Abstract
Development of multidrug resistance (MDR) is a major deterrent in the effective treatment of metastatic cancers by chemotherapy. Even though MDR and cancer invasiveness have been correlated, the molecular basis of this link remains obscure. We show here that treatment with chemotherapeutic drugs increases the expression of several ATP binding cassette transporters (ABC transporters) associated with MDR, as well as epithelial–mesenchymal transition (EMT) markers, selectively in invasive breast cancer cells, but not in immortalized or non-invasive cells. Interestingly, the mere induction of an EMT in immortalized and non-invasive cell lines increased their expression of ABC transporters, migration, invasion, and drug resistance. Conversely, reversal of EMT in invasive cells by downregulating EMT-inducing transcription factors reduced their expression of ABC transporters, invasion, and rendered them more chemosensitive. Mechanistically, we demonstrate that the promoters of ABC transporters carry several binding sites for EMT-inducing transcription factors, and overexpression of Twist, Snail, and FOXC2 increases the promoter activity of ABC transporters. Furthermore, chromatin immunoprecipitation studies revealed that Twist binds directly to the E-box elements of ABC transporters. Thus, our study identifies EMT inducers as novel regulators of ABC transporters, thereby providing molecular insights into the long-standing association between invasiveness and MDR. Targeting EMT transcription factors could hence serve as novel strategies to curb both metastasis and the associated drug resistance.
Keywords: ABC transporters, multidrug resistance, epithelial–mesenchymal transition, EMT transcription factors, Twist
The highly orchestrated molecular circuitries that ensure normal functioning of each cell are disrupted in cancer, making it a disease of uncontrolled cell proliferation. Although decades of research have gone into understanding the causes and mechanisms of cancer, yet relatively little progress has been made to effectively cure this disease. The development of therapies to cure cancer is further complicated by the emergence of drug resistance. For an effective treatment of cancers, conventional chemotherapy employs the use of a combination of drugs.1 However, instead of succumbing, cancer cells often remain refractory to chemotherapeutic drugs owing to the development of drug resistance.
Cancers can either be inherently drug resistant before drug exposure or can acquire drug resistance after the administration of drugs.2 Those that respond to chemotherapy at the start of the treatment can often simultaneously become resistant to different classes of drugs resulting in multidrug resistance (MDR).2 Development of MDR is also the leading cause of cancer recurrence, with the resultant cancers behaving more aggressive than at the start of treatment. Several adaptive mechanisms contribute to drug resistance, including activation of DNA repair pathways, alteration of drug targets, decreased uptake of chemotherapeutic drugs,2, 3 and most significantly, an increased active efflux of drugs mediated by transporters belonging to the ATP binding cassette (ABC transporters) superfamily of proteins.4
ABC transporters form the largest transportome in the human genome.4 They are ubiquitously and heterogeneously expressed in various body tissues and important pharmacological barriers where they have a pivotal role in host cell detoxification and protection of the body against xenobiotics.1, 2 ABC transporters are also overexpressed in several cancers.1, 2 These transporters actively efflux a wide spectrum of commonly employed chemotherapeutic drugs like estramustine, mitoxantrone, anthracyclines, vinca alkaloids, taxanes, thiopurines, and so on.5 Recently, as many as 16 ABC transporters have been implicated in MDR.5 Therefore, understanding the mechanisms that regulate ABC transporter expression becomes imperative in addressing the problem of MDR.
Increased ABC transporter expression has been correlated with aggressive and invasive cancers, which also tend to be more chemoresistant.5, 6, 7 One of the early steps in the invasion-metastasis cascade includes epithelial–mesenchymal transition (EMT) through which polarized epithelial cells undergo major phenotypic changes and acquire a more mesenchymal morphology.8, 9, 10 EMT can be triggered by a myriad of stimuli,9 several of which ultimately culminate in the overexpression of transcription factors like Snail, Twist, Slug, and so on.10 Overexpression of some of these transcription factors has also been associated with chemoresistance, although their depletion has been shown to increase drug sensitivity.11, 12, 13 Even though these studies establish a strong correlation between EMT and drug resistance, a clear understanding of the molecular basis for the association between invasion and drug resistance is still lacking.
In this study, we have investigated the role of EMT-inducing transcription factors in drug resistance mediated by ABC transporters in breast cancer cells. We demonstrate that the very transcription factors that lead to EMT and invasion also orchestrate the overexpression of drug transporters by directly modulating their promoter activity, thus providing a novel molecular mechanism for the long-standing association between invasiveness and drug resistance.
Results
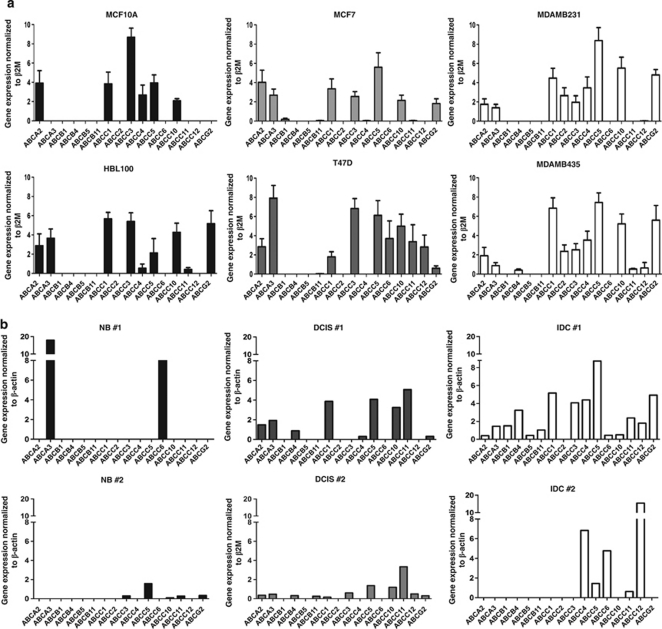
Heterogeneous expression of ABC transporters in breast cells
To assess the basal level of expression of the 16 ABC transporters implicated in drug resistance,5 we undertook a detailed reverse transcriptase polymerase chain reaction (RT-PCR)-based analysis in nine breast epithelial cell lines comprising of immortalized, non-invasive and invasive cell types for our study. We found a large heterogeneity in the number and level of expression of ABC transporters, but not a significant difference across cell types (Figure 1a and Supplementary Table S1 and Figure S1A). Expression analysis at the protein level for three ABC transporters, viz, ABCB1, ABCC1, and ABCG2, also revealed a similar heterogeneity (Supplementary Figure S1B). To assess if a similar heterogeneity exists in vivo, we analyzed primary tissues from untreated normal, ductal carcinoma in situ (DCIS), and invasive ductal carcinoma (IDC) patient samples. Among the primary samples also, heterogeneity in both number and level of expression of ABC transporters was observed (Figure 1b and Supplementary Figure S1C). In addition, the DCIS and the IDC samples expressed a higher number of ABC transporters in comparison to the normal tissue (Figure 1b and Supplementary Figure S1C), suggesting that the process of tumor progression in vivo may lead to the upregulation of ABC transporter expression.
Figure 1.
