Abstract
Background. Pro-inflammatory monocytes/macrophages may contribute to increased atherosclerosis in human immunodeficiency virus (HIV)–infected patients. We investigate—to our knowledge, for the first time—sCD163 and other markers of monocyte activation in relationship to atherosclerotic plaque in HIV-infected patients.
Methods. One hundred two HIV-infected and 41 HIV-seronegative men with equivalent cardiovascular risk factors and without history of coronary artery disease were prospectively recruited and underwent computed tomography coronary angiography.
Results. sCD163 levels and presence of plaque were significantly higher among antiretroviral-treated subjects with undetectable HIV RNA levels, compared with seronegative controls (1172 ± 646 vs. 883 ± 561 ng/mL [P = .02] for sCD163 and 61% vs. 39% [P = .03] for presence of plaque). After adjusting for age, race, lipids, blood pressure, glucose, smoking, sCD14, and HIV infection, sCD163 remained independently associated with noncalcified plaque (P = .008). Among HIV-infected patients, sCD163 was associated with coronary segments with noncalcified plaque (r = 0.21; P = .04), but not with calcium score. In contrast, markers of generalized inflammation, including C-reactive protein level, and D-dimer were not associated with sCD163 or plaque among HIV-infected patients.
Conclusions. sCD163, a monocyte/macrophage activation marker, is increased in association with noncalcified coronary plaque in men with chronic HIV infection and low or undetectable viremia. These data suggest a potentially important role of chronic monocyte/macrophage activation in the development of noncalcified vulnerable plaque.
Clinical Trial Registration. NCT00455793.
We previously observed that patients with human immunodeficiency virus (HIV) infection have a higher prevalence of subclinical coronary atherosclerosis and greater burden of coronary atherosclerotic plaque, particularly noncalcified plaque, than HIV-seronegative subjects with similar cardiovascular risk factors, even if they are young, have a low Framingham risk score, and are free of symptoms of cardiovascular disease [1]. The plaque characteristics in this young, asymptomatic HIV-infected patient population were more likely to be noncalcified rather than calcified plaque lesions. Plaques with necrotic lipid rich cores composed of pro-inflammatory immune cells are more vulnerable to rupture and are often noncalcified, with low attenuation on computerized tomography (CT) [2, 3].
Pro-inflammatory monocyte and macrophage subsets are an integral component of the atherosclerotic process and have been shown to migrate to atherosclerotic plaque [4].
Hypothesizing that activation of the innate immune system contributes to cardiovascular disease (CVD), we investigated markers of macrophage activation in association with atherosclerotic disease among HIV-infected patients, with a particular focus on those with undetectable viral loads. CD163, a scavenger receptor specifically expressed on the surface of macrophages and monocytes, is involved in the uptake of hemoglobin-haptoglobin and mediates the clearance of hemoglobin [5]. Soluble CD163 (sCD163) is shed via proteolytic cleavage at the cell surface to a greater extent by activated monocytes/macrophages and is found in plasma. We previously demonstrated that plasma sCD163 levels correlated with monocyte expansion from bone marrow, the rate of AIDS progression, and severity of macrophage-mediated AIDS pathogenesis in simian immunodeficiency virus (SIV)–infected rhesus macaques [6]. sCD163 was also recently shown to be a novel marker of HIV disease activity in early infected and chronically infected patients [7].
Levels of sCD163 are increased in HIV-uninfected patients with angiographically significant coronary artery disease and related to the extent of coronary artery disease [8], but to our knowledge, the relationship of sCD163 to atherosclerosis has not been studied among HIV-infected patients in whom activation of the innate immune system is an important process. In this study, we investigated the relationship of activated monocyte/macrophages, as measured by sCD163 in plasma as well as other markers of monocyte/macrophage activation using flow cytometry, to atherosclerotic plaque and plaque characteristics in HIV-infected patients.
METHODS
Study Participants
One hundred forty-three men participated in the current study. One hundred two men with HIV infection were prospectively recruited from HIV clinics and community health care centers in the Boston area as well as by newspaper advertisements. Forty-one HIV-seronegative control men were simultaneously prospectively recruited from the same communities. Neither group was recruited on the basis of the presence of heart disease, and in fact, history or symptoms of heart disease were similarly exclusionary for both groups. Family members, partners, and friends of the HIV-infected patients were especially encouraged to participate as control subjects to ensure similar characteristics between the 2 groups. Other than HIV disease, inclusion and exclusion criteria were identical for both groups. Participants aged 18–58 years without known cardiac disease or symptoms suggestive of cardiac disease (any current or prior heart disease, including angina, arrhythmias, valvular heart disease, pericarditis, congestive heart failure, or any prior treatment for coronary artery disease [CAD] or any heart disease) were recruited. Participants underwent cardiac CT only as part of the study protocol and not for clinical indications. HIV-infected and control participants were not recruited with regard to any changes in body composition, weight, or metabolic criteria. HIV-infected and control participants with known renal disease or creatinine levels >1.5 mg/dL or estimated creatinine clearance <70 mL/min were excluded to minimize risk of contrast nephropathy. HIV-infected patients receiving combination antiretroviral therapy (ART) at the time of the study were required to have been receiving stable therapy for >3 months. All participants provided informed consent. This study was approved by the institutional review boards of Massachusetts General Hospital and Massachusetts Institute of Technology. Cardiac CT data were previously published in a subset of the patients [1]. We have continued to expand this cohort and now report novel data not previously published on monocyte activation using sCD163, flow cytometry, and other markers, including LPS, sCD14, osteopontin, hsIL-6, and D-dimer.
Study Procedures and Assessment of Cardiovascular Risk Factors
Data on sociodemographic factors, cardiovascular risk factors, medical history, family history, behavior (including smoking), use of medications, and prior nadir CD4 cell count were obtained. For diabetes mellitus, both clinical history and prevalence based on fasting glucose were determined. All participants fasted at least 12 hours before blood draws. The Framingham risk score was calculated [9]. Cardiac multidetector row CT and CT angiography imaging were performed using a 64-slice CT scanner (Sensation 64; Siemens Medical Solutions). For full description, see Lo et al [1]. Assessment of coronary atherosclerotic plaque burden and stenosis were determined by a consensus reading between 2 investigators, including a cardiologist and a radiologist with significant experience in the interpretation of cardiac CTs. The presence of any coronary atherosclerotic plaque, whether calcified or noncalcified, was determined. Discrete coronary segments affected by plaque were counted, and these segments were characterized as to whether they were calcified or noncalcified. The number of segments with noncalcified or calcified plaque per patient was determined. The percentage of noncalcified plaque was also determined by dividing the number of noncalcified segments by the total number of segments affected with any plaque per subject. Only those subjects with plaque could be included in this analysis. Presence of severe coronary artery stenosis was defined as luminal obstruction >70% diameter in any coronary segment. Physicians analyzing the scans were blinded to the participants’ clinical history or HIV status. Agatston calcium score was calculated using the noncontrast CT images using standardized techniques [10].
