Abstract
The aim of the present study was to examine spinal processing of cardiac and somatic nociceptive input in rats with STZ-induced diabetes.
Type 1 diabetes was induced with streptozotocin (50 mg/kg) in 14 male Sprague-Dawley rats and citrate buffer was injected in 14 control rats. After 4–11 weeks, the rats were anesthetized with pentobarbital, ventilated and paralyzed. A laminectomy enabled extracellular recording of T3 spinal cord neuronal activity. Intrapericardial administration of a mixture of algogenic chemicals (bradykinin, serotonin, prostaglandin E2 (all at 10−5 M), and adenosine (10−3 M)) was applied to activate nociceptors of cardiac afferent nerve endings. Furthermore, somatic receptive properties were examined by applying innocuous (brush and light pressure) and noxious (pinch) cutaneous mechanical stimuli.
Diabetes-induced increases in spontaneous activity were observed in subsets of neurons exhibiting long-lasting excitatory responses to administration of the algogenic mixture.
Algogenic chemicals altered activity of a larger proportion of neurons from diabetic animals (73/111) than control animals (55/115, P < 0.05). Some subtypes of neurons exhibiting long-lasting excitatory responses, elicited prolonged duration and others, had a shortened latency. Some neurons exhibiting short-lasting excitatory responses in diabetic animals elicited a shorter latency and some a decreased excitatory change. The size of the somatic receptive field was increased for cardiosomatic neurons from diabetic animals. Cutaneous somatic mechanical stimulation caused spinal neurons to respond with a mixture of hyper- and hypoexcitability.
In conclusion, diabetes induced changes in the spinal processing of cardiac input and these might contribute to cardiovascular autonomic neuropathy in patients with diabetes.
Keywords: Diabetes, streptozotocin, thoracic spinal cord, spinal neurons, extracellular recording, angina, autonomic neuropathy
1. INTRODUCTION
Diabetic neuropathy is a common complication of diabetes mellitus that can affect autonomic, sensory, as well as motor nerves and significantly impact quality of life of these patients (Thomas and Tomlinson, 1993). Cardiovascular autonomic neuropathy (CAN) is found in patients with diabetes at a prevalence of 7–90 % (Vinik et al., 2003) and involves cardiomotor and vasomotor efferents as well as cardiac sensory afferent fibers (Ahluwalia et al., 1995; Koistinen et al., 1996; Thomas and Tomlinson, 1993; Vinik et al., 2003). CAN in diabetic patients is associated with increased risk of developing cardiovascular disease and silent myocardial ischemia, a decreased heart rate variation, orthostatic hypotension, resting tachycardia, exercise intolerance, and these patients furthermore have a higher overall mortality (Johnstone and Kinzfogl, 2005; Thomas and Tomlinson, 1993; Vinik et al., 2003; Vinik and Ziegler, 2007). Silent myocardial ischemia is found more commonly in diabetic patients with coronary artery disease compared to the corresponding non-diabetic patients (Ditchburn et al., 2001; Schultz, 2003; Vinik et al., 2003), and a few studies show that this occurs even in the absence of autonomic neuropathy (Ahluwalia et al., 1995), suggesting that autonomic neuropathy is not the only factor affecting the cardiac nociception during diabetes. It seems likely that altered pain perception might also be related to a sensory disorder or alterations of the central integration of signals from the heart. Despite the huge impact altered pain perception potentially can have on the morbidity and mortality in diabetic patients, as yet, very little is known about cardiac sensory function and its central processing during diabetes. Just a few studies have shown that cardiac vagal afferent function and cardiosomatic reflexes are impaired in streptozotocin (STZ)-induced diabetic rats when compared to control animals (Gouty et al., 2001; Liu et al., 2011; Schultz, 2001).
A characteristic of neuropathy is decreased nerve conduction velocity, in humans as well as in animals, and has been shown to develop both centrally (Suzuki et al., 2000) and peripherally (Biessels et al., 1996; Kalichman et al., 1998; Nemeth et al., 1999; Nemeth et al., 2001; Selvarajah et al., 2006; Walker et al., 1999). In rats, nerve conduction velocity is decreased 4 weeks after the onset of type 1 diabetes (Biessels et al., 1996; Kalichman et al., 1998; Nemeth et al., 2001) and decreases with duration of diabetes (Nemeth et al., 1999). Animal experiments concerning sensory nerves during diabetes typically focus on painful diabetic neuropathy, a sensory neuropathy, which is a common cause of morbidity in diabetic patients. Behavioral studies using type 1 diabetic animals have shown the presence of this form of neuropathy, with a consensus on the presence of mechanical hyperalgesia and tactile allodynia in the paw, but conflicting results as to whether the thermal threshold is altered (Chen and Pan, 2002; Khan et al., 2002; Morgado and Tavares, 2007; Pertovaara et al., 2001). Studies on primary afferent somatosensory fibers show that the mechanical threshold is decreased for Aδ fibers and their responsiveness augmented during diabetes; whereas for C-fibers, the mechanical threshold is unchanged and the responsiveness ranges from no alteration to hyperresponsiveness (Ahlgren et al., 1992; Chen and Levine, 2001; Chen and Levine, 2003; Khan et al., 2002).
Changes have also been found in the spinal cord during diabetes. Selvarajah et al. found a decreased cross-sectional area of the 2nd and 3rd cervical segments of the spinal cord in diabetic patients with subclinical and clinical diabetic neuropathy (Selvarajah et al., 2006). Furthermore, chemogenic hypoalgesia was associated with a decrease in Fos expression in the dorsal horn of the spinal cord in STZ-diabetic C57BL/6 mice (Johnson et al., 2007), whereas mechanical hyperalgesia was found to be associated with an increase in Fos expression in STZ-diabetic rats (Morgado and Tavares, 2007). Furthermore, spinothalamic tract (STT) cells and spinal neurons from STZ-diabetic animals appeared to be hypersensitive following innocuous and noxious mechanical stimulation, and have higher neuronal spontaneous activity and enlarged somatic receptive fields (Chen and Pan, 2002; Pertovaara et al., 2001). However, the above studies are all based on somatosensory input. A recent electrophysiological study from our laboratory has investigated spinal neurons receiving visceral esophageal sensory afferent input in diabetic rats (Qin et al., 2009). In that study, altered activity of upper thoracic neurons receiving esophageal input was observed in STZ-diabetic rats. Tanabe et al. (2005) found changes in the intrinsic inhibitory system in the spinal cord that contributed to an increased spinal motor output in STZ-diabetic rats. Altered excitability of spinal neurons might be ascribed to functional changes of inhibitory descending pathways (Chen and Pan, 2002; Kimura et al., 2005; Tanabe et al., 2005), alterations in the intrinsic inhibitory system (Tanabe et al., 2005), sensitization of afferent fibers (Ahlgren et al., 1992; Chen and Levine, 2001; Chen and Levine, 2003; Khan et al., 2002), wind-up of C-fibers ((Kimura et al., 2005)), as well as receptor and neuropeptide changes (Cloutier and Couture, 2000; Kamei et al., 1990).
