Abstract
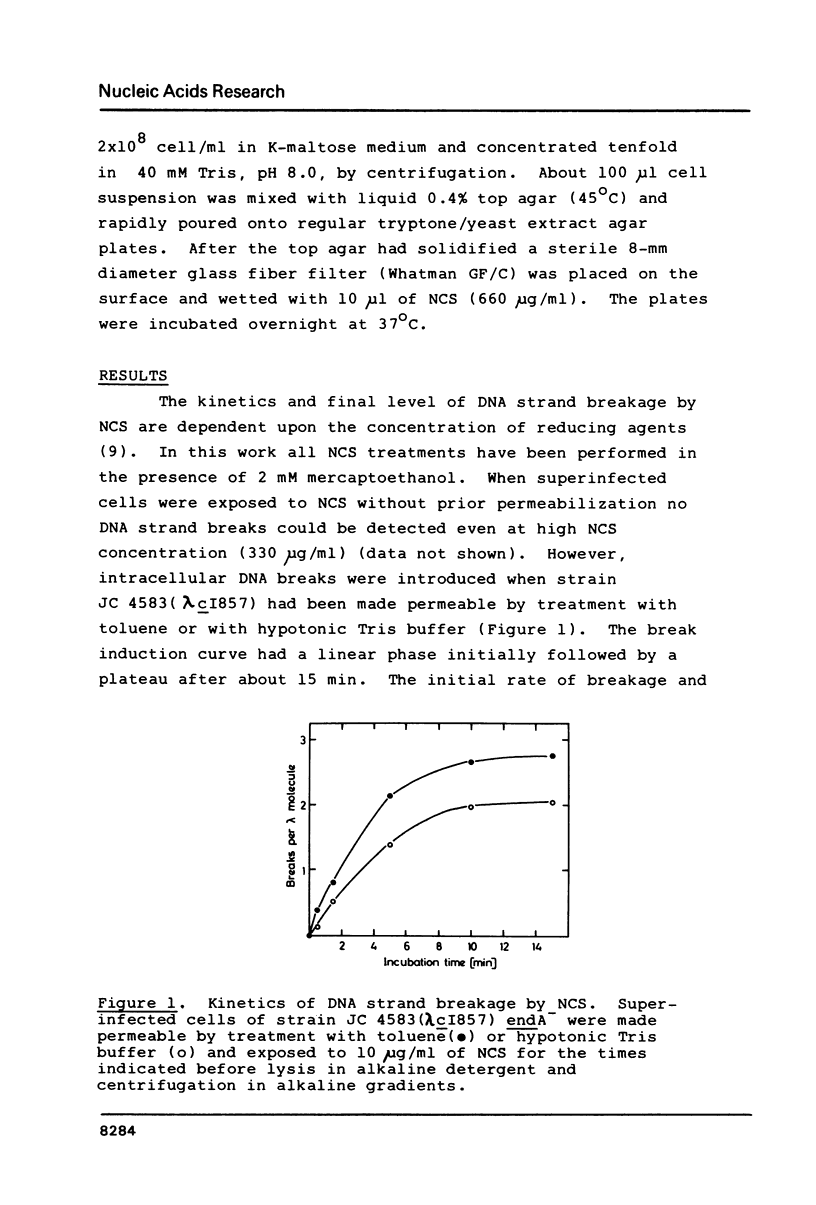
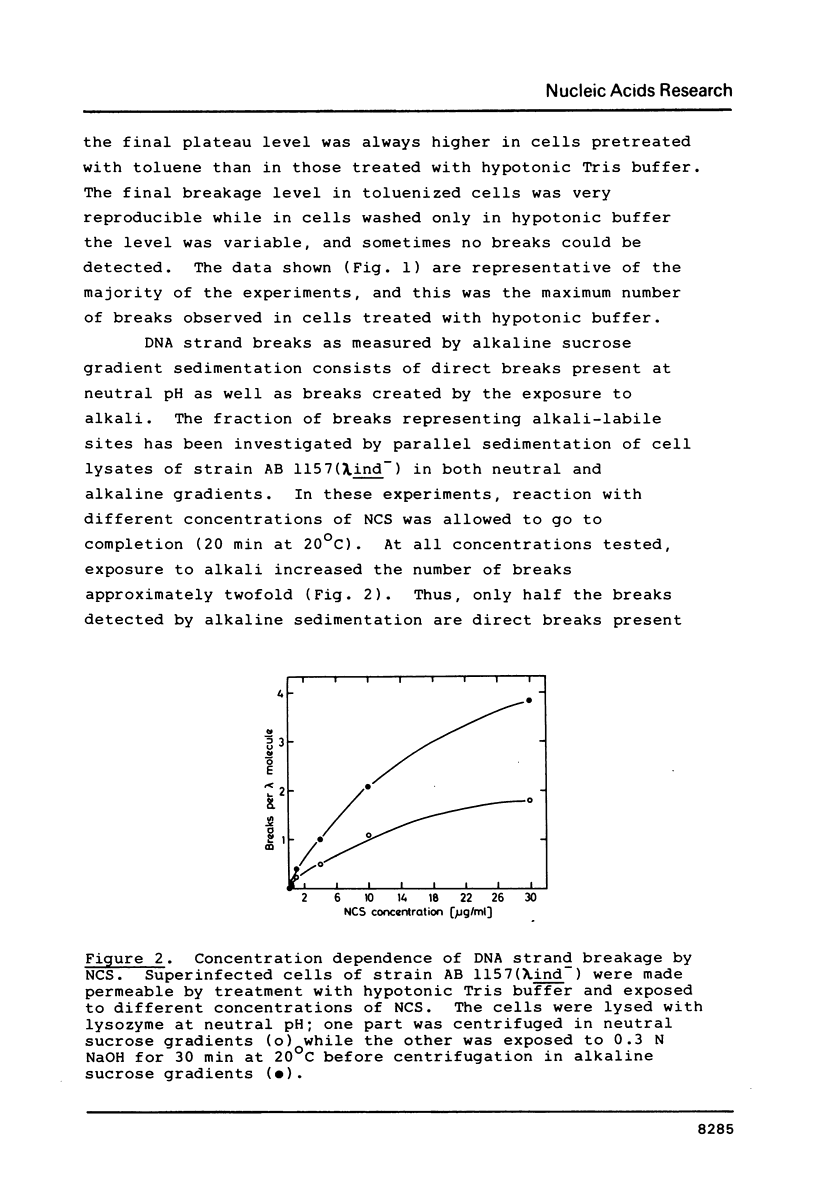
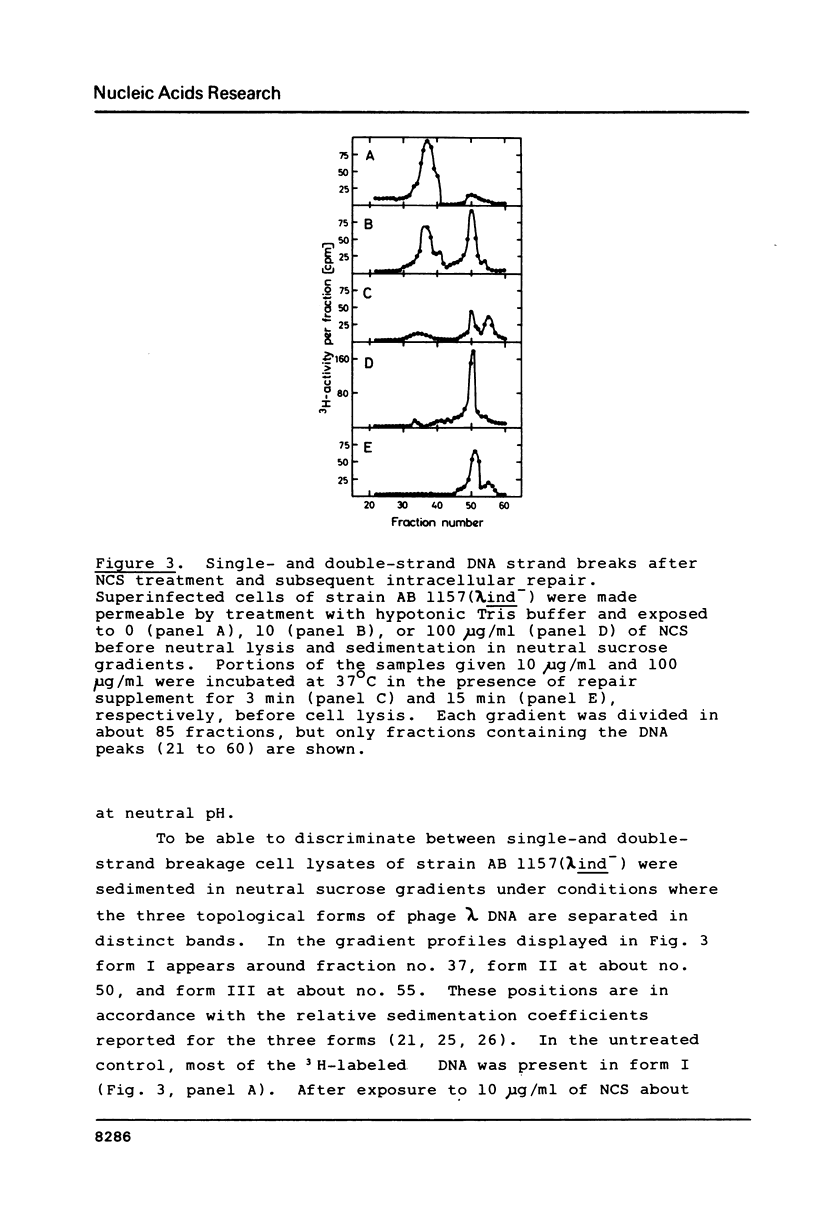
DNA strand breaks induced by Neocarzinostatin in Escherichia coli cells have been characterized. Radioactively labeled phage lambda DNA was introduced into lysogenic host bacteria allowing the phage DNA to circularize into superhelical molecules. After drug treatment DNA single- and double-strand breaks were measured independently after neutral sucrose gradient sedimentation. The presence of alkali-labile lesions was measured in parallel in alkaline sucrose gradients. The cell envelope provided an efficient protection towards the drug, since no strand breaks were detected unless the cells were made permeable with toluene or with hypotonic Tris buffer. In permeable cells, no double strand breaks could be detected, even at high NCS concentration (100 micrograms/ml). Induction of single-strand breaks leveled off after 15 min at 20 degrees C in the presence of 2 mM mercaptoethanol. Exposure to 0.3N NaOH doubled the number of strand breaks. No enzymatic repair of the breaks could be observed.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bachmann B. J. Pedigrees of some mutant strains of Escherichia coli K-12. Bacteriol Rev. 1972 Dec;36(4):525–557. doi: 10.1128/br.36.4.525-557.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beerman T. A., Goldberg I. H. DNA strand scission by the antitumor protein neocarzinostatin. Biochem Biophys Res Commun. 1974 Aug 19;59(4):1254–1261. doi: 10.1016/0006-291x(74)90449-5. [DOI] [PubMed] [Google Scholar]
- Beerman T. A., Poon R., Goldberg I. H. Single-strand nicking of DNA in vitro by neocarzinostatin and its possible relationship to the mechanism of drug action. Biochim Biophys Acta. 1977 Mar 18;475(2):294–306. doi: 10.1016/0005-2787(77)90020-x. [DOI] [PubMed] [Google Scholar]
