Abstract
Allergic diseases including asthma and food allergies are an increasing health concern. Immunotherapy is an effective therapeutic approach for many allergic diseases but requires long dose escalation periods and has a high risk of adverse reactions, particularly in food allergy. New methods to safely induce antigen-specific tolerance could improve the clinical approach to allergic disease. We hypothesized that antigen-specific tolerance induced by the i.v. injection of antigens attached to the surface of syngeneic splenic leukocytes (Ag-SP) with the chemical cross-linking agent ethylene-carbodiimide (ECDI), which effectively modulate Th1/Th17 diseases, may also safely and efficiently induce tolerance in Th2-mediated mouse models of allergic asthma and food allergy. Mice were tolerized with Ag-SP before or after initiation of ovalbumin/alum-induced allergic airway inflammation or peanut-induced food allergy. The effects on disease pathology and Th2-directed cytokine and antibody responses were studied. Ag-SP tolerance prevented disease development in both models and safely tolerized T cell responses in an antigen-specific manner in pre-sensitized animals. Prophylactically, Ag-SP efficiently decreased local and systemic Th2 responses, eosinophilia and Ag-specific IgE. Interestingly, Ag-SP induced Th2 tolerance was found to be partially dependent on the function of CD25+ Tregs in the food allergy model, but was Treg independent in the model of allergic airway inflammation. We demonstrate that Ag-SP tolerance can be rapidly, safely and efficiently induced in murine models of allergic disease highlighting a potential new antigen-specific tolerance immunotherapy for Th2-associated allergic diseases.
Keywords: Immunotherapy, tolerance, allergy, asthma, food allergy, regulatory T cell, peanut, Th2 cells
INTRODUCTION
Allergic diseases such as asthma and food allergies are increasing health concerns in developed nations. CD4+ Th2 cells are important regulators of allergy through production of cytokines that induce B cell class-switching to IgE, thereby priming mast cells, as well as eosinophil and basophil maturation – cells important in the effector phase of the allergic cascade (1–3). Avoidance and symptom control are currently the primary clinical therapies for most allergies. However, recent attempts to target the underlying Th2 response have employed specific immunotherapies (SITs) that use gradually increasing doses of soluble antigen via either subcutaneous or sublingual routes to induce tolerance and reduce clinical symptoms of disease (4). However, SITs have a number of drawbacks – requiring many treatments over a long time period and pose a significant risk of inducing adverse reactions, including anaphylaxis. This is particularly concerning in food allergy, wherein patients may exhibit severe reactions to extremely small amounts of antigen. In a recent study using oral immunotherapy for peanut allergy, 93% of patients had adverse reactions during the initial escalation period (5). New methods to more safely and efficiently induce antigen-specific tolerance could improve the clinical approach to allergic disease.
We have previously demonstrated that i.v. administration of peptides attached to the surface of syngeneic splenic leukocytes (Ag-SP) with the chemical cross-linking agent ethylene-carbodiimide (ECDI) safely and efficiently induces antigen-specific immune tolerance (6). Ag-SP tolerance has been shown to prevent and treat pre-established symptoms of Th1/Th17-mediated autoimmune disease including experimental autoimmune encephalomyelitis (EAE) (7) and Type-1 diabetes (8), and promote potent alloantigen-specific tolerance for specific long-term protection of transplanted allogeneic pancreatic islets in the absence of immunosuppressive drugs (9). However, Ag-SP tolerance has yet to be investigated in Th2-associated models. ECDI-fixation covalently attaches antigen to splenocytes and induces apoptosis of Ag-SP, which are engulfed, processed and re-presented in a tolerogenic fashion by host splenic marginal zone antigen-presenting cells (APCs) (10, 11). The involvement of clonal anergy, via co-stimulatory molecule blockade and/or activation of negative costimulatory molecules such as PD-1 and CTLA-4, and activation of Treg populations, which regulate tolerance induction and maintenance, have been described as potential mechanisms of Ag-SP tolerance (8, 9, 11–13). Relevant to the potential use of Ag-SP tolerance for allergic diseases, we have shown that Ag-SP appears to be non-toxic and safe in EAE unlike soluble peptides, which often induce fatal anaphylaxis in myelin peptide-sensitized animals (14, 15). Additionally, a phase I/IIA clinical trial is being carried out by Dr. S. Miller in collaboration with Dr. Roland Martin at the University of Hamburg, Germany, (EudraCT #2008-004408-29) to examine the safety and efficacy of tolerance to a cocktail of myelin-derived peptides ECDI-fixed to autologous cells for treatment of new-onset multiple sclerosis.
Here we employ two murine models of allergy, a food allergy model to whole peanut extract (WPE) (16) and an ovalbumin (OVA)-induced allergic airway inflammation model (17), to investigate the efficacy of Ag-SP-induced tolerance to regulate allergic responses. In each model, Ag-SP tolerance prevented disease development and tolerized T cell responses in an antigen-specific manner in pre-sensitized animals. Prophylactically, Ag-SP decreased local and systemic Th2 responses, eosinophilia and Ag-specific IgE. Additionally, short-term induction of Ag-SP-induced tolerance was found to be independent of the function of CD25+ Tregs in the allergic airway inflammation, but partially dependent on Tregs in the food allergy model. The current experiments demonstrate that Ag-SP tolerance can be rapidly, safely and efficiently induced in murine models of Th2-associated disease, highlighting a potential new antigen-specific immunotherapy approach.
MATERIALS AND METHODS
Animals
4–8 week-old C3H/HeJ (Jackson Laboratory, Bar Harbor, ME) and BALB/c mice (Taconic Farms, Hudson, NY) were maintained in specific pathogen-free conditions at Northwestern University Center for Comparative Medicine. C3H/HeJ mice were maintained on a peanut-free diet. All protocols were approved by the Northwestern University Animal Care and Use Committee.
Reagents
PC61 antibody (anti-CD25, rIgG1) and isotype control (rIgG1) were purchased from Bio X Cell (West Lebanon, NH). Treg depletion/CD25 receptor downregulation was assessed by staining with 7D4 antibody (anti-CD25, rIgM) (18). WPE was prepared from unsalted, uncooked peanuts, as previously described (19). Staphylococcal enterotoxin B (SEB; Toxin Technology, Sarasota, FL), covered under federal select agents requirements, was used as approved by the Northwestern University Office of Research Safety under the requirements of the Centers for Disease Control and Prevention and the Department of Agriculture and according to necessary containment and use reporting mandates.
Food Allergy Model
Murine food allergy model was performed as previously described (16). Briefly, mice were given 50µg SEB with or without 100µg WPE by oral gavage once a week for eight weeks. Mice were orally challenged with 5mg/mouse WPE on week 9. Symptom scores and body temperature were determined after challenge as previously described (20, 21). Briefly, a score of 0 was assigned if no symptoms were evident, 1 represents mild scratching, rubbing, or both of the nose, head, or feet; 2 and 3 represent intermediate symptoms (e.g., edema around the eyes or mouth, pilar erection, and/or labored breathing); 4 represents significantly reduced motility, tremors, and/or significant respiratory distress; and 5 represents death. Symptom scores were determined by two investigators who were blinded to the experimental conditions.
Allergic Airway Inflammation Model
Mice were immunized i.p. with two doses of 10µg OVA (grade V, Sigma-Aldrich, St. Louis, MO) in alum (3mg) or alum and PBS alone. Mice were challenged for 20 min with aerosolized OVA (10mg/mL) for three consecutive days prior to tissue harvest (20).
