Abstract
The cyclic β-(1→3),β-(1→6)-d-glucan synthesis locus of Bradyrhizobium japonicum is composed of at least two genes, ndvB and ndvC. Mutation in either gene affects glucan synthesis, as well as the ability of the bacterium to establish a successful symbiotic interaction with the legume host soybean (Glycine max). B. japonicum strain AB-14 (ndvB::Tn5) does not synthesize β-glucans, and strain AB-1 (ndvC::Tn5) synthesizes a cyclic β-glucan lacking β-(1→6)-glycosidic bonds. We determined that the structure of the glucan synthesized by strain AB-1 is cyclodecakis-(1→3)-β-d-glucosyl, a cyclic β-(1→3)-linked decasaccharide in which one of the residues is substituted in the 6 position with β-laminaribiose. Cyclodecakis-(1→3)-β-d-glucosyl did not suppress the fungal β-glucan-induced plant defense response in soybean cotyledons and had much lower affinity for the putative membrane receptor protein than cyclic β-(1→3),β-(1→6)-glucans produced by wild-type B. japonicum. This is consistent with the hypothesis presented previously that the wild-type cyclic β-glucans may function as suppressors of a host defense response.
Many bacteria in the family Rhizobiaceae enter the roots of their legume host plants via specialized structures known as infection threads. This leads to the formation of a new organ, the root nodule, in which rhizobia undergo differentiation to become bacteroids that fix nitrogen into ammonia. Rhizobial extracellular polysaccharides are critical for an effective symbiosis in many rhizobial species and in many cases appear to have a structure-specific role in symbiosis (Leigh and Walker, 1994; Becker and Pühler, 1998). EPS, LPS, β-glucans, and K-antigen-type polysaccharides are the major categories of extracellular carbohydrates that have been studied (Hotter and Scott, 1991; Petrovics et al., 1993; Gonzalez et al., 1996; Breedveld and Miller, 1998). In several instances, rhizobia with alterations in the structure of one of these carbohydrates are defective in their ability to invade the plant. Such mutants typically fail to form abundant infection threads and/or fail to persist and differentiate inside of the host (Leigh and Walker, 1994; Cheng and Walker, 1998). The abortion of infection threads appears to be caused by the accumulation of plant phenolic compounds such as flavonoids and in some cases phytoalexins (Niehaus et al., 1993; Vasse et al., 1993; Parniske et al., 1994). On the other hand, no significant induction in chalcone synthase, chalcone isomerase, or isoflavone reductase transcripts was observed during the early stages of the alfalfa-Sinorhizobium meliloti symbiosis when either wild-type or EPS mutants were used as inoculum (McKhann et al., 1997).
Why more than one carbohydrate entity is required for the development of a successful symbiosis is not understood. Some are required for initiation and elongation of infection threads (Battisti et al., 1992; Cheng and Walker, 1998), whereas other extracellular molecules may be important for avoiding elicitation of a host defense (Ahlborn and Werner, 1991; Niehaus et al., 1993, 1998; McKhann and Hirsch, 1994; Spaink, 1995). Cyclic β-(1→3),(1→6)-glucans of Bradyrhizobium japonicum are predominantly periplasmic molecules that are required for this bacterial species' growth under hypoosmotic conditions (Tully et al., 1990; Rolin et al., 1992; Pfeffer et al., 1994). They also may have a specific function during symbiotic interactions with the legume hosts (Breedveld and Miller, 1994, 1998).
We have isolated and characterized the cyclic glucan synthesis locus from B. japonicum, and have identified two genes (ndvB and ndvC) by creating site-specific Tn5 mutations in the chromosome (Bhagwat and Keister, 1995; Bhagwat et al., 1996). Mutations within the ndvB locus (strain AB-14) resulted in total absence of cyclic glucan synthesis, whereas mutations within the ndvC locus (strain AB-1) resulted in synthesis of cyclic glucans with predominantly β-(1→3)-glycosyl linkages. Strain AB-14 was defective in motility and growth in hypoosmotic medium, and formed ineffective but differentiated nodules on soybean plants (Bhagwat and Keister, 1995). The β-(1→3)-linked cyclic glucans produced by strain AB-1 supported motility and growth under low osmolarity but were unable to promote effective symbiosis with soybean. An 8- to 10-d delay in nodulation was observed with the ndvC mutant of B. japonicum, and very small nodule-like structures (pseudonodules) that were devoid of viable bacteria were formed (Bhagwat et al., 1996; Dunlap et al., 1996). This suggested that the structure of the cyclic β-glucan molecule is important for a successful symbiotic interaction and that the cyclic β-glucans may have a specific function in addition to their role in hypoosmotic adaptation.
The cyclic β-(1→3),(1→6)-glucans of B. japonicum share some structural features with the noncyclic hepta β-(1→3),(1→6)-glucan fragments derived from the mycelial walls of fungal pathogens of soybean (Ayers et al., 1976). These molecules are potent elicitors of phytoalexins (glyceollins in soybean). However, the cyclic β-glucans of wild-type B. japonicum are only very weak elicitors of glyceollin production in soybean (Miller et al., 1994; Mithöfer et al., 1996a). The structural similarity between the two glucan species prompted us to investigate the ability of these compounds to interact with the putative β-glucan membrane-receptor protein and also to induce a defense response in the soybean host. We previously observed that induction of phytoalexin synthesis by fungal β-glucans was suppressed by bradyrhizobial glucans (Mithöfer et al., 1996a). Ultrastructural analysis of nodule-like structures induced on soybean roots by strain AB-1 (ndvC) revealed morphological features similar to those found in plants exhibiting a defense response to pathogen attack (Dunlap et al., 1996). These observations lead us to propose that cyclic β-(1→3),β-(1→6)-glucans of B. japonicum may have the novel role of suppressing a host defense response during rhizobial invasion.