Breast epithelial cells have a heterogeneous expression of ABC transporters. (a) Representative graphs showing relative mRNA expression of ABC transporters in immortalized (MCF10A and HBL100), non-invasive (MCF7 and T47D), and invasive (MDAMB231 and MDAMB435) breast epithelial cell lines as analyzed by RT-PCR, n=3. (b) Representative graphs showing relative mRNA expression of ABC transporters in primary normal (NB), DCIS, and IDC breast samples as analyzed by RT-PCR. n=3 for NB, n=6 for DCIS, and n=3 for IDC samples
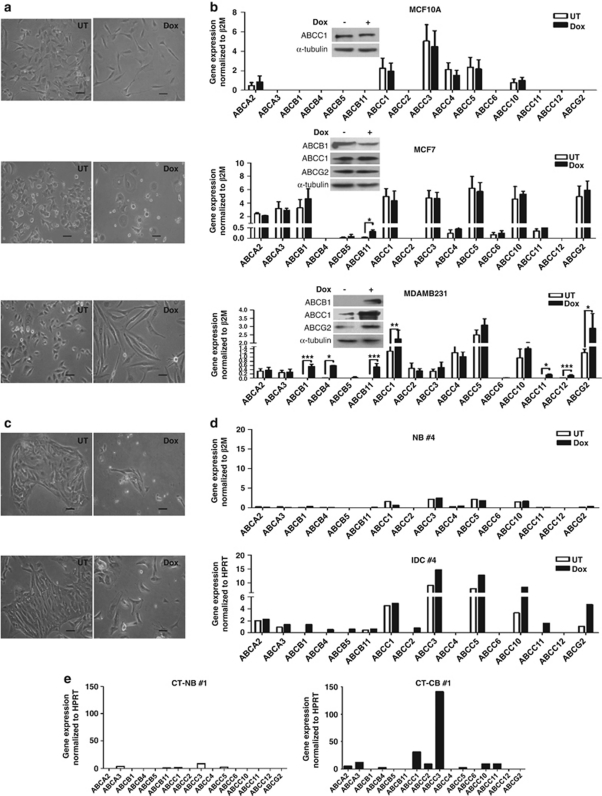
Effect of chemotherapeutic drug treatment on ABC transporter expression
Previous studies have shown an increase in drug resistance phenotype on chemotherapeutic drug treatment.2 To understand the effect of chemotherapeutic treatment on the expression of ABC transporters, we exposed the panel of immortalized, non-invasive and invasive cells to an 1-week treatment with doxorubicin (Dox), a commonly used chemotherapeutic drug in breast cancer treatment regimens, and examined their expression of 16 ABC transporters.5 The time of treatment and the concentration of drug used were determined based on time-course analyses and 3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) cytotoxicity assays, respectively, as detailed in Materials and Methods.
Dox treatment of two immortalized cell lines, MCF10A (Figure 2b) and HBL100 (Supplementary Figure S2A), did not significantly alter the expression of any of the 16 ABC transporters. Dox treatment of the two non-invasive cell types, MCF7 and T47D, led to an increase in only one ABC transporter in each cell line, viz, ABCB11 in MCF7 (Figure 2b) and ABCC5 in T47D (Supplementary Figure S2A) cells. Interestingly, Dox treatment of the invasive cell types, MDAMB231 (Figure 2b) and MDAMB435 (Supplementary Figure S2A), led to a significant increase in as many as 7–9 ABC transporters. To assess if this effect was specific to Dox, we treated MCF10A, MCF7 and MDAMB231 cells with yet another drug, mitoxantrone. Although no change was observed in either MCF10A or MCF7 cells, a significant increase in as many as six ABC transporters was observed in MDAMB231 cells (Supplementary Figure S2B).
Figure 2.
Dox treatment upregulates ABC transporter expression only in invasive cells. (a) Phase-contrast microscopic images taken after 1 week of untreated (UT) and doxorubicin-treated (Dox) MCF10A, MCF7, and MDAMB231 breast epithelial cell lines. Scale bar=100 μm. (b) Graphs represent relative mRNA expression of 16 ABC transporters after 1-week Dox treatment in MCF10A, MCF7, and MDAMB231 breast epithelial cell lines, n=3. Statistical significance for all the experiments was determined using ratio t-test. Error bars denote ±S.E.M. *P<0.05; **P<0.01; ***P<0.001. The insets represent immunoblot analysis of whole-cell lysates for the expression of three ABC transporters, viz, ABCB1, ABCC1, and ABCG2 in the absence or presence of Dox. (c) Phase-contrast microscopic images of primary breast tissue-derived cells in the presence (Dox) or absence (UT) of Dox for 7 days; scale bar=100 μm. (d) Representative graph showing relative mRNA expression of 16 ABC transporters after 1-week Dox treatment in primary normal- and tumor-derived breast epithelial cells, n=4. (e) Representative graph showing relative mRNA expression of ABC transporters in chemotherapy-treated patient-derived primary normal (CT-NB) and tumor (CT-CB) breast samples. n=2 for normal and n=3 for tumor samples
To assess the behavior of primary samples, cells derived from patient biopsies were treated with Dox for a week in culture (Figure 2c) and then assessed for ABC transporter expression. Although an appreciable increase in ABC transporters was not observed in Dox-treated primary normal breast cells, many ABC transporters were upregulated in the Dox-treated primary breast cancer cells (Figure 2d and Supplementary Figure S2C). To further assess if these in vitro drug treatments mimicked in vivo behavior, we undertook the expression analysis of ABC transporters in several chemotherapy-treated primary patient samples. In concordance with previous reports,1 our results revealed that chemotherapeutic treatment significantly upregulated the expression of many ABC transporters in the cancer samples (Figure 2e and Supplementary Figure S2D).
Taken together, our data demonstrated that the presence of chemotherapeutic drugs causes an increased expression of ABC transporters only in invasive breast cancer cells, revealing a strong correlation between the invasive phenotype and ABC transporter-mediated drug resistance phenotype. These data further suggested that the cellular machinery required to orchestrate the upregulation of ABC transporters in response to drug treatment is inherent within the invasive cells.
Effect of chemotherapeutic drug treatment on EMT and invasion
Recent studies have suggested a correlation between EMT and chemoresistance.14 To assess if 1-week Dox treatment has an effect on EMT in our system, we undertook RT-PCR and western blot analysis of several markers of EMT. Upon Dox treatment, no significant change in EMT markers was observed in immortalized (MCF10A) and non-invasive (MCF7) cell lines; however, a significant increase was observed in the invasive cell line (MDAMB231) (Figures 3a and b). Similar results were obtained for another set of immortalized (HBL100), non-invasive (T47D) and invasive (MDAMB435) cell lines (Supplementary Figure S3A). This was also accompanied with the acquisition of a more mesenchymal phenotype in the invasive cells (Figure 2a). Another hallmark feature of EMT is an increase in the migratory capability of cells.8 In comparison to the untreated cells, a previous treatment with Dox for 72 h caused a threefold increase in motility in MDAMB231 cells (Figures 3c and d) and a twofold increase in motility in MDAMB435 cells (Supplementary Figure S3B) in wound healing assays. To test whether Dox treatment can also cause an increase in the invasive properties of MDAMB231 cells, we undertook an RT-PCR-based analysis of several matrix metalloproteinase (MMP) markers that are known to be overexpressed during metastasis of cancer cells. A significant increase in the expression of several MMPs (MMP1, MMP2, MMP3 and MMP13) was observed on Dox treatment (Figure 3e). To further validate this, a functional in vitro assay of invasion through Matrigel was undertaken. As seen in Figure 3f, Dox treatment significantly increased the invasive potential of only MDAMB231 cells and not that of MCF10A or MCF7 cells. Collectively these results showed that drug treatment further potentiates the migratory and invasive potential of invasive breast cancer cell lines.