To the best of our knowledge, no prior studies have investigated the activity of upper thoracic spinal neurons receiving cardiac input in diabetic animals to determine if spinal neuronal processing is affected by this disease. Previously, our laboratory has performed electrophysiological investigations on upper thoracic T3–T4 spinal neurons in healthy rats after cutaneous somatic and chemical cardiac stimulation (Qin et al., 2002; Qin et al., 2003). Intrapericardial administration of inflammatory substances, known to be released during myocardial ischemia, changed activity of spinal neurons in the dorsal horn through activation of spinal cardiac sensory afferents in the heart. The majority of responsive neurons were excited and the remaining exhibited inhibitory or biphasic responses (Qin et al., 2003). Spinal neurons receiving cardiac input are modulated by descending – primarily inhibitory – input, interneurons and convergent somatic or visceral input (Qin et al., 2002; Qin et al., 2004b). The spinal neurons recorded are possibly projecting neurons, interneurons, and propriospinal neurons and therefore could be involved in intraspinal integration of nociceptive input, affecting both perception of anginal pain and autonomic cardiocardiac reflexes (Hobbs et al., 1992; Qin et al., 2001; Qin et al., 2002; Schultz, 2003). The purpose of the present study was to characterize the activity of upper thoracic (T3) spinal neurons after noxious chemical stimulation of cardiac afferents and cutaneous somatic mechanical stimulation in rats with STZ-induced diabetes (type 1 diabetes). A preliminary report of parts of this work has been published in abstract form (Foreman et al., 2007).
2. MATERIALS AND METHODS
2.1. Induction of diabetes
A total of 28 Sprague Dawley rats (Harlan Inc., USA) were used in the present study. Type 1 diabetes was induced with a single injection of streptozotocin (STZ, 50 mg/kg in 0.1 M citrate buffer pH 4.5, i.p.in 14 rats at the age of 7 weeks). Citrate buffer was injected in the eight rats as an age- and weight-matched control group. An additional six animals were also used as controls. Two to three days later, the presence of diabetes was assessed by determining that blood glucose concentration was ≥ 20 mM in blood samples obtained from the tail using a strip-operated blood glucose sensor (OneTouch® Ultra®, LifeScan). Rats were weighed three times a week and blood glucose concentrations were measured once a week. All experimental protocols were approved by the Institutional Animal Care and Use Committee at The University of Oklahoma Health Sciences Center.
2.2 Surgical procedure
The surgical procedure has previously been described by Qin et al. (Qin et al., 2003). In short, 4–12 weeks after injection of STZ, rats were anaesthetized with pentobarbital (60 mg/kg, i.p.). Sustained anesthesia was accomplished by continuous infusion of pentobarbital (15–25 mg/kg/h, i.v.) through a catheter in the left jugular vein. Mean arterial blood pressure was measured through a catheter in the right carotid artery. Artificial ventilation with a constant-volume pump (55–60 strokes/min, 3.0–5.0 ml stroke volume) was provided after tracheotomy. During spinal neuronal recordings, the animals were paralyzed with pancuronium bromide (0.4 mg/kg, i.v.) and supplementary doses were used to maintain muscle relaxation (0.2 mg/kg/hr, i.p.). A thermostatically controlled heating pad and overhead infrared lamps kept rectal temperature between 36.7°C and 37.3°C throughout the experiment.
For chemical activation of the sensory nerve endings in the heart, a silicone tubing (0.020 cm i.d., 0.037 cm o.d., 14–16 cm in length) was passed through the thoracic thymus gland and inserted into the pericardial sac over the left ventricle.
To record extracellular action potentials rats were placed in the prone position and mounted in a stereotaxic headholder and stabilized with vertebral clamps at T2 and T6–T8. A laminectomy was performed to expose the T3 spinal segment. Then, dura mater and arachnoid were cut and the exposed spinal cord was covered with warm agar (3–4 % in saline) for a stabile recording. Extracellular potentials of single T3 spinal neurons were recorded with a carbon-filament glass microelectrode. The area of searching for neurons was approximately 0.5–2.0 mm lateral from midline of the spinal cord. Superficial neurons were recorded within 0.30 mm and deeper neurons within 0.31–1.20 mm from the dorsal surface of the spinal cord, corresponding, in rats, to lamina I–III and IV–VII + X, respectively (Molander et al., 1989; Ness and Gebhart, 1989).
2.3 Experimental protocol
Sensory nerve endings were activated with a mixture of algogenic chemicals including bradykinin (10−5 M), serotonin (10−5 M), prostaglandin E2 (10−5 M) and adenosine (10−3 M), which are released during myocardial ischemia and previously shown to stimulate spinal neurons after intrapericardial administration (Pan and Longhurst, 1995; Qin et al., 2001; Qin et al., 2003). All drugs were dissolved in saline. Algogenic chemicals (0.2 ml) was administered through the silicone tubing into the pericardial sac (intrapericardial algogenic chemicals, IAC), withdrawn after 60 s, and after that the pericardial sac was rinsed 2–3 times with 0.2 ml saline. Spinal neuronal activity before, during and after intrapericardial injections of chemicals was observed. At least 20 min elapsed between each injection of IAC.
Spinal neurons were also examined for responses to innocuous (brush and light pressure) and noxious (pinch) mechanical stimuli of somatic receptive fields. Neurons were divided into low-threshold (LT), wide dynamic range (WDR) and high-threshold (HT) neurons according to their somatic field properties. Low-threshold (LT) neurons were activated by applying brush or light pressure to their cutaneous somatic field and activity was not changed to a greater extent during noxious pinch. Activity of wide dynamic range (WDR) neurons changed during brushing or light pressure of somatic fields, and even more during noxious pinch. Activity of high-threshold (HT) neurons changed only with noxious pinching of the somatic field with a blunt forceps. Mechanical stimulation of somatic fields was applied for 10 s, followed by a rest period of at least 60 s to allow complete recovery of the neuronal activity. For neurons receiving somatic input, the sizes of the somatic field were categorized as small (ipsilateral, long axis ≤ 4 cm), medium (ipsilateral, long axis > 4 cm) and large (bilateral fields) (Qin et al., 2009).
2.4 Data analysis
Cell activity was recorded online with Spike 2 data acquisition system (CED, Cambridge, UK). Excitatory and inhibitory changes in neuronal activity during a chemical stimulus were calculated by subtracting the mean of 10 s of control activity (i.e. the spontaneous activity) from the mean of 10 s of the maximal responses. For neurons with somatic inputs, background activity as well as the response was measured for periods of 5 s. Increases or decreases in firing rate of ≥ 20 % were considered a neuronal response. Duration of response was measured from the onset of a response to the point of recovery to control activity. Based on the recovery time (time from offset of stimulation to point of recovery to control activity), the neurons responding to IAC were divided into short-lasting (SL, recovery time ≤ 50 s) or long-lasting (LL, recovery time > 50 s) neurons. Latency of responses was measured as the time from onset of stimulation to onset of response. Cardioreceptive (“cardiac”) neurons responded to IAC but not to somatic mechanical stimuli, somatoreceptive (“somatic”) neurons responded to somatic mechanical stimuli but not to IAC, and cardiosomatic neurons responded to IAC as well as mechanical stimuli.
For neurons responding to IAC, an electrolytic lesion (50 μA DC, 20 s) was made after completing the protocol. At the end of each experimental day, the animal was euthanized with an overdose of i.v. pentobarbital (> 200 mg/kg). The thoracic spinal cord was removed and placed in 10 % buffered formalin solution. After ≥ 3 days, frozen sections (55–60 μm) were made and the location of lesion sites in the laminae were identified using the cytoarchitectonic sketch of the spinal cord (Molander et al., 1989).
Statistics
Quantitative results from diabetic and control animals were compared using either student’s t-test (or the corresponding non-parametric Mann-Whitney Rank Sum test), or two-way ANOVA followed by Tukey test. P < 0.05 was used as a level of significance. Data are presented as mean ± SEM. Populations of response patterns, somatic field size and somatic field characteristics were compared for diabetic and control animals using Chi-square.