MATERIALS AND METHODS
Bacteria and Culture Conditions
Bradyrhizobium japonicum strains USDA 110, AB-1, and AB-14 (Bhagwat and Keister, 1995; Bhagwat et al., 1996) were grown on arabinose gluconate medium (Cole and Elkan, 1973).
Glucan Isolation and Purification
Glucans from B. japonicum were extracted from cell pellets as described previously (Bhagwat et al., 1996), with minor modifications. Cell pellets were extracted with 75% ethanol at 70°C and the cell debris was pelleted by centrifugation at 12,000g for 10 min. The glucans in the supernatant were precipitated by increasing the ethanol concentration to 90% (final concentration) and incubating at −20°C overnight. The glucans were pelleted at 12,000g for 10 min, dissolved in water, and dialyzed for 3 d against distilled water at 4°C. The glucans were purified by passage through a DEAE-cellulose column and absorption on C18 silica gel (Bhagwat et al., 1996).
The fungal β-glucan elicitor was prepared by partial acid hydrolysis from purified cell walls of Phytophthora sojae according to the method of Schmidt and Ebel (1987). The branched β-glucan elicitor fraction used had 420 μg of Glc equivalents and 25 μg protein mg−1.
Bacteroids were isolated from nodules of 4-week-old soybean (Glycine max) cv 9007 plants grown in Leonard jars (Keister and Marsh, 1990). Glucans were extracted from bacteroids with 75% ethanol and purified as described above.
LPS Analysis
LPS B. japonicum strains were isolated by hot-phenol extraction (Johnson and Perry, 1976) and were analyzed by SDS-PAGE (deMaagd et al., 1988). The LPS bands were visualized by silver staining (Tsai and Frasch, 1982).
Oligosaccharide Analysis by HPAEC-PAD
Conditions for HPAEC-PAD were reported previously (Pfeffer et al., 1996). An HPLC system (model 4500, Dionex, Sunnyvale, CA) that included a CarboPac PA1 column (4 × 250 mm), a CarboPac PA Guard column (4 × 50 mm), and a pulsed electrochemical detector using a linear sodium acetate gradient (5–375 mm) in a 100 mm NaOH mobile phase was used for analysis of cyclic β-glucan samples.
Fast-Atom Bombardment Mass-Spectral Analysis
Mass spectra were obtained with a magnetic-sector mass spectrometer (model VGZAB-2SE/FPD, VG Analytical, Manchester, UK) by fast-atom bombardment ionization at 8 keV using a thioglycerol-m-nitrobenzoic acid matrix (Pfeffer et al., 1996).
NMR Spectroscopy
13C-NMR spectroscopy was performed using a spectrometer (Unity Plus 400, Varian, Sugarland, TX) at 35°C. The samples were exchanged at least twice in 2H2O before obtaining the spectra in 2H2O. Approximately 20,000 scans were obtained at a 70° pulse for one-dimensional spectra, with a repetition time of 1.8 s (Pfeffer et al., 1996).
Nodulation Studies
Soybean seeds obtained from Pioneer (Ames, IA) were surface sterilized using 1.25% NaHClO4. Plants were grown in Leonard jar assemblies (Bhagwat et al., 1992) and were watered with N-free nutrient solution. Nodules were harvested 4 weeks after inoculation with B. japonicum.
Binding Assays
Soybean root membrane proteins were prepared by Zwittergent 3-12 solubilization (Mithöfer et al., 1996b). Solubilized root membrane protein (150 μg) was incubated with increasing concentrations of either cyclic β-(1→3),(1→6)-glucans from the wild-type strain USDA 110 or cyclic β-(1→3)-glucans from the ndvC mutant strain AB-1 in the presence of 3 nm 125I-labeled HG-APEA in a final volume of 200 μL of a buffer consisting of 25 mm Tris-HCl, pH 8.0, 100 mm NaCl, 10 mm MgCl2, and 5 mm d-gluconic acid lactone for 2 h at 4°C (Cosio et al., 1990a). Values for half-maximal displacement were calculated by nonlinear regression using the Marquardt-Levenberg algorithm (Cosio et al., 1996). Synthesis of HG-APEA was performed as described by Cosio et al. (1990b).
Biological Activity Assays
Detached cotyledons from 5-d-old soybean seedlings were cut, and an aliquot of either fungal β-glucan, bradyrhizobial cyclic β-glucan, or a mixture of fungal and bradyrhizobial glucans was placed on wounded areas (Ayers et al., 1976). Each measurement was done at least in triplicate using 10 to 12 cotyledons per data point. The cotyledons were incubated for 22 h at 27°C on moist filter paper in Petri dishes in the dark. Phytoalexin accumulation in the wound-droplet solutions was determined by measuring the A285. The response of the cotyledons was calculated as A/Amax, where Amax represents the value obtained from 200 μg mL−1 fungal β-glucans.
Analysis of Glyceollin
Lyophilized roots and nodules from 9 to 12 plants were extracted twice with ethyl acetate (15 mL). The organic phase was dried over Na2SO4 and removed under reduced pressure. The residue was dissolved in 200 μL of ethanol and chromatographed by reverse-phase HPLC using a LiChrosorb RP-18 column as described by Kraus et al. (1995). Identification and quantification of glyceollins from nodules were done using reference compounds by GC and mass-spectrometric analysis (Kraus et al., 1995).