- Bose K. K., Tatsumi K., Strauss B. S. Apurinic/apyrimidinic endonuclease sensitive sites as intermediates in the in vitro degradation of deoxyribonucleic acid by neocarzinostatin. Biochemistry. 1980 Oct 14;19(21):4761–4766. doi: 10.1021/bi00562a007. [DOI] [PubMed] [Google Scholar]
- Boye E. DNA repair and replication in cells of Escherichia coli made permeable with hypotonic buffers. Radiat Res. 1980 Oct;84(1):35–45. [PubMed] [Google Scholar]
- Boye E. Formation and repair of DNA double-strand breaks in superinfecting phage lambda after ionizing irradiation of Escherichia coli host cells. Radiat Res. 1980 Mar;81(3):427–440. [PubMed] [Google Scholar]
- Boye E., Johansen I., Brustad T. The yield of radiation-induced DNA single-strand breaks in Escherichia coli and superinfecting phage lambda at different dose rates. Repair of strand breaks in different buffers. Radiat Res. 1976 Dec;68(3):499–508. [PubMed] [Google Scholar]
- Boye E., Krisch R. E. Induction and repair of double- and single-strand DNA breaks in bacteriophage lambda superinfecting Escherichia coli. Int J Radiat Biol Relat Stud Phys Chem Med. 1980 Feb;37(2):119–133. doi: 10.1080/09553008014550191. [DOI] [PubMed] [Google Scholar]
- D'Andrea A. D., Haseltine W. A. Sequence specific cleavage of DNA by the antitumor antibiotics neocarzinostatin and bleomycin. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3608–3612. doi: 10.1073/pnas.75.8.3608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Lucia P., Cairns J. Isolation of an E. coli strain with a mutation affecting DNA polymerase. Nature. 1969 Dec 20;224(5225):1164–1166. doi: 10.1038/2241164a0. [DOI] [PubMed] [Google Scholar]
- Falkson G., Von Hoff D., Klaassen D., Du Plessis H., Van Der Merwe C. F., Van Der Merwe A. M., Carbone P. P. A phase II study of neocarzinostatin (NSC 157365) in malignant hepatoma. An Eastern Cooperative Oncology Group pilot study. Cancer Chemother Pharmacol. 1980;4(1):33–36. doi: 10.1007/BF00255455. [DOI] [PubMed] [Google Scholar]
