Abstract
Inherited loss-of-function mutations in BRCA1 and BRCA2 and other tumor suppressor genes predispose to ovarian carcinomas, but the overall burden of disease due to inherited mutations is not known. Using targeted capture and massively parallel genomic sequencing, we screened for germ-line mutations in 21 tumor suppressor genes in genomic DNA from women with primary ovarian, peritoneal, or fallopian tube carcinoma. Subjects were consecutively enrolled at diagnosis and not selected for age or family history. All classes of mutations, including point mutations and large genomic deletions and insertions, were detected. Of 360 subjects, 24% carried germ-line loss-of-function mutations: 18% in BRCA1 or BRCA2 and 6% in BARD1, BRIP1, CHEK2, MRE11A, MSH6, NBN, PALB2, RAD50, RAD51C, or TP53. Six of these genes were not previously implicated in inherited ovarian carcinoma. Primary carcinomas were generally characterized by genomic loss of normal alleles of the mutant genes. Of women with inherited mutations, >30% had no family history of breast or ovarian carcinoma, and >35% were 60 y or older at diagnosis. More patients with ovarian carcinoma carry cancer-predisposing mutations and in more genes than previously appreciated. Comprehensive genetic testing for inherited carcinoma is warranted for all women with ovarian, peritoneal, or fallopian tube carcinoma, regardless of age or family history. Clinical genetic testing is currently done gene by gene, with each test costing thousands of dollars. In contrast, massively parallel sequencing allows such testing for many genes simultaneously at low cost.
Ovarian carcinoma is the most deadly of gynecological malignancies; the majority of women are diagnosed with advanced stage disease when the chance of cure is small. Inherited mutations in BRCA1 and BRCA2 create a lifetime risk of ovarian carcinoma of between 20% (for BRCA2) and 50% or even higher (for BRCA1) (1). It has been previously estimated that 13–15% of patients with ovarian carcinoma in North America carry germ-line mutations in BRCA1 or BRCA2 (2, 3). Hereditary ovarian carcinoma also occurs in the context of Lynch syndrome [hereditary nonpolyposis colorectal cancer (HNPCC)], but the proportion of ovarian carcinoma explained by germ-line mutations in the mismatch repair genes has not been determined. Inherited mutations in RAD51C, RAD51D, and PALB2 have also been reported in patients with familial ovarian carcinoma (4–6). The overall proportion of ovarian carcinoma due to germ-line mutations in these genes and the roles of other tumor suppressor genes, particularly those implicated in inherited breast cancer, remain unknown.
Women with early stage ovarian carcinoma have far better survival than women whose carcinomas are diagnosed at later stages, but current methods of early detection have not proven effective (7). In contrast, risk-reducing salpingo-oophorectomy in women with BRCA1 or BRCA2 mutations dramatically reduces risk of ovarian carcinoma and significantly decreases overall mortality (8–10). It is critically important, therefore, to identify inherited risk for ovarian carcinoma to allow effective and targeted prevention. The recent development of poly-ADP ribose polymerase (PARP) inhibitors in cancer therapeutics has added another incentive to identify inherited risk (11, 12). PARP inhibitors are selectively lethal to cells deficient in BRCA1 or BRCA2 and are proving effective in the treatment of ovarian carcinomas associated with mutations in these genes (13).
We recently validated a massively parallel sequencing approach, which we name BROCA, in honor of Paul Broca, the 19th Century neurosurgeon, oncologist, anatomist, and evolutionist, who elegantly described inherited breast and ovarian cancer (14). BROCA is highly sensitive for sequencing a panel of 21 tumor suppressor genes, including BRCA1, BRCA2, and other genes known to cause inherited breast or ovarian carcinoma (15). BROCA accurately identifies all classes of mutations including single-base substitutions, small insertions and deletions, and large gene rearrangements. In the present study, we apply BROCA to analyze genomic DNA from a large consecutive series of women with primary ovarian, peritoneal, or fallopian tube carcinoma. Subjects were enrolled at diagnosis and not selected for age at onset or family history.
Results
Mutation Detection and Characterization.
Subjects were 360 women undergoing primary surgery at University of Washington between 2001 and 2010. Subjects included 273 women with ovarian carcinomas, 48 with peritoneal carcinomas, 31 with fallopian tube carcinomas, and 8 with synchronous endometrial and ovarian carcinomas. Targeted capture by BROCA baits and genomic sequencing yielded median 449-fold coverage; 98.4% of bases had >50-fold coverage, our threshold for variant detection (Fig. S1).
