Summary
Circadian clocks organize our inner physiology with respect to the external world providing life with the ability to anticipate and thereby better prepare for major fluctuations in its environment. Circadian systems are widely represented in nearly all major branches of life except archaebacteria, and within the eukaryotes the filamentous fungus Neurospora crassa has served for nearly half a century as a durable model organism for uncovering the basic circadian physiology and molecular biology. Studies using Neurospora have clarified our fundamental understanding of the clock as nested positive and negative feedback loops regulated through transcriptional and post-transcriptional processes. These feedback loops are centered on a limited number of proteins that form molecular complexes, and their regulation provides a physical explanation for nearly all clock properties. This review will introduce the basics of circadian rhythms, the model filamentous fungus Neurospora crassa, and provide an overview of the molecular components and regulation of the circadian clock.
Keywords: Circadian rhythms, Negative feedback, Post-translational modification, Protein phosphorylation, Frequency, White collar
Introduction
Organization is an underlying theme in the study of living systems. This truth is evident in any number of basic cellular functions. By necessity, the execution of all biological functions falls within the persistent influence of an oscillatory external environment. It is of little surprise then that the organization of living systems extends into temporal perception, and even anticipation. This review focuses on the circadian system of internal daily organization, introducing the core biological clock and the rhythms that it drives. Because excellent reviews have recently appeared detailing a variety of fine points on circadian regulation (de Paula et al., 2007, Dunlap et al., 2007, Loros et al., 2007, Brunner & Kaldi, 2008, Jinhu & Yi, 2010), this review is aimed more towards providing a historical context and introducing the field, the key molecular questions and players, and focuses on regulation of these players through phosphorylation.
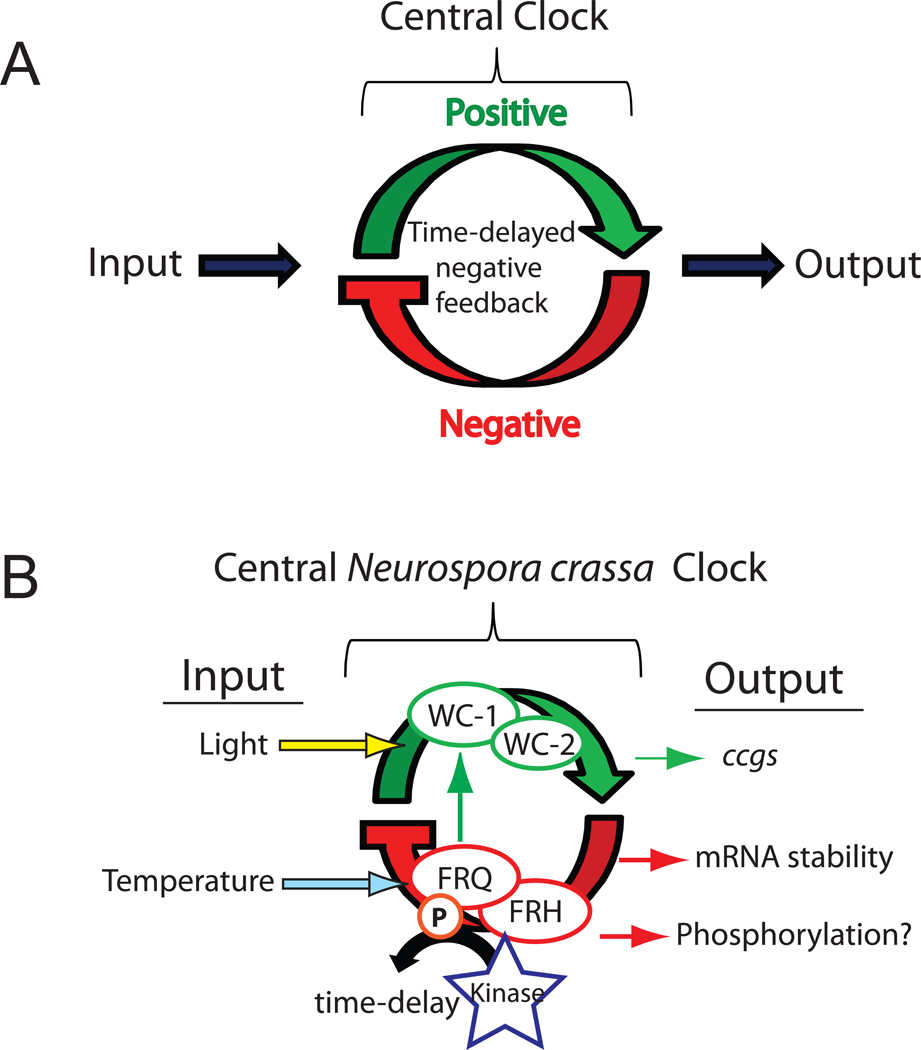
Circadian rhythms have been reported in almost all branches in the universal tree of life (Dunlap, 1999). At its most basic, the circadian system can be thought of in three parts as input, a central oscillator, and output (Figure 1A, Pittendrigh & Bruce, 1957). The central oscillator functions as an endogenous self-sustaining rhythm generator with a period approximating that of the 24-hour cycle of earth’s rotation. Circadian rhythms manifest at all levels of tissue organization, yet they are generated at the level of individual cells and persist in the absence of external stimuli. Furthermore, the period length of the rhythms controlled by circadian clocks is largely the same at different ambient temperatures reflecting a mechanism for compensation. This last feature is central to the concept of a ‘clock’ and sets circadian clocks apart from other kinds of observable biochemical rhythms such as the cell cycle. However, clocks are not temperature-insensitive; they are universally associated with the perception of light and temperature cues that provide time information about the environment. This allows internal time to be appropriately phased with respect to external time so that appropriate biochemical, physiological or behavioral activities, regulated clock output, happen at appropriate times of day. These features – circa 24 hour period length, persistence in the absence of environmental cues, a compensation mechanism to keep period length similar under different conditions of temperature and nutritional, and ability to use changes in the environment as time signals – are the cardinal characteristics that make a biological rhythm a circadian rhythm.
Figure 1. Oscillator model for the circadian clock.
(A) Circadian systems comprise three essential elements: an endogenous self-sustaining oscillator, an ability to sense environmental time cues (Input), and physiological output tied to the oscillator at distinct phases. Environmental variables such as light and temperature can entrain or couple the core clock via input pathways. The central oscillator is based on the interplay between positive acting factors driving the expression of negative factors which feedback to inhibit the positive complex. A critical component to this feedback is a mechanism for time delay of the negative state variable that reflects the ~24 hour nature of the physiological rhythms. (B) The organization of molecular components of the feedback loop in Neurospora crassa. The entraining variables of light and temperature impact different parts of the oscillator, light acting through the photoreceptor function of WC-1 to induce transcription of frq and temperature acting to modulate amounts of FRQ. WC-1 and WC-2 form the WCC complex. FRQ and FRH form the FFC complex. Rhythmic output is primarily generated through rhythmic expression of clock-controlled genes (ccgs) but can also be due to changes in mRNA stability and possibly phosphorylation. Kinases contribute to the time-delay by influencing the stability of FRQ.
This review will focus on contributions to the field of chronobiology gleaned from the study of the circadian system in Neurospora crassa, with a detailed look at the central clock protein FREQUENCY (FRQ), its protein-protein interactions and its regulation through post-translational modification. While oscillatory behavior has been observed in strains lacking FRQ (see, de Paula et al., 2006, Shi et al., 2007, Schneider et al., 2009) these noncircadian rhythms will not be covered here. Finally a brief comparison between the fungal and animal clocks will be made to highlight the many conserved features between these two Kingdoms.
Introduction to Neurospora crassa
Neurospora crassa has a long history as a genetic model dating from when Bernard O. Dodge recognized the potential of Neurospora genetics and encouraged Thomas H. Morgan and Carl Lindegren to pursue further work (Tatum, 1961). It was through Dodge’s enthusiasm that George W. Beadle, along with research associate Edward Tatum, began their biochemical genetic studies by isolating metabolic mutants of Neurospora (Beadle & Tatum, 1941), fully establishing the organism as a modern genetic model. Neurospora is a filamentous fungus classified under the Phylum Ascomycota and is widely distributed in nature (Davis, 2000). Neurospora is commonly found in its vegetative state growing as a mycelium of haploid hyphae. The tubular hyphae form incomplete septae, undergo cytoplasmic streaming, and contain multiple nuclei that can be of different genotypes, comprising a heterokaryon. Neurospora crassa undergoes both sexual and asexual reproductive cycles. Upon certain cues, mycelia can undergo a developmental switch and begin to form aerial hyphae that segment into the asexual spores called macroconidia. Macroconidia, also known simply as conidia, are characteristically bright orange due to carotenoid pigments and are strongly hydrophobic. This process of conidia production is controlled in a daily manner and peaks just before dawn.
The physical manifestation of the internal clock through conidial production is perhaps the primary reason that Neurospora helped to found the molecular era of clock research and continues to contribute to our understanding of clock biology. These rhythms manifest as alternating zones of mycelia and conidia production that form a linear pattern down a simple glass tube (so called race tubes, Figure 2A). Briefly, a small amount of an agar-based defined growth medium is injected into hollow tube, and after the medium is solidified a culture is inoculated with conidia at one end. This culture is allowed to grow under constant light (LL) prior to transfer to constant darkness (DD) in order to synchronize the clocks in the growing hyphae and set them to subjective dusk. Under these conditions, Neurospora grows with a quasi-linear growth rate down the tube where, under control of the circadian oscillator, a developmental switch is thrown into one of two positions. In the late subjective night the mycelia that are laid down are determined to differentiate and they do so by forming patches of aerial hyphae leading to conidial production. As time and growth proceed, mycelia laid down later in the subjective day and early evening are determined never to differentiate. This growth pattern is determined by the circadian time of day when the mycelia at the growth front are laid down and is fixed once the growth front has passed. Because distance along the tube correlates with time since the light to dark transition, this time-history of asexual differentiation potential easily allows conversation of spatial information into temporal information for the calculation of period and phase (Figure 2A). This self-reporting assay has facilitated mutant screens that identified many central circadian clock components and thereby contributed to our understanding of the basic mechanism behind all circadian rhythms. More recently other molecular reporter systems have been developed to visualize and automate the high-throughput recording of rhythms, and even individual molecules, within the clock (Figure 2B, Morgan et al., 2003, Gooch et al., 2008, Schafmeier et al., 2008, Castro-Longoria et al., 2010, Chen et al., 2010).
Figure 2. Recording and analysis of rhythms.