RESULTS
Structure of Cellular Glucans Produced by the ndvC Mutant
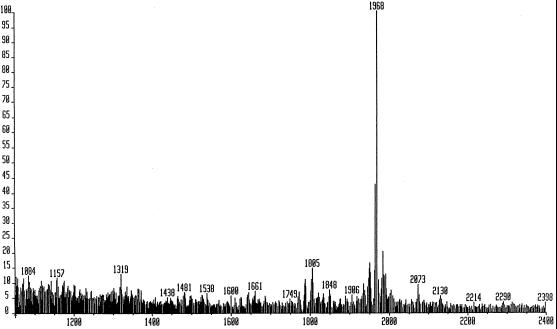
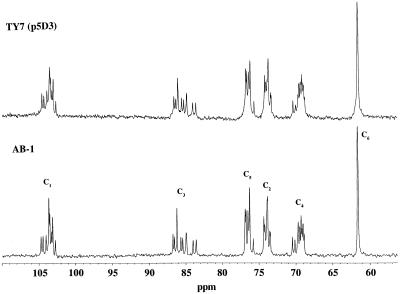
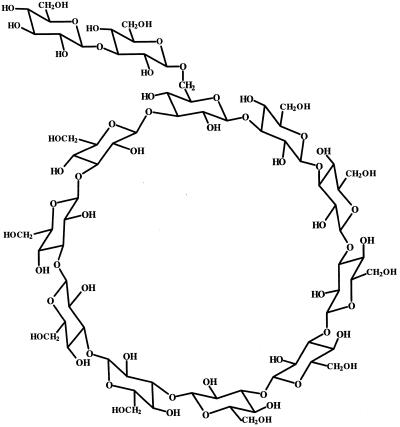
The fast-atom bombardment mass spectrum of the purified glucan displayed an [M+Na]+ at 1968 (Fig. 1), consistent with a molecule containing 12 glucosidically linked hexose units with no reducing end. The one-dimensional 13C-NMR spectrum of glucans from strain AB-1 (Fig. 2) showed a dispersion of each Glc carbon type (C1 to C6) in the molecule. The C1 resonances were observed in the range from δ 102.8 to δ 104.8. That all linkages are of the δ configuration was verified because no 13C anomeric resonances at a field higher than δ 102.8 were observed. Typically, anomeric carbon resonances of α-1,3 Glc units are observed at approximately δ 100.0 in acyclic and cyclic glucans (Usui et al., 1973; Cote and Biely, 1994). The 13C-NMR spectrum of glucans from strain AB-1 (Fig. 2) is identical to the spectrum observed for the recently characterized cyclic β-(1→3)-glucan produced by the Sinorhizobium meliloti ndvB mutant TY7 carrying the β-glucan synthesis locus p5D3 from B. japonicum USDA 110 (also shown in Fig. 2 for comparison) (Pfeffer et al., 1996). In addition, elution profiles of these glucans were identical using HPAEC-PAD, with one major peak eluting at approximately 69 min. Elution of mixed samples gave a single peak at 69 min as well (data not shown). Based on these observations, the glucan synthesized by strain AB-1 appears to be identical to the branched β-(1→3)-linked cyclic glucan cyclodecakis-(1→3)-β-d-glucosyl recently described by us (previously named cyclolaminarinose; Pfeffer et al., 1996). The proposed structure of the molecule (Fig. 3) is a cyclic decasaccharide in which one of the residues is substituted in the 6 position with β-laminaribiose.
Figure 1.
Fast-atom bombardment mass ionization spectrum of glucans from B. japonicum ndvC strain AB-1. The m/z values are reported as the nominal masses of the molecular ions plus Na (M+23). degree of polymerization11 = 1805; degree of polymerization12 = 1968.
Figure 2.
13C-NMR spectra (100 MHz) of glucans from B. japonicum ndvC strain AB-1 and from S. meliloti TY7 (ndvB) carrying the glucan locus from B. japonicum on cosmid clone p5D3. The assignment of each carbon resonance is indicated.
Figure 3.
Proposed structure of the β-(1→3) cyclic glucan, cyclodecakis-(1→3)-β-d-glucosyl, from B. japonicum ndvC mutant AB-1.
LPS Analysis of ndv Mutants
The nodulation phenotype of the ndv mutants is similar to that of some EPS and LPS mutants of rhizobia (Niehaus et al., 1993; Parniske et al., 1994). Therefore, we examined the LPS content of ndvB and ndvC mutants. Electrophoresis of LPS on SDS-polyacrylamide gels revealed no significant differences between USDA 110 and ndv mutants (Fig. 4). The LPS from all of the strains fractionated into approximately 12 bands, as visualized by silver staining. As observed previously, the colony morphology and the synthesis of capsular polysaccharides (EPS) by strains AB-1 and AB-14 were not significantly different from those of USDA 110 (Bhagwat et al., 1996).
Figure 4.
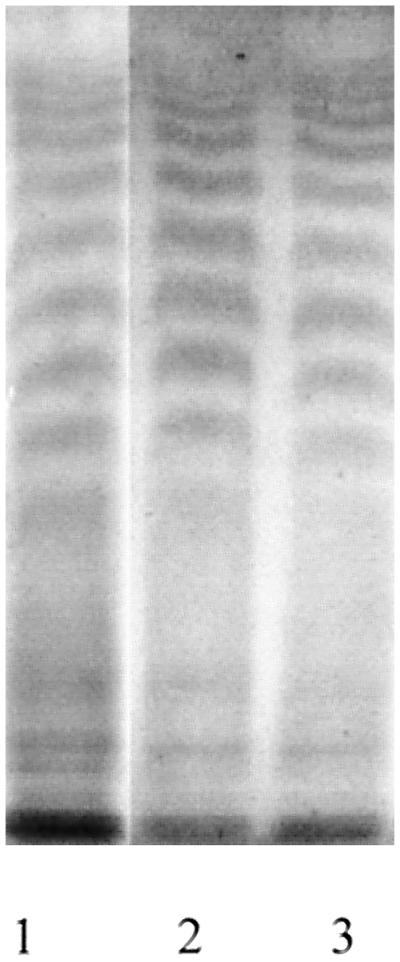
Profile of LPS after SDS-PAGE visualized by silver staining. B. japonicum strains AB-1 (lane 1), AB-14 (lane 2), and USDA 110 (lane 3).