The 360 subjects harbored 85 germ-line loss-of-function mutations in 12 genes: 40 (11.1%) in BRCA1, 23 (6.4%) in BRCA2, and 22 (6.1%) in 10 other genes (Fig. 1 and Table S1). Three subjects carried mutations in two genes, so the total proportion of subjects with at least one germ-line mutation was 0.23 (82/360). Features of the mutations and of neoplastic histology, family history, and age of diagnosis for each of the subjects with a mutation are provided in Table S2.
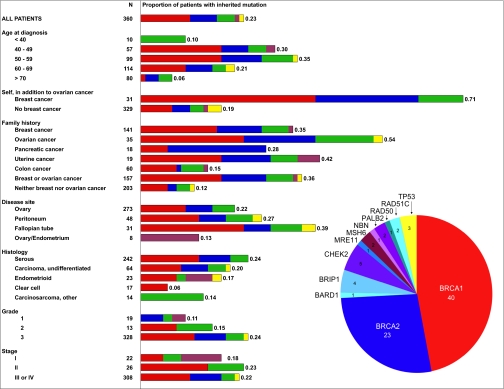
Fig. 1.
Proportions of patients with primary ovarian, fallopian tube, or peritoneal cancers with germ-line loss-of-function mutations in BRCA1 (red); BRCA2 (blue); BARD1, BRIP1, CHEK2, MRE11, NBN, PALB2, RAD50, or RAD51C (green); MSH6 (purple); or p53 (yellow).
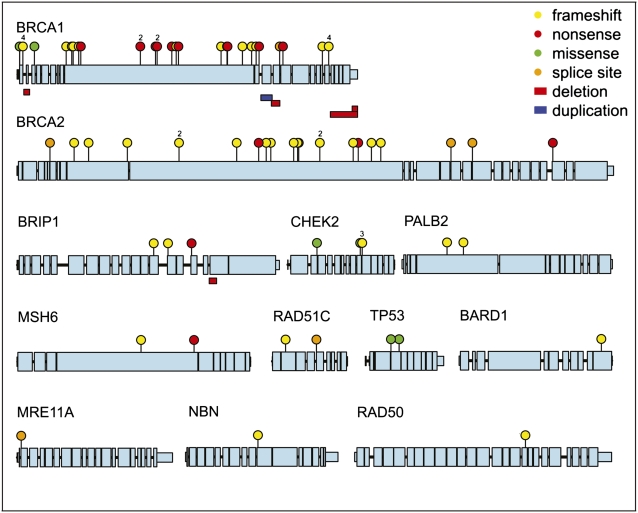
Classes of damaging events included protein-truncating mutations, missense mutations with demonstrated effect on protein function, and large genomic deletions and duplications (Fig. 2). Six mutations (five in BRCA1 and one in BRIP1) were structural genomic deletions or duplications ranging in size from 1 to 10 kb. All structural mutations generated mutant transcripts confirmed by Sanger sequence of cDNA generated from RNA from patients’ lymphoblasts. Large deletions or duplications accounted for 13% of BRCA1 mutations and 7% of all loss-of-function mutations. Missense mutations were considered to lead to loss-of-function only if so demonstrated by functional assay. BRCA1 p.M1I, p.M1V, and p.C61G and TP53 p.Y126D, p.R175C, and pR175H were included as loss-of-function alleles on the basis of previous experimental results (Table S1). For CHEK2, five missense mutations and one in-frame deletion were tested by a yeast-based assay that assesses repair after DNA damage (Table S3). CHEK2 p.H186R and p.S357F were damaging on the basis of this assay.
Fig. 2.
Genes harboring germ-line loss-of-function mutations in patients with primary ovarian, fallopian tube, or peritoneal cancers. Coding regions of all genes are on the same scale. Untranslated regions are at 1:10 scale and introns are at 1:200 scale relative to coding exons. Mutations occurring in multiple individuals are indicated with the number of affected individuals above the small balloon.
Two deleterious germ-line mutations in MSH6, one frameshift and one nonsense, were detected in two subjects diagnosed with ovarian carcinomas of endometrioid histology, one of whom also had a synchronous endometrial carcinoma. Neoplasms of both patients had microsatellite instability phenotypes and no detectable MSH6 protein by immunohistochemistry. No clearly deleterious mutations were detected in MLH1, MSH2, or PMS2. Seventeen missense mutations in MLH1, MSH2, and MSH6 and a 3-bp deletion in MSH6 were predicted to be damaging by bioinformatics tools, but none of these tumors had evidence of microsatellite instability or of loss of MLH1, MSH2, PMS2, or MSH6 protein (Table S4). Conservatively, therefore, the proportion of subjects with a deleterious germ-line mutation in a Lynch syndrome gene was 0.5% (2/360).