Inflammatory, Metabolic, Biochemical, and Immunologic Parameters
Plasma sCD163, MCP-1, interleukin (IL)–6, sCD14, and osteopontin levels were quantified by enzyme-linked immunosorbent assay (ELISA) in accordance with the manufacturer's protocol (Trillium Diagnostics [sCD163], R&D Systems [high sensitive IL-6, MCP-1, and sCD14], and IBL-America (osteopontin). The endpoint LAL assay (Associates of Cape Cod) was used to measure lipopolysaccharide levels, as previously described [6]. Total cholesterol, high- and low-density lipoprotein, triglycerides, glucose, and creatinine levels were determined using standard techniques. C-reactive protein (CRP) levels were measured using ELISA. Quantitative determination of D-Dimer in plasma was performed by immuno-turbidometric method. CD4+ and CD8+ T cell counts were assessed by flow cytometry. HIV RNA levels were determined by ultrasensitive reverse-transcription polymerase chain reaction (RT-PCR; Roche Amplicor Monitor; lower limit of detection, 50 copies/mL). HIV testing was performed by ELISA (Abbott), and results were confirmed by western blot. Serum immunoglobulin (Ig) G antibodies to cytomegalovirus (CMV) were measured using ELISA (Wampole Laboratories) in the Massachusetts General Hospital clinical microbiology laboratory. The CMV ELISA antibody measurement is an ELISA index value based on optical density (OD) ratio, not a titer. The OD ratio is calculated by dividing the raw OD value by the cutoff OD. The cutoff OD is determined by multiplying the kit-specific correction factor, provided by the manufacturer, by the mean OD of the calibrator, which is run in triplicate along with a positive and negative control each time the assay is run. EBV viral capsid antigen IgG titers were measured by indirect fluorescent antibody and image analysis (Quest Diagnostics Nichols Institute of Valencia, CA).
Flow Cytometry
Peripheral blood samples were collected in EDTA and prepared by standard methods and techniques previously established in the Flow Cytometry Laboratory at Massachusetts General Hospital [11, 12]. Eight fluorochrome-conjugated antibodies were combined with 100 μL of whole blood, incubated for 15 min, then washed and fixed with 2 mL of 1X BD FACSM Lysing solution (Beckton Dickinson) followed by 500 μL 1% paraformaldehyde (Sigma). Samples were processed on a FACSCanto II (Beckton Dickinson), and 50000 events were collected and analyzed using FACSDiva software. Data analysis was performed using FACSDiva software with side light scatter versus CD14 gating used to identify monocytes and forward-versus-side light scatter gating used to identify lymphocytes. T lymphocytes were identified using populations of brightly stained CD4+ or CD8+ cells. HLA-DR and CD38 expression on CD4+ or CD8+ T cells was assessed by using negative lymphocyte populations to define negative controls.
Statistical Analysis
Data are presented as mean ± standard deviation or median (interquartile range), depending on normality of distribution. Comparisons between 2 groups were performed using the Student t test for normally distributed continuous variables and Wilcoxon rank sum test if the distribution was nonnormal. Pearson correlation coefficients were used to assess correlations for normally distributed data. For nonnormally distributed end points, data were either log-transformed or Spearman rho was used to assess correlation. Due to values of zero, log transformation of (calcium score + 1) and (number of segments with noncalcified plaque +1) were performed. Linear regression modeling was used in adjusted analyses for continuous outcome variables. Linear regression models were constructed with the natural log of noncalcified segments and the percentage of noncalcified plaque as the dependent variables of interest. Independent variables entered into the model included known traditional cardiovascular risk markers (age, race, smoking, blood pressure, and levels of cholesterol, high-density lipoprotein, triglycerides, and fasting glucose), in addition to markers of immune activation or inflammation that were significantly related on univariate regression analysis, including sCD163 and sCD14. A sensitivity analysis was performed including lipid-lowering therapy in the model, because of the known tendency of lipid-lowering therapy to affect immune activation [13]. Two-tailed probability values are reported, and statistical significance was assumed when P < .05. All statistical analyses were performed using SAS JMP (SAS Institute).
RESULTS
Comparisons of Traditional Risk Factors and Markers of Monocyte/Macrophage Activation, Inflammation, and Atherosclerosis, by HIV and HIV RNA Status
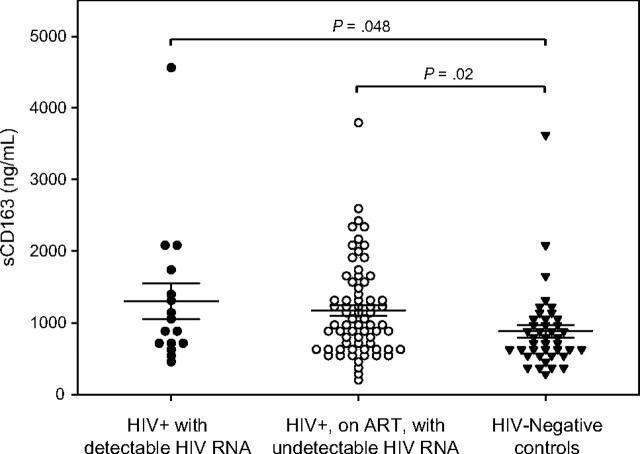
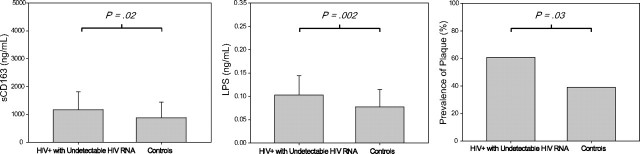
Demographic characteristics, overall Framingham risk score, prevalence of diabetes mellitus, and smoking were similar between HIV-infected subjects and HIV-seronegative controls (Table 1). Use of antihypertensive medications and lipid-lowering therapy were more common among HIV subjects. HIV RNA level ranged from undetectable to 49700 copies/mL. The majority (81%) of HIV-infected patients had undetectable HIV RNA levels, and all but 2 had HIV RNA levels <15000 copies/mL. HIV-infected patients also had an increased prevalence of plaque and a greater number of coronary segments with noncalcified plaque noted by coronary CT angiography, compared with control subjects (Table 1). In contrast, calcium score and number of segments with calcified plaque were similar between the groups. HIV-infected men demonstrated higher sCD163 levels in plasma than did HIV-seronegative men (P = .006) (Table 1). LPS, osteopontin, and hsIL-6 levels in plasma were higher in HIV-infected patients than HIV-seronegative control subjects, whereas C-reactive protein and D-dimer levels did not differ (Table 1). Among the subset of HIV-infected patients treated with ART and with undetectable HIV RNA levels, again presence of plaque, sCD163, LPS, and osteopontin, remained significantly higher in the HIV-infected patients versus HIV-seronegative control subjects (Table 1; Figures 1 and 2), whereas traditional risk factors, including smoking, diabetes, hypertension, and overall Framingham risk score were again similar between the groups.