3. RESULTS
3.1 The animals
All rats injected with STZ developed diabetes within two days as assessed by a blood glucose value above 20 mM. Throughout the examination period, diabetic rats maintained high values of blood glucose compared to those measured for control animals. On the day of experiment the blood glucose value was 26.2 ± 3.7 mM (n = 14) for diabetic animals and 5.9 ± 0.7 mM (n = 14) for control animals. The diabetic animals experienced only a slight weight gain throughout the experimental period with a body weight of 230 ± 7.6 g prior to STZ injection and 260 ± 36.2 g (n = 14) on the day of experiment compared to an increment from 231 ± 9.4 to 384 ± 41.8 g (n = 8) in the same period for age-matched control animals. In addition, 6 control animals were weighed on the day of experiment with a mean body weight of 442 ± 16.0 g
3.2 Cardiac and somatic receptive properties
Overall, neurons from diabetic rats exhibited an altered distribution of receptive properties (P < 0.05, Figure 1). Administration of IAC changed the activity (i.e. cardiac and cardiosomatic neurons) of 73/111 tested neurons from diabetic animals compared to only 55/115 tested neurons from control animals (66% and 48 %, respectively, P < 0.05, Figure 1). Somatic receptive fields were found for 91/111 (82 %) diabetic neurons and 98/115 (85 %) control neurons (Figure 1). 57/111 (51 %) of the tested neurons from diabetic animals received convergent cardiosomatic input, and this was seen in 47/115 (41 %) of the tested neurons from control animals. Among neurons receiving cardiac input, cardiosomatic convergence was found in 78 % (57/73) of neurons from diabetic animals compared to 85 % (47/55) of neurons from control animals. The diabetes-induced alteration in receptive properties was found throughout the dorsal horn, and thus, a tendency towards an increased proportion of cardiac and cardiosomatic neurons (62 vs. 44 % in superficial (P > 0.05), and 69 vs. 51 % in deeper lamina (P > 0.05) and a decreased proportion of somatic neurons (38 vs. 50 % in superficial (P > 0.05), and 25 vs. 40 % in deeper lamina (P > 0.05)) was found for diabetic neurons both in superficial as well as in deeper lamina.
Figure 1.
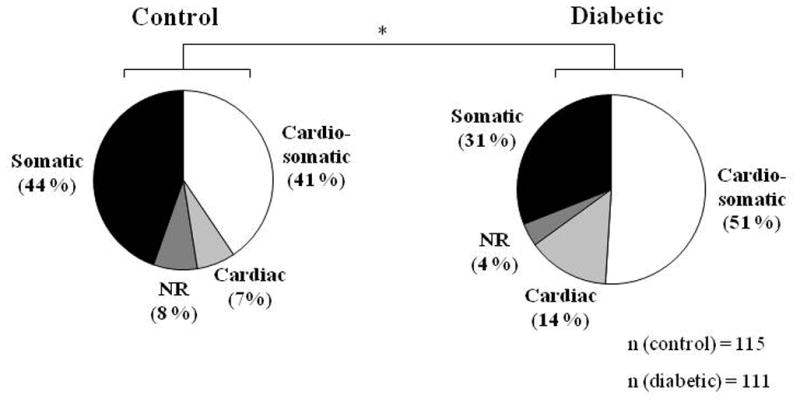
Response pattern of all neurons tested with intrapericardial algogenic chemicals and mechanical stimuli. *P < 0.05. Chi-square test performed on the distribution in the four categories. NR; No response.
3.3 Response to IAC
Neurons from diabetic and control animals exhibited similar response patterns that included excitatory, inhibitory and biphasic (excitatory-inhibitory/inhibitory-excitatory) responses. Representative traces of response patterns for single neurons exhibiting short-lasting excitatory (SL-E), long-lasting excitatory (LL-E) and inhibitory responses are shown in Figure 2. An excitatory response was exhibited by 75 % (55/73) of IAC-responding neurons from diabetic animals compared to 80 % (44/55) from control animals. Only 21 % (15/73) of the IAC-responding neurons from diabetic animals exhibited an inhibitory response compared to 13 % (7/55) of neurons from control animals. Because of the low number of inhibitory (I), excitatory-inhibitory (E-I) and inhibitory-excitatory (I-E) neurons, these were excluded from further analyses.
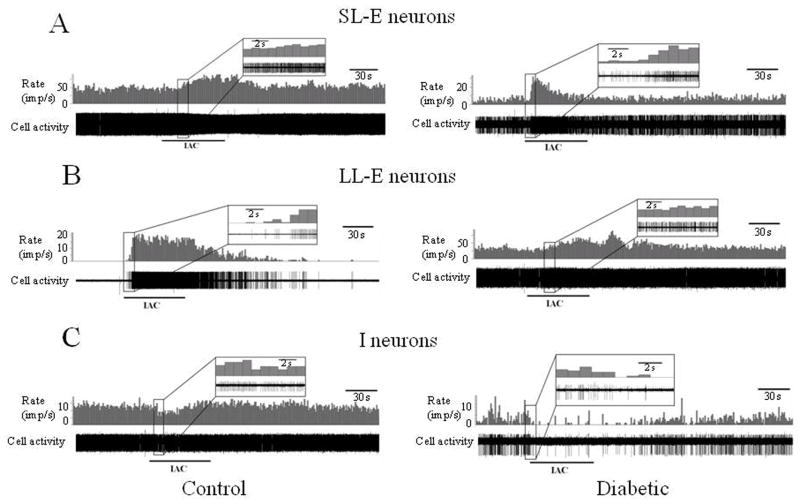
Figure 2.
Representative traces of response patterns of neurons to intrapericardial algogenic chemicals (IAC). A. Short-lasting excitatory (SL-E) responses in control (left panel) and diabetic (right panel) rats. B. Long-lasting excitatory (LL-E) responses in control (left panel) and diabetic (right panel) rats. C. Inhibitory (I) responses in control (left panel) and diabetic (right panel) rats. For each neuron, the trace of individual action potentials as well as a histogram displaying the activity (impulses/s) is depicted. An expanded view of the trace and histogram showing the initial part of the response is included for all neurons.
A comparison of cells recorded from different laminae showed that 62 % (31/50) of the superficial neurons and 69 % (42/61) of the deeper neurons received cardiac input in the diabetic animals, whereas 44 % (22/50) of superficial neurons and 51 % (33/65) of deeper neurons received cardiac input in the control animals. Figure 3 shows histologically identified lesion sites of 26 neurons from control animals and 28 neurons from diabetic animals responding to IAC in the T3 spinal cord segment as well as summaries of the response patterns for IAC-responding neurons in the superficial and deeper laminae, respectively. The average duration of all excitatory responses to IAC was significantly longer in diabetic rats than in control rats (170.7 ± 16.5 s vs. 126.9 ± 12.0 s, P < 0.05). Further analysis showed that this prolonged duration in neurons from diabetic animals was caused by superficial neurons exhibiting LL-E responses (264.0 ± 32.3 s vs. 167.1 ± 22.5 s, P < 0.05, Table 1). Furthermore, the spontaneous activity observed for superficial LL-E neurons was higher in diabetic animals than the corresponding neurons from control animals (9.1 ± 1.4 impulses (imp)/s vs. 4.0 ± 1.6 imp/s, P < 0.05). No statistical differences were found in latency or excitatory changes between diabetic and control neurons exhibiting LL-E responses (Table 1). In contrast, the latency was decreased for the diabetic superficial SL-E neurons compared to the corresponding control neurons. No other differences were found for SL-E neurons (Table 2).