Figure 3.
Dox treatment of invasive cells leads to the upregulation of EMT markers and invasiveness. (a) Graphs represent relative mRNA expression of EMT markers after 1-week Dox treatment in MCF10A, MCF7, and MDAMB231 cells, n=3. Error bars denote ±S.E.M. *P<0.05. (b) Immunoblot analysis of whole-cell lysates of MCF10A, MCF7, and MDAMB231 cells for the expression of EMT markers in the absence or presence of Dox. (c) Photomicrographs represent cell migration of untreated (UT) or doxorubicin-treated (Dox) MDAMB231 cells in wound healing assay at 0 and 24 h. Scale bar=500 μm. (d) Graph represents distance migrated by cells in the experiment (c) calculated as a difference of distance between the two edges of the wounds at 0 and 24 h measured in μm using the ProgRes capture software, n=8. Error bars denote ±S.E.M. **P<0.01. (e) Graph represents relative mRNA expression of MMP markers after 1 week of Dox treatment in MDAMB231 cells, n=3. Error bars denote ±S.E.M. *P<0.05; **P<0.01. (f) Graph represents invasion index calculated as ratio of percentage cell invasion of Dox-treated cells and -untreated control cells through Matrigel in Boyden chamber assay; n=2 for MCF10A and MCF7, and n=3 for MDAMB231. Error bars denote ±S.E.M. *P<0.05
Effect of EMT induction on ABC transporter expression
As our earlier results demonstrated that drug treatment leads to an increase in the expression of both ABC transporters and the invasive phenotype in only invasive cells, we hypothesized that the molecular mechanisms that regulate EMT and invasiveness could also co-regulate ABC transporter expression. To test this, we asked if induction of EMT in a non-invasive cell line will elicit an increase in ABC transporter expression. To address this, we induced EMT in non-invasive MCF7 cells by TGFβ treatment.9 One week of TGFβ treatment led to a significant increase in the levels of TGFBIp, a read out of TGFβ signaling (Figure 4a), and a significant increase in levels of several mesenchymal markers (Figure 4a), indicative of EMT induction. A subsequent analysis of ABC transporters in these cells revealed a significant increase in the levels of five ABC transporters compared with untreated controls (Figure 4b). To further strengthen our hypothesis that EMT regulates ABC transporter-mediated drug resistance, we chose to induce EMT in MCF7 cells by a second approach involving the overexpression of EMT-inducing transcription factors. MCF7 cells were transiently transfected with Twist expressing vector. An RT-PCR analysis after 48 h of transfection confirmed the overexpression of Twist, and the upregulation of various mesenchymal markers, thereby confirming the induction of EMT (Figure 4c). Further, we observed that the mere overexpression of Twist led to a concomitant upregulation of several of the 16 ABC transporters (Figure 4d). Similar results were obtained with transient transfections of other EMT-inducing transcription factors such as Snail (Supplementary Figures S4A and B) and FOXC2 (Supplementary Figures S4C and D).
Figure 4.
EMT induction upregulates ABC transporter expression. Graphs represent relative mRNA expression of EMT markers (a) and ABC transporters (b) after 1-week treatment of MCF7 cells with TGFβ, n=4. Error bars denote ±S.E.M. *P<0.05; **P<0.01. Graphs represent relative mRNA expression of EMT markers (c) and ABC transporters (d) in MCF7 cells transiently overexpressing mTwist, n=4. Error bars denote ±S.E.M. *P<0.05; **P<0.01. (e) Phase-contrast images of parent HMLE cells and HMLE cells stably overexpressing mTwist (HMLE-mTwist); scale bar=100 μm. (f) Graph represents distance migrated by cells in wound migration assay calculated as a difference of distance between the two edges of the wounds at 0 and 24 h measured in μm using the ProgRes capture software, n=4. Error bars denote ±S.E.M. **P<0.01. (g) Graph represents cell invasion in arbitrary units (AU) calculated as a ratio of cells invaded through Matrigel to the total number of cells plated, n=3. Error bars denote ±S.E.M. **P<0.01. Graphs represent relative mRNA expression of EMT markers (h) and ABC transporters (i) in HMLE cells stably overexpressing mTwist, n=3. Error bars denote ±S.E.M. *P<0.05. (j) Dose–response curves of parent HMLE cells versus HMLE cells stably expressing mTwist/FOXC2/Snail treated with doxorubicin for 48 h. The dotted lines represent the IC50 values. (k) Graph represents relative mRNA expression of 16 ABC transporters in HMLE-mTwist cells after 1 week of Dox treatment, n=3. Error bars denote ±S.E.M. *P<0.05; **P<0.01; ***P<0.001
To address if this upregulation of ABC transporters in response to EMT induction is restricted to non-invasive cancer cells, we tested yet another cell type, viz, immortalized human mammary epithelial cells (HMLE).15 HMLE cells stably overexpressing Twist (HMLE-Twist) were generated by retroviral transduction. These cells showed a mesenchymal morphology, increased migration and invasion, upregulation of various EMT markers, and downregulation of E-cadherin (Figures 4e–h). Concomitantly, we observed an increase in the levels of five ABC transporters (Figure 4i). Similar results were obtained with HMLE cells transduced with Snail- and FOXC2-expressing retroviruses (Supplementary Figures S4E–I). In addition, HMLE cells stably overexpressing Twist, FOXC2, or Snail tolerated a 10-fold higher Dox concentration in comparison with the uninfected control HMLE cells, as assessed by an MTT cytotoxicity assay (Figure 4j). Furthermore, similar to invasive cell types, HMLE-Twist cells showed further upregulation of several ABC transporters on treatment with Dox (Figure 4k). Thus, an immortalized cell line that otherwise is sensitive to chemotherapeutic drugs can be rendered drug resistant by the mere introduction of EMT-inducing transcription factors. Taken together, these results clearly demonstrated that EMT induction simultaneously upregulated the expression of several ABC transporters in non-invasive cancer and immortalized cell lines.
