Abstract
Background
Low regeneration ability limits biotechnological breeding approaches. The influence of genotype in the regeneration response is high in both tomato and other important crops. Despite the various studies that have been carried out on regeneration genetics, little is known about the key genes involved in this process. The aim of this study was to localize the genetic factors affecting regeneration in tomato.
Results
We developed two mapping populations (F2 and BC1) derived from a previously selected tomato cultivar (cv. Anl27) with low regeneration ability and a high regeneration accession of the wild species Solanum pennellii (PE-47). The phenotypic assay indicated dominance for bud induction and additive effects for both the percentage of explants with shoots and the number of regenerated shoots per explant. Two linkage maps were developed and six QTLs were identified on five chromosomes (1, 3, 4, 7 and 8) in the BC1 population by means of the Interval Mapping and restricted Multiple QTL Mapping methods. These QTLs came from S. pennellii, with the exception of the minor QTL located on chromosome 8, which was provided by cv. Anl27. The main QTLs correspond to those detected on chromosomes 1 and 7. In the F2 population, a QTL on chromosome 7 was identified on a similar region as that detected in the BC1 population. Marker segregation distortion was observed in this population in those areas where the QTLs of BC1 were detected. Furthermore, we located two tomato candidate genes using a marker linked to the high regeneration gene: Rg-2 (a putative allele of Rg-1) and LESK1, which encodes a serine/threonine kinase and was proposed as a marker for regeneration competence. As a result, we located a putative allele of Rg-2 in the QTL detected on chromosome 3 that we named Rg-3. LESK1, which is also situated on chromosome 3, is outside Rg-3. In a preliminary exploration of the detected QTL peaks, we found several genes that may be related to regeneration.
Conclusions
In this study we have identified new QTLs related to the complex process of regeneration from tissue culture. We have also located two candidate genes, discovering a putative allele of the high regeneration gene Rg-1 in the QTL on chromosome 3. The identified QTLs could represent a significant step toward the understanding of this process and the identification of other related candidate genes. It will also most likely facilitate the development of molecular markers for use in gene isolation.
Background
In vitro regeneration of cultivated tomato (Solanum lycopersicum L.) has been a constant subject of research because of the commercial value of the crop. Consequently, numerous studies on plant regeneration from a wide range of tissues and organs of wild and cultivated tomato germplasm have been published [1]. These studies demonstrate that organogenesis, the common tomato regeneration pathway, is strongly influenced by genotype as well as by several physical and chemical factors. These reports also document the existence of recalcitrance (partial or total inability to respond to in vitro culture), which greatly limits biotechnological breeding. High regeneration is crucial to the success of techniques such as haploid regeneration, genetic transformation, propagation, somatic hybridization, mutation selection and germplasm storage [2,3]. For example, the low efficiency of tomato transformation has been associated with the low regeneration potential of the cultivars used [4,5]. In addition, in some cultivars, buds may be induced but do not develop into shoots [6]. In order to increase regeneration ability in low regenerating tomato cultivars, several introgression programs have been documented [7-10].
The process of in vitro shoot organogenesis usually involves a hormonal response of somatic cells, the dedifferentiation of differentiated cells in order to acquire organogenic competence, cell division of the responding cell(s) and initiation and development of new shoots from the newly dividing cell(s), either directly or indirectly through a callus stage [11,12]. Thus, many genes may be involved at different steps of this complex process. For instance, the cdc2 gene expression, which encodes p34, a key cell cycle regulator, has been proposed as an indicator of the state of competence to divide [13]. Genes that encode or regulate cytokinins and auxin may clearly influence regeneration. Both types of growth regulators act synergistically to promote cell division and antagonistically to promote shoot and root initiation from callus cultures [14]. In Arabidopsis, a Histidine Kinase (AHK) gene that encodes a cytokinin receptor (CRE1/AHK4) has been identified [15,16] and linked, like other AHKs, to cell division and regulation [17]. With regard to the initiation of shoot formation, the most characterized gene reported is ESR1, which confers, when overexpressed, cytokinin-independent shoot formation in Arabidopsis root explants [18]. ESR1 encodes a transcription factor belonging to the ethylene-responsive factor (ERF) family and is classified in subgroup VIII-b. The ESR2 gene that encodes a protein that is very similar to ESR1 appears to have redundant functions that regulate shoot regeneration [19]. The expression patterns of other Arabidopsis ERF VIII-b subgroup genes may also be involved in early events of shoot regeneration [20].
Genetic analysis of regeneration in tomato suggests that dominant alleles determine high regeneration capacity [7,21-24]. However, there is no consensus about the number of genes involved. For instance, Koorneef et al. [25] obtained regeneration segregation ratios in accordance with either a monogenic, digenic or trigenic model depending on the tester tomato line, despite the fact that none of the lines themselves were able to regenerate shoots from root explants. In this study, a dominant allele of S. peruvianum L. (Rg-1), which determines efficient shoot regeneration in tomato root explants, was mapped near the middle of chromosome 3. In addition, a putative allele of Rg-1 from S. chilense (Dunal) Reiche (Rg-2) was reported by Takashina et al. [9] and Satoh et al. [22]. Both alleles may act in combination with other alleles of either tomato or the wild relatives S. peruvianum or S. chilense [22,25]. On the other hand, Torelli et al. [26] identified a cDNA by mRNA-differential display that corresponded to the LESK1 gene and whose expression is specifically and transiently enhanced by the exposure to the hormonal treatment leading to caulogenesis (shoot induction). This gene encodes a putative serine-threonine kinase and has been reported as an in vitro caulogenesis marker in tomato [27,28].
Despite ongoing research into the genetic control of in vitro culture traits in tomato and other crops, there is still not enough information regarding which key genes are responsible for low or high regeneration ability, nor even the number of genes involved. The study and characterization of the reported genes and others that might be identified could greatly improve our understanding of the molecular mechanism underlying the different phases of tomato in vitro organogenesis. In the present study, we developed two mapping populations (F2 and BC1) from S. lycopersicum (as the recurrent parent) and S. pennellii Correll (as the regenerating parent) and conducted a QTL-based analysis. We hereby report the identification of six QTLs on five chromosomes. These QTLs present high significant LOD scores and together represent a high percentage of phenotypic variance. We also report markers associated with QTL peaks. In addition, we located two candidate genes, Rg-2 and LESK1, and performed a preliminary search for genes situated at QTL peaks. Our findings will complement the current knowledge of the genetics of regeneration and facilitate the development of molecular markers for use in tomato breeding and gene isolation.
Results
Development of populations and evaluation of the regeneration ability
Two mapping populations, F2 and BC1, were obtained from a low regenerating cultivar of tomato (cv. Anl27) and the organogenic accession of S. pennellii (PE-47). The BC1 population was obtained using the tomato cultivar as the recurrent parent. In the first assay, the regeneration ability of the parents and the F1 plant used for obtaining the mapping populations was checked by culturing leaf explants on shoot induction medium. Regeneration occurred with little callus development and can be considered as direct. As expected, S. pennellii and F1 explants manifested a higher regeneration potential versus S. lycopersicum explants (P <0.001). The percentage of explants with buds (B) in S. pennellii was 100%, whereas only 10% was obtained in tomato cv. Anl27 (Table 1). Data obtained in F1 for B do not significantly differ from those obtained for S. pennellii. The percentage of explants with shoots (R) and the number of regenerated plants per explant with shoots, considered to be the productivity rate (PR), was also higher in S. pennellii and F1 than in cv. Anl27. However, for these traits (R and PR), the F1 values differ significantly from those of S. pennellii (Table 1).
Table 1.
Phenotyping parental genotypes and mapping population
| First Assay Phenotyping parental genotypes and F1 |
Second Assay Phenotyping mapping populations |
|||||
|---|---|---|---|---|---|---|
| Ba, c | Ra, c | PRb, c | Ba, c | Ra, c | PRb, c | |
| S. pennellii | 100 b | 96 c | 6.36 c | 100.00 c | 95.00 d | 6.74 c |
| S. lycopersicum | 10 a | 6 a | 0.30 a | 7.50 a | 2.50 a | 0.12 a |
| F1 | 90 b | 78 b | 3.17 b | 87.50 c | 70.0 c | 3.08 b |
| F2 | - | - | - | 76.91 bc | 63.92 c | 2.65 b |
| BC1 | - | - | - | 59.48 b | 36.65 b | 1.67 b |
Means of the traits: percentage of explants with buds (B), percentage of explants with shoots (R) and number of shoots per explant with shoots (PR) for the parent genotypes (S. pennellii and S. lycopersicum), F1, F2 and BC1.
a B and R are the percentages of explants able to develop buds and shoots, respectively. b PR is the number of shoots per explant with shoots. c Mean values within a column separated by different letters are significantly different (P < 0.05) according to Duncan's multiple range test.
