Abstract
Background
In select patient populations, Doppler echocardiographic indices may be used to estimate left sided filling pressures. It is not known, however, whether changes in these indices track changes in left-sided filling pressures within individual healthy subjects or patients with heart failure with preserved ejection fraction (HFpEF). This knowledge is important as it would support, or refute, the serial use of these indices to estimate changes in filling pressures associated with the titration of medical therapy in patients with heart failure.
Methods and Results
Forty seven volunteers were enrolled: 11 highly screened elderly outpatients with a clear diagnosis of HFpEF, 24 healthy elderly subjects and 12 healthy young subjects. Each patient underwent right heart catheterization with simultaneous transthoracic echo. Pulmonary capillary wedge pressure (PCWP) and key echo indices (E/e’ & E/Vp) were measured at two baselines and during four preload altering maneuvers: lower body negative pressure (LBNP) -15 mmHg; LBNP -30 mmHg; rapid saline infusion of 10-15 ml/kg; and rapid saline infusion of 20-30 ml/kg. A random coefficient mixed model regression of PCWP vs. E/e’ and PCWP vs. E/Vp was performed for: 1) a composite of all data points; 2) a composite of all data points within each of the three groups. Linear regression analysis was performed for individual subjects. With this protocol, PCWP was manipulated from 0.8 to 28.8 mmHg. For E/e’, the composite random effects mixed model regression was PCWP = 0.58×E/e’+7.02 (p<0.001) confirming the weak but significant relationship between these two variables. Individual subject linear regression slopes (range: -6.76 to 11.03) and r2 (0.00 to 0.94) were highly variable and often very different than those derived for the composite and group regressions. For E/Vp, the composite random coefficient mixed model regression was PCWP = 1.95×E/Vp +7.48 (p=0.005); once again, individual subject linear regression slopes (range: -16.42 to 25.39) and r2 (range: 0.02 to 0.94) were highly variable and often very different than those derived for the composite and group regressions.
Conclusions
Within individual subjects the non-invasive indices E/e’ and E/Vp do not reliably track changes in left-sided filling pressures as these pressures vary, precluding the use of these techniques in research studies with healthy volunteers or the titration of medical therapy in patients with HFpEF.
Keywords: echocardiography, pressure, ultrasonics, Doppler, heart failure
Several modalities of echocardiography including Doppler, tissue Doppler and color-M-mode have shown promise in the estimation of left-sided filling pressures. Two indices: a) the ratio of the peak early mitral inflow velocity over the early diastolic mitral annular velocity (E/e’) 1; and b) the ratio of the peak early mitral inflow velocity over the color M-mode Doppler flow propagation velocity (E/Vp) 2 have received recognition for their utility in the one-time estimation of left sided filling pressures in healthy subjects 3 as well as select patient populations with cardiovascular disease 4,5,6,7,8. Accordingly, their application in the diagnosis of diastolic heart failure has been recommended by the European Society of Cardiology 9. Conversely, these indices have proven to be less reliable in alternate populations including primary mitral regurgitation 8, mitral stenosis 10, symptomatic hypertrophic cardiomyopathy 11 and most recently, decompensated advanced systolic heart failure 12, calling into question their broad applicability.
It has been proposed that non-invasive indices of left-sided filling pressures be used to titrate medical therapy in patients with heart failure 4, 7, 13. Doing so would require demonstration that changes in non-invasive indices accurately track changes in left-sided filling pressures within individuals as filling pressures vary. To accomplish this goal, we sought to make multiple, simultaneous measures within individual subjects as preload was manipulated using lower body negative pressure and rapid saline infusion. Study groups included healthy subjects across a broad age range and elderly outpatients with heart failure with preserved ejection fraction (HFpEF).
Methods
Subjects
Three study groups were evaluated: 1) elderly outpatients with heart failure with preserved ejection fraction (HFpEF) (n=11); 2) healthy elderly subjects (n=24); and 3) healthy young subjects (n=12).
Patients over the age of 65 years with a discharge diagnosis of congestive heart failure (CHF) were considered for the HFpEF group if they had a clear history of CHF by Framingham criteria 14 with an ejection fraction (EF) > 50% documented at the time of their index hospitalization. In addition, all HFpEF patients were required to have either an elevated brain natriuretic peptide level (BNP), documented pulmonary congestion by chest X-ray or evidence of elevated left sided filling pressures by right heart catheterization at the time of their index hospitalization. Exclusion criteria included other conditions that might explain the hospitalization, including the presence of atrial fibrillation at the time of the hospitalization, prior coronary artery bypass grafting (CABG), active myocardial ischemia, unrevascularized obstructive coronary disease (>50% lesions by prior angiography), stable angina, recent (< 1 year) myocardial infarction, renal failure (creatinine > 2.5 g/dl) or dialysis dependence, severe COPD/or pulmonary disease, active malignancy, moderate or severe valvular heart disease, and warfarin use. HFpEF patients enrolled in the study also participated in additional testing beyond the scope of this manuscript; additional details regarding the screening of HFpEF patients are available elsewhere15, 16.
The healthy elderly study group consisted of subjects over the age of 65 used previously in our research 17, 18. All elderly subjects were carefully screened for hypertension and cardiac disease, including structural heart disease and hemodynamically significant obstructive coronary disease, using a history and physical examination as well as resting and post-exercise transthoracic echocardiograms. Additional exclusion criteria for this group included the presence of valvular heart disease, atrial flutter or fibrillation, renal insufficiency, chronic lung disease, regular cigarette smoking within the past 10 years or the use of cardiovascular medications. The healthy young study group consisted of subjects under the age of 50 used previously in our research 18. These subjects underwent a similar screening protocol as the healthy elderly subjects described above.
All subjects signed an informed consent approved by the institutional review boards of the University of Texas-Southwestern Medical Center at Dallas, Medical City Hospital, Dallas, Texas, Doctors Hospital Dallas, Texas or Presbyterian Hospital of Dallas.
Experimental Protocol
Subjects were studied in the resting supine or left lateral position. A 6 Fr balloon-tipped fluid-filled catheter (Edwards Lifesciences, Irvine, California) was placed with fluoroscopic guidance through an antecubital vein into the pulmonary artery. The catheter was connected to a physiologic pressure transducer with the zero reference point placed 5.0 cm below the sternal angle. The wedge position of the catheter tip was confirmed fluoroscopically and by the presence of an appropriate pulmonary capillary wedge pressure (PCWP) waveform. The PCWP balloon was inflated during data collection at each loading condition; in between data collections, the balloon was deflated.