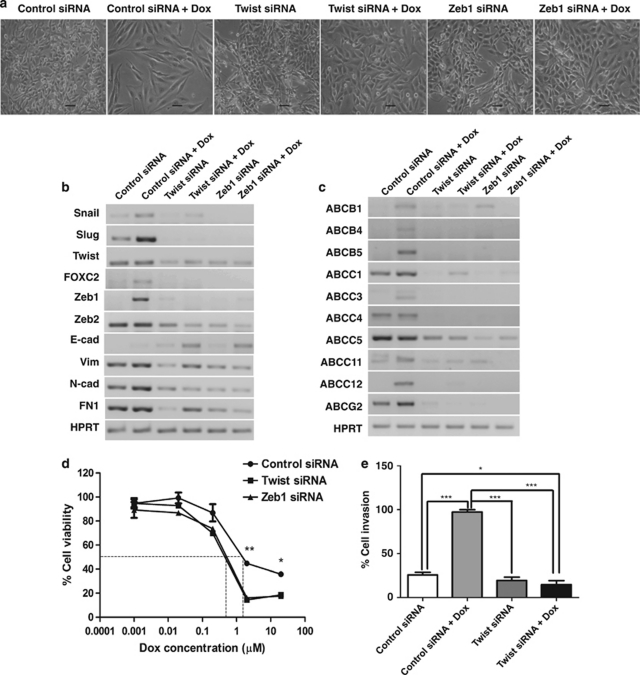
Reversal of EMT leads to increased drug sensitivity
To further test the specificity of the link between EMT and ABC transporter expression, we analyzed the effect of downregulating EMT-inducing transcription factors using the RNAi approach. We found that transfection of invasive MDAMB231 cells with siRNA oligos targeting Twist (Twist siRNA) or Zeb1 (Zeb1 siRNA) led to a reversal of the EMT phenotype, as judged by the emergence of epithelial looking colonies (Figure 5a), upregulation of E-cadherin, as well as reduction in the levels of several EMT markers (Figure 5b). Twist and Zeb1 siRNA also prevented Dox-mediated upregulation of EMT-inducing transcription factors like Snail, Slug, FOXC2, and Zeb2 (Figure 5b). Further, both Twist (Figure 5c and Supplementary Figure S5) and Zeb1 siRNA (Figure 5c) decreased the basal levels of ABC transporters in MDAMB231 cells. In addition, Twist and Zeb1 siRNA significantly prevented the Dox-mediated induction of several ABC transporters compared with control siRNA transfections (Figure 5c). Twist and Zeb1 knockdown also reduced their chemoresistance (Figure 5d). Furthermore, on Twist knockdown, MDAMB231 cells failed to show Dox-mediated increase in invasion (Figure 5e). Thus, these set of experiments confirmed that the upregulation of ABC transporters increase in migration, invasion, and chemoresistance in response to Dox treatment, via, EMT-inducing transcription factors.
Figure 5.
Reversal of EMT leads to increased drug sensitivity. (a) Photomicrographs of MDAMB231 cells transfected with control, Twist, or Zeb1 siRNA in the presence and absence of Dox treatment for 72 h. Scale bar=100 μm. Gel pictures depict RT-PCR analysis of EMT markers (b) and ABC transporters (c) in MDAMB231 cells transfected with control, Twist, or Zeb1 siRNA in the presence and absence of Dox treatment for 72 h. (d) Dose–response curves of MDAMB231 cells transfected with control, Twist, or Zeb1 siRNA, and subsequently treated with varying concentrations of Dox for 48 h. The dotted lines represent the IC50 values. Error bars denote ±S.E.M. *P<0.05; **P<0.01. (e) Graph represents percentage cell invasion through Matrigel in Boyden chamber assay for MDAMB231 cells transfected with control or Twist siRNA, and subsequently treated with Dox for 72 h, n=3. Error bars denote ±S.E.M. *P<0.05; ***P<0.001
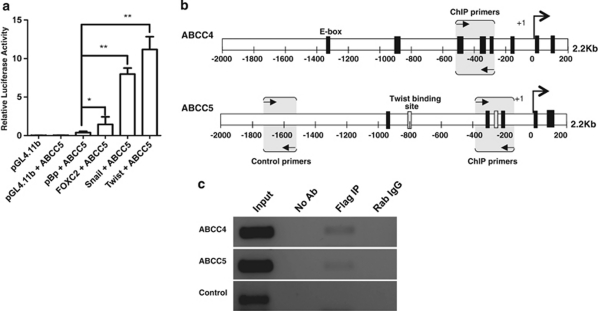
EMT transcription factors directly modulate ABC transporter expression
Snail, Twist, and FOXC2 have been implicated as transcriptional activators for genes like CDK inhibitor p15INK4b, Akt2 and plasminogen activator inhibitor type I, respectively.16, 17, 18, 19 To assess whether these EMT factors could act as transcriptional activators for ABC transporters as well, we undertook a promoter–reporter assay for ABCC5. MCF7 cells were co-transfected with constructs encoding for ABCC5-Luc along with one of the following vectors: pBp-empty vector, pBp-Snail, pBp-mTwist, or pBp-FOXC2. Our results demonstrated that all the three EMT-inducing transcription factors, Snail, Twist, and FOXC2, were able to significantly increase the promoter activity of ABCC5 in comparison to the pBp-empty vector control (Figure 6a).
Figure 6.
EMT-inducing transcription factors regulate ABC transporter expression. (a) Graph represents luciferase assay undertaken in MCF7 cells to assess the ABCC5 promoter activity (ABCC5-Luc; firefly luciferase) on co-transfection with pRL-TK (Renilla luciferase) and pBp-empty vector/Snail/mTwist/FOXC2. After 48 h culture, firefly luciferase activity was measured and normalized to Renilla luciferase activity and depicted as relative luciferase units (RLU), n=3. Error bars denote ±S.E.M. *P<0.05; **P<0.01. (b) Schematic of E-box elements in 2.2 kb long promoter regions of ABCC4 and ABCC5 genes. (c) Gel picture represents PCR carried out on ChIP undertaken in MCF7 cells transiently overexpressing Flag-Twist using anti-Flag Ab. PCR was carried out with the eluted DNA fragments using primers depicted in (b). No antibody (No Ab) served as a negative control and rabbit IgG (Rab IgG) served as an isotype control. Control primers were designed ∼700 bp upstream of E-box elements in the ABCC5 promoter and served as a specificity control
Further, a bioinformatic-based analysis of the promoter regions of the 16 ABC transporters revealed the presence of several binding sites for EMT-inducing transcription factors like Snail, Slug, Twist (E-boxes), FOXC2 (forkhead domains), E12, and E47 (Table 1). To assess whether the EMT-inducing transcription factors can directly bind to the consensus binding elements present in the promoters of ABC transporters (Figure 6b), we undertook a chromatin immunoprecipitation (ChIP) assay with MCF7 cells transiently transfected with pCDNA3-Flag-Twist construct. The ChIP assay revealed that Flag-Twist indeed binds specifically to E-boxes present in the promoter region of ABCC4 and ABCC5 genes (Figure 6c). Taken together, these results revealed that EMT-inducing transcription factors can directly bind to the promoters of ABC transporters and thus regulate their expression.
Table 1. ABC transporter promoters possess binding sites for EMT transcription factors.
|
Binding sites for EMT-inducing transcription factors |
|||||
|---|---|---|---|---|---|
| ABC genes | E-Box | Twist | FKHD | E12 | E47 |
| ABCA2 | 15 | 9 | 2 | 2 | 3 |
| ABCA3 | 16 | 3 | 6 | 2 | 2 |
| ABCB1 | 3 | 1 | 22 | 0 | 1 |
| ABCB4 | 6 | 6 | 7 | 2 | 3 |
| ABCB5 | 1 | 4 | 13 | 0 | 3 |
| ABCB11 | 1 | 4 | 12 | 0 | 2 |
| ABCC1 | 2 | 11 | 12 | 1 | 6 |
| ABCC2 | 1 | 3 | 13 | 0 | 1 |
| ABCC3 | 4 | 4 | 2 | 1 | 2 |
| ABCC4 | 11 | 3 | 3 | 0 | 1 |
| ABCC5 | 6 | 2 | 9 | 0 | 2 |
| ABCC6 | 4 | 2 | 11 | 1 | 0 |
| ABCC10 | 3 | 2 | 14 | 0 | 1 |
| ABCC11 | 0 | 8 | 23 | 1 | 2 |
| ABCC12 | 3 | 5 | 9 | 2 | 1 |
| ABCG2 | 6 | 1 | 12 | 1 | 1 |
Bioinformatic analysis (using the MatInspector software) of promoters of ABC transporters for the presence of binding sites for EMT-inducing transcription factors.