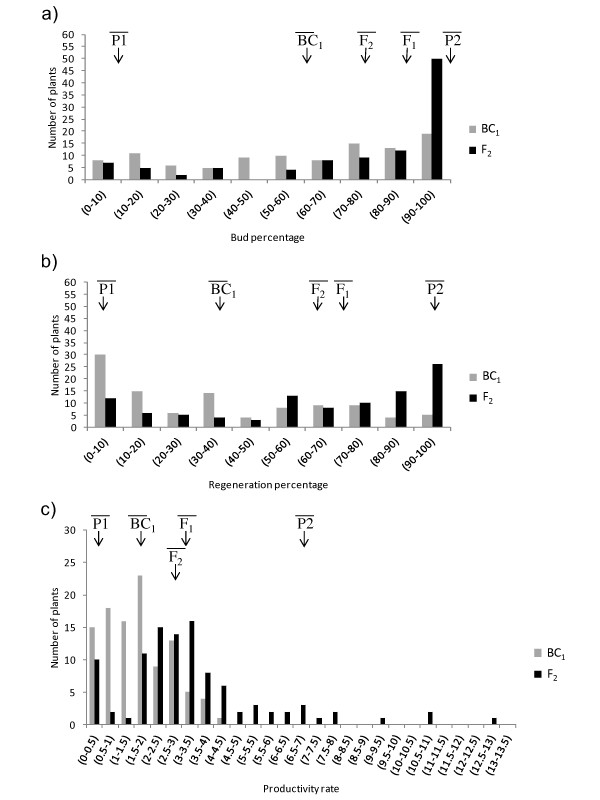
The F2 and BC1 populations were evaluated for regeneration using explants from the parents and F1 plants as controls (Table 1). The phenotypes are shown in Additional File 1. The distribution obtained for each individual trait as well as the means for controls in this assay are presented in Figure 1. Mean values for B, R and PR in the F2 population are between F1 and tomato (P1), but skewed towards F1. For the PR trait, some F2 plants were in a range higher than the S. pennellii parent (P2). This can be considered transgressive segregation. BC1 yielded mean values for B, R and PR that were intermediate between F1 and cv. Anl27 (Figure 1).
Figure 1.
Population distributions for regeneration traits. a) The percentage of explants with buds (B), b) The percentage of explants with plants (R) and c) The percentage of plants per explant with shoots (PR). The F2 population (dark) is derived from selfing an F1, the result of a cross between the tomato cv. Anl27 (P1) and S. pennellii PE-47 (P2). The BC1 population (grey) is the result of crossing the tomato cv. Anl27 and the F1 plants. Maternal (P1), Paternal (P2), F1, F2 and BC1 mean values are indicated by arrows.
B and R show a high correlation (r = 0.88/0.79 p < 0, 001 for F2 and BC1 data, respectively), which suggests common or linked genes controlling these traits. The correlation between PR and both B and R was lower (r = 0.56/0.52 p < 0, 001; 0.66/0.66 p < 0, 001 for PR and B and R for F2 and BC1, respectively) indicating that different genes may influence the PR trait and/or variations between different biological samples are higher in PR.
Linkage maps
Genetic linkage maps were constructed from 106 F2 and 113 BC1 plants genotyped with SSR, COSI, COSII, CAPS and AFLP markers (Figure 2). Of the 149 SSR and 97 other markers (86 COSII, 6 COSI, 5 CAPS) assayed, 78 SSR and 59 (51 COSII, 4 COSI, 4 CAPS) markers exhibited codominant polymorphisms. These markers were obtained from the Sol Genomics Network (SGN) webpage at http://www.sgn.cornell.edu/with the exception of 60 SSRs that were designed following the procedure described in Materials and Methods (see Additional File 2).
Figure 2.
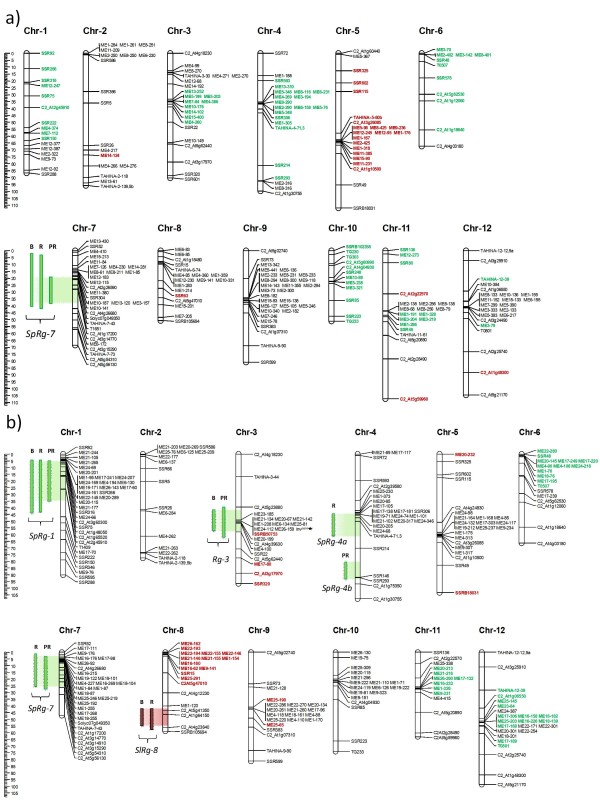
a) Tomato genetic linkage map of F2 population derived from S. lycopersicum (cv. Anl27) × S. pennellii (PE-47) and QTLs detected for regeneration traits by IM. b) Tomato genetic linkage map of BC1 population derived from S. lycopersicum (cv. Anl27) × F1 (cv. Anl27 × PE-47) and QTLs detected for regeneration traits by rMQM. The segregated data were classified into 12 linkage groups, which corresponded to the Tomato-EXPEN 2000 map; italics indicate markers with segregation significantly skewed (P < 0.05) in favour of parent alleles. The colors specify the direction of the segregation distortion (red: markers skewed toward the alleles of cultivated tomato; green: markers skewed toward the alleles of the wild parent). Green bars reflect QTLs from S. pennellii: SpRg-1, Rg-3, SpRg-4a, SpRg-4b and SpRg-7; the red bar reflects the SlRg-8 QTL from S. lycopersicum. Regeneration traits: B (Bud percentage), R (Regeneration percentage) and PR (Productivity rate). The black star labels the acid invertase gene (invpenn) mapped on chromosome 3 included in the Rg-3 QTL range.
For the F2 linkage map (Figure 2a), a total of 246 polymorphic loci were used, including 151 AFLP, 53 SSR, 35 COSII, 3 COSI and 4 CAPS markers. The markers were aligned in 12 linkage groups, with LOD scores ≥ 3.0. The average number of markers per linkage group was 20 and markers were well distributed over all the 12 linkage groups. The F2 map spans 963.85 cM with an average interval of 3.72 cM between adjacent markers. There were five intervals > 25 cM in chromosomes 2, 4, 5 and 11. A total of 268 polymorphic loci were used to assemble the genetic linkage map of BC1 (Figure 2b), including 174 AFLP, 46 SSR, 43 COSII, 3 COSI and 2 CAPS markers. The markers were distributed over 12 linkage groups with LOD scores ≥ 3.0. The average number of markers per linkage group was 22. The total genetic distance covered by the markers was 1014.94 cM, with an average interval of 4.12 cM between adjacent markers. The markers were well distributed over all the 12 linkage groups with only two intervals ≥ 25 cM in chromosomes 5 and 10. Marker distribution in both maps indicates that they will be useful for tagging the traits studied.
The order and placement of SSR markers were in agreement with the S. lycopersicum x S. pennellii reference tomato-EXPEN 2000 map (SGN) with the exception of TAHINA-6-64 (in silico designed), which was expected to be positioned on chromosome 6 (position 64) but is positioned on chromosome 8 (position 8.85) in our F2 map.
Distorted segregation
42.45% of the mapped markers deviated significantly from the expected 1:2:1 segregation ratio for the F2 generation at P < 0.05 (Figure 2a). Segregation distorted markers (SDMs) were mainly observed on chromosomes 1 (0.00-63.17 cM), 3 (33.24-38.85 cM), 4 (19.74-92.09 cM), 5 (12.60-72.26 cM), 6 (0.00-55.38 cM) and 10 (0.00-51.24 cM). SDMs were generally caused by a surplus of S. pennellii homozygotes, with the exception of that observed on chromosome 5.
In the BC1 population (Figure 2b), SDMs were fewer (30.3%) than in F2, and were observed mainly on chromosomes 6 (0.00-6.75 cM), 8 (0.00-15.40 cM), 11 (25.80-27.48 cM) and 12 (28.23-60.63 cM). The distortion on chromosome 8 was caused by a surplus of tomato homozygotes, whereas distortions on the other chromosomes were caused by an excess of hybrid genotypes.