After 30 minutes of quiet supine rest, baseline data were collected. Cardiac filling was then decreased using lower body negative pressure (LBNP) as previously described 19. The levels of LBNP used were -15 and -30 mm Hg. Due to large body habitus limiting use of the LBNP apparatus in 2 of the HFpEF subjects, head-up tilt at (20 and 40 or 60 degrees) was used instead of LBNP, with the pressure transducer zero position carefully adjusted to the level of the right atrium documented by flouroscopy and echocardiography. Five minutes into each level of cardiac unloading, three measures of the mean PCWP were obtained at end-expiration using a previously described technique 20. These measurements were then averaged; Doppler echocardiography immediately followed.
After release of the negative pressure (or return to supine position) and confirmed return to hemodynamic baseline, cardiac filling was increased through a rapid infusion of warm (37°C) isotonic saline solution at 100-200 mL/min. The above measures were repeated after the infusion of 10-15 mL/kg and 20-30 mL/kg with smaller volumes generally used in the HFpEF patients to keep the PCWP < 25 mmHg and to avoid inducing pulmonary edema.
Echocardiography
For all subjects, at each level of cardiac loading and unloading, a transthoracic echocardiogram was obtained using an ATL (Advanced Technology Laboratories, Bothell, Washington) HDI 5000CV (software version 10.1) or an iE33 (Philips Healthcare, Bothell, WA) ultrasound machine. Traditional apical 4- and 5- chamber views were used for all measures. Care was taken to obtain all images in the traditional imaging planes by an experienced echocardiographer. All images were evaluated blindly offline by an experienced sonographer in the Cleveland Clinic echo core laboratory; this reader was unaware of the resultant changes in PCWP associated with preload manipulation.
Doppler Measurements
Similar to measurements of the PCWP, all Doppler indices were obtained at end-expiration with three values averaged per stage of preload manipulation. Pulse-waved Doppler, using a sample volume of 2.0 mm placed at the tips of the mitral valve leaflets, was used to determine peak velocities of mitral inflow (E and A velocities). For tissue Doppler measurements, the septal and lateral walls were highlighted in the apical 4-chamber view. Using pulse-wave Doppler, a sample volume of 4.0 mm was placed at the septal side of the mitral annulus; the resulting early diastolic waveform velocity was recorded. This process was then repeated for the lateral side of the mitral annulus. Values were averaged to obtain TDI emean 21 which is abbreviated as e’ throughout the rest of the manuscript. A color M-mode image of left ventricular inflow was obtained with the sampling area positioned to extend from mid-atrium to the apex, directly through the mitral valve orifice. The scale was reduced sufficiently to result in clear aliasing within the early portion of the mitral inflow. The resulting mitral inflow spatiotemporal velocity profile pattern was used to derive the early propagation velocity of mitral inflow (Vp) as previously described 22.
Statistical Analysis
Numerical data are presented as mean ± standard deviation. Random coefficient mixed models analysis was performed to estimate regression parameters between non-invasive estimates of left-sided filling pressure (i.e. E/e’ and E/Vp) and invasive measures of left-sided filling pressure (PCWP) in instances in which group data was comprised of multiple data points from individual subjects, thereby accounting for the correlation within individual subjects. Linear regression analysis was performed to estimate regression parameters between non-invasive estimates of left-sided filling pressure and invasive measures of left-sided filling pressure in instances in which: 1) group data was comprised of single data points from individual subjects; or 2) a single subject’s data was being regressed. Standard error of the estimate (SE) was determined for the slope coefficient in each simple linear regression. As clinicians and researchers are generally interested in estimating the PCWP by the non-invasive measures of E/e’ or E/Vp, PCWP was set as the dependent variable and E/e’ and E/Vp as the independent variables in the analyses except in the group data for each stage of preload manipulation. All analyses were performed by use of commercial software (SigmaPlot 11.0, Systat Software. Inc, Chicago, IL and SAS version 9.2, SAS Institute, Cary, NC).
Results
Characteristics for each of the three study groups are presented in Table 1. Additional characteristics of the HFpEF patients 15, 16, healthy elderly subjects 17, 18 and healthy young subjects 18 can be found elsewhere.
Table 1.
Group Characteristics
| Variable | HFpEF (n=11) | Healthy Elderly (n=24) | Healthy Young (n=12) |
|---|---|---|---|
| Age (years) | 73.0 ± 6.8 | 69.2 ± 3.4 | 32.3 ± 9.0 |
| Female (%) | 64% | 46% | 25% |
| Height (cm) | 162.4 ± 9.6 | 169.3 ± 10.5 | 179.2 ± 8.2 |
| Weight (kg) | 88.9 ± 21.3 | 69.5 ± 12.5 | 74.9 ± 15.9 |
| Body Mass Index (kg/m2) | 33.6 ± 6.7 | 24.1 ± 2.8 | 23.1 ± 3.4 |
| Body Surface Area (m2) | 1.99 ± 0.27 | 1.80 ± 0.21 | 1.90 ± 0.27 |
| Peak VO2 (ml/kg/min) | 13.7 ± 3.4 | 29.9 ± 9.8 | 40.6 ± 10.1 |
Data are presented as mean ± standard deviation
Alterations in Preload
The mean PCWP, mean E/e’ and mean E/Vp (± standard deviation) by group for each stage of preload manipulation are presented in Figure 1. The application of LBNP at levels of -15 and -30 mmHg lowered PCWP in each group; the rapid infusion of warm saline at 10-15 and 20-30 mL/kg increased PCWP in each group. With this protocol, a range of PCWP from 0.8 mmHg to 28.8 mmHg was achieved.
Figure 1.