Table 1.
Demographic and Clinical Characteristics of Study Population, by Human Immunodeficiency Virus (HIV) Status
| Characteristic | HIV-negative controls (n = 41) | All HIV-infected subjects (n = 102) | Pa | HIV+ on ART, with undetectable viral load (n = 69) | Pb |
| Demographic | |||||
| Age, years | 44.6 ± 7.6 | 46.6 ± 6.4 | .10 | 46.8 ± 6.3 | .10 |
| Race | |||||
| White | 61 | 63 | .19 | 67 | .12 |
| Black | 17 | 22 | 19 | ||
| Hispanic | 7 | 12 | 12 | ||
| Asian | 7 | 1 | 0 | ||
| Native American | 5 | 3 | 3 | ||
| Family history of premature CHD by NCEP (%) | 13 | 22 | .24 | 19 | .46 |
| Framingham risk score | 6.2 ± 5.1 | 7.8 ± 5.0 | .08 | 7.6 ± 4.9 | .15 |
| Diabetes mellitus (%) | 2 | 7 | .26 | 7 | .25 |
| Hypertension (%) | 12 | 26 | .06 | 27 | .06 |
| Use of antihypertensive medications (%) | 8 | 29 | .003 | 29 | .004 |
| Current smoker (%) | 29 | 41 | .20 | 40 | .27 |
| Use of lipid-lowering medications (%) | 13 | 27 | .05 | 31 | .03 |
| HIV disease–related parameters | |||||
| Duration since diagnosis of HIV infection, years | N/A | 13.8 ± 6.4 | N/A | 13.6 ± 6.7 | N/A |
| Currently receiving antiretroviral therapy (%) | N/A | 95 | N/A | 100 | N/A |
| Duration of antiretroviral therapy, years | N/A | 7.2 ± 5.0 | N/A | 7.8 ± 4.6 | N/A |
| Current PI treatment (%) | N/A | 52 | N/A | 49 | N/A |
| Current NRTI treatment (%) | N/A | 92 | N/A | 97 | N/A |
| Current NNRTI treatment (%) | N/A | 47 | N/A | 54 | N/A |
| Current CD4+ T lymphocyte count, cells/mm3 | N/A | 530 ± 287 | N/A | 544 ± 288 | N/A |
| Nadir CD4+ T lymphocyte count, cells/mm3 | N/A | 202 ± 173 | N/A | 203 ± 175 | N/A |
| HIV RNA level, copies/mL | N/A | <50 (<50 to <50) | N/A | <50 (<50 to <50) | N/A |
| Undetectable HIV RNA level (<50 copies/mL) (%) | N/A | 81 | N/A | 100 | N/A |
| Traditional cardiovascular risk factors | |||||
| Body mass index, kg/m2 | 26.9 ± 4.9 | 26.3 ± 4.7 | .51 | 26.5 ± 4.3 | .70 |
| Systolic blood pressure, mm Hg | 117 ± 12 | 120 ± 12 | .23 | 121 ± 11 | .14 |
| Diastolic blood pressure, mm Hg | 76 ± 9 | 77 ± 9 | .59 | 77 ± 9 | .39 |
| Fasting glucose level, mg/dL | 92 ± 10 | 92 ± 12 | .89 | 93 ± 11 | .75 |
| Total cholesterol level, mg/dL | 177 ± 40 | 180 ± 41 | .77 | 182 ± 42 | .59 |
| HDL cholesterol level, mg/dL | 48 ± 12 | 48 ± 15 | .97 | 49 ± 15 | .62 |
| LDL colesterol, mg/dL | 110 ± 33 | 101 ± 31 | .14 | 101 ± 30 | .17 |
| Triglyceride leve, mg/dL | 100 ± 58 | 155 ± 129 | .009 | 158 ± 142 | .01 |
| Creatinine level, mg/dL | 1.07 ± 0.15 | 1.04 ± 0.19 | .35 | 1.02 ± 0.19 | .13 |
| Markers of monocyte/macrophage activation | |||||
| sCD163, ng/mL | 883 ± 561 | 1232 ± 716 | .006 | 1172 ± 646 | .02 |
| LPS, ng/mL | 0.08 ± 0.04 | 0.10 ± 0.04 | .001 | 0.10 ± 0.04 | .002 |
| sCD14, ng/mL | 211 (121–374) | 305 (157–440) | .08 | 296 (156–449) | .11 |
| Osteopontin, ng/mL | 328 (269–455) | 482 (348–616) | .0002 | 464 (358–674) | .001 |
| MCP-1, pg/mL | 235 (190–299) | 275 (179–363) | .13 | 281 (197–366) | .06 |
| Markers of generalized inflammation, and hemostasis | |||||
| hs interleukin-6, pg/mL | 0.6 (0.5–1.0) | 0.9 (0.7–1.5) | .01 | 0.9 (0.6–1.3) | .05 |
| C-reactive protein, mg/L | 1.3 (0.7–3.3) | 1.6 (0.7–3.8) | .49 | 1.5 (0.7–3.8) | .66 |
| D-dimer, ng/mL | <220 (<220 to 333) | <220 (<220 to 322) | .93 | <220 (<220 to 333) | .81 |
| Cytomegalovirus IgG titers | 36 (3–68) | 93 (65–194) | <.0001 | 86 (64–194) | <.0001 |
| Epstein-Barr virus viral capsid antigen IgG titers | 160 (40–160) | 160 (160–160) | .02 | 160 (160–160) | .08 |
| Plaque characteristics | |||||
| Presence of plaque, % | 39 | 60 | .02 | 61 | .03 |
| Agatston calcium score | 0 (0–9.9) | 0 (0–17.9) | .29 | 0 (0–22.5) | .19 |
| No. of segments with noncalcified plaque | 0 (0–1);0.46 ± 0.98 | 0 (0–1);0.99 ± 1.57 | .049 | 0 (0–1); 0.84 ± 1.39 | .12 |
| No. of segments with calcified plaque | 0 (0–0);0.29 ± 0.81 | 0 (0–0);0.26 ± 0.77 | .87 | 0 (0–0); 0.25 ± 0.76 | .82 |
| Participants with coronary stenosis >70% (%) | 0 | 5 | .07 | 3 | .17 |
Data are percentage of subjects, mean ± standard deviation, or median (interquartile range).
