Abstract
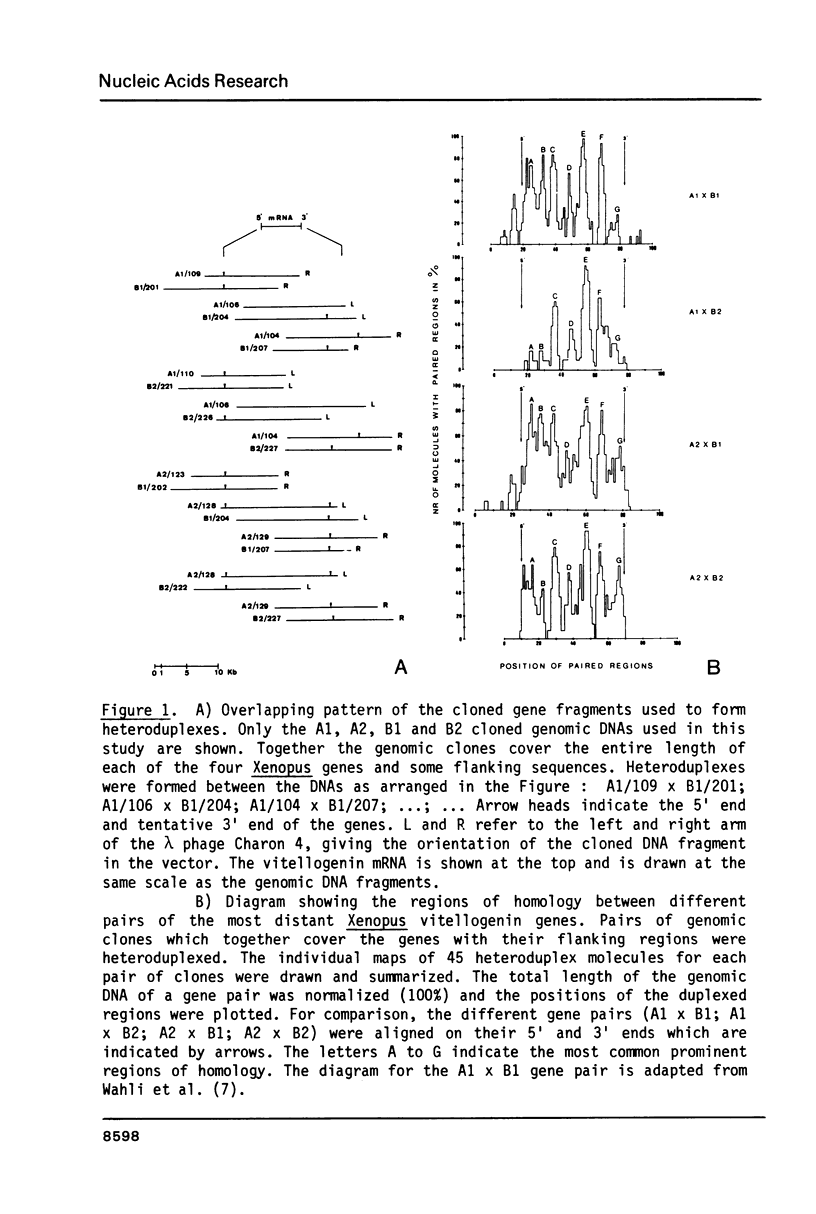
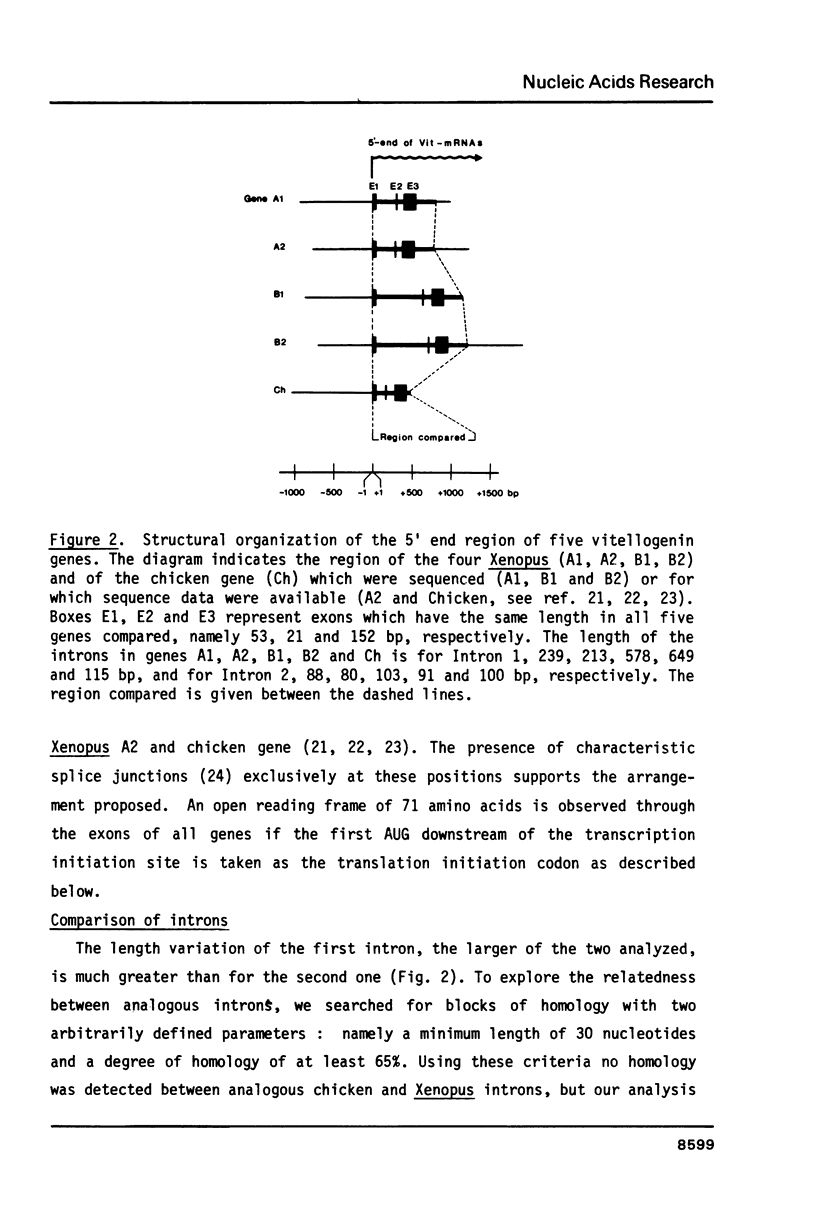
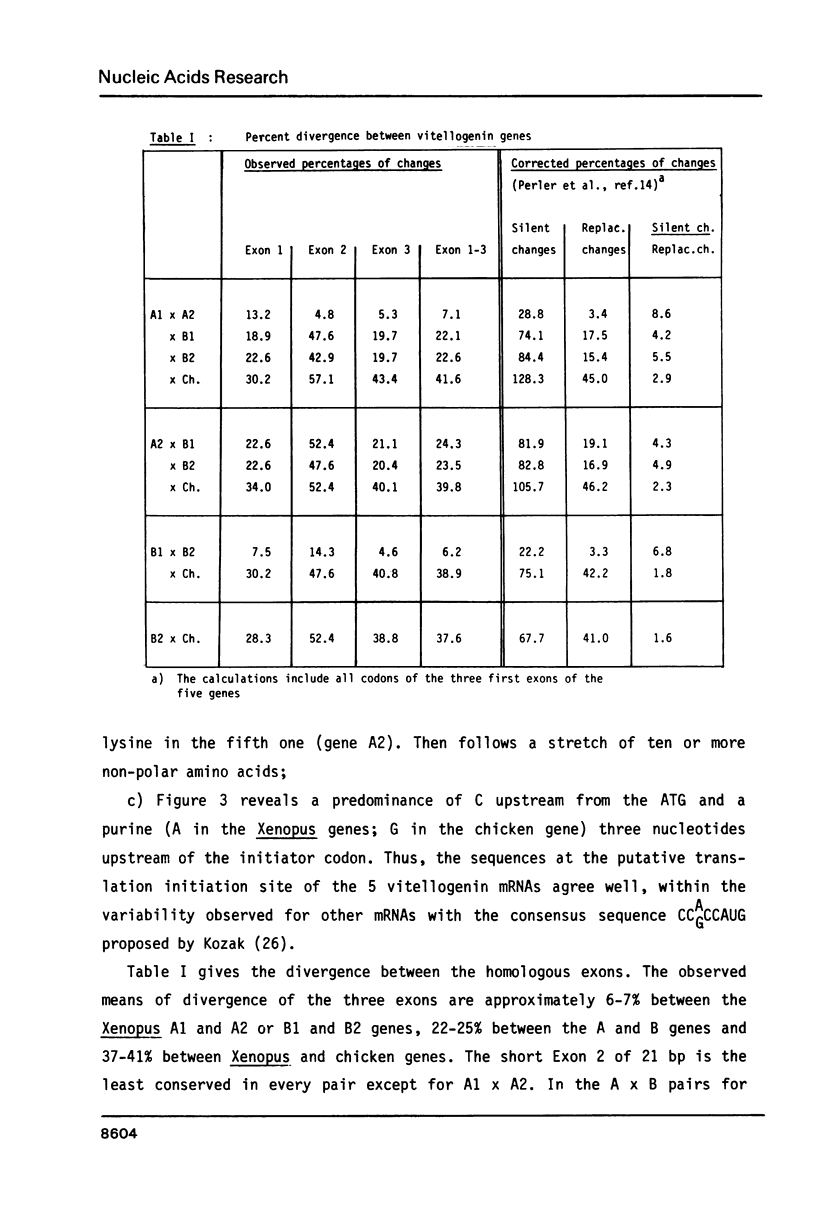
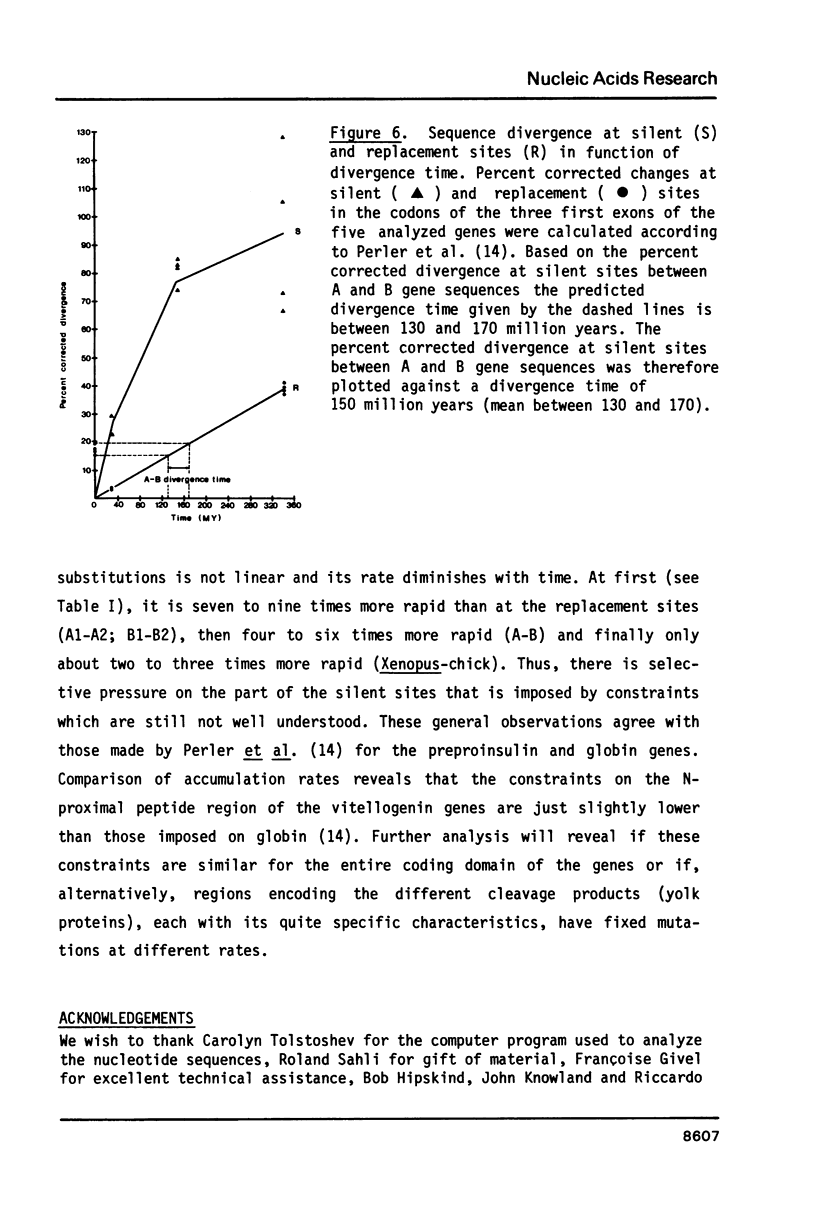
Electron microscopic analysis of heteroduplexes between the most distantly related Xenopus vitellogenin genes (A genes X B genes) has revealed the distribution of homologous regions that have been preferentially conserved after the duplication events that gave rise to the multigene family in Xenopus laevis. DNA sequence analysis was limited to the region downstream of the transcription initiation site of the Xenopus genes A1, B1 and B2 and a comparison with the Xenopus A2 and the major chicken vitellogenin gene is presented. Within the coding regions of the first three exons, nucleotide substitutions resulting in amino acid changes accumulate at a rate similar to that observed in globin genes. This suggests that the duplication event which led to the formation of the A and B ancestral genes in Xenopus laevis occurred about 150 million years ago. Homologous exons of the A1-A2 and B1-B2 gene pairs, which formed about 30 million years ago, show a quite similar sequence divergence. In contrast, A1-A2 homologous introns seem to have evolved much faster than their B1-B2 counterparts.
Full text
PDF










Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnberg A. C., Meijlink F. C., Mulder J., van Bruggen E. F., Gruber M., Geert A. B. Isolation and characterization of genomic clones covering the chicken vitellogenin gene. Nucleic Acids Res. 1981 Jul 24;9(14):3271–3286. doi: 10.1093/nar/9.14.3271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisbee C. A., Baker M. A., Wilson A. C., Haji-Azimi I., Fischberg M. Albumin phylogeny for clawed frogs (Xenopus). Science. 1977 Feb 25;195(4280):785–787. doi: 10.1126/science.65013. [DOI] [PubMed] [Google Scholar]
- Blanchetot A., Wilson V., Wood D., Jeffreys A. J. The seal myoglobin gene: an unusually long globin gene. Nature. 1983 Feb 24;301(5902):732–734. doi: 10.1038/301732a0. [DOI] [PubMed] [Google Scholar]
- Burch J. B. Identification and sequence analysis of the 5' end of the major chicken vitellogenin gene. Nucleic Acids Res. 1984 Jan 25;12(2):1117–1135. doi: 10.1093/nar/12.2.1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burch J. B., Weintraub H. Temporal order of chromatin structural changes associated with activation of the major chicken vitellogenin gene. Cell. 1983 May;33(1):65–76. doi: 10.1016/0092-8674(83)90335-5. [DOI] [PubMed] [Google Scholar]
