Abstract
Dietary folate is essential in all tissues to maintain several metabolite pools and cellular proliferation. Prostate cells, due to specific metabolic characteristics, have increased folate demand to support proliferation and prevent genetic and epigenetic damage. Although several studies found that dietary folate interventions can affect colon cancer biology in rodent models, impact on prostate is unknown. The purpose of this study was to determine if dietary folate manipulation, possibly being of primary importance for prostate epithelial cell metabolism, could significantly affect prostate cancer (CaP) progression. Strikingly, mild dietary folate depletion arrested CaP progression in 25/26 transgenic TRAMP mice, where tumorigenesis is prostate specific and characteristically aggressive. The significant effect on CaP growth was characterized by size, grade, proliferation and apoptosis analyses. Folate supplementation had a mild, non significant beneficial effect on grade. In addition, characterization of folate pools (correlated with serum), metabolite pools (polyamines, nucleotides), genetic and epigenetic damage, and expression of key biosynthetic enzymes in prostate tissue revealed interesting correlations with tumor progression. These findings indicate that CaP is highly sensitive to folate manipulation and suggest that antifolates, paired to current therapeutic strategies, might significantly improve treatment of CaP, the most commonly diagnosed cancer in American men.
Keywords: Prostate cancer, TRAMP, folate, one-carbon metabolism, polyamine biosynthesis
Introduction
Folate (vitamin B9) is essential to sustain proliferation in all cell types because it is part of a complex metabolism that ultimately provides de novo deoxythymidine triphosphate (dTTP) and s-adenosylmethionine (AdoMet) (Fig. 1). In turn, dTTP is necessary for DNA synthesis, while AdoMet is the universal intracellular methyl donor for methylases including DNA and histone methylases. For this reason antifolate therapy has been used for decades to treat certain types of cancer (1). Conversely, folate supplementation, by supporting the maintenance of these pools, might prevent genetic and epigenetic damage and the consequent development of neoplasia. Importantly, AdoMet is also utilized, through decarboxylation, for the biosynthesis of polyamines (2, 3) (Fig. 1). We previously demonstrated that heightened polyamine biosynthesis, consequent to prostate epithelial cells’ secretory function, draws on AdoMet pools (Fig. 1) and makes prostate cancer (CaP) cell growth in vitro extremely dependent on folate (4). Nonetheless, the effects of dietary manipulation of folate on the prostate in vivo are currently unknown.
Figure 1. Folate, one-carbon metabolism, methionine cycle and polyamine biosynthesis overview.
Dietary folate (left) is necessary for de novo synthesis of deoxythymidine monophosphate (dTMP) and adenosylmethionine (AdoMet), which in turn are necessary for DNA synthesis and intracellular methylation reactions, respectively. AdoMet is also used for polyamine biosynthesis (top), which is prominent in prostate cells. dTMP can also be salvaged as thymidine from DNA degradation. The key enzyme responsible for de novo synthesis of dTMP is thymidylate kinase (TS), while the one for conversion of thymidine into dTMP is thymidine kinase (TK).
Folate fortification of foods became mandatory in the US in 1998 to benefit pregnant women and prevent neural tube defects in newborns. However, folate supplementation might be detrimental for men at risk for prostate cancer as suggested by the results of a recent clinical trial where supplementation resulted in a significant increase in the incidence of prostate cancer (5, 6). Indeed, dietary supplementation of folate has been referred to as a “double edged sword” (7) since epidemiologic studies over the past decade suggest an inverse association of dietary folate intake with the risk of several malignancies, including colon and liver cancer (8, 9), yet supplementation could also sustain and accelerate the growth of pre-existing cancer cells (7, 8).
Given that prostate cancer is the most commonly diagnosed and second most lethal cancer in men in the US (10), and in light of our findings that prostate metabolism increases demand on folate intake (4, 11), there is a pressing need to have a better understanding of how dietary folate influences prostate cancer in vivo. We therefore studied the effects of dietary folate manipulation in the Transgenic Adenoma of Mouse Prostate (TRAMP) model, where tumorigenesis is driven by androgen dependent expression of the SV40 large and small T antigen specifically in the prostate at puberty (12). The results indicated that dietary folate restriction, at a level sufficient to sustain normal mouse growth, dramatically and significantly blocked CaP progression in TRAMP, where tumorigenesis is considered to be very aggressive. Conversely, dietary folate supplementation had no significant effect on CaP growth or disease progression, though there is some suggestion of moderation of the phenotype. Further investigation into mechanisms associated with growth arrest and recovery consequent to folate deficiency suggested that upregulation of the enzymes involved in dTTP biosynthesis (thymidylate synthase, TS) and salvage (thymidylate kinase, TK) may be important.
Our study suggests that antifolate treatment and/or specific inhibitors aimed at counteracting upregulation of key biosynthetic/salvage enzymes could be a new promising therapeutic approach for prostate cancer.
Materials and methods
Mice and dietary intervention
All the mouse work was carried out at the Department of Laboratory Animal Research at Roswell Park Cancer Institute. Male TRAMP (Transgenic Adenoma of Mouse Prostate) mice (12), heterozygous for the Pb-Tag transgene, ([C57BL/6J X FVB]F1 background), were bred in the Roswell Park Cancer Institute animal housing facility in accordance with an Institutional Animal Care and Use Committee–approved protocol. Mice were weaned at 3 weeks of age. At the time of weaning the mice were randomly assigned to three cohorts characterized by different folate concentrations in their diet until they reached 22 weeks of age, at which time the mice were sacrificed. The same was applied to three cohorts of mice carried out only till 12 weeks of age, but with larger cohort size due to the small size of the prostate at this age (~25 mg for lateral, dorsal and ventral lobe combined) to ensure enough prostatic tissue to carry out the presented analyses. Each cohort at both time points had at least 25 mice. The time points of 12 and 22 weeks were selected in order to assess a possible effect of the diets on both early (12 weeks) and late (22 weeks) prostate tumorigenesis. A priori power analysis determined that with a cohort size of 20 we would have 80% power to detect an effect size of 0.96 at a significance level of 0.05 using the Wilcoxon Rank Sum test to determine if the diets affect various aspects of disease progression. Starting with a cohort size of 25, we achieved a minimum of 20 samples per diet for most measurements.