Abbreviations: CHD, coronary heart disease; HDL, high-density lipoprotein; Ig, immunoglobulin, LDL, low-density lipoprotein; N/A, not available; NCEP, National Cholesterol Education Program; NNRTI, nonnucleoside reverse-transcriptase inhibitor; NRTI, nucleoside/nucleotide reverse-transcriptase inhibitor; PI, protease inhibitor.
HIV-infected subjects versus control subjects.
HIV-infected subjects who are receiving antiretroviral therapy who have undetectable viral loads versus control subjects.
Figure 1.
sCD163 levels in human immunodeficiency virus (HIV)–patients with or without detectable HIV RNA levels versus HIV-seronegative controls. Results are mean values ± standard errors of the mean.
Figure 2.
Comparison of sCD163, lipopolysaccharide (LPS), and prevalence of coronary plaque in human immunodeficiency virus (HIV)–seronegative controls versus HIV-infected patients receiving antiretroviral therapy who have viral suppression. Results are mean values ± standard deviations.
Relationship of sCD163 to Markers of HIV Disease Progression, Monocyte/Macrophage Activation, Generalized Inflammation, Immune Parameters, and Plaque Characteristics Among HIV-Infected Patients
sCD163 was significantly associated with noncalcified plaque (r = 0.21; P = .04) but not with calcium score (r = 0.06, P = .52) (Table 2). sCD163 was associated with plasma HIV load (r = 0.22; P = .04) and negatively correlated with CD4+/CD8+ ratio (r =−0.22; P = .02) among all HIV-infected patients (Table 2). sCD163 was associated with neither nadir CD4 cell count (r = −0.13; P = .24) nor current CD4 cell count (r = −0.12; P = .23). sCD163 also correlated with LPS (r = 0.27; P = .007), sCD14 (ρ = 0.22; P = .03), hsIL-6 (ρ = 0.27, P = .01), and CMV IgG titers (ρ = 0.27, P = .01). sCD163 was not associated with EBV viral capsid IgG titers (ρ = 0.08, P = .41). Furthermore, sCD163 was significantly associated with percentago of CD14+CD16+ monocytes (r = 0.35, P = .04) and with percentage of HLA-DR+ CD4+ T cells (r = 0.45, P = .006) determined by flow cytometry. In contrast to sCD163, sCD14 did not correlate with LPS (ρ = 0.02, P = .87).
Table 2.
Relationships to sCD163 (Pearson and Spearman Correlation Coefficients)
| Characteristic | All patients (n = 143) | All HIV-infected patients (n = 102) | HIV-infected patients with undetectable viral load (n = 69) |
| Traditional risk factors | |||
| Age | r = 0.15, P =.08 | r = 0.16, P =.11 | r = 0.18, P =.14 |
| Systolic blood pressure | r = 0.09, P =.29 | r = 0.05, P =.60 | r = 0.10, P =.43 |
| Diastolic blood pressure | r = 0.05, P =.54 | r = −0.009, P =.93 | r = 0.06, P =.62 |
| Total cholesterol | r = −0.11, P =.20 | r = −0.14, P =.16 | r = −0.007, P =.95 |
| Low-density lipoprotein cholesterol | r = −0.13, P =.13 | r = −0.13, P =.20 | r = −0.02, P =.90 |
| Triglycerides | r = 0.17, P =.045 | r = 0.15, P =.14 | r = 0.13, P =.28 |
| Measurements of coronary atherosclerosis | |||
| Natural log segments with noncalcified plaque | r = 0.22, P =.01 | r = 0.21, P =.04 | r = 0.24, P =.046 |
| Natural log calcium score | r = −0.04, P =.62 | r = −0.06, P =.52 | r = 0.05, P =.67 |
| Monocyte/macrophage activation markers | |||
| Lipopolysaccharide | r = 0.28, P =.0007 | r = 0.27, P =.007 | r = 0.21, P =.08 |
| sCD14 | ρ = 0.06, P =.46 | ρ = 0.22, P =.03 | ρ = 0.14, P =.28 |
| Osteopontin | ρ = 0.16, P =.06 | ρ = 0.03, P =.75 | ρ = 0.17, P =.15 |
| Percentage of CD14+CD16+ monocytes | r = 0.20, P =.14 | r = 0.35, P =.04 | r = 0.29, P =.13 |
| Markers of generalized inflammation, coagulation, and cytomegalovirus | |||
| hs Interleukin-6 | ρ = 0.26, P =.005 | ρ = 0.27, P =.01 | ρ = 0.32, P =.01 |
| C-reactive protein | ρ = −0.04, P =.59 | ρ = −0.03, P =.75 | ρ = 0.13, P =.27 |
| D-dimer | ρ = 0.09, P =.29 | ρ = 0.13, P =.18 | ρ = 0.16, P =.19 |
| Cytomegalovirus immunoglobulin G titers | ρ = 0.30, P =.001 | ρ = 0.27, P =.01 | ρ = 0.23, P =.10 |
| Epstein-Barr virus viral capsid antigen immunoglobulin G titers | ρ = 0.07, P =.40 | ρ = 0.08, P =.41 | ρ = 0.08, P =.53 |
| Markers of HIV disease | |||
| Natural log (viral load) | N/A | r = 0.22, P =.04 | N/A |
| Current CD4+ T lymphocyte count | N/A | r = −0.12, P =.23 | r = −0.13, P =.30 |
| Nadir CD4+ T lymphocyte count | N/A | r = −0.13, P =.24 | r = −0.23, P =.08 |
| CD4+/CD8+ ratio | N/A | r = −0.22, P =.02 | r = −0.29, P =.02 |
| Duration of HIV Infection | N/A | r = 0.19, P =.06 | r = 0.23, P =.05 |
Abbreviations: HIV, human immunodeficiency virus; N/A, not available
Among HIV-infected patients receiving ART with undetectable HIV RNA levels, sCD163 was positively associated with number of coronary segments with noncalcified plaque (r = 0.24; P = .046), hsIL-6 (ρ = 0.32; P = .01), and duration of HIV infection (r = 0.23; P = .05) and was negatively associated with CD4+/CD8+ ratio (r = −0.29; P = .02).