Antigen Coupled-Cell Tolerance
Tolerance was induced by i.v. injection of ECDI-fixed, Ag-coupled splenocytes (Ag-SP), as previously described (10). Splenocytes from syngeneic mice were coupled to OVA, WPE, MBP (in all allergic airway inflammation studies) or BSA (in all food allergy model studies) (1mg/mL) via incubation with ECDI (150mg/3.2×108 cells; Calbiochem, San Diego, CA) for 1 h at 4°C followed by washing. 5×107 Ag-SP in 200µL PBS were injected i.v. per mouse.
Analysis of Bronchoalveolar Lavage and Peripheral Blood Cells
Lungs were flushed with bronchoalveolar lavage (BAL) fluid (1mM EDTA and 10%FCS in PBS). Total cell counts were determined and samples cytospun onto slides and DiffQuik (Siemens, Newark, DE) stained for differential cell counts (21). Blood was collected into EDTA-coated tubes, stained with Discombe’s fluid and eosinophils were counted using a hemocytometer (22).
Histology
Tissues were collected, fixed in formalin, embedded in paraffin and stained with hematoxylin and eosin (H&E) or periodic acid-Schiff (PAS) by Histo-Scientific Research Laboratories (Mount Jackson, VA).
Antigen-Specific Antibodies
Serum was collected upon sacrifice and tissue harvest and antigen-specific antibodies were quantified by sandwich ELISA as previously described (16).
OVA-specific Recall Responses
Mediastinal lymph nodes were harvested and incubated with OVA in serum-free HL-1 culture medium at 37°C for 48 h and pulsed with 1µCi/well [3H]TdR for the last 24 h of culture. Proliferation was determined by [3H]TdR incorporation as detected by a Topcount Microplate Scintillation Counter.
WPE-specific Recall Responses
Spleens were harvested and incubated with 100 µg/ml WPE in complete RPMI at 37°C for 72 h.
Cytokine Quantification
Cell-free BAL fluid and supernatants from recall response cultures were assayed for production of IL-4, IL-5, IL-13 and IFN-γ by cytometric bead array (CBA; BD Biosciences, San Jose, CA), Milliplex (Millipore, Billerica, MA) or ELISA. Serum mMCP-1 was quantified by mMCP-1 ELISA kit (eBioscience, CA).
Statistics
Statistics were performed on GraphPad Prism 5 software (GraphPad Software, La Jolla, CA) by using Student t test or 1-way ANOVA to determine significance.
RESULTS
Ag-SP tolerance prevents induction of allergic disease
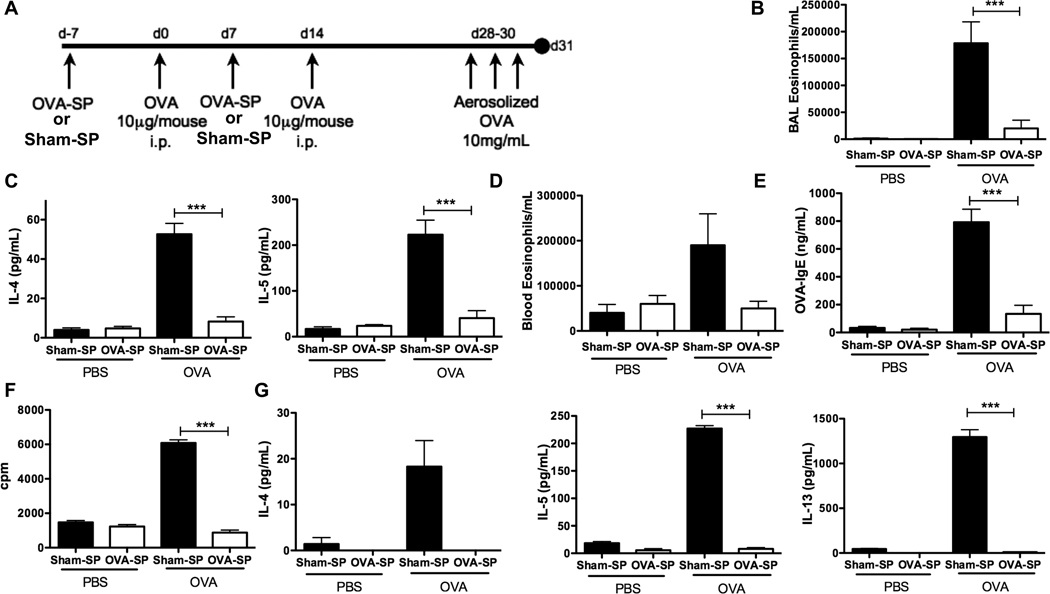
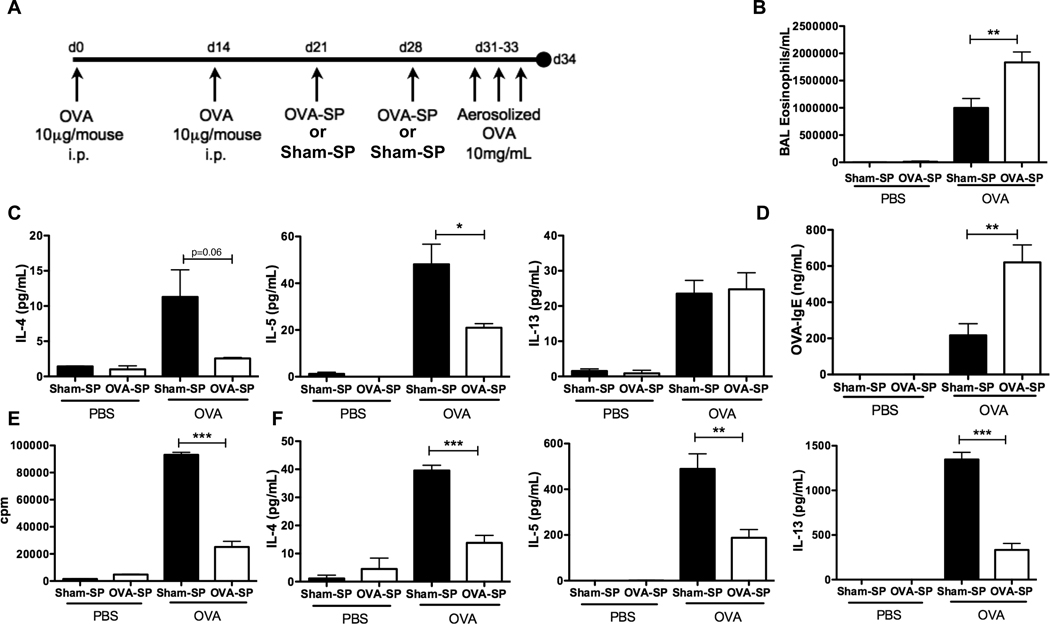
We investigated the ability of Ag-SP tolerance to prevent allergic responses by pre-treating mice with ECDI-fixed Ag-coupled splenocytes in two murine models of allergy: an OVA-induced allergic airway inflammation model and a food allergy model to whole peanut extract (WPE). In the allergic airway inflammation model, we administered OVA-coupled splenocytes (OVA-SP) prior to each of two sensitizations with OVA in alum adjuvant (Fig. 1A). We first examined the effect of OVA-SP as compared to Sham-SP on local allergic responses induced by an inhalation challenge of aerosolized OVA. Analysis of BAL fluid showed that OVA-SP treated mice had significantly reduced lung eosinophilia (Fig. 1B). Additionally, the Th2-associated cytokines IL-4 and IL-5 were significantly reduced in the BAL fluid of OVA-SP treated mice (Fig. 1C). The systemic allergic response was also inhibited, as blood eosinophil concentrations were reduced (Fig. 1D) and serum OVA-specific IgE levels were also dramatically reduced (Fig. 1E) in OVA-SP treated mice.