The three amino acid defined diets were A) a folate control (FC) diet containing the recommended amount of folic acid (FA) for rodents (2 mg/Kg diet)(13, 14), B) a folate deficient (FD) diet (0.3 mg/Kg diet)(14, 15) and C) a folate supplemented (FS) diet (20 mg/Kg diet), all supplemented with 1% succinylsulfanthiozole (SST) to inhibit FA synthesis by intestinal flora, and all containing 3.3 mg/kg of methionine, 2.5 g/kg choline with energy provided as 15.5% L-amino acids, 66% carbohydrates, and 18.5% fat (14, 15) All three diets are identical with the exception of the amount of folic acid. The diets were purchased from Harlan Teklad, (Madison WI), stored at 4°C and fed without restriction with replacement every second day. The diets were TD.01369 (control), TD.01546 (folate deficient), and TD.08149 (folate supplemented). Urogenital tract weight and body weight were determined at sacrificing. Tissue samples were obtained as previously described (16).
Tissue and serum folate measurements
Serum and tissue folate was quantified using the L. Casei microbiological assay like previously described (4, 17). Tissue folates were digested with recombinant rat conjugase (a kind gift of Karen Chave, Research scientist at Wadsworth Center, NYSDOH) prior analysis with L. Casei. Complete digestion of poly-gamma-glutamylfolates into the monoglutamate form (folic acid) by the conjugase was verified comparing standard curves generated with folic acid and pteroylheptaglutamate (PteGlu7, a kind gift of Dr. John McGuire, Roswell Park Cancer Institute, Buffalo, NY) before and after digestion.
Histopathology on TRAMP tissues was carried out as previously described (18). The dorsal, lateral, and ventral lobes of TRAMP prostates were scored for tumor grade by hematoxylin & eosin (H&E) in mice on all three diets at both 12 and 22 weeks of age. The previously established scoring system in which the prostate lobes are graded 1–6 was used as follows: 1) Normal mouse prostate; 2) Low-grade prostatic intraepithelial neoplasia (PIN); 3) High-grade PIN; 4) Well-differentiated adenocarcinoma; 5) Moderately differentiated adenocarcinoma; 6) Poorly differentiated adenocarcinoma with sheets of anaplastic cells(18). Disease index for each mouse at 22 weeks was calculated by averaging the predominant H&E grade for each lobe plus the average worst grade for each lobe multiplied by the UG weight (19). 25 slides were analyzed per diet per time point for immunohistochemical (IHC) staining of KI67 and caspase 3. Analyses were carried out on the entire section with the percent positively staining cells counted manually and each section categorized as having <5%, 5–25%, 26–50%, 51–75%, or >75% positive cells, as shown in figure 3B. Analyses were performed blind to both diet and age. Statistical analysis was carried out using a two-tailed Fischer’s exact test for the comparisons described.
Figure 3. Representative images of H&E staining on 22 weeks old TRAMP mice fed the folate defined diets as indicated.
Dorsal (DP), lateral (LP), and ventral (VP) prostate lobes are shown from the same mouse per each diet.
HPLC analyses
HPLC analyses for polyamine, nucleotide and AdoMet pools measurements were carried out as previously described (11, 20–22). All analyses were carried out on a reverse-phase Econosil (C18) column (5 µm particle size, 4.6×250 mm) (Fisher Scientific) with a C18 guard column assembled on the Waters 2796 Bioseparation module of the Biopolymer Facility, at Roswell Park Cancer Institute (Buffalo, NY).
Quantitative RT-PCR
1 µg RNA was retro-transcribed with the single strand cDNA synthesis kit (Fermentas) in a 20 µl reaction using random hexamers. 1 µl of the so-obtained cDNA was used for real-time RT PCR analyses, in triplicate, with the iTaq SYBR Green supermix with ROX (Bio-Rad) on an ABI 7900-HT (Applied Biosystems). Primers for TS, TK and 18S are shown in Supplemental Table 1
Random Oligonucleotide Primed Synthesis (ROPS)
ROPS was carried out as previously described (11, 23). Briefly, DNA was denatured and rapidly cooled down causing single DNA strand breaks to provide random priming for successive DNA synthesis with labeled nucleotides. Radioactivity incorporation provides a reproducible, quantitative assessment of single strand DNA breaks. Pre-treating DNA with uracil glycosidase and Exo III allows quantifying uracil misincorporation. At least five independent replicates per sample were analyzed. Uracil misincorporation was normalized, as recommended, by subtracting the background due to pre-existing single strand breaks and apyrimidinic sites. The so-obtained data expressed as deoxcytidine (dCTP) incorporated per microgram of DNA (dCTP/µg).
MAQMA by Sequenom
Briefly, the DNA was bisulphite treated (EZ DNA methylation kit, Zymo Research, CA, USA) and polymerase chain reaction amplified (HotStarTaq DNA Polymerase, Qiagen, CA, USA) with bisulphite sequencing primers flanking the CpGs of interest at loci identified by RLGS. MAQMA was performed using the MassARRAY Compact system developed by the Sequenome Company (CA, USA) as previously described by Ehrich et al. (24). This system utilizes MALDI-TOF mass spectrometry for the detection and quantitative analysis of DNA methylation. This approach has been shown to be a highly accurate and reproducible way to quantitate DNA methylation (25). Primers for the MAQMA assays are shown in Supplemental Table 1.