Relationship of sCD163 With Noncalcified Plaque in Unadjusted and Adjusted Analyses Among All Patients
Among all patients, sCD163 was significantly associated with total number of noncalcified segments (r = 0.22, P = .01) (Table 2), the percentage of noncalcified plaque (ρ= 0.23; P = .047), but not calcium score (r = −0.04, P = .62) (Table 3). In addition, sCD14 was significantly associated with percentage of noncalcified plaque (ρ= 0.26; P = .03). In contrast, markers of generalized inflammation, such as C-reactive proteine (ρ = 0.14, P = .22) and hsIL-6 (ρ = 0.17, P = .17) level, and D-Dimer (ρ=−0.12, P = .29) were not associated with percentage of noncalcified plaque. In contrast to the association seen between markers of monocyte/macrophage activation and noncalcified plaque, these markers were not significantly related to calcium score. In contrast, traditional cardiovascular markers were more strongly associated with calcium score, as opposed to noncalcified plaque (Table 3).
Table 3.
Relationships to Plaque Characteristics (Pearson and Spearman Correlation Coefficient) in All Participants
| Characteristic | Percentage of noncalcified plaque | Natural log (Ca score +1) |
| Traditional risk factors | ||
| Age | ρ = −0.12, P =.32 | r = 0.36, P < .0001 |
| Framingham risk score | ρ = −0.21, P =.08 | r = 0.37, P < .0001 |
| Systolic blood pressure | ρ = 0.04, P =.73 | r = 0.12, P =.15 |
| Diastolic blood pressure | ρ = 0.19, P =.10 | r = 0.12, P =.15 |
| Total cholesterol | ρ =−0.24, P =.04 | r = 0.32, P < .0001 |
| Low-density lipoprotein cholesterol | ρ =−0.18, P =.14 | r = 0.25, P =.003 |
| High-density lipoprotein cholesterol | ρ =−0.03, P =.78 | r = −0.10, P =.25 |
| Triglycerides | ρ =−0.20, P =.09 | r = 0.33, p < .0001 |
| Monocyte/macrophage activation markers | ||
| sCD163 | ρ =0.23, P =.047 | r = −0.04, P =.62 |
| Lipopolysaccharide | ρ =0.05, P =.68 | r = 0.06, P =.47 |
| sCD14 | ρ =0.26, P =.03 | r = −0.07, P =.39 |
| Osteopontin | ρ =−0.01, P =.94 | r = −0.06, P =.47 |
| Markers of generalized inflammation, coagulation, and cytomegalovirus | ||
| hs interleukin-6 | ρ =0.17, P =.17 | r = 0.03, P =.77 |
| C-reactive protein | ρ =0.14, P =.22 | r = −0.07, P =.37 |
| D-dimer | ρ =−0.12, P =.29 | r = −0.06, P =.46 |
| Cytomegalovirus immunoglobulin G titers | ρ =−0.10, P =.44 | r = 0.16, P =.09 |
In multivariate regression analysis, after adjusting for HIV status and traditional risk factors, as well as for sCD14, sCD163 remained independently associated with the percentage of noncalcified plaque (P = .02) and with number of segments with noncalcified plaque (P = .008) among all subjects (Table 4). Similarly, sCD163 remained independently and significantly associated with percentage of noncalcified plaque (P = .05) and segments with noncalcified segments (P = .048), after controlling for use of lipid-lowering therapy in sensitivity analyses.
Table 4.
Multivariate Models for Relationship of sCD163 With Noncalcified Segments and Percent Noncalcified Plaque in All Participants, Adjusting for Traditional Cardiovascular Disease Risk Factors
| Characteristic | Natural log segments with noncalcified plaque |
Percentage of noncalcified plaque |
||
| β | P | β | P | |
| HIV status | −.12 | .03 | −4.93 | .39 |
| Age, years | .01 | .25 | −.66 | .42 |
| Race | −.14 | .60 | −41.73 | .18 |
| Ever smoked | .03 | .61 | 2.73 | .60 |
| Diastolic blood pressure, mm Hg | .03 | .13 | 1.05 | .03 |
| Fasting glucose level, mg/dL | .02 | .04 | .38 | .25 |
| Total cholesterol, mg/dL | .002 | .16 | −.11 | .49 |
| High-density lipoprotein cholesterol, mg/dL | −.006 | .12 | .23 | .55 |
| Triglycerides, mg/dL | −.001 | .02 | −.06 | .14 |
| sCD14 | .00005 | .67 | .01 | .17 |
| sCD163, ng/mL | .0002 | .008 | .01 | .02 |
Subanalyses performed in HIV-infected and HIV-uninfected subjects demonstrated that sCD163 remained significantly associated with noncalcified plaque segments in HIV (P = .045) (Table 5). No relationship was seen between sCD163 and noncalcified segments in HIV-uninfected patients (P = .89) (Table 6).
Table 5.