Figure 3.
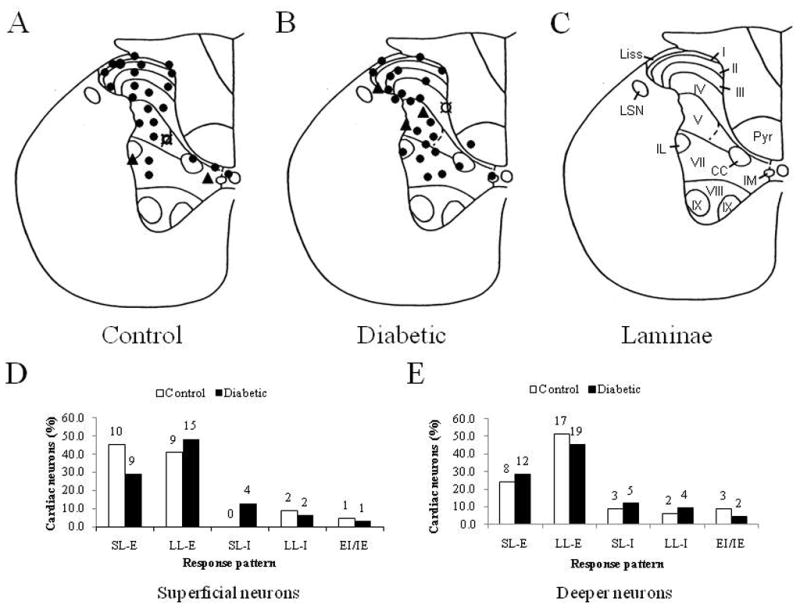
Lesion sites and response pattern of T3 spinal neurons excited (●), inhibited (▲), or excited-inhibited/inhibited-excited (¤) by intrapericardial algogenic chemicals (IAC). A. Lesion sites for neurons from control rats and B. from diabetic rats. C. Laminae of the T3 spinal cord segment. D, E. Response patterns for neurons receiving cardiac input located in the superficial (D) and the deeper (E) laminae. The digits above each column represent the number of neurons accounting for that particular column. SL-E; Short-lasting excitatory, LL; Long-lasting, I; Inhibitory. E-I; Excitatory and inhibitory, I-E; Inhibitory and excitatory.
Table 1.
Comparison of long-lasting excitatory responses of superficial and deeper thoracic spinal neurons receiving noxious cardiac input in diabetic and control animals.
| Groups | Neuron classes | n | Spontaneous activity (imp/s | Latency (s) | Excitatory change (imp/s) | Duration (s) |
|---|---|---|---|---|---|---|
| Control | Su | 9 | 4.0 ± 1.6 | 9.7 ± 2.7 | 23.7 ± 4.8 | 167.1 ± 22.5 |
| De | 17 | 9.4 ± 1.7 | 7.6 ± 1.7 | 18.5 ± 2.0 | 183.8 ± 14.5 | |
| Diabetic | Su | 15 | 9.1 ± 1.4* | 5.8 ± 0.8 | 29.0 ± 6.5 | 264.0 ± 32.3* |
| De | 19 | 11.0 ± 2.5 | 8.3 ± 1.9 | 25.1 ± 3.9 | 221.8 ± 18.8 |
Results are shown as mean ± SEM.
P < 0.05 compared to corresponding activity of diabetic group. Imp; impulse
Table 2.
Comparison of short-lasting excitatory responses of superficial and deeper thoracic spinal neurons receiving noxious cardiac input in diabetic and control animals.
| Groups | Neuron classes | n | Spontaneous activity (imp/s | Latency (s) | Excitatory change (imp/s) | Duration (s) |
|---|---|---|---|---|---|---|
| Control | Su | 10 | 12.7 ± 5.1 | 14.7 ± 3.3 | 23.8 ± 7.5 | 53.1 ± 7.6 |
| De | 8 | 7.1 ± 1.9 | 8.8 ± 2.0 | 24.6 ± 11.4 | 52.9 ± 10.7 | |
| Diabetic | Su | 9 | 6.0 ± 3.2 | 5.1 ± 1.3* | 13.8 ± 3.5 | 70.8 ± 8.5 |
| De | 12 | 9.3 ± 2.5 | 10.2 ± 2.5 | 11.3 ± 1.5 | 48.2 ± 7.8 |
Results are shown as mean ± SEM. P < 0.05 compared to corresponding activity of diabetic group. Imp; impulse
Neurons exhibiting SL-E and LL-E responses were furthermore categorized according to the levels of spontaneous activity of 0–5 imp/s or 5–30 imp/s (Tables 3 and 4). At both levels of spontaneous activity, diabetic neurons with LL-E responses exhibited a higher spontaneous activity compared to control neurons (Table 3). Duration of response was increased only for diabetic LL-E neurons exhibiting lower spontaneous activity (P < 0.05) and the latency was decreased for these neurons (P < 0.05). Diabetic SL-E neurons with high spontaneous activity had a significantly lower excitatory change after administration of IAC than the corresponding control neurons (P < 0.05). Furthermore, the excitatory change of control SL-E neurons was larger at higher spontaneous activity compared to lower spontaneous activity (P < 0.01) (Table 4). This difference in excitatory change based on the level of spontaneous activity was not observed in diabetic animals.
Table 3.
Comparison of long-lasting excitatory responses of thoracic spinal neurons receiving noxious cardiac input and exhibiting low or high spontaneous activity in diabetic and control animals.
| Level of SA (imp/s) | Animal | n | SA (imp/s) | Latency (s) | Excitatory change (imp/s) | Duration (s) |
|---|---|---|---|---|---|---|
| 0–5 | Control | 9 | 0.4 ± 0.1 | 10.9 ± 2.4 | 22.6 ± 4.7 | 161.5 ± 21.5 |
| Diabetic | 12 | 1.7 ± 0.5* | 5.8 ± 0.9* | 20.8 ± 3.5 | 268.4 ± 35.8* | |
| 5–30 | Control | 17 | 11.3 ± 1.3# | 7.0 ± 1.7 | 19.1 ± 2.1 | 186.8 ± 14.6 |
| Diabetic | 22 | 14.8 ± 1.6*,# | 8.0 ± 1.7 | 30.1 ± 5.1 | 225.2 ± 19.3 |
Results are shown as mean ± SEM.
P < 0.05 compared to corresponding control neurons,
P < 0.001 compared to corresponding neurons with SA 0–5 imp/s Imp; impulse
Table 4.
Comparison of short-lasting excitatory responses of thoracic spinal neurons receiving noxious cardiac input and exhibiting low or high spontaneous activity in diabetic and control animals.
| Level of SA (imp/s) | Animal | n | SA (imp/s) | Latency (s) | Excitatory change (imp/s) | Duration (s) |
|---|---|---|---|---|---|---|
| 0–5 | Control | 7 | 0.9 ± 0.5 | 15.0 ± 2.2 | 8.5 ± 1.7 | 47.1 ± 8.4 |
| Diabetic | 12 | 1.5 ± 0.4 | 7.8 ± 2.5 | 11.5 ± 2.7 | 58.1 ± 8.4 | |
| 5–30 | Control | 11 | 16.1 ± 3.9# | 10.2 ± 3.1 | 34.2 ± 9.3† | 56.8 ± 8.6 |
| Diabetic | 9 | 16.4 ± 2.5# | 8.2 ± 1.9 | 13.4 ± 1.7* | 57.6 ± 9.4 |
Results are shown as mean ± SEM.