(A) Race tube assay - glass tubes containing growth media are inoculated at one end and placed in constant light (LL) for one day prior to shifting to constant darkness (DD) which synchronizes the clocks in the culture by setting them all to subjective dusk. Mycelia then ‘race’ down the tube growing at a semi-constant rate such that distance grown approximates time since the light to dark transfer in a linear fashion. Production of the asexual conidia is gated in a daily, phase-specific manner by the circadian clock. These bands of conidia provide a self-reporting time-history of development. This information can be digitized and densitometric analysis of the images result in raw numerical data. Peak of conidial production is often defined as the phase reference point allowing calculation of the period with high precision. (τ = mean period calculation, SD = standard deviation, n = number of tubes for which period was determined). (B) Synthetic engineering of the firefly luciferase enzyme connected to the frq promoter provides a highly quantitative, automated measurement of rhythmic gene expression readily adaptable to high throughput methods. Recording of rhythms of the central FRQ oscillator via luciferase activity also allows investigators to make a distinction between circadian regulation within the core clock and circadian or other regulation of growth wherein different metabolic rhythms might control conidiation and mask circadian output. This system can be adopted to follow regulation of any ccg.
The circadian clock in Neurospora crassa
In Neurospora, circadian rhythms were first documented in 1959 (Pittendrigh et al., 1959). These developmental rhythms fulfilled the criteria discussed above for defining them as circadian; they had a sustained period of ~22.5 hours in constant conditions; the phase was set by a single transition from light to dark; they could be entrained by a 24-hour light cycle; the period was not very dependent upon ambient temperature. A variety of Neurospora strains that helped to visualize rhythms appeared contemporaneously in the literature, all with rhythmic-inspired nomenclature including patch (Stadler, 1959), clock and wrist watch (Sussman et al., 1965), timex and band (Sargent et al., 1966), all revealing underlying metabolic rhythms through production of conidia. Interestingly though, not all of the rhythms exposed in these strains were subsequently shown to be truly circadian in nature and so most of the strains have fallen out of use. However, band proved very useful, and the band phenotype has subsequently allowed isolation of mutant strains with altered clock properties including period and temperature compensation (Feldman & Hoyle, 1973, Gardner & Feldman, 1980, Gardner & Feldman, 1981, Loros et al., 1986). Complementation analysis identified a single locus identified early on by Feldman, called frequency (frq), that displayed long, short, and arrhythmic periods some of which also altered or disrupted temperature compensation (Gardner & Feldman, 1980, Loros et al., 1986). These early analyses demonstrated the genetic basis of circadian rhythms and showed that a single allele could control many of the core circadian properties in Neurospora.
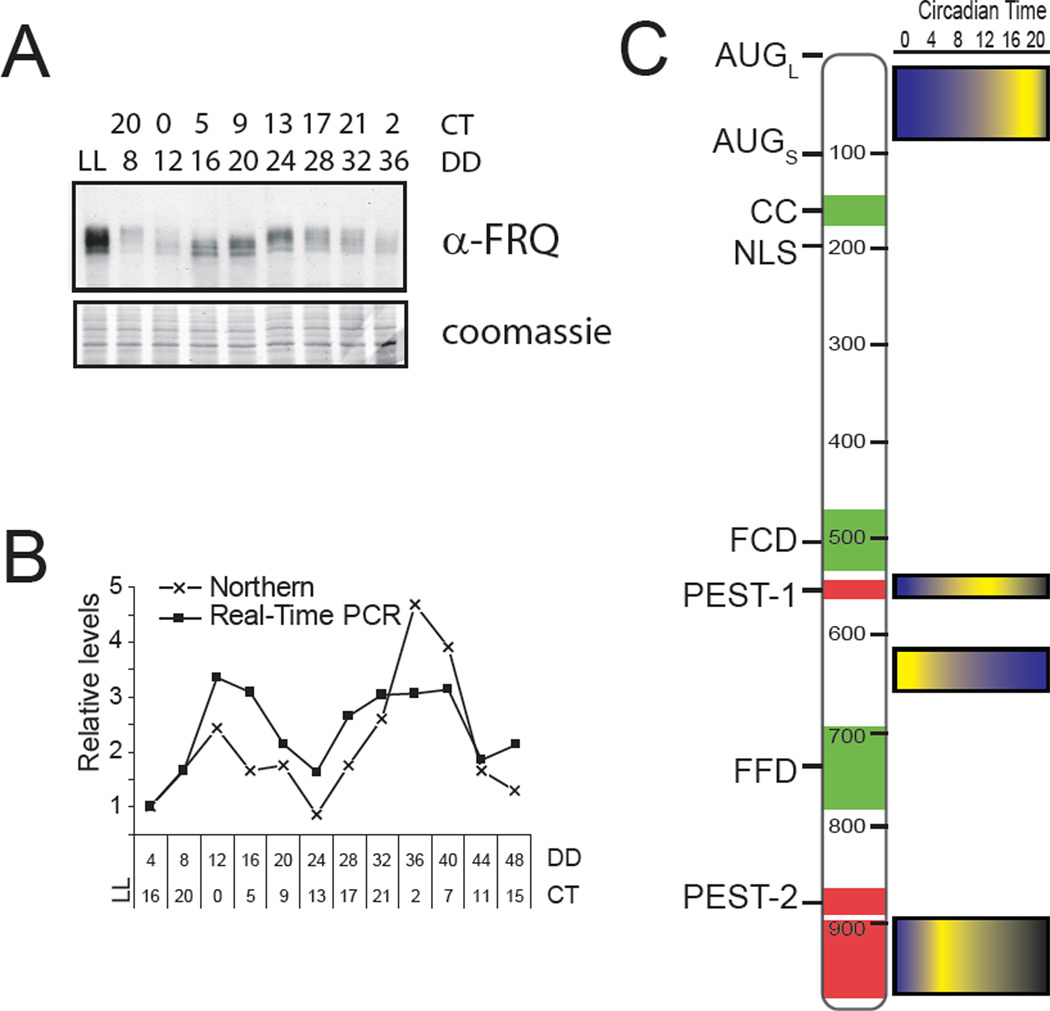
The cloning of frq in 1986 and its subsequent publication (McClung et al., 1989) opened up the clock in Neurospora to molecular dissection, leading to a concrete understanding of the mechanism behind circadian rhythms (Dunlap, 2008). The molecular mechanism underlying circadian rhythms in this system revolves around a central time-delayed negative feedback loop (Figure 1B, Aronson et al., 1994). This negative feedback, first demonstrated in Neurospora to be essential for rhythms, is now known to be a conserved feature of all circadian rhythms (Dunlap, 1999). Within the circadian system positive elements drive activation of negative elements, which feed back to limit their own activation. Key to this auto regulatory feedback is a built in system for time-delay, thus extending the period to approximately 24-hours. Both frq message and a long and short form of (see below) the FRQ protein are rhythmically expressed in a 22.5-hour cycle under constant conditions with an approximately four-hour phase difference (Figure 3, Aronson et al., 1994, Garceau et al., 1997). FRQ is also phosphorylated in a time-dependent manner as more fully described below (Garceau et al., 1997, Baker et al., 2009, Tang et al., 2009). The period of the molecular rhythms seen in frq/FRQ match that of the conidiation rhythm, frq mutants that alter the banding period also alter the molecular period (Garceau et al., 1997), and conditions that stop FRQ cycles also result in arrhythmic strains. frq expression is regulated by white collar-1 and white collar-2 (Crosthwaite et al., 1997). These GATA-family transcription factors form a heterodimeric complex through interaction with their PAS domains, named the White Collar Complex (WCC, Linden & Macino, 1997, Ballario et al., 1998, Cheng et al., 2002).
Figure 3. FREQUENCY, a central molecular component in the Neurospora crassa circadian clock.
(A) Representative Western blot of FRQ to visualize dynamic protein levels and phosphorylation. Each lane contains equal portions of protein extracted from cultures grown in constant darkness (DD) over one circadian day and harvested at four-hour intervals (LL represents cultures from constant light). FRQ undergoes dual molecular rhythms in protein abundance and phosphorylation peaking in the late subjective day (CT = circadian time where hour 0 is defined as subjective dawn). (B) Similar to the protein, frq message undergoes molecular rhythms in abundance. mRNA levels were measured both by densitometric analysis of Northern blots and quantitative real-time PCR and show similar phase of maximum expression slightly after subjective dawn (CT0-4). This rhythmic production of mRNA is driven by the WCC. (C) Schematic of the primary structure of FRQ showing the position of the two translational start sites and domain structure and time-of-day-specific phosphorylation of FRQ as revealed by tandem mass spectroscopy. CC – coiled-coiled domain, NLS – nuclear localization signal, FCD – FRQ-CKI interacting domain, FFD – FRQ-FRH interacting domain. Domains in green provide protein-protein interaction giving FRQ a scaffold-like function and domains in red regulates stability. Phosphorylation is seen nearly over the entire protein however, here spatial and temporal domains that change phosphorylation state in concert are indicated by the blue/yellow colored gradients on the right. Degrees of phosphorylation are color coded such that the highest intensity yellow represents the time-of-day peak in phosphorylation for the corresponding region of the protein. This panel of the figure is modified from Baker et al. (2009).
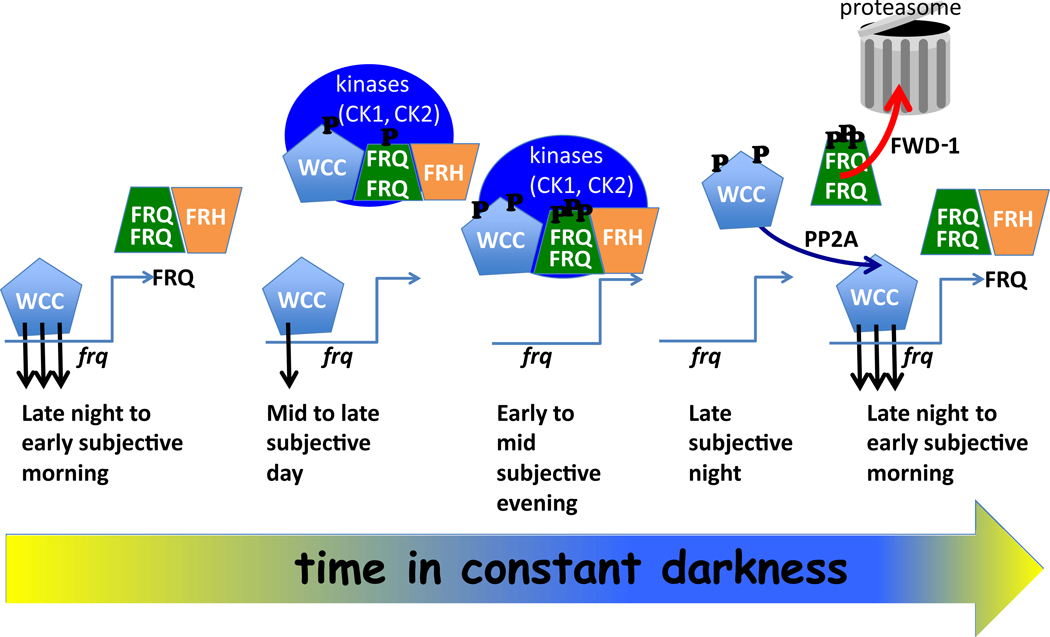
A good way to understand the operation of the clock is to follow the events in time (Figure 4). In Neurospora, the core oscillation occurs in two major steps (Merrow et al., 1997). Starting in the late subjective night the WCC binds to the frq promoter to induce frq expression that peaks in the early subjective morning; this is the positive arm in the feedback process (Crosthwaite et al., 1997, He et al., 2002, Belden et al., 2007). FRQ synthesis begins, and within the first 3–6 hours of expression FRQ forms functional homodimers through a coiled-coil domain (Cheng et al., 2001) and enters the nucleus (Luo et al., 1998) to rapidly repress frq transcription via a time-of-day-specific increase in its interaction with the WCC (Denault et al., 2001, Hong et al., 2008, Baker et al., 2009). The interaction of FRQ with the WCC facilitates phosphorylation of the WCC that inactivates it, removing the WCC from the frq promoter, and this negative feedback is further helped by eventual clearance of WCC from the nucleus (Schafmeier et al., 2005, He et al., 2006, Hong et al., 2008, Schafmeier et al., 2008). As negative feedback reaches completion in the mid to late day, frq expression drops as does the rate of FRQ synthesis; FRQ levels peak in the late subjective day, and the cycle is completed by the delayed release of FRQ-mediated repression, a process that requires the extensive phosphorylation of FRQ over the next 14–18 hours (Merrow et al., 1997, Schafmeier et al., 2006). Eventually FRQ phosphorylation makes it attractive to its ubiquitin ligase FWD-1 (F-box/WD-40 repeat-containing protein, He et al., 2003), FRQ is ubiquitylated and targeted to the proteasome where it is turned over. The cycle can then restart.
