Abstract
Objective
The aim of this study was to determine the effect of DRIs on hot flash symptoms in menopausal women.
Design
This was a randomized, double-blind, placebo-controlled trial of menopausal women, aged 38 to 60 years, who experienced 4 to 14 hot flashes per day. After a 1-week run-in period, a total of 190 menopausal women were randomized to receive a placebo or 40 or 60 mg/day of a DRI for 12 weeks. The primary outcome was the mean changes from baseline to week 12 in the frequency of hot flashes recorded in the participant diary. The secondary outcomes included changes in quality of life and hormonal profiles.
Results
A total of 147 women (77%) completed the study. It was found that 40 and 60 mg of DRI improved hot flash frequency and severity equally. At 8 weeks hot flash frequency was reduced by 43% in the 40-mg DRI group and by 41% in the 60-mg DRI group, compared with 32% in the placebo group (P = not significant vs placebo). The corresponding numbers for 12 weeks were 52%, 51%, and 39%, respectively (P = 0.07 and 0.09 vs placebo). When comparing the two treatment groups with the placebo group, there were significant reductions in mean daily hot flash frequency. The supplement (either 40 or 60 mg) reduced hot flash frequency by 43% at 8 weeks (P = 0.1) and 52% at 12 weeks (P = 0.048) but did not cause any significant changes in endogenous sex hormones or thyroid hormones. Menopausal quality of life improved in all three groups, although there were no statistically significant differences between groups.
Conclusions
DRI supplementation may be an effective and acceptable alternative to hormone treatment for menopausal hot flashes.
Keywords: Hot flashes, Menopausal symptoms, Soybean germ, Isoflavones, Aglycones, Menopause
Hot flashes occur in approximately 75% of menopausal women in Western countries and are the main reason women entering menopause seek treatment. They are thought to be due to thermoregulatory dysfunction caused by estrogen withdrawal and initiated at the level of the hypothalamus.1 Hot flashes have been shown to critically affect quality of life, including work ability, social life, daily activities, and the perception of health. In one study hot flashes were correlated with an exacerbation of other symptoms, such as depression, insomnia, and joint pain.2
Although hormone therapy (HT) is the most effective treatment for the relief of hot flashes to date, evidence suggests that long-term use of HT may increase the risk of developing certain medical disorders. The data from the Women's Health Initiative showed an increased risk of coronary heart disease, stroke, pulmonary embolism, and breast cancer with estrogen + progestogen therapy (conjugated equine estrogens + medroxyprogesterone acetate) after a mean of 5.2 years of follow-up.3 Estrogen+ progestogen therapy was associated with a 24% increased risk of coronary heart disease.4 Thus, safe and effective alternatives are needed. Recently interest has arisen in isoflavones as a potential therapy for menopausal hot flashes.
Isoflavones are one of several classes of phytoestrogens, compounds that exert both estrogenic and antiestrogenic properties. Daidzein and genistein are isoflavones that are found in rich supply in soybeans and soy products. Available data on isoflavones for the treatment of menopausal symptoms have been inconclusive; studies with positive results have reported only a slight improvement. Nonetheless, there are no studies showing harmful effects of isoflavones on menopausal symptoms. This inconsistency may, in part, be due to differences in methods used to isolate isoflavones, concentrations of bioavailable isoflavones, and the composition of isoflavones used. In this study we used an isoflavone-rich supplement made from soy germ. The supplement is rich in daidzein, a precursor of equol, which also has estrogenic properties.
In our nonrandomized, open-label pilot study, we found that 40 to 60 mg of daidzein-rich isoflavone aglycones (DRIs) improved the hot flash composite score (hot flash frequency × hot flash severity) by 54% at 8 weeks.5 In the present study we conducted a randomized, double-blind, placebo-controlled study to evaluate the changes in menopausal symptoms in response to DRI intake of 40 or 60 mg/day for 12 weeks.
METHODS
Two hundred thirty-five women were recruited from referring physicians in the Beth Israel Deaconess Medical Center and from newspaper advertisements. Of those, 191 women met inclusion criteria and were randomized. The inclusion criteria were being postmenopausal with no menses for at least the previous 6 months, between the ages of 38 and 60, and experiencing hot flashes four or more times per day but 15 times or less per day. Women were excluded if they were active smokers (more than one pack of cigarettes per day); had taken dietary supplements containing soy isoflavones, vitamin E, flaxseed, or red clover within the past 6 weeks; or were on HT or any medications for treatment of hot flashes (clonidine, selective serotonin reuptake inhibitors) within the past 6 weeks. Women were also excluded if they had a body mass index greater than 40 kg/m2; had a history of breast, endometrial, or cervical cancer; had a positive pregnancy test; or had a history of undiagnosed abnormal vaginal bleeding, thromboembolic disease, cardiovascular disease, liver or kidney disease, diabetes mellitus, or other major illnesses. The Institutional Review Board of the Beth Israel Deaconess Medical Center approved the study protocol, and all participants gave written informed consent.
The study took place over 13 weeks in an ambulatory care setting. At the initial visit a detailed medical history and a menstrual and menopausal history were completed. A physical examination including height, weight, and breast and pelvic examinations was performed. Blood was obtained for hematology (complete blood count) and serum chemistries (aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, bilirubin, blood urea nitrogen, creatinine, glucose, and electrolytes). Urine was obtained for a pregnancy test. The Menopause-Specific Quality of Life Questionnaire6 and a questionnaire on current soy intake were completed. The Menopause Quality of Life summary score was calculated. Women were then instructed to record their hot flash frequency and severity in a provided daily diary for the duration of the study. Using a 10-point scale (1 = mild to 10 = very severe), women recorded hot flash severity twice per day (the average severity of hot flashes experienced during the morning and again in the evening) and an approximate number of hot flashes for the entire day. The 10-point severity scale was first translated into a 4-point scale (1, 2, 3 = 1; 4, 5, 6 = 2; 7, 8, 9 = 3; 10 = 4). Daily hot flash composite scores (average hot flash severity × hot flash frequency) were also averaged to produce a single baseline score for each participant.7 The women were not given any supplements during this 1-week run-in period.