Cyclodecakis-(1→3)-β-d-Glucosyl and Host Defense Response
Next, we analyzed soybean nodules for glyceollins I, II, and III after inoculation with B. japonicum strains USDA 110, AB-1, and AB-14 (Table I). Nodules induced by wild-type strain USDA 110 and uninoculated roots had low levels of glyceollin. The nodules induced by strain AB-14, although well differentiated and morphologically advanced, had almost a 3-fold higher glyceollin level than nodules induced by USDA 110. The glyceollin level was more than 5-fold higher in the pseudonodules induced by strain AB-1. Strain AB-1 also was delayed in nodulation, and nodule morphogenesis was aborted at a much earlier stage compared with strain AB-14.
Table I.
Glyceollin contents of soybean nodules formed by various B. japonicum strains
| Strain | Cyclic β-Glucans
|
Glyceollins (I, II, and III) | |
|---|---|---|---|
| Cultured cells | Bacteroids | ||
| μg mg−1 proteina | μg g−1 nodule dry wt | ||
| Uninoculated roots | –b | –b | 2.1 ± 1.9 |
| USDA 110 | 76 | 32.4 | 3.7 ± 0.8 |
| AB-14 (ndvB) | 0 | 0 | 9.9 ± 0.6 |
| AB-1 (ndvC) | 51 | –b | 18.5 ± 2.6 |
Average of two experiments.
Bacteroids could not be isolated.
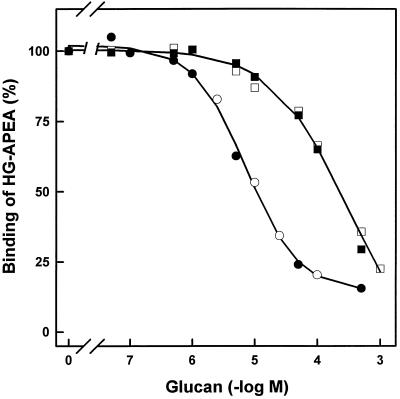
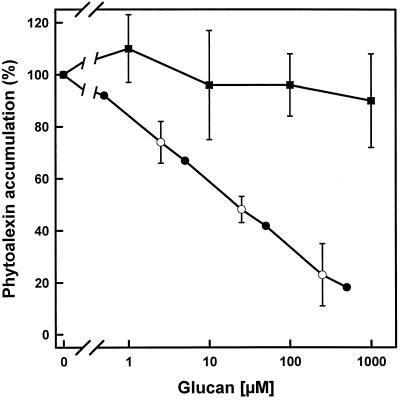
The binding of cyclodecakis-(1→3)-β-d-glucosyl from AB-1 to the soybean β-glucan receptor was analyzed in competition experiments with the 125I-labeled HG-APEA. HG-APEA has been shown to have characteristics of fungal β-glucans, such as specific, reversible, and saturable binding to the putative receptor protein from soybean (Ebel and Cosio, 1994). As shown in Figure 5, cyclic β-(1→3),(1→6)-glucans from USDA 110, as well as cyclodecakis-(1→3)-β-d-glucosyl from strain AB-1, competed with the radiolabeled ligand for binding to a solubilized membrane fraction from soybean roots. The glucans from the wild-type strain were 40 times more effective at inhibiting fungal hepta-β-glucoside binding than cyclodecakis-(1→3)-β-d-glucosyl. The values for half-maximal displacement were 7.3 μm for bradyrhizobial glucans from the wild-type strain and 308 μm for cyclodecakis-(1→3)-β-d-glucosyl. The ability of cyclodecakis-(1→3)-β-d-glucosyl to suppress the fungal β-glucan-induced phytoalexin synthesis was very poor compared with bradyrhizobial wild-type glucans. As illustrated in Figure 6, the concentration of cyclic β-(1→3),(1→6)-glucans giving 50% inhibition of the phytoalexin response elicited by the fungal β-glucans was about 35 μm, whereas even with 1 mm cyclodecakis-(1→3)-β-d-glucosyl, the suppression was less than 25%.
Figure 5.
Displacement of 125I-labeled HG-APEA by increasing concentrations of B. japonicum cyclic glucans from solubilized soybean glucan-binding proteins. Maximal HG-APEA binding was set to 100%, representing an average value of 0.36 pmol mg−1 protein. Independent experiments are denoted by different symbols. Cyclic β-(1→3),(1→6)-glucans from B. japonicum USDA 110 (•, ○) or cyclodecakis-(1→3)-β-d-glucosyl from strain AB-1 (ndvC) (▪, □). Values for half-maximal displacement were calculated by nonlinear regression using the Marquardt-Levenberg algorithm (see Methods).
Figure 6.
Effect of increasing concentrations of cyclic glucans of B. japonicum on phytoalexin accumulation in soybean cotyledons induced by a fixed concentration of fungal β-(1→3),(1→6)-branched glucans (2 μg mL−1, set to 100%). The average value of A/Amax at this concentration of fungal glucan alone was 0.81. Cyclic β-(1→3),(1→6)-glucans from B. japonicum USDA 110 (•, ○) or cyclodecakis-(1→3)-β-d-glucosyl from strain AB-1 (ndvC) (▪). Independent experiments are denoted by different symbols.