QTL Identification
In order to identify QTLs, we first used Interval Mapping (IM) analysis that resulted in the identification of one QTL in the F2 population and six in the BC1 population (See Additional Files 3, 4, 5 and 6). The QTL identified in F2, located on chromosome 7, overlapped for the three traits. In the BC1 analysis, this QTL also appeared for the R and PR traits. However, in this population, another five QTLs were identified on chromosomes 1, 3, 4 (at two different areas: 4a and 4b) and 8. All these QTLs were confirmed by restricted Multiple QTL Mapping (rMQM) analysis (Figure 2b, Table 2). With the exception of the QTL on chromosome 8, all QTLs come from S. pennellii. These QTLs were named by their origin, Sp for S. pennellii or Sl for S. lycopersicum, followed by Rg (referring to regeneration) and the number of the chromosome on which they were located.
Table 2.
QTLs for shoot regeneration traits (Bud percentage (B), Regeneration percentage (R) and Productivity Rate (PR)) found to be significant at the empirical genome wide mapping threshold by restricted Multiple QTL Mapping (rMQM) in BC1 and Interval Mapping (IM) in F2
| Test | QTL analysis | Trait | QTL | Genome wide significant threshold level (P < 0.05) | Chr | Start (cM) |
Finish (cM) |
Coverage (cM) | LOD Peak | Position of LOD peak (cM) | Peak marker a | %variance explained | Estimated additive effect | Estimated dominance effect |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BC1 | rMQM | B | SpRg-1 | 2.7 | 1 | 3.87 | 44.42 | 40.55 | 7.12 | 22.47 | C2_At1g65520/C2_At2g45910 | 23.9 | -31.56 | |
| BC1 | rMQM | R | SpRg-1 | 2.7 | 1 | 3.87 | 43.42 | 39.55 | 5.52 | 24.47 | C2_At1g65520/C2_At2g45910 | 15.0 | -24.10 | |
| BC1 | rMQM | PR | SpRg-1 | 2.8 | 1 | 3.87 | 34.42 | 30.55 | 4.19 | 22.47 | C2_At1g65520/C2_At2g45910 | 10.2 | -0.70 | |
| BC1 | rMQM | B | Rg-3 | 2.7 | 3 | 42.41 | 55.80 | 13.39 | 4.64 | 50.47 | ME20-199 | 12.2 | -21.60 | |
| BC1 | rMQM | PR | Rg-3 | 2.8 | 3 | 32.77 | 63.10 | 30.33 | 4.26 | 50.47 | ME20-199 | 10.6 | -0.68 | |
| BC1 | rMQM | R | SpRg-4a | 2.7 | 4 | 44.39 | 61.24 | 16.85 | 4.94 | 50.24 | TAHINA-4-71, 3 | 13.3 | -22.29 | |
| BC1 | rMQM | PR | SpRg-4b | 2.8 | 4 | 81.33 | 93.18 | 11.85 | 3.08 | 86.33 | SSR214/SSR146 | 7.4 | -0.63 | |
| F2 | IM | B | SpRg-7 | 3.7 | 7 | 2.20 | 40.28 | 38.08 | 6.84 | 19.51 | ME10-141/C2_At4g26680 | 27.0 | -22.20 | 12.32 |
| F2 | IM | R | SpRg-7 | 3.6 | 7 | 4.50 | 40.28 | 35.78 | 6.18 | 19.51 | ME10-141/C2_At4g26680 | 24.8 | -23.29 | 13.63 |
| F2 | IM | PR | SpRg-7 | 4.4 | 7 | 19.51 | 36.28 | 16.77 | 5.72 | 28.28 | C2_At1g17200 | 23.1 | -1.53 | -0.55 |
| BC1 | rMQM | R | SpRg-7 | 2.7 | 7 | 0.00 | 25.08 | 25.08 | 5.47 | 13.44 | TAHINA-7-43 | 14.9 | -23.13 | |
| BC1 | rMQM | PR | SpRg-7 | 2.8 | 7 | 3.54 | 28.23 | 24.69 | 5.28 | 13.44 | TAHINA-7-43 | 13.5 | -0.77 | |
| BC1 | rMQM | B | SpRg-8 | 2.7 | 8 | 41.18 | 53.37 | 12.19 | 3.84 | 46.37 | C2_At1g64150 | 12.2 | 21.25 | |
| BC1 | rMQM | R | SpRg-8 | 2.7 | 8 | 42.18 | 58.90 | 16.72 | 4.25 | 53.37 | C2_At1g64150/C2_At4g23840 | 9.3 | 19.35 | |
aIn case of the absence of a peak marker, loci flanking the likely peak of a QTL are shown.
QTLs for regeneration traits in the BC1 population
Bud percentage (B)
IM analysis identified two QTLs on chromosomes 1 and 8 (SpRg-1 and SlRg-8; Additional File 4). SpRg-1 has a maximum LOD score of 5.87 and is spanned by markers SSR316 and ME17-70. This QTL explained 22.9% of the phenotypic variation of the B trait. SpRg-8, with a maximum LOD score of 2.8, including just the C2_At1g64150 marker, explained 11.7% of the phenotypic variation in B. rMQM analysis, using C2_At2g45910 (chromosome 1) and C2_At1g64150 (chromosome 8) markers as cofactors, confirmed those QTLs detected by IM and detected a new one on chromosome 3 (Figure 2b, Additional File 4). QTL characteristics are shown in Table 2. Collectively, these QTLs explained 34.6% and 48.3% of phenotypic variance in IM and rMQM, respectively.
Regeneration percentage (R)
IM analysis identified three QTLs located on chromosomes 1, 4 and 7 denominated SpRg-1, SpRg-4a and SpRg-7, respectively. The three QTLs had maximum LOD scores of 4.20, 3.92 and 3.86, and each explained around 16-17% of the phenotypic variation (see Additional File 5). rMQM analysis, using C2_At2g45910 (chromosome 1), TAHINA-4-71.3 (chromosome 4) and TAHINA-7-43 (chromosome 7) markers as cofactors, confirmed all QTLs detected by IM and detected the SlRg-8 QTL (Figure 2b, Additional File 5, Table 2). In this case, the percentage of the phenotypic variation explained by each QTL was 15% for SpRg-1, 13.3% for SpRg-4a, 14.9% for SpRg-7 and 9.3% for SlRg-8. Collectively, these QTLs explained 48.7% and 52.5% of the phenotypic variance in IM and rMQM, respectively.
Productivity rate (PR)
IM detected the QTLs located previously for B and R on chromosomes 1, 3 and 7 (Figure 2b, Additional File 6), as well as another QTL on chromosome 4, denominated SpRg-4b. The maximum phenotypic variation for PR (17.4%) is explained by SpRg-7, and the lowest (11.9%) by a QTL on chromosome 3. rMQM analysis, using SSR92 (chromosome 1), ME20-199 (chromosome 3), SSR146 (chromosome 4) and TAHINA-7-43 (chromosome 7) markers as cofactors, confirmed the QTLs detected by IM (Table 2).
Mapping tomato candidate genes
We selected the acid invertase gene linked to the Rg-2 regeneration gene of S. chilense [22] and the LESK1 gene, described as a marker in tomato for in vitro regeneration competence [27], as the tomato candidate genes.
The amplification products of the acid invertase gene marker (invpenn) produce fragments of different sizes: 162 bp for S. lycopersicum cv. Anl27 and 173 bp for S. pennellii (see Additional File 7). Thus, invpenn was used for mapping the BC1 population (Figure 2b, Additional Files 4 and 6). It was located in the QTL detected on chromosome 3, between the C2_At5g23880 and SSRB50753 markers, at positions 49.9 cM and 49.93 cM, respectively. For this reason, we named this QTL Rg-3 (a putative allele of Rg-2).
The LESK1 gene is located in the SGN Tomato-EXPEN 2000 map on chromosome 3 between markers C2_At4g18230 and cLPT-5-e7 (7 - 15 cM). As a result, in our BC1 map, LESK1 must be placed between C2_At4g18230 and TAHINA-3-44 (7 - 44 cM). Thus, this candidate gene is outside the located Rg-3 QTL.
Exploring QTLs
The official annotation for the tomato genome provided by the International Tomato Annotation Group at the SGN was used to carry out a preliminary search for related regeneration genes near the identified QTL peaks. We found a histidine kinase in SpRg-7, several serine/threonine kinases in all identified QTLs, ethylene response factors (ERFs) in all identified QTLs with the exception of SpRg-4b, cyclines in SpRg-1, Rg-3, SpRg-4a and SpRg-7 and MADS-box in SpRg-1, SpRg-4a and SpRg-7.