At the second visit women returned their run-in week hot flash diaries, and those who fulfilled the entry criteria were included. Blood was obtained for baseline fasting total cholesterol, triglycerides, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, and isoflavone levels. In addition, several hormone levels were measured, including estradiol (third-generation estradiol enzyme immunoassay), luteinizing hormone, follicle-stimulating hormone, sex hormone–binding globulin, estrone, insulin, leptin, thyroid-stimulating hormone, thyroglobulin, free and total triiodothyronine, and free and total thyroxine. All hormone levels were measured by enzyme-linked immunosorbent assays (Diagnostic System Laboratories, Inc, Webster, TX).
Extraction and analysis of isoflavonoids as aglycones from serum samples after enzymatic hydrolysis of conjugates followed a previously applied high-performance liquid chromatography protocol with modifications.8,9 In brief, 1 mL of plasma equilibrated to room temperature was mixed with 250 μL of 1 M NH4Ac buffer, 10 μL of β-glucuronidase (10 U/mL), 40 μL of arylsulfatase (5 U/mL), and 50 μL of estriol as internal standard, and the mixture was incubated at 37°C for 16 hours. After addition of 100 μL of glacial acid, the sample was extracted with 5 mL of hexane to remove fat content. The remaining sample was extracted three times with 2 mL of diethyl ether. The diethyl ether extracts were combined and evaporated to dryness under nitrogen gas, and the residue was redissolved in 100 μL of methanol/water (80:20, vol/vol). After centrifugation, 25 μL of clear sample supernatants was injected into the high-performance liquid chromatography system. The reverse-phase high-performance liquid chromatography system (Waters 2695 Separation Module) with 2996 Photodiode Array Detector and a C18 reverse-phase column (Phenomenex Ultremex 5, 250 × 4.6 mm) were used.
A second menopause quality of life questionnaire was also completed at the second visit. All women were then randomized to placebo or 40 or 60 mg of a DRI every morning. This DRI (AglyMax, Nichimo Co, Ltd, Shinagawa, Tokyo, Japan) is a concentrated isoflavone product prepared from soybean germ fermentation with Koji fungus (Aspergliius awamori), producing β-glycosidase efficiency, followed by ethanol and water extraction and purification using a proprietary extraction procedure. The product is rich in daidzein (70% daidzein, 10% genistein, and 20% glycitein). The women were instructed to take a single soft-gel capsule daily that contained either 0 (placebo), 40, or 60 mg.
Women returned to the clinic after 4, 8, and 12 weeks on the supplement. At each visit women completed the Menopause-Specific Quality of Life survey, hot flash diaries were reviewed, and vital signs and adverse events were monitored. Blood samples were drawn for a lipid panel and hormone levels at every clinic visit and at 12 weeks for isoflavone levels and routine chemistry.
Statistical methods
Data from the placebo arms of the previous hot flash studies indicated that differences in hot flash activity between baseline and the end of the treatment period had an SD of two hot flashes and five score units per patient per day.7 The sample size of 50 women per group provided 80% power to detect differences in average hot flash activity of 0.58 SD via a standard two-sample t test using a two-sided type I error rate of 5%. Hence, 50 women with complete data per treatment arm provide 80% power to detect an average shift of 1.2 hot flashes per day or a hot flash score of three units per day. For the generation of the randomization list, a permuted block algorithm for the three treatments in a 1:1:1 ratio was used. Stratification was not used in this study. Study supplement bottles had unique numbers to allow double-blind dispensing.
The primary measure of efficacy was the percentage of change in hot flash frequency recorded in daily diaries. Frequency and severity were analyzed separately as well as together as a hot flash composite score. The SAS statistical package (SAS Institute, Inc, Cary, NC) and the Minitab statistical package (version 14.2, Minitab, State College, PA) for personal computers were used for statistical analysis. The analysis for the percentage of change in hot flashes was based on the two-way fixed-effect analysis of variance model. Repeated-measures analysis was performed to determine whether there was a significant treatment effect of the reduction in hot flash frequency over time. Correlations between changes in hot flash frequency and score and serum isoflavone levels were studied by linear regression. Data are presented as mean ± SD. A P value less than 0.05 was considered significant.
RESULTS
The demographic characteristics of the women enrolled in the study are summarized in Table 1. A total of 147 women completed 12 weeks of supplementation. At baseline there were no significant differences among women in body mass index or hot flash activity. The main reason for withdrawal from the study was that the women were either unable to comply with the study protocol or were lost to follow-up (n = 30). Eight women withdrew because they started taking medications or other supplements that might affect hot flashes (including HT, selective serotonin reuptake inhibitors, and vitamin E). Two women resumed their menses after randomization and were withdrawn from the study. Two women were randomized but then withdrawn early because of abnormal findings on baseline liver function tests. Two women in the 60-mg DRI group discontinued supplement use because of gastrointestinal side effects (one stomach upset and one diarrhea). Five women did not complete their hot flash diaries and were also excluded from analysis. A total of 142 women had analyzable data. Of those who completed the study, 119 were postmenopausal and 28 women were perimenopausal. Sixty-three women had previous exposure to HT with the average time between last HT use and time of enrollment in the study of 1.5 years. Women who did not complete the study were younger than those who completed the study (51.0 ± 4.8 vs 53.5 ± 5.2 y; P = 0.002). However, there were no differences in terms of body mass index, number of years after menopause, previous use of HT, or baseline hot flash activity between women who completed or did not complete the study.