DISCUSSION
Two genes from B. japonicum, ndvB and ndvC, that are required for cyclic β-(1→3),(1→6)-glucan synthesis have been identified. Mutation in either ndvB or ndvC results in a symbiotically defective microsymbiont. Mutation in ndvB abolishes glucan synthesis and mutation in ndvC results in synthesis of a structurally altered glucan (Bhagwat and Keister, 1995; Bhagwat et al., 1996). Based on HPAEC-PAD, 13C-NMR, and MS analysis, the cyclic glucan produced by strain AB-1 (ndvC−) is identical to cyclodecakis-(1→3)-β-d-glucosyl, the cyclic β-(1→3)-linked glucan produced by the ndvB mutant of S. meliloti [TY7(p5D3)] carrying the glucan synthesis locus from B. japonicum (Pfeffer et al., 1996). Soybeans inoculated with strain AB-1 developed pseudonodules with some ultrastructural features (thickened cell walls, dense cytoplasm, and large vacuoles) that frequently are observed in tissues as a result of pathogen invasion (Dunlap et al., 1996). Although some of the ultrastructural features of the nodules are similar to those produced by B. japonicum mutants defective in capsular polysaccharides or LPS, ndv mutant strains AB-1 and AB-14 appear to synthesize normal LPS (Fig. 4). There was also no significant difference observed in the amount of capsular polysaccharides synthesized by the ndv mutants and USDA 110 (Bhagwat et al., 1996). Similarly, no discernible differences with respect to polysaccharides other than the cyclic β-(1→2)-glucans were reported for S. meliloti ndv mutants (Ielpi et al., 1990). Thus, we believe that the nodule phenotypes of ndv mutants are primarily attributable to the effects of the alterations in glucan synthesis or structure.
As calculated from the data shown in Figure 5, cyclodecakis-(1→3)-β-d-glucosyl was about 40 times less effective (half-maximal displacement was 308 μm) than the wild-type glucans (half-maximal displacement was 7.3 μm) in displacing a radiolabeled heptaglucoside ligand from solubilized soybean root membrane protein. A protein has been identified and characterized from soybean cell membranes that binds the fungal β-(1→3),(1→6)-heptaglucan elicitor rather specifically. Thus, it meets several of the criteria for a receptor-specific binding protein (Hahn, 1996; Ebel and Mithöfer, 1998). Cyclodecakis-(1→3)-β-d-glucosyl reversed the binding of the radiolabeled heptaglucoside, although much less effectively than the wild-type glucans (Fig. 5). This glucan was even less effective in suppressing the fungal β-glucan-induced defense response in cotyledons (Fig. 6). This may be attributable to differences in the signal-perception pathways in roots versus cotyledons. The interaction of cyclic β-(1→3),(1→6)-glucans from wild-type B. japonicum with this receptor protein and the inhibition of the defense response in cotyledons suggest that the soybean perception system that recognizes β-glucans may be generally involved in the recognition of foreign organisms. Several laboratories are currently working to characterize the receptor and signaling system, but at this time a role for this receptor is unknown.
One paradox for which we do not have a good explanation is that cyclodecakis-(1→3)-β-d-glucosyl synthesis in the S. meliloti genetic background [TY7(p5D3)] complemented the ndvB mutant phenotype, resulting in effective nodulation of alfalfa (Bhagwat et al., 1993; Pfeffer et al., 1996), whereas the same cyclic glucan produced by B. japonicum strain AB-1 did not result in an effective symbiosis with soybean (Bhagwat et al., 1996). This suggests that there is a different (and less specific) structural requirement for the glucan molecule in alfalfa compared with soybean nodule morphogenesis. This difference may be related to the host plant. Extracellular polysaccharide-deficient mutants of a broad host range Rhizobium result in different symbiotic phenotypes on different hosts with either indeterminant or determinant nodulation characteristics (Djordjevic et al., 1987; Hotter and Scott, 1991; Becker and Pühler, 1998).
Wild-type rhizobia do not elicit a significant host-plant defense response. Whether the lack of response is caused by structural isolation (of rhizobia in infection threads and symbiosomes), the inability of host plants to perceive elicitor compounds from rhizobia, or the production of a suppressor is controversial. Recently, we reported that the cyclic β-(1→3),(1→6)-glucans produced by wild-type B. japonicum were poor elicitors of a host defense response in soybean cotyledons compared with fungal β-glucan elicitors (Mithöfer et al., 1996a; see also Miller et al., 1994). Perhaps more significant is the observation that the cyclic β-(1→3),(1→6)-glucans of wild-type USDA 110 suppressed the host defense response induced by fungal β-glucans in soybean cotyledons (Mithöfer et al., 1996a). This led us to postulate that these molecules function as suppressors. The data presented in Table I indicate that nodules induced by the AB-14 mutant, which does not make any glucans, accumulated glyceollin. Surprisingly, nodules induced by the AB-1 mutant, which makes cyclodecakis-(1→3)-β-d-glucosyl, accumulated even more glyceollin. One explanation for this finding is that cyclodecakis-(1→3)-β-d-glucosyl may function as an elicitor, albeit a weak one. This could also explain why the nodules induced by AB-1 are arrested at an earlier stage of development.