P < 0.05 compared to corresponding control neurons,
P < 0.01 compared to control neurons with SA 0–5 imp/s,
P < 0.001 compared to corresponding neurons with SA 0–5 imp/s. SA; spontaneous activity. Imp; impulse
3.4 Response to somatic stimuli
Qualitative analysis showed that the distribution of somatic field properties of LT, WDR, and HT were not altered by diabetes (Figure 4). Spontaneous activity of cardiosomatic WDR and HT neurons was higher than of the corresponding somatic neurons, and this was seen for both control and diabetic neurons (P < 0.05, Table 5). The response of WDR neurons to brush was higher for diabetic somatic neurons (34.8 ± 11.6 imp/s) than for diabetic cardiosomatic (9.3 ± 1.5 imp/s, P < 0.01) and also higher than for somatic neurons from control animals (15.4 ± 3.8 imp/s, P < 0.05) (Table 5). Furthermore, when applying noxious pinch to the WDR neurons, diabetic somatic neurons responded with a greater excitatory change than the diabetic cardiosomatic neurons (54.4 ± 14.5 vs. 26.6 ± 3.4 imp/s, P < 0.05). The excitatory change of the diabetic cardiosomatic WDR neurons was also significantly reduced in comparison to the control neurons (26.6 ± 3.4 vs. 43.2 ± 6.3 imp/s, P < 0.05). The excitatory change of HT neurons caused by noxious pinch was decreased in diabetic somatic neurons (12.3 ± 1.5 imp/s) compared to both the diabetic cardiosomatic neurons (24.4 ± 2.8 imp/s, P < 0.05) and the control somatic neurons (25.3 ± 5.0 imp/s, P < 0.05). No differences were found between neurons from diabetic and control animals when light pressure was applied to the somatic receptive fields (data not shown).
Figure 4.

Characteristics of somatic receptive fields of spinal neurons from diabetic and control animals. A. A spinal neuron with low threshold (LT) has a response to brush (Br) and a smaller (or comparative) response to pinch (Pi). B. A wide dynamic range (WDR) neuron responds to brush and to a higher degree to pinch. C. A high-threshold (HT) neurons responds to pinch but not to brush. D, E. Somatic field properties for diabetic and control neurons responding to somatic (D) and cardiosomatic stimuli (E). The digits above each column represent the number of neurons accounting for that particular column.
Table 5.
Comparison of the excitatory change in activity of excitatory neurons by stimulation with brush and/or pinch.

|
Results are shown as mean ± S.E.M.
“ P< 0.05,
P < 0.01, and
P < 0.001 compared to the corresponding somatic neurons.
P < 0.05 compared to corresponding control neurons,
P < 0.05 and
P < 0.01 compared to somatic diabetic neurons. n; number of neurons, imp; impulse, LT; low-threshold, WDR; wide-dynamic range, HT; high-threshold.
Only few LT neurons were found among the tested neurons, leading to less statistical strength and quantitative analysis found no statistically significant changes in diabetic animals.
3.5 Somatic field size
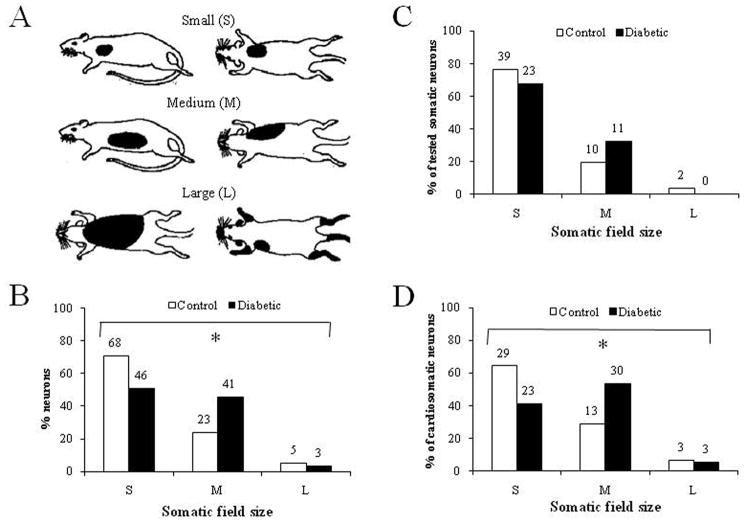
Somatic field sizes were categorized as small (S), medium (M) or large (L) in accordance to earlier determined descriptions (Qin et al., 2003), and examples are shown in Figure 5. Overall, somatic field size was found to be larger in diabetic animals than control animals (P < 0.01). Analyses of subgroups revealed that the somatic field size was increased for cardiosomatic neurons from diabetic animals compared to control animals (P < 0.05), whereas this difference in size was not found for purely somatic neurons (P > 0.05) (Figure 5). When cardiosomatic neurons were categorized according to whether they elicited SL-E or LL-E responses after stimulation with IAC, it was found that superficial, but not deeper LL-E neurons, had an enlarged somatic field size (P < 0.05, n = 8 (control), n = 11 (diabetes), data not shown). Moreover, the somatic field size was enlarged for LL-E neurons at low, but not high levels of spontaneous activity (P < 0.01, n = 7 (control), n = 9 (diabetes), data not shown). The somatic field size was unaltered for SL-E neurons (P > 0.05, data not shown). No difference was seen for the deeper neurons. Finally, it was investigated whether diabetes affected the somatic field size differently depending on the somatic receptive properties of the neuron. LT neurons from diabetic animals had significantly larger somatic field sizes than control animals (p < 0.05, data not shown); whereas, no differences were found among WDR and HT neurons (data not shown).
Figure 5.
Somatic field sizes of spinal neurons from diabetic and control animals. A. Areas of somatic receptive fields for small- (S), medium- (M) and large-sized (L) cells. B. Distribution of somatic field sizes for all neurons receiving somatic input, i.e. somatic and cardiosomatic neurons. * P < 0.05 compared to control neurons. C, D. Distribution of somatic field sizes for somatic (C) and cardiosomatic (D) neurons. * P < 0.05 compared to control neurons. The digits above each column represent the number of neurons accounting for that particular column.
4. DISCUSSION
To the best of our knowledge, this is the first study to investigate the activity of upper thoracic spinal neurons receiving cardiac input in diabetic animals. The major findings of the study were: 1) Significantly more spinal neurons from diabetic animals received noxious cardiac input (66 vs. 48 %); 2) Diabetes caused an increase in spontaneous activity in subsets of neurons exhibiting LL-E responses after stimulation with IAC; 3) Some neurons exhibiting LL-E responses elicited a faster onset of response and some a prolonged duration after stimulation with IAC; 4) Superficial neurons exhibiting SL-E responses in diabetic animals had a faster onset of response; whereas, SL-E neurons from diabetic animals with a high level of spontaneous activity had a decreased excitatory change following stimulation with IAC; 5) The size of the somatic receptive field was increased for cardiosomatic neurons from diabetic animals, more specifically for those exhibiting LL-E responses following stimulation with IAC, and 6) Diabetic cardiosomatic WDR neurons stimulated with pinch showed hyposensitivity as did diabetic somatic HT neurons; however, diabetic somatic WDR neurons in contrast showed hypersensitivity when stimulated with innocuous brush. Overall, the changes seen in the upper thoracic spinal neurons from STZ-diabetic rats suggest that diabetes affects subgroups of spinal neurons in a differentiated manner, causing hypersensitivity in some neurons and hyposensitivity in others.