TABLE 1.
Baseline characteristics of study participants
| Placebo (n = 45) | 40 mg DRI (n = 48) | 60 mg DRI (n = 49) | |
|---|---|---|---|
| Age, y (mean ± SD) | 53.8 ± 5.1 | 52.2 ± 4.8 | 53.2 ± 5.6 |
| Postmenopause status, y (mean ± SD) | 5.6 ± 5.9 | 4.0 ± 4.9 | 5.8 ± 6.1 |
| BMI, kg/m2 | 27.4 ± 4.1 | 29.9 ± 6.1 | 28.3 ± 5.2 |
| Alcohol, drinks/wk | 1.7 ± 2.6 | 1.8 ± 3.2 | 1.7 ± 2.7 |
| Hot flash severity | 2.1 ± 0.6 | 2.2 ± 0.6 | 2.1 ± 0.6 |
| Hot flash frequency | 7.3 ± 2.5 | 8.5 ± 3.5 | 7.6 ± 2.6 |
| Hot flash score | 15.9 ± 8.4 | 20.2 ± 12.2 | 16.3 ± 8.0 |
| Race, % | |||
| White | 66 | 60 | 60 |
| African-American | 24 | 36 | 30 |
| Other | 10 | 4 | 10 |
| Education, % | |||
| Postgraduate | 28 | 25 | 28 |
| College | 53 | 56 | 59 |
| High school | 19 | 19 | 13 |
| Smokers (n) | 11 | 9 | 6 |
Values are means ± SD. There was no significant difference among the three groups at baseline.
DRI, daidzein-rich isoflavone aglycone; BMI, body mass index.
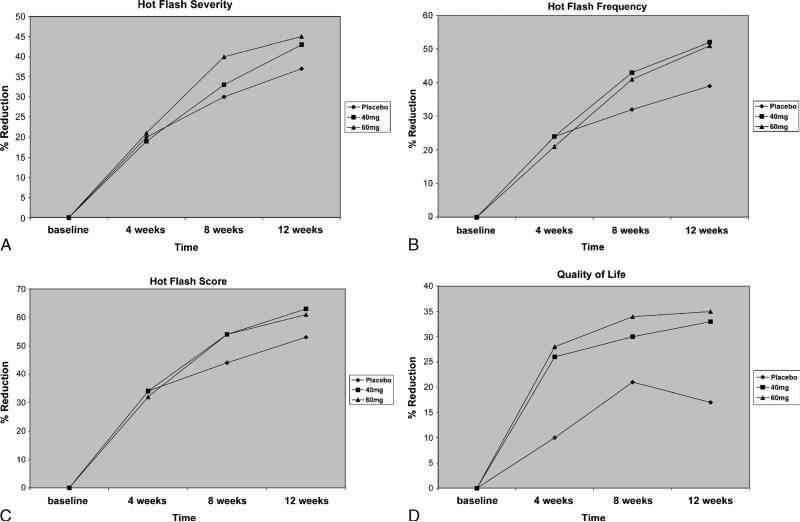
Our results showed that 12-week supplementation with a DRI significantly reduced hot flash frequency. Hot flash frequency decreased by 43% in the 40-mg DRI group and by 41% in the 60-mg DRI group at 8 weeks, compared with 32% in the placebo group (P = not significant). At 12 weeks 40 and 60 mg of DRI produced 52% and 51%, respectively, reductions in hot flash frequency, compared with 39% in the placebo group (P = 0.07 and 0.09) (Fig. 1). When comparing the two treatment groups together as one group (DRI supplement) against placebo, there were significant reductions in mean daily hot flash frequency. The supplement reduced hot flash frequency by 43% at 8 weeks (P = 0.1) and 52% at 12 weeks (P = 0.048). When perimenopausal and menopausal women were analyzed separately, we did not find significant differences between the treatment groups and placebo. This is probably due to small sample sizes. Menopausal quality of life as measured by the Menopause Quality of Life summary score showed that women reported significant decreases of menopausal symptoms from baseline in all three groups, but there were no statistically significant differences between groups. All women reported no major side effects. The main side effects of the DRI were gastrointestinal in nature, including stomach upset (one woman), constipation (two women), flatulence (two women), and diarrhea (one woman), although only two women on 60 mg of DRI withdrew from the study because of gastrointestinal side effects. Serum chemistry levels, including liver function tests, fasting blood glucose levels, electrolytes, blood urea nitrogen, creatinine, and complete blood counts, were all within normal limits at the end of the study.
FIG. 1.
A: Percentage of decrease in hot flash severity from baseline at 4, 8, and 12 weeks. B: Percentage of decrease in hot flash frequency from baseline at 4, 8, and 12 weeks. C: Percentage of decrease in hot flash composite score from baseline at 4, 8, and 12 weeks. D: Percentage of decrease in menopausal symptoms from baseline at 4, 8, and 12 weeks.
To assess the effect of isoflavone supplementation on cardiovascular risk profiles, fasting blood glucose, total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, triglycerides, blood pressure, and body weight were analyzed. During 12 weeks of supplement use, women had no statistically significant changes in their blood pressure, body weight, or lipid, glucose, leptin, or insulin levels as compared with the control group (Tables 2 and 3).
TABLE 2.