Germ-Line Mosaicism in TP53.
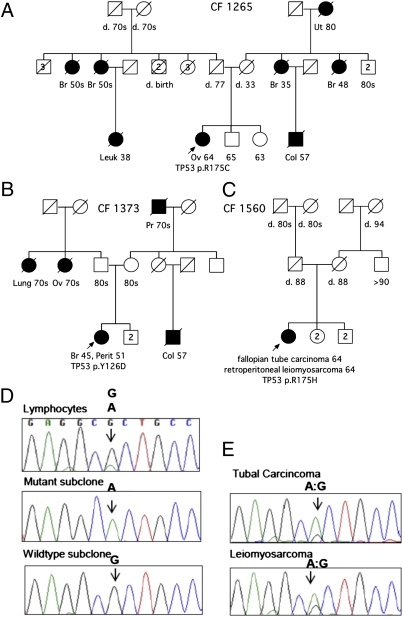
Three subjects (CF1265.01, CF1373.01, and CF1560.01) carried germ-line mutations in TP53. None of their families met criteria for Li–Fraumeni syndrome, although both CF1265.01 and CF1373.01 had positive personal or family history of premenopausal breast cancer (Fig. 3 A and B). CF1560.01 was particularly intriguing as she appears to harbor a germ-line mosaic mutation. This patient was diagnosed at age 64 with two synchronous primary cancers, a high-grade serous tubal carcinoma and a retroperitoneal leiomyosarcoma, but no family history of cancer (Fig. 3C). By BROCA analysis of this patient’s lymphocyte DNA, the number of mutant reads at TP53 p.R175H constituted only 20% of total reads at that base (Table S2). Sanger sequencing confirmed that the mutant allele accounted for only 20% of sequences in lymphocytes (Fig. 3D), both from the original blood sample and from a second blood sample obtained 4 y later. The PCR amplicon from lymphocyte DNA containing the TP53 mutation site was subcloned. Of 65 clones sequenced, 14 (21.5%) contained the mutant sequence. To rule out contamination of blood by free neoplastic DNA, DNA was also obtained from multiply passaged cultured lymphoblasts and the TP53 mutation was confirmed. DNA isolated from both the tubal carcinoma and the retroperitoneal leiomyosarcoma contained TP53 p.R175H with loss of the normal allele (Fig. 3E). The negative family history was consistent with a de novo mosaic germ-line event, as described in one previous patient (16).
Fig. 3.
Pedigrees of subjects with germ-line TP53 mutations. Subjects with carcinomas are indicated with solid symbols. Sites of carcinoma are breast (Br), Colon (Col), leukemia (Leuk), lung (Lung), peritoneum (Perit), ovary (Ov),and uterus (Ut). Ages under symbols indicate age at death (d.) for deceased individuals, age at cancer diagnosis for affected individuals, or present age for unaffected individuals. Probands are indicated with arrows. Other family members have not been tested. (A) CF1265.01 carries TP53 p.R175C. (B) CF1373.01 carries TP53 p.Y126D. (C) CF 1560.01 carries TP53 p.R175H as a mosaic mutation. (D) Sequences at the TP53 c.524G > A (p.R175H) mutant site for lymphocytes of CF1560.01. Electropherogram peaks indicate that DNA from pooled lymphocytes included ∼80% WT and 20% mutant sequences. The amplicon from lymphocyte DNA was subcloned and 65 clones were sequenced, with 51 subclones containing WT sequence and 14 subclones containing mutant sequence. Representative subclones are shown. (E) Sequences at the TP53 c.524G > A (p.R175H) mutant site for two distinct cancers of CF1560.01. DNA sequences from the fallopian tube carcinoma and from the retroperitoneal leimyosarcoma revealed primarily mutant sequence, consistent with loss of the WT allele in neoplastic tissue.
Loss of the WT Allele in Tumor Tissue.