4.1 Spontaneous activity
In the present study, an increased spontaneous activity was found in diabetic animals only in subsets of neurons exhibiting LL-E response to IAC but not those exhibiting an SL-E response. The observed increase in spontaneous activity is in accordance with findings from electrophysiological and c-fos studies; however, these studies have in addition also found increases in deeper laminae than this study and in somatic neurons, for which no changes in spontaneous activity was seen in the present study (Chen and Pan, 2002; Morgado and Tavares, 2007; Pertovaara et al., 2001) The variation in results between the studies might be explained by differences in receptive properties, and segmental and laminal orientation of the spinal neurons.
A possible explanation for the increase in spontaneous activity seen in the present study for neurons exhibiting LL-E responses could be ectopic discharges from the primary afferents providing input to these neurons, as ectopic discharges has previously been found in diabetic animals (Burchiel et al., 1985; Zochodne, 1996). Furthermore, upper thoracic spinal cord neurons responsive to IAC are subject to tonic descending modulation (Qin et al., 2003; Qin et al., 2004b), and it is possible that diabetes may have caused an altered balance in the facilitatory and inhibitory descending pathways, that might also contribute to the increased spontaneous activity. Rewiring of synapses in the dorsal horn might also contribute as spinal reorganization has been found in the dorsal horn of rats after peripheral nerve damage (Woolf et al., 1992).
4.2 Response to IAC
In the present study, 66 % of diabetic neurons and 48% of control neurons responded to IAC. The proportion of responsive control neurons is comparable to a study conducted by Qin et al. where 42 % of spinal neurons from non-diabetic rats responded to IAC (Qin et al., 2003). The shift in receptive properties of neurons from the diabetic animals seen in the present study (more cardiac/cardiosomatic neurons, but fewer somatic and non-responsive neurons) might be explained by diabetes-induced changes in the primary afferents. Silent C-fiber afferents insensitive to mechanical stimuli innervate the heart and can be activated during myocardial ischemia (Pan and Chen, 2002). A decreased proportion of mechanically and thermally silent C-fibers have been found in the saphenous nerve of STZ-induced diabetic rats, suggesting that a larger number of fibers are transmitting nociceptive information in diabetic rats (Ahlgren et al., 1992). In the present study, the larger fraction of spinal neurons from diabetic animals receiving nociceptive input after stimulation with IAC is therefore possibly caused by activation of a larger fraction of normally silent cardiac afferent fibers. This suggested an increased sensitivity towards IAC and might induce a change in spinal, intraspinal and/or supraspinal processing which could help explain the decreased latency found in the present study for two subsets of spinal neurons following IAC administration. These changes could be of importance to the manner in which an individual responds to myocardial ischemia, e.g. autonomic reflexes and pain perception.
LL-E neurons in the superficial laminae as well as those exhibiting low levels of spontaneous activity were found to have enlarged somatic receptive fields in diabetic animals, and following IAC administration, their responses were of prolonged duration compared to control animals. LL-E neurons with a low level of spontaneous activity furthermore had a faster onset of response. All changes, including the augmented spontaneous activity suggest an increased activity and responsiveness of these subsets of neurons. Neurons exhibiting SL-E responses in the superficial laminae had a faster onset of response in the diabetic animals compared to control animals. Furthermore, neurons with SL-E responses exhibiting high spontaneous activity showed an attenuated excitatory change after stimulation with IAC, the decreased excitatory change suggesting hyposensitivity in this subset of spinal cord neurons. The present results shows that the diabetes-induced neuronal changes are differentiated in the spinal cord and this view is supported by Qin et al. (2009) who found both hypo- and hypersensitivity in spinal neurons receiving esophageal mechanical input in STZ-diabetic rats (Qin et al., 2009). The hypersensitivity found in the present study for neurons with LL-E responses combined with the hyposensitivity of neurons with SL-E responses is analogous to findings by Ness and Gebhart. They have characterized two groups of spinal neurons from L6-S1 segments exhibiting excitatory responses following colorectal distension in rats with experimentally induced inflamed colon. “Sustained” neurons show afterdischarges for > 4 s whereas “abrupt” neurons show a quick return to baseline after removing stimulus and differences in responses to non-segmental stimulation are also apparent (Ness and Gebhart, 1987). Ness and Gebhart found hypersensitivity in the sustained and hyposensitivity in the abrupt neurons following colorectal distension (Ness and Gebhart, 2001). Neurons with LL-E responses have been suggested to play an important role in the transmission of visceral pain (Ness and Gebhart, 2000; Ness and Gebhart, 2001; Qin et al., 2001) but SL-E as well as LL-E neurons might also be involved in cardiocardiac (or other cardiovisceral) and cardiosomatic reflexes which would be of great importance during myocardial ischemia. Thus, the hyposensitivity of SL-E neurons might correlate to the decreased cardiosomatic reflex that Liu et al. found in STZ-diabetic rats (Liu et al., 2011).
Algogenic mixtures that includes bradykinin are known to activate/sensitize cardiac primary afferents causing alteration in activity of upper thoracic spinal cord neurons and are widely used in the study of cardiac nociception in spinal neurons (Euchner-Wamser et al., 1994; Qin et al., 2003). One of the molecular sensors of ischemia is thought to be the transient receptor potential vanilloid 1 (TRPV1), a ligand-gated cation channel, whose activity is activated/modulated by inflammatory mixtures (Pan and Chen, 2004). Studies show that TRPV1 can be affected by diabetes in multiple ways depending on the tissue investigated (Hong and Wiley, 2005; Liu et al., 2011; Song et al., 2009; Wei et al., 2009). As TRPV1, located on cardiac afferents, are activated by IAC administration (Caterina et al., 1997; Pan and Chen, 2004; Zahner et al., 2003), diabetes-induced changes in TRPV1 expression or function might have altered the nociceptive information reaching the spinal cord and thus contributed to the changes found in activity of spinal cord neurons following IAC administration seen in the present study.
The changes caused by diabetes in the present study were apparent in the superficial laminae of the dorsal horn and this might be especially important for the further processing of the nociceptive cardiac input because neurons in the superficial laminae is thought to 1) be involved in visceral nociceptive processing leading to the perception of visceral pain, including referred pain (Cervero, 1991; Ness and Gebhart, 1989; Qin et al., 2003), and 2) act as interneurons, thereby modulating the activity of deeper neurons (Biella et al., 1997; Qin et al., 2003).
Overall, the results obtained by stimulation with IAC shows a central sensitization (indicated by LL-E neurons) and at the same time a hyposensitivity of SL-E neurons. It is likely that these changes are caused by an altered balance between inhibitory and facilitatory pathways or processes, either directly or indirectly. Further studies will need to be done to determine the role of descending pathways in diabetic animals.
4.3 Somatic input
Diabetes did not change the relative distribution of somatic receptive field properties (LT, WDR, HT) in the spinal cord of neither somatic nor cardiosomatic neurons and this is in accordance with the results found for the esophagosomatic spinal neurons in the study by Qin et al. (Qin et al., 2009). Chen and Pan, however, found a decreased proportion of HT STT neurons in the lumbar spinal cord of STZ-induced diabetic rats, (Chen and Pan, 2002) which could be explained by the difference in recorded neuron population.