Figure 4. A time line of biochemical and molecular processes in the Neurospora clock.
In the late night at the left hand side, the WCC is rapidly cycling through the frq promoter (black arrows), actively driving transcription of frq and being rendered unstable in the process so that it rapidly turns over. WCC in the figure denotes the active complex only. FRQ is translated, dimerizes, and makes a stable complex with FRH. This complex interacts with the WCC and serves as a platform to introduce kinases, especially CK1 which stably associates with the complex; CK2 and other kinases are transiently associated. By mid to late subjective day CK1 and the other kinases are phosphorylating the WCC to inactivate it, and FRQ to change its structure. By the early subjective night WCC is inactivated, frq transcription is nearly stopped, and FRQ is becoming highly phosphorylated, so that by the late subjective night it transiently interacts with its ubiquitin ligase FWD-1 leading to turnover in the proteasome. After FRQ turns over in the late night, the frq promoter can be reactivate by newly synthesized WCC or by WCC reactivated through the action of protein phosphatases such as PP2A.
In addition to the negative feedback loop there is a nested positive feedback loop. Positive feedback was first observed when loss of FRQ was seen to result in reduced WC-1 levels through a post-translational mechanism (Lee et al., 2000, Cheng et al., 2001). This occurs independently of the transcriptional autoregulation of wc-1 and wc-2 (Kaldi et al., 2006, Neiss et al., 2008). The basis for this is that WC-1 is unstable when it is transcriptionally active; that is, WC-1 appears to be turned over as a natural part of its activity cycle in a manner similar to that known for transcription factors in yeast (Tansey, 2001). Thus, early in the cycle when WC-1 can bind to DNA and activate transcription it is unstable because it is active, but later in the cycle when WCC has been inactivated via phosphorylation and can no longer bind to DNA, WC-1 is stable so its levels increase (He et al., 2005, Schafmeier et al., 2005, Shi et al., 2010). This stabilization is thus observed late in the negative cycle when FRQ levels are high and hyperphosphorylated (Schafmeier et al., 2006) and is an indirect result of FRQ’s repressive activity on the WCC. This continued inhibition is in part mediated through subcellular location by maintaining inactive WCC in the cytosol and also keeping chromatin in a repressed state at the frq promoter (Belden et al., 2007, Cha et al., 2008, Schafmeier et al., 2008). Interestingly, WC-1 can be found with the promoter throughout the day, even when it is inactivated whereas WC-2 binds cyclically (Belden et al., 2007).
Throughout this cycle FRQ interacts with several proteins other than the WCC, many in a phase-dependent manner (Hong et al., 2008, Baker et al., 2009). Upon translation FRQ binds stoichiometrically to FRH (for FRQ-Interacting RNA Helicase, Cheng et al., 2005). FRH is essential for negative feedback in the Neurospora clock as strains with disrupted function are arrhythmic and express high levels of frq as a result of uninterrupted WCC binding to the frq promoter (Guo et al., 2009, Shi et al., 2010). FRH was first identified biochemically, as the name suggests, through its interaction with FRQ (Cheng et al., 2005) and its importance later confirmed and clarified through a genetic screen (Shi et al., 2010). Together FRQ and FRH form the FRQ-FRH Complex, FFC, that acts as part of the negative arm of the clock. Formation of the FFC is independent of FRQ dimerization but is crucial for stability of FRQ as knock down of FRH decreases levels of FRQ (Cheng et al., 2005, Guo et al., 2010). Interestingly, FRQ’s degradation in the absence of FRH does not require FWD-1 (Guo et al., 2010) potentially suggesting a structural component to FRQ stability. FRH also interacts with the WCC in the absence of FRQ (Cheng et al., 2005) but, cryptically, FRQ mutants that lack FRQ-FRH interaction also block FRH-WCC interaction (Guo et al., 2010). In Neurospora FRH is essential; however, one non-lethal allele frhR806H results in arrhythmicity while maintaining normal growth (Shi et al., 2010). This shows that the essential role of FRH for survival can be functionally separated from its role in the core circadian clock. This point mutation results in a protein that maintains interactions with FRQ but yields a FRQ/FRH complex that can no longer meaningfully interact with the WCC to bring about its inhibition. WCC levels are thus very low in strains bearing frhR806H while frq levels are high, consistent with the model where transcriptionally active WC-1 is turned over rapidly, (Guo et al., 2010, Shi et al., 2010).
FRH is homologous to the yeast RNA-binding protein Dob1p/Mtr4p. Mtr4p is a component of the TRAMP complex involved in RNA metabolism and turnover and interacts with the exosome complex. While no clear evidence exists that FRH binds the Neurospora homologs of the TRAMP complex, it does bind the exosome components RRP6 and RRP44 and is proposed to play a conserved role in the Neurospora exosome (Guo et al., 2009). Knock down of the exosome catalytic subunit RRP44 also results in loss of the overt conidiation rhythm and results in long period rhythms in FRQ protein. FRH can also bind frq mRNA and influence its message stability and poly(A) tail length (Guo et al., 2009). These data suggest an added layer to the mechanism of negative feedback in addition to simple inhibition of the WCC through post transcriptional activity directly on frq.
Post-translational modification in circadian rhythms
FRQ has no known enzymatic activity. This leads to the prediction that, in agreement with the protein interactions described above, the primary function of the post-translational modifications (PTMs) of FRQ may be to regulate its use as a platform for recruiting, in a temporal fashion, other factors involved in the negative feedback process required to sustain rhythms (He et al., 2006, Baker et al., 2009). The circadian circuitries for both the positive and negative complexes are broadly controlled by PTMs (Mehra et al., 2009). As previously mentioned, phosphorylation of the negative factors is typical, rhythmic, and coincident with their degradation, and it appears to be the major contribution determining the time constant of the feedback loop. Phosphorylation of WCC components also plays a role in the positive loop where it helps to regulate DNA binding and transcriptional activation. While other PTMs such as ubiquitination, sumoylation, and acetylation are involved in the circadian clock in other systems (Mehra et al., 2009), phosphorylation and the kinases and phosphatases that regulate this are the primary controllers of circadian period length in all systems. Indeed even the prokaryotic circadian system in Synechococcus is based on phosphorylation and can cycle without the aid of transcription or translation (Nakajima et al., 2005). Our understanding of phosphorylation has expanded to include both degradative and non-degradative consequences which are often dependent on where and when the protein is phosphorylated.
Phosphorylation of FRQ
FRQ is phosphorylated immediately after synthesis and becomes increasingly so throughout the circadian day in a remarkably choreographed manner (Garceau et al., 1997, Baker et al., 2009). This phosphorylation is regulated, as the shifts in the mobility of FRQ on SDS gels caused by phosphorylation are limited in number, are phase-specific, and are reproducible in magnitude across experiments (Figure 3A). FRQ is also phosphorylated in constant light, although under these conditions FRQ is heterogeneously phosphorylated across a range of greater than 85 individual locations (Baker et al., 2009, Tang et al., 2009). Mechanistically phosphorylation primarily influences FRQ stability and period length, as most mutations that reduce the level of phosphorylated FRQ also tend to increase its stability and lead to long periods (Liu et al., 2000, Gorl et al., 2001, Ruoff et al., 2005, He et al., 2006). By the end of the circadian day the longest unmodified region of the 998 amino acid full length FRQ is less than 150 amino acids. So it is probable that by the end of the FRQ phosphorylation cycle the protein is in a very different conformation than when it was first translated, consistent with a model where FRQ serves as a time-dependent platform to facilitate interactions among clock components.
Experiments designed to understand the roles of different domains are consistent with this view. FRQ contains two PEST domains characterized by their amino acid content (Figure 3C, Merrow & Dunlap, 1994). Deletion or site-directed mutation of PEST-1 leads to a reduction in FRQ phosphorylation and degradation as well as loss of conidial banding (Gorl et al., 2001, Baker et al., 2009, Tang et al., 2009). Among sites in PEST-2 phosphorylated in vitro are S885 and S887 (Schafmeier et al., 2006). Mutation of these sites to asparagine to prevent phosphorylation resulted in loss of circadian rhythmicity on race tubes and had no effect on FRQ turnover or negative feedback. Instead, it was proposed that these sites help to stabilize the positive arm through a nested positive feedback loop. However, this non-degradative function is not entirely clear as mutation of S885 and S887 to alanine resulted in no clock phenotype (Tang et al., 2009). The phosphorylation status of FRQ is also related to the subcellular steady-state distribution of FRQ (Diernfellner et al., 2009). As FRQ progresses through the cycle and picks up increasing phosphate it switches from being predominantly nuclear, where it represses WCC activity, to predominantly cytoplasmic (Luo et al., 1998).