Discussion
Cancer invasiveness has long been associated with increased drug resistance; yet, little is known about the molecular mechanisms linking the two phenomena. We show here that the transcription factors that mediate EMT, a process intricately associated with cancer invasion-metastasis cascade, also regulate the expression of ABC family of drug transporters that mediate MDR.
Heterogeneous expression of ABC transporters by cancer cells
Several studies have identified ABC transporters as important players in cancer chemoresistance5 leading to the development of anticancer strategies based on targeting these transporters.1 Despite addressing the toxicity issues, inhibitors of ABC transporters have proved to be largely disappointing in clinical trials.5 One possible reason for this could be that most therapies till date have attempted to target only a small subset of ABC transporters, namely ABCB1, ABCC1, or ABCG2,1, 5, 20 whereas recent studies have demonstrated the involvement of as many as 16 ABC transporters in cancer drug resistance.5 Consistent with this, our study reveals the existence of a large heterogeneity in the expression of several of these transporters across established breast cancer cell lines and primary tumor-derived cells. Also, in response to chemotherapeutic drugs, several breast cancer cells showed an increased expression of a large number of ABC transporters simultaneously. Furthermore, different cancer cell lines and primary samples responded differently to the same drug by upregulating diverse types of ABC transporters. As ABC transporters have broad substrate recognition and redundancy in function, our results suggest that future therapies should be designed targeting several of these 16 ABC transporters as an armamentarium of ‘MDR' proteins.
EMT factors: link between invasion and drug resistance
EMT is an evolutionarily conserved mechanism characterized by the attainment of a mesenchymal morphology, invasiveness, and changes in the expression of epithelial and mesenchymal genes.8 Cells undergoing EMT become more migratory and invasive,8 and invasive cancers are known to be more drug resistant.14, 21 Furthermore, the mesenchymal-like cells within a cancer have been shown to be more drug resistant than the epithelial-like cells.21 On the other hand, drug-resistant cancer cells derived by long-term exposure to drugs have been shown to acquire mesenchymal morphology, increased migratory and invasive potential, and an increased expression of EMT-regulating transcription factors.22, 23, 24, 25 Our data revealed that even an 1-week treatment of invasive cancer cells suffices to increase their expression of EMT markers. Furthermore, induction of EMT in immortalized/non-invasive cells led to increased expression of ABC transporters and drug resistance, whereas a reversal of EMT in invasive cells led to a concomitant decrease in ABC transporter expression and chemoresistance, thus establishing a strong link between EMT and ABC transporter expression. Similar to our findings, a recent study has reported that Dox treatment of cancer cells enhances their invasion and drug-resistant phenotype selectively in cells undergoing EMT.26 We additionally demonstrate that several ABC transporters contain binding sites for EMT regulators like Twist, Snail, Slug, FOXC2, and E12/E47, and at least three of these factors, Snail, Twist, and FOXC2, can modulate the promoter activity of ABC transporters. A ChIP experiment further confirmed the role of Twist in upregulating the expression of ABCC4 and ABCC5 transporters by directly binding to their promoters. Thus, our data demonstrate for the first time that the very transcription factors that lead to cancer invasiveness by triggering EMT directly modulate the expression of ABC family of drug transporters, thereby contributing to chemoresistance.
EMT, cancer stem cells, and drug resistance
Although our data provide a mechanistic link between EMT and drug resistance, the question of how exposure to drugs induces EMT remains unclear. One possibility is that the drugs influence signal-transduction pathways upstream of EMT. Indeed activation of TGFβ signaling, a potent inducer of EMT, in the presence of chemotherapeutic drugs has recently been reported.27 An alternate hypothesis, and an emerging theme in cancer biology, is that the drugs kill most of the cells in the population leaving behind a small subset of cells, called the cancer stem cells (CSCs), characterized by their mesenchymal phenotype and increased expression of ABC transporters and stemness markers.9, 21, 28, 29 Consistent with this, our data revealed that drug-treated cells appeared more mesenchymal at the end of a week, and revealed enhanced expression of stemness markers that are currently being investigated. Furthermore, several signal-transduction pathways involved in the regulation of stem cell self-renewal (such as Notch, Wnt, and TGFβ) have also been associated with EMT and chemoresistance.27, 30, 31
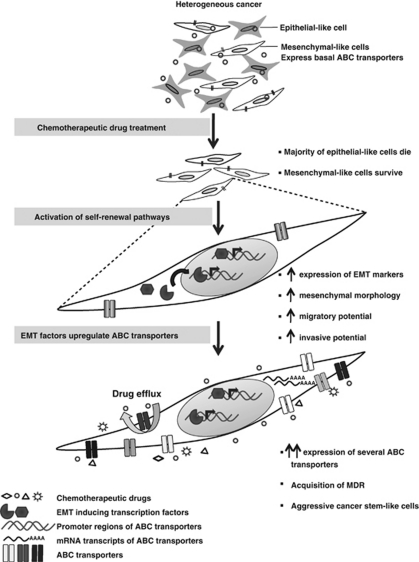
More recently, EMT markers and mesenchymal phenotype have also been correlated with stemness.32, 33 Based on all this, and in light of our observation that the previous presence of an EMT machinery within a cell is a prerequisite for upregulating their ABC transporters in response to drugs, we suggest the following model: when a heterogeneous population of cancer cells is treated with chemotherapeutic drugs, the majority of the epithelial-like cells die. The mesenchymal-like cells survive owing to a basal expression of ABC transporters. Drug-induced activation of self-renewal pathways within these cells causes further increase in the expression of EMT factors, which in turn upregulate the expression of a large number of ABC transporters simultaneously, resulting in the generation of multidrug-resistant, cancer stem-like cells (Figure 7). This model also explains why cancers that tend to initially respond to chemotherapy show a much aggressive and multidrug-resistant phenotype on relapse.
Figure 7.
A schematic representation of the effect of chemotherapy on invasive cancer cells. Chemotherapeutic treatment of a heterogeneous population of cancer cells kills majority of the epithelial-like cells, whereas the mesenchymal-like cells survive owing to a basal expression of ABC transporters. Drug-induced activation of self-renewal pathways (such as TGFβ) causes an increase in the expression of EMT transcription factors, mesenchymal morphology, migratory, and invasive potential of the surviving cells. These EMT factors can then upregulate the expression of a large number of ABC transporters simultaneously, resulting in the generation of cancer stem-like cells that are multidrug resistant
Role of ABC transporters in migration and invasion
Although on the one hand ABC transporters appear to help cancer cells in evading chemotherapy, these transporters may additionally have a direct role in cancer cell migration and invasion during tumor progression. In support of this, recent experiments have demonstrated that dendritic cells lacking specific ABC transporters fail to migrate to lymph nodes.34 In addition, an elegant study in Drosophila demonstrated that ABC transporters are essential for controlling the export of a germ cell attractant during directional cell migration during embryonic development.35 Hence, increased expression of ABC transporters by migratory cancer cells may additionally confer survival advantage against harmful chemicals encountered in the transiting microenvironments, and also regulate selective export of signaling molecules that may aid their directional migration. Thus, EMT-mediated upregulation of ABC transporters could be an evolutionarily conserved mechanism, which the cancer cells have exploited for their benefit.