Glyceollins are known to be antibacterial for B. japonicum in micromolar concentrations (Parniske et al., 1991a). Similarly, exudate preparations from roots infected with P. sojae had a strong bactericidal effect on B. japonicum. Thus, suppression of phytoalexin synthesis may be necessary for the formation of an effective symbiosis. The levels of glyceollin in nodules induced by the B. japonicum ndv mutants are much lower compared with the level (43 μg/g fresh weight) in fungal elicitor-treated roots (Kraus et al., 1995). We hypothesize that bacteroids in the developing nodules may be more sensitive to phytoalexins than free-living cells, and thus the observed glyceollin levels may be adequate to prevent normal nodule development when a suppressor is absent. Studies are planned to examine this possibility.
In nodules induced by mutants of Rhizobium leguminosarum bv viciae defective in LPS, chalcone synthase genes were expressed at high levels in the invasion zone, whereas in the wild-type nodules only very low levels of chalcone synthase mRNA were detected (Yang et al., 1992). In pseudonodules induced by the EPS I− mutants of S. meliloti, Niehaus et al. (1993) observed enhanced levels of phenolic compounds. This led the authors to suggest that Sinorhizobium meliloti EPS I acted as a suppressor of plant defense responses (Niehaus et al., 1998). Some mutants and ineffective B. japonicum strains are reported to induce higher levels of defense responses (Werner et al., 1985; Parniske et al., 1991b). This implies that there is an endogenous elicitor, but the role and the nature of a putative endogenous elicitor from rhizobia have not been investigated. Considering the genetic diversity between Sinorhizobium fredii and B. japonicum strains, it is likely that the endogenous elicitor, the corresponding host receptor, and the putative suppressor in these strains would be different. Based on our results, we believe that cyclic β-glucans may serve as suppressors in the interaction with soybean.
ACKNOWLEDGMENTS
We thank Susan Fogarty and Ramin Samadani for excellent technical assistance.
Abbreviations:
- EPS
exopolysaccharide(s)
- HG-APEA
2-(4-aminophenyl)ethylamine conjugate of the hepta-β-glucoside
- HPAEC-PAD
high-performance anion-exchange chromatography-pulsed amperometric detection
- LPS
lipopolysaccharide(s)
Footnotes
This research was supported in part by award 96 35305 3731 to A.A.B. and D.L.K. from the U.S. Department of Agriculture-National Research Initiative Competitive Research Grants Program, by the Deutsche Forschungsgemeinschaft (SFB 369) to J.E., and by the Binational National Science Foundation-Deutsche Academic Exchange Service (Germany) visiting scientist exchange program (J.E., A.M., and A.A.B.).
LITERATURE CITED
- Ahlborn B, Werner D. Inhibition of 1,3-β-glucan synthase from Glycine max and Pisum sativum by exopolysaccharides of Bradyrhizobium japonicum and Rhizobium leguminosarum. Physiol Mol Plant Pathol. 1991;39:299–307. [Google Scholar]
- Ayers AR, Ebel J, Finelli F, Berger N, Albersheim P. Host-pathogen interactions. IX. Quantitative assays of elicitor activity and characterization of the elicitor present in the extracellular medium of cultures of Phytophthora megasperma var. sojae. Plant Physiol. 1976;57:751–759. doi: 10.1104/pp.57.5.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Battisti L, Lara JC, Leigh JA. A specific oligosaccharide form of the Rhizobium meliloti exopolysaccharide promotes nodule invasion in alfalfa. Proc Natl Acad Sci USA. 1992;89:5625–5629. doi: 10.1073/pnas.89.12.5625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker A, Pühler A. Production of exopolysaccharides. In: Spaink HP, Kondorosi A, Hooykaas JJ, editors. The Rhizobiaceae. Dordrecht, The Netherlands: Kluwer Academic Publishers; 1998. pp. 97–118. [Google Scholar]
- Bhagwat AA, Gross KC, Tully RE, Keister DL. β-Glucan synthesis in Bradyrhizobium japonicum: characterization of a new locus (ndvC) influencing β-(1→6)-linkages. J Bacteriol. 1996;178:4635–4642. doi: 10.1128/jb.178.15.4635-4642.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhagwat AA, Keister DL. Site-directed mutagenesis of the cyclic β-(1→3)(1→6)-glucan synthesis locus of Bradyrhizobium japonicum. Mol Plant Microbe Interact. 1995;8:366–370. [Google Scholar]
- Bhagwat AA, Tully RE, Keister DL. Isolation and characterization of an ndvB locus from Rhizobium fredii. Mol Microbiol. 1992;6:2159–2165. doi: 10.1111/j.1365-2958.1992.tb01389.x. [DOI] [PubMed] [Google Scholar]
- Bhagwat AA, Tully RE, Keister DL. Identification and cloning of cyclic β-(1→3),β-(1→6)-d-glucan synthesis locus from Bradyrhizobium japonicum. FEMS Microbiol Lett. 1993;114:139–144. doi: 10.1111/j.1574-6968.1993.tb06564.x. [DOI] [PubMed] [Google Scholar]
- Breedveld MW, Miller KJ. Cyclic β-glucans of members of the family Rhizobiaceae. Microbiol Rev. 1994;58:145–161. doi: 10.1128/mr.58.2.145-161.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breedveld MW, Miller KJ. Cell-surface β-glucans. In: Spaink HP, Kondorosi A, Hooykaas JJ, editors. The Rhizobiaceae. Dordrecht, The Netherlands: Kluwer Academic Publishers; 1998. pp. 81–96. [Google Scholar]