Recently the time course of FRQ phosphorylation has been followed through the use of heavy isotope labeling in conjunction with tandem mass spectrometry (Baker et al., 2009). In general many individual phosphorylation events seem to lack unique roles. Instead most phosphorylations occur in spatial and temporal clusters with the result that they probably form charged regions on the FRQ protein, and these lead to changes in the activity of FRQ. FRQ is of course unmodified immediately after its synthesis but phosphorylation events occur rapidly in the middle part of the proteins between the PEST-1 and FFD domain (Figure 3C). The roles of these events are not well understood as mutational analysis did not alter circadian rhythms at steady state. Soon after this, phosphorylations are clustered near to the C-terminus, and serine to alanine mutations of these residues, or deletion of the entire C-terminus (FRQSHORT), results in FRQ protein that is less stable and that supports a short period rhythm (Baker et al., 2009, Tang et al., 2009). This indicates that phosphorylations here act to stabilize FRQ. During the middle of the cycle the PEST-1 domain shows a dramatic increase in phosphorylation. Functional studies into the role of phosphorylation at this region results in a period length that is longer than normal and more stable FRQ, suggesting that phosphorylation of these residues is needed to promote turnover of FRQ. Still later other domains of FRQ, predominantly residues specific to the long FRQ isoform (Figure 3) become modified and serine to alanine mutations in this region result in long period clocks suggesting again a role in promoting turnover. In all, the sequential modifications appear to be acting to modulate the structure of FRQ with the goal of regulating interactions with other proteins as further described below, among these the ubiquitin ligase FWD-1 that will lead to the turnover of FRQ.
Phosphorylation of the WCC
WC-1 and WC-2 are both regulated by phosphorylation and several sites on these proteins have been identified using methods in mass-spectrometry (He et al., 2005, Huang et al., 2007, Baker et al., 2009, Sancar et al., 2009). In the dark WC-2 is phosphorylated rhythmically throughout the circadian day with maximal phosphorylation around late subjective day (CT8-12, Schafmeier et al., 2005). Because WC-2 levels are nearly constant, the cycle in phosphorylation must be mediated by kinases and phosphatases. This light-independent phosphorylation is dependent on FRQ and dephosphorylation of the WCC increases its binding to DNA as noted above (He et al., 2005, Schafmeier et al., 2005). Although to date MS/MS analysis of WC-2 has only identified one phosphorylated site at S433 (Baker et al., 2009, Sancar et al., 2009), 2D gels of WC-2 suggest at least 8 possible phosphorylation states (Schafmeier et al., 2005). Mutation of S433 to alanine results in a moderate decrease in circadian period length and a slight increase in transcriptional activity (Sancar et al., 2009). WC-1 is also phosphorylated in the dark but it is not clear if this is time-dependent (He et al., 2005). Upon exposure to light WC-1 becomes hyperphosphorylated after 15 minutes, a time corresponding to a reduction in light-induced transcription and degradation of WC-1 (Talora et al., 1999). To date at least 10 phosphorylation sites have been identified on WC-1 (He et al., 2005, Baker et al., 2009, Sancar et al., 2009) with at least six clustered in the C-terminus of WC-1 near its DNA-binding domain (He et al., 2005). Phosphorylation of this region is sequential, FRQ-dependent, and required for negative feedback (Huang et al., 2007). This shows that the core negative feedback loop of FRQ inhibiting its own transcription is closed around FRQ promoting phosphorylation of the WCC.
Regulatory enzymes in the clock
All of the phosphorylation events that regulate the ticking of the clock require the activity of kinases and phosphatases. Molecular dissection has revealed several important players in these processes. Kinases included on this list are the acidic-directed casein kinases 1 and 2 (CK1a and CK2), the Neurospora homolog of checkpoint kinase-2 (PERIOD-4, PRD-4), as well as CAMK-1, and the basophilic Protein kinase A (PKA, Gorl et al., 2001, Yang et al., 2001, Yang et al., 2002, Yang et al., 2003, Pregueiro et al., 2006, Huang et al., 2007, Mehra et al., 2009). FRQ physically interacts with several of these kinases including CK1a, CK2, and PRD-4 (Gorl et al., 2001, Pregueiro et al., 2006, Baker et al., 2009). This group of kinases is joined by several known phosphatases including Protein Phosphatase-1 (PP1), PP2a, and PP4 (Yang et al., 2004, Cha et al., 2008). Many of these same enzymes perform similar regulatory roles in animal circadian clocks (Table 1).
Table 1.
Functional classes of clock proteinsa
| Class | Function | Neurospora | Drosophila | Chordata (vertebrates) |
|---|---|---|---|---|
| Negative arm | Phosphoprotein scaffold | FRQ | ||
| PER | PER1-3 | |||
| Phosphoprotein binding partner |
FRH | |||
| TIM | ||||
| CRY1-2 | ||||
| Positive arm | PAS-domain transcription factor |
WC-1 | CYC | BMAL |
| WC-2 | CLK | CLOCK | ||
| Regulatory enzymes |
Protein kinase | CK1a | DBT | CK1δ/ε |
| CK2 | CK2 | CK2 | ||
| SGG | GSK3 | |||
| PRD-4 | Chk2 | |||
| CAMK-1 | ||||
| PKA | PKA | |||
| Protein phosphatases | PP1 | PP1 | PP1 | |
| PP2a | PP2a | PP2a | ||
| PP4 | ||||
| Degradation (F-Box domains) |
||||
| FWD-1 | SLIMB | Β-TRCP | ||
| JETLAG |
Proteins in the same row share sequence/domain structure and serve orthologous functions
Out of the kinases several lines of data suggest FRQ has the most intimate physical interaction with casein kinase 1 (Gorl et al., 2001, He et al., 2006, Querfurth et al., 2007, Baker et al., 2009). Neurospora contains two casein kinase 1 orthologs, ck1a and ck1b. Of these ck1a is more similar to the mammalian CK1ε, functionally conserved in mammalian circadian rhythms. CK1a participates in regulation of both FRQ and the WCC whereas CK1b appears to have no role in the clock (Gorl et al., 2001, He et al., 2006). FRQ interacts with CK1a through the FRQ-CK1a interacting domain (FCD, He et al., 2006) that is positioned on FRQ just N-terminal to the heavily phosphorylated PEST-1 domain responsible for FRQ degradation (Figure 3C, Gorl et al., 2001, Baker et al., 2009). FRQ acts as a substrate recruiting scaffold by bringing CK1a to the WCC (He et al., 2006, Baker et al., 2009). FRQ-CK1a interaction is continuous throughout the circadian day but increases specifically early in the cycle when hypophosphorylated FRQ is in complex with the WCC (Baker et al., 2009). This timing is consistent with the role of CK1a in phosphorylating the WCC leading to its inactivation and repression (Schafmeier et al., 2005, He et al., 2006). FRQ is also a target of CK1a phosphorylation. FRQ can be phosphorylated at a minimum of 41 serine/threonine by CK1a in vitro (Tang et al., 2009) and many of the same sites are found to be phosphorylated in vivo (Baker et al., 2009, Tang et al., 2009). Introduction into the Neurospora ck1a of a mutation shown in Drosophila to increase period in flies also increases period length in Neurospora. Since circadian period length is tied to FRQ degradation rate this results suggests FRQ is a direct target of ck1a and reveals a conserved mechanism across widely divergent phyla (He et al., 2006). Furthermore, placing just the FCD and PEST-1 regions of FRQ into GFP is sufficient to increase the degradation rate of the recipient protein (Querfurth et al., 2007), consistent with the idea that CK1a binds to and phosphorylates FRQ leading to its turnover through these domains. CK1a describes several different protein isoforms that vary in their C-terminal tail length as a result of mRNA splicing. At least two major isoforms interact with FRQ but are not found in complex with each other (Querfurth et al., 2007). Since FRQ functions as a dimer, with both molecules containing a FCD, it becomes an interesting question as to how these protein complexes are assembled.
Casein kinase 2 was biochemically purified from Neurospora extracts based on its ability to phosphorylate FRQ (Yang et al., 2002). CK2 is functions as a heterodimer with one subunit encoded by a single catalytic domain, cka, and one of two regulatory domains encoded by ckb-1 and ckb-2. Of the two regulatory subunits only ckb-1 is known to be involved in the circadian clock (Yang et al., 2003) whereas ckb-2 appears to have no role in the clock. CK2 physically associates with FRQ although this interaction appears to be weak and possibly transient suggesting FRQ is primarily a substrate of CK2 (Baker et al., 2009). Mutation of cka abolishes rhythms in frq and several ccgs in constant darkness (Yang et al., 2003). Similar to CK1a, many of the phosphorylation sites identified on FRQ in vivo can also be phosphorylated by CK2 in vitro (Tang et al., 2009). Mutation of in vivo identified phosphorylation sites on FRQ that conform to the predicted motif of CK2 also resulted in lengthening of the circadian period (Yang et al., 2003, Baker et al., 2009, Tang et al., 2009). CK2 is unique in its regulation of FRQ stability differing from CK1 in one major functional role. Cloning of two classic circadian mutants defective in temperature compensation, period-3 and chrono, identified separate point mutations in both cka and ckb-1 respectively (Mehra et al., 2009). Mutation of specific residues on FRQ phosphorylated by CK2 in vitro phenocopies the chrono circadian behavior across a range of temperatures, thus identifying the first molecular process involved in regulating temperature compensation.
Finally, whenever there is kinase activity, phosphatases are readily available to counterbalance the equation. PP1 and PP2a represent two major families of protein phosphatases widely conserved in eukaryotes (Gallego & Virshup, 2005), and both are important for circadian rhythms. Mutation of ppp-1 (the catalytic subunit of PP1) destabilizes FRQ and results in a slightly reduced period, while PP2a activity influences frq transcription (Yang et al., 2004). As mentioned WC-2 undergoes rhythmic phosphorylation while levels of the protein do not cycle, and mutation of rgb-1, the catalytic subunit of PP2a, causes constitutive hyperphosphorylation of WC-2 (Schafmeier et al., 2005). A recent report showed that WC-2 rapidly shuttles between the nucleus and cytoplasm and that this is required for dephosphorylation by rgb-1 of inactive, hyperphosphorylated species in the cytoplasm (Schafmeier et al., 2008). One last PP2a-related phosphatase, PP4, plays a similar role in regulating WCC localization and transcriptional control. Knock-out of PP4 also results in a short rhythm and hyperphosphorylated FRQ (Cha et al., 2008). Which of these proteins plays the major role in WCC localization is still under debate and it is possible that both contribute equally because loss of function of neither rgb-1 nor pp4 completely abolishes circadian rhythmicity.
Input and output in the clock
The central clock components described above are also sufficient to explain the molecular bases of other features of the oscillator such as input and output. WC-1 is also a blue-light photoreceptor and responsible directly or indirectly for all known light responses in Neurospora (Ballario et al., 1996, Froehlich et al., 2002, He et al., 2002). Light activation results in the WCC directly binding to and inducing transcription of hundreds of genes (Chen et al., 2009, Smith et al., 2010). WC-1 light activity is due its association with flavin adenine dinucleotide which becomes covalently linked to the conserved light sensing domain (LOV domain for Light Oxygen Voltage. For a comprehensive review of photosensing in Neurospora see, Dunlap & Loros, 2005, Chen et al., 2010). Light induction by the WCC drives an increase in frq levels. Additionally, because frq cycles, the impact of light on the oscillator is dependent on time. When frq levels are low and on the rise (before subjective dawn) brief light pulses will drive frq mRNA to peak levels and thereby advance the clock to the time corresponding to highest frq expression, mid to late morning. Conversely, when frq levels are decreasing light exposure will again induce frq mRNA to peak levels, this time setting the clock back leading to phase delays. This provides a molecular explanation for light entrainment and phase resetting (Crosthwaite et al., 1997).