Taken together, our study revealed that in addition to invasion, EMT inducers can promote drug resistance by upregulating ABC transporters, which efflux chemotherapeutic drugs. Our data provide novel insights into the molecular mechanism behind the long-term association between cancer invasiveness and drug resistance, and suggests that targeting regulators of EMT may simultaneously tackle the intertwined phenomena of metastasis, drug resistance, and CSCs, and prevent cancer relapse. Thus, EMT regulators like Twist, Snail, or FOXC2 transcription factors, self-renewal pathways like TGFβ, Wnt, and Notch that are known to induce EMT, or microRNAs like miR200, miR155 involved with EMT21 may prove to be opportune therapeutic targets.
Materials and Methods
Cell culture and reagents
Breast epithelial cell lines HBL100, T47D, MCF7, MDAMB453, MDAMB435, MDAMB231, and BT549 (from ATCC) were cultured in Dulbecco's modified Eagle's medium (DMEM) (Sigma-Aldrich, St. Louis, MO, USA) supplemented with 10% fetal bovine serum (FBS), penicillin (1 kU/ml), and streptomycin (0.1 mg/ml). HMLE was cultured as described previously15 in DMEM-F12 media containing 10 ng/ml hEGF, 0.5 μg/ml hydrocortisone, and 10 μg/ml insulin (Sigma-Aldrich). MCF10A cells were grown in DMEM-F12 supplemented with 10% FBS, epidermal growth factor (20 ng/ml), hydrocortisone (0.5 μg/ml), and insulin (10 μg/ml). The primary breast normal and cancer tissues were processed as described earlier.36 After digestion and trypsinization, single cells were seeded in attachment cultures for 1 week for drug treatment. Dox hydrochloride solution and mitoxantrone were obtained from Sigma-Aldrich.
Primary samples
Normal, DCIS, and IDC primary tissue (untreated and post-chemotherapy treated) samples were obtained from Kidwai Memorial Institute of Oncology (KMIO), Bangalore, as per the Institutional Review Board and in compliance with the ethical guidelines of KMIO and IISc.
Time-course drug assays
To determine the duration of drug treatment, a time-course experiment was undertaken in MDAMB435 and MDAMB231 cells. A total of 2 × 105 cells each were seeded in five 60 mm dishes. After 24 h of seeding, one plate was harvested as a 0 h minus Dox control. To all the other plates, Dox was added to a final concentration of 0.2 μM. The media were changed every third day and fresh drug was added. The plates were harvested in TRI reagent at respective time points of 24, 48, and 72 h and 1 week for subsequent RT-PCR-based analysis (Supplementary Figure S6).
MTT assay
MTT assay was performed in triplicates in 96-well microtiter plates (Greiner Bio-One, Frickenhausen, Germany). At 24 h after seeding, specific concentrations of Dox or mitoxantrone was added to independent wells and the plates were incubated for another 48 h in the incubator. MTT (5 mg/ml) reagent (Sigma-Aldrich) was added to each well and the plate was incubated for 4 h until the formation of formazan crystals. After dissolving the crystals in DMSO, the plate was read in ELISA reader at 540 nm. Cell viability was expressed as the percentage of the absorbance of drug-treated cells, relative to that of the untreated controls (Supplementary Table S2).
RNA extraction and RT-PCR
Total RNA was isolated using the guanidine isothiocyanate and phenol/chloroform method from TRI reagent (Sigma-Aldrich) harvested cells. RT of mRNA was carried out using Gene-Amp RNA PCR cDNA synthesis kit (Applied Biosystems, Carlsbad, CA, USA) according to the manufacturer's instructions. Specific primers used in the study were designed using the Primer 3.0 software and sequences are mentioned in Supplementary Table S3. Either β2-microglobulin, β-actin, or HPRT were used as normalizing controls as mentioned in the figures.
Wound healing assay
Equal numbers of live cells were seeded in two 60 mm dishes (Greiner Bio-One). Dox treatment was carried out for 72 h when cells were 90% confluent. Cells were treated with 10 μg/ml of mitomycin C (Calbiochem, Gibbstown, NJ, USA) for 2 h to arrest proliferation. Two wounds were made using a P-200 pipette tip. Microphotographs were taken at 0 and 24 h. The distance migrated was quantified using the ProgRes capture software.
Matrigel invasion assay
In vitro tumor cell invasion assay was performed using the BD BioCoat Matrigel Invasion Chambers as per the manufacturer's instructions. MDAMB231 cells were grown in the absence or presence of Dox for 72 h at which time they were trypsinized and 50 000 total live cells were used for the experiment. At 24 h after invasion, total cells were counted and plotted as invasion index or % cell invasion.
EMT induction
The MCF7 cells were transiently transfected with pBp-empty vector, pBp-Snail, pBp-mTwist, or pBp-FOXC2 expression constructs (using Lipofectamine 2000; Invitrogen, Carlsbad, CA, USA) and subsequently harvested at 48 h post-transfection for further experimentation.
HMLE cells overexpressing Twist (HMLE-Twist) were generated by retroviral transduction. Retroviruses were produced by transient transfection of 293T cells with retroviral vectors (pBp-Snail, pBp-mTwist, or pBp-FOXC2) and packaging plasmids (encoding pCMV-VSV-G and pUMVC3-gag-pol) using Fugene 6 (Roche Molecular Biochemicals, Mannheim, Germany). HMLE cells growing in a 60 mm dish were infected at 40% confluency with viral supernatants along with 8 μg/ml protamine sulfate and 10 mM HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) for 6 h, following which the viral particles were removed and fresh media were added. After 48 h of infection, drug selection with 0.5 μg/ml of puromycin was initiated. Clones of cells that emerged in the 60 mm dish following drug selection all showed similar morphological changes (mesenchymal appearance). These clones were pooled and the experiments were performed in the polyclonal population.
RNAi experiments
MDAMB231 cells were seeded in 60 mm dishes and at 40% confluency transfected with ON-TARGET plus SMART pool of non-targeting control siRNA, Twist siRNA, or Zeb1 siRNA (Dharmacon, Lafayette, CO, USA) using oligofectamine (Invitrogen) as per the manufacturer's instructions in Opti-MEM media. After 24 h of transfection, Dox was added for another 72 h following which cells were taken for either RT-PCR-based analysis or Matrigel invasion assay.
For Dox chemosensitivity experiments, MDAMB231 cells were seeded in 96-well microplates. After 24 h, cells were transfected with non-targeting control siRNA, Twist siRNA, or Zeb1 siRNA. After another 24 h, specific concentrations of Dox were added and the plates incubated for 48 h at which time MTT reagent was added and reading taken as described above.
Promoter analysis
The 2.2 kb (2000 bp upstream and 200 bp downstream of TSS) of promoter sequences were obtained using the DBTSS software and promoter analysis was carried out using the MatInspector software, which identifies all potential binding sites for transcription factors.