Cardiovascular risk factors
| Placebo |
40 mg DRI |
60 mg DRI |
||||
|---|---|---|---|---|---|---|
| Baseline | 12 wk | Baseline | 12 wk | Baseline | 12 wk | |
| Total cholesterol, mg/dL | 214 ± 42 | 227 ± 86 | 214 ± 42 | 215 ± 44 | 213 ± 34 | 210 ± 34 |
| Triglycerides, mg/dL | 99 ± 62 | 116 ± 105 | 115 ± 61 | 113 ± 61 | 108 ± 67 | 113 ± 71 |
| HDL cholesterol, mg/dL | 74 ± 17 | 74 ± 20 | 68 ± 19 | 69 ± 17 | 71 ± 22 | 71 ± 20 |
| LDL cholesterol, mg/dL | 120 ± 37 | 119 ± 31 | 123 ± 38 | 123 ± 41 | 120 ± 33 | 117 ± 35 |
| Glucose, mg/dL | 88 ± 15 | 86 ± 9 | 88 ± 19 | 92 ± 17 | 88 ± 14 | 87 ± 13 |
| SBP, mm Hg | 120 ± 13 | 117 ± 15 | 122 ± 14 | 119 ± 13 | 121 ± 15 | 116 ± 15 |
| DBP, mm Hg | 76 ± 9 | 74 ± 10 | 76 ± 10 | 75 ± 11 | 78 ± 10 | 74 ± 11 |
| Weight, kg | 73.4 ± 12 | 72.6 ± 12 | 79.1 ± 17 | 78.9 ± 17 | 75.9 ± 14 | 75.4 ± 14 |
| BMI, kg/m2 | 27.4 ± 4.1 | 27.1 ± 4.3 | 29.9 ± 6.1 | 29.9 ± 6.3 | 28.3 ± 5.2 | 28.1 ± 5.2 |
Values are means ± SD. There was no significant difference among the three groups at baseline and at 12 weeks. DRI, daidzein-rich isoflavone aglycone; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SBP, systolic blood pressure; DBP, diastolic blood pressure; BMI, body mass index.
TABLE 3.
Quantification of female sex hormones, thyroid hormones, leptin, and insulin
| Hormones | Baseline | 4 wk | 8 wk | 12 wk |
|---|---|---|---|---|
| LH, mIU/mL | ||||
| Placebo | 44.5 ± 18.0 | 45.4 ± 18.9 | 44.4 ± 18.2 | 44.6 ± 17.5 |
| 40 mg DRI | 39.8 ± 28.1 | 38.4 ± 31.0 | 37.2 ± 28.3 | 36.8 ± 27.9 |
| 60 mg DRI | 45.5 ± 22.5 | 45.1 ± 23.9 | 41.8 ± 25.3 | 43.8 ± 21.0 |
| Estrone, pg/mL | ||||
| Placebo | 173.8 ± 115.4 | 182.5 ± 129.7 | 186.8 ± 135.4 | 180.2 ± 133.7 |
| 40 mg DRI | 203.9 ± 105.7 | 193.4 ± 102.0 | 215.7 ± 134.9 | 176.1 ± 92.9 |
| 60 mg DRI | 273.0 ± 249.9 | 272.6 ± 223.1 | 293.6 ± 217.9 | 234.0 ± 213.2 |
| Estradiol, pg/mL | ||||
| Placebo | 24.3 ± 15.0 | 25.4 ± 15.3 | 21.6 ± 12.9 | 24.3 ± 15.7 |
| 40 mg DRI | 28.6 ± 18.7 | 30.0 ± 19.6 | 35.1 ± 23.4 | 27.3 ± 17.5 |
| 60 mg DRI | 28.4 ± 16.6 | 28.7 ± 17.0 | 35.8 ± 21.5 | 24.2 ± 15.8 |
| SHBG, nmol/L | ||||
| Placebo | 112.4 ± 77.2 | 111.7 ± 67.9 | 109.7 ± 69.1 | 108.2 ± 64.4 |
| 40 mg DRI | 131.2 ± 89.7 | 133.9 ± 87.9 | 132.7 ± 87.0 | 134.4 ± 96.0 |
| 60 mg DRI | 116.9 ± 61.2 | 128.5 ± 76.5 | 127.0 ± 74.3 | 126.0 ± 66.7 |
| FSH, mIU/mL | ||||
| Placebo | 104.5 ± 44.2 | 100.5 ± 40.4 | 97.7 ± 38.9 | 99.3 ± 37.6 |
| 40 mg DRI | 85.1 ± 46.2 | 79.3 ± 49.0 | 77.9 ± 48.7 | 80.0 ± 51.4 |
| 60 mg DRI | 108.6 ± 48.3 | 100.7 ± 51.3 | 98.2 ± 54.5 | 103.8 ± 51.3 |
| Insulin, μIU/mL | ||||
| Placebo | 15.1 ± 21.8 | 15.9 ± 19.0 | 18.3 ± 24.0 | 15.7 ± 27.4 |
| 40 mg DRI | 19.1 ± 35.4 | 26.7 ± 53.3 | 20.8 ± 47.5 | 26.3 ± 46.9 |
| 60 mg DRI | 18.0 ± 32.7 | 18.4 ± 37.3 | 17.9 ± 29.7 | 18.6 ± 31.7 |
| Leptin, ng/mL | ||||
| Placebo | 48.2 ± 40.6 | 47.3 ± 42.5 | 45.2 ± 39.3 | 46.6 ± 40.3 |
| 40 mg DRI | 58.1 ± 40.3 | 63.4 ± 44.4 | 61.0 ± 44.1 | 61.2 ± 40.4 |
| 60 mg DRI | 52.8 ± 38.3 | 47.2 ± 32.6 | 50.8 ± 34.8 | 53.2 ± 37.0 |
| TSH, μIU/mL | ||||
| Placebo | 3.2 ± 3.6 | 3.2 ± 3.0 | 3.1 ± 2.8 | 3.5 ± 3.8 |
| 40 mg DRI | 4.3 ± 11.0 | 3.7 ± 10.8 | 4.4 ± 10.9 | 4.9 ± 11.6 |
| 60 mg DRI | 3.6 ± 3.2 | 3.6 ± 3.9 | 3.6 ± 3.1 | 3.4 ± 2.6 |
| Thyroglobulin, ng/mL | ||||
| Placebo | 17.6 ± 19.1 | 21.0 ± 30.9 | 20.5 ± 30.1 | 24.8 ± 32.1 |
| 40 mg DRI | 25.6 ± 24.2 | 25.1 ± 24.9 | 23.1 ± 21.3 | 26.3 ± 23.9 |
| 60 mg DRI | 38.8 ± 93.3 | 35.6 ± 93.2 | 38.2 ± 93.4 | 41.0 ± 88.6 |
| Free T3, pg/mL | ||||
| Placebo | 2.8 ± 0.9 | 2.8 ± 1.0 | 2.7 ± 1.1 | 2.6 ± 1.0 |
| 40 mg DRI | 2.7 ± 0.8 | 2.7 ± 0.9 | 2.6 ± 0.9 | 2.7 ± 0.9 |