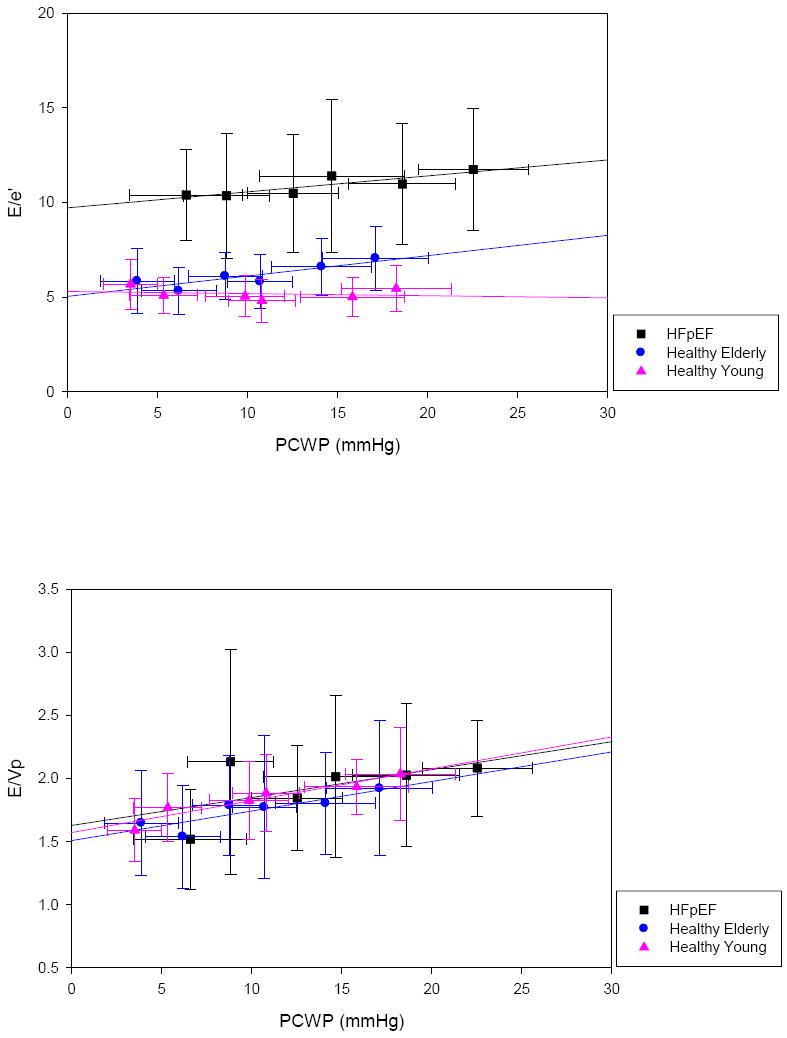
E/e’ (top) and E/Vp (bottom) across filling pressures. Data represent group means ± standard deviations at each stage of preload manipulation.
E/e’ and PCWP
In the composite of all subjects, the random coefficient mixed model regression was PCWP = 0.58×E/e’+7.02 (p<0.001) (Table 2); this relationship was based on 267 paired measurements of E/e’ and PCWP (Figure 2). The linear regression slopes of the 47 individual subjects who comprised this composite varied greatly, ranging from -6.76 to 11.03. Individual coefficients of determination (r2) also varied greatly, ranging from 0.00 to 0.94.
Table 2.
Summary Data for Non-Invasive Index E/e’
| Group (n = subjects) | Paired Data Points (E/e’ & PCWP) | Group Random Coefficient Mixed Model Regression | p Value | Range of Individual Linear Regression Slopes Within Group | Range of Individual r2 Within Group | ||
|---|---|---|---|---|---|---|---|
| Average | (Min - Max) | Average | (Min - Max) | ||||
| HFpEF (n=11) | 60 | PCWP = 0.83*E/e’ + 3.93 | 0.04 | 1.46 | (-1.44 - 4.42) | 0.38 | (0.01 - 0.82) |
| Healthy Elderly (n=24) | 137 | PCWP = 0.93*E/e’ + 4.60 | 0.008 | 3.12 | (-2.37 - 11.03) | 0.42 | (0.00 - 0.94) |
| Healthy Young (n=12) | 70 | PCWP = 0.07*E/e’ + 10.33 | 0.91 | -1.31 | (-6.76 - 6.35) | 0.27 | (0.00 - 0.76) |
| All Subjects (n=47) | 267 | PCWP = 0.58*E/e’ + 7.02 | <0.0001 | 1.60 | (-6.76 - 11.03) | 0.37 | (0.00 - 0.94) |
Figure 2.
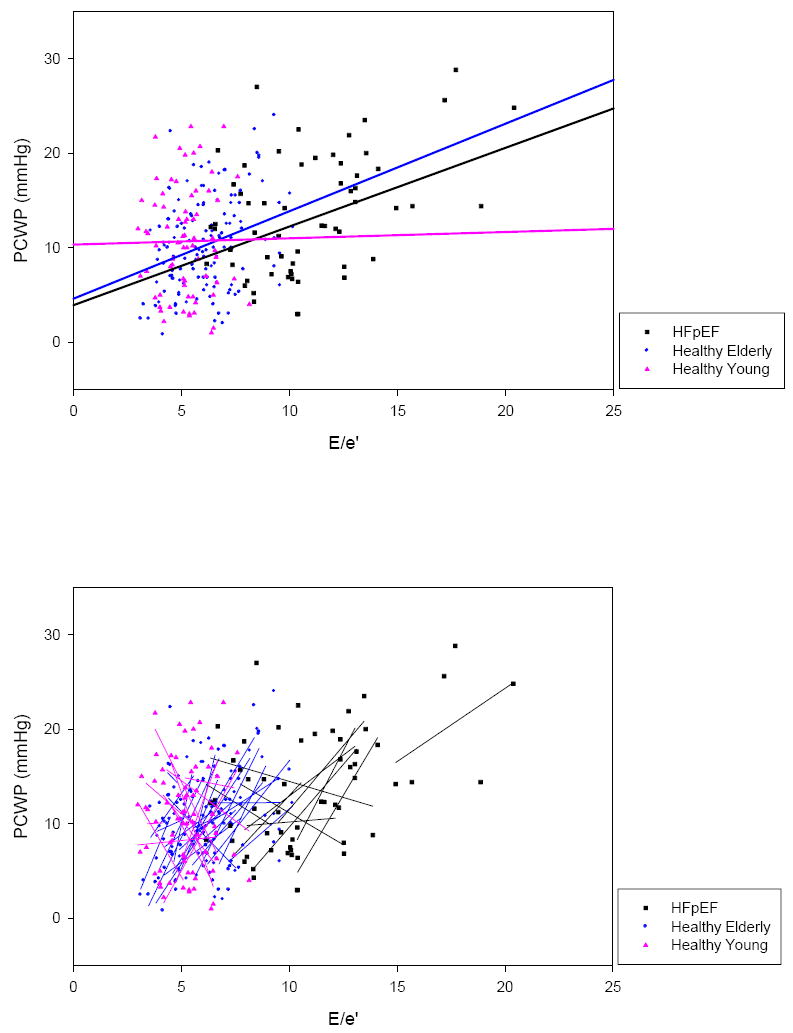
Composite of E/e’ vs. PCWP in all subjects. Group random coefficient mixed model regressions (top) and individual linear regressions (bottom) are superimposed. Individual linear regressions were highly variable in regards to slope and r2.