Multivariate Models for Relationship of sCD163 With Noncalcified Segments in Human Immunodeficiency Virus (HIV)–Infected Patients
| Natural log segments with noncalcified plaque |
||
| Characteristic | β | P |
| Age, years | .01 | .13 |
| Race | .04 | .94 |
| Ever smoked | .08 | .29 |
| Diastolic blood pressure, mm Hg | .01 | .06 |
| Fasting glucose , mg/dL | .006 | .20 |
| Total cholesterol, mg/dL | .001 | .54 |
| High-density lipoprotein cholesterol, mg/dL | −.007 | .13 |
| Triglycerides, mg/dL | −.001 | .08 |
| sCD14 | .00009 | .49 |
| sCD163, ng/mL | .0002 | .045 |
Table 6.
Multivariate Models for Relationship of sCD163 With Noncalcified Segments in Human Immunodeficiency Virus (HIV)–Seronegative Control Subjects
| Natural log segments with noncalcified plaque |
||
| Characteristic | β | P |
| Age, years | −.0001 | .99 |
| Race | −.38 | .09 |
| Ever smoked | −.18 | .02 |
| Diastolic blood pressure, mm Hg | .02 | .03 |
| Fasting glucose, mg/dL | −.006 | .31 |
| Total cholesterol, mg/dL | .007 | .002 |
| High-density lipoprotein cholesterol, mg/dL | −.008 | .18 |
| Triglycerides, mg/dL | −.005 | .01 |
| sCD14 | −.0004 | .04 |
| sCD163, ng/mL | .00001 | .89 |
DISCUSSION
Data from this study support the hypothesis that monocyte and macrophage activation in HIV disease may contribute to the formation of vulnerable atherosclerotic plaque. Specifically, we demonstrate that the monocyte/macrophage-specific marker, sCD163, is uniquely associated with increased noncalcified plaque among young, asymptomatic, HIV-infected men with low or undetectable levels of viremia and a long duration of HIV disease. sCD163 was significantly associated with increased noncalcified plaque burden, independent of traditional risk factors. These data lend insight into the dynamic process by which cardiovascular disease develops among HIV-infected patients with low or undetectable plasma virus and suggest that macrophage activation, independent of traditional risk factors, contributes to a unique phenotype of inflammatory noncalcified coronary plaque among patients with long-term HIV infection.
In this study, we show that traditional risk factors, including age, Framingham risk score, and cholesterol and low-density lipoprotein levels, were significantly associated with increased calcium score, a measure of calcified plaque burden. In contrast, sCD163 and duration of known HIV infection were positively associated with noncalcified plaque. In addition, sCD14, another marker of monocyte activation, was associated with sCD163 and with noncalcified plaque in univariate analysis. However, sCD163, but not sCD14, remained significantly associated with plaque in the multivariate regression analysis that also controlled for traditional risk factors. This was true in a combined analysis of all patients, controlling for HIV infection, and in the HIV-infected subgroup alone. In contrast, the relationship between sCD163 and noncalcified plaque was not significant in multivariate regression among HIV-uninfected patients.
In contrast to our data on sCD163, markers of generalized inflammation, such as C-reactive protein level, did not show a relationship with plaque in our study, suggesting that, among the large number of patients with controlled viremia, monocyte/macrophage activation—and not generalized inflammation—contributes to increased plaque.
Taken together, these data are significant in that they suggest that specific types of atherosclerotic disease have different etiologies in the HIV-infected population. Those patients with traditional risk factors resulting from dyslipidemia and insulin resistance, hypertension, and other traditional risk factors will be expected to have more calcified lesions. However, we now show that even those with few traditional risk factors can have noncalcified disease and that this may be related to monocyte/macrophage activation, a point not previously appreciated in the HIV-infected population. Plaques with necrotic cores are known to be more prone to rupture, and histologically, increased macrophage infiltration is seen in these necrotic cores [3]. Taken together, our data suggest a potential mechanism by which HIV infection may produce a phenotype of lipid-rich inflamed atherosclerotic plaques potentially via up-regulated monocyte activation that may be more vulnerable to rupture.
Inflammatory and immunologic factors may play important roles contributing to increased coronary artery disease in the HIV population, as suggested by studies like SMART [14, 15]. Triant et al [16] and Lichtenstein et al [17] recently showed, among large cohorts, that acute myocardial infarction rates are related to low CD4 cell count, after controlling for traditional risk factors and ART . In addition, Kaplan et al [18] recently showed that HIV-associated T cell activation is related to subclinical carotid atherosclerosis. The current data suggest—to our knowledge, for the first time—that monocyte/macrophage activation may also contribute to subclinical atherosclerosis.
The current data extend the data from prior studies, which showed endothelial dysfunction by brachial artery flow mediated vasodilation among those with long-standing low viral loads [19]. We now demonstrate that, among patients receiving stable ART who have undetectable viremia, monocyte activation is significant and relates most to noncalcified coronary plaque, the level of which is increased among HIV-infected patients. Atherosclerosis is an inflammatory process in which monocytes and T lymphocytes play important roles [20, 21]. We and others have previously shown that CMV-induced immune responses may also be contributory to atherosclerosis in HIV-infected patients [1, 22–24]. In the current study, we found that sCD163 related not only to HIV RNA levels but also with CMV IgG titers, and this may be another factor contributing to monocyte activation in our study population. Indeed, animal data suggest acute CMV infection may mediate trafficking of inflammatory monocyte/macrophages from the bone marrow via CCR2 [25]. In contrast, sCD163 did not relate to EBV viral capsid IgG titers in our study. Taken together, these data suggest that CMV titers may be uniquely related to monocyte activation, as measured by sCD163, rather than a reflection of a nonspecific B cell activation.
sCD163 has been previously described as a plasma marker of atherosclerosis in HIV-uninfected patients [8, 26] Activated macrophages expressing CD163 have been found in atherosclerotic plaque in HIV-uninfected patients [27]. These activated macrophages are thought to participate in the atherosclerotic process and contribute to the pathogenesis of cardiovascular disease. However, the specific role played by CD163 itself in the development of atherosclerosis is unknown [28, 29]. In this study, the first among HIV-infected patients to investigate the relationship between sCD163 and atherosclerotic plaque, we find a significant association of sCD163 with the pro-inflammatory CD14+CD16+ monocyte subsets as well as with sCD14. In addition, we demonstrate that levels of LPS in plasma, a measure of circulating microbial products that have crossed the gastrointestinal tract, are also elevated in HIV-infected patients in association with sCD163. LPS and bacterial products have been found in humans with chronic HIV infection and SIV-infected monkeys and correlate with development of AIDS [30, 31]. LPS is a potent stimulant with other macrophage activation products of CD163 expression and secretion by monocyte/macrophages [32, 33]. However, LPS was not itself associated with plaque. In addition, chronicity of HIV infection related to degree of monocyte activation as indicated by sCD163 level. Together, these data suggest that HIV-infected individuals have an increased risk of atherosclerosis due to macrophage-mediated mechanisms, likely resulting from chronic immune activation despite low or undetectable plasma virus.
