ml NM prewarmed to 37°C. Centrifuge at 100g and remove supernatant.
Resuspend cells with NM + EGF + FGF2 and continuously culture in a flask as previously described.
A. Maturation of astrocytes. Timing 6 days
Prepare a 24-well plate containing PLO and laminin coated coverslips as described in equipment setup. Without laminin, cells will attach but do not flatten out on the glass, making proper detection of proteins difficult. Alternatively, 1% serum may be added in the initial plating (<24 hours) to induce attachment.
Transfer ~50 astrospheres to a 15-ml tube. Centrifuge at 100g, remove the supernatant, and digest with 0.5 ml of prewarmed Accutase at 37°Cfor 5 minutes. Immediately wash 2X with prewarmed D-MEM/F-12 to remove Accutase. Resuspend with 1 ml of NM.
Break astrospheres into single cells by triturating through a small pipette tip 3–5 times. Critical: If clusters still remain, they may be transferred to a second tube for additional trituration with ~50 μl of solution, and then returned to the original tube. This allows for full dissociation of clusters, while avoiding excessive trituration of dissociated cells, which may cause cell death. Count the number of single live cells using a standard hemocytometer.
For one 24-well plate containing laminin-coated coverslips, resuspend 240,000 cells into 1.2 ml of NM and add 50 μl directly onto each coverslip containing 50 μl of laminin solution, for a plating density of 10,000 cells/cm2. Do not allow the cell suspension to spill out of the coverslip. If fewer cells are obtained after digestion, recalculate to maintain a similar plating density for fewer coverslips.
Cells will attach between 3–5 hours after plating. After attachment, carefully add 0.5 ml of NM + CNTF (10 ng/ml) to each well and change media every 2 days for a total of 6 days. Cells will not proliferate during this period because of the absence of EGF and FGF2.
B. Preparing cells for immunocytochemistry and quality control. Timing 1 day
After 6–7 days of maturation (step B), remove media and wash 1X with prewarmed D-PBS. To fix cells, remove D-PBS and add ice cold 4% PFA prepared as described in Reagent setup. Transfer the plate to ice for 30 minutes.
-
Wash 3X with cold PBS.
PAUSEPOINT Plates can be stored at 4° up to one month or immediately used.
-
Proceed with immunocytochemistry with standard techniques to determine regional patterning efficiency and stage of differentiation.
TROUBLESHOOTING
Astrosphere maintenance and maturation. Timing 30+ days. Days 90 –120+
-
10
By day 90, astrospheres are primarily composed of cells with the ability to generate S100β+ immature astrocytes. Up to this stage, cells are maintained as spheres in order to easily select out any non-neural cells that attach to the flask. TROUBLESHOOTING
-
11
As described in steps 9Aii–iii, transfer all astrospheres from one flask to a tube and centrifuge, digest with Accutase, and dissociate to single cells to accelerate proliferation. Critical: do not use trypsin, as cells do not recover well from the digestion.
-
12
Resuspend cells at a concentration of at least 100,000 cells/ml and transfer to new T25 flasks to allow cells to attach. Astroglial progenitors will not proliferate well at a lower density. Astroglial progenitors tend to cluster together in the next few days, forming aggregates that w ill eventually detach and again generate free floating astrospheres (Fig. 2e, g, i). If rapidly dividing fibroblast-like cells are observed ( Fig. 2f, h), they produce a confluent layer without reforming spheres. Such a culture likely contains non neural or neural crest derivatives and will not generate astrocytes efficiently. TROUBLESHOOTING
-
13
Expand the astrospheres for at least 6–8 months in total, thus generating large quantities of astrocytes. The expansion rate usually slows down after 6 months of cultures. The maturation state of the astroglial progenitors in the astrospheres differs depending on the time of expansion. When measured at day 90, ~80% of cells will become S100β+ and ~10% are GFAP+ after removal of mitogens (Fig. 3). The S100β+/GFAP-cells are likely immature/non reactive progenitors since this population decreases with time. When the astrospheres after day 120 are differentiated upon removal of mitogens, nearly all the cells are positive for S100β and>90% of them are S100β/GFAP + within a week, and no neurons are present (Fig. 3). In addition, further quality control measures as described for day 90 (step 9, option B)may be taken to confirm lack of contaminating neuroepithelial cells, neurons, and oligodendrocytes with immunocytochemical detection of markers such as Pax6, Map2, and O4, respectively( Fig. 1).