Input to the clock can be modified VVD, a small (21 kDa) flavin-binding blue-light photoreceptor consisting of a PAS domain variant called a LOV (light, oxygen or voltage) domain, and an N-terminal cap (Heintzen et al., 2001, Zoltowski et al., 2007) that is responsible for conferring photoadaptation in Neurospora (Heintzen et al., 2001; Schwerdtfeger and Linden, 2001; Schwerdtfeger and Linden, 2003; Shrode et al., 2001). Photoadaptation refers to the phenomenon in which, following light exposure in wild type Neurospora, elevated levels of light-induced gene transcription are transient and generally return to pre-induction levels within two to four hours, and Neurospora can detect changes in light intensity rather than simply seeing “on versus off” (Schwerdtfeger and Linden, 2001, Schwerdtfeger and Linden, 2003). The molecular basis of photoadaptation is that VVD is rapidly and highly light induced; light also causes the N-terminal cap to come off the LOV domain with the result that, in solution, VVD forms a rapidly exchanging dimer in light (Zoltowski et al., 2007). This suggested that the VVD LOV domain might interact in vivo with PAS domains from other proteins like those in WC-1 or WC-2 (Lamb et al., 2009; Zoltowski and Crane, 2008), and recent studies have shown this to be the case: Light induced VVD rapidly moves to the nucleus where it physically interacts with the WC-1 in the WCC to reduce its ability to activate transcription, and because VVD induction is graded with increasing light, incremental light exposures yield more VVD to inactivate the newly activated WCC, in all cases leading to photoadaptation (Chen et al., 2010; Hunt et al., 2010, Malzahn et al., 2010). VVD is itself clock regulated which leads to circadian gating of light response (Heintzen et al., 2001), and along with repressing the WCC activity in light, VVD modulates various WCC-mediated circadian clock properties to keep the light response the clock running, appropriately entrained. Specifically, VVD allows the clock to run through the dawn transition and take its principle phase cues from dusk (Elvin et al., 2005), and it contributes to temperature compensation of circadian clock phase (Hunt et al., 2007). A nice added touch is that although VVD decays with darkness, the elevated production of VVD during the day ensures that some remains at night to inactivate any WCC that is light induced due to moonlight, thereby ensuring that the clock can run (Malzahn et al., 2010).
Temperature can also act as an environment input to entrain the clock; however, it seems to have an influence primarily on FRQ (Liu et al., 1997, Pregueiro et al., 2005). At different ambient temperatures, the average level of frq expression and amplitude of frq cycling is quite similar whereas FRQ levels are seen to cycle around a higher mean level at higher temperatures. This means of course that the absolute number of molecules of FRQ corresponds to one time of day at one temperature but corresponds to a different time of day at another temperature: If a culture is rapidly shifted between temperatures, FRQ levels will not change as quickly as the temperature, so the new time of day after the shift will be the time corresponding to that level of FRQ at that temperature. As we saw before, the low point of FRQ is near to dawn, and in practice, the high point of FRQ when the clock is running at about 20–22°C is still lower than the low point when the clock is running at 28°C. Thus, explicitly then, after a discreet shift up in temperature from 21°C to 28°C at any time of day, the clock will be reset to subjective dawn, the new phase representing the low point in the FRQ cycle at the new temperature (Liu et al., 1998).
Another influence of temperature on FRQ and the clock is seen at the post-translational level. frq pre-mRNA undergoes complex temperature-sensitive alternative splicing in the 5’UTR that produces either a short (sFRQ) or long (lFRQ) form of the protein differing by 100 amino acids (Figure 3, Liu et al., 1997, Colot et al., 2005, Diernfellner et al., 2007). Under standard laboratory conditions both of these proteins are expressed together but at low temperatures (~20°C) there are roughly comparable levels of sFRQ and lFRQ while high temperatures (~28°C) result in relatively more lFRQ as well as more FRQ altogether. Either FRQ form can support circadian rhythmicity although both are required for robust rhythms across the typical temperature range of 20–30°C (Liu et al., 1997). Mutation of key phosphorylation sites that are found solely in lFRQ can decrease period length, even in the presence of sFRQ, to that found in strains only expressing sFRQ (Diernfellner et al., 2007, Baker et al., 2009). However, the function of these lFRQ-only phosphorylation sites is currently not well understood.
Output refers to the regulatory pathways and mechanisms whereby time information generated by the clock is used to regulate the time-of-day-specific expression of the overt rhythms in the cell. It is thus of singular importance since output is the true source of the biology that makes rhythms themselves important, yet it remains poorly understood at the molecular level (Vitalini et al., 2006). Recalling from above that the operation of the core clock results in cyclical activation and deactivation of the WCC transcription factor, circadian output is best understood largely as a consequence of the daily cycle of gene expression brought about by this negative feedback in the central clock, where it impacts expression of genes that do not participate in the feedback loop. In fact, in addition to frq, a large number of transcripts have been identified that undergo circadian regulation. These are referred to as clock controlled genes (ccgs, Loros et al., 1989) and they can loosely be thought of as primary ccgs that are acted upon directly by the WCC, and secondary ccgs that are directly regulated by factors regulated directly or indirectly by the WCC and are thus further downstream from the clock. Primary ccgs share the conserved DNA binding motif for the WCC and are regulated directly through the WCC binding to their promoter. An additional level of regulation can occur when the FRQ-FRH complex interacts with mRNA and the exosome through FRH (Guo et al., 2009). This allows control at the mRNA level through regulation of message stability rather than production.
Recently, a genetic screen was undertaken with the objective to identify components of the output pathway. This effort uncovered several new alleles of rrg-1, a member of the p38-type MAP Kinase involved in the osmotic stress (Vitalini et al., 2004, Vitalini et al., 2007). Osmotic sensing-2, OS-2, is a downstream kinase in this pathway and undergoes circadian regulated phosphorylation leading to its activation that is dependent on rrg-1. The circadian rhythm in phosphorylation of OS-2 is also dependent on FRQ (Vitalini et al., 2007). This system of daily activation of a kinase cascade sets up an output pathway connected to the core clock that may allow anticipation of osmotic stress associated with changes in the environment. This type of output regulation through phosphorylation also raises the possibility that the FFC in association with CK1a might have targets other than just the WCC. These examples provide a direct transcriptional and a potential post-transcriptional link between the core clock and output.
Conserved mechanisms across phyla
Concurrent with the effort to unravel the Neurospora crassa molecular clock was a concerted effort focused on Drosophila, and in the mid 1990s genetic screens and homology based searches identified potential components and drove work on vertebrate clocks. In Drosophila the first genetic screen for circadian mutants revealed a single locus, named period, having short, long, and arrhythmic phenotypes (Konopka & Benzer, 1971). Subsequent cloning and characterization of per (Bargiello & Young, 1984, Reddy et al., 1984) found that, similar to frq, the mRNA and protein are expressed in a rhythmic fashion (Siwicki et al., 1988, Hardin et al., 1990). Per also undergoes time-dependent multisite phosphorylation (Edery et al., 1994). Vertebrates have three homologous per genes, all of which are involved in the negative complex and are the target of timed phosphorylation (Tei et al., 1997, Bae et al., 2001, Lee et al., 2001). Similar to FRQ, the PER phosphoproteins in both flies and vertebrates are found in complex with other proteins required to execute negative feedback (Table 1). These binding partners are divergent among animal models (Gallego & Virshup, 2007). Similar to the Neurospora negative complex, animal PER proteins are also found associated with CK1 (Gallego & Virshup, 2007). Therefore, in all animal and fungal systems a primary phosphoprotein mediates circadian period length, is found in complex with a divergent binding partner, and acts as a scaffold for interaction with CK1.
In addition to similarities in the organization of the negative arm repressor complex, animal circadian systems were found to have striking similarities in the positive arm of the feedback loop (Table 1). per expression in both Drosophila and mammalian systems is induced by a PAS-domain containing heterodimeric transcription factor complex (Dunlap, 1999). One of these transcription factors, CLOCK, was first identified in mice and is part of the positive arm in both flies and mammals (King et al., 1997, Allada et al., 1998). In vertebrates, CLOCK binds BMAL (Hogenesch et al., 1998), a protein that shares sequence homology to WC-1 and that occupies the same place and plays the same role in the mammalian clock that WC-1 plays in Neurospora (Lee et al., 2003). In flies, CLOCK is found in association with CYCLE, an ortholog to BMAL1 (Rutila et al., 1998). These transcription factors are inhibited by the negative complex containing the PER proteins and their binding partners in the manner just described for the Neurospora feedback loop (Dunlap, 1999). Therefore, while some of the exact constituents of the positive and negative complexes are not orthologous proteins, the functional logic of the clock architecture is conserved.
While the logic is conserved, it is interesting to see the degree of variation among the components. Mammals use PER proteins in the negative complex, but the principal repressors in this complex are the CRY proteins that, in Drosophila, serve only as photoreceptors with no role in the oscillator itself (Dunlap, 1999). Among the fungi, WC-1 and WC-2 are broadly and highly conserved in the fungi, but FRQ is much less conserved (Dunlap & Loros, 2006, Salichos & Rokas, 2010) so it is likely that other protein(s) assume the role of a negative element. However the conserved nature of the regulatory logic suggests that the negative element(s) will work in a manner similar to that of FRQ and PER.
Perspectives
The rich history of genetics (Perkins et al., 2001), standardization of mutant selection (Beadle & Tatum, 1941), well characterized growth and reproduction cycle (Davis, 2000), and centralized strain collection (FGSC, Kansas City, Kansas) provide reasons for why after 70 years researchers continue to turn to Neurospora crassa to help answer new questions in eukaryotic biology. Further, an array of molecular techniques such as targeted homologous recombination (Ninomiya et al., 2004), a fully sequenced haploid genome (Borkovich et al., 2004), and a knock-out (KO) collection approaching nearly all ~10,000 open reading frames (Colot et al., 2006) contribute to make Neurospora crassa and ideal platform for the study of numerous biological functions. Recently developed tools include methods for whole proteome labeling for quantitative mass-spectrometry (Huang et al., 2007, Baker et al., 2009), a high density SNP map (Lambreghts et al., 2009), real time luciferase-based reporters (Gooch et al., 2008), and methods for functional genomics (Honda & Selker, 2009, Larrondo et al., 2009). Together these research tools help continue to keep Neurospora a premiere fungal model.