Results
Dietary folate status affects serum and tissue folates and disease progression
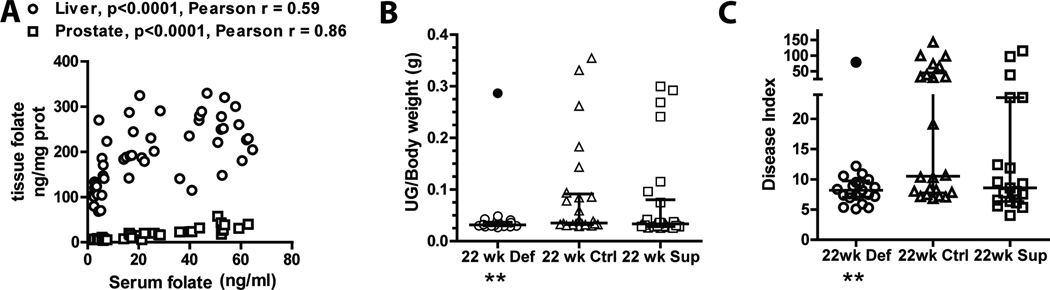
TRAMP mice were placed on the experimental diets immediately after weaning (3 weeks old), and kept on the diets until either 12 or 22 weeks. The diets had no significant effect on body weight (Fig. S1B), yet significantly affected serum and prostate folate levels at both 12 and 22 weeks and liver folates at 22 weeks (Fig. S1). Tissue folate levels significantly correlated with serum levels as shown in Fig. 2A.
Figure 2. Dietary folate depletion prevents CaP development in the TRAMP model.
(A) Pearson correlation analysis between serum and tissue folate levels as quantified by Lactobacillus Casei assay at 22 weeks (mice on all three diets are shown). A similar correlation was observed at 12 weeks (not shown). Liver (purple circles) and prostate (orange squares) folate levels from mice on the three folate defined diets are represented on the Y axis, while serum level of folate in the same animals is represented on the X axis. (B) Prostate tumor burden analysis. The ratio between the weight of the urogenital tract (UG) and the whole body of each mouse is represented on the Y axis according to the three folate defined diets. (C) Histopathological analysis of prostate tumors. Disease index, calculated as ( (average predominant H&E grade for each lobe+ average worst grade for each lobe) * UG weight), is represented on the Y axis for the cohorts of TRAMP mice.
To assess how the dietary interventions affected the course of disease in this model, we measured the urogenital tract weight normalized to body weight (UG/Body weight) at 12 (n=28/diet; not shown) and 22 weeks (Fig. 2B). At 12 weeks, the distribution of UG/body weights was equal across the three diets (not shown). At 22 weeks, however, the data are striking. The distribution of UG/body weights in the control group exhibited significant heterogeneity. This distribution into two distinct populations (mice that developed large tumors versus mice that did not) is typical of this model (18). There was no difference in distribution for mice fed the folate supplemented diet. Mice fed the deficient diet, however, showed a remarkable reduction (p<0.01) and uniformity in UG/body weights, with the exception of a single animal in which the tumor escaped the growth suppressive effects of low dietary folate (Fig. 2B). 10/24 animals had a normalized UG weight greater than 50mg when fed the control diet compared to only 1/26 in mice fed the folate deficient diet. These data suggest that the folate deficient diet caused a dramatic block in tumor growth, while folate supplementation had little or no effect on overall tumor growth.
Folate deficiency improves prostate cancer pathology in TRAMP
Histological examination of TRAMP prostate revealed additional effects of the dietary manipulation. The dorsal, lateral, and ventral lobes of TRAMP prostates were scored for tumor grade by hematoxylin & eosin (H&E) in mice on all three diets at both 12 and 22 weeks of age as described in the methods section. Representative H&E sections from TRAMP prostate lobes at 22 weeks are shown in Figure 3. Figure 4A shows the number of cases scored as each grade for ventral prostate at 22 weeks. TRAMP prostates at 12 weeks were nearly all characterized by early lesions of intraepithelial neoplasia, with no significant difference among the diets (Fig. S2). Strikingly, at 22 weeks on the folate deficient diet, there was little or no progression from what was observed at 12 weeks, with still the majority of prostates scoring as grade 3 and no grade 6 tumors. In the control and supplemented groups, however, there were 10 and 7, respectively, grade 6 tumors (Fig. 4A). Thus, dietary folate deficiency resulted in a significant reduction in mice that progressed beyond grade 3, high grade PIN (p=0.02).
Figure 4. Dietary folate depletion arrests CaP progression in the TRAMP model.
Tabulation of immunohistochemical analysis of 22 weeks old TRAMP mouse ventral prostate (A) Hematoxylin and Eosin to assess tumor grading (H&E). Statistical analyses tested the difference in cases with a grade >3. (B) KI-67, to assess cellular proliferation. Statistical analyses tested the difference in cases with >25% positive cells. (C) Cleaved caspase 3, to assess apoptosis. Statistical analyses tested the difference in cases with >25% positive cells. (D) E-cadherin to assess differentiation. Statistical analyses tested the difference in cases with normal versus abnormal (weak or absent) staining. (E) Transgene staining (SV-40 large T antigen) of the lymph node to assess presence or absence of lymph node metastasis. Statistical analyses tested the difference in cases that were positive or negative for lymph node metastases. All statistical comparisons were made between the control diet and each of the experimental diets individually using a two-tailed Fisher’s Exact test.
Neither diet affected transgene or AR expression (not shown) with all mice showing a very high percentage of strongly staining cells specifically in the prostate. No evidence of distant micro-metastatic spread to the liver or kidney was found in any animal. However, folate deficiency significantly reduced spread to the peri-aortic lymph node (p=0.05). Lymph node metastases were assessed by both H&E staining and IHC using antibody to the transgene as exemplified in figure S3. Lymph node metastases were found in 10/20 mice on the control diet compared to 4/21 in mice on the deficient diet and 8/18 mice on the supplemented diet (Fig. 4E). To quantify overall UG disease for each animal a disease index was derived (19). Folate deficiency significantly reduced the disease index with only 1/21 animals having a disease index greater than 25 compared to 9/22 animals on the control diet (Fig. 2C; p=0.01). Folate supplementation had a slight, yet non-significant (p=0.1) moderating effect with only 3/19 animals having a disease index greater than 25 compared to 9/22 in the control diet. There were no differences in disease index among the three diets at 12 weeks of age (not shown).