FIGURE 1. OVA-SP tolerance specifically inhibits Th2 responses in OVA/alum immunized mice.
A, BALB/c mice (n=5) were i.v. tolerized with OVA-SP or Sham (MBP)-SP, sensitized with OVA/alum and challenged as shown. B, BAL eosinophils were quantified by differential staining. C, IL-4 and IL-5 in BAL fluid were quantified by Milliplex. D, Peripheral blood eosinophils were quantified. E, Serum OVA-specific IgE was quantified by ELISA. F and G, Mediastinal lymphocytes were restimulated with 25µg/ml OVA, (F) proliferation was quantified by [3H]TdR incorporation and (G) IL-4, IL-5 and IL-13 were quantified by CBA. Results are mean ± SEM and representative of three experiments. ***p<0.001.
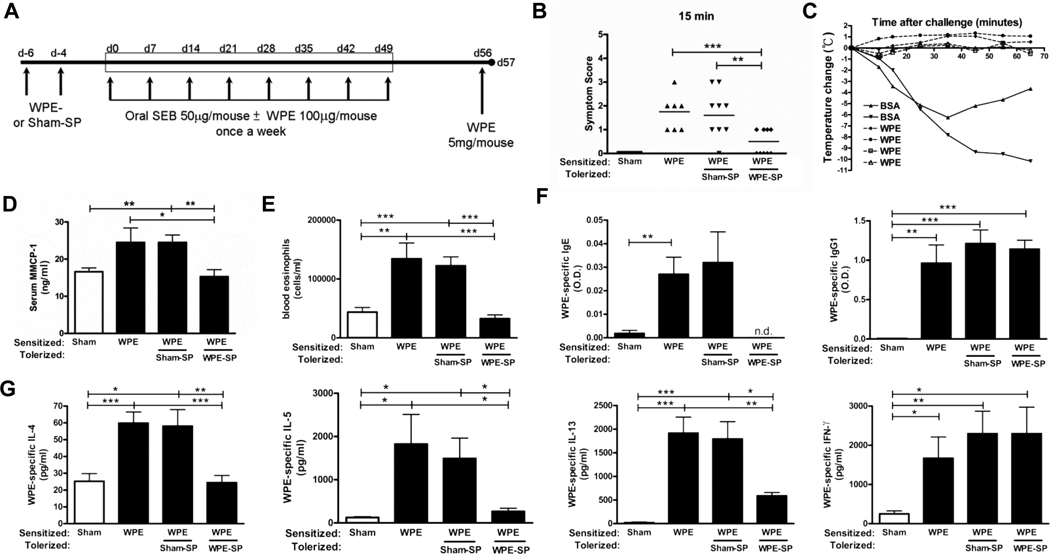
In the food allergy model, a model with a strong mast cell-mediated anaphylactic component, we administered WPE-coupled splenocytes (WPE-SP) prior to sensitization (Fig. 2A). 15 min after oral challenge with WPE, mice tolerized with WPE-SP showed significantly decreased symptom scores compared to Sham-SP tolerized mice (Fig. 2B). Furthermore, core body temperature was also measured after challenge since systemic anaphylaxis leads to vascular leak and hypothermia as a consequence of hypotension. Serum levels of mouse mast cell protease -1 (mMCP-1), a major protease released following mast cell activation, were measured after challenge as well.. We found that WPE-SP significantly prevented core temperature decreases (Fig. 2C) and mMCP-1 releas into serum (Fig. 2D) compared to Sham-SP controls. This indicated that WPE-SP prevents WPE-specific anaphylaxis in this model. As in the allergic airway inflammation model, WPE-SP tolerization also significantly reduced systemic allergic responses. Peripheral blood eosinophil numbers were significantly reduced (Fig. 2E) and serum WPE-specific IgE was undetectable in WPE-SP treated mice while WPE-specific IgG1 was unchanged (Fig. 2F).
FIGURE 2. WPE-SP tolerance specifically inhibits disease symptoms and Th2 responses in a food allergy model.
A, C3H/HeJ mice (n=7–10) were i.v. tolerized with WPE-SP or Sham (BSA-SP), sensitized and challenged with WPE as shown. B, 15 min after challenge, clinical symptoms were scored and (C) mouse body temperature was measured for 1 hr. D, Serum mMCP-1 collected 24 h after challenge was quantified by ELISA. E, Peripheral blood eosinophils were quantified. F, Serum WPE-specific IgE and IgG1 and (G) IL-4, IL-5, IL-13 and IFN-γ production from splenocytes restimulated with 100µg WPE were quantified by ELISA. Results are means ± SEM. from three experiments *p<0.05, **p<0.01, ***p<0.001, n.d.= not detectable.
Taken together, these results clearly demonstrate that, in addition to inhibiting Th1 and Th17 associated responses as previously published, Ag-SP tolerance can also prevent the induction of Ag-specific, Th2-associated allergic diseases.
Ag-SP tolerize antigen-specific Th2 cytokine responses
In the OVA-induced allergic airway inflammation model, restimulation of cells from mediastinal lymph nodes demonstrated that OVA-SP effectively tolerized OVA-specific T cells and prevented the induction of a Th2 response. Following OVA restimulation, lymphocytes from OVA-SP treated mice had significantly reduced proliferation compared to Sham-SP controls (Fig. 1F). These cells also produced significantly lower levels of IL-4, IL-5 and IL-13 upon OVA restimulation (Fig. 1G) with no effect on IFN-γ (data not shown). Similar data was seen after WPE restimulation of splenocytes from WPE-SP tolerized mice compared to Sham-SP controls in the food allergy model (Fig. 2G). These results indicate that Ag-SP tolerization inhibits the induction of Th2 cytokine responses in an antigen-specific manner.