In contrast to the data from the SMART study, our data in carefully phenotyped patients assessed by coronary angiography do not suggest a relationship between D-dimer and subclinical atherosclerotic disease. The lack of an association between D-dimer and coronary atherosclerosis in our study may be due to the fact that we investigated early asymptomatic cardiovascular disease before an acute coronary event, in contrast to data from the SMART study [34].
The current study has limitations but a number of strengths. We cannot fully determine the contribution of prior inflammation or risk factors to current plaque burden from an assessment at a single point in time in this cross-sectional study. However, nadir CD4 cell count, a marker of prior disease severity, was not related to plaque in the HIV-infected patients. We were unable to study women, and our data cannot be generalized to that group, and future studies will be critical in that population. Nonetheless, our study prospectively enrolled a large number of patients with longstanding HIV infection, largely well controlled, with minimal traditional risks and without known coronary disease and a well-matched control group of HIV-uninfected patients. We used detailed phenotyping of plaque morphology and measurements of monocyte/macrophage activation markers to find a novel relationship with monocyte activation and noncalcified plaque in HIV-infected patients.
Taken together, these data demonstrate monocyte activation in association with increased plaque comprise predominantly noncalcified plaque in this population. In particular, our data demonstrate that sCD163, a marker of monocyte activation, is strongly and uniquely associated with non-calcified coronary lesions in HIV-infected patients. Development of non-calcified lesions related to persistent monocyte activation may predispose HIV-infected patients to plaque rupture and premature CAD. Our data do not negate the impact and need to modify traditional risk factors, which do appear to be associated with increased calcified lesions. The data suggest the need for additional studies to assess how monocyte activation may contribute to atherosclerosis and ways in which this can be minimized among patients with longstanding HIV disease.
Acknowledgments
We thank the patients and the nursing staff on the Massachusetts General Hospital Clinical and Translational Science Award Center for their dedicated patient care.
Financial support. This work was supported by Bristol Myers Squibb; National Institutes of Health (K23 HL092792 to J. L., NS37654 and NS40237 to K. C. W., K24 DK064545 and R01 HL 095123 to S. G., R01 AI071915 to E. S. R., and M01 RR01066-25S1); and the Mary Fisher Clinical AIDS Research and Education Fund. Funding sources had no role in the design of the study, data analysis, or the writing of the manuscript.
Potential conflicts of interest. S. G. received research funding for this investigator-initiated research project through Bristol Myers Squibb, Inc. All other authors report no potential conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Lo J, Abbara S, Shturman L, et al. Increased prevalence of subclinical coronary atherosclerosis detected by coronary computed tomography angiography in HIV-infected men. AIDS. 2010;24:243–53. doi: 10.1097/QAD.0b013e328333ea9e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Motoyama S, Kondo T, Sarai M, et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J Am Coll Cardiol. 2007;50:319–26. doi: 10.1016/j.jacc.2007.03.044. [DOI] [PubMed] [Google Scholar]
- 3.van der Wal A, Becker A, van der Loos C, Das P. Site of intimal rupture or erosion of thrombosed coronary atherosclerotic plaques is characterized by an inflammatory process irrespective of the dominant plaque morphology. Circulation. 1994;89:36–44. doi: 10.1161/01.cir.89.1.36. [DOI] [PubMed] [Google Scholar]
- 4.Swirski FK, Libby P, Aikawa E, et al. Ly-6chi monocytes dominate hypercholesterolemia-associated monocytosis and give rise to macrophages in atheromata. J Clin Invest. 2007;117:195–205. doi: 10.1172/JCI29950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fabriek BO, Dijkstra CD, van den Berg TK. The macrophage scavenger receptor cd163. Immunobiology. 2005;210:153–60. doi: 10.1016/j.imbio.2005.05.010. [DOI] [PubMed] [Google Scholar]
- 6.Burdo TH, Soulas C, Orzechowski K, et al. Increased monocyte turnover from bone marrow correlates with severity of SIV encephalitis and cd163 levels in plasma. PLoS Pathog. 2010;6:e1000842. doi: 10.1371/journal.ppat.1000842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Burdo TH, Lentz MR, Autissier P, et al. Soluble cd163 made by monocyte/macrophages is a novel marker of HIV activity in early and chronic infection prior to and after anti-retroviral therapy. J Infect Dis. 2011;204:154–63. doi: 10.1093/infdis/jir214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Aristoteli LP, Moller HJ, Bailey B, Moestrup SK, Kritharides L. The monocytic lineage specific soluble cd163 is a plasma marker of coronary atherosclerosis. Atherosclerosis. 2006;184:342–7. doi: 10.1016/j.atherosclerosis.2005.05.004. [DOI] [PubMed] [Google Scholar]
- 9.Wilson PWF, D'Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB. Prediction of coronary heart disease using risk factor categories. Circulation. 1998;97:1837–47. doi: 10.1161/01.cir.97.18.1837. [DOI] [PubMed] [Google Scholar]
- 10.Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M, Jr, Detrano R. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol. 1990;15:827–32. doi: 10.1016/0735-1097(90)90282-t. [DOI] [PubMed] [Google Scholar]
- 11.Cserti-Gazdewich CM, Dzik WH, Dorn ME, et al. Quantitation of cd36 (platelet glycoprotein iv) expression on platelets and monocytes by flow cytometry: application to the study of Plasmodium falciparum malaria. Cytometry B Clin Cytom. 2009;76:127–34. doi: 10.1002/cyto.b.20443. [DOI] [PubMed] [Google Scholar]