Discussion
The wild tomato species S. peruvianum, S. pimpinellifolium L. and S. chilense were used as sources of regeneration genes in order to study the genetics of the in vitro regeneration in tomato [7,9,21]. In this study, we used one accession of S. pennellii (PE-47) as the high regeneration parent [29]. This accession, along with a previously selected low regenerating tomato cultivar (cv. Anl27), was used to develop two mapping populations (F2 and BC1). The use of the introgression lines of S. pennellii in the M82 tomato background [30] had been previously ruled out for this analysis because of the high regeneration ability of both parent lines (data not shown). Data in Figure 1 and Table 1 seem to indicate complete dominance for B, partial dominance for R and additive effects for PR. This is in agreement with other reported studies on tomato where dominance, to different degrees, depending on the regeneration trait studied, was also reported [21,22,24,25]. B and R traits show a high correlation in both populations, suggesting that common or linked genes control these traits. The correlation between PR and both B and R was lower. This could imply that other genes may be influencing the PR trait and/or variations between different biological samples are higher in PR (for instance, competition for development due to the presence of different shoots in a similar explant area). Thus, the low sample size may be also a possible explanation for the lower correlation.
Some descendants in the F2 population showed phenotypes for the PR trait that are more extreme that those shown by the regenerating parent line (Figure 1). Transgressive segregation has already been described in other reports in relation to the genetic control of plant regeneration [31-33], and suggests poligenic inheritance [34]. It also suggests the existence of alleles that promote, and others that inhibit, in vitro regeneration, with only some of the alleles with positive effects occurring in the same parent [34]. In fact, in this study, the SlRg-8 QTL that contributes to regeneration came from the low regenerating parent.
Plant regeneration from cultured tissues is assumed to fall under quantitative genetics [34], although evidence in tomato [22,25] and other vegetables [35-37] indicates that just a few genes could be responsible for regeneration. We identified 6 QTLs in the BC1 analysis, which is indicative of the participation of a large number of genes in this character. These QTLs are situated on chromosomes 1, 3, 4, 7 and 8 (Figure 2b). The percentage of variance explained by each QTL ranges from 7.4 to 27%, which is in accordance with the most common range (6-26%) reported in the genetic mapping of QTLs for tissue culture response in plants [34]. We used three traits (B, R and PR) as a measurement of regeneration capability that could be useful for detecting chromosome regions that act at different times.
In the F2 population, only the QTL of chromosome 7 was identified for all analyzed traits (Additional File 3); the SDMs observed in most chromosome areas where QTLs were detected in the BC1 population are most likely the cause (Figure 2). The SDMs on chromosomes 1, 3, 6, 10 and 11 were also observed in similar areas in the Tomato-EXPEN 2000 map [38]. SDMs affect the detection power of QTLs when QTLs and SDMs are closely linked [39], as occurred in our case. Deviation from the expected segregation ratio is a common feature of inter-specific tomato crosses [40]. To wit: in a F2 population from S. lycopersicum x S. pennellii, De Vicente and Tanskley [41] reported a skewness rate of up to 80%.
In the BC1 population, three QTLs were detected for B: SpRg-1, SpRg-3 and SlRg-8. These QTLs may be associated with the first stages of regeneration, that is, hormonal induction response and bud formation. SpRg-1, which explained the highest percentage of variation for B (23.9%), was also identified for the R and PR traits. Given that bud formation is a necessary prerequisite for the production of shoots, it was expected that this major QTL for B would be found for R and PR, which in fact turned out to be the case (Table 2). For R and PR, a common QTL on chromosome 7 (SpRg-7) was also identified. In addition, two QTLs were detected for R (SpRg-4a and SlRg-8) and PR (SpRg-4b and Rg-3). All these QTLs seem to be involved in the development of buds into shoots. As can also be observed in this study, common QTLs for the different regeneration traits, as well as a higher number of QTLs for traits related to plant development compared to those associated with bud induction, have been reported in different studies [42,43]. For instance, in Arabidopsis, Schianterelli et al. [43] found a common area of chromosome 1 in all analyzed parameters, a peak in chromosome 4 and another in chromosome 5 when they analyzed the total number of regenerated shoots. In wheat, Ben Amer et al. [42] identified three QTLs, two that affect green spot initiation and shoot regeneration and a third that only influences plant formation.
A partial common genetic system controlling the regeneration frequency of diverse types of explants has been reported by Molina and Nuez [36] in melon. This indicates that using different explants for loci detection may lead to the identification of some common QTLs, but also to the possible identification of other new QTLs. Root explants were used by Koornneef et al. [25] and Satoh et al. [22] for phenotyping, at which point two alleles for regeneration ability were located on chromosome 3 of tomato. In the present study, leaves were used for phenotyping and a QTL (Rg-3) in a similar area of chromosome 3 was detected in addition to other QTLs that influence regeneration and were identified on chromosomes 1, 4, 7 and 8. Differences in root and leaf explants for QTL identification were also found in Arabidopsis thaliana [43].
Koornneef et al. [25] located a dominant allele from S. peruvianum (Rg-1) near the middle of chromosome 3 that determines efficient shoot regeneration in tomato root explants. Satoh et al. [22] mapped a putative allele (Rg-2) from S. chilense on this chromosome. The acid invertase gene, reported as a marker linked to Rg-2, was chosen for mapping Rg-2 in our population derived from S. pennellii. The polymorphisms detected in our parents allow us to map this gene in the QTL detected on chromosome 3 that we named Rg-3. We consider Rg-3 to be a putative allele of the Rg-2 gene. Allelism must be confirmed.
The other gene chosen as a candidate was LESK1, which encodes a serine/threonine kinase, and was reported as a marker of competence for in vitro regeneration in tomato [27,28]. This gene was positioned on chromosome 3, but it is not located in the Rg-3 QTL.
The recent release of the entire genome sequence of tomato provides a powerful tool for interrogating QTL data. In this respect, we have taken a preliminary look at genes located at the peak areas of the detected QTLs, and which could be related to organogenesis. Histidine kinases were reported as cytokinin receptors [15-17]. In our QTL peaks, only one histidine kinase is located in the SpRg-7 QTL. The candidate tomato gene, LESK1, which has been described as a marker for in vitro competence, encodes a serine/threonine kinase. We looked for serine/threonine kinases and found this kind of protein in all identified QTLs. Other putative candidate genes could be ESR1 and its paralogue, ESR2, from Arabidopsis, which are the best-characterized genes related to regeneration [18,19]. These genes code for ethylene response factors (ERF). We found ERFs, which contain the AP2 domain, in all analysed QTLs with the exception of SpRg-4b. Cyclines related to cell division [13] were found in SpRg-1, Rg-3, SpRg-4a and SpRg-7. MADS-box genes, which have been correlated to adventitious regeneration induction and regulation [44,45], were found in the SpRg-1, SpRg-4a and SpRg-7 QTL peaks.
Conclusions
The results obtained in this study may very well represent a significant step toward the goal of understanding the processes underlying tomato tissue culture and regeneration responses. We have situated six QTLs on chromosomes 1, 3, 4, 7 and 8, five from S. pennellii and one from S. lycopersicum. The most important QTLs are SpRg-1, which is most likely associated with the morphogenetic response, and SpRg-7, which promotes bud development. A QTL detected on chromosome 3, Rg-3, likely contains a putative allele of the Rg-1 and Rg-2 genes, as is shown by mapping the acid invertase gene linked to Rg-2. QTLs detected on chromosomes 8 and 4 most likely contain genes influencing bud formation and development, respectively.
Methods
Plant materials and growing conditions
S. pennellii PE-47, which showed a high ability for regeneration [29], and the tomato cultivar Anl27 (cv. Anl27), with a low ability for regeneration, were chosen for obtaining the mapping population. The initial genotypes were established in vitro, starting with the sterilization of seeds by immersion for 10 min in a solution of 25% commercial bleach (40 g L-1 active chlorine), being then washed twice with sterile deionized water for 5 min each and then sown in Petri dishes containing nutrient medium (Murashige and Skoog [46] salts including vitamins, 2% sucrose, 0.6% plant agar (DUCHEFA, the Netherlands). The pHs of the media were adjusted to 5.8 before sterilization at 121°C for 20 min. Cultures were incubated in a growth chamber at 26°C ± 2°C under a 16h photoperiod with cool white light provided by Sylvania cool white F37T8/CW fluorescent lamps (90 μmol m-2 s-1). Clones of one plant of each genotype were obtained and maintained in in vitro culture. The clones were multiplied by transferring nodes to tubes with fresh basal medium (BM: Murashige and Skoog -[46]- salts including vitamins, 1.5% sucrose and 7 g L-1 plant agar) every 3-4 weeks. The tubes were 15 cm in length and 22 mm in diameter, with 15 ml of medium per tube.
Mapping population
One clone of tomato and another of S. pennellii were transferred to a greenhouse in order to obtain the F1 plant that was reintroduced in vitro by disinfection of shoots following a similar procedure as that carried out for seed sterilization. F2 and BC1 populations were obtained and seeds were germinated in vitro as described above.
The F2 mapping population was composed of 106 individuals obtained from selfing one F1 plant, the result of a cross between the tomato cv. Anl27 (P1) and S. pennellii PE-47 (P2). The backcross (BC1) mapping population, composed of 113 plants, was obtained by crossing the cv. Anl27 and the F1 plant. To allow the test to be reproduced, the F1 plant and F2 and BC1 individuals were clonally replicated and maintained in vitro as described above.