In healthy subjects, group random coefficient mixed model regression slopes ranged from 0.07 in the young group to 0.93 in the elderly group. Within the individual subjects who comprised these groups, linear regression slopes ranged from negative to steeply positive and r2 ranged from low to high. Based on linear regression slopes, in 10 healthy subjects changes in E/e’ and PCWP were discordant (E/e’ decreased as PCWP increased) whereas in the other 24, changes were concordant.
Using only baseline measures, the HFpEF group’s linear regression was PCWP = 0.52×E/e’ + 6.85 (p=0.04, r2 = 0.42, SE = 1.99) (Figure 3). However, with preload manipulations the linear regression slopes of the individual HFpEF patients varied greatly, ranging from -1.44 to 4.42. Likewise, individual HFpEF patient r2 varied from 0.01 to 0.82. Based on linear regression slopes, in 3 HFpEF patients changes in E/e’ and PCWP were discordant whereas in the other 8, changes were concordant. To further characterize these relationships, the linear regression slopes of E vs. PCWP and e’ vs. PCWP in individual HFpEF patients are presented in Figure 4.
Figure 3.
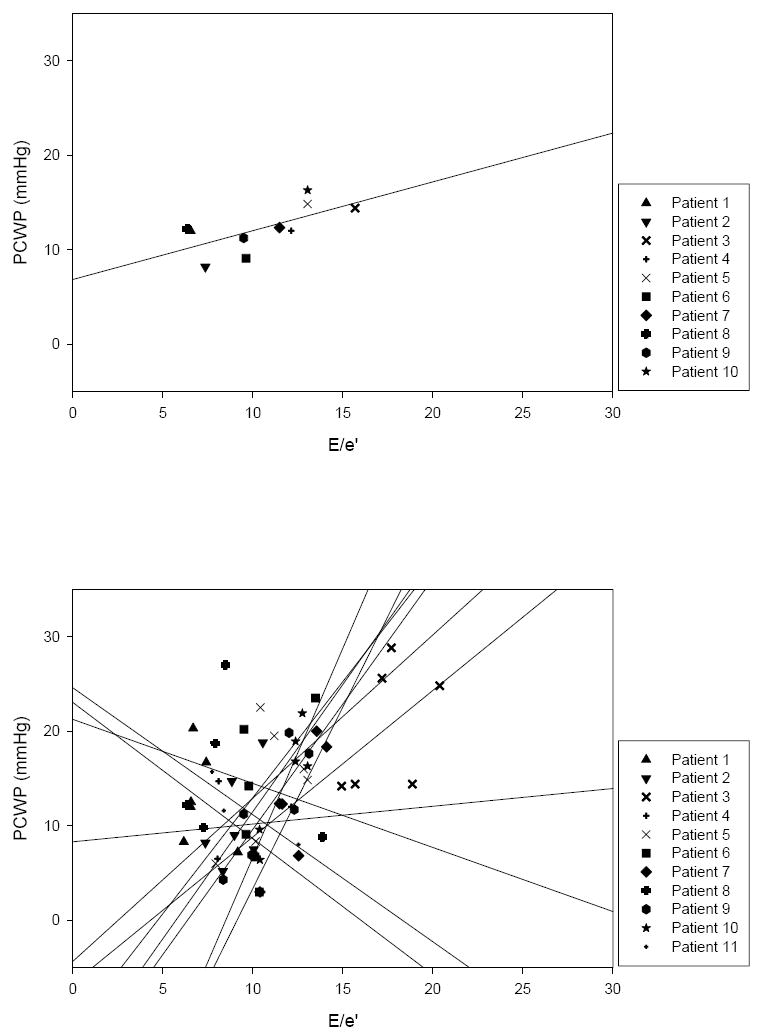
Top: E/e’ vs. PCWP in HFpEF patients at baseline with superimposed group linear regression (one HFpEF patient could not adequately imaged at baseline). Bottom: E/e’ vs. PCWP in individual HFpEF patients with superimposed linear regressions (note: linear regressions are extended to axes). Again, individual linear regressions were highly variable in regards to slope and r2.
Figure 4.
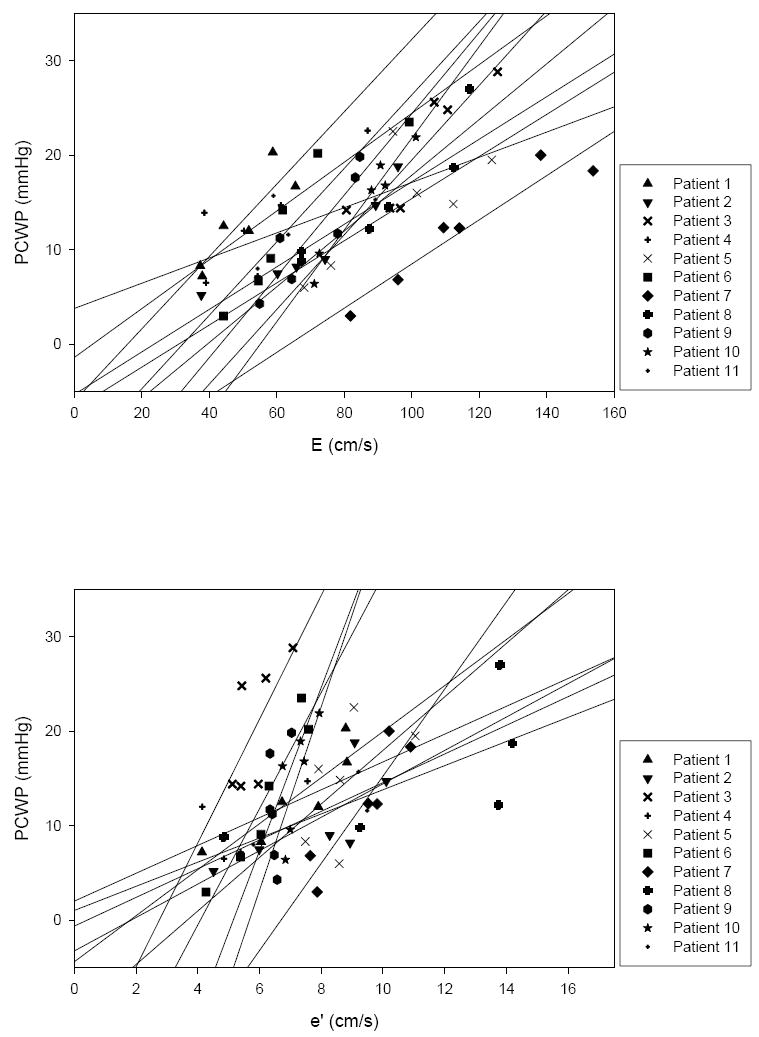
Top: E vs. PCWP in individual HFpEF patients with superimposed linear regressions. E demonstrated a degree of preload dependence which was similar between individual HFpEF patients. Bottom: e’ vs. PCWP in individual HFpEF patients with superimposed linear regressions. e’ varied greatly between individual HFpEF patients accounting for the broad variability observed in individual E/e’ slopes as preload was manipulated.
