Abstract
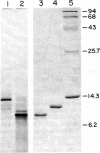
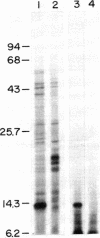
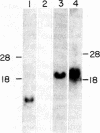
Major changes in the mRNA population of murine liver occur after administration of bacterial lipopolysaccharide, an agent that causes increases in the concentrations of acute-phase serum proteins. The mRNA for one of these, serum amyloid A, is increased at least 500-fold compared to the normal level. It becomes one of the most abundant hepatic mRNAs, and serum amyloid A synthesis comprises about 2.5% of total hepatic protein synthesis in the acute-phase response. Its synthesis is tissue-specific in that amyloid A mRNA was not detected in the kidney, an important site of amyloid fibril accumulation. The protein synthesized in largest amount by acute-phase liver tissue in culture is cytoplasmic actin. Its relative rate of synthesis is increased about 5-fold compared to the normal tissue; that of serum albumin is decreased to about one-third of its normal rate. The concentration of mRNA for serum albumin is decreased by a similar amount. Starting with induced liver RNA, we have constructed a recombinant plasmid containing most of the DNA sequence encoding the serum amyloid A polypeptide.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anders R. F., Natvig J. B., Sletten K., Husby G., Nordstoga K. Amyloid-related serum protein SAA from three animal species: comparison with human SAA. J Immunol. 1977 Jan;118(1):229–234. [PubMed] [Google Scholar]
- Anderson L., Anderson N. G. High resolution two-dimensional electrophoresis of human plasma proteins. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5421–5425. doi: 10.1073/pnas.74.12.5421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benditt E. P., Eriksen N., Hanson R. H. Amyloid protein SAA is an apoprotein of mouse plasma high density lipoprotein. Proc Natl Acad Sci U S A. 1979 Aug;76(8):4092–4096. doi: 10.1073/pnas.76.8.4092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benson M. D., Aldo-Benson M. Effect of purified protein SAA on immune response in vitro: mechanisms of suppression. J Immunol. 1979 May;122(5):2077–2082. [PubMed] [Google Scholar]
- Benson M. D., Kleiner E. Synthesis and secretion of serum amyloid protein A (SAA) by hepatocytes in mice treated with casein. J Immunol. 1980 Feb;124(2):495–499. [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Garrels J. I., Gibson W. Identification and characterization of multiple forms of actin. Cell. 1976 Dec;9(4 Pt 2):793–805. doi: 10.1016/0092-8674(76)90142-2. [DOI] [PubMed] [Google Scholar]
- Glenner G. G. Amyloid deposits and amyloidosis. The beta-fibrilloses (first of two parts). N Engl J Med. 1980 Jun 5;302(23):1283–1292. doi: 10.1056/NEJM198006053022305. [DOI] [PubMed] [Google Scholar]
- Goldberg D. A. Isolation and partial characterization of the Drosophila alcohol dehydrogenase gene. Proc Natl Acad Sci U S A. 1980 Oct;77(10):5794–5798. doi: 10.1073/pnas.77.10.5794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorevic P. D., Levo Y., Frangione B., Franklin E. C. Polymorphism of tissue and serum amyloid A (AA and SAA) proteins in the mouse. J Immunol. 1978 Jul;121(1):138–140. [PubMed] [Google Scholar]
- Grunstein M., Hogness D. S. Colony hybridization: a method for the isolation of cloned DNAs that contain a specific gene. Proc Natl Acad Sci U S A. 1975 Oct;72(10):3961–3965. doi: 10.1073/pnas.72.10.3961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hastie N. D., Held W. A., Toole J. J. Multiple genes coding for the androgen-regulated major urinary proteins of the mouse. Cell. 1979 Jun;17(2):449–457. doi: 10.1016/0092-8674(79)90171-5. [DOI] [PubMed] [Google Scholar]
- Kaplan M. H., Volanakis J. E. Interaction of C-reactive protein complexes with the complement system. I. Consumption of human complement associated with the reaction of C-reactive protein with pneumococcal C-polysaccharide and with the choline phosphatides, lecithin and sphingomyelin. J Immunol. 1974 Jun;112(6):2135–2147. [PubMed] [Google Scholar]
- Kioussis D., Hamilton R., Hanson R. W., Tilghman S. M., Taylor J. M. Construction and cloning of rat albumin structural gene sequences. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4370–4374. doi: 10.1073/pnas.76.9.4370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurtz D. T., Chan K. M., Feigelson P. Glucocorticoid induction of hepatic alpha2u-globulin synthesis and messenger RNA level in castrated male rats in vivo. J Biol Chem. 1978 Nov 10;253(21):7886–7890. [PubMed] [Google Scholar]
- Kushner I., Feldmann G. Control of the acute phase response. Demonstration of C-reactive protein synthesis and secretion by hepatocytes during acute inflammation in the rabbit. J Exp Med. 1978 Aug 1;148(2):466–477. doi: 10.1084/jem.148.2.466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laskey R. A., Mills A. D. Quantitative film detection of 3H and 14C in polyacrylamide gels by fluorography. Eur J Biochem. 1975 Aug 15;56(2):335–341. doi: 10.1111/j.1432-1033.1975.tb02238.x. [DOI] [PubMed] [Google Scholar]