Protective tolerance induced by Ag-SP is Treg independent in OVA-induced allergic airway inflammation but Tregs contribute to tolerance in peanut-induced food allergy
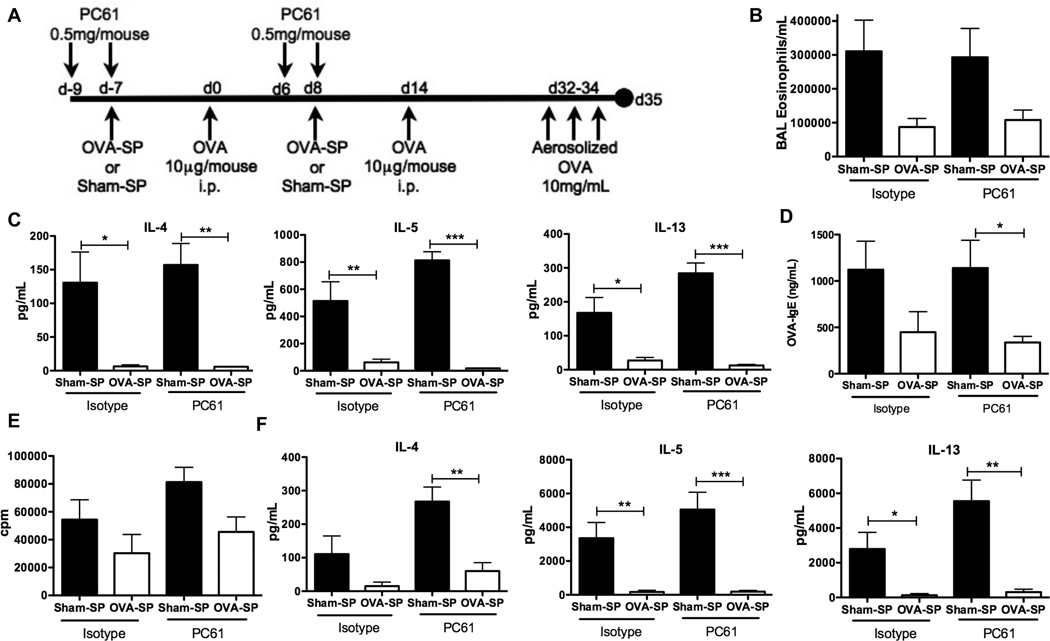
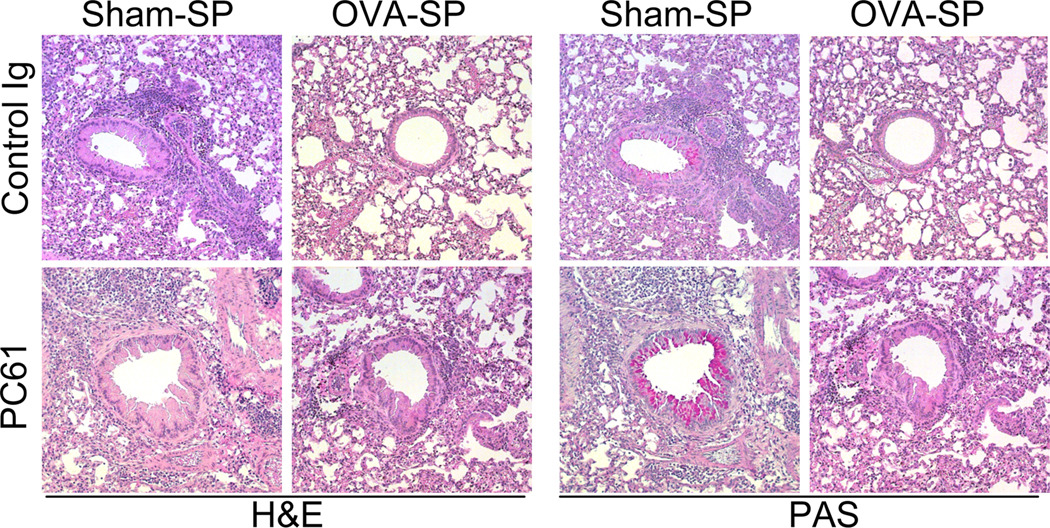
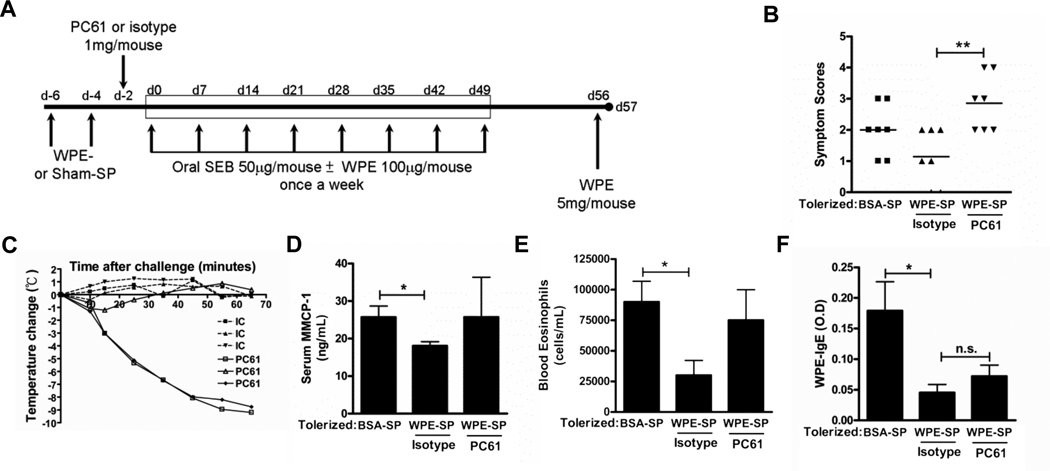
Our previous work in a model of allogeneic islet transplant rejection indicated an important role for Tregs in the induction of tolerance by Ag-SP (9). We thus investigated whether Tregs were also involved in the induction of tolerance by Ag-SP in the two murine models of allergic disease by treating mice with PC61 to functionally inactivate Tregs, using a similar treatment regimen to that we previously established will alter Ag-Sp responses in EAE (18). In the OVA-induced allergic airway inflammation model, we injected mice with PC61 around the time of tolerance (Fig. 3A). PC61-treated mice showed a trend of exacerbated allergic airway inflammation, as has been previously reported (23). However, OVA-SP tolerance reduced local inflammation in PC61-treated mice as effectively as in isotype control Ig-treated mice since eosinophils in the BAL fluid of PC61-treated mice were reduced by OVA-SP (Fig. 3B), indicating a Treg-independent mechanism of tolerance induction. Additionally, IL-4, IL-5 and IL-13 were also significantly reduced by OVA-SP in the BAL fluid of PC61-treated animals (Fig. 3C). Histological analysis of lung sections indicated that OVA-SP reduced cellular infiltrate and mucus secretion as shown by H&E and PAS staining, respectively (Fig. 4). Inhibition of the sensitization response by OVA-SP was also Treg-independent since OVA-SP significantly reduced serum OVA-specific IgE in PC61-treated animals (Fig. 3D). Restimulation of cells from mediastinal lymph nodes further demonstrated that OVA-SP induced tolerance of OVA-specific T cells in a Treg-independent manner. In PC61 treated mice, OVA-SP reduced proliferation following OVA restimulation of lymphocytes (Fig. 3E). These cells also produced significantly less IL-4, IL-5 and IL-13 upon OVA restimulation (Fig. 3F). Conversely, in the food allergy model, where we injected mice with PC61 shortly after the induction of tolerance (Fig. 5A), mice that received PC61 had significantly higher symptom scores (Fig. 5B) and, while one mouse exhibited only a modest decrease in body temperature, the other 2 mice tested had robust decreases in body temperature (Fig. 5C) compared to none of the isotype-treated controls. Mice receiving PC61 also exhibited a trend towards enhanced mMCP-1 in the serum (Fig 5D) and increased peripheral blood eosinophilia (Fig. 5E) versus isotype-treated controls, although not quite statistically significant. However, serum WPE-specific IgE (Fig. 5F) and IgG1 (data not shown) and IL-4, IL-5 and IL-13 produced from WPE-restimulated splenocytes (data not shown) were unaffected by Treg inactivation compared to isotype-treated controls. While the symptom scores between sham-treated and isotype-treated controls also failed to reach significance (p=0.08 by unpaired t-test), in all other readouts these groups were significantly different, reflective of our similar findings in Figure 2. Collectively, these results indicate that Tregs are partially responsible for the induction of tolerance by Ag-SP in the WPE-induced food allergy model, but not in an allergic OVA-induced airway inflammation model and imply that Tregs are one of several mechanism of tolerance induced by Ag-SP that control the allergic effector response. It also supports the mechanisms of allergic responses being different between models of airway disease and food allergy, with food allergy exhibiting a stronger dependence on Treg-mediated tolerance.