E/Vp and PCWP
In the composite of all subjects, the random coefficient mixed model regression was PCWP = 1.95×E/Vp +7.48 (p=0.005) (Table 3); this relationship was based on 256 paired measurements (Figure 5). Within this composite, the linear regression slopes of 46 individual subjects ranged from -16.42 to 25.39 while r2 ranged from 0.00 to 0.94.
Table 3.
Summary Data for Non-Invasive Index E/Vp
| Group (n = subjects) | Paired Data Points (E/Vp & PCWP) | Group Random Coefficient Mixed Model Regression | p Value | Range of Individual Linear Regression Slopes Within Group | Range of Individual r2 Within Group | ||
|---|---|---|---|---|---|---|---|
| Average | (Min - Max) | Average | (Min - Max) | ||||
| HFpEF (n=11) | 59 | PCWP = 2.51*E/Vp + 9.02 | 0.16 | 4.70 | (-16.33 - 24.65) | 0.41 | (0.02 - 0.94) |
| Healthy Elderly (n=24) | 135 | PCWP =1.64*E/Vp +7.54 | 0.06 | 4.40 | (-12.54 - 25.39) | 0.27 | (0.00 - 0.66) |
| Healthy Young (n=11) | 62 | PCWP = 7.07*E/Vp - 1.90 | 0.003 | 3.89 | (-16.42 - 20.04) | 0.30 | (0.00 - 0.87) |
| All Subjects (n=46) | 256 | PCWP = 1.94*E/Vp + 7.48 | 0.005 | 4.35 | (-16.42 - 25.39) | 0.31 | (0.00 - 0.94) |
Figure 5.
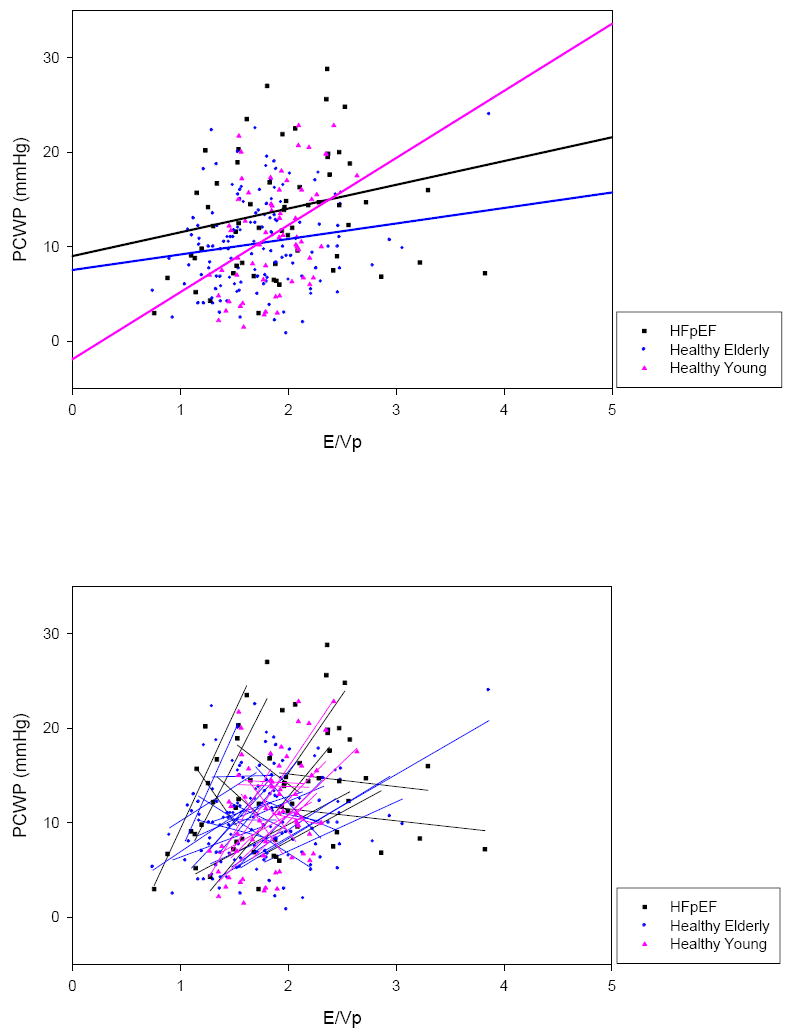
Composite of E/Vp vs. PCWP in all subjects. Group random coefficient mixed model regressions (top) and individual linear regressions (bottom) are superimposed. Again, individual linear regressions were highly variable in regards to slope and r2.
In healthy subjects, group random coefficient mixed model regression slopes ranged from 1.64 in the elderly group to 7.07 in the young group; in HFpEF patients, this group regression slope was 2.51. Once again, within the individual subjects who comprised these groups, linear regression slopes ranged from steeply negative to steeply positive and r2 ranged from low to high. Based on linear regression slopes, in 12 healthy subjects changes in E/Vp and PCWP were discordant whereas in the other 24, changes were concordant; in HFpEF patients these numbers were 5 and 6, respectively.
Discussion
The key finding of this study is that as left-sided filling pressures are manipulated in healthy individuals or in outpatients with HFpEF, the relationship between non-invasive indices (E/e’ and E/Vp) and the PCWP is highly variable, with individual subject linear regression slopes ranging from steeply negative to steeply positive and coefficients of determination ranging from very low to very high. Consequently, non-invasive indices do not adequately track changes in left-sided filling pressures as these pressures vary within individual subjects.