Proliferative and apoptotic indices as measured by KI67 and cleaved caspase 3 staining, respectively, showed that prostates of mice fed the deficient diet were characterized by a significantly lower number of proliferating cells (Fig. S4 and 4B; p=0.02) and apoptotic cells (Fig S4 and Fig. 4C; p=0.05), while folate supplementation had no effect. Cell surface E-cadherin staining, a marker of differentiation that is often lost during the process of malignant progression (26), was significantly retained in mice on the deficient diet compared to mice fed the control or supplemented diets (Fig. 4D; p=0.02). We conclude that while folate supplementation had no significant effect on prostate tumorigenesis in TRAMP, the folate deficient diet effectively prevented CaP growth by blocking proliferation, lymph node metastasis, and acquisition of a poorly differentiated phenotype, but not by increasing apoptosis.
Genomic effects of dietary folate manipulation in TRAMP
Folate depletion hinders cell proliferation by decreasing the availability of AdoMet and dTTP, yet might induce genetic and epigenetic damage and worsen the phenotype of cells that are able to proliferate. Specifically, folate deficiency limits the ability of the cells to convert dUMP to dTMP (Fig. 1), and ultimately results in an increased dUTP:dTTP ratio (U:T), which in turn may lead to futile cycles of uracil misincorporation into the DNA followed by its excision and consequent single strand DNA breaks (11, 27–30). Similarly, decreased AdoMet pools may affect DNA and histone methylation.
Neither AdoMet nor s-adenosylhomocysteine (AdoHcy) pools were affected by the diets in the diseased prostate (not shown). However CpG island hypermethylation was dramatically prevented by the folate depleted diet. CpG island methylation status was determined by MassArray Quantitative Methylation Analysis (MAQMA) (Fig. 5A) at four CpG islands previously found to be commonly methylated in TRAMP tumors (31). The data in Fig 5A demonstrate that these four CpG islands were not hypermethylated in mice on the folate deficient diet, but were hypermethylated on the control diet. For all four CpG islands there was one clear exception in the folate deficient group; the one animal that developed a large tumor. As was found concerning disease index (Fig. 2C), folate supplementation moderated the phenotype. For all four CpG islands there were fewer tumors with high level methylation in the supplemented group than in the control group.
Figure 5. Genetic and epigenetic damage assessment in TRAMP prostate.
A) Mass array quantitative methylation analysis of four loci frequently methylated during tumor progression in the TRAMP model. The average level of methylation detected by MAQMA across the sequenced fragment of the CpG islands for each samples is shown on the Y axis. This value comes from taking the average of MAQMA values for each CpG dinucleotide sequenced for each sample. Each symbol represents a single sample. B) HPLC analysis of dTTP, dUTP, and the dUTP: dTTP ratio in 22 weeks old TRAMP mice fed the folate defined diets. C) Random oligo-primed synthesis (ROPS) analysis in 22 weeks old TRAMP prostate on the folate restricted diets quantifies DNA single strand breaks before and after the removal of misincorporated uracil. Single strand DNA breaks are directly proportional to the amount of radioactive dCTP incorporated (Y axis). Statistical comparisons were made between the control diet and each of the experimental diets individually using a two-tailed Mann Whitney test. Statistical significance is indicated as *=P<0.05, **=P<0.01, ***=P<0.001.
Measurement of nucleotide pools (Fig. 5B) and genetic damage (Fig. 5C) revealed a significant increase in dUTP, but no change in Uracil mis-incorporation into the DNA or single strand breaks in the prostates of mice on the folate deficient diet at 22 weeks. We found no significant change in uracil misincorporation and nucleotide pools at 12 weeks (data not shown). Interestingly, the prostate of the one TRAMP mouse that developed a large tumor on the deficient diet (Fig. 2C) had the highest levels of both Uracil mis-incorporation and single strand breaks as compared to any other mouse on this diet (Fig. 5C, solid symbol). In contrast, significant changes in nucleotide pools were detected in the liver (Fig. S5), confirming previous reports and suggesting that prostate’s response to dietary manipulation of folate might indeed be different.
We assessed the mRNA levels of the enzymes involved in dTTP biosynthesis (TS) and salvage (TK) (Fig.1) in the prostates of 22 week old TRAMP mice. Both TS and TK levels significantly correlated with UG/body weight (Fig. 6A). TS expression was not significantly affected by either diet, but TK expression was significantly reduced in mice on the folate deficient and the folate supplemented diets (Fig. 6B). Interestingly, the only prostate overexpressing both enzymes in the folate deficient group was from the one mouse that overcame the growth suppressive effects of low dietary folate and developed a large tumor (Fig. 6B; solid symbol).
Figure 6. Expression of biosynthetic and salvage pathway enzymes for dTTP under the influence of dietary folate manipulation.
(A) Correlation of TS (left) and TK (right) expression with the UG/body weight at 22 weeks, regardless of the diet. (B) Real-time RT PCR analysis of TS ( top) and TK (bottom) expression normalized to 18s in 22 week old TRAMP prostate categorized by the folate defined diets. The full dot represents data from the one animal that developed a large tumor on the folate deficient diet.
Discussion
This study addresses how dietary folate manipulation impacts prostate cancer progression in an initiated mouse model. Our previous findings linking polyamine production to increased demand on folate intake to sustain prostate cancer cell proliferation (4) and maintain the genome (11) provided a compelling rationale to expect that CaP would be extremely sensitive to availability of folate. Indeed, we found that mild dietary folate deficiency strongly blocked disease progression in TRAMP despite continued expression of the SV40 large T-antigen and androgen receptor. Lack of polyamine production did not explain the lack of tumor growth on the deficient diet as polyamine pools were not reduced by the diet (Fig. S6). Indeed, polyamine levels are strictly maintained intracellularly, as demonstrated by the large body of publications that in the past 20 years reported that targeting of polyamine biosynthesis to arrest cancer growth is largely ineffective due to complex compensatory mechanisms (2, 32). High levels of polyamine synthesis required by the prostate are indeed maintained under conditions of low folate, at the expense of other critical pathways. Specifically, folate depletion in our model led to growth arrest of prostate cells, even under the strong proliferation drive of the antigen (SV40-Tag).