FIGURE 3. OVA-SP-induced tolerance is Treg-independent.
A, BALB/c mice (n=5) were treated with PC61 or control Ig i.p., tolerized, sensitized and challenged as shown. B, BAL eosinophils were quantified. C, IL-4, IL-5 and IL-13 in BAL supernatant were quantified by Milliplex. D Serum OVA-specific IgE was determined by ELISA. E and F, Mediastinal lymphocytes were restimulated with 25µg/mL OVA, (E) proliferation was quantified by [3H]TdR incorporation and (F) IL-4, IL-5 and IL-13 were quantified by Milliplex. Results are mean ± SEM and representative of three experiments. *p<0.05, **p<0.01, ***p<0.001.
FIGURE 4. OVA-SP tolerance inhibits OVA-induced airway inflammation in a Treg-independent fashion.
Mice were treated as indicated in Fig. 3A. Representative histology sections from mice sacrificed at day 35, stained with H&E or PAS as indicated, are shown at 200×.
FIGURE 5. WPE-SP-induced tolerance is partially Treg-dependent.
A, C3H/HeJ mice were tolerized, sensitized and challenged as shown. Mice were treated with 1mg/mouse PC61 or isotype at day -2. B, Symptom scores were determined 30 min after challenge (n=7, pooled from 3 independent experiments). C, mouse body temperature (n=3 per group, measured during one experiment from Panel B) and D, serum mMCP-1 (n=4–5, measured during one experiment from Panel B) were determined after challenge. E, Peripheral blood eosinophils were quantified (n=4–5, measured during one experiment from Panel B). F, Serum WPE-specific IgE was quantified by ELISA (n=8–9, pooled from 2 independent experiments). Results are mean ± SEM from three experiments. *p<0.05, **<0.01 by unpaired t-test, n.d.= not detectable.
Treatment with Ag-SP tolerizes the antigen-specific T cell response after disease onset
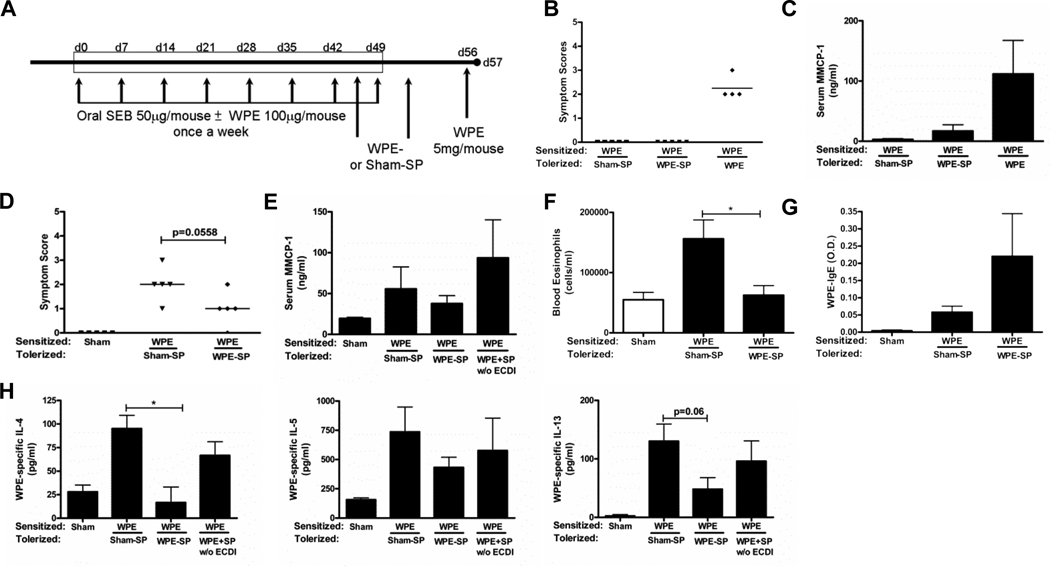
The primary clinical interest in allergic immunotherapy is in treating established hypersensitivity in an antigen-specific manner. Previous work in models of autoimmune disease has shown Ag-SP to be capable of inducing tolerance for amelioration of ongoing disease. Importantly, as anaphylaxis remains a major concern of immunotherapy therapy for food allergy, the inability of Ag-SP to trigger anaphylactic responses in sensitized mice has been established in autoimmune models of disease (14). We therefore sought to determine if Ag-SP could be similarly useful in a therapeutic context in allergic disease. To this end, we administered Ag-SP post-sensitization in the OVA-induced allergic airway inflammation model (Fig. 6A) and the food allergy model (Fig. 7A). In both models, Ag-SP treatment demonstrated significant reduction of most of the Th2-mediated cytokine release. Analysis of cytokine concentrations in the BAL fluid from the allergic airway sensitization model showed a significant reduction in IL-5, a reduction in IL-4 but no change in IL-13 of OVA-SP treated mice (Fig. 6C). Restimulation of cells from mediastinal lymph nodes in this model further demonstrated that, administered post-sensitization, OVA-SP tolerized the T cell response to OVA since lymphocytes from OVA-SP treated animals had significantly reduced proliferation to OVA restimulation compared to Sham-SP treated controls (Fig. 6E). These cells also produced significantly less IL-4, IL-5 and IL-13 upon OVA restimulation (Fig. 6F), demonstrating successful tolerization of Th2 cells in pre-sensitized mice. Similarly, WPE restimulation of splenocytes from WPE-SP tolerized mice in the food allergy model produced significantly less IL-4, IL-5 and IL-13 than seen in Sham-SP or soluble WPE controls (Fig. 7H). In response to tolerance induction, mice previously sensitized with WPE showed no anaphylactic symptoms and less mMCP-1 in serum while those receiving splenocytes with soluble WPE developed significant anaphylactic responses within 30 min (Fig. 7B) and higher mMCP-1 within 1 h (Fig. 7C). Upon subsequent challenge with WPE, mice tolerized with WPE-SP had only modestly lower symptom scores (Fig. 7D) and serum levels of mMCP-1 (Fig. 7E) when compared to Sham-SP controls. Peripheral blood eosinophil counts were also significantly lower in WPE-SP tolerized mice 24 h after challenge (Fig. 7F). Surprisingly, there was no correlative reduction in, and sometimes higher levels of, systemic Ag-specific IgE levels in both models (Fig. 6D, Fig. 7G) as well as an increase in BAL fluid lung eosinophilia in the allergic airway inflammation model relative to Sham-SP controls (Fig. 6B). However, WPE-specific IgG1 levels were unaltered (data not shown). Despite these findings, these studies indicate that Ag-SP treatment can tolerize antigen-specific Th2 responses and significant reduction of symptom scores in pre-sensitized recipients.
FIGURE 6. Treatment with OVA-SP post-OVA/alum sensitization specifically inhibits T cell responses.
A, BALB/c mice (n=5) were sensitized, tolerized and challenged as shown. B, BAL eosinophils were quantified. C, IL-4, IL-5 and IL-13 in BAL supernatant were quantified by CBA. D, Serum OVA-specific IgE was quantified by ELISA. F and G, Mediastinal lymphocytes were restimulated with 25ug/mL OVA, (F) proliferation was quantified by [3H]TdR incorporation and (G) IL-4, IL-5 and IL-13 were quantified by CBA. Results are mean ± SEM and representative of two experiments. *p<0.05, **p<0.01, ***p<0.001.