- Hatayama T., Goldberg I. H. DNA damage and repair in relation to cell killing in neocarzinostatin-treated HeLa cells. Biochim Biophys Acta. 1979 Jun 20;563(1):59–71. doi: 10.1016/0005-2787(79)90007-8. [DOI] [PubMed] [Google Scholar]
- Hatayama T., Goldberg I. H. Deoxyribonucleic acid sugar damage in the action of neocarzinostatin. Biochemistry. 1980 Dec 9;19(25):5890–5898. doi: 10.1021/bi00566a035. [DOI] [PubMed] [Google Scholar]
- Hensens O. D., Dewey R. S., Liesch J. M., Napier M. A., Reamer R. A., Smith J. L., Albers-Schönberg G., Goldberg I. H. Neocarzinostatin chromophore: presence of a highly strained ether ring and its reaction with mercaptan and sodium borohydride. Biochem Biophys Res Commun. 1983 Jun 15;113(2):538–547. doi: 10.1016/0006-291x(83)91759-x. [DOI] [PubMed] [Google Scholar]
- Howard-Flanders P., Boyce R. P., Theriot L. Three loci in Escherichia coli K-12 that control the excision of pyrimidine dimers and certain other mutagen products from DNA. Genetics. 1966 Jun;53(6):1119–1136. doi: 10.1093/genetics/53.6.1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ISHIDA N., MIYAZAKI K., KUMAGAI K., RIKIMARU M. NEOCARZINOSTATIN, AN ANTITUMOR ANTIBIOTIC OF HIGH MOLECULAR WEIGHT. ISOLATION, PHYSIOCHEMICAL PROPERTIES AND BIOLOGICAL ACTIVITIES. J Antibiot (Tokyo) 1965 Mar;18:68–76. [PubMed] [Google Scholar]
- Ishida R., Takahashi T. In vitro release of thymine from DNA by neocarzinostatin. Biochem Biophys Res Commun. 1976 Jan 12;68(1):256–261. doi: 10.1016/0006-291x(76)90037-1. [DOI] [PubMed] [Google Scholar]
- Kiger J. A., Jr, Young E. T., 2nd, Sinsheimer R. L. Purification and properties of intracellular lamba DNA rings. J Mol Biol. 1968 Apr 28;33(2):395–413. doi: 10.1016/0022-2836(68)90197-6. [DOI] [PubMed] [Google Scholar]
- Kikuchi M., Shoji M., Ishida N. Pre-neocarzinostatin, a specific antagonist of neocarzinostatin. J Antibiot (Tokyo) 1974 Oct;27(10):766–774. doi: 10.7164/antibiotics.27.766. [DOI] [PubMed] [Google Scholar]
- Kuo W. L., Meyn R. E., Haidle C. W. Neocarzinostatin-mediated DNA damage and repair in wild-type and repair-deficient Chinese hamster ovary cells. Cancer Res. 1984 May;44(5):1748–1751. [PubMed] [Google Scholar]