Evaluation of the regeneration capacity
A first assay was performed with cloned P1, P2 and F1 plants. Leaf disks (0.6-0.8 cm2) obtained from in vitro cultured plants that were at a similar growing stage were placed with the abaxial side in contact with the shoot induction medium (SIM) containing Murashige and Skoog salts [46], 3% sucrose, 7% plant agar and 0.2 mg L-1 zeatin riboside (ZR). This growth regulator was sterilized by filtration and added to the sterile SIM. After 30 days of culture on SIM, the explants were transferred to BM for 20 days. In this medium, buds develop into shoots. For each genotype, five explants per plate (90 × 25 mm with 40 ml of medium per plate) and 10 repetitions per genotype were evaluated. At the end of the experiment, the following variables were analyzed:
-Bud percentage (B): number of explants with buds × 100/total number of cultured explants.
-Regeneration percentage (R): number of cultures that differentiated into completely developed shoots × 100/total number of cultured explants.
-Productivity rate (PR): total number of completely developed shoots/total number of cultured explants that regenerated plants.
In a second assay, leaf explants of F2, BC1, P1, P2 and F1 plants were tested as explained above. In this case, for each genotype, five explants per plate and 4 repetitions per genotype were evaluated. Data for regeneration was obtained for 102 genotypes of the F2 population and 104 genotypes of BC1. The average value for each trait and genotype was used for QTL analysis.
To assess the effect of genotype on regeneration ability, data from the genetically uniform classes (P1, P2 and F1) were subjected to a unifactorial analysis of variance (ANOVA), and then means for the different traits were separated by a Duncan test. The correlations between the different traits were calculated using the Statgraphics Plus 4.0 software.
Genotyping
Preparation of genomic DNA
Young leaves from in vitro-cultured plants were collected and immediately frozen with liquid nitrogen and then stored at -80°C. DNA was prepared based on the modified CTAB method of Doyle and Doyle [47]. Subsequently, quality and quantity of the DNA was evaluated on 0.8% agarose gel stained with ethidium bromide and using the NanoDrop® ND-1000 Spectrophotometer.
Amplified fragment length polymorphism (AFLP) procedure
AFLPs were obtained following de Vos et al. [48] procedure. Fifteen and sixteen selective combinations of primers were used for the F2 and BC1 populations, respectively. The code of each selective combination is specified in Table 3. Each code followed by the number corresponding to each obtained band (size in bp) is used to name the polymorphic AFLPs. Electrophoresis of the PCR products was conducted using an ABI PRISM 310 Genetic Analyzer (PerkinElmer Applied Biosystems, Foster City, California, USA). GeneScan™ 600 LIZ® Size Standard, with fluorophore LIZ, was used as a molecular size marker. Raw data were analyzed with the GeneScan 3.1.2 analysis software (PerkinElmer Applied Biosystems) and the resulting GeneScan trace files were imported into Genographer 1.6.0. The AFLP fragments between 60 to 380 bp were scored in Genographer as present (1) or absent (0).
Table 3.
Selective combinations of primers used for F2 and BC1 genotyping
| Code | Mapping population | Selective primers combination |
|---|---|---|
| ME1 | F2, BC1 | MseI CTA-EcoRI AAC |
| ME2 | F2 | MseI CAA-EcoRI ACC |
| ME3 | F2 | MseI CAA-EcoRI ACG |
| ME4 | F2, BC1 | MseI CAA-EcoRI AGC |
| ME5 | F2 | MseI CAC-EcoRI ACA |
| ME6 | F2, BC1 | MseI CAC-EcoRI ACG |
| ME7 | F2 | MseI CAC-EcoRI AGC |
| ME8 | F2, BC1 | MseI CAA-EcoRI ACA |
| ME9 | F2, BC1 | MseI CAA-EcoRI AAC |
| ME10 | F2 | MseI CTA-EcoRI AGC |
| ME11 | F2 | MseI CTC-EcoRI AGC |
| ME12 | F2 | MseI CCG-EcoRI AAC |
| ME13 | F2 | MseI CCG-EcoRI ACC |
| ME14 | F2 | MseI CCG-EcoRI ACG |
| ME15 | F2 | MseI CTC-EcoRI AGG |
| ME16 | BC1 | MseI CAA-EcoRI ACT |
| ME17 | BC1 | MseI CTA-EcoRI ACC |
| ME18 | BC1 | MseI CTA-EcoRI ATG |
| ME19 | BC1 | MseI CTA-EcoRI ACA |
| ME20 | BC1 | MseI CCT-EcoRI ACC |
| ME21 | BC1 | MseI CCT-EcoRI AAC |
| ME22 | BC1 | MseI CCT-EcoRI ATG |
| ME23 | BC1 | MseI CCT-EcoRI ACA |
| ME24 | BC1 | MseI CAC-EcoRI ACC |
| ME25 | BC1 | MseI CAC-EcoRI ATG |
| ME26 | BC1 | MseI CAC-EcoRI AGG |
Microsatellites (SSRs)
One hundred and forty-nine SSR markers were used to detect polymorphism between P1 and P2, which included 89 SSRs previously reported and mapped onto the Tomato-EXPEN 2000 available at SGN [49,50], along with 60 new SSRs: 18 from the COMAV research group "Aprovechamiento de la variabilidad estraespecífica en la mejora del tomate" and 42 designed from sequences deposited in Genbank (see Additional File 2). Primer pairs were designed from these sequences using the SSR Primer 3 tool http://frodo.wi.mit.edu/[51]. The criteria used for designing the primers were as follows: the primer Tm ranged from 55 to 65°C and GC content was 50%. The presence of G or C bases within the last five bases from the 3' end of primers (GC clamp), which helps promote specific binding at the 3' end, was taken into account. In order to design the SSRs, wherever possible the AT/TA repetitions were selected based on the results obtained by Frary et al. [49].
All the SSRs, with the exception of those specified below, were labelled following the M13-tail method described by Schuelke et al. [52]. DNA amplification was carried out in volumes of 15 μL using a sample of 10 ng of DNA. The reaction mixture contained 1.5 μL 10 × PCR buffer [75 mM Tris-HCl (Ph 9.0), 50 mM KCl, 20 mM (NH4)2SO4 and 0.001% BSA], 2 mM MgCl2, 200 μM dNTPs, 0.133 μM of primers, 0.2 μM of fluorescent labelled M13 primer and 0.3 units of TaqI DNA polymerase (Need S. L., Valencia, Spain). An Eppendorf 5333 Thermal Cycler was used. The PCR parameters included the following: an initial 3 min at 94°C; 35 cycles, each with 30 s DNA denaturation at 94°C; 45 s at an annealing temperature (depending on the primer combination Tm) and a 1 min extension at 72°C, and a final extension of 10 min at 72°C. Amplified bands were visualized using a LI-COR sequencing gel (DNA LI-COR 4300; LI-COR Biosciences, Lincoln, Nebraska, USA); 10 μl of loading buffer (95% formamide, 2 mM EDTA, 0.001% bromophenol blue) and 5 μl of deionized water were added to the 5 μl PCR mix (2.5 μl of each IRDye 700 or IRDye800-labeled) samples which were denatured at 96°C for 8 min. Electrophoresis was performed in denaturing conditions at 50°C, using 6% acrylamide gels in TBE buffer.
The SSR356, SSR73, SSR248, SSR46 markers in which polymorphisms were visible in the agarose gels were amplified in volumes of 23.32 μl with: 10 ng of DNA, 1.6 mM MgCl2, 171.52 μM dNTPs, 0.214 μM of primers, 2.5 μl of 10 × PCR buffer, and 0.6 U TaqI DNA polymerase. The PCR conditions were similar to those applied before, with the exception of a final extension of 30 min in this case. Amplified bands were run in standard agarose gels (1 or 2%) in TAE buffer at 100V and visualized by ethidium bromide staining.
Conserved ortholog set (COS) and cleaved amplified polymorphic sequence (CAPS) markers
Ninety-six markers (86 COSII, 6 COSI, 4 CAPS) from the Tomato-EXPEN 2000 map [53,54] and one developed CAPS marker (Solyc07g049350) were tested for polymorphism between the P1 and P2 parents. The restriction enzymes used when required were those indicated in the SGN database. When restriction enzymes was needed, the protocol described in the commercial product's instructions (Fermentas, York, UK or Biolabs, Takara, Japan) was followed.
The PCR reaction was performed in a total volume of 12 μL using a sample of 10 ng of DNA. The reaction mixture contained 1.5 mM MgCl2, 200 μM dNTPs, 0.25 μm of primers, 1.2 μL PCR buffer 10X, and 0.3 U TaqI DNA polymerase. Amplification was performed using an Eppendorf 5333 Thermal Cycler, which was programmed as follows: 5 min at 94°C, 35 30-s cycles each at 94°C, 1 min at Ta (depending on the primer combination Tm) and a 2 min extension at 72°C, with a final stage of 10 min at 72°C. Amplified bands were separated by 1 or 2% agarose electrophoresis in TAE buffer at 100V, and visualized by ethidium bromide staining.