FIGURE 7. Treatment with WPE-SP post-WPE/SEB-sensitization safely alleviates disease severity.
A, WPE sensitized C3H/HeJ mice (n=4–5) were sensitized, tolerized and challenged as shown. B, Symptom scores were determined 30 min after the first WPE-SP tolerization. C, Serum mMCP-1 collected 1 h after tolerization was quantified by ELISA. D, 45 min after oral challenge, clinical symptoms were scored and (E) Serum mMCP-1 collected 24 h after challenge was quantified by ELISA. F, Peripheral blood eosinophils were quantified. G, Serum WPE-specific IgE and (H) IL-4, IL-5 and IL-13 production from splenocytes restimulated with 100µg WPE were quantified by ELISA. Results are mean ± SEM and representative of two experiments. *p<0.05.
DISCUSSION
The development of efficient, safe means of inducing specific tolerance to antigens is important in allergic disease research because many patients end conventional immunotherapy due to side effects and the long treatment courses, especially food allergy patients (24). We, and others, have previously demonstrated the ability of Ag-SP to prevent and treat Th1/Th17-dependent autoimmune disease and transplant rejection (7–9), including showing that Ag-SP tolerance inhibited the induction of allo-specific antibody production. The present study is the first to show the ability of Ag-SP to tolerize Th2 responses. Here we demonstrate the efficacy of Ag-SP treatment both in the prevention of allergic responses and the tolerization of pre-sensitized Th2-associated responses in two murine models of allergic disease. Our results indicate several key advantages of Ag-SP-induced tolerance over existing SITs including its exquisite antigen specificity and efficiency, requiring only two treatments with a total of only 30–40 µg of cell-bound Ag (data not shown), and, critically in the context of anaphylaxis, that it does not trigger adverse responses when administered to pre-sensitized animals.
We first examined the ability of Ag-SP to prevent the induction of Th2-mediated allergic disease using two different models that share some aspects of immune reactivity but differ in others. In a well-established OVA/alum-induced model of allergic airway inflammation, a strongly T cell-dependent model will little or no mast cell involvement, OVA-SP prevented local allergic inflammation. Recruitment of eosinophils to the lungs was inhibited, as was the production of local Th2 cytokines. In the WPE/SEB-induced food allergy model, a model with Th2-associated responses, but wherein mast cell-dependent anaphylaxis is a hallmark response, WPE-SP effectively inhibited the anaphylatic symptoms and drops in body temperature seen after oral WPE challenge. In both models, Ag-SP inhibited systemic eosinophilia and Ag-specific IgE. Interestingly, Ag-specific IgG1 (data not shown) was also reduced in the airway inflammation model but not in the food allergy model, perhaps relating to the different routes of immunization and adjuvants employed in the two models. Since IgE and IgG1 can induce anaphylactic responses by binding Fc receptors on mast cells (25), the presence of IgG1 in the food allergy model could explain the mild anaphylactic symptoms observed in the WPE-SP tolerized group. Indeed, IgE-deficient mice still exhibit anaphylactic-like reactivity (26). In addition to changes in responsiveness and antibodies, T cell cytokine responses were profoundly affected since restimulated lymphocytes failed to proliferate and did not produce Th2 cytokines. Importantly, Th1 or Th17 cytokines were not enhanced (Fig 2G and data not shown), indicating that Ag-SP induced Ag-specific tolerance to Th2 responses, rather than skewing responses towards an alternative T helper phenotype. This is consistent with our earlier work showing that Ag-SP inhibited Th1/Th17 cytokine production in EAE (27) and suggests that Ag-SP exerts tolerance towards a specific antigen regardless of the type of effector T cell response being generated in that model.
The mechanisms of Ag-SP-induced tolerance have been previously investigated in our Th1/Th17-mediated models of autoimmunity and transplant rejection. Tregs are critical for the induction of tolerance by Ag-SP in a model of alloantigen-specific islet cell transplantation (9), and for long-term tolerance maintenance in the EAE model (11, 27). Employing a similar Treg-inhibiting PC61 antibody treatment approach here, we show some evidence of Treg dependence of Ag-SP tolerance induction in allergic disease as well, most notably in our mast-cell dependent model of food allergy.
Interestingly, the two models showed significant differences in the dependence of Treg responses. In the OVA-induced model of allergic airway inflammation, tolerance of local inflammation by Ag-SP was Treg-independent, as was inhibition of a Th2 recall response from draining lymph nodes. These results in this acute model of inflammation are consistent with previous observations that the induction of tolerance by Ag-SP in an acute model of EAE is Treg-independent. In autoimmunity, we have showed that Ag-SP undergo apoptosis and are taken up and re-presented by host APCs in a tolerogenic fashion (10) and clonal anergy induced by costimulatory blockade as well as negative costimulation by molecules such as PD-1 and CTLA-4 are also important in the induction of tolerance (8, 9, 11–13). These mechanisms may contribute in allergic airway inflammation tolerance. However, we have also found that Tregs were critical for long-term maintenance of tolerance in EAE (11). Therefore, future studies will address the possibility that Tregs, while dispensable in the short-term for the induction of tolerance, may be required for long-term maintenance of tolerance in this model of allergic airway inflammation.
Conversely, in the peanut food allergy model, Ag-SP tolerance is Treg-dependent, as anaphylactic symptom scores were restored, body temperature drops observed again and slightly higher serum mMCP-1 was detected in Ag-SP treated mice receiving Treg inactivation. This model of allergy, and these readouts in particular, are mast cell dependent (16) while the OVA-induced model of airway inflammation is thought to be relatively mast cell independent (28). Mast cell degranulation releases several mediators that can cause anaphylaxis. Recently, it has been shown that Tregs can inhibit mast cell degranulation via OX40-OX40L interactions (29). Therefore, the enhanced symptom scores seen in PC61-treated mice may be due to the loss of mast cell inhibition by Tregs but this remains to be determined.
Similar in both models, we surprisingly observed that inhibition of Ag-specific IgE levels was unchanged by PC61 treatment, indicating that B cell tolerance by Ag-SP is Treg independent. One potential mechanism for this Treg independent B cell tolerance is the recognition of Ag in the absence of appropriate CD40 stimulation. CD40/CD40L interactions are required for B cell proliferation and class-switching (30). We recently demonstrated that CD4+ T cells isolated from Ag-SP treated mice fail to upregulate CD40L following in vitro Ag challenge and that treatment with a CD40 agonist abrogates Ag-SP-induced tolerance (31). We hypothesize that, in the absence of CD40L-mediated costimulation from T cells in Ag-SP treated mice, Ag-specific B cells will encounter Ag but fail to undergo class-switching, thus accounting for the Treg-independent reduction in IgE production.