Limitations of Non-Invasive Indices in the One-Time Estimation of Left-Sided Filling Pressures
There are several known limitations of E/e’ and E/Vp in the one-time estimation of left-sided filling pressures. Published linear regressions relating E/e’ and E/Vp with invasive measures are generally derived from a composite of data points from individual subjects, each data point representing a single, simultaneous measure of the non-invasive index and PCWP. As each subject contributes only one paired measurement to the composite, these regressions consequently reflect the relationship for the population rather than for the individual subjects which comprise the population. For this reason population based linear regressions often poorly predict left-sided filling pressures in individual subjects as reflected by their wide confidence intervals. Furthermore, the utility of non-invasive estimates of left sided filling pressure appears to be highly dependent on the population studied. While non-invasive indices have been validated in populations of healthy subjects as well as in select patients depressed systolic heart failure 2, cardiac transplantation 4, atrial fibrillation 5, sinus tachycardia 6 and hypertrophic cardiomyopathy 7, others have shown that they fail to accurately estimate left-sided filling pressures in patients with mitral valve disease 8, 10, symptomatic hypertrophic cardiomyopathy 11 and decompensated advanced systolic heart failure 12.
Non-Invasive Indices Do Not Accurately Track Changes in Left-Sided Filling Pressures
Despite these limitations, some have suggested that serial measures of non-invasive indices might allow for the estimation of directional changes in left-sided filling pressures in response to therapy or worsening of heart failure within an individual subject. This thought is based on the presumption that changes in non-invasive indices should track changes in invasive measures within individuals in accordance with population based regressions 4, 7, 13. Our data refute this presumption. The group means (of E/e’ and E/Vp) tended to track preload manipulation well (Figure 1) and our population-based random coefficient mixed model regressions suggested a weak but significant relationship between PCWP and E/e’ and PCWP and E/Vp (Figures 2, 4). However, as filling pressures were manipulated in the individual subjects who comprised these groups, significant variability was observed within individual subjects with regression slopes ranging from steeply negative to steeply positive and coefficients of determination ranging from very low to very high. This broad variability demonstrates that E/e’ and E/Vp are not able to accurately track changes in left-sided filling pressures in individual patients.
To illustrate this point, our HFpEF group’s baseline linear regression (PCWP = 0.52×E/e’ + 6.85 [p=0.04, r2 = 0.42, SE = 1.99]; Figure 3) is similar to previously published regressions with the exception of a lower standard error of the estimate (SE), which has ranged from 3.4 7 to 3.9 mmHg 1, 6 in prior studies. Yet despite this favorable group baseline regression, as filling pressures were manipulated within the individual HFpEF patients who comprised the group, E/e’ responded unpredictably with individual linear regressions varying profoundly in terms of slope (patient slopes: -1.44, -1.34, -0.68, 0.19, 1.55, 1.73, 2.45, 2.65, 2.69, 3.83, 4.42) and coefficient of determination (range: 0.00 – 0.82). Examples of the primary data for PCWP, E and e’ in two HFpEF patients are presented in Figure 6. In the top example, PCWP and E/e’ were concordant (the patient with a slope of 4.42) while in the bottom example, the two were discordant (the patient with a slope of -1.44). These examples illustrate the broad individual variability observed with this index as preload is manipulated. Similar findings were observed between PCWP and E/Vp in these HFpEF patients and between PCWP and E/e’ and PCWP and E/Vp in our healthy subjects.
Figure 6.
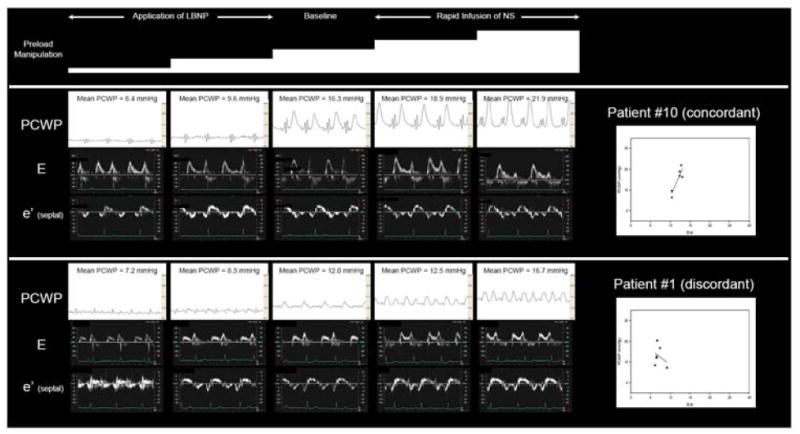
Effects of preload manipulation on PCWP, E, e’ and E/e’ slope in two HFpEF patients. In the top example, increasing PCWP results in a positive E/e’ slope demonstrating concordance between these measures. In the bottom example, increasing PCWP results in a negative E/e’ slope demonstrating discordance. In 8/11 HFpEF patients, PCWP and E/e’ were concordant whereas in the other 3, the two measures were discordant. Mean PCWP was determined during the period of diastasis, thereby minimizing the impact of large V waves on the mean PCWP observed in the top example.
To better understand the limitations of E/e’ as an index to track changes in left-sided filling pressures, individual linear regression slopes for E vs. PCWP and e’ vs. PCWP were examined in HFpEF patients. As demonstrated in Figure 4, E showed a degree of preload dependence that was generally similar among individual HFpEF patients. Conversely, the response of e’, which also demonstrated a degree of preload dependence, was highly variable between individual HFpEF patients with steeply elevated slopes in 5 patients and more mildly elevated slopes in the other 6. This degree of preload dependence contrasts prior reports of the preload independence of e’ 1 falling more in line with reports demonstrating a varying strength of relationship between e’ and left sided filling pressures 3, 6, 7, 23. Of the two measures, the broad variability observed in the denominator of E/e’ would appear to primarily account for the broad variability observed in individual E/e’ slopes. As a collective, these observations illustrate the point that the inter-individual slope of the preload manipulated E/e’ relationship is so highly variable that it is impossible to reliably track changes in filling pressures in a given individual without first establishing the slope of the relationship in that given individual. This is, of course, impractical to perform in patients with clinical heart failure and hence, demonstrates an important limitation of this index.
A limited number of studies have previously attempted to assess the feasibility of serial measures of non-invasive indices to track changes in left-sided filling pressures associated with medical therapy or exercise. Mullens et al examined the ability of the non-invasive index E/Ea to track changes in filling pressures between admission and the 48 hour time point in 51 patients admitted to the intensive care unit with advanced decompensated heart failure. Over the course of these 48 hours, the change in PCWP ranged from -24 to 16 mmHg; however, the authors found no correlation between absolute changes in the E/Ea ratio and PCWP 12. More recently, Maeder et al demonstrated that E/e’ failed to reflect PCWP in exercising patients with heart failure with normal ejection fraction 24. In another study, Dokainish et al correlated the change in E/E’ with the change in PCWP in 9 patients hospitalized in the intensive care unit before and after 48 hours of treatment with intravenous diuretics (n=5) and/or ionotropes (n=8) 13. While these authors did show a correlation between change in PCWP and change in E/E’ based on the group’s composite data, the linear regression slopes of the individual subjects were not presented. Additional studies have also reported correlations between changes in PCWP and changes in E/E’ 4, 7; however, again, the linear regression slopes of the individual subjects who made up these populations were not presented, calling into question the ability of these population based indices to accurately predict changes in left-sided filling pressures within individual subjects.