| 60 mg DRI | 2.9 ± 1.1 | 2.8 ± 1.0 | 2.8 ± 0.9 | 2.8 ± 1.0 |
| Free T4, pg/dL | ||||
| Placebo | 1.1 ± 0.2 | 1.1 ± 0.2 | 1.0 ± 0.2 | 1.0 ± 0.2 |
| 40 mg DRI | 1.0 ± 0.2 | 1.1 ± 0.2 | 1.1 ± 0.3 | 1.0 ± 0.3 |
| 60 mg DRI | 1.1 ± 0.3 | 1.1 ± 0.3 | 1.1 ± 0.3 | 1.1 ± 0.3 |
| Total T3, ng/dL | ||||
| Placebo | 85.6 ± 31.3 | 83.4 ± 26.1 | 82.7 ± 31.3 | 83.4 ± 30.5 |
| 40 mg DRI | 82.7 ± 30.2 | 82.7 ± 31.2 | 78.9 ± 27.5 | 81.9 ± 29.5 |
| 60 mg DRI | 84.5 ± 25.4 | 82.0 ± 24.7 | 82.6 ± 24.5 | 83.1 ± 25.0 |
| Total T4, μg/dL | ||||
| Placebo | 6.3 ± 1.7 | 6.4 ± 1.6 | 6.2 ± 1.6 | 6.0 ± 1.6 |
| 40 mg DRI | 6.4 ± 2.2 | 6.7 ± 2.3 | 6.4 ± 1.9 | 6.3 ± 1.6 |
| 60 mg DRI | 6.6 ± 1.8 | 6.7 ± 2.4 | 6.7 ± 2.3 | 6.4 ± 2.1 |
Values are means ± SD. There was no significant difference among the three groups at baseline, 4, 8 and 12 weeks. LH, luteinizing hormone; DRI, daidzein-rich isoflavone aglycone; SHBG, sex hormone–binding hormone; FSH, follicle-stimulating hormone; TSH, thyroid-stimulating hormone; T3, triiodothyronine; T4, thyroxine.
The DRI supplement did not produce any significant changes in endogenous sex hormone levels compared with the placebo. There were also no changes in serum thyroid-stimulating hormone, free thyroxine, free triiodothyronine, total thyroxine, total triiodothyronine, or thyroglobulin level with the supplements. The data for the hormone levels are summarized in Table 3.
Serum isoflavone levels at the beginning of the study were similarly low among the three treatment groups. At the end of the study, serum daidzein, glycetein, and equol, but not genistein, significantly increased in the 40- and 60-mg DRI groups compared with the control group (Table 4).
TABLE 4.
Isoflavones and metabolites
| Placebo |
40 mg DRI |
60 mg DRI |
||||
|---|---|---|---|---|---|---|
| Baseline | 12 wk | Baseline | 12 wk | Baseline | 12 wk | |
| Daidzein, ng/mL | 4.11 ± 10.61 | 9.37 ± 33.13 | 5.84 ± 20.08 | 22.77 ± 32.98a | 2.19 ± 4.78 | 60.90 ± 104.65b |
| Glycetein, ng/mL | 1.21 ± 2.51 | 1.83 ± 4.24 | 2.49 ± 4.01 | 4.24 ± 5.38a | 2.64 ± 5.60 | 15.80 ± 22.78b |
| Genistein, ng/mL | 12.35 ± 43.28 | 15.05 ± 76.40 | 4.01 ± 5.54 | 11.19 ± 22.48 | 5.34 ± 7.52 | 16.04 ± 21.37 |
| Equol, ng/mL | 3.31 ± 8.88 | 3.72 ± 6.41 | 5.70 ± 8.55 | 8.89 ± 11.67a | 7.11 ± 11.60 | 13.19 ± 20.55a |
Values are means ± SD. DRI, daidzein-rich isoflavone aglycone.
P < 0.05 vs control.
P < 0.0005 vs control.
There were no correlations between either the changes in hot flash frequency or hot flash score at the end of the study and serum levels of daidzein or equol (data not shown).
DISCUSSION
This study observed that in postmenopausal women with hot flashes, dietary supplementation with 40 to 60 mg of DRI per day significantly decreased hot flash frequency. Hot flash frequency was reduced by 43% to 52% at 8 to 12 weeks with the supplement, compared with a 32% to 39% change in the placebo group. Although there was only a trend toward an improvement with supplements when analyzing the three groups separately in this study, the two treatment groups combined produced slightly better improvement in hot flash symptoms than did the placebo. The degree of improvement with the DRI supplement is similar to that of the selective serotonin reuptake inhibitor/serotonin and norepinephrine reuptake inhibitor treatment family (50%-60%) but without their significant side effects.10 We also saw a similar improvement in menopausal quality of life in the two treatment groups; however, the sample size was insufficient to detect a statistically significant difference in this secondary outcome.