While many molecular players in the clock have been characterized, the full list of proteins, genes, and other control mechanisms are far from complete. Furthermore the only partly conserved nature of biological clock proteins within fungi (Dunlap & Loros, 2006, Salichos & Rokas, 2010), including many plant and animal pathogens, leave many unexplored avenues for research into the potential roles of clock regulated biology in various ecological niches. New methods in full genome and proteome dissection will open our understanding to the systems level and potentially offer clock biologists exciting new insights. Certainly experiments utilizing Neurospora crassa paved the way for our current understanding, and going forward will continue to reveal new answers into this fascinating example of biological organization.
Acknowledgments
This work was supported by grants from the National Institutes of Health to J.C.D. (GM34985 and GM68087) to J.J.L. and J.C.D (GM083336). An NIH predoctoral training grant (NIGMS 5T32GM008704) supported C.L.B.
References
- Allada R, White NE, So WV, Hall JC, Rosbash M. A mutant Drosophila homolog of mammalian Clock disrupts circadian rhythms and transcription of period and timeless. Cell. 1998;93:791–804. doi: 10.1016/s0092-8674(00)81440-3. [DOI] [PubMed] [Google Scholar]
- Aronson BD, Johnson KA, Loros JJ, Dunlap JC. Negative feedback defining a circadian clock: autoregulation of the clock gene frequency. Science. 1994;263:1578–1584. doi: 10.1126/science.8128244. [DOI] [PubMed] [Google Scholar]
- Bae K, Jin X, Maywood ES, Hastings MH, Reppert SM, Weaver DR. Differential functions of mPer1, mPer2, and mPer3 in the SCN circadian clock. Neuron. 2001;30:525–536. doi: 10.1016/s0896-6273(01)00302-6. [DOI] [PubMed] [Google Scholar]
- Baker CL, Kettenbach AN, Loros JJ, Gerber SA, Dunlap JC. Quantitative proteomics reveals a dynamic interactome and phase-specific phosphorylation in the Neurospora circadian clock. Mol Cell. 2009;34:354–363. doi: 10.1016/j.molcel.2009.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballario P, Talora C, Galli D, Linden H, Macino G. Roles in dimerization and blue light photoresponse of the PAS and LOV domains of Neurospora crassa white collar proteins. Mol Microbiol. 1998;29:719–729. doi: 10.1046/j.1365-2958.1998.00955.x. [DOI] [PubMed] [Google Scholar]
- Ballario P, Vittorioso P, Magrelli A, Talora C, Cabibbo A, Macino G. White collar-1, a central regulator of blue light responses in Neurospora, is a zinc finger protein. EMBO J. 1996;15:1650–1657. [PMC free article] [PubMed] [Google Scholar]
- Bargiello TA, Young MW. Molecular genetics of a biological clock in Drosophila. Proc Natl Acad Sci U S A. 1984;81:2142–2146. doi: 10.1073/pnas.81.7.2142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beadle GW, Tatum EL. Genetic Control of Biochemical Reactions in Neurospora. Proc Natl Acad Sci U S A. 1941;27:499–506. doi: 10.1073/pnas.27.11.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belden WJ, Loros JJ, Dunlap JC. Execution of the circadian negative feedback loop in Neurospora requires the ATP-dependent chromatin-remodeling enzyme CLOCKSWITCH. Mol Cell. 2007;25:587–600. doi: 10.1016/j.molcel.2007.01.010. [DOI] [PubMed] [Google Scholar]
- Borkovich KA, Alex LA, Yarden O, et al. Lessons from the genome sequence of Neurospora crassa: tracing the path from genomic blueprint to multicellular organism. Microbiol Mol Biol Rev. 2004;68:1–108. doi: 10.1128/MMBR.68.1.1-108.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunner M, Kaldi K. Interlocked feedback loops of the circadian clock of Neurospora crassa. Mol Microbiol. 2008;68:255–262. doi: 10.1111/j.1365-2958.2008.06148.x. [DOI] [PubMed] [Google Scholar]
- Castro-Longoria E, Ferry M, Bartnicki-Garcia S, Hasty J, Brody S. Circadian rhythms in Neurospora crassa: dynamics of the clock component frequency visualized using a fluorescent reporter. Fungal Genet Biol. 2010;47:332–341. doi: 10.1016/j.fgb.2009.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cha J, Chang SS, Huang G, Cheng P, Liu Y. Control of WHITE COLLAR localization by phosphorylation is a critical step in the circadian negative feedback process. EMBO J. 2008;27:3246–3255. doi: 10.1038/emboj.2008.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen CH, Dunlap JC, Loros JJ. Neurospora illuminates fungal photoreception. Fungal Genet Biol. 2010;47:922–929. doi: 10.1016/j.fgb.2010.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen CH, Ringelberg CS, Gross RH, Dunlap JC, Loros JJ. Genome-wide analysis of light-inducible responses reveals hierarchical light signalling in Neurospora. EMBO J. 2009;28:1029–1042. doi: 10.1038/emboj.2009.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen CH, Demay BS, Gladfelter AS, Dunlap JC, Loros JJ. Physical interaction between VIVID and white collar complex regulates photoadaptation in Neurospora. Proc Natl Acad Sci U S A. 2010;107:16715–16720. doi: 10.1073/pnas.1011190107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng P, Yang Y, Liu Y. Interlocked feedback loops contribute to the robustness of the Neurospora circadian clock. Proc Natl Acad Sci U S A. 2001;98:7408–7413. doi: 10.1073/pnas.121170298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng P, Yang Y, Heintzen C, Liu Y. Coiled-coil domain-mediated FRQ-FRQ interaction is essential for its circadian clock function in Neurospora. EMBO J. 2001;20:101–108. doi: 10.1093/emboj/20.1.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng P, Yang Y, Gardner KH, Liu Y. PAS domain-mediated WC-1/WC-2 interaction is essential for maintaining the steady-state level of WC-1 and the function of both proteins in circadian clock and light responses of Neurospora. Mol Cell Biol. 2002;22:517–524. doi: 10.1128/MCB.22.2.517-524.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng P, He Q, Wang L, Liu Y. Regulation of the Neurospora circadian clock by an RNA helicase. Genes Dev. 2005;19:234–241. doi: 10.1101/gad.1266805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colot HV, Loros JJ, Dunlap JC. Temperature-modulated alternative splicing and promoter use in the circadian clock gene frequency. Mol Biol Cell. 2005;16:5563–5571. doi: 10.1091/mbc.E05-08-0756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colot HV, Park G, Turner GE, et al. A high-throughput gene knockout procedure for Neurospora reveals functions for multiple transcription factors. Proc Natl Acad Sci U S A. 2006;103:10352–10357. doi: 10.1073/pnas.0601456103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crosthwaite SK, Dunlap JC, Loros JJ. Neurospora wc-1 and wc-2: transcription, photoresponses, and the origins of circadian rhythmicity. Science. 1997;276:763–769. doi: 10.1126/science.276.5313.763. [DOI] [PubMed] [Google Scholar]
- Davis RH. Neurospora: Contributions of a Model Organism. Oxford: University Press; 2000. [Google Scholar]
- de Paula RM, Vitalini MW, Gomer RH, Bell-Pedersen D. Complexity of the Neurospora crassa circadian clock system: multiple loops and oscillators. Cold Spring Harb Symp Quant Biol. 2007;72:345–351. doi: 10.1101/sqb.2007.72.002. [DOI] [PubMed] [Google Scholar]
- de Paula RM, Lewis ZA, Greene AV, et al. Two circadian timing circuits in Neurospora crassa cells share components and regulate distinct rhythmic processes. J Biol Rhythms. 2006;21:159–168. doi: 10.1177/0748730406288338. [DOI] [PubMed] [Google Scholar]
- Denault DL, Loros JJ, Dunlap JC. WC-2 mediates WC-1-FRQ interaction within the PAS protein-linked circadian feedback loop of Neurospora. EMBO J. 2001;20:109–117. doi: 10.1093/emboj/20.1.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diernfellner A, Colot HV, Dintsis O, Loros JJ, Dunlap JC, Brunner M. Long and short isoforms of Neurospora clock protein FRQ support temperature-compensated circadian rhythms. FEBS Lett. 2007;581:5759–5764. doi: 10.1016/j.febslet.2007.11.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diernfellner AC, Querfurth C, Salazar C, Hofer T, Brunner M. Phosphorylation modulates rapid nucleocytoplasmic shuttling and cytoplasmic accumulation of Neurospora clock protein FRQ on a circadian time scale. Genes Dev. 2009;23:2192–2200. doi: 10.1101/gad.538209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunlap JC. Molecular bases for circadian clocks. Cell. 1999;96:271–290. doi: 10.1016/s0092-8674(00)80566-8. [DOI] [PubMed] [Google Scholar]
- Dunlap JC. Salad days in the rhythms trade. Genetics. 2008;178:1–13. doi: 10.1534/genetics.104.86496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunlap JC, Loros JJ. Neurospora Photoreceptors. Wiley-VCH Verlag GmbH & Co. KGaA; 2005. [Google Scholar]
- Dunlap JC, Loros JJ. How fungi keep time: circadian system in Neurospora and other fungi. Curr Opin Microbiol. 2006;9:579–587. doi: 10.1016/j.mib.2006.10.008. [DOI] [PubMed] [Google Scholar]
- Dunlap JC, Loros JJ, Colot HV, et al. A circadian clock in Neurospora: how genes and proteins cooperate to produce a sustained, entrainable, and compensated biological oscillator with a period of about a day. Cold Spring Harb Symp Quant Biol. 2007;72:57–68. doi: 10.1101/sqb.2007.72.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edery I, Zwiebel LJ, Dembinska ME, Rosbash M. Temporal phosphorylation of the Drosophila period protein. Proc Natl Acad Sci U S A. 1994;91:2260–2264. doi: 10.1073/pnas.91.6.2260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elvin M, Loros JJ, Dunlap JC, Heintzen C. The PAS/LOV protein VIVID supports a rapidly dampened daytime oscillator that facilitates entrainment of the Neurospora circadian clock. Genes Dev. 2005;19:2593–2605. doi: 10.1101/gad.349305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldman JF, Hoyle MN. Isolation of circadian clock mutants of Neurospora crassa. Genetics. 1973;75:605–613. doi: 10.1093/genetics/75.4.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Froehlich AC, Liu Y, Loros JJ, Dunlap JC. White Collar-1, a circadian blue light photoreceptor, binding to the frequency promoter. Science. 2002;297:815–819. doi: 10.1126/science.1073681. [DOI] [PubMed] [Google Scholar]
- Gallego M, Virshup DM. Protein serine/threonine phosphatases: life, death, and sleeping. Curr Opin Cell Biol. 2005;17:197–202. doi: 10.1016/j.ceb.2005.01.002. [DOI] [PubMed] [Google Scholar]