For 80 of the 85 mutations, it was possible to evaluate genotype at the critical sites from patients’ tumor specimens (Table S2). For 82% of tumors (66/80), the WT allele was lost at the mutant site of the critical gene. Exceptions were inherited loss-of-function mutations in RAD50 and BARD1, each occurring in one patient, and neither associated with loss of the WT allele in the corresponding tumors. This observation parallels reports of germ-line loss-of-function mutations in RAD51D (4) and in CHEK2 (17) that were not accompanied by detectable loss of the WT allele in tumor DNA. CHEK2 mutations have been postulated to function via haploinsufficiency (18); we speculate that an alternative explanation is allelic inactivation by promoter methylation or another epigenetic mechanism. Although it is possible in principle that some mutations without corresponding loss of the WT allele in tumors were not causative in ovarian carcinogenesis, it is interesting to note that with complete exome analysis of ovarian carcinomas, 10 of 44 unambiguously deleterious germ-line mutations in BRCA1 and BRCA2 were not accompanied by genomic loss of the WT allele (19).
Characteristics of Patients with Germ-Line Loss-of-Function Mutations.
Overall, 82 of 360 subjects carried a germ-line mutation in ≥1 of the 21 genes (Fig. 1 and Table S1). As expected, personal prior history of breast cancer was associated with an extremely high likelihood of harboring an inherited mutation. Among women with a personal history of breast carcinoma in addition to ovarian carcinoma, >70% (22/31) had a germ-line mutation in one of the tested genes. Family histories of breast, ovarian, uterine, and pancreatic carcinomas (but not colon carcinomas) were each associated with inherited mutations. The converse observation, with implications for clinical practice, was that 31% (25/82) of women with inherited mutations had no prior personal history of breast cancer or family history of breast or ovarian cancer in their first- or second-degree relatives A high proportion of mutation carriers without family history of breast or ovarian cancer can be explained by the combined effects of small family size, inheritance of mutations from unaffected fathers, and Mendelian chance (1).
Age at ovarian cancer diagnosis was not generally associated with the likelihood of harboring an inherited mutation or with the gene in which a mutation was found, although women diagnosed younger than age 40 or at age 70 or older were less likely to carry mutant alleles. Median age at diagnosis of women with mutations in BRCA1 was 55 y (range 42–72 y), in BRCA2 was 58 y (range 45–78 y), and in another gene was 53 y (range 31–84 y), whereas median age for women with no identified mutation was 62 y (range 27–91 y). None of these differences were significant. The median age at diagnosis of patients with BRCA1 mutations in this series was older than that generally reported. This difference may be due to the enrollment of patients in this study without regard to age at diagnosis, whereas genetic testing of patients with ovarian cancer has generally focused on younger patients. Mutations were identified in patients of all deciles of age at diagnosis (Table S1). Of the 82 women with inherited mutations, 37% (30/82) were diagnosed with their cancer after age 60.
Patients with serous carcinomas were more likely to carry mutations specifically in BRCA1 and BRCA2 than were patients with tumors of other histologies; one in four serous ovarian carcinomas was associated with a germ-line BRCA1 or BRCA2 mutation. Nonetheless, the genetics of inherited predisposition is heterogeneous even for patients with serous carcinomas, in that an additional 6% of women with serous carcinomas carried germ-line mutations in any of 10 other genes (Fig. 1). Germ-line mutations in the entire spectrum of genes were also found in patients with carcinomas of nonserous histologies. Tumor grade was related to source of mutation. Patients with grade 2 or 3 carcinomas were more likely to carry mutations in BRCA1 and BRCA2, whereas patients with grade 1 carcinomas were more likely to carry mutations in one of the other genes (Fig. 1).
Previous studies have documented better survival for patients with ovarian cancer with BRCA1 or BRCA2 mutations compared with survival of patients of other genotypes (20). In this series, survival differences by genotype were in the same direction as previously reported, but of smaller effect. Differences in survival by genotype were not significant in this cohort. For all subjects combined, median survival was 47 mo for women with no identified mutation, compared with 49 mo for women with a mutation in a gene other than BRCA1 or BRCA2 (two-sided log-rank P = 0.76) and 53 mo for women with a BRCA1 or BRCA2 mutation (two-sided log-rank P = 0.30). The same trend appeared for all subjects combined and specifically for those with stage III and IV carcinomas.
Discussion
More than one in five ovarian carcinomas are associated with germ-line mutations in tumor suppressor genes, and these mutations are distributed in a larger number of genes than previously appreciated. BRCA1 and BRCA2 account for most inherited risk for ovarian carcinoma, with 18% of patients carrying germ-line loss-of-function mutations in 1 of these genes. In addition, 6% of subjects carried an inherited loss-of-function mutation in ≥1 of 10 other tumor suppressor genes. Mutation frequency may be underestimated in this series of patients, because we included only mutations with a clearly damaging impact on protein function. Some not-yet-characterized missense mutations may prove damaging as well. Also, we did not examine RAD51D, for which inherited mutations have recently been reported in familial ovarian cancer (4).