Luciferase reporter assay
A total of 2 × 105 MCF7 cells were plated in 12-well plates (Greiner Bio-One). Cells were co-transfected with 800 ng of pGL4.11b-ABCC5 promoter-luciferase plasmid37 and 800 ng of pBp-empty vector, pBp-Snail, pBp-mTwist, or pBp-FOXC2 in triplicates. To normalize transfection efficiency, cells were also co-transfected with 50 ng of the pRL-TK (Renilla luciferase). At 48 h after transfection, luciferase activity was measured using the Dual-Luciferase Assay kit (Promega, Madison, WI, USA) in a scintillation counter for 10 s. Firefly luciferase activity was expressed as relative light units compared with Renilla luciferase activity.
ChIP assay
ChIP assay was carried out essentially as per the standard protocols.38 In brief, MCF7 cells transfected with pCNDA3-Flag-Twist were harvested after 72 h, crosslinked with 1% formaldehyde for 10 min at room temperature, and then sonicated to obtain chromatin fragments ranging between 300 and 700 bp. After pre-clearing with blocked protein-A beads (Bangalore Genei, Bangalore, India), the soluble chromatin was equally divided and immunoprecipitated with either rabbit anti-Flag antibody (Cell Signaling Technology, Beverly, MA, USA) or a rabbit anti-IgG antibody (isotype control) (Bangalore Genei) by incubation at 4°C overnight. ‘No antibody' control was also incubated at 4°C. Following washes, the antibody–protein–DNA complex was eluted from the beads, followed by reverse crosslinking at 65°C with 200 mM NaCl. After RNase (Sigma-Aldrich) and proteinase K (Sigma-Aldrich) treatment, the DNA was purified by phenol–chloroform extraction. Equal amount of DNA was subjected to PCR with primers specific for E-box sites within the ABCC4 and ABCC5 promoters. The sequences of the PCR primers used are mentioned in Supplementary Table S4.
Statistical analysis
All statistical analysis was performed using the GraphPad Prism 5.0 software. All data are presented as mean±S.E.M. P-values <0.05 were considered to be statistically significant. Ratio t-test was performed for statistical analysis. All experiments were repeated thrice, unless otherwise stated.
Acknowledgments
We thank Dr. Robert Weinberg for pBp-Snail, pBp-mTwist, pBp-FOXC2, and breast cancer cell lines; Dr. Kathleen Giacomini and Dr. Deanna Kroetz for pGL4.11b-ABCC5 promoter-luciferase plasmid; and Dr. Kimitoshi Kohno for pCNDA3-Flag-Twist constructs. We also thank Dr. Geetashree Mukherjee and Umaira Tabassum for help with procuring primary breast tissue samples from Kidwai Memorial Institute of Oncology, Bangalore; Ms Divya M Pillai and Mr H Sravanth Kumar for help with manuscript revision and experiments; and Mr Shubham Pandey, Mr Sravan GS, and Mr A Sai Balaji for technical assistance. We acknowledge funding from IISc and Dept. of Biotechnology, Govt. of India, to AR; support from Dept. of Science and Technology, Govt. of India to the Department of MRDG; MS is a CSIR Senior Research Fellow; AR is a Wellcome Trust/ DBT India Alliance Senior Research Fellow.
Glossary
- ABC transporters
ATP binding cassette transporters
- ChIP assay
chromatin immunoprecipitation assay
- DCIS
ductal carcinoma in situ
- Dox
doxorubicin
- EMT
epithelial–mesenchymal transition
- IDC
invasive ductal carcinoma
- MDR
multidrug resistance
- MMP
matrix metalloproteinase
- MTT
3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- RT-PCR
reverse transcriptase polymerase chain reaction
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on Cell Death and Disease website (http://www.nature.com/cddis)
Edited by P Salomoni
Supplementary Material
References
- Szakacs G, Paterson JK, Ludwig JA, Booth-Genthe C, Gottesman MM. Targeting multidrug resistance in cancer. Nat Rev Drug Discov. 2006;5:219–234. doi: 10.1038/nrd1984. [DOI] [PubMed] [Google Scholar]
- Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer: role of ATP-dependent transporters. Nat Rev Cancer. 2002;2:48–58. doi: 10.1038/nrc706. [DOI] [PubMed] [Google Scholar]
- Simon SM, Schindler M. Cell biological mechanisms of multidrug resistance in tumors. Proc Natl Acad Sci USA. 1994;91:3497–3504. doi: 10.1073/pnas.91.9.3497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean M, Hamon Y, Chimini G. The human ATP-binding cassette (ABC) transporter superfamily. J Lipid Res. 2001;42:1007–1017. [PubMed] [Google Scholar]
- Fletcher JI, Haber M, Henderson MJ, Norris MD. ABC transporters in cancer: more than just drug efflux pumps. Nat Rev Cancer. 2010;10:147–156. doi: 10.1038/nrc2789. [DOI] [PubMed] [Google Scholar]
- Walsh N, Kennedy S, Larkin AM, Tryfonopoulos D, Eustace AJ, Mahgoub T, et al. Membrane transport proteins in human melanoma: associations with tumour aggressiveness and metastasis. Br J Cancer. 2010;102:1157–1162. doi: 10.1038/sj.bjc.6605590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank NY, Margaryan A, Huang Y, Schatton T, Waaga-Gasser AM, Gasser M, et al. ABCB5-mediated doxorubicin transport and chemoresistance in human malignant melanoma. Cancer Res. 2005;65:4320–4333. doi: 10.1158/0008-5472.CAN-04-3327. [DOI] [PubMed] [Google Scholar]
- Kalluri R, Weinberg RA. The basics of epithelial–mesenchymal transition. J Clin Invest. 2009;119:1420–1428. doi: 10.1172/JCI39104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polyak K, Weinberg RA. Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer. 2009;9:265–273. doi: 10.1038/nrc2620. [DOI] [PubMed] [Google Scholar]
- Gavert N, Ben-Ze′ev A. Epithelial–mesenchymal transition and the invasive potential of tumors. Trends Mol Med. 2008;14:199–209. doi: 10.1016/j.molmed.2008.03.004. [DOI] [PubMed] [Google Scholar]
- Hoshino H, Miyoshi N, Nagai K, Tomimaru Y, Nagano H, Sekimoto M, et al. Epithelial–mesenchymal transition with expression of SNAI1-induced chemoresistance in colorectal cancer. Biochem Biophys Res Commun. 2009;390:1061–1065. doi: 10.1016/j.bbrc.2009.10.117. [DOI] [PubMed] [Google Scholar]
- Zhuo W, Wang Y, Zhuo X, Zhang Y, Ao X, Chen Z. Knockdown of Snail, a novel zinc finger transcription factor, via RNA interference increases A549 cell sensitivity to cisplatin via JNK/mitochondrial pathway. Lung Cancer. 2008;62:8–14. doi: 10.1016/j.lungcan.2008.02.007. [DOI] [PubMed] [Google Scholar]
- Kurrey NK, Jalgaonkar SP, Joglekar AV, Ghanate AD, Chaskar PD, Doiphode RY, et al. Snail and slug mediate radioresistance and chemoresistance by antagonizing p53-mediated apoptosis and acquiring a stem-like phenotype in ovarian cancer cells. Stem Cells. 2009;27:2059–2068. doi: 10.1002/stem.154. [DOI] [PubMed] [Google Scholar]