- Gallego M, Virshup DM. Post-translational modifications regulate the ticking of the circadian clock. Nat Rev Mol Cell Biol. 2007;8:139–148. doi: 10.1038/nrm2106. [DOI] [PubMed] [Google Scholar]
- Garceau NY, Liu Y, Loros JJ, Dunlap JC. Alternative initiation of translation and time-specific phosphorylation yield multiple forms of the essential clock protein FREQUENCY. Cell. 1997;89:469–476. doi: 10.1016/s0092-8674(00)80227-5. [DOI] [PubMed] [Google Scholar]
- Gardner GF, Feldman JF. The frq locus in Neurospora crassa: a key element in circadian clock organization. Genetics. 1980;96:877–886. doi: 10.1093/genetics/96.4.877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner GF, Feldman JF. Temperature Compensation of Circadian Period Length in Clock Mutants of Neurospora crassa. Plant Physiol. 1981;68:1244–1248. doi: 10.1104/pp.68.6.1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gooch VD, Mehra A, Larrondo LF, Fox J, Touroutoutoudis M, Loros JJ, Dunlap JC. Fully codon-optimized luciferase uncovers novel temperature characteristics of the Neurospora clock. Eukaryot Cell. 2008;7:28–37. doi: 10.1128/EC.00257-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorl M, Merrow M, Huttner B, Johnson J, Roenneberg T, Brunner M. A PEST-like element in FREQUENCY determines the length of the circadian period in Neurospora crassa. EMBO J. 2001;20:7074–7084. doi: 10.1093/emboj/20.24.7074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo J, Cheng P, Liu Y. Functional significance of FRH in regulating the phosphorylation and stability of Neurospora circadian clock protein FRQ. J Biol Chem. 2010;285:11508–11515. doi: 10.1074/jbc.M109.071688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo J, Cheng P, Yuan H, Liu Y. The exosome regulates circadian gene expression in a posttranscriptional negative feedback loop. Cell. 2009;138:1236–1246. doi: 10.1016/j.cell.2009.06.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardin PE, Hall JC, Rosbash M. Feedback of the Drosophila period gene product on circadian cycling of its messenger RNA levels. Nature. 1990;343:536–540. doi: 10.1038/343536a0. [DOI] [PubMed] [Google Scholar]
- He Q, Cheng P, Yang Y, Yu H, Liu Y. FWD1-mediated degradation of FREQUENCY in Neurospora establishes a conserved mechanism for circadian clock regulation. EMBO J. 2003;22:4421–4430. doi: 10.1093/emboj/cdg425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He Q, Cha J, Lee HC, Yang Y, Liu Y. CKI and CKII mediate the FREQUENCY-dependent phosphorylation of the WHITE COLLAR complex to close the Neurospora circadian negative feedback loop. Genes Dev. 2006;20:2552–2565. doi: 10.1101/gad.1463506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He Q, Cheng P, Yang Y, Wang L, Gardner KH, Liu Y. White collar-1, a DNA binding transcription factor and a light sensor. Science. 2002;297:840–843. doi: 10.1126/science.1072795. [DOI] [PubMed] [Google Scholar]
- He Q, Shu H, Cheng P, Chen S, Wang L, Liu Y. Light-independent phosphorylation of WHITE COLLAR-1 regulates its function in the Neurospora circadian negative feedback loop. J Biol Chem. 2005;280:17526–17532. doi: 10.1074/jbc.M414010200. [DOI] [PubMed] [Google Scholar]
- Heintzen C, Loros JJ, Dunlap JC. The PAS protein VIVID defines a clock-associated feedback loop that represses light input, modulates gating, and regulates clock resetting. Cell. 2001;104:453–464. doi: 10.1016/s0092-8674(01)00232-x. [DOI] [PubMed] [Google Scholar]
- Hogenesch JB, Gu YZ, Jain S, Bradfield CA. The basic-helix-loop-helix-PAS orphan MOP3 forms transcriptionally active complexes with circadian and hypoxia factors. Proc Natl Acad Sci U S A. 1998;95:5474–5479. doi: 10.1073/pnas.95.10.5474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honda S, Selker EU. Tools for fungal proteomics: multifunctional neurospora vectors for gene replacement, protein expression and protein purification. Genetics. 2009;182:11–23. doi: 10.1534/genetics.108.098707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong CI, Ruoff P, Loros JJ, Dunlap JC. Closing the circadian negative feedback loop: FRQ-dependent clearance of WC-1 from the nucleus. Genes Dev. 2008;22:3196–3204. doi: 10.1101/gad.1706908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang G, Chen S, Li S, et al. Protein kinase A and casein kinases mediate sequential phosphorylation events in the circadian negative feedback loop. Genes Dev. 2007;21:3283–3295. doi: 10.1101/gad.1610207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt SM, Elvin M, Crosthwaite SK, Heintzen C. The PAS/LOV protein VIVID controls temperature compensation of circadian clock phase and development in Neurospora crassa. Genes Dev. 2007;21:1964–1974. doi: 10.1101/gad.437107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt SM, Thompson S, Elvin M, Heintzen C. The role of VIVID in light and temperature responses of the Neurospora circadian system. Proc Natl Acad Sci U S A. 2010;107:16709–16714. doi: 10.1073/pnas.1009474107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jinhu G, Yi L. Molecular mechanism of the Neurospora circadian oscillator. Protein Cell. 2010;1:331–341. doi: 10.1007/s13238-010-0053-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaldi K, Gonzalez BH, Brunner M. Transcriptional regulation of the Neurospora circadian clock gene wc-1 affects the phase of circadian output. EMBO Rep. 2006;7:199–204. doi: 10.1038/sj.embor.7400595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King DP, Zhao Y, Sangoram AM, et al. Positional cloning of the mouse circadian clock gene. Cell. 1997;89:641–653. doi: 10.1016/s0092-8674(00)80245-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konopka RJ, Benzer S. Clock mutants of Drosophila melanogaster. Proc Natl Acad Sci U S A. 1971;68:2112–2116. doi: 10.1073/pnas.68.9.2112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb JS, Zoltowski BD, Pabit SA, Li L, Crane BR, Pollack L. Illuminating solution responses of a LOV domain protein with photocoupled small-angle X-ray scattering. J Mol Biol. 2009;393:909–919. doi: 10.1016/j.jmb.2009.08.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambreghts R, Shi M, Belden WJ, et al. A high-density single nucleotide polymorphism map for Neurospora crassa. Genetics. 2009;181:767–781. doi: 10.1534/genetics.108.089292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larrondo LF, Colot HV, Baker CL, Loros JJ, Dunlap JC. Fungal Functional Genomics: Tunable Knockout-Knockin-expression and tagging strategies. Eukaryot Cell. 2009 doi: 10.1128/EC.00072-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee C, Etchegaray JP, Cagampang FR, Loudon AS, Reppert SM. Posttranslational mechanisms regulate the mammalian circadian clock. Cell. 2001;107:855–867. doi: 10.1016/s0092-8674(01)00610-9. [DOI] [PubMed] [Google Scholar]
- Lee K, Loros JJ, Dunlap JC. Interconnected feedback loops in the Neurospora circadian system. Science. 2000;289:107–110. doi: 10.1126/science.289.5476.107. [DOI] [PubMed] [Google Scholar]
- Lee K, Dunlap JC, Loros JJ. Roles for WHITE COLLAR-1 in circadian and general photoperception in Neurospora crassa. Genetics. 2003;163:103–114. doi: 10.1093/genetics/163.1.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linden H, Macino G. White collar 2, a partner in blue-light signal transduction, controlling expression of light-regulated genes in Neurospora crassa. EMBO J. 1997;16:98–109. doi: 10.1093/emboj/16.1.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Loros J, Dunlap JC. Phosphorylation of the Neurospora clock protein FREQUENCY determines its degradation rate and strongly influences the period length of the circadian clock. Proc Natl Acad Sci U S A. 2000;97:234–239. doi: 10.1073/pnas.97.1.234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Garceau NY, Loros JJ, Dunlap JC. Thermally regulated translational control of FRQ mediates aspects of temperature responses in the Neurospora circadian clock. Cell. 1997;89:477–486. doi: 10.1016/s0092-8674(00)80228-7. [DOI] [PubMed] [Google Scholar]
- Liu Y, Merrow M, Loros JJ, Dunlap JC. How temperature changes reset a circadian oscillator. Science. 1998;281:825–829. doi: 10.1126/science.281.5378.825. [DOI] [PubMed] [Google Scholar]
- Loros JJ, Richman A, Feldman JF. A recessive circadian clock mutation at the frq locus of Neurospora crassa. Genetics. 1986;114:1095–1110. doi: 10.1093/genetics/114.4.1095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loros JJ, Denome SA, Dunlap JC. Molecular cloning of genes under control of the circadian clock in Neurospora. Science. 1989;243:385–388. doi: 10.1126/science.2563175. [DOI] [PubMed] [Google Scholar]
- Loros JJ, Dunlap JC, Larrondo LF, et al. Circadian output, input, and intracellular oscillators: insights into the circadian systems of single cells. Cold Spring Harb Symp Quant Biol. 2007;72:201–214. doi: 10.1101/sqb.2007.72.067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo C, Loros JJ, Dunlap JC. Nuclear localization is required for function of the essential clock protein FRQ. EMBO J. 1998;17:1228–1235. doi: 10.1093/emboj/17.5.1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malzahn E, Ciprianidis S, Káldi K, Schafmeier T, Brunner M. Photoadaptation in Neurospora by competitive interaction of activating and inhibitory LOV domains. Cell. 2010;142:762–772. doi: 10.1016/j.cell.2010.08.010. [DOI] [PubMed] [Google Scholar]
- McClung CR, Fox BA, Dunlap JC. The Neurospora clock gene frequency shares a sequence element with the Drosophila clock gene period. Nature. 1989;339:558–562. doi: 10.1038/339558a0. [DOI] [PubMed] [Google Scholar]
- Mehra A, Baker CL, Loros JJ, Dunlap JC. Post-translational modifications in circadian rhythms. Trends Biochem Sci. 2009;34:483–490. doi: 10.1016/j.tibs.2009.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehra A, Shi M, Baker CL, Colot HV, Loros JJ, Dunlap JC. A role for casein kinase 2 in the mechanism underlying circadian temperature compensation. Cell. 2009;137:749–760. doi: 10.1016/j.cell.2009.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merrow MW, Dunlap JC. Intergeneric complementation of a circadian rhythmicity defect: phylogenetic conservation of structure and function of the clock gene frequency. EMBO J. 1994;13:2257–2266. doi: 10.1002/j.1460-2075.1994.tb06507.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merrow MW, Garceau NY, Dunlap JC. Dissection of a circadian oscillation into discrete domains. Proc Natl Acad Sci U S A. 1997;94:3877–3882. doi: 10.1073/pnas.94.8.3877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan LW, Greene AV, Bell-Pedersen D. Circadian and light-induced expression of luciferase in Neurospora crassa. Fungal Genet Biol. 2003;38:327–332. doi: 10.1016/s1087-1845(02)00562-5. [DOI] [PubMed] [Google Scholar]