Functionally, isoflavones can exert both estrogenic and antiestrogenic effects depending on their concentration, target tissue, receptor status of the tissue, and the level of endogenous estrogen. The structure of the isoflavone molecule resembles that of many estrogenic and antiestrogenic compounds, including 17β-estradiol and tamoxifen. Daidzein has been found to have both weak estrogenic and weak anti-estrogenic effects. In vivo its estrogenic activity is the lowest of the soy isoflavones, but its metabolite equol can also exert estrogenic activity.11 Other effects of isoflavones include effects on adenosine triphosphatase, glucose transport, and a variety of ion transport systems; antioxidant effects on lipids, lipoprotein, and DNA; and specific actions on protein synthesis, cell proliferation, and cell differentiation.
Isoflavones are biotransformed extensively in the intestine by the action of bacterial enzymes. The extent of intestinal bacterial metabolism influences the bioavailability of isoflavones and therefore their potential for physiologic effects, although the degree of influence varies among individuals. In soybeans, genistein and daidzein are present as β-glycosides, which are inactive forms of isoflavones. When consumed they undergo metabolic conversion in the intestine whereby a sugar molecule is removed from each, and conversion to metabolically active aglycones occurs. After the conversion approximately one third of aglycones are absorbed as free isoflavones, and two thirds undergo further metabolism by intestinal flora and are converted to isoflavone metabolites such as equol and p-ethylphenol, which are then absorbed into the bloodstream. Both intact and metabolized isoflavones freely circulate in the blood and are excreted in the urine within 24 hours.12
Previous randomized controlled clinical trials of isoflavone supplementation in women with no history of breast cancer that lasted more than 6 weeks have shown either no improvement or a slight reduction in hot flashes in women who consumed soy or isoflavones compared with control women. A randomized, controlled trial of 104 postmenopausal Italian women with severe hot flashes showed that 76 mg/day of soy isoflavones was superior to placebo in reducing the mean number of hot flashes per 24 hours after 4, 8, and 12 weeks of treatment (45% vs 30% at 12 wk). Gastrointestinal side effects (constipation and food intolerance) were the most common causes of premature withdrawal from the study (seven women in each group).13 In another placebo-controlled trial of 80 postmenopausal women, menopausal symptoms assessed by the Kupperman index significantly improved with 100 mg/day of isoflavone (70% genistein, 19% daidzein, and 11% glycitein) supplementation for 4 months compared with the placebo. In that study, however, hot flash frequency decreased only from 11.3 ± 0.3 to 8.2 ± 0.5 per day in the isoflavone group, compared with 10.4 ± 0.4 to 9.9 ± 0.4 per day in the placebo group.14 In a 3-month study of oral and transdermal isoflavones, a dose-dependent effect of transdermal but not oral isoflavones was seen. Both forms were more effective than placebo in attenuating mild to moderate hot flashes and other menopausal symptoms.15 In contrast, a recent study found that soy isoflavones were effective in reducing hot flashes only when combined with a supplement of polyunsaturated fatty acids.16 A recent meta-analysis of 17 randomized clinical studies on isoflavone supplementation and hot flash frequency found statistically significant results,17 whereas another earlier published meta-analysis of 17 randomized isoflavone trials showed no reduction in hot flashes for red clover isoflavones and inconsistent results for soy isoflavones.18 Another recent analysis of 11 studies found that studies using predominantly genistein compositions showed benefit, whereas predominantly daidzein compositions generally did not.19 However, two other previous randomized, controlled studies using daidzein-rich isoflavone extracted from soy germ were conducted in Brazil and in Japan, and both studies showed significant improvement in hot flashes compared with placebo.11,20
The effort to determine the effects of isoflavones is complicated by the variety of different soy foods and an absence of standardized nomenclature.21 For instance, the chemical methods used to isolate isoflavones (eg, ethanol extraction) may alter their physiologic effects. In addition, striking compositional differences are found between types of soy foods commonly used in Southeast Asia and those used in Western countries. In the United States soy has been used mostly for the production of vegetable oils, whereas in Southeast Asia many of the soy foods used are fermented; the microorganisms used in their preparation are capable of hydrolyzing the inactive isoflavone glycosides to active isoflavone aglycones. These differences may be important with regard to metabolism and bioavailability.22 There is also considerable interindividual variation in gut bacterial metabolism of isoflavones, which could further contribute to inconsistencies.23,24
In contrast to isoflavone products used in other studies, the supplement used in this study is a DRI. This is a standardized, concentrated isoflavone ingredient made by extracting the isoflavones from soy germ fermented with Koji fungus, which transforms the glycoside isoflavones in the soy germ to aglycone isoflavones. In the United States this isoflavone extract is available as an over-the-counter dietary supplement under the trade name Effisoy. Koji is a microorganism that possesses numerous enzymes, and soy germ contains other physiologically active components besides isoflavones, such as saponins. It is possible that the functionality of numerous components in soy germ are improved by the collective enzymatic breakdown during the fermentation stage and that such physiologically active components are captured in the supplement during the isoflavone extraction and concentration process. In our study it is possible that the fermentation process, along with the higher concentration of daidzein relative to other isoflavone supplements, accounted for the efficacy of the supplement. Starting with the aglycone form may have eliminated some of the inconsistency related to interindividual variation in gut bacterial metabolism of isoflavones. Previous studies have also shown that the aglycone form may lead to faster and greater absorption of isoflavones than their glycoside counterpart.25,26
In agreement with other studies, we found no significant changes in endogenous sex hormone levels. We also found no association between hot flash frequency or severity and serum isoflavone levels. A previous clinical trial similarly reported that the increase in blood or urine phytoestrogen levels in the soy supplement group did not correlate with fewer hot flashes.27 This is probably due in part to large interindividual and intraindividual variations of isoflavone levels; additionally, there may be other ways that isoflavones affect hot flashes.