Timing
Steps 1–6, Regional specificaton of neuroepithelial cells (optional) and formation of free floating aggregates:11 days.
Steps 7–9, induction of gliogenesis: 69 days.
Steps 10–13, Astrosphere maintenance and maturation: 30+days.
TROUBLESHOOTING
See Table 2 for troubleshooting guidance
Table 2.
Troubleshooting
| Step | Problem | Possible reason | Solution |
|---|---|---|---|
| Setup | PSCs are spontaneously differentiating | Poor quality MEF, inactive FGF2, inefficient iPSC generation | Quality control MEF and FGF2, remove all differentiating colonies |
| 6 | Majority of spheres tightly attaching to flask | Insufficient neural induction | Remove all non neuroepithelial cells from days 8–15 |
| 9 | Low percentage of S100β cells | Insufficient neural induction, non-neuronal expansion, poor immunostaining | Start with undifferentiated PSCs, remove all non neuroepithelial cells, quality control EGF and FGF2, use positive control for staining |
| 10 | Astrospheres exhibit dark/black interiors, indicative of cell death | Single astrospheres are allowed to grow too large | Remove astrospheres with dark interiors and triturate to break into smaller clusters |
| 13 | Astrocytes are not proliferating and not reforming astrospheres | Use of trypsin or serum, too much cell death during trituration, low plating density | Increase plating denstity to greater than 100,000 cells/ml, dissociate more gently |
Anticipated results
This protocol makes use of developmental principles for stepwise differentiation of Pax6+ neuroepithelial cells from human PSCs 19, which later bypass neuronal differentiation to develop into astroglial progenitor cells by expanding the progenitors with mitogens and by reducing cell to cell contact, and then finally become mature functional astrocytes. These astrocytes will, by default, display dorsal telencephalic properties unless instructed otherwise by addition of morphogens during the neuroepithelial stage. The majority of astroglial progenitors express S100β and CD44 and they can differentiate to GFAP-expressing maturing astrocytes (Fig. 3). Efficient neural induction is critical for this protocol because unwanted non neural cells will quickly proliferate and eventually overtake the culture (Fig. 2f, h). In the final stage, CNTF is applied during terminal differentiation as it is known to induce maturation through the janus kinase signaling pathway, though it may be optional to generate functional distinct astrocytes through activation of alternate pathways such as through bone morphogenetic protein signaling26,27.
The timeline of each stage reported here, though very reproducible with human ESC lines, may have discrepancies with the timing of human iPSC differentiation due to variations of neuroepithelial cell differentiation between lines15. Differentiation of disease specific iPSC lines with genetic variations of astroglial regulatory factors may also lead to differences, which would be advantageous for studying human disease mechanisms. Regardless, with efficient neural induction and subsequent proliferation of astroglial progenitors, a large number of astroglia at similar stages of development can be generated in a simplified manner. The temporal expression of astrocyte markers during differentiation of PSCs recapitulates the timing of human development28,29, highlighting its advantages as a model system for development. This protocol maybe combined with other methods, e.g., sorting of CD44 -expressing astroglial progenitors after the second month of differentiation, if desired.
Astrocytes generated with this protocol will exhibit functional and regional specific attributes, allowing a new system to study developmental processes of astrocytes, disease related mechanisms, and possible therapeutic discovery. In order to study the functions of these astrocytes in an in vivo system, PSC -derived astrocytes may be transplanted into a normal or diseased nervous system at different stages of development. By applying specific sets of morphogens at day 10–21 of this protocol, many more diverse subtypes of astrocytes may be generated17.