Folate supplementation did not accelerate tumor growth or disease progression, despite a demonstrable increase in prostate tissue folate. However, the supplemented diet did significantly reduce the numbers of mice with aberrant methylation of three of the four CpG islands tested (Fig. 5), suggesting that there was some effect on the disease process. Additionally, folate supplementation significantly reduced the expression of TK, the key regulator of the salvage pathway for maintaining dTTP pools (Fig. 6). Furthermore, supplementation appears to have reduced the overall disease index for the TRAMP mice (Fig. 2C). The lack of statistical significance (p=0.1) is likely driven by the large amount of heterogeneity of the TRAMP model on the control diet, as the observed variance was larger than expected (Fig. 2B, C). These data suggest that folate supplementation may have some beneficial effects on CaP progression in this very aggressive model. However, the aggressiveness and heterogeneity of the TRAMP model may have overwhelmed the potential for folate supplementation to improve or worsen tumor progression and/or limited our ability to observe such changes without doubling the sample size. Further studies with larger samples sizes using different models of CaP will be necessary to elucidate this point.
The effect of dietary deficiency of folate on CaP progression in the TRAMP model (blocking progression) is particularly striking because this same diet initiated intestinal tumors when fed to wild type mice for 12 months (14) and increased adenoma number in APCmin/+ mice (15). A key relevant difference between prostate tissue and colon tissue might be polyamine biosynthesis. Prostate cells are characterized by an unusually high rate of polyamine biosynthesis (33–36), which is maintained even in the face of low levels of intracellular folates (Fig S6). This renders prostate exquisitely sensitive to folate depletion, because a significant amount of one-carbon units has to be devoted to maintain the high polyamine biosynthesis characteristic of this organ (4, 11). Their acetylation and consequent secretion due to the activity of Spermidine/spermine N1-acetyltransferase (SSAT) draws on AdoMet pools in order to maintain intracellular polyamine levels (37). This leads to increased demand for folate input to maintain AdoMet pools, polyamines and proliferation (4). When transgenic mice over expressing SSAT were crossed with Apcmin/+ mice, the result was a 6-fold increase in colon adenoma number (38). However, when crossed with the TRAMP mouse, the result was the opposite - greatly reduced prostate tumor growth (19). This was likely due to Acetyl Co-A pool depletion, as suggested by the authors, but perhaps also due to metabolic stress on the methionine cycle and one-carbon metabolism because of enhanced flux to maintain intracellular polyamine pools. Although there are caveats to these comparisons due to the different genetic backgrounds and transgenes, the findings nevertheless suggest that differences in metabolic processes between colon and prostate tissue might significantly impact the biological consequences of their manipulation.
Antifolate cancer therapy has been widely used in the past 30 years to treat various malignancies, but the effects of antifolates on primary CaP is unknown. Positive results treating castration recurrent CaP with methotrexate (39, 40), were not confirmed by later studies (41–43). We suggest that the high level of polyamine biosynthesis due to the secretory characteristics of prostate, uniquely stresses one-carbon metabolism and the methionine cycle, and is therefore central to the rationale behind applying antifolate therapy to CaP. The enzymes responsible for synthesizing and exporting polyamines, are regulated by androgens, which keep their expression levels high in prostate tissue (35, 44–46). Although it is known that intracellular polyamine concentration increases upon tumor progression, the effects of androgen deprivation therapy on these enzymes and therefore overall prostate polyamine synthesis are unknown and might in fact strongly weaken the potential of antifolates in the castration recurrent setting. Therefore, its failure in this advanced setting should not predict failure at earlier time points where polyamine biosynthesis is known to be high and driven by androgens.
Our findings are significant from both a dietary recommendation perspective as well as a therapeutic perspective. Folate supplementation, as well as fortification of certain foods, has the potential to protect from cancer development, but may also accelerate the growth of neoplasms that may already be present, though subclinical (7, 47). However, the rate of indolent CaP in men over 50 is high (48) and it is possible that folate supplementation in these men might in fact accelerate CaP growth. Conversely, our findings suggest that CaP, due to its high dependence on folate, might be sensitive to antifolate based approaches; perhaps not in the context of castration recurrence and chemotherapy resistance, but rather in the context of earlier treatment.
Supplementary Material
We measured the following parameters in TRAMP mice fed the folate deficient (Def), control (Ctrl), and supplemented (Sup) diet at 12 (left) and 22 (right) weeks: (A) serum folate, (B) body weight, (C) prostate levels of folate, (D) liver levels of folate. Folate levels were measured by a microbiological assai (L. Casei). Statistical significance was measured with the Mann Whitney U test. * = p<0.05, ** = p<0.01, *** = p<0.001.
Representative images of H&E staining on 12 weeks old TRAMP mice fed the folate defined diets as indicated.
Example of a normal lymph node (top) and a metastatic lymph node (bottom) stained with an antibody specific to the transgene (SV40-Tag) that is expressed only in the nucleus of prostate cells. Notice that the brown stains visible in the normal lynph node are not nuclear and are likely due to aspecific background.
Representative images of KI-67 and caspase 3 IHC on 22 weeks old ventral prostate from TRAMP mice fed the folate defined diets as indicated.
HPLC analysis of nucleotide pools in the liver of 22 weeks old TRAMP mice fed the folate defined diet as indicated.
HPLC analysis of total polyamines (PAs, spermine + spermidine) in the prostate of TRAMP mice fed the folate defined diets as specified in the Figures, at 12 (top) and 22 weeks (bottom).
Acknowledgments
We would like to acknowledge Dr. Karen Chave from the Wadsworth Center, NYSDOH, who kindly donated to us the recombinant rat conjugase and Dr. John McGuire, from the Roswell Park Cancer Institute, who kindly donate to us the poly-{gamma}-glutamate7.