- Legha S. S., Von Hoff D. D., Rozencweig M., Abraham D., Slavik M., Muggia M. Neocarzinostatin (NSC 157365) a new cancerostatic compound. Oncology. 1976;33(5-6):265–270. doi: 10.1159/000225161. [DOI] [PubMed] [Google Scholar]
- Lewis R. S., Jung G., Köhnlein W. A model for the activation and inactivation of neocarzinostatin, an antitumor protein. Biochim Biophys Acta. 1980 Jun 27;608(1):138–146. doi: 10.1016/0005-2787(80)90141-0. [DOI] [PubMed] [Google Scholar]
- Ljungquist S., Lindahl T., Howard-Flanders P. Methyl methane sulfonate-sensitive mutant of Escherichia coli deficient in an endonuclease specific for apurinic sites in deoxyribonucleic acid. J Bacteriol. 1976 May;126(2):646–653. doi: 10.1128/jb.126.2.646-653.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meienhofer J., Maeda H., Glaser C. B., Czombos J., Kuromizu K. Primary structure of neocarzinostatin, an antitumor protein. Science. 1972 Nov 24;178(4063):875–876. doi: 10.1126/science.178.4063.875. [DOI] [PubMed] [Google Scholar]
- Mousstacchi E., Favaudon V. Cytotoxic and mutagenic effects of neocarzinostatin in wild-type and repair-deficient yeasts. Mutat Res. 1982 Apr;104(1-3):87–94. doi: 10.1016/0165-7992(82)90125-7. [DOI] [PubMed] [Google Scholar]
- Ogawa H., Tomizawa J. Bacteriophage lambda DNA with different structures found in infected cells. J Mol Biol. 1967 Jan 28;23(2):265–276. doi: 10.1016/s0022-2836(67)80032-9. [DOI] [PubMed] [Google Scholar]
- Ohtsuki K., Ishida N. Mechanism of DNA degradation induced by neocarzinostatin in Bacillus subtilis. J Antibiot (Tokyo) 1975 Mar;28(3):229–236. doi: 10.7164/antibiotics.28.229. [DOI] [PubMed] [Google Scholar]
- Ono Y., Watanabe Y., Ishida N. Mode of action of neocarzinostatin: inhibition of DNA synthesis and degradation of DNA in Sarcina lutea. Biochim Biophys Acta. 1966 Apr 18;119(1):46–58. doi: 10.1016/0005-2787(66)90036-0. [DOI] [PubMed] [Google Scholar]