Map construction and QTL mapping
Linkage analysis for both mapping populations was performed with the JoinMap® 4.0 software [55]. Markers were grouped into linkage groups at LOD ≥ 3, with the exception of those in chromosomes 9 and 10 of the BC1 mapping population with LOD ≥ 2. Order was determined with a recombination threshold of 0.40 and distances were calculated using the Kosambi mapping function (Kosambi 1944). For the genetic map construction, AFLP, SSR, COS and CAPS markers were used (Additional File 8). The segregation ratio of alleles was evaluated for each locus by the Chi-square test with a significance threshold of P < 0.05. The expected segregation ratios were 3:1 and 1:1 for F2 and BC1, respectively. Visual representations of the marker maps were created with the MapChart software [56].
QTL analysis on the F2 and BC1 phenotypic data sets was performed with the MapQTL® 6.0 software [57]. Significance thresholds for the LOD values, corresponding to a genome-wide false discovery rate of 5% (p < 0.05) were calculated by genome-wide permutation tests using 1, 000 permutations. Firstly, IM analysis was performed (simple Interval Mapping). Then, if many putative QTLs were detected by IM, markers close to the likelihood peaks of the detected QTLs were used as cofactors for rMQM (also called composite Interval Mapping) analyses.
Locating candidate genes and looking for other regeneration-related genes
The S. pennellii acid invertase gene (invpenn) was analyzed and mapped as a marker. Primers described by Harada et al. [58] were used for DNA amplification using conditions previously described for COS and CAPS markers. Amplified bands were separated using the multicapillary electrophoresis QIAxcel System (Qiagen, Valencia, California, USA). We searched for the location of the LESK1 gene at SGN and for its nearest markers at International Tomato Annotation Group. This database was also used for looking for genes putatively related to organogenesis.
Authors' contributions
CG obtained the mapping populations. CT conducted the population phenotyping and genotyping and participated in the drafting. CT and SV performed the map construction and QTL mapping. CG collaborated in the phenotyping and genotyping and conceived, supervised and drafted the manuscript. FN conceived of the study and contributed to critically reviewing the manuscript. All authors read and approved the final manuscript.
Supplementary Material
Regeneration response of leaf explants. Regeneration response of leaf explants from parents [tomato (cv. Anl27); S. pennellii (PE-47)], F1, F2 and BC1 populations, cultured on shoot induction medium (SIM) for 30 days and transferred to basal medium (BM) for 20 days.
In silico-designed SSR markers. Table with the name, band size, repeat motif, temperature of annealing and primers sequences of in silico-designed SSR markers.
Genetic location and LOD score profile of the F2-QTLs for regeneration components detected by Interval Mapping on chromosome 7 (SpRg-7). Genetic location and LOD score profile of the F2-QTLs for regeneration components (Bud percentage (B), Regeneration percentage (R) and Productivity Rate (PR)). On the left, projections of QTLs as black bars indicate the SpRg-7 for B, R and PR traits. The vertical dotted line indicates the 95% significant threshold value for declaring a QTL (B LOD threshold = 3.7) (R LOD threshold = 3.6) (PR LOD threshold = 4.4). Map position (cM) and distances are based on the genetic linkage map developed in this study. QTLs characteristics in attached table.
Genetic location and LOD score profile of the BC1-QTLs for Bud percentage (B), detected on chromosomes 1 (SpRg-1), 3 (SpRg-3) and 8 (SlRg-8). Results from the Interval Mapping (IM) and restricted Multiple QTL Mapping (rMQM) approaches. On the left, projections as black bars (IM) and grey bars (rMQM) indicate the range of SpRg-1, SpRg-3 and SlRg-8 QTLs for B. The vertical dotted line indicates the 95% significant threshold value for declaring a QTL (B LOD threshold = 2.7). The horizontal dotted line indicates the position of the acid invertase gene (invpenn) marker included in the chromosome 3 QTL range. Map position (cM) and distances are based on the genetic linkage map developed in this study.
Genetic location and LOD score profile of the BC1-QTLs for Regeneration percentage (R), detected in this study on chromosomes 1 (SpRg-1), 4 (SpRg-4a), 7 (SpRg-7) and 8 (SlRg-8). Results from the Interval Mapping (IM) and restricted Multiple QTL Mapping (rMQM) approaches. On the left, projections as black bars (IM) and grey bars (rMQM) indicate the range of SpRg-1, SpRg-4a, SpRg-7 and SlRg-8 QTLs for R. The vertical dotted line indicates the 95% significant threshold value for declaring a QTL (R LOD threshold = 2.7). Map position (cM) and distances are based on the genetic linkage map developed in this study.
Genetic location and LOD score profile of the BC1-QTLs for Productivity Rate (PR), detected in this study on chromosomes 1 (SpRg-1), 3 (SpRg-3), 4 (SpRg-4b) and 7 (SpRg-7). Results from the Interval Mapping (IM) and restricted Multiple QTL Mapping (rMQM) approaches. On the left, projections as black bars (IM) and grey bars (rMQM) indicate the range of SpRg-1, SpRg-3, SpRg-4b and SpRg-7 for PR. The vertical dotted line indicates the 95% significant threshold value for declaring a QTL (PR LOD threshold = 2.8). Horizontal dotted lines indicate the position of the acid invertase gene (invpenn) marker included in the chromosome 3 QTL range. Map position (cM) and distances are based on the genetic linkage map developed in this study.
Polymorphic acid invertase gene marker (invpenn). Amplified bands separated using the multicapillary electrophoresis QIAxcel System. Lane 1: S. lycopersicum L. (Anl27), band size (~162bp). Lane 2: S. pennellii PE-47, band size (~173bp). Lane 3: F1 Hybrid S. lycopersicum L. (Anl27) × S. pennellii PE-47, both bands (~162bp-~173bp). Lane 4: negative control.
Markers used for genotyping the F2 and BC1 population. SSR, COS, COSII, CAP markers used for genotyping the F2 and BC1 population.
Contributor Information
Carlos Trujillo-Moya, Email: cecaltrujillo@yahoo.es.
Carmina Gisbert, Email: cgisbert@btc.upv.es.
Santiago Vilanova, Email: sanvina@btc.upv.es.
Fernando Nuez, Email: fnuez@btc.upv.es.
Acknowledgements
CG and CT thank the Spanish 'Ministerio de Educación y Ciencia' for a Ramón y Cajal contract and a predoctoral fellowship, respectively. The authors acknowledge the financial support of the Instituto de Conservación y Mejora de la Agrodiversidad Valenciana (COMAV), suggestions from Dr. J. Cañizares, SSRs designed by O. Julián and the technical assistance of N. Palacios. The revision and the English revision of the manuscript by A. Monforte and J. Bergen, respectively, are also acknowledged.