- Efstratiadis A., Posakony J. W., Maniatis T., Lawn R. M., O'Connell C., Spritz R. A., DeRiel J. K., Forget B. G., Weissman S. M., Slightom J. L. The structure and evolution of the human beta-globin gene family. Cell. 1980 Oct;21(3):653–668. doi: 10.1016/0092-8674(80)90429-8. [DOI] [PubMed] [Google Scholar]
- Geiser M., Mattaj I. W., Wilks A. F., Seldran M., Jost J. P. Structure and sequence of the promoter area and of a 5' upstream demethylation site of the estrogen-regulated chicken vitellogenin ii gene. J Biol Chem. 1983 Jul 25;258(14):9024–9030. [PubMed] [Google Scholar]
- Hong G. F. A systemic DNA sequencing strategy. J Mol Biol. 1982 Jul 5;158(3):539–549. doi: 10.1016/0022-2836(82)90213-3. [DOI] [PubMed] [Google Scholar]
- Kreil G. Transfer of proteins across membranes. Annu Rev Biochem. 1981;50:317–348. doi: 10.1146/annurev.bi.50.070181.001533. [DOI] [PubMed] [Google Scholar]
- Messing J., Crea R., Seeburg P. H. A system for shotgun DNA sequencing. Nucleic Acids Res. 1981 Jan 24;9(2):309–321. doi: 10.1093/nar/9.2.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mount S. M. A catalogue of splice junction sequences. Nucleic Acids Res. 1982 Jan 22;10(2):459–472. doi: 10.1093/nar/10.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perler F., Efstratiadis A., Lomedico P., Gilbert W., Kolodner R., Dodgson J. The evolution of genes: the chicken preproinsulin gene. Cell. 1980 Jun;20(2):555–566. doi: 10.1016/0092-8674(80)90641-8. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shapiro D. Steroid hormone regulation of vitellogenin gene expression. CRC Crit Rev Biochem. 1982 Mar;12(3):187–203. doi: 10.3109/10409238209108706. [DOI] [PubMed] [Google Scholar]
- Wahli W., Dawid I. B. Isolation of two closely related vitellogenin genes, including their flanking regions, from a Xenopus laevis gene library. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1437–1441. doi: 10.1073/pnas.77.3.1437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wahli W., Dawid I. B., Ryffel G. U., Weber R. Vitellogenesis and the vitellogenin gene family. Science. 1981 Apr 17;212(4492):298–304. doi: 10.1126/science.7209528. [DOI] [PubMed] [Google Scholar]
- Wahli W., Dawid I. B., Wyler T., Jaggi R. B., Weber R., Ryffel G. U. Vitellogenin in Xenopus laevis is encoded in a small family of genes. Cell. 1979 Mar;16(3):535–549. doi: 10.1016/0092-8674(79)90028-x. [DOI] [PubMed] [Google Scholar]
- Wahli W., Dawid I. B., Wyler T., Weber R., Ryffel G. U. Comparative analysis of the structural organization of two closely related vitellogenin genes in X. laevis. Cell. 1980 May;20(1):107–117. doi: 10.1016/0092-8674(80)90239-1. [DOI] [PubMed] [Google Scholar]