The substantial proportion of subjects harboring germ-line mutations is particularly noteworthy given that ages at diagnosis were not selected to be young, and most patients had no family history of breast or ovarian cancer in first- or second-degree relatives. Indeed, >30% of patients with mutations (25/82) had no family history of breast or ovarian cancer, and >35% of patients with mutations (30/82) were diagnosed after age 60. Therefore, selecting patients for genetic testing on the basis of family history or young age would miss a significant fraction of mutation carriers. Germline mutations were identified in six genes not previously associated with inherited ovarian carcinoma. These genes are BARD1, BRIP1, CHEK2, MRE11A, NBN, and RAD50. Previous reports linking BARD1 and BRIP1 with ovarian cancer were based on missense variants of unknown consequence (21, 22), whereas patients in this series carry mutations in these genes with definitive loss of function.
The prevalence of inherited mutations in BRCA1 and BRCA2 in this series is similar to that reported in comparable patients in other series drawn from the general population. Among those patients in our series with cancers classified as ovarian in origin, BRCA1 and BRCA2 mutation prevalence was 16%. Previous population-based studies of mutation prevalence, which evaluated only patients with tumors of ovarian origin, found BRCA1 and BRCA2 prevalence rates of 13% and 15% in Ontario, Canada, and in Florida, respectively (2, 3). Similar mutation prevalence rates for comparable subsets of patients suggest that the consecutive series of patients from our tertiary center did not differ substantially from patients with ovarian cancer from the North American population as a whole. Current genomic technologies will facilitate the large case–control studies necessary to estimate relative risks for ovarian carcinoma associated with inherited mutations in the more moderate penetrance genes. Robust penetrance estimates will be important before counseling women about early detection and prevention options.
The prevalence of germ-line mutations in our series is significantly higher than that published recently from The Cancer Genome Atlas (TCGA) Research Network (19). On the basis of exome sequencing, TCGA reported 44 germ-line BRCA1 or BRCA2 mutations in 316 women: 14%, after subtracting three reported occurrences of BRCA2 p.K3326X, a benign polymorphism found in 2% of the general population (23). TCGA reported no germ-line mutations in any other gene, although all were sequenced from constitutional DNA. In contrast, among the 282 women with serous carcinoma in our series, we identified 62 women with germ-line mutations (24%, P = 0.0007). Explanations of this discrepancy likely include (i) low depth of coverage by the TCGA analysis of many exonic regions, even among those targeted for exome sequencing, and (ii) virtually no intronic coverage, precluding assessment of TGCA cases for gene rearrangements, which comprised 7% of damaging events in our series. Whereas exome sequencing is very well suited for the identification of new cancer susceptibility genes, the overall much higher read depth of hybridization to a smaller number of target genes, following by multiplexed sequencing, confers greater sensitivity at lower cost, making it more suitable as a clinical test.
The Fanconi anemia pathway controls DNA repair via homologous recombination. Multiple genes in the Fanconi anemia pathway are known to be associated with breast cancer risk (reviewed in ref. 24). In this series of patients with ovarian carcinoma, we identified deleterious germ-line mutations in every gene that we evaluated in the Fanconi anemia pathway, including NBN, MRE11, RAD50, RAD51C, PALB2, BARD1, and BRIP1. Mutation rates in these genes in patients with ovarian carcinoma in this series (who were not selected for family history) are similar to rates previously reported for patients with breast cancer from high-incidence families (25–28). Our data suggest that this pathway is as broadly involved in ovarian carcinogenesis as in breast carcinogenesis. Carcinomas with mutations in any gene in this pathway are likely to be deficient in homologous recombination and thereby susceptible to killing by PARP inhibitors. Perhaps some of the clinical responses to PARP inhibitors observed in women with serous ovarian carcinomas and triple negative breast cancers without mutations in BRCA1 or BRCA2 could be explained by mutations in other genes in this pathway.