Clinically, allergic immunotherapy must induce Ag-specific tolerance in sensitized subjects. A recent trial using an oral immunotherapy treatment for peanut allergy showed that most patients (93%) had adverse reactions to the treatment (5). We previously demonstrated that Ag-SP treatment can successfully ameliorate established EAE and that the treatment is safe and non-anaphylactic, unlike the administration of soluble Ag to mice with ongoing EAE (14). Crucially, in our food allergy model, Ag-SP did not induce anaphylaxis or release of mMCP-1 in sensitized mice. In both models of disease, Ag-SP successfully down-regulated the Th2 response in pre-sensitized mice in an antigen-specific manner, as indicated by decreased proliferation and Th2 cytokine production. However, within the time frame we examined, anaphylactic symptoms and serum level of mMCP-1 in response to Ag challenge in the food allergy model were only slightly reduced by Ag-SP treatment. Future experiments will address whether this efficient blockade of Th2 responses might lead to more significant losses in mast cell reactivity if more time and/or additional treatments with Ag-SP are provided before challenge. Interestingly, while blood eosinophilia was suppressed in food allergy, in our allergic airway inflammation model, however, lung eosinophilia was enhanced by Ag-SP treatment while blood eosinophilia was not significantly affected (data not shown). These differences may be due to the different sensitization protocols in each model. Alternatively, apoptotic Ag-SP traffic to the lungs in large numbers post i.v. injection (unpublished results) and may, in such close proximity to aerosolized Ag challenge, be responsible for enhanced eosinophilia.
In both models, Ag-SP treatment resulted in higher serum concentrations of Ag-specific IgE. This observation mirrors that of a recent oral immunotherapy study in which treatment initially enhanced serum IgE levels (32). However, IgE decreased in the following months. Due to the short time course of our studies, we are currently determining if a similar decrease in IgE will be observed over time. It is also possible that the increase in Ag-specific IgE is due to pre-sensitized B cells secreting IgE in response to direct or indirect re-presentation of Ag derived from apoptotic Ag-SP in a T-independent manner. Despite the increased IgE and eosinophilia seen here, we do not see a corresponding anaphylaxis response. The profound tolerization of the Ag-specific Th2 response under therapeutic conditions should effectively ameliorate the T cell help required for sustained production of allergic antibodies. Indeed, in other models, Ag-SP treatment has been demonstrated to induce long-lasting tolerance (27). Over a sufficient time course, as primed B cells turn over and as FcR-bound IgE and IgG1 antibodies decay, the lack of help from the tolerized T cell compartment should contribute to amelioration of disease. Additionally, Ag-SP treatment would be predicted to be more effective when used in combination with treatments such as anti-IgE therapies that target non-T cell-dependent arms of the allergic response (3).
In conclusion, our data establish that, in two models of Th2-associated allergic disease, Ag-SP treatment effectively prevents the induction of disease and safely and specifically tolerizes the Th2 response after onset of disease. Mechanistically, tolerance in the two models is differentially dependent on Tregs, with airway responses being largely unaffected by Treg inactivation while the prevention of mast cell-dependent anaphylaxis to peanut was Treg dependent. Importantly, the ability of Ag-SP to induce tolerance to mixed protein products such as WPE demonstrates its utility to tolerize responses against multiple allergic antigens, similar to our previously published demonstration that tolerization with Ag-SP linked to mouse spinal cord homogenate can effectively prevent EAE (27,33, 34). Collectively, our data demonstrate that Ag-SP treatment can induce protective Ag-specific tolerance in models of Th2-mediated immune responses and presents an attractive means of developing a safe and effective therapy to prevent and treat allergic disease.
Acknowledgments
This work was supported by NIH R01 Grants NS-026543 (SDM), NS-030871 (SDM), AI-076456 (PJB) and support from the Food Allergy Initiative (PJB).
ABBREVIATIONS
- Ag-SP
antigen-coupled splenocytes
- ECDI
1-ethyl-3-(3’-dimethylaminopropyl)-carbodiimide
- LN
lymph nodes
- SEB
Staphylococcal enterotoxin B
- Treg
regulatory T cell
- WPE
whole peanut extract
- BAL
bronchoalveolar lavage
REFERENCES
- 1.Cousins DJ, Lee TH, Staynov DZ. Cytokine coexpression during human Th1/Th2 cell differentiation: direct evidence for coordinated expression of Th2 cytokines. J. Immunol. 2002;169:2498–2506. doi: 10.4049/jimmunol.169.5.2498. [DOI] [PubMed] [Google Scholar]
- 2.Georas SN, Guo J, De Fanis U, Casolaro V. T-helper cell type-2 regulation in allergic disease. Eur. Respir. J. 2005;26:1119–1137. doi: 10.1183/09031936.05.00006005. [DOI] [PubMed] [Google Scholar]
- 3.Holgate ST, Djukanovic R, Casale T, Bousquet J. Anti-immunoglobulin E treatment with omalizumab in allergic diseases: an update on anti-inflammatory activity and clinical efficacy. Clin. Exp. Allergy. 2005;35:408–416. doi: 10.1111/j.1365-2222.2005.02191.x. [DOI] [PubMed] [Google Scholar]
- 4.Holgate ST, Polosa R. Treatment strategies for allergy and asthma. Nat. Rev. Immunol. 2008;8:218–230. doi: 10.1038/nri2262. [DOI] [PubMed] [Google Scholar]
- 5.Hofmann AM, Scurlock AM, Jones SM, Palmer KP, Lokhnygina Y, Steele PH, Kamilaris J, Burks AW. Safety of a peanut oral immunotherapy protocol in children with peanut allergy. J. Allergy Clin. Immunol. 2009;124:286–291. doi: 10.1016/j.jaci.2009.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Miller SD, Wetzig RP, Claman HN. The induction of cell-mediated immunity and tolerance with protein antigens coupled to syngeneic lymphoid cells. J. Exp. Med. 1979;149:758–773. doi: 10.1084/jem.149.3.758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vanderlugt CL, Eagar TN, Neville KL, Nikcevich KM, Bluestone JA, Miller SD. Pathologic role and temporal appearance of newly emerging autoepitopes in relapsing experimental autoimmune encephalomyelitis. J. Immunol. 2000;164:670–678. doi: 10.4049/jimmunol.164.2.670. [DOI] [PubMed] [Google Scholar]
- 8.Fife BT, Guleria I, Gubbels Bupp M, Eagar TN, Tang Q, Bour-Jordan H, Yagita H, Azuma M, Sayegh MH, Bluestone JA. Insulin-induced remission in new-onset NOD mice is maintained by the PD-1-PD-L1 pathway. J. Exp. Med. 2006;203:2737–2747. doi: 10.1084/jem.20061577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Luo X, Pothoven KL, McCarthy D, DeGutes M, Martin A, Getts DR, Xia G, He J, Zhang X, Kaufman DB, Miller SD. ECDI-fixed allogeneic splenocytes induce donor-specific tolerance for long-term survival of islet transplants via two distinct mechanisms. Proc. Natl. Acad. Sci. USA. 2008;105:14527–14532. doi: 10.1073/pnas.0805204105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Turley DM, Miller SD. Peripheral tolerance Induction using ethylenecarbodiimide-fixed APCs uses both direct and indirect mechanisms of antigen presentation for prevention of experimental autoimmune encephalomyelitis. J. Immunol. 2007;178:2212–2220. doi: 10.4049/jimmunol.178.4.2212. [DOI] [PubMed] [Google Scholar]