Soy consumption has been linked to the development of goiter, an effect independent of estrogenicity.28 Soy antithyroid effects have been shown to increase in people with iodine deficiency, whereas iodine supplementation is protective. In our study we found no effects of isoflavone supplementation on thyroid function tests. This is not surprising because iodine intake is usually sufficient among women in the United States. Additionally, the supplement used in this study contains mainly daidzein, whereas the studies that demonstrated decreased activity of rat and human thyroid peroxidase in vitro had focused on genistein-fortified diets.
Previous animal studies29 and clinical trials30 have demonstrated the beneficial effects of soy protein on plasma cholesterol levels. However, an analysis of recent randomized controlled trials showed soy protein with isoflavone supplement (average 50 g/d) was associated with a very small reduction in low-density lipoprotein cholesterol (mean 3%) with no change in high-density lipoprotein cholesterol, triglycerides, lipoprotein (a), and blood pressure.31 In our study we did not find any significant changes in serum cholesterol, other lipids, blood pressure, or body weight.
There were some limitations to this study. The washout period for the supplement or medication treatment of hot flashes of 6 weeks may not have been enough. However, most of the women had never used HT in the past or had stopped HT for more than 6 months before enrollment in the study. A high dropout rate (fewer than 50 women per group completed the study) may also have led to underestimation of the efficacy and side effects of the supplement. Last, the inclusion of both perimenopausal and postmenopausal women in the study might have affected our results because hot flashes tend to improve over time even without treatment. Although there were no significant differences between the supplement groups and the control group when perimenopausal and postmenopausal women were analyzed separately, this similarity may be the result of the small sample size.
CONCLUSION
In conclusion, this study suggests that DRI supplements may be an effective alternative treatment for menopausal hot flashes. The supplements do not have any effect on endogenous sex hormones, thyroid hormones, or lipid levels.
Acknowledgments
The authors greatly appreciate Nichimo Co, Ltd, Tokyo, Japan, for providing the funding for this investigator-initiated study.
Funding/support: This investigator-initiated study was supported by a research grant by Nichimo Co, Ltd, Tokyo, Japan, who made daidzein-rich isoflavone aglycone extract from soy germ fermentation with Koji fungus Effisoy.
Footnotes
Financial disclosure: None reported.
REFERENCES
- 1.Freedman RR. Physiology of hot flashes. Am J Hum Biol. 2001;13:453–464. doi: 10.1002/ajhb.1077. [DOI] [PubMed] [Google Scholar]
- 2.Pansini F, Albertazzi P, Bonaccorsi G, et al. The menopausal transition: a dynamic approach to the pathogenesis of neurovegetative complaints. Eur J Obstet Gynecol Reprod Biol. 1994;57:103–109. doi: 10.1016/0028-2243(94)90051-5. [DOI] [PubMed] [Google Scholar]
- 3.Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women's Health Initiative randomized controlled trial. JAMA. 2002;288:321–333. doi: 10.1001/jama.288.3.321. [DOI] [PubMed] [Google Scholar]
- 4.Manson JE, Hsia J, Johnson KC, et al. Estrogen plus progestin and the risk of coronary heart disease. N Engl J Med. 2003;349:523–534. doi: 10.1056/NEJMoa030808. [DOI] [PubMed] [Google Scholar]
- 5.Ricciotti HA, Khaodhiar L, Blackburn GL. Daidzein-rich isoflavone-aglycones for menopausal symptoms. Int J Gynaecol Obstet. 2005;89:65–66. doi: 10.1016/j.ijgo.2005.01.028. [DOI] [PubMed] [Google Scholar]
- 6.Hilditch JR, Lewis J, Peter A, et al. A menopause-specific quality of life questionnaire: development and psychometric properties. Maturitas. 1996;24:161–175. doi: 10.1016/s0378-5122(96)82006-8. [DOI] [PubMed] [Google Scholar]
- 7.Sloan JA, Loprinzi CL, Novotny PJ, Barton DL, Lavasseur BI, Windschitl H. Methodologic lessons learned from hot flash studies. J Clin Oncol. 2001;19:4280–4290. doi: 10.1200/JCO.2001.19.23.4280. [DOI] [PubMed] [Google Scholar]
- 8.Singh AV, Franke AA, Blackburn GL, Zhou JR. Soy phytochemicals prevent orthotopic growth and metastasis of bladder cancer in mice by alterations of cancer cell proliferation and apoptosis and tumor angiogenesis. Cancer Res. 2006;66:1851–1858. doi: 10.1158/0008-5472.CAN-05-1332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhou JR, Yu L, Zhong Y, et al. Inhibition of orthotopic growth and metastasis of androgen-sensitive human prostate tumors in mice by bioactive soybean components. Prostate. 2002;53:143–153. doi: 10.1002/pros.10141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stearns V. Serotonergic agents as an alternative to hormonal therapy for the treatment of menopausal vasomotor symptoms. Treat Endocrinol. 2006;5:83–87. doi: 10.2165/00024677-200605020-00002. [DOI] [PubMed] [Google Scholar]