Women with mutations in genes associated with Lynch syndrome are estimated to have an 8–10% lifetime risk of ovarian carcinoma (29, 30), but the proportion of ovarian carcinoma explained by Lynch syndrome mutations has not been previously defined. In this series, only 0.5% (2/360) of cases carried loss-of-function mutations in genes associated with Lynch syndrome. Both mutations were in MSH6, and both women with MSH6 mutations had endometrioid ovarian carcinomas, a histology associated with this syndrome. Our series included lower proportions of endometrioid and mucinous carcinomas compared with previous series (2, 3). Additional mutations in DNA mismatch repair genes might occur in a series with a higher proportion of endometrioid ovarian carcinomas. Nevertheless, the overall contribution of Lynch syndrome to ovarian carcinoma appears to be relatively low.
Mutations not associated with “typical phenotypes” are of particular interest. For example, the three TP53 mutations occurred in patients without a family history of Li–Fraumeni syndrome and the two MSH6 mutations occurred in patients without a family history of Lynch syndrome. As comprehensive genetic testing is undertaken for individuals not selected for established syndromic phenotypes, a wider range of expressivity associated with germ-line mutations of cancer susceptibility genes will become increasingly apparent.
Currently, the vast majority of genetic testing in the United States for inherited mutations in BRCA1 and BRCA2 is done via a single company at a cost of ∼$4,000 for a noncomprehensive test that is complemented by an additional test to identify gene rearrangements. Far less clinical genetic testing for ovarian cancer risk is carried out for the other tumor suppressor genes. Targeted capture and massively parallel sequencing accurately identify mutations of all classes in all tumor suppressor genes in a single test. There is an increasing discrepancy between the falling cost of genomic sequencing and the current high cost of clinical gene-by-gene testing. As more cancer susceptibility genes are identified, it is not economically sustainable to assess risk by sequentially assessing individual genes, given that any one gene is mutant in only a small percentage of cases. For the BROCA test, the reagent cost is ∼$200 per sample and decreases as more samples are multiplexed per lane. Sensitivity is high, demonstrated by the detection of a mosaic germ-line TP53 mutation present in only 20% of sequenced alleles. BROCA capture baits have been recently supplemented to include all known colon, pancreatic, and melanoma cancer genes, yielding a single test applicable to broader cancer susceptibility testing. The speed and cost of BROCA will allow immediate translation into clinical laboratories, and throughput is sufficient for larger epidemiological studies. The BROCA test is not patented and the design of capture baits is freely available.
In conclusion, we identified 85 germ-line mutations in 12 different genes in 360 patients with ovarian, peritoneal, or fallopian tube carcinoma. More than one-fifth of ovarian carcinoma appears to be associated with inherited risk. As the cost of multigene testing continues to fall, it may become cost effective to offer population testing to allow targeted prevention.
Materials and Methods
Study Subjects.
Of patients from whom consent was requested, 95% consented to genomics studies. To minimize referral bias inherent in a tertiary care center, we excluded cases referred for recurrence of disease and cases referred for genetic risk, including cancers identified at the time of risk-reducing surgery. The study was approved by the Human Subjects Division of University of Washington (protocol 34173).
Library Construction, Hybridization, and Massively Parallel Sequencing.
From 3 μg constitutional DNA of each patient, paired-end libraries with 150-bp inserts were prepared and hybridized to a custom pool of oligonucleotides targeting 21 genomic regions as previously described (15), using the SureSelectXT enrichment system on a Bravo liquid-handling instrument (Agilent). Following capture, samples were barcoded with 12 different indexed primers (31), pooled 12 per lane, and sequenced with 2 × 76 bp paired end reads on a GAIIx (Illumina). All suspected deleterious mutations were verified with Sanger sequencing. Carcinomas were evaluated for loss of heterozygosity (LOH) and microsatellite instability, as appropriate. Genes evaluated by BROCA were BRCA1, BRCA2, BRIP1, BARD1, CHEK2, MRE11A, NBN, RAD50, RAD51C, PALB2, TP53, PTEN, STK11, CDH1, ATM, MLH1, MSH2, MSH6, PMS1, PMS2, and MUTYH.
Mutation Analysis.
Single-nucleotide variants and insertions and deletions were detected as previously described (15). Deletions and duplications of exons were detected by a combination of depth of coverage and split-read analysis as previously described (31), supplemented with additional alignments generated by SLOPE (32). All frameshift, nonsense, missense, or splice site mutations predicted to be deleterious to protein function were validated by PCR amplification and Sanger sequencing. Potential splice site mutations within 12 nucleotides of the consensus exon–intron boundaries were also tested for aberrant transcription by RT-PCR of RNA from patients’ lymphoblasts (Table S5) (6). Exonic deletions and duplications were validated by determining genomic breakpoints with Sanger sequencing, as previously described (33). Missense mutations and in-frame deletions were classified as deleterious only if a specific functional assessment could be or previously had been carried out (e.g., BRCA1 M1V). Missense mutations in CHEK2, MLH1, MSH2, and MSH6 were initially evaluated using bioinformatics tools and conservation considerations and, if predicted to be damaging, were assessed by a yeast functional assay for CHEK2 or by assessing neoplasms for microsatellite instability (MLH1, MSH2, and MLH6).