- Cheng HP, Walker GC. Succinoglycan is required for initiation and elongation of infection threads during nodulation of alfalfa by Rhizobium meliloti. J Bacteriol. 1998;180:5183–5191. doi: 10.1128/jb.180.19.5183-5191.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole MA, Elkan GH. Transmissible resistance to penicillin G, neomycin, chloramphenicol in Rhizobium japonicum. Antimicrob Agents Chemother. 1973;4:248–253. doi: 10.1128/aac.4.3.248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cosio EG, Feger M, Miller CJ, Antelo L, Ebel J. High-affinity binding of fungal β-glucan elicitors to cell membranes of species of the plant family Fabaceae. Planta. 1996;200:92–99. [Google Scholar]
- Cosio EG, Frey T, Ebel J. Solubilization of soybean membrane binding sites for β-glucans that elicit phytoalexin accumulation. FEBS Lett. 1990a;264:235–238. doi: 10.1016/0014-5793(90)80256-i. [DOI] [PubMed] [Google Scholar]
- Cosio EG, Frey T, Verduyn R, van Boom J, Ebel J. High-affinity binding of a synthetic heptaglucoside and fungal glucan phytoalexin elicitors to soybean membranes. FEBS Lett. 1990b;271:223–226. doi: 10.1016/0014-5793(90)80411-b. [DOI] [PubMed] [Google Scholar]
- Cote G, Biely P. Enzymically produced cyclic β-1,3-linked and β-1,6-linked oligosaccharides of d-glucose. Eur J Biochem. 1994;226:641–648. doi: 10.1111/j.1432-1033.1994.tb20091.x. [DOI] [PubMed] [Google Scholar]
- deMaagd RA, vanRossum C, Lugtenberg BJJ. Recognition of individual strains of fast-growing rhizobia by using profiles of membrane proteins and lipopolysaccharides. J Bacteriol. 1988;170:3782–3785. doi: 10.1128/jb.170.8.3782-3785.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Djordjevic SP, Chen H, Bately M, Redmond JW, Rolfe BG. Nitrogen fixation ability of exopolysaccharide synthesis mutants of Rhizobium sp. strain NGR234 and Rhizobium trifolii is restored by the addition of homologous exopolysaccharides. J Bacteriol. 1987;169:53–60. doi: 10.1128/jb.169.1.53-60.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunlap J, Minami E, Bhagwat AA, Keister DL, Stacey G. Nodulin gene expression by nodule development (ndv) mutants of Bradyrhizobium japonicum. Mol Plant Microbe Interact. 1996;9:546–555. doi: 10.1094/mpmi-9-0546. [DOI] [PubMed] [Google Scholar]
- Ebel J, Cosio EG. Elicitors of plant defense responses. Int Rev Cytol. 1994;148:1–36. [Google Scholar]
- Ebel J, Mithöfer A. Early events in the elicitation of plant defense. Planta. 1998;206:335–348. [Google Scholar]
- Gonzalez JE, Reuhs BL, Walker GC. Low molecular weight EPS II of Rhizobium meliloti allows nodule invasion in Medicago sativa. Proc Natl Acad Sci USA. 1996;93:8636–8641. doi: 10.1073/pnas.93.16.8636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hahn MG. Microbial elicitors and their receptors in plants. Annu Rev Plant Physiol Plant Mol Biol. 1996;34:387–412. doi: 10.1146/annurev.phyto.34.1.387. [DOI] [PubMed] [Google Scholar]
- Hotter GS, Scott DB. Exopolysaccharide mutants of Rhizobium loti are fully effective on a determinate nodulating host but are ineffective on an indeterminate nodulating host. J Bacteriol. 1991;173:851–859. doi: 10.1128/jb.173.2.851-859.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ielpi L, Dylan T, Ditta GS, Helinski DR, Stanfield SW. The ndvB locus of Rhizobium meliloti encodes a 319-kDa protein involved in the production of β-(1→2)-glucan. J Biol Chem. 1990;265:2843–2851. [PubMed] [Google Scholar]
- Johnson KG, Perry MB. Improved techniques for the preparation of bacterial lipopolysaccharides. Can J Microbiol. 1976;22:29–34. doi: 10.1139/m76-004. [DOI] [PubMed] [Google Scholar]
- Keister DL, Marsh SS. Hemoproteins of Bradyrhizobium japonicum cultured cells and bacteroids. Appl Environ Microbiol. 1990;56:2736–2741. doi: 10.1128/aem.56.9.2736-2741.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraus C, Spiteller G, Mithöfer A, Ebel J. Quantification of glyceollins in non-elicited seedlings of Glycine max by gas chromatography-mass spectrometry. Phytochemistry. 1995;40:739–743. [Google Scholar]
- Leigh JA, Walker GC. Exopolysaccharides of Rhizobium: synthesis, regulation and symbiotic function. Trends Genet. 1994;10:63–67. doi: 10.1016/0168-9525(94)90151-1. [DOI] [PubMed] [Google Scholar]
- McKhann HI, Hirsch AM. Does Rhizobium avoid the host response? In: Glick BR, Thompson JE, editors. Methods in Plant Molecular Biology and Biotechnology. Boca Raton, FL: CRC Press; 1994. pp. 179–205. [Google Scholar]
- McKhann HI, Paiva NL, Dixon RA, Hirsch AM. Chalcone synthase transcripts are detected in alfalfa root hairs following inoculation with wild-type Rhizobium meliloti. Mol Plant Microbe Interact. 1997;10:50–58. [Google Scholar]