Study Limitations
Although great care was taken to obtain all echocardiographic measures in traditional imaging planes, subtle variations are often unavoidable and occasionally, representative measures could not obtained or interpreted. Overall, subtle variation in imaging planes was felt to be a random phenomenon which was equally distributed amongst all measures obtained. There were few instances of unobtainable or uninterpretable measures. For E/e’, only 15 of the 282 possible measures (5%) could not be obtained or interpreted; for E/Vp, this number was 20 of the 276 possible measures (7%). Finally, we used PCWP as a measure of left-sided filling pressures rather than directly measured left atrial pressure (LAP). However previous studies have shown that in the absence of anatomic obstruction between the pulmonary capillary and the left ventricle, PCWP correlates closely with left ventricular end-diastolic pressure and is widely used as a surrogate for LAP 23, 24.
Conclusion
As filling pressures are manipulated, the relationship between non-invasive indices (E/e’ and E/Vp) and the pulmonary capillary wedge pressure is highly variable in healthy patients and patients with HFpEF, with individual subject linear regression slopes ranging from steeply negative to steeply positive and coefficients of determination ranging from very low to very high. Consequently, within individual subjects, non-invasive indices do not reliably track changes in left-sided filling pressures, precluding the use of these techniques in research studies with healthy volunteers or to titrate medical therapy in patients with HFpEF.
Acknowledgments
We would like to acknowledge our research team for the invaluable contributions: Mrs. Diane Bedenkop, Mr. Colin Connor, Mr. Daniel Creson, Mrs. Peggy Fowler, Mr. Cyrus Oufi, Ms. Tiffany VanGundy and Dr. Murugappan Ramanathan.
Sources of Funding This project was supported by the NIH (Grant R01 AG17479). Additional support for statistical analyses was provided by the National Institutes of Health Clinical and Translational Science Award (Grant UL1 RR024982).
Abbreviations
- CHF
congestive heart failure
- EF
ejection fraction
- E/e’
the ratio of the peak early mitral inflow velocity over the mean of the early diastolic mitral annular velocity as measured at the septal and lateral mitral annulus
- E/Vp
the ratio of the peak early mitral inflow velocity over the color M-mode Doppler flow propagation velocity
- HFpEF
heart failure with preserved ejection fraction
- LAP
left atrial pressure
- LBNP
lower body negative pressure
- PCWP
pulmonary capillary wedge pressure
- TDI
tissue Doppler imaging
- r2
coefficient of determination
- SE
standard error of the estimate
Footnotes
Disclosures None.
Contributor Information
Paul S. Bhella, John Peter Smith Health Network, Fort Worth, TX.
Eric L. Pacini, Univ of Texas-Southwestern, Dallas, TX.
Anand Prasad, Univ of Texas-Southwestern, Dallas, TX.
Jeffrey L. Hastings, Univ of Texas-Southwestern, Dallas, TX.
Beverley Adams-Huett, Univ of Texas-Southwestern, Dallas, TX.
James D. Thomas, The Cleveland Clinic, Cleveland, OH.
Paul A. Grayburn, Baylor University Medical Center, Dallas, TX.
Benjamin D. Levine, Univ of Texas-Southwestern, Dallas, TX.
References
- 1.Nagueh SF, Middleton KJ, Kopelen HA, Zoghbi WA, Quiñones MA. Doppler tissue imaging: A noninvasive technique for evaluation of left ventricular relaxation and estimation of filling pressures. Journal of the American College of Cardiology. 1997;30:1527–1533. doi: 10.1016/s0735-1097(97)00344-6. [DOI] [PubMed] [Google Scholar]
- 2.Garcia M, Ares M, Asher C, Rodriguez L, Vandervoort P, Thomas J. An index of early left ventricular filling that combined with pulsed doppler peak e velocity may estimate capillary wedge pressure. Journal of the American College of Cardiology. 1997;29:448–454. doi: 10.1016/s0735-1097(96)00496-2. [DOI] [PubMed] [Google Scholar]
- 3.Firstenberg MS, Levine BD, Garcia MJ, Greenberg NL, Cardon L, Morehead AJ, Zuckerman J, Thomas JD. Relationship of echocardiographic indices to pulmonary capillary wedge pressures in healthy volunteers. Journal of the American College of Cardiology. 2000;36:1664–1669. doi: 10.1016/s0735-1097(00)00909-8. [DOI] [PubMed] [Google Scholar]
- 4.Sundereswaran L, Nagueh SF, Vardan S, Middleton KJ, Zoghbi WA, Quiñones MA, Torre-Amione G. Estimation of left and right ventricular filling pressures after heart transplantation by tissue doppler imaging. The American Journal of Cardiology. 1998;82:352–357. doi: 10.1016/s0002-9149(98)00346-4. [DOI] [PubMed] [Google Scholar]
- 5.Sohn D-W, Song J-M, Zo J-H, Chai I-H, Kim H-S, Chun H-G, Kim H-C. Mitral annulus velocity in the evaluation of left ventricular diastolic function in atrial fibrillation. J Am Soc Echocardiogr. 1999;12:927–931. doi: 10.1016/s0894-7317(99)70145-8. [DOI] [PubMed] [Google Scholar]
- 6.Nagueh SF, Mikati I, Kopelen HA, Middleton KJ, Quinones MA, Zoghbi WA. Doppler estimation of left ventricular filling pressure in sinus tachycardia : A new application of tissue doppler imaging. Circulation. 1998;98:1644–1650. doi: 10.1161/01.cir.98.16.1644. [DOI] [PubMed] [Google Scholar]
- 7.Nagueh SF, Lakkis NM, Middleton KJ, Spencer WH, III, Zoghbi WA, Quinones MA. Doppler estimation of left ventricular filling pressures in patients with hypertrophic cardiomyopathy. Circulation. 1999;99:254–261. doi: 10.1161/01.cir.99.2.254. [DOI] [PubMed] [Google Scholar]