Acknowledgments
This study was supported by the ALS Association, National Institute of Neurological Disorders and Stroke (NS045926, NS057778, NS064578), National MS Society (NMSS TR-3761), NYSTEM (C024406), Bleser Family Foundation, Busta Family Foundation, Neuroscience Training Program (T32 GM007507) and partly by a core grant to the Waisman Center from the National Institute of Child Health and Human Development (P30 HD03352).
Footnotes
The authors declare that they have no competing financial interests.
R.K. and S.-C.Z. designed the protocol and wrote the manuscript. R.K. collected the data for figures.
BIBLIOGRAPHY
- 1.Matyash V, Kettenmann H. Heterogeneity in astrocyte morphology and physiology. Brain Res Rev. 2010;63:2–10. doi: 10.1016/j.brainresrev.2009.12.001. [DOI] [PubMed] [Google Scholar]
- 2.Zhang Y, Barres BA. Astrocyte heterogeneity: an underappreciated topic in neurobiology. Curr Opin Neurobiol. 2010;20:588–594. doi: 10.1016/j.conb.2010.06.005. [DOI] [PubMed] [Google Scholar]
- 3.Zhang SC. Defining glial cells during CNS development. Nat Rev Neurosci. 2001;2:840–843. doi: 10.1038/35097593. [DOI] [PubMed] [Google Scholar]
- 4.Rao MS, Mayer-Proschel M. Glial-restricted precursors are derived from multipotent neuroepithelial stem cells. Dev Biol. 1997;188:48–63. doi: 10.1006/dbio.1997.8597. [DOI] [PubMed] [Google Scholar]
- 5.Carpenter MK, et al. Enrichment of neurons and neural precursors from human embryonic stem cells. Exp Neurol. 2001;172:383–397. doi: 10.1006/exnr.2001.7832. [DOI] [PubMed] [Google Scholar]
- 6.Liu Y, et al. CD44 expression identifies astrocyte -restricted precursor cells. Dev Biol. 2004;276:31–46. doi: 10.1016/j.ydbio.2004.08.018. [DOI] [PubMed] [Google Scholar]
- 7.Morrison RS, de Vellis J. Growth of purified astrocytes in a chemically defined medium. Proc Natl Acad Sci U S A. 1981;78:7205–7209. doi: 10.1073/pnas.78.11.7205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mi H, Barres BA. Purification and characterization of astrocyte precursor cells in the developing rat optic nerve. J Neurosci. 1999;19:1049–1061. doi: 10.1523/JNEUROSCI.19-03-01049.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim HJ, McMillan E, Han F, Svendsen CN. Regionally specified human neural progenitor cells derived from the mesencephalon and forebrain undergo increased neurogenesis following overexpression of ASCL1. Stem Cells. 2009;27:390–398. doi: 10.1634/stemcells.2007-1047. [DOI] [PubMed] [Google Scholar]
- 10.Zhang SC, Wernig M, Duncan ID, Brustle O, Thomson JA. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat Biotechnol. 2001;19:1129–1133. doi: 10.1038/nbt1201-1129. [DOI] [PubMed] [Google Scholar]
- 11.Reubinoff BE, et al. Neural progenitors from human embryonic stem cells. Nat Biotechnol. 2001;19:1134–1140. doi: 10.1038/nbt1201-1134. [DOI] [PubMed] [Google Scholar]
- 12.Itsykson P, et al. Derivation of neural precursors from human embryonic stem cells in the presence of noggin. Mol Cell Neurosci. 2005;30:24–36. doi: 10.1016/j.mcn.2005.05.004. [DOI] [PubMed] [Google Scholar]
- 13.Tabar V, et al. Migration and differentiation of neural precursors derived from human embryonic stem cells in the rat brain. Nat Biotechnol. 2005;23:601–606. doi: 10.1038/nbt1088. [DOI] [PubMed] [Google Scholar]