- Nakajima M, Imai K, Ito H, et al. Reconstitution of circadian oscillation of cyanobacterial KaiC phosphorylation in vitro. Science. 2005;308:414–415. doi: 10.1126/science.1108451. [DOI] [PubMed] [Google Scholar]
- Neiss A, Schafmeier T, Brunner M. Transcriptional regulation and function of the Neurospora clock gene white collar 2 and its isoforms. EMBO Rep. 2008;9:788–794. doi: 10.1038/embor.2008.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ninomiya Y, Suzuki K, Ishii C, Inoue H. Highly efficient gene replacements in Neurospora strains deficient for nonhomologous end-joining. Proc Natl Acad Sci U S A. 2004;101:12248–12253. doi: 10.1073/pnas.0402780101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perkins DD, Radford A, Sachs MS. The Neurospora Compendium - Chromosomal Loci. Academic Press; 2001. [Google Scholar]
- Pittendrigh CS, Bruce VG. An oscillator model for biological clocks. In: Rudnick D, editor. Rhythmic and Synthetic Processes in Growth. Princeton, New Jersey: Princeton Univ Press; 1957. pp. 75–109. [Google Scholar]
- Pittendrigh CS, Bruce VG, Rosensweig NS, Rubin ML. A Biological Clock in Neurospora. Nature. 1959;184:169–170. [Google Scholar]
- Pregueiro AM, Liu Q, Baker CL, Dunlap JC, Loros JJ. The Neurospora checkpoint kinase 2: a regulatory link between the circadian and cell cycles. Science. 2006;313:644–649. doi: 10.1126/science.1121716. [DOI] [PubMed] [Google Scholar]
- Pregueiro AM, Price-Lloyd N, Bell-Pedersen D, Heintzen C, Loros JJ, Dunlap JC. Assignment of an essential role for the Neurospora frequency gene in circadian entrainment to temperature cycles. Proc Natl Acad Sci U S A. 2005;102:2210–2215. doi: 10.1073/pnas.0406506102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Querfurth C, Diernfellner A, Heise F, et al. Posttranslational regulation of Neurospora circadian clock by CK1a-dependent phosphorylation. Cold Spring Harb Symp Quant Biol. 2007;72:177–183. doi: 10.1101/sqb.2007.72.025. [DOI] [PubMed] [Google Scholar]
- Reddy P, Zehring WA, Wheeler DA, Pirrotta V, Hadfield C, Hall JC, Rosbash M. Molecular analysis of the period locus in Drosophila melanogaster and identification of a transcript involved in biological rhythms. Cell. 1984;38:701–710. doi: 10.1016/0092-8674(84)90265-4. [DOI] [PubMed] [Google Scholar]
- Ruoff P, Loros JJ, Dunlap JC. The relationship between FRQ-protein stability and temperature compensation in the Neurospora circadian clock. Proc Natl Acad Sci U S A. 2005;102:17681–17686. doi: 10.1073/pnas.0505137102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rutila JE, Suri V, Le M, So WV, Rosbash M, Hall JC. CYCLE is a second bHLH-PAS clock protein essential for circadian rhythmicity and transcription of Drosophila period and timeless. Cell. 1998;93:805–814. doi: 10.1016/s0092-8674(00)81441-5. [DOI] [PubMed] [Google Scholar]
- Salichos L, Rokas A. The diversity and evolution of circadian clock proteins in fungi. Mycologia. 2010;102:269–278. doi: 10.3852/09-073. [DOI] [PubMed] [Google Scholar]
- Sancar G, Sancar C, Brunner M, Schafmeier T. Activity of the circadian transcription factor White Collar Complex is modulated by phosphorylation of SP-motifs. FEBS Lett. 2009;583:1833–1840. doi: 10.1016/j.febslet.2009.04.042. [DOI] [PubMed] [Google Scholar]
- Sargent ML, Briggs WR, Woodward DO. Circadian nature of a rhythm expressed by an invertaseless strain of Neurospora crassa. Plant Physiol. 1966;41:1343–1349. doi: 10.1104/pp.41.8.1343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafmeier T, Kaldi K, Diernfellner A, Mohr C, Brunner M. Phosphorylation-dependent maturation of Neurospora circadian clock protein from a nuclear repressor toward a cytoplasmic activator. Genes Dev. 2006;20:297–306. doi: 10.1101/gad.360906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafmeier T, Haase A, Kaldi K, Scholz J, Fuchs M, Brunner M. Transcriptional feedback of Neurospora circadian clock gene by phosphorylation-dependent inactivation of its transcription factor. Cell. 2005;122:235–246. doi: 10.1016/j.cell.2005.05.032. [DOI] [PubMed] [Google Scholar]
- Schafmeier T, Diernfellner A, Schafer A, Dintsis O, Neiss A, Brunner M. Circadian activity and abundance rhythms of the Neurospora clock transcription factor WCC associated with rapid nucleo-cytoplasmic shuttling. Genes Dev. 2008;22:3397–3402. doi: 10.1101/gad.507408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider K, Perrino S, Oelhafen K, Li S, Zatsepin A, Lakin-Thomas P, Brody S. Rhythmic conidiation in constant light in vivid mutants of Neurospora crassa. Genetics. 2009;181:917–931. doi: 10.1534/genetics.108.097808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwerdtfeger C, Linden H. Blue light adaptation and desensitization of light signal transduction in Neurospora crassa. Mol Microbiol. 2001;39:1080–1087. doi: 10.1046/j.1365-2958.2001.02306.x. [DOI] [PubMed] [Google Scholar]
- Schwerdtfeger C, Linden H. VIVID is a flavoprotein and serves as a fungal blue light photoreceptor for photoadaptation. EMBO J. 2003;22:4846–4855. doi: 10.1093/emboj/cdg451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi M, Larrondo LF, Loros JJ, Dunlap JC. A developmental cycle masks output from the circadian oscillator under conditions of choline deficiency in Neurospora. Proc Natl Acad Sci U S A. 2007;104:20102–20107. doi: 10.1073/pnas.0706631104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi M, Collett M, Loros JJ, Dunlap JC. FRQ-interacting RNA helicase mediates negative and positive feedback in the Neurospora circadian clock. Genetics. 2010;184:351–361. doi: 10.1534/genetics.109.111393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shrode LB, Lewis ZA, White LD, Bell-Pedersen D, Ebbole DJ. VVD is required for light adaptation of conidiation-specific genes of Neurospora crassa, but not circadian conidiation. Fungal Genet Biol. 2001;32:169–181. doi: 10.1006/fgbi.2001.1264. [DOI] [PubMed] [Google Scholar]
- Siwicki KK, Eastman C, Petersen G, Rosbash M, Hall JC. Antibodies to the period gene product of Drosophila reveal diverse tissue distribution and rhythmic changes in the visual system. Neuron. 1988;1:141–150. doi: 10.1016/0896-6273(88)90198-5. [DOI] [PubMed] [Google Scholar]
- Smith KM, Sancar G, Dekhang R, et al. Transcription factors in light and circadian clock signaling networks revealed by genomewide mapping of direct targets for Neurospora white collar complex. Eukaryot Cell. 2010;9:1549–1556. doi: 10.1128/EC.00154-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stadler DR. Genetic Control of a Cyclic Growth Pattern in Neurospora. Nature. 1959;184:170–171. [Google Scholar]
- Sussman AS, Durkee TL, Lowry RJ. A model for rhythmic and temperature-independent growth in 'clock' mutants of Neurospora. Mycopathol Mycol Appl. 1965;25:381–396. doi: 10.1007/BF02049924. [DOI] [PubMed] [Google Scholar]
- Talora C, Franchi L, Linden H, Ballario P, Macino G. Role of a white collar-1 white collar-2 complex in blue-light signal transduction. EMBO J. 1999;18:4961–4968. doi: 10.1093/emboj/18.18.4961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang CT, Li S, Long C, et al. Setting the pace of the Neurospora circadian clock by multiple independent FRQ phosphorylation events. Proc Natl Acad Sci U S A. 2009;106:10722–10727. doi: 10.1073/pnas.0904898106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tansey WP. Transcriptional activation: risky business. Genes Dev. 2001;15:1045–1050. doi: 10.1101/gad.896501. [DOI] [PubMed] [Google Scholar]
- Tatum EL. Contributions of Bernard O. Dodge to Biochemical Genetics. Bulletin of the Torrey Botanical Club. 1961;88:115–118. [Google Scholar]
- Tei H, Okamura H, Shigeyoshi Y, Fukuhara C, Ozawa R, Hirose M, Sakaki Y. Circadian oscillation of a mammalian homologue of the Drosophila period gene. Nature. 1997;389:512–516. doi: 10.1038/39086. [DOI] [PubMed] [Google Scholar]
- Vitalini MW, Morgan LW, March IJ, Bell-Pedersen D. A genetic selection for circadian output pathway mutations in Neurospora crassa. Genetics. 2004;167:119–129. doi: 10.1534/genetics.167.1.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitalini MW, de Paula RM, Park WD, Bell-Pedersen D. The rhythms of life: circadian output pathways in Neurospora. J Biol Rhythms. 2006;21:432–444. doi: 10.1177/0748730406294396. [DOI] [PubMed] [Google Scholar]
- Vitalini MW, de Paula RM, Goldsmith CS, Jones CA, Borkovich KA, Bell-Pedersen D. Circadian rhythmicity mediated by temporal regulation of the activity of p38 MAPK. Proc Natl Acad Sci U S A. 2007;104:18223–18228. doi: 10.1073/pnas.0704900104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, Cheng P, Liu Y. Regulation of the Neurospora circadian clock by casein kinase II. Genes Dev. 2002;16:994–1006. doi: 10.1101/gad.965102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, Cheng P, Zhi G, Liu Y. Identification of a Calcium/Calmodulin-dependent protein kinase that phosphorylates the Neurospora circadian clock protein FREQUENCY. J Biol Chem. 2001;276:41064–41072. doi: 10.1074/jbc.M106905200. [DOI] [PubMed] [Google Scholar]
- Yang Y, Cheng P, He Q, Wang L, Liu Y. Phosphorylation of FREQUENCY protein by casein kinase II is necessary for the function of the Neurospora circadian clock. Mol Cell Biol. 2003;23:6221–6228. doi: 10.1128/MCB.23.17.6221-6228.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, He Q, Cheng P, Wrage P, Yarden O, Liu Y. Distinct roles for PP1 and PP2A in the Neurospora circadian clock. Genes Dev. 2004;18:255–260. doi: 10.1101/gad.1152604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zoltowski BD, Crane BR. Light activation of the LOV protein vivid generates a rapidly exchanging dimer. Biochemistry. 2008;47:7012–7019. doi: 10.1021/bi8007017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zoltowski BD, Schwerdtfeger C, Widom J, Loros JJ, Bilwes AM, Dunlap JC, Crane BR. Conformational switching in the fungal light sensor Vivid. Science. 2007;316:1054–1057. doi: 10.1126/science.1137128. [DOI] [PMC free article] [PubMed] [Google Scholar]