Grant Support
This work was supported by an American Institute for Cancer Research Fellowship (GB), an American-Italian Cancer Foundation Fellowship (GB), a Department Of Defense post-doctoral fellowship (GB) and NCI R21 CA121216 (GB, BAF, EK, BG, JM, VKD, and DJS, ) This research was supported, in part, by the NCI Cancer Center Support Grant to the Roswell Park Cancer Institute [CA016056].
References
- 1.Gangjee A, Jain HD, Kurup S. Recent advances in classical and non-classical antifolates as antitumor and antiopportunistic infection agents: part I. Anticancer Agents Med Chem. 2007 Sep;7(5):524–542. doi: 10.2174/187152007781668724. [DOI] [PubMed] [Google Scholar]
- 2.Gerner EW, Meyskens FL., Jr Polyamines and cancer: old molecules, new understanding. Nat Rev Cancer. 2004 Oct;4(10):781–792. doi: 10.1038/nrc1454. [DOI] [PubMed] [Google Scholar]
- 3.Loenen WA. S-adenosylmethionine: jack of all trades and master of everything? Biochem Soc Trans. 2006 Apr;34(Pt 2):330–333. doi: 10.1042/BST20060330. [DOI] [PubMed] [Google Scholar]
- 4.Bistulfi G, Diegelman P, Foster BA, Kramer DL, Porter CW, Smiraglia DJ. Polyamine biosynthesis impacts cellular folate requirements necessary to maintain S-adenosylmethionine and nucleotide pools. FASEB J. 2009 Sep;23(9):2888–2897. doi: 10.1096/fj.09-130708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cole BF, Baron JA, Sandler RS, Haile RW, Ahnen DJ, Bresalier RS, McKeown-Eyssen G, Summers RW, Rothstein RI, Burke CA, Snover DC, Church TR, Allen JI, Robertson DJ, Beck GJ, Bond JH, Byers T, Mandel JS, Mott LA, Pearson LH, Barry EL, Rees JR, Marcon N, Saibil F, Ueland PM, Greenberg ER. Folic acid for the prevention of colorectal adenomas: a randomized clinical trial. Jama. 2007 Jun 6;297(21):2351–2359. doi: 10.1001/jama.297.21.2351. [DOI] [PubMed] [Google Scholar]
- 6.Figueiredo JC, Grau MV, Haile RW, Sandler RS, Summers RW, Bresalier RS, Burke CA, McKeown-Eyssen GE, Baron JA. Folic acid and risk of prostate cancer: results from a randomized clinical trial. J Natl Cancer Inst. 2009 Mar 18;101(6):432–435. doi: 10.1093/jnci/djp019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim YI. Folate: a magic bullet or a double edged sword for colorectal cancer prevention? Gut. 2006 Oct;55(10):1387–1389. doi: 10.1136/gut.2006.095463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.James SJ, Pogribny IP, Pogribna M, Miller BJ, Jernigan S, Melnyk S. Mechanisms of DNA damage, DNA hypomethylation, and tumor progression in the folate/methyl-deficient rat model of hepatocarcinogenesis. J Nutr. 2003 Nov;133(11 Suppl 1):3740S–3747S. doi: 10.1093/jn/133.11.3740S. [DOI] [PubMed] [Google Scholar]
- 9.Kim YI. Nutritional epigenetics: impact of folate deficiency on DNA methylation and colon cancer susceptibility. J Nutr. 2005 Nov;135(11):2703–2709. doi: 10.1093/jn/135.11.2703. [DOI] [PubMed] [Google Scholar]
- 10.Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009 Jul–Aug;59(4):225–249. doi: 10.3322/caac.20006. [DOI] [PubMed] [Google Scholar]
- 11.Bistulfi G, Vandette E, Matsui SI, Smiraglia DJ. Mild folate deficiency induces genetic and epigenetic instability and phenotype changes in prostate cancer cells. BMC Biol. 2010 Jan 21;8(1):6. doi: 10.1186/1741-7007-8-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Greenberg NM, DeMayo F, Finegold MJ, Medina D, Tilley WD, Aspinall JO, Cunha GR, Donjacour AA, Matusik RJ, Rosen JM. Prostate cancer in a transgenic mouse. Proc Natl Acad Sci U S A. 1995 Apr 11;92(8):3439–3443. doi: 10.1073/pnas.92.8.3439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Reeves PG, Nielsen FH, Fahey GC., Jr AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr. 1993 Nov;123(11):1939–1951. doi: 10.1093/jn/123.11.1939. [DOI] [PubMed] [Google Scholar]
- 14.Knock E, Deng L, Wu Q, Leclerc D, Wang XL, Rozen R. Low dietary folate initiates intestinal tumors in mice, with altered expression of G2-M checkpoint regulators polo-like kinase 1 and cell division cycle 25c. Cancer Res. 2006 Nov 1;66(21):10349–10356. doi: 10.1158/0008-5472.CAN-06-2477. [DOI] [PubMed] [Google Scholar]
- 15.Lawrance AK, Deng L, Brody LC, Finnell RH, Shane B, Rozen R. Genetic and nutritional deficiencies in folate metabolism influence tumorigenicity in Apc(min/+) mice. J Nutr Biochem. 2006 Sep 8; doi: 10.1016/j.jnutbio.2006.06.001. [DOI] [PubMed] [Google Scholar]
- 16.Morey Kinney SR, Smiraglia DJ, James SR, Moser MT, Foster BA, Karpf AR. Stage-specific alterations of DNA methyltransferase expression, DNA hypermethylation, and DNA hypomethylation during prostate cancer progression in the transgenic adenocarcinoma of mouse prostate model. Mol Cancer Res. 2008 Aug;6(8):1365–1374. doi: 10.1158/1541-7786.MCR-08-0040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Horne DW, Patterson D. Lactobacillus casei microbiological assay of folic acid derivatives in 96-well microtiter plates. Clin Chem. 1988 Nov;34(11):2357–2359. [PubMed] [Google Scholar]