- Miller KJ, Hadley JA, Gustine DL. Cyclic β-(1,6)(1,3)-glucans of Bradyrhizobium japonicum USDA 110 elicit isoflavonoid production in the soybean host. Plant Physiol. 1994;104:917–923. doi: 10.1104/pp.104.3.917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mithöfer A, Bhagwat AA, Feger M, Ebel J. Suppression of fungal β-glucan-induced plant defence in soybean (Glycine max L.) by cyclic 1,3–1,6-β-glucans from the symbiont Bradyrhizobium japonicum. Planta. 1996a;199:270–275. [Google Scholar]
- Mithöfer A, Lottspeich F, Ebel J. One-step purification of the β-glucan elicitor-binding protein from soybean (Glycine max L.) roots and characterization of an anti-peptide antiserum. FEBS Lett. 1996b;381:203–207. doi: 10.1016/0014-5793(96)00126-3. [DOI] [PubMed] [Google Scholar]
- Niehaus K, Albus U, Baier R, Schiene K, Schröder S, Pühler A. Symbiotic suppression of the Medicago sativa plant defence system by Rhizobium meliloti oligosaccharides. In: Elmerich C, Kondorosi A, Newton WE, editors. Biological Nitrogen Fixation for the 21st Century. Dordrecht, The Netherlands: Kluwer Academic Publishers; 1998. pp. 225–226. [Google Scholar]
- Niehaus K, Kapp D, Pühler A. Plant defense and delayed infection of alfalfa pseudonodules induced by an exopolysaccharide (EPS-I)-deficient Rhizobium meliloti mutant. Planta. 1993;190:415–425. [Google Scholar]
- Parniske M, Ahlborn B, Werner D. Isoflavonoid-inducible resistance to the phytoalexin glyceollin in soybean rhizobia. J Bacteriol. 1991a;173:3432–3439. doi: 10.1128/jb.173.11.3432-3439.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parniske M, Fischer HM, Hennecke H, Werner D. Accumulation of the phytoalexin glyceollin I in soybean nodules infected by a Bradyrhizobium japonicum nifA mutant. Z Naturforsch. 1991b;46c:318–320. [Google Scholar]
- Parniske M, Schmidt PE, Kosch E, Müller P. Plant defense responses of host plants with determinate nodules induced by EPS-defective exoB mutants of Bradyrhizobium japonicum. Mol Plant Microbe Interact. 1994;5:631–638. [Google Scholar]
- Petrovics G, Putnoky P, Reuhs B, Kim J, Thorp TA, Noel KD, Carlson RW, Kondorosi A. The presence of a novel type of surface polysaccharide in Rhizobium meliloti requires a new fatty acid synthase-like gene cluster involved in symbiotic nodule development. Mol Microbiol. 1993;8:1083–1094. doi: 10.1111/j.1365-2958.1993.tb01653.x. [DOI] [PubMed] [Google Scholar]
- Pfeffer PE, Becard G, Rolin DB, Uknalis J, Cook P, Tu S. In vivo nuclear magnetic resonance study of the osmoregulation of phosphocholine-substituted β-1,3;1,6 cyclic glucan and its associated carbon metabolism in Bradyrhizobium japonicum USDA 110. Appl Environ Microbiol. 1994;60:2137–2146. doi: 10.1128/aem.60.6.2137-2146.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeffer PE, Osman SF, Hotchkiss A, Bhagwat AA, Keister DL, Valentine KM. Cyclolaminarinose: a new biologically active β-1,3 cyclic glucan. Carbohydr Res. 1996;296:23–37. doi: 10.1016/s0008-6215(96)00238-8. [DOI] [PubMed] [Google Scholar]
- Rolin DB, Pfeffer PE, Osman SF, Szwergold BS, Kappler F, Benesi AJ. Structural studies of a phosphocholine substituted β-(1,3);(1,6) macrocyclic glucan from Bradyrhizobium japonicum USDA 110. Biochim Biophys Acta. 1992;1116:215–225. doi: 10.1016/0304-4165(92)90014-l. [DOI] [PubMed] [Google Scholar]
- Schmidt WE, Ebel J. Specific binding of a fungal glucan phytoalexin elicitor to membrane fractions from soybean Glycine max. Proc Natl Acad Sci USA. 1987;84:4117–4121. doi: 10.1073/pnas.84.12.4117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spaink HP. The molecular basis of infection and nodulation by rhizobia: the ins and outs of sympathogenesis. Annu Rev Phytopathol. 1995;33:345–368. doi: 10.1146/annurev.py.33.090195.002021. [DOI] [PubMed] [Google Scholar]
- Tsai C-M, Frasch CE (1982) A sensitive silver stain for detecting lipopolysaccharides in polyacrylamide gels. Anal Biochem 119: 115–119 [DOI] [PubMed]
- Tully RE, Keister DL, Gross KC. Fractionation of the β-linked glucans of Bradyrhizobium japonicum and their response to osmotic potential. Appl Environ Microbiol. 1990;56:1518–1522. doi: 10.1128/aem.56.6.1518-1522.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Usui T, Yamaoka N, Matsuda K, Tuzimura K, Sugiyama H, Seto S (1973) 13C nuclear magnetic resonance spectra of glucobiose, glucotriose, and glucans. J Chem Soc Perkin Trans 2425–2432
- Vasse J, deBilly F, Truchet G. Abortion of infection during the Rhizobium meliloti-alfalfa symbiotic interaction is accompanied by a hypersensitive reaction. Plant J. 1993;4:555–566. [Google Scholar]
- Werner D, Mellor RB, Hahn MG, Grisebach H. Soybean root response to symbiotic infection glyceollin I accumulation in an ineffective type of soybean nodules with an early loss of the peribacteroid membrane. Z Naturforsch. 1985;40c:179–181. [Google Scholar]
- Yang WC, Canter-Cremers HCJ, Hogendrijk P, Katinakis P, Wijffelman CA, Franssen H, van Kammen A, Bisseling T. In-situ localization of chalcone synthase mRNA in pea root nodule development. Plant J. 1992;2:143–151. [Google Scholar]