Survival Analysis.
Survival was determined from date of diagnosis to date of death or date of last follow-up and was censored for patients alive at last follow-up according to the methods of Kaplan and Meier.
Supplementary Material
Acknowledgments
This work was supported by National Institutes of Health Grants R01CA131965, R01CA157744, and P50CA083636, the Breast Cancer Research Foundation, Susan G. Komen for the Cure, and Department of Defense Ovarian Cancer Research Program OC093285.
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1115052108/-/DCSupplemental.
References
- 1.King MC, Marks JH, Mandell JB. New York Breast Cancer Study Group Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science. 2003;302:643–646. doi: 10.1126/science.1088759. [DOI] [PubMed] [Google Scholar]
- 2.Zhang S, et al. Frequencies of BRCA1 and BRCA2 mutations among 1,342 unselected patients with invasive ovarian cancer. Gynecol Oncol. 2011;121:353–357. doi: 10.1016/j.ygyno.2011.01.020. [DOI] [PubMed] [Google Scholar]
- 3.Pal T, et al. BRCA1 and BRCA2 mutations account for a large proportion of ovarian carcinoma cases. Cancer. 2005;104:2807–2816. doi: 10.1002/cncr.21536. [DOI] [PubMed] [Google Scholar]
- 4.Loveday C, et al. Germline mutations in RAD51D confer susceptibility to ovarian cancer. Nat Genet. 2011;43:879–882. doi: 10.1038/ng.893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Meindl A, et al. Germline mutations in breast and ovarian cancer pedigrees establish RAD51C as a human cancer susceptibility gene. Nat Genet. 2010;42:410–414. doi: 10.1038/ng.569. [DOI] [PubMed] [Google Scholar]
- 6.Casadei S, et al. Contribution of inherited mutations in the BRCA2-interacting protein PALB2 to familial breast cancer. Cancer Res. 2011;71:2222–2229. doi: 10.1158/0008-5472.CAN-10-3958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Partridge E, et al. PLCO Project Team. Results from four rounds of ovarian cancer screening in a randomized trial. Obstet Gynecol. 2009;113:775–782. doi: 10.1097/AOG.0b013e31819cda77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rebbeck TR, Kauff ND, Domchek SM. Meta-analysis of risk reduction estimates associated with risk-reducing salpingo-oophorectomy in BRCA1 or BRCA2 mutation carriers. J Natl Cancer Inst. 2009;101:80–87. doi: 10.1093/jnci/djn442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kauff ND, et al. Risk-reducing salpingo-oophorectomy for the prevention of BRCA1- and BRCA2-associated breast and gynecologic cancer: A multicenter, prospective study. J Clin Oncol. 2008;26:1331–1337. doi: 10.1200/JCO.2007.13.9626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Domchek SM, et al. Mortality after bilateral salpingo-oophorectomy in BRCA1 and BRCA2 mutation carriers: A prospective cohort study. Lancet Oncol. 2006;7:223–229. doi: 10.1016/S1470-2045(06)70585-X. [DOI] [PubMed] [Google Scholar]
- 11.Bryant HE, et al. Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature. 2005;434:913–917. doi: 10.1038/nature03443. [DOI] [PubMed] [Google Scholar]
- 12.Farmer H, et al. Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature. 2005;434:917–921. doi: 10.1038/nature03445. [DOI] [PubMed] [Google Scholar]
- 13.Fong PC, et al. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med. 2009;361:123–134. doi: 10.1056/NEJMoa0900212. [DOI] [PubMed] [Google Scholar]
- 14.Broca P. Traité des Tumeurs. Paris: Asselin; 1866. [Google Scholar]
- 15.Walsh T, et al. Detection of inherited mutations for breast and ovarian cancer using genomic capture and massively parallel sequencing. Proc Natl Acad Sci USA. 2010;107:12629–12633. doi: 10.1073/pnas.1007983107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Prochazkova K, et al. Somatic TP53 mutation mosaicism in a patient with Li-Fraumeni syndrome. Am J Med Genet A. 2009;149A:206–211. doi: 10.1002/ajmg.a.32574. [DOI] [PubMed] [Google Scholar]