- 14.Johnson MA, Weick JP, Pearce RA, Zhang SC. Functional neural development from human embryonic stem cells: accelerated synaptic activity via astrocyte coculture. J Neurosci. 2007;27:3069–3077. doi: 10.1523/JNEUROSCI.4562-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hu BY, et al. Neural differentiation of human induced pluripotent stem cells follows developmental principles but with variable potency. Proc Natl Acad Sci U S A. 2010;107:4335–4340. doi: 10.1073/pnas.0910012107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ruiz S, et al. High-efficient generation of induced pluripotent stem cells from human astrocytes. PLoS One. 2010;5:e15526. doi: 10.1371/journal.pone.0015526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Krencik R, Weick JP, Liu Y, Zhang ZJ, Zhang SC. Specification of transplantable astroglial subtypes from human pluripotent stem cells. Nat Biotechnol. 2011;29:528–534. doi: 10.1038/nbt.1877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yuan SH, et al. Cell-surface marker signatures for the isolation of neural stem cells, glia and neurons derived from human pluripotent stem cells. PLoS One. 2011;6:e17540. doi: 10.1371/journal.pone.0017540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hu BY, Zhang SC. Differentiation of spinal motor neurons from pluripotent human stem cells. Nat Protoc. 2009;4:1295–1304. doi: 10.1038/nprot.2009.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hu BY, Du ZW, Zhang SC. Differentiation of human oligodendrocytes from pluripotent stem cells. Nat Protoc. 2009;4:1614–1622. doi: 10.1038/nprot.2009.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xia X, Zhang SC. Differentiation of neuroepithelia from human embryonic stem cells. Methods Mol Biol. 2009;549:51–58. doi: 10.1007/978-1-60327-931-4_4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen G, et al. Chemically defined conditions for human iPSC derivation and culture. Nat Methods. 2011;8:424–429. doi: 10.1038/nmeth.1593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chojnacki A, Weiss S. Production of neurons, astrocytes and oligodendrocytes from mammalian CNS stem cells. Nat Protoc. 2008;3:935–940. doi: 10.1038/nprot.2008.55. [DOI] [PubMed] [Google Scholar]
- 24.Caldwell MA, et al. Growth factors regulate the survival and fate of cells derived from human neurospheres. Nat Biotechnol. 2001;19:475–479. doi: 10.1038/88158. [DOI] [PubMed] [Google Scholar]
- 25.Li XJ, et al. Coordination of sonic hedgehog and Wnt signaling determines ventral and dorsal telencephalic neuron types from human embryonic stem cells. Development. 2009;136:4055–4063. doi: 10.1242/dev.036624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rajan P, McKay RD. Multiple routes to astrocytic differentiation in the CNS. J Neurosci. 1998;18:3620–3629. doi: 10.1523/JNEUROSCI.18-10-03620.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Davies JE, et al. Transplanted astrocytes derived from BMP -or CNTF -treated glial-restricted precursors have opposite effects on recovery and allodynia after spinal cord injury. J Biol. 2008;7:24. doi: 10.1186/jbiol85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wilkinson M, Hume R, Strange R, Bell JE. Glial and neuronal differentiation in the human fetal brain 9–23 weeks of gestation. Neuropathol Appl Neurobiol. 1990;16:193–204. doi: 10.1111/j.1365-2990.1990.tb01156.x. [DOI] [PubMed] [Google Scholar]
- 29.Pal U, Chaudhury S, Sarkar PK. Tubulin and glial fibrillary acidic protein gene expression in developing fetal human brain at midgestation. Neurochem Res. 1999;24:637–641. doi: 10.1023/a:1021096224161. [DOI] [PubMed] [Google Scholar]