- Poon R., Beerman T. A., Goldberg I. H. Characterization of DNA strand breakage in vitro by the antitumor protein neocarzinostatin. Biochemistry. 1977 Feb 8;16(3):486–493. doi: 10.1021/bi00622a023. [DOI] [PubMed] [Google Scholar]
- Povirk L. F., Dattagupta N., Warf B. C., Goldberg I. H. Neocarzinostatin chromophore binds to deoxyribonucleic acid by intercalation. Biochemistry. 1981 Jul 7;20(14):4007–4014. doi: 10.1021/bi00517a009. [DOI] [PubMed] [Google Scholar]
- Shiloh Y., van der Schans G. P., Lohman P. H., Becker Y. Induction and repair of DNA damage in normal and ataxia-telangiectasia skin fibroblasts treated with neocarzinostatin. Carcinogenesis. 1983;4(7):917–921. doi: 10.1093/carcin/4.7.917. [DOI] [PubMed] [Google Scholar]
- Tatsumi K., Bose K. K., Ayres K., Strauss B. S. Repair of neocarzinostatin-induced deoxyribonucleic acid damage in human lymphoblastoid cells: possible involvement of apurinic/apyrimidinic sites as intermediates. Biochemistry. 1980 Oct 14;19(21):4767–4772. doi: 10.1021/bi00562a008. [DOI] [PubMed] [Google Scholar]
- Tatsumi K., Nakamura T., Wakisaka G. Damage of mammalian cell DNA in vivo and in vitro induced by neocarzinostatin. Gan. 1974 Oct;65(5):459–461. [PubMed] [Google Scholar]
- Tatsumi K., Nishioka H. Effect of DNA Repair systems on antibacterial and mutagenic activity of an antitumor protein, neocarzinostatin. Mutat Res. 1977 Apr;48(2):195–203. doi: 10.1016/0027-5107(77)90161-0. [DOI] [PubMed] [Google Scholar]
- Tilby M. J., Loverock P. S., Fielden E. M. An effect of elevated postirradiation pH on the yield of double-strand breaks in DNA from irradiated bacterial cells. Radiat Res. 1984 May;98(2):284–292. [PubMed] [Google Scholar]
- White B. J., Hochhauser S. J., Cintron N. M., Weiss B. Genetic mapping of xthA, the structural gene for exonuclease III in Escherichia coli K-12. J Bacteriol. 1976 Jun;126(3):1082–1088. doi: 10.1128/jb.126.3.1082-1088.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]