- Linder E., Lehto V. P., Virtanen I., Stenman S., Natvig J. B. Localization of amyloid-related serum protein SAA-like material to intermediate (10 nm) filaments of cultured human embryonal fibroblasts. J Exp Med. 1977 Oct 1;146(4):1158–1163. doi: 10.1084/jem.146.4.1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lux S. E., John K. M., Ronan R., Brewer H. B., Jr Isolation and characterization of the tryptic and cyanogen bromide peptides of apoLp-Gln-II (apoA-II), plasma high density apolipoprotein. J Biol Chem. 1972 Dec 10;247(23):7519–7527. [PubMed] [Google Scholar]
- McAdam K. P., Sipe J. D. Murine model for human secondary amyloidosis: genetic variability of the acute-phase serum protein SAA response to endotoxins and casein. J Exp Med. 1976 Oct 1;144(4):1121–1127. doi: 10.1084/jem.144.4.1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan E. H. Factors affecting the synthesis of transferrin by rat tissue slices. J Biol Chem. 1969 Aug 10;244(15):4193–4199. [PubMed] [Google Scholar]
- Nelson T., Brutlag D. Addition of homopolymers to the 3'-ends of duplex DNA with terminal transferase. Methods Enzymol. 1979;68:41–50. doi: 10.1016/0076-6879(79)68005-9. [DOI] [PubMed] [Google Scholar]
- Oberg B., Saborio J., Persson T., Everitt E., Philipson L. Identification of the in vitro translation products of adenovirus mRNA by immunoprecipitation. J Virol. 1975 Jan;15(1):199–207. doi: 10.1128/jvi.15.1.199-207.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmiter R. D., Oka T., Schimke R. T. Modulation of ovalbumin synthesis by estradiol-17 beta and actinomycin D as studied in explants of chick oviduct in culture. J Biol Chem. 1971 Feb 10;246(3):724–737. [PubMed] [Google Scholar]
- Pollard T. D., Thomas S. M., Niederman R. Human platelet myosin. I. Purification by a rapid method applicable to other nonmuscle cells. Anal Biochem. 1974 Jul;60(1):258–266. doi: 10.1016/0003-2697(74)90152-3. [DOI] [PubMed] [Google Scholar]
- Prentki M., Chaponnier C., Jeanrenaud B., Gabbiani G. Actin microfilaments, cell shape, and secretory processes in isolated rat hepatocytes. Effect of phalloidin and cytochalasin D. J Cell Biol. 1979 Jun;81(3):592–607. doi: 10.1083/jcb.81.3.592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ricciardi R. P., Miller J. S., Roberts B. E. Purification and mapping of specific mRNAs by hybridization-selection and cell-free translation. Proc Natl Acad Sci U S A. 1979 Oct;76(10):4927–4931. doi: 10.1073/pnas.76.10.4927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts B. E., Paterson B. M. Efficient translation of tobacco mosaic virus RNA and rabbit globin 9S RNA in a cell-free system from commercial wheat germ. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2330–2334. doi: 10.1073/pnas.70.8.2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenthal C. J., Sullivan L. Serum amyloid A: evidence for its origin in polymorphonuclear leukocytes. J Clin Invest. 1978 Dec;62(6):1181–1186. doi: 10.1172/JCI109237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selinger M. J., McAdam K. P., Kaplan M. M., Sipe J. D., Vogel S. N., Rosenstreich D. L. Monokine-induced synthesis of serum amyloid A protein by hepatocytes. Nature. 1980 Jun 12;285(5765):498–500. doi: 10.1038/285498a0. [DOI] [PubMed] [Google Scholar]
- Shapiro S. G., Cousins R. J. Induction of rat liver metallothionein mRNA and its distribution between free and membrane-bound polyribosomes. Biochem J. 1980 Sep 15;190(3):755–764. doi: 10.1042/bj1900755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sipe J. D. Induction of the acute-phase serum protein SAA requires both RNA and protein synthesis. Br J Exp Pathol. 1978 Jun;59(3):305–310. [PMC free article] [PubMed] [Google Scholar]
- Storti R. V., Scott M. P., Rich A., Pardue M. L. Translational control of protein synthesis in response to heat shock in D. melanogaster cells. Cell. 1980 Dec;22(3):825–834. doi: 10.1016/0092-8674(80)90559-0. [DOI] [PubMed] [Google Scholar]
- Vandekerckhove J., Weber K. Actin amino-acid sequences. Comparison of actins from calf thymus, bovine brain, and SV40-transformed mouse 3T3 cells with rabbit skeletal muscle actin. Eur J Biochem. 1978 Oct 16;90(3):451–462. doi: 10.1111/j.1432-1033.1978.tb12624.x. [DOI] [PubMed] [Google Scholar]
- Wickens M. P., Buell G. N., Schimke R. T. Synthesis of double-stranded DNA complementary to lysozyme, ovomucoid, and ovalbumin mRNAs. Optimization for full length second strand synthesis by Escherichia coli DNA polymerase I. J Biol Chem. 1978 Apr 10;253(7):2483–2495. [PubMed] [Google Scholar]
- Wicks W. D. Induction of hepatic enzymes by adenosine 3',5'-monophosphate in organ culture. J Biol Chem. 1969 Jul 25;244(14):3941–3950. [PubMed] [Google Scholar]