- Wahli W., Germond J. E., ten Heggeler B., May F. E. Vitellogenin genes A1 and B1 are linked in the Xenopus laevis genome. Proc Natl Acad Sci U S A. 1982 Nov;79(22):6832–6836. doi: 10.1073/pnas.79.22.6832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker P., Brown-Luedi M., Germond J. E., Wahli W., Meijlink F. C., van het Schip A. D., Roelink H., Gruber M., Ab G. Sequence homologies within the 5' end region of the estrogen-controlled vitellogenin gene in Xenopus and chicken. EMBO J. 1983;2(12):2271–2279. doi: 10.1002/j.1460-2075.1983.tb01734.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S. Y., Williams D. L. Identificiation, purification, and characterization of two distinct avian vitellogenins. Biochemistry. 1980 Apr 15;19(8):1557–1563. doi: 10.1021/bi00549a004. [DOI] [PubMed] [Google Scholar]
- Wilks A., Cato A. C., Cozens P. J., Mattaj I. W., Jost J. P. Isolation and fine structure organisation of an avian vitellogenin gene coding for the major estrogen-inducible mRNA. Gene. 1981 Dec;16(1-3):249–259. doi: 10.1016/0378-1119(81)90081-0. [DOI] [PubMed] [Google Scholar]
- Wilson A. C., Carlson S. S., White T. J. Biochemical evolution. Annu Rev Biochem. 1977;46:573–639. doi: 10.1146/annurev.bi.46.070177.003041. [DOI] [PubMed] [Google Scholar]
- von Heijne G. How signal sequences maintain cleavage specificity. J Mol Biol. 1984 Feb 25;173(2):243–251. doi: 10.1016/0022-2836(84)90192-x. [DOI] [PubMed] [Google Scholar]