References
- Bhatia P, Ashwath N, Senaratna T, David M. Tissue culture studies of tomato (Lycopersicon esculentum) Plant Cell Tissue Organ Cult. 2004;78:1–21. [Google Scholar]
- Engelmann F. In vitro conservation of tropical plant germplasm. Euphytica. 1991;57:227–243. doi: 10.1007/BF00039669. [DOI] [Google Scholar]
- Benson EE. Special symposium: In vitro plant recalcitrance: An introduction. In Vitro Cell Dev Biol Plant. 2000;36:141–148. doi: 10.1007/s11627-000-0029-z. [DOI] [Google Scholar]
- Hamza S, Chupeau Y. Re-evaluation of conditions for plant regeneration and Agrobacterium-mediated transformation from tomato (Lycopersicon esculentum) J Exp Bot. 1993;44:1837–1845. doi: 10.1093/jxb/44.12.1837. [DOI] [Google Scholar]
- Frary A, Earle ED. An examination of factors affecting the efficiency of Agrobacterium-mediated transformation of tomato. Plant Cell Rep. 1996;16:235–240. doi: 10.1007/BF01890875. [DOI] [PubMed] [Google Scholar]
- Peres LEP, Morgante PG, Vecchi C, Kraus JE, Sluys MAv. Shoot regeneration capacity from roots and transgenic hairy roots of tomato cultivars and wild related species. Plant Cell Tissue Organ Cult. 2001;65:37–44. doi: 10.1023/A:1010631731559. [DOI] [Google Scholar]
- Koornneef M, Hanhart CJ, Martinelli L. A genetic analysis of cell culture traits in tomato. Theor Appl Genet. 1987;74:633–641. doi: 10.1007/BF00288863. [DOI] [PubMed] [Google Scholar]
- Wijbrandi J, Vos JGM, Koornneef M. Transfer of regeneration capacity from Lycopersicon peruvianum to L. esculentum by protoplast fusion. Plant Cell Tissue Organ Cult. 1988;12:193–196. doi: 10.1007/BF00040085. [DOI] [Google Scholar]
- Takashina T, Suzuki T, Egashira H, Imanishi S. New molecular markers linked with the high shoot regeneration capacity of the wild tomato species Lycopersicon chilense. Breed Sci. 1998;48:109–113. doi: 10.1270/jsbbs1951.48.109. [DOI] [Google Scholar]
- Faria RT, Destro D, Bespalhok JC, Illg RD. Introgression of in vitro regeneration capability of Lycopersicon pimpinellifolium Mill. into recalcitrant tomato cultivars. Euphytica. 2002;124:59–63. doi: 10.1023/A:1015693902836. [DOI] [Google Scholar]
- Sugiyama M. Organogenesis in vitro. Curr Opin Plant Biol. 1999;2:61–64. doi: 10.1016/S1369-5266(99)80012-0. [DOI] [PubMed] [Google Scholar]
- Zhang S, Lemaux PG. Molecular analysis of in vitro shoot organogenesis. Crit Rev Plant Sci. 2004;23:325–335. doi: 10.1080/07352680490484569. [DOI] [Google Scholar]
- Hemerly AS, Ferreira P, de Almeida Engler J, Van Montagu M, Engler G, Inze D. cdc2a expression in Arabidopsis is linked with competence for cell division. Plant Cell. 1993;5:1711–1723. doi: 10.1105/tpc.5.12.1711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skoog F, Miller CO. Chemical regulation of growth and organ formation in plant tissue cultures in vitro. Symp Soc Exp Biol. 1957;11:118–131. [PubMed] [Google Scholar]
- Inoue T, Higuchi M, Hashimoto Y, Seki M, Kobayashi M, Kato T, Tabata S, Shinozaki K, Kakimoto T. Identification of CRE1 as a cytokinin receptor from Arabidopsis. Nature. 2001;409:1060–1063. doi: 10.1038/35059117. [DOI] [PubMed] [Google Scholar]
- Ueguchi C, Sato S, Kato T, Tabata S. The AHK4 gene involved in the cytokinin-signalling pathway as a direct receptor molecule in Arabidopsis thaliana. Plant and Cell Physiology. 2001;42:751–755. doi: 10.1093/pcp/pce094. [DOI] [PubMed] [Google Scholar]
- Nishimura C, Ohashi Y, Sato S, Kato T, Tabata S, Ueguchi C. Genetic analysis of Arabidopsis histidine kinase genes encoding cytokinin receptors reveals their overlapping biological functions in the regulation of shoot and root growth in Arabidopsis thaliana. Plant Cell. 2004;16:1365–1377. doi: 10.1105/tpc.021477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banno H, Ikeda Y, Niu QW, Chua NH. Overexpression of Arabidopsis ESR1 induces initiation of shoot regeneration. Plant Cell. 2001;12:2609–2618. doi: 10.1105/tpc.010234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda Y, Banno H, Niu QW, Howell SH, Chua NH. The ENHANCER OF SHOOT REGENERATION 2 gene in Arabidopsis Regulates CUP-SHAPED COTYLEDON 1 at the transcriptional level and controls cotyledon development. Plant Cell Physiol. 2006;47:1443–1456. doi: 10.1093/pcp/pcl023. [DOI] [PubMed] [Google Scholar]
- Mase H, Hashiba M, Matsuo N, Banno H. Expression patterns of Arabidopsis ERF VIII-b subgroup genes during in vitro shoot regeneration and effects of their overexpression on shoot regeneration efficiency. Plant Biotechnol. 2007;24:481–486. doi: 10.5511/plantbiotechnology.24.481. [DOI] [Google Scholar]
- Faria RT, Illg RD. Inheritance of in vitro plant regeneration ability in the tomato. Braz J Genet. 1996;19:113–116. [Google Scholar]
- Satoh H, Takashina T, Escalante A, Egashira H, Imanishi S. Molecular markers mapped around the high shoot regeneration capacity gene Rg-2 in Lycopersicon chilense. Breed Sci. 2000;50(4):251–256. [Google Scholar]
- Pratta G, Cánepa LN, Zorzoli R, Picardi LA. Diallel analysis of in vitro culture traits in the genus Lycopersicon. HortScience. 2003;38:110–112. [Google Scholar]
- Marchionni BE, Pratta GR, Zorzoli R. Genetic analysis of the in vitro culture response in tomato. Plant Cell Tissue Organ Cult. 2007;88:233–239. doi: 10.1007/s11240-006-9195-y. [DOI] [Google Scholar]
- Koornneef M, Bade J, Hanhart C, Horsman K, Schel J, Soppe W, Vekerk R, Zabel P. Characterization and mapping of a gene controlling shoot regeneration in tomato. Plant J. 1993;3:131–141. doi: 10.1111/j.1365-313X.1993.tb00016.x. [DOI] [Google Scholar]
- Torelli A, Soragni E, Bolchi A, Petrucco S. New potential markers of in vitro tomato morphogenesis identified by mRNA differential display. Plant Mol Biol. 1996;32:891–900. doi: 10.1007/BF00020486. [DOI] [PubMed] [Google Scholar]
- Torelli A, Soragni E, Borinato M, Branca C. The expression of LESK1 morphogenetic marker along the tomato hypocotyl axis is linked to a position-dependent competence for shoot regeneration. Plant Sci. 2004;166(1):179–190. doi: 10.1016/j.plantsci.2003.09.006. [DOI] [Google Scholar]
- Torelli A, Borinato M, Soragni E, Bolpagni R, Bottura C, Branca C. The delay in hormonal treatment modulates the expression of LESK1, a gene encoding a putative serine-threonine kinase, marker of in vitro caulogenesis in tomato (Lycopersicon esculentum Mill.) Plant Sci. 2004;167(3):607–620. doi: 10.1016/j.plantsci.2004.05.007. [DOI] [Google Scholar]
- Gisbert C, Arrillaga I, Roig LA, Moreno V. Adquisition of a collection of Lycopersicon pennellii (Corr. D'Arcy) transgenic plants with uidA and nptII marker genes. J Hortic Sci Biotechnol. 1999;74(1):105–109. [Google Scholar]
- Eshed Y, Abu-Abied M, Saranga Y, Zamir D. Lycopersicon esculentum lines containing small overlapping introgressions from L. pennellii. Theor Appl Genet. 1992;83:1027–1034. doi: 10.1007/BF00232968. [DOI] [PubMed] [Google Scholar]
- Komatsuda T, Annaka T, Oka S. Genetic mapping of a quantitative trait locus (QTL) that enhances the shoot differentiation rate in Hordeum vulgare L. Theor Appl Genet. 1993;86:713–720. doi: 10.1007/BF00222661. [DOI] [PubMed] [Google Scholar]
- Taguchi-Shiobara F, Lin SY, Tanno K, Komatsuda T, Yano M, Sasaki T, Oka S. Mapping quantitative trait loci associated with regeneration ability of seed callus in rice, Oryza sativa L. Theor Appl Genet. 1997;95:828–833. doi: 10.1007/s001220050632. [DOI] [Google Scholar]
- Flores Berrios E, Gentzbittel L, Mokrani L, Alibert G, Sarrafi A. Genetic control of early events in protoplast division and regeneration pathways in sunflower. Theor Appl Genet. 2000;101:606–612. doi: 10.1007/s001220051522. [DOI] [Google Scholar]
- Bolibok H, Rakoczy-Trojanowska M. Genetic mapping of QTLs for tissue-culture response in plants. Euphytica. 2006;149:73–83. doi: 10.1007/s10681-005-9055-6. [DOI] [Google Scholar]
- Molina RV, Nuez F. Respuesta correlacionada de la capacidad de regeneración en distintos tipos de explante en Cucumis melo. Actas Horticultura. 1989;3:111–118. [Google Scholar]
- Molina RV, Nuez F. Correlated response of in vitro regeneration capacity from different source of explants in Cucumis melo. Plant Cell Rep. 1995;15:129–132. doi: 10.1007/BF01690269. [DOI] [PubMed] [Google Scholar]
- Molina RV, Nuez F. Sexual transmission of the in vitro regeneration capacity via caulogenesis of Cucumis melo L. in a medium with a high auxin/cytokinin ratio. Sci Hortic. 1997;70:237–241. doi: 10.1016/S0304-4238(97)00024-1. [DOI] [Google Scholar]