- 18.Kaplan-Lefko PJ, Chen TM, Ittmann MM, Barrios RJ, Ayala GE, Huss WJ, Maddison LA, Foster BA, Greenberg NM. Pathobiology of autochthonous prostate cancer in a pre-clinical transgenic mouse model. Prostate. 2003 May 15;55(3):219–237. doi: 10.1002/pros.10215. [DOI] [PubMed] [Google Scholar]
- 19.Kee K, Foster BA, Merali S, Kramer DL, Hensen ML, Diegelman P, Kisiel N, Vujcic S, Mazurchuk RV, Porter CW. Activated polyamine catabolism depletes acetyl-CoA pools and suppresses prostate tumor growth in TRAMP mice. J Biol Chem. 2004 Sep 17;279(38):40076–40083. doi: 10.1074/jbc.M406002200. [DOI] [PubMed] [Google Scholar]
- 20.Cross DR, Miller BJ, James SJ. A simplified HPLC method for simultaneously quantifying ribonucleotides and deoxyribonucleotides in cell extracts or frozen tissues. Cell Prolif. 1993 Jul;26(4):327–336. doi: 10.1111/j.1365-2184.1993.tb00328.x. [DOI] [PubMed] [Google Scholar]
- 21.Kramer D, Mett H, Evans A, Regenass U, Diegelman P, Porter CW. Stable amplification of the S-adenosylmethionine decarboxylase gene in Chinese hamster ovary cells. J Biol Chem. 1995 Feb 3;270(5):2124–2132. doi: 10.1074/jbc.270.5.2124. [DOI] [PubMed] [Google Scholar]
- 22.Kramer D, Stanek J, Diegelman P, Regenass U, Schneider P, Porter CW. Use of 4-fluoro-L-ornithine to monitor metabolic flux through the polyamine biosynthetic pathway. Biochem Pharmacol. 1995 Oct 26;50(9):1433–1443. doi: 10.1016/0006-2952(95)02037-3. [DOI] [PubMed] [Google Scholar]
- 23.Basnakian AG, James SJ. Quantification of 3'OH DNA breaks by random oligonucleotide-primed synthesis (ROPS) assay. DNA Cell Biol. 1996 Mar;15(3):255–262. doi: 10.1089/dna.1996.15.255. [DOI] [PubMed] [Google Scholar]
- 24.Ehrich M, Nelson MR, Stanssens P, Zabeau M, Liloglou T, Xinarianos G, Cantor CR, Field JK, van den Boom D. Quantitative high-throughput analysis of DNA methylation patterns by base-specific cleavage and mass spectrometry. Proc Natl Acad Sci U S A. 2005 Nov 1;102(44):15785–15790. doi: 10.1073/pnas.0507816102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Coolen MW, Statham AL, Gardiner-Garden M, Clark SJ. Genomic profiling of CpG methylation and allelic specificity using quantitative high-throughput mass spectrometry: critical evaluation and improvements. Nucleic Acids Res. 2007;35(18):e119. doi: 10.1093/nar/gkm662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schmalhofer O, Brabletz S, Brabletz T. E-cadherin, beta-catenin, and ZEB1 in malignant progression of cancer. Cancer Metastasis Rev. 2009 Jun;28(1–2):151–166. doi: 10.1007/s10555-008-9179-y. [DOI] [PubMed] [Google Scholar]
- 27.Blount BC, Mack MM, Wehr CM, MacGregor JT, Hiatt RA, Wang G, Wickramasinghe SN, Everson RB, Ames BN. Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: implications for cancer and neuronal damage. Proc Natl Acad Sci U S A. 1997 Apr 1;94(7):3290–3295. doi: 10.1073/pnas.94.7.3290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.James SJ, Miller BJ, Basnakian AG, Pogribny IP, Pogribna M, Muskhelishvili L. Apoptosis and proliferation under conditions of deoxynucleotide pool imbalance in liver of folate/methyl deficient rats. Carcinogenesis. 1997 Feb;18(2):287–293. doi: 10.1093/carcin/18.2.287. [DOI] [PubMed] [Google Scholar]
- 29.Pogribny IP, Muskhelishvili L, Miller BJ, James SJ. Presence and consequence of uracil in preneoplastic DNA from folate/methyl-deficient rats. Carcinogenesis. 1997 Nov;18(11):2071–2076. doi: 10.1093/carcin/18.11.2071. [DOI] [PubMed] [Google Scholar]
- 30.Duthie SJ, Narayanan S, Blum S, Pirie L, Brand GM. Folate deficiency in vitro induces uracil misincorporation and DNA hypomethylation and inhibits DNA excision repair in immortalized normal human colon epithelial cells. Nutr Cancer. 2000;37(2):245–251. doi: 10.1207/S15327914NC372_18. [DOI] [PubMed] [Google Scholar]
- 31.Camoriano M, Kinney SR, Moser MT, Foster BA, Mohler JL, Trump DL, Karpf AR, Smiraglia DJ. Phenotype-specific CpG island methylation events in a murine model of prostate cancer. Cancer Res. 2008 Jun 1;68(11):4173–4182. doi: 10.1158/0008-5472.CAN-07-6715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pegg AE. Polyamine metabolism and its importance in neoplastic growth and a target for chemotherapy. Cancer Res. 1988 Feb 15;48(4):759–774. [PubMed] [Google Scholar]
- 33.Harrison GA. Spermine in human tissues. Biochem J. 1931;25(6):1885–1892. doi: 10.1042/bj0251885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Harrison GA. The approximate determination of spermine in single human organs. Biochem J. 1933;27(4):1152–1156. doi: 10.1042/bj0271152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pegg AE, Lockwood DH, Williams-Ashman HG. Concentrations of putrescine and polyamines and their enzymic synthesis during androgen-induced prostatic growth. Biochem J. 1970 Mar;117(1):17–31. doi: 10.1042/bj1170017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Williams-Ashman HG, Canellakis ZN. Polyamines in mammalian biology and medicine. Perspect Biol Med. 1979 Spring;22(3):421–453. doi: 10.1353/pbm.1979.0013. [DOI] [PubMed] [Google Scholar]