- Hollier BG, Evans K, Mani SA. The epithelial-to-mesenchymal transition and cancer stem cells: a coalition against cancer therapies. J Mammary Gland Biol Neoplasia. 2009;14:29–43. doi: 10.1007/s10911-009-9110-3. [DOI] [PubMed] [Google Scholar]
- Elenbaas B, Spirio L, Koerner F, Fleming MD, Zimonjic DB, Donaher JL, et al. Human breast cancer cells generated by oncogenic transformation of primary mammary epithelial cells. Genes Dev. 2001;15:50–65. doi: 10.1101/gad.828901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu CT, Chang TY, Cheng CC, Liu CS, Wu JR, Li MC, et al. Snail associates with EGR-1 and SP-1 to upregulate transcriptional activation of p15. FEBS J. 2010;277:1202–1218. doi: 10.1111/j.1742-4658.2009.07553.x. [DOI] [PubMed] [Google Scholar]
- Cheng GZ, Chan J, Wang Q, Zhang W, Sun CD, Wang LH. Twist transcriptionally up-regulates AKT2 in breast cancer cells leading to increased migration, invasion, and resistance to paclitaxel. Cancer Res. 2007;67:1979–1987. doi: 10.1158/0008-5472.CAN-06-1479. [DOI] [PubMed] [Google Scholar]
- Fujita H, Kang M, Eren M, Gleaves LA, Vaughan DE, Kume T. Foxc2 is a common mediator of insulin and transforming growth factor beta signaling to regulate plasminogen activator inhibitor type I gene expression. Circ Res. 2006;98:626–634. doi: 10.1161/01.RES.0000207407.51752.3c. [DOI] [PubMed] [Google Scholar]
- Reece-Hoyes JS, Deplancke B, Barrasa MI, Hatzold J, Smit RB, Arda HE, et al. The C. elegans Snail homolog CES-1 can activate gene expression in vivo and share targets with bHLH transcription factors. Nucleic Acids Res. 2009;37:3689–3698. doi: 10.1093/nar/gkp232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ponte-Sucre A. Availability and applications of ATP-binding cassette (ABC) transporter blockers. Appl Microbiol Biotechnol. 2007;76:279–286. doi: 10.1007/s00253-007-1017-6. [DOI] [PubMed] [Google Scholar]
- Singh A, Settleman J. EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene. 2010;29:4741–4751. doi: 10.1038/onc.2010.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kajiyama H, Shibata K, Terauchi M, Yamashita M, Ino K, Nawa A, et al. Chemoresistance to paclitaxel induces epithelial–mesenchymal transition and enhances metastatic potential for epithelial ovarian carcinoma cells. Int J Oncol. 2007;31:277–283. [PubMed] [Google Scholar]
- Arumugam T, Ramachandran V, Fournier KF, Wang H, Marquis L, Abbruzzese JL, et al. Epithelial to mesenchymal transition contributes to drug resistance in pancreatic cancer. Cancer Res. 2009;69:5820–5828. doi: 10.1158/0008-5472.CAN-08-2819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah AN, Summy JM, Zhang J, Park SI, Parikh NU, Gallick GE. Development and characterization of gemcitabine-resistant pancreatic tumor cells. Ann Surg Oncol. 2007;14:3629–3637. doi: 10.1245/s10434-007-9583-5. [DOI] [PubMed] [Google Scholar]
- Yang AD, Fan F, Camp ER, van Buren G, Liu W, Somcio R, et al. Chronic oxaliplatin resistance induces epithelial-to-mesenchymal transition in colorectal cancer cell lines. Clin Cancer Res. 2006;12 (Part 1:4147–4153. doi: 10.1158/1078-0432.CCR-06-0038. [DOI] [PubMed] [Google Scholar]
- Li QQ, Xu JD, Wang WJ, Cao XX, Chen Q, Tang F, et al. Twist1-mediated adriamycin-induced epithelial–mesenchymal transition relates to multidrug resistance and invasive potential in breast cancer cells. Clin Cancer Res. 2009;15:2657–2665. doi: 10.1158/1078-0432.CCR-08-2372. [DOI] [PubMed] [Google Scholar]
- Bandyopadhyay A, Wang L, Agyin J, Tang Y, Lin S, Yeh IT, et al. Doxorubicin in combination with a small TGFbeta inhibitor: a potential novel therapy for metastatic breast cancer in mouse models. PLoS One. 2010;5:e10365. doi: 10.1371/journal.pone.0010365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean M. ABC transporters, drug resistance, and cancer stem cells. J Mammary Gland Biol Neoplasia. 2009;14:3–9. doi: 10.1007/s10911-009-9109-9. [DOI] [PubMed] [Google Scholar]
- Dean M, Fojo T, Bates S. Tumour stem cells and drug resistance. Nat Rev Cancer. 2005;5:275–284. doi: 10.1038/nrc1590. [DOI] [PubMed] [Google Scholar]
- Wang Z, Li Y, Kong D, Banerjee S, Ahmad A, Azmi AS, et al. Acquisition of epithelial–mesenchymal transition phenotype of gemcitabine-resistant pancreatic cancer cells is linked with activation of the notch signaling pathway. Cancer Res. 2009;69:2400–2407. doi: 10.1158/0008-5472.CAN-08-4312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chikazawa N, Tanaka H, Tasaka T, Nakamura M, Tanaka M, Onishi H, et al. Inhibition of Wnt signaling pathway decreases chemotherapy-resistant side-population colon cancer cells. Anticancer Res. 2010;30:2041–2048. [PubMed] [Google Scholar]
- Gupta PB, Onder TT, Jiang G, Tao K, Kuperwasser C, Weinberg RA, et al. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell. 2009;138:645–659. doi: 10.1016/j.cell.2009.06.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, et al. The epithelial–mesenchymal transition generates cells with properties of stem cells. Cell. 2008;133:704–715. doi: 10.1016/j.cell.2008.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Randolph GJ, Beaulieu S, Pope M, Sugawara I, Hoffman L, Steinman RM, et al. A physiologic function for p-glycoprotein (MDR-1) during the migration of dendritic cells from skin via afferent lymphatic vessels. Proc Natl Acad Sci USA. 1998;95:6924–6929. doi: 10.1073/pnas.95.12.6924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ricardo S, Lehmann R. An ABC transporter controls export of a Drosophila germ cell attractant. Science. 2009;323:943–946. doi: 10.1126/science.1166239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dey D, Saxena M, Paranjape AN, Krishnan V, Giraddi R, Kumar MV, et al. Phenotypic and functional characterization of human mammary stem/progenitor cells in long term culture. PLoS One. 2009;4:e5329. doi: 10.1371/journal.pone.0005329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hesselson SE, Matsson P, Shima JE, Fukushima H, Yee SW, Kobayashi Y, et al. Genetic variation in the proximal promoter of ABC and SLC superfamilies: liver and kidney specific expression and promoter activity predict variation. PLoS One. 2009;4:e6942. doi: 10.1371/journal.pone.0006942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyd KE, Farnham PJ. Coexamination of site-specific transcription factor binding and promoter activity in living cells. Mol Cell Biol. 1999;19:8393–8399. doi: 10.1128/mcb.19.12.8393. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.