- 17.Sodha N, et al. CHEK2 variants in susceptibility to breast cancer and evidence of retention of the wild type allele in tumours. Br J Cancer. 2002;87:1445–1448. doi: 10.1038/sj.bjc.6600637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jekimovs CR, et al. kConFab Investigators Low frequency of CHEK2 1100delC allele in Australian multiple-case breast cancer families: Functional analysis in heterozygous individuals. Br J Cancer. 2005;92:784–790. doi: 10.1038/sj.bjc.6602381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cancer Genome Atlas Research Network Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609–615. doi: 10.1038/nature10166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chetrit A, et al. Effect of BRCA1/2 mutations on long-term survival of patients with invasive ovarian cancer: The national Israeli study of ovarian cancer. J Clin Oncol. 2008;26:20–25. doi: 10.1200/JCO.2007.11.6905. [DOI] [PubMed] [Google Scholar]
- 21.Ratajska M, et al. Cancer predisposing BARD1 mutations in breast-ovarian cancer families. Breast Cancer Res Treat. 2011 doi: 10.1007/s10549-011-1403-8. [DOI] [PubMed] [Google Scholar]
- 22.Sauer MK, Andrulis IL. Identification and characterization of missense alterations in the BRCA1 associated RING domain (BARD1) gene in breast and ovarian cancer. J Med Genet. 2005;42:633–638. doi: 10.1136/jmg.2004.030049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mazoyer S, et al. A polymorphic stop codon in BRCA2. Nat Genet. 1996;14:253–254. doi: 10.1038/ng1196-253. [DOI] [PubMed] [Google Scholar]
- 24.D’Andrea AD. Susceptibility pathways in Fanconi’s anemia and breast cancer. N Engl J Med. 2010;362:1909–1919. doi: 10.1056/NEJMra0809889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guénard F, et al. INHERIT BRCAs Mutational analysis of the breast cancer susceptibility gene BRIP1 /BACH1/FANCJ in high-risk non-BRCA1/BRCA2 breast cancer families. J Hum Genet. 2008;53:579–591. doi: 10.1007/s10038-008-0285-z. [DOI] [PubMed] [Google Scholar]
- 26.Kuusisto KM, Bebel A, Vihinen M, Schleutker J, Sallinen SL. Screening for BRCA1, BRCA2, CHEK2, PALB2, BRIP1, RAD50, and CDH1 mutations in high-risk Finnish BRCA1/2-founder mutation-negative breast and/or ovarian cancer individuals. Breast Cancer Res. 2011 doi: 10.1186/bcr2832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McInerney NM, et al. Evaluation of variants in the CHEK2, BRIP1 and PALB2 genes in an Irish breast cancer cohort. Breast Cancer Res Treat. 2010;121:203–210. doi: 10.1007/s10549-009-0540-9. [DOI] [PubMed] [Google Scholar]
- 28.Wong MW, et al. BRIP1, PALB2, and RAD51C mutation analysis reveals their relative importance as genetic susceptibility factors for breast cancer. Breast Cancer Res Treat. 2011;127:853–859. doi: 10.1007/s10549-011-1443-0. [DOI] [PubMed] [Google Scholar]
- 29.Aarnio M, et al. Cancer risk in mutation carriers of DNA-mismatch-repair genes. Int J Cancer. 1999;81:214–218. doi: 10.1002/(sici)1097-0215(19990412)81:2<214::aid-ijc8>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 30.Watson P, Lynch HT. Extracolonic cancer in hereditary nonpolyposis colorectal cancer. Cancer. 1993;71:677–685. doi: 10.1002/1097-0142(19930201)71:3<677::aid-cncr2820710305>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- 31.Nord AS, Lee M, King MC, Walsh T. Accurate and exact CNV identification from targeted high-throughput sequence data. BMC Genomics. 2011 doi: 10.1186/1471-2164-12-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Abel HJ, et al. SLOPE: A quick and accurate method for locating non-SNP structural variation from targeted next-generation sequence data. Bioinformatics. 2010;26:2684–2688. doi: 10.1093/bioinformatics/btq528. [DOI] [PubMed] [Google Scholar]
- 33.Walsh T, et al. Spectrum of mutations in BRCA1, BRCA2, CHEK2, and TP53 in families at high risk of breast cancer. JAMA. 2006;295:1379–1388. doi: 10.1001/jama.295.12.1379. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.