- 8.Bruch C, Stypmann J, Gradaus R, Breithardt G, Wichter T. Usefulness of tissue doppler imaging for estimation of filling pressures in patients with primary or secondary pure mitral regurgitation. The American Journal of Cardiology. 2004;93:324–328. doi: 10.1016/j.amjcard.2003.10.012. [DOI] [PubMed] [Google Scholar]
- 9.Paulus WJ, Tschöpe C, Sanderson JE, Rusconi C, Flachskampf FA, Rademakers FE, Marino P, Smiseth OA, De Keulenaer G, Leite-Moreira AF, Borbãly A, Ãdes IN, Handoko ML, Heymans S, Pezzali N, Pieske B, Dickstein K, Fraser AG, Brutsaert DL. How to diagnose diastolic heart failure: A consensus statement on the diagnosis of heart failure with normal left ventricular ejection fraction by the heart failure and echocardiography associations of the european society of cardiology. Eur Heart J. 2007;28:2539–2550. doi: 10.1093/eurheartj/ehm037. [DOI] [PubMed] [Google Scholar]
- 10.Diwan A, McCulloch M, Lawrie GM, Reardon MJ, Nagueh SF. Doppler estimation of left ventricular filling pressures in patients with mitral valve disease. Circulation. 2005;111:3281–3289. doi: 10.1161/CIRCULATIONAHA.104.508812. [DOI] [PubMed] [Google Scholar]
- 11.Geske JB, Sorajja P, Nishimura RA, Ommen SR. Evaluation of left ventricular filling pressures by doppler echocardiography in patients with hypertrophic cardiomyopathy: Correlation with direct left atrial pressure measurement at cardiac catheterization. Circulation. 2007;116:2702–2708. doi: 10.1161/CIRCULATIONAHA.107.698985. [DOI] [PubMed] [Google Scholar]
- 12.Mullens W, Borowski AG, Curtin RJ, Thomas JD, Tang WH. Tissue doppler imaging in the estimation of intracardiac filling pressure in decompensated patients with advanced systolic heart failure. Circulation. 2009;119:62–70. doi: 10.1161/CIRCULATIONAHA.108.779223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dokainish H, Zoghbi WA, Lakkis NM, Al-Bakshy F, Dhir M, Quinones MA, Nagueh SF. Optimal noninvasive assessment of left ventricular filling pressures: A comparison of tissue doppler echocardiography and b-type natriuretic peptide in patients with pulmonary artery catheters. Circulation. 2004;109:2432–2439. doi: 10.1161/01.CIR.0000127882.58426.7A. [DOI] [PubMed] [Google Scholar]
- 14.McKee PA, Castelli WP, McNamara PM, Kannel WB. The natural history of congestive heart failure: The framingham study. N Engl J Med. 1971;285:1441–1446. doi: 10.1056/NEJM197112232852601. [DOI] [PubMed] [Google Scholar]
- 15.Drazner MH, Prasad A, Ayers C, Markham DW, Hastings J, Bhella PS, Shibata S, Levine BD. The relationship of right- and left-sided filling pressures in patients with heart failure and a preserved ejection fraction. Circ Heart Fail. 2010;3:202–206. doi: 10.1161/CIRCHEARTFAILURE.108.876649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Prasad A, Hastings JL, Shibata S, Popovic ZB, Arbab-Zadeh A, Bhella PS, Okazaki K, Fu Q, Berk M, Palmer D, Greenberg NL, Garcia MJ, Thomas JD, Levine BD. Characterization of static and dynamic left ventricular diastolic function in patients with heart failure with a preserved ejection fraction / clinical perspective. Circulation: Heart Failure. 2010;3:617–626. doi: 10.1161/CIRCHEARTFAILURE.109.867044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Arbab-Zadeh A, Dijk E, Prasad A, Fu Q, Torres P, Zhang R, Thomas JD, Palmer D, Levine BD. Effect of aging and physical activity on left ventricular compliance. Circulation. 2004;110:1799–1805. doi: 10.1161/01.CIR.0000142863.71285.74. [DOI] [PubMed] [Google Scholar]
- 18.Prasad A, Popovic ZB, Arbab-Zadeh A, Fu Q, Palmer D, Dijk E, Greenberg NL, Garcia MJ, Thomas JD, Levine BD. The effects of aging and physical activity on doppler measures of diastolic function. Am J Cardiol. 2007;99:1629–1636. doi: 10.1016/j.amjcard.2007.01.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Levine BD, Lane LD, Buckey JC, Friedman DB, Blomqvist CG. Left ventricular pressure-volume and frank-starling relations in endurance athletes. Implications for orthostatic tolerance and exercise performance. Circulation. 1991;84:1016–1023. doi: 10.1161/01.cir.84.3.1016. [DOI] [PubMed] [Google Scholar]
- 20.Prasad A, Okazaki K, Zadeh AA, Dijk E, Fu Q, Thomas JD, Levine BD. Abnormalities of doppler measures of diastolic function in the healthy elderly are not related to alterations of left atrial pressure. Circulation. 2005;111:1499–1503. doi: 10.1161/01.CIR.0000159353.31271.F2. [DOI] [PubMed] [Google Scholar]
- 21.Sohn D-W, Chai I-H, Lee D-J, Kim H-C, Kim H-S, Oh B-H, Lee M-M, Park Y-B, Choi Y-S, Seo J-D, Lee Y-W. Assessment of mitral annulus velocity by doppler tissue imaging in the evaluation of left ventricular diastolic function. Journal of the American College of Cardiology. 1997;30:474–480. doi: 10.1016/s0735-1097(97)88335-0. [DOI] [PubMed] [Google Scholar]
- 22.Popovic ZB, Prasad A, Garcia MJ, Arbab-Zadeh A, Borowski A, Dijk E, Greenberg NL, Levine BD, Thomas JD. Relationship among diastolic intraventricular pressure gradients, relaxation, and preload: Impact of age and fitness. Am J Physiol Heart Circ Physiol. 2006;290:H1454–1459. doi: 10.1152/ajpheart.00902.2005. [DOI] [PubMed] [Google Scholar]
- 23.Nagueh SF, Sun H, Kopelen HA, Middleton KJ, Khoury DS. Hemodynamic determinants of the mitral annulus diastolic velocities by tissue doppler. J Am Coll Cardiol. 2001;37:278–285. doi: 10.1016/s0735-1097(00)01056-1. [DOI] [PubMed] [Google Scholar]
- 24.Maeder MT, Thompson BR, Brunner-La Rocca H-P, Kaye DM. Hemodynamic basis of exercise limitation in patients with heart failure and normal ejection fraction. J Am Coll Cardiol. 2010;56:855–863. doi: 10.1016/j.jacc.2010.04.040. [DOI] [PubMed] [Google Scholar]