- 12.Cannizzo E, Bellio E, Sohani AR, et al. Multiparameter immunophenotyping by flow cytometry in multiple myeloma: the diagnostic utility of defining ranges of normal antigenic expression in comparison to histology. Cytometry B Clin Cytom. 2010;78:231–8. doi: 10.1002/cyto.b.20517. [DOI] [PubMed] [Google Scholar]
- 13.Ganesan A, Crum-Cianflone N, Higgins J, et al. High dose atorvastatin decreases cellular markers of immune activation without affecting HIV-1 RNA levels: results of a double-blind randomized placebo controlled clinical trial. J Infect Dis. 2011;203:756–64. doi: 10.1093/infdis/jiq115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.El-Sadr WM, Lundgren JD, Neaton JD, et al. CD4+ count-guided interruption of antiretroviral treatment. N Engl J Med. 2006;355:2283–96. doi: 10.1056/NEJMoa062360. [DOI] [PubMed] [Google Scholar]
- 15.Stein JH. Cardiovascular risks of antiretroviral therapy. N Engl J Med. 2007;356:1773–5. doi: 10.1056/NEJMe078037. [DOI] [PubMed] [Google Scholar]
- 16.Triant VA, Lee H, Hadigan C, Grinspoon SK. Increased acute myocardial infarction rates and cardiovascular risk factors among patients with human immunodeficiency virus disease. J Clin Endocrinol Metab. 2007;92:2506–12. doi: 10.1210/jc.2006-2190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lichtenstein KA, Armon C, Buchacz K, et al. Low CD4+ T cell count is a risk factor for cardiovascular disease events in the HIV Outpatient Study. Clin Infect Dis. 2010;51:435–47. doi: 10.1086/655144. [DOI] [PubMed] [Google Scholar]
- 18.Kaplan RC, Sinclair E, Landay AL, et al. T cell activation and senescence predict subclinical carotid artery disease in HIV-infected women. J Infect Dis. 2011;203:452–63. doi: 10.1093/infdis/jiq071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hsue PY, Hunt PW, Wu Y, et al. Association of abacavir and impaired endothelial function in treated and suppressed HIV-infected patients. AIDS. 2009;23:2021–7. doi: 10.1097/QAD.0b013e32832e7140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Libby P. Inflammation in atherosclerosis. Nature. 2002;420:868–74. doi: 10.1038/nature01323. [DOI] [PubMed] [Google Scholar]
- 21.Ross R. Atherosclerosis–an inflammatory disease. N Engl J Med. 1999;340:115–26. doi: 10.1056/NEJM199901143400207. [DOI] [PubMed] [Google Scholar]
- 22.Melnick JL, Adam E, DeBakey ME. Possible role of cytomegalovirus in atherogenesis. JAMA. 1990;263:2204–7. [PubMed] [Google Scholar]
- 23.Hsue PY, Hunt PW, Sinclair E, et al. Increased carotid intima-media thickness in hiv patients is associated with increased cytomegalovirus-specific T-cell responses. AIDS. 2006;20:2275–83. doi: 10.1097/QAD.0b013e3280108704. [DOI] [PubMed] [Google Scholar]
- 24.Epstein SE, Zhu J, Najafi AH, Burnett MS. Insights into the role of infection in atherogenesis and in plaque rupture. Circulation. 2009;119:3133–41. doi: 10.1161/CIRCULATIONAHA.109.849455. [DOI] [PubMed] [Google Scholar]
- 25.Crane MJ, Hokeness-Antonelli KL, Salazar-Mather TP. Regulation of inflammatory monocyte/macrophage recruitment from the bone marrow during murine cytomegalovirus infection: role for type i interferons in localized induction of ccr2 ligands. J Immunol. 2009;183:2810–7. doi: 10.4049/jimmunol.0900205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Moreno JA, Munoz-Garcia B, Martin-Ventura JL, et al. The cd163-expressing macrophages recognize and internalize tweak: potential consequences in atherosclerosis. Atherosclerosis. 2009;207:103–10. doi: 10.1016/j.atherosclerosis.2009.04.033. [DOI] [PubMed] [Google Scholar]
- 27.Ratcliffe NR, Kennedy SM, Morganelli PM. Immunocytochemical detection of fcgamma receptors in human atherosclerotic lesions. Immunol Lett. 2001;77:169–74. doi: 10.1016/s0165-2478(01)00217-6. [DOI] [PubMed] [Google Scholar]
- 28.Philippidis P, Mason JC, Evans BJ, et al. Hemoglobin scavenger receptor cd163 mediates interleukin-10 release and heme oxygenase-1 synthesis: antiinflammatory monocyte-macrophage responses in vitro, in resolving skin blisters in vivo, and after cardiopulmonary bypass surgery. Circ Res. 2004;94:119–26. doi: 10.1161/01.RES.0000109414.78907.F9. [DOI] [PubMed] [Google Scholar]
- 29.Schaer CA, Schoedon G, Imhof A, Kurrer MO, Schaer DJ. Constitutive endocytosis of cd163 mediates hemoglobin-heme uptake and determines the noninflammatory and protective transcriptional response of macrophages to hemoglobin. Circ Res. 2006;99:943–50. doi: 10.1161/01.RES.0000247067.34173.1b. [DOI] [PubMed] [Google Scholar]
- 30.Brenchley JM, Price DA, Schacker TW, et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat Med. 2006;12:1365–71. doi: 10.1038/nm1511. [DOI] [PubMed] [Google Scholar]
- 31.Jiang W, Lederman MM, Hunt P, et al. Plasma levels of bacterial DNA correlate with immune activation and the magnitude of immune restoration in persons with antiretroviral-treated HIV infection. J Infect Dis. 2009;199:1177–85. doi: 10.1086/597476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Weaver LK, Hintz-Goldstein KA, Pioli PA, et al. Pivotal advance: activation of cell surface toll-like receptors causes shedding of the hemoglobin scavenger receptor cd163. J Leukoc Biol. 2006;80:26–35. doi: 10.1189/jlb.1205756. [DOI] [PubMed] [Google Scholar]
- 33.Hintz KA, Rassias AJ, Wardwell K, et al. Endotoxin induces rapid metalloproteinase-mediated shedding followed by up-regulation of the monocyte hemoglobin scavenger receptor cd163. J Leukoc Biol. 2002;72:711–7. [PubMed] [Google Scholar]
- 34.Kuller LH, Tracy R, Belloso W, et al. Inflammatory and coagulation biomarkers and mortality in patients with hiv infection. PLoS Med. 2008;5:e203. doi: 10.1371/journal.pmed.0050203. [DOI] [PMC free article] [PubMed] [Google Scholar]