- Shirasawa K, Asamizu E, Fukuoka H, Ohyama A, Sato S, ·Nakamura Y, ·Tabata S, Sasamoto S, Wada T, Kishida Y. An interspecific linkage map of SSR and intronic polymorphism markers in tomato. Theor Appl Genet. 2010;121:731–739. doi: 10.1007/s00122-010-1344-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Wang S, Li H, Deng Q, Zheng A, Li S, Li P, Li Z, Wang J. Effects of missing marker and segregation distortion on QTL mapping in F2 populations. Theor Appl Genet. 2010;121:1071–1082. doi: 10.1007/s00122-010-1372-z. [DOI] [PubMed] [Google Scholar]
- Chaerani R, Smulders MJ, van der Linden CG, Vosman B, Stam P, Voorrips RE. QTL identification for early blight resistance (Alternaria solani) in a Solanum lycopersicum x S. arcanum cross. Theor Appl Genet. 2006;114:439–450. doi: 10.1007/s00122-006-0442-8. [DOI] [PubMed] [Google Scholar]
- de Vicente MC, Tanksley SD. QTL analysis of transgressive segregation in an interspecific tomato cross. Genetics. 1993;134:585–596. doi: 10.1093/genetics/134.2.585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben Amer IM, Korzun V, Worland AJ, Börner A. Genetic mapping of QTLs controlling tissue-culture response on chromosome 2B of wheat (Triticum aestivum L.) in relation to major genes and RFLP markers. Theor Appl Genet. 1997;94:1047–1052. doi: 10.1007/s001220050513. [DOI] [Google Scholar]
- Schiantarelli E, De La Pena A, Candela M. Use of recombinant inbred lines (RILs) to identify, locate and map major genes and quantitative trait loci involved with in vitro regeneration ability in Arabidopsis thaliana. Theor Appl Genet. 2001;102:335–341. doi: 10.1007/s001220051650. [DOI] [Google Scholar]
- Prakash AP, Kumar PP. PkMADS1 is a novel MADS box gene regulating adventitious shoot induction and vegetative shoot development in Paulownia kawakamii. Plant J. 2002;29:141–151. doi: 10.1046/j.0960-7412.2001.01206.x. [DOI] [PubMed] [Google Scholar]
- Bielenberg DG, Wang Y, Li Z, Zhebentyayeva T, Fan S, Reighard GL. et al. Sequencing and annotation of the evergrowing locus in peach [Prunus persica (L.) Batsch] reveals a cluster of six MADS-box transcription factors as candidate genes for regulation of terminal bud formation. Tree Genet Genomes. 2008;4(3):495–507. doi: 10.1007/s11295-007-0126-9. [DOI] [Google Scholar]
- Murashige T, Skoog F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant. 1962;15:473–497. doi: 10.1111/j.1399-3054.1962.tb08052.x. [DOI] [Google Scholar]
- Doyle JJ, Doyle JL. Isolation of plant DNA from fresh tissue. Focus. 1990;12:13–15. [Google Scholar]
- Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M. et al. AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res. 1995;23(21):4407–4414. doi: 10.1093/nar/23.21.4407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frary A, Xu Y, Liu J, Mitchell S, Tedeschi E, Tanksley S. Development of a set of PCR-based anchor markers encompassing the tomato genome and evaluation of their usefulness for genetics and breeding experiments. Theor Appl Genet. 2005;111:291–312. doi: 10.1007/s00122-005-2023-7. [DOI] [PubMed] [Google Scholar]
- Mueller LA, Solow TH, Taylor N, Skwarecki B, Buels R, Binns J. et al. The SOL Genomics Network: a comparative resource for Solanaceae biology and beyond. Plant Physiol. 2005;138:1310–1317. doi: 10.1104/pp.105.060707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rozen S, Skaletsky HJ. In: Bioinformatics Methods and Protocols: Methods in Molecular Biology. Krawetz S, Misener S, editor. Totowa, NJ: Humana Press; 2000. Primer3 on the WWW for general users and for biologist programmers; pp. 365–386. [DOI] [PubMed] [Google Scholar]
- Schuelke M. An economic method for the fluorescent labelling of PCR fragments. Nature Biotechnology. 2000;18:233–234. doi: 10.1038/72708. [DOI] [PubMed] [Google Scholar]
- Fulton TM, Van der Hoeven R, Eannetta NT, Tanksley SD. Identification, analysis and utilization of a conserved ortholog set markers for comparative genomics in Higher Plants. Plant Cell. 2002;14:1457–1467. doi: 10.1105/tpc.010479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu F, Mueller LA, Crouzillat D, Petiard V, Tanksley SD. Combining Bioinformatics and Phylogenetics to Identify Large Sets of Single Copy, Orthologous Genes (COSII) for Comparative, Evolutinonary and Systematics Studies: A Test Case in the Euasterid Plant Clade. Genetics. 2006;174(3):1407–1420. doi: 10.1534/genetics.106.062455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Ooijen JW. JoinMap 4, Software for the calculation of genetic linkage maps in experimental populations. Wageningen, Netherlands: Kyazma BV; 2006. [Google Scholar]
- Voorrips RE. MapChart: Software for the graphical presentation of linkage maps and QTLs. J Hered. 2002;93(1):77–78. doi: 10.1093/jhered/93.1.77. [DOI] [PubMed] [Google Scholar]
- Van Ooijen JW MapQTL ® 6, Software for the mapping of quantitative trait loci in experimental populations of diploid species 2009Wageningen, Netherlands: Kyazma BV; 20234834 [Google Scholar]
- Harada S, Fukuta S, Tanaka H, Ishiguro Y, Sato T. Genetic analysis of the trait of sucrose accumulation in tomato fruit using molecular marker. Breeding Sci. 1995;45:429–434. doi: 10.1270/jsbbs1951.45.429. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Regeneration response of leaf explants. Regeneration response of leaf explants from parents [tomato (cv. Anl27); S. pennellii (PE-47)], F1, F2 and BC1 populations, cultured on shoot induction medium (SIM) for 30 days and transferred to basal medium (BM) for 20 days.
In silico-designed SSR markers. Table with the name, band size, repeat motif, temperature of annealing and primers sequences of in silico-designed SSR markers.
Genetic location and LOD score profile of the F2-QTLs for regeneration components detected by Interval Mapping on chromosome 7 (SpRg-7). Genetic location and LOD score profile of the F2-QTLs for regeneration components (Bud percentage (B), Regeneration percentage (R) and Productivity Rate (PR)). On the left, projections of QTLs as black bars indicate the SpRg-7 for B, R and PR traits. The vertical dotted line indicates the 95% significant threshold value for declaring a QTL (B LOD threshold = 3.7) (R LOD threshold = 3.6) (PR LOD threshold = 4.4). Map position (cM) and distances are based on the genetic linkage map developed in this study. QTLs characteristics in attached table.
Genetic location and LOD score profile of the BC1-QTLs for Bud percentage (B), detected on chromosomes 1 (SpRg-1), 3 (SpRg-3) and 8 (SlRg-8). Results from the Interval Mapping (IM) and restricted Multiple QTL Mapping (rMQM) approaches. On the left, projections as black bars (IM) and grey bars (rMQM) indicate the range of SpRg-1, SpRg-3 and SlRg-8 QTLs for B. The vertical dotted line indicates the 95% significant threshold value for declaring a QTL (B LOD threshold = 2.7). The horizontal dotted line indicates the position of the acid invertase gene (invpenn) marker included in the chromosome 3 QTL range. Map position (cM) and distances are based on the genetic linkage map developed in this study.
Genetic location and LOD score profile of the BC1-QTLs for Regeneration percentage (R), detected in this study on chromosomes 1 (SpRg-1), 4 (SpRg-4a), 7 (SpRg-7) and 8 (SlRg-8). Results from the Interval Mapping (IM) and restricted Multiple QTL Mapping (rMQM) approaches. On the left, projections as black bars (IM) and grey bars (rMQM) indicate the range of SpRg-1, SpRg-4a, SpRg-7 and SlRg-8 QTLs for R. The vertical dotted line indicates the 95% significant threshold value for declaring a QTL (R LOD threshold = 2.7). Map position (cM) and distances are based on the genetic linkage map developed in this study.
Genetic location and LOD score profile of the BC1-QTLs for Productivity Rate (PR), detected in this study on chromosomes 1 (SpRg-1), 3 (SpRg-3), 4 (SpRg-4b) and 7 (SpRg-7). Results from the Interval Mapping (IM) and restricted Multiple QTL Mapping (rMQM) approaches. On the left, projections as black bars (IM) and grey bars (rMQM) indicate the range of SpRg-1, SpRg-3, SpRg-4b and SpRg-7 for PR. The vertical dotted line indicates the 95% significant threshold value for declaring a QTL (PR LOD threshold = 2.8). Horizontal dotted lines indicate the position of the acid invertase gene (invpenn) marker included in the chromosome 3 QTL range. Map position (cM) and distances are based on the genetic linkage map developed in this study.
Polymorphic acid invertase gene marker (invpenn). Amplified bands separated using the multicapillary electrophoresis QIAxcel System. Lane 1: S. lycopersicum L. (Anl27), band size (~162bp). Lane 2: S. pennellii PE-47, band size (~173bp). Lane 3: F1 Hybrid S. lycopersicum L. (Anl27) × S. pennellii PE-47, both bands (~162bp-~173bp). Lane 4: negative control.
Markers used for genotyping the F2 and BC1 population. SSR, COS, COSII, CAP markers used for genotyping the F2 and BC1 population.