- 37.Casero RA, Jr, Pegg AE. Spermidine/spermine N1-acetyltransferase--the turning point in polyamine metabolism. Faseb J. 1993 May;7(8):653–661. [PubMed] [Google Scholar]
- 38.Tucker JM, Murphy JT, Kisiel N, Diegelman P, Barbour KW, Davis C, Medda M, Alhonen L, Janne J, Kramer DL, Porter CW, Berger FG. Potent modulation of intestinal tumorigenesis in Apcmin/+ mice by the polyamine catabolic enzyme spermidine/spermine N1-acetyltransferase. Cancer Res. 2005 Jun 15;65(12):5390–5398. doi: 10.1158/0008-5472.CAN-05-0229. [DOI] [PubMed] [Google Scholar]
- 39.Murphy GP, Slack NH. Response criteria for the prostate of the USA National Prostatic Cancer Project. Prostate. 1980;1(3):375–382. doi: 10.1002/pros.2990010313. [DOI] [PubMed] [Google Scholar]
- 40.Loening SA, Beckley S, Brady MF, Chu TM, deKernion JB, Dhabuwala C, Gaeta JF, Gibbons RP, McKiel CF, McLeod DG, Pontes JE, Prout GR, Scardino PT, Schlegel JU, Schmidt JD, Scott WW, Slack NH, Soloway MS, Murphy GP. Comparison of estramustine phosphate, methotrexate and cis-platinum in patients with advanced, hormone refractory prostate cancer. J Urol. 1983 May;129(5):1001–1006. doi: 10.1016/s0022-5347(17)52509-4. [DOI] [PubMed] [Google Scholar]
- 41.Jones WG, Fossa SD, Verbaeys AC, Droz JP, Klijn JG, Boven E, de Pauw M, Sylvester R. Low-dose fortnightly methotrexate in advanced prostate cancer. The EORTC Genito-Urinary Tract Cancer Cooperative Group. Eur J Cancer. 1990;26(5):646. doi: 10.1016/0277-5379(90)90099-f. [DOI] [PubMed] [Google Scholar]
- 42.Saxman S, Ansari R, Drasga R, Miller M, Wheeler B, McClean J, Einhorn L. Phase III trial of cyclophosphamide versus cyclophosphamide, doxorubicin, and methotrexate in hormone-refractory prostatic cancer. A Hoosier Oncology Group study. Cancer. 1992 Nov 15;70(10):2488–2492. doi: 10.1002/1097-0142(19921115)70:10<2488::aid-cncr2820701016>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]
- 43.Hahn NM, Zon RT, Yu M, Ademuyiwa FO, Jones T, Dugan W, Whalen C, Shanmugam R, Skaar T, Sweeney CJ. A phase II study of pemetrexed as second-line chemotherapy for the treatment of metastatic castrate-resistant prostate cancer (CRPC); Hoosier Oncology Group GU03-67. Ann Oncol. 2009 Dec;20(12):1971–1976. doi: 10.1093/annonc/mdp244. [DOI] [PubMed] [Google Scholar]
- 44.Blackshear PJ, Manzella JM, Stumpo DJ, Wen L, Huang JK, Oyen O, Young WS., 3rd High level, cell-specific expression of ornithine decarboxylase transcripts in rat genitourinary tissues. Mol Endocrinol. 1989 Jan;3(1):68–78. doi: 10.1210/mend-3-1-68. [DOI] [PubMed] [Google Scholar]
- 45.Cyriac J, Haleem R, Cai X, Wang Z. Androgen regulation of spermidine synthase expression in the rat prostate. Prostate. 2002 Mar 1;50(4):252–261. doi: 10.1002/pros.10052. [DOI] [PubMed] [Google Scholar]
- 46.Fjosne HE, Ostensen MA, Haarstad H, Sunde A. Androgen regulation of polyamine synthesis in seminal vesicle and in different lobes of the rat prostate. Prostate. 1990;17(1):1–11. doi: 10.1002/pros.2990170102. [DOI] [PubMed] [Google Scholar]
- 47.Ulrich CM, Potter JD. Folate and cancer--timing is everything. Jama. 2007 Jun 6;297(21):2408–2409. doi: 10.1001/jama.297.21.2408. [DOI] [PubMed] [Google Scholar]
- 48.Breslow N, Chan CW, Dhom G, Drury RA, Franks LM, Gellei B, Lee YS, Lundberg S, Sparke B, Sternby NH, Tulinius H. Latent carcinoma of prostate at autopsy in seven areas. The International Agency for Research on Cancer, Lyons, France. Int J Cancer. 1977 Nov 15;20(5):680–688. doi: 10.1002/ijc.2910200506. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
We measured the following parameters in TRAMP mice fed the folate deficient (Def), control (Ctrl), and supplemented (Sup) diet at 12 (left) and 22 (right) weeks: (A) serum folate, (B) body weight, (C) prostate levels of folate, (D) liver levels of folate. Folate levels were measured by a microbiological assai (L. Casei). Statistical significance was measured with the Mann Whitney U test. * = p<0.05, ** = p<0.01, *** = p<0.001.
Representative images of H&E staining on 12 weeks old TRAMP mice fed the folate defined diets as indicated.
Example of a normal lymph node (top) and a metastatic lymph node (bottom) stained with an antibody specific to the transgene (SV40-Tag) that is expressed only in the nucleus of prostate cells. Notice that the brown stains visible in the normal lynph node are not nuclear and are likely due to aspecific background.
Representative images of KI-67 and caspase 3 IHC on 22 weeks old ventral prostate from TRAMP mice fed the folate defined diets as indicated.
HPLC analysis of nucleotide pools in the liver of 22 weeks old TRAMP mice fed the folate defined diet as indicated.
HPLC analysis of total polyamines (PAs, spermine + spermidine) in the prostate of TRAMP mice fed the folate defined diets as specified in the Figures, at 12 (top) and 22 weeks (bottom).