- 11.Uesugi S, Watanabe S, Ishiwata N, Uehara M, Ouchi K. Effects of isoflavone supplements on bone metabolic markers and climacteric symptoms in Japanese women. Biofactors. 2004;22:221–228. doi: 10.1002/biof.5520220145. [DOI] [PubMed] [Google Scholar]
- 12.Vincent A, Fitzpatrick LA. Soy isoflavones: are they useful in menopause? Mayo Clin Proc. 2000;75:1174–1184. doi: 10.4065/75.11.1174. [DOI] [PubMed] [Google Scholar]
- 13.Albertazzi P, Pansini F, Bonaccorsi G, Zanotti L, Forini E, De Aloysio D. The effect of dietary soy supplementation on hot flushes. Obstet Gynecol. 1998;91:6–11. doi: 10.1016/s0029-7844(97)00597-8. [DOI] [PubMed] [Google Scholar]
- 14.Han KK, Soares JM, Jr, Haidar MA, de Lima GR, Baracat EC. Benefits of soy isoflavone therapeutic regimen on menopausal symptoms. Obstet Gynecol. 2002;99:389–394. doi: 10.1016/s0029-7844(01)01744-6. [DOI] [PubMed] [Google Scholar]
- 15.Colacurci N, Zarcone R, Borrelli A, et al. Effects of soy isoflavones on menopausal neurovegetative symptoms. Minerva Ginecol. 2004;56:407–412. [PubMed] [Google Scholar]
- 16.Campagnoli C, Abba C, Ambroggio S, Peris C, Perona M, Sanseverino P. Polyunsaturated fatty acids (PUFAs) might reduce hot flushes: an indication from two controlled trials on soy isoflavones alone and with a PUFA supplement. Maturitas. 2005;51:127–134. doi: 10.1016/j.maturitas.2004.11.002. [DOI] [PubMed] [Google Scholar]
- 17.Howes LG, Howes JB, Knight DC. Isoflavone therapy for menopausal flushes: a systematic review and meta-analysis. Maturitas. 2006;55:203–211. doi: 10.1016/j.maturitas.2006.03.008. [DOI] [PubMed] [Google Scholar]
- 18.Nelson HD, Vesco KK, Haney E, et al. Nonhormonal therapies for menopausal hot flashes: systematic review and meta-analysis. JAMA. 2006;295:2057–2071. doi: 10.1001/jama.295.17.2057. [DOI] [PubMed] [Google Scholar]
- 19.Williamson-Hughes PS, Flickinger BD, Messina MJ, Empie MW. Isoflavone supplements containing predominantly genistein reduce hot flash symptoms: a critical review of published studies. Menopause. 2006;13:831–839. doi: 10.1097/01.gme.0000227330.49081.9e. [DOI] [PubMed] [Google Scholar]
- 20.Petri Nahas E, Nahas Neto J, De Luca L, Traiman P, Pontes A, Dalben I. Benefits of soy germ isoflavones in postmenopausal women with contraindication for conventional hormone replacement therapy. Maturitas. 2004;48:372–380. doi: 10.1016/j.maturitas.2003.09.026. [DOI] [PubMed] [Google Scholar]
- 21.Erdman JW, Jr, Badger TM, Lampe JW, Setchell KD, Messina M. Not all soy products are created equal: caution needed in interpretation of research results. J Nutr. 2004;134:1229S–1233S. doi: 10.1093/jn/134.5.1229S. [DOI] [PubMed] [Google Scholar]
- 22.The role of isoflavones in menopausal health: consensus opinion of The North American Menopause Society. Menopause. 2000;7:215–229. [PubMed] [Google Scholar]
- 23.Wiseman H, Casey K, Bowey EA, et al. Influence of 10 wk of soy consumption on plasma concentrations and excretion of isoflavonoids and on gut microflora metabolism in healthy adults. Am J Clin Nutr. 2004;80:692–699. doi: 10.1093/ajcn/80.3.692. [DOI] [PubMed] [Google Scholar]
- 24.Hendrich S. Bioavailability of isoflavones. J Chromatogr B Analyt Technol Biomed Life Sci. 2002;777:203–210. doi: 10.1016/s1570-0232(02)00347-1. [DOI] [PubMed] [Google Scholar]
- 25.Izumi T, Piskula MK, Osawa S, et al. Soy isoflavone aglycones are absorbed faster and in higher amounts than their glucosides in humans. J Nutr. 2000;130:1695–1699. doi: 10.1093/jn/130.7.1695. [DOI] [PubMed] [Google Scholar]
- 26.Kano M, Takayanagi T, Harada K, Sawada S, Ishikawa F. Bioavailability of isoflavones after ingestion of soy beverages in healthy adults. J Nutr. 2006;136:2291–2296. doi: 10.1093/jn/136.9.2291. [DOI] [PubMed] [Google Scholar]
- 27.Albertazzi P, Pansini F, Bottazzi M, Bonaccorsi G, De Aloysio D, Morton MS. Dietary soy supplementation and phytoestrogen levels. Obstet Gynecol. 1999;94:229–231. doi: 10.1016/s0029-7844(99)00275-6. [DOI] [PubMed] [Google Scholar]
- 28.Doerge DR, Sheehan DM. Goitrogenic and estrogenic activity of soy isoflavones. Environ Health Perspect. 2002;110(Suppl 3):349–353. doi: 10.1289/ehp.02110s3349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Anthony MS, Clarkson TB, Bullock BC, Wagner JD. Soy protein versus soy phytoestrogens in the prevention of diet-induced coronary artery atherosclerosis of male cynomolgus monkeys. Arterioscler Thromb Vasc Biol. 1997;17:2524–2531. doi: 10.1161/01.atv.17.11.2524. [DOI] [PubMed] [Google Scholar]
- 30.Potter SM, Baum JA, Teng H, Stillman RJ, Shay NF, Erdman JW., Jr Soy protein and isoflavones: their effects on blood lipids and bone density in postmenopausal women. Am J Clin Nutr. 1998;68(Suppl 6):1375S–1379S. doi: 10.1093/ajcn/68.6.1375S. [DOI] [PubMed] [Google Scholar]
- 31.Sacks FM, Lichtenstein A, Van Horn L, Harris W, Kris-Etherton P, Winston M. Soy protein, isoflavones, and cardiovascular health: an American Heart Association science advisory for professionals from the Nutrition Committee. Circulation. 2006;113:1034–1044. doi: 10.1161/CIRCULATIONAHA.106.171052. [DOI] [PubMed] [Google Scholar]