In the present study, diabetes caused changes in the excitability of several subsets of neurons responding to somatic stimuli and this diversity of diabetes-induced changes is also seen in other studies on somatic spinal neurons. The increased response of WDR neurons to innocuous brushing of the skin is in accordance with the study by Chen and Pan which showed the same hypersensitivity of STT neurons towards brush (Chen and Pan, 2002). Chen and Pan, however, also found hypersensitivity of STT neurons after noxious pinch which contrasts the present study, where somatic HT and cardiosomatic WDR neurons from diabetic animals rather showed hyposensitivity following noxious pinch. Pertovaara et al. found hyposensitivity of T12-L2 WDR spinal neurons following innocuous and noxious somatic mechanical stimulation in STZ-diabetic rats after 3–4 weeks of diabetes (Pertovaara et al., 2001). In contrast, they found no changes in the response of nociceptive-specific spinal neurons but increased responses of LT neurons to innocuous mechanical stimuli (Pertovaara et al., 2001). The c-fos study by Morgado and Tavares also showed differentiated responses to different stimuli as noxious but not innocuous mechanical stimulation caused larger increases in Fos expression in the spinal cord dorsal horn of STZ-diabetic rats 4 weeks after induction of diabetes (Morgado and Tavares, 2007).
Studies have shown that diabetes causes alterations in mechanical threshold (Ahlgren et al., 1992) and responsiveness (Ahlgren et al., 1992; Chen and Levine, 2001; Chen and Levine, 2003) of subsets of Aδ- and C-fibers and these complex changes might be responsible for the differentiated changes seen in WDR and HT neurons receiving somatic or cardiosomatic input in the present study.
LT neurons, but not WDR or HT neurons, were found to exhibit an increased somatic field size. In comparison, in STZ-diabetic rats Chen and Pan found an enlarged somatic field size for L4–L5 STT neurons which consisted mostly of WDR and HT neurons (Chen and Pan, 2002), emphasizing the selective neuronal effect of diabetes.
The differentiated responses seen within these studies and differences in results between the studies might be caused by variations in duration of diabetes, segmental orientation, subgroup of neurons investigated, type of primary afferent supplying the spinal cord neuron, and differences in influence of inhibitory descending control.
4.4 Cardiosomatic convergence
Viscerovisceral and viscerosomatic convergence have been found for many tissues, e.g. the heart and lungs (Qin et al., 2007), the heart and esophagus (Qin et al., 2004a), and the heart and left upper arm (somatic structure) (McNeill and Burden, 1986). Cardiosomatic convergence is an important mechanism for anginal pain as this kind of pain is not felt in the heart per se, but rather is referred to somatic structures, such as the left arm and the neck (Foreman, 1999). In the present study, 78 % of neurons from diabetic animals receiving cardiac input received convergent input from cutaneous receptive fields, and this was similar to the 85 % convergence found in control animals. This finding is in agreement with a study by Qin et al. where no difference in esophagosomatic convergence was found in spinal neurons from STZ-diabetic rats and control rats (Qin et al., 2009).
The present study found an enlarged somatic receptive field in diabetic rats, specifically for the neurons receiving cardiosomatic convergent input and this is in accordance with the findings by Qin et al., where the size of the somatic receptive field for esophagosomatic spinal neurons in diabetic rats was also increased (Qin et al., 2009). Patients suffering from type 1 diabetes have been shown to have an increased pain referral area after esophageal and duodenal distension (Frokjaer et al., 2007). The increase in somatic receptive field size found in the present study is indicative of central sensitization (Arendt-Nielsen et al., 2000; Woolf and Mannion, 1999) and this could have an impact on the localization of anginal pain.
4.5 Concluding remarks
Diabetes altered the receptive and response characteristics of upper thoracic spinal cord neurons receiving cardiac and/or cutaneous somatic input. The changes were selective and differentiated, presenting challenges in interpretation of the results. Neurons with LL-E responses to IAC displayed hypersensitivity; whereas, neurons with SL-E responses showed hyposensitivity following IAC administration. Further, variable effects were found in somatic and cardiosomatic neurons that responded to cutaneous mechanical stimulation. The results show the complexity of upper thoracic spinal neurons, which probably in part is caused by the various visceral and somatic input that these neurons receive, reflecting the diversity of physiological roles.
Among possible explanations for the observed diabetes-induced alterations are an altered balance between facilitatory and inhibitory pathways (Chen and Pan, 2002; Kimura et al., 2005; Tanabe et al., 2005), changes in the intrinsic inhibitory system (Tanabe et al., 2005), wind-up of C-fibers (Kimura et al., 2005), spinal rewiring, changes in receptor and neuropeptides (Cloutier and Couture, 2000; Kamei et al., 1990), impaired cardiac vagal function (Schultz, 2001), and finally peripheral sensitization of primary afferents (Ahlgren et al., 1992; Chen and Levine, 2001; Chen and Levine, 2003; Khan et al., 2002).
Overall, the alterations found in the spinal processing of cardiac input likely contribute to the cardiovascular autonomic neuropathy frequently found among patients with diabetes and could therefore interfere with the cardiovisceral reflexes and development of pain following myocardial ischemia.
Future studies might include animals with longer-term diabetes as well as recordings of distinct neuron populations, e.g. STT neurons, in order to further evaluate the differentiated diabetes-induced effects that were found in the spinal processing of noxious cardiac and/or somatic input.
Acknowledgments
The authors thank D. Holston and R. Mott for excellent technical assistance. This work was supported by NIH grants NS-035471 and HL-075524, and The Danish Heart Association grant 05-04-B120-A476-22223.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ahlgren SC, White DM, Levine JD. Increased responsiveness of sensory neurons in the saphenous nerve of the streptozotocin-diabetic rat. J Neurophysiol. 1992;68(6):2077–2085. doi: 10.1152/jn.1992.68.6.2077. [DOI] [PubMed] [Google Scholar]
- Ahluwalia G, Jain P, Chugh SK, Wasir HS, Kaul U. Silent myocardial ischemia in diabetics with normal autonomic function. Int J Cardiol. 1995;48(2):147–153. doi: 10.1016/0167-5273(94)02233-9. [DOI] [PubMed] [Google Scholar]
- Arendt-Nielsen L, Laursen RJ, Drewes AM. Referred pain as an indicator for neural plasticity. Prog Brain Res. 2000;129:343–356. doi: 10.1016/s0079-6123(00)29026-2. [DOI] [PubMed] [Google Scholar]