- 11.Getts DR, Turley DM, Smith CE, Harp CT, McCarthy D, Feeney EM, Getts MT, Martin AJ, Luo X, Terry RL, King NJ, Miller SD. Tolerance induced by apoptotic antigen-coupled leukocytes is induced by PD-L1+ and IL-10-producing splenic macrophages and maintained by T regulatory cells. J. Immunol. 2011;187:2405–2417. doi: 10.4049/jimmunol.1004175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eagar TN, Turley DM, Padilla J, Karandikar NJ, Tan LJ, Bluestone JA, Miller SD. CTLA-4 regulates expansion and differentiation of Th1 cells following induction of peripheral T cell tolerance. J. Immunol. 2004;172:7442–7450. doi: 10.4049/jimmunol.172.12.7442. [DOI] [PubMed] [Google Scholar]
- 13.Jenkins MK, Schwartz RH. Antigen presentation by chemically modified splenocytes induces antigen-specific T cell unresponsiveness in vitro and in vivo. J. Exp. Med. 1987;165:302–319. doi: 10.1084/jem.165.2.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Smith CE, Eagar TN, Strominger JL, Miller SD. Differential induction of IgE-mediated anaphylaxis after soluble vs. cell-bound tolerogenic peptide therapy of autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. USA. 2005;102:9595–9600. doi: 10.1073/pnas.0504131102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Smith CE, Miller SD. Multi-peptide coupled-cell tolerance ameliorates ongoing relapsing EAE associated with multiple pathogenic autoreactivities. J. Autoimmunity. 2006;27:218–231. doi: 10.1016/j.jaut.2006.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ganeshan K, Neilsen CV, Hadsaitong A, Schleimer RP, Luo X, Bryce PJ. Impairing oral tolerance promotes allergy and anaphylaxis: A new murine food allergy model. J. Allergy Clin. Immunol. 2008;123:231–238. doi: 10.1016/j.jaci.2008.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gueders MM, Paulissen G, Crahay C, Quesada-Calvo F, Hacha J, Van Hove C, Tournoy K, Louis R, Foidart JM, Noel A, Cataldo DD. Mouse models of asthma: a comparison between C57BL/6 and BALB/c strains regarding bronchial responsiveness, inflammation, and cytokine production. Inflamm. Res. 2009;58:845–854. doi: 10.1007/s00011-009-0054-2. [DOI] [PubMed] [Google Scholar]
- 18.Kohm AP, McMahon JS, Podojil JR, Smith Begolka W, DeGutes M, Kasprowicz DJ, Ziegler SF, Miller SD. Cutting Edge: Anti-CD25 mAb injection results in the functional inactivation, not depletion of CD4+CD25+ Treg cells. J. Immunol. 2006;176:3301–3305. doi: 10.4049/jimmunol.176.6.3301. [DOI] [PubMed] [Google Scholar]
- 19.Koppelman SJ, Knol EF, Vlooswijk RA, Wensing M, Knulst AC, Hefle SL, Gruppen H, Piersma S. Peanut allergen Ara h 3: isolation from peanuts and biochemical characterization. Allergy. 2003;58:1144–1151. doi: 10.1034/j.1398-9995.2003.00259.x. [DOI] [PubMed] [Google Scholar]
- 20.Bryce PJ, Mathias CB, Harrison KL, Watanabe T, Geha RS, Oettgen HC. The H1 histamine receptor regulates allergic lung responses. J. Clin. Invest. 2006;116:1624–1632. doi: 10.1172/JCI26150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bryce PJ, Geha R, Oettgen HC. Desloratadine inhibits allergen-induced airway inflammation and bronchial hyperresponsiveness and alters T-cell responses in murine models of asthma. J. Allergy Clin. Immunol. 2003;112:149–158. doi: 10.1067/mai.2003.1616. [DOI] [PubMed] [Google Scholar]
- 22.Colley DG. Schistosoma mansoni: eosinophilia and the development of lymphocyte blastogenesis in response to soluble egg antigen in inbred mice. Exp. Parasitol. 1972;32:520–526. doi: 10.1016/0014-4894(72)90070-7. [DOI] [PubMed] [Google Scholar]
- 23.Lewkowich IP, Herman NS, Schleifer KW, Dance MP, Chen BL, Dienger KM, Sproles AA, Shah JS, Kohl J, Belkaid Y, Wills-Karp M. CD4+CD25+ T cells protect against experimentally induced asthma and alter pulmonary dendritic cell phenotype and function. J. Exp. Med. 2005;202:1549–1561. doi: 10.1084/jem.20051506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Thyagarajan A, Varshney P, Jones SM, Sicherer S, Wood R, Vickery BP, Sampson H, Burks AW. Peanut oral immunotherapy is not ready for clinical use. J. Allergy Clin. Immunol. 2010;126:31–32. doi: 10.1016/j.jaci.2010.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Silva SR, Casabuono A, Jacysyn JF, Favoretto BC, Fernandes I, Macedo MS, Couto AS, Faquim-Mauro EL. Sialic acid residues are essential for the anaphylactic activity of murine IgG1 antibodies. J. Immunol. 2008;181:8308–8314. doi: 10.4049/jimmunol.181.12.8308. [DOI] [PubMed] [Google Scholar]
- 26.Oettgen HC, Martin TR, Wynshaw-Boris A, Deng C, Drazen JM, Leder P. Active anaphylaxis in IgE-deficient mice. Nature. 1994;370:367–370. doi: 10.1038/370367a0. [DOI] [PubMed] [Google Scholar]
- 27.Miller SD, Turley DM, Podojil JR. Antigen-specific tolerance strategies for the prevention and treatment of autoimmune disease. Nat. Rev. Immunol. 2007;7:665–677. doi: 10.1038/nri2153. [DOI] [PubMed] [Google Scholar]
- 28.Takeda K, Hamelmann E, Joetham A, Shultz LD, Larsen GL, Irvin CG, Gelfand EW. Development of eosinophilic airway inflammation and airway hyperresponsiveness in mast cell-deficient mice. J. Exp. Med. 1997;186:449–454. doi: 10.1084/jem.186.3.449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gri G, Piconese S, Frossi B, Manfroi V, Merluzzi S, Tripodo C, Viola A, Odom S, Rivera J, Colombo MP, Pucillo CE. CD4+CD25+ regulatory T cells suppress mast cell degranulation and allergic responses through OX40-OX40L interaction. Immunity. 2008;29:771–781. doi: 10.1016/j.immuni.2008.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kawabe T, Naka T, Yoshida K, Tanaka T, Fujiwara H, Suematsu S, Yoshida N, Kishimoto T, Kikutani H. The immune responses in CD40-deficient mice: impaired immunoglobulin class switching and germinal center formation. Immunity. 1994;1:167–178. doi: 10.1016/1074-7613(94)90095-7. [DOI] [PubMed] [Google Scholar]
- 31.Martin AJ, McCarthy D, Waltenbaugh C, Goings G, Luo X, Miller SD. Ethylenecarbodiimide-treated splenocytes carrying male CD4 epitopes confer Hya transplant protection by inhibiting CD154 upregulation. J. Immunol. 2010;185:3326–3336. doi: 10.4049/jimmunol.1000802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jones SM, Pons L, Roberts JL, Scurlock AM, Perry TT, Kulis M, Shreffler WG, Steele P, Henry KA, Adair M, Francis JM, Durham S, Vickery BP, Zhong X, Burks AW. Clinical efficacy and immune regulation with peanut oral immunotherapy. J. Allergy Clin. Immunol. 2009;124:292–300. doi: 10.1016/j.jaci.2009.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kennedy MK, Dal Canto MC, Trotter JL, Miller SD. Specific immune regulation of chronic-relapsing experimental allergic encephalomyelitis in mice. J. Immunol. 1988;141:2986–2993. [PubMed] [Google Scholar]
- 34.Kennedy MK, Tan LJ, Dal Canto MC, Miller SD. Regulation of the effector stages of experimental autoimmune encephalomyelitis via neuroantigen-specific tolerance induction. J. Immunol. 1990;145:117–126. [PubMed] [Google Scholar]