- Biella G, Riva L, Sotgiu ML. Interaction between neurons in different laminae of the dorsal horn of the spinal cord. A correlation study in normal and neuropathic rats. Eur J Neurosci. 1997;9(5):1017–1025. doi: 10.1111/j.1460-9568.1997.tb01452.x. [DOI] [PubMed] [Google Scholar]
- Biessels GJ, Stevens EJ, Mahmood SJ, Gispen WH, Tomlinson DR. Insulin partially reverses deficits in peripheral nerve blood flow and conduction in experimental diabetes. Journal of the Neurological Sciences. 1996;140(1–2):12–20. doi: 10.1016/0022-510x(96)00080-9. [DOI] [PubMed] [Google Scholar]
- Burchiel KJ, Russell LC, Lee RP, Sima AA. Spontaneous activity of primary afferent neurons in diabetic BB/Wistar rats. A possible mechanism of chronic diabetic neuropathic pain. Diabetes. 1985;34(11):1210–1213. doi: 10.2337/diab.34.11.1210. [DOI] [PubMed] [Google Scholar]
- Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature. 1997;389(6653):816–824. doi: 10.1038/39807. [DOI] [PubMed] [Google Scholar]
- Cervero F. Mechanisms of acute visceral pain. Br Med Bull. 1991;47(3):549–560. doi: 10.1093/oxfordjournals.bmb.a072492. [DOI] [PubMed] [Google Scholar]
- Chen SR, Pan HL. Hypersensitivity of spinothalamic tract neurons associated with diabetic neuropathic pain in rats. J Neurophysiol. 2002;87(6):2726–2733. doi: 10.1152/jn.2002.87.6.2726. [DOI] [PubMed] [Google Scholar]
- Chen X, Levine JD. Hyper-responsivity in a subset of C-fiber nociceptors in a model of painful diabetic neuropathy in the rat. Neuroscience. 2001;102(1):185–192. doi: 10.1016/s0306-4522(00)00454-1. [DOI] [PubMed] [Google Scholar]
- Chen X, Levine JD. Altered temporal pattern of mechanically evoked C-fiber activity in a model of diabetic neuropathy in the rat. Neuroscience. 2003;121(4):1007–1015. doi: 10.1016/s0306-4522(03)00486-x. [DOI] [PubMed] [Google Scholar]
- Cloutier F, Couture R. Pharmacological characterization of the cardiovascular responses elicited by kinin B(1) and B(2) receptor agonists in the spinal cord of streptozotocin-diabetic rats. Br J Pharmacol. 2000;130(2):375–385. doi: 10.1038/sj.bjp.0703319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ditchburn CJ, Hall JA, de BM, Davies A, Kelly W, Bilous R. Silent myocardial ischaemia in patients with proved coronary artery disease: a comparison of diabetic and non-diabetic patients. Postgrad Med J. 2001;77(908):395–398. doi: 10.1136/pmj.77.908.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Euchner-Wamser I, Meller ST, Gebhart GF. A model of cardiac nociception in chronically instrumented rats: behavioral and electrophysiological effects of pericardial administration of algogenic substances. Pain. 1994;58(1):117–128. doi: 10.1016/0304-3959(94)90191-0. [DOI] [PubMed] [Google Scholar]
- Foreman RD. Mechanisms of cardiac pain. Annu Rev Physiol. 1999;61:143–167. doi: 10.1146/annurev.physiol.61.1.143. [DOI] [PubMed] [Google Scholar]
- Foreman RD, Ghorbani MLM, Qin C, Sheykhzade M, Farber J. Altered discharge characteristics of upper thoracic spinal neurons receiving noxious cardiac input in diabetic rats. Annual meeting for Society for Neuroscience Abstract 699.13/S13 2007 [Google Scholar]
- Frokjaer JB, Andersen SD, Ejskaer N, Funch-Jensen P, rendt-Nielsen L, Gregersen H, Drewes AM. Gut sensations in diabetic autonomic neuropathy. Pain. 2007;131(3):320–329. doi: 10.1016/j.pain.2007.04.009. [DOI] [PubMed] [Google Scholar]
- Gouty S, Regalia J, Helke CJ. Attenuation of the afferent limb of the baroreceptor reflex in streptozotocin-induced diabetic rats. Auton Neurosci. 2001;89(1–2):86–95. doi: 10.1016/S1566-0702(01)00256-9. [DOI] [PubMed] [Google Scholar]
- Hobbs SF, Chandler MJ, Bolser DC, Foreman RD. Segmental organization of visceral and somatic input onto C3-T6 spinothalamic tract cells of the monkey. J Neurophysiol. 1992;68(5):1575–1588. doi: 10.1152/jn.1992.68.5.1575. [DOI] [PubMed] [Google Scholar]
- Hong S, Wiley JW. Early painful diabetic neuropathy is associated with differential changes in the expression and function of vanilloid receptor 1. J Biol Chem. 2005;280(1):618–627. doi: 10.1074/jbc.M408500200. [DOI] [PubMed] [Google Scholar]
- Johnson MS, Ryals JM, Wright DE. Diabetes-induced chemogenic hypoalgesia is paralleled by attenuated stimulus-induced fos expression in the spinal cord of diabetic mice. Journal of Pain. 2007;8(8):637–649. doi: 10.1016/j.jpain.2007.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnstone MT, Kinzfogl GP. Diabetes and Heart Disease. In: Johnstone MT, Veves A, editors. Diabetes and Cardiovascular Disease. 2. Chapter 28. Humana Press; 2005. pp. 579–628. In series: Contemporary Cardiology, Series edited by Cannon, C. P. [Google Scholar]
- Kalichman MW, Dines KC, Bobik M, Mizisin AP. Nerve conduction velocity, laser Doppler flow, and axonal caliber in galactose and streptozotocin diabetes. Brain Res. 1998;810(1–2):130–137. doi: 10.1016/s0006-8993(98)00898-1. [DOI] [PubMed] [Google Scholar]
- Kamei J, Ogawa M, Kasuya Y. Development of Supersensitivity to Substance-P in the Spinal-Cord of the Streptozotocin-Induced Diabetic Rats. Pharmacology Biochemistry and Behavior. 1990;35(2):473–475. doi: 10.1016/0091-3057(90)90188-n. [DOI] [PubMed] [Google Scholar]
- Khan GM, Chen SR, Pan HL. Role of primary afferent nerves in allodynia caused by diabetic neuropathy in rats. Neuroscience. 2002;114(2):291–299. doi: 10.1016/s0306-4522(02)00372-x. [DOI] [PubMed] [Google Scholar]
- Kimura S, Tanabe M, Honda M, Ono H. Enhanced wind-up of the C-fiber-mediated nociceptive flexor reflex movement following painful diabetic neuropathy in mice. J Pharmacol Sci. 2005;97(2):195–202. doi: 10.1254/jphs.fp0040785. [DOI] [PubMed] [Google Scholar]
- Koistinen MJ, Airaksinen KE, Huikuri HV, Linnaluoto MM, Heikkila J, Torniainen P, Ahonen A. No difference in cardiac innervation of diabetic patients with painful and asymptomatic coronary artery disease. Diabetes Care. 1996;19(3):231–233. doi: 10.2337/diacare.19.3.231. [DOI] [PubMed] [Google Scholar]
- Liu XH, Qin C, Du JQ, Xu Y, Sun N, Tang JS, Li Q, Foreman RD. Diabetic rats show reduced cardiac-somatic reflex evoked by intrapericardial capsaicin. Eur J Pharmacol. 2011;651(1–3):83–88. doi: 10.1016/j.ejphar.2010.10.073. [DOI] [PubMed] [Google Scholar]
- McNeill DL, Burden HW. Convergence of sensory processes from the heart and left ulnar nerve onto a single afferent perikaryon: a neuroanatomical study in the rat employing fluorescent tracers. Anat Rec. 1986;214(4):441–447. doi: 10.1002/ar.1092140416. [DOI] [PubMed] [Google Scholar]
- Molander C, Xu Q, Rivero-Melian C, Grant G. Cytoarchitectonic organization of the spinal cord in the rat: II. The cervical and upper thoracic cord. J Comp Neurol. 1989;289(3):375–385. doi: 10.1002/cne.902890303. [DOI] [PubMed] [Google Scholar]
- Morgado C, Tavares I. C-fos expression at the spinal dorsal horn of streptozotocin-induced diabetic rats. Diabetes-Metabolism Research and Reviews. 2007;23(8):644–652. doi: 10.1002/dmrr.751. [DOI] [PubMed] [Google Scholar]
- Nemeth J, Szilvassy Z, Oroszi G, Porszasz R, Jakab B, Szolcsanyi J. Impaired capsaicin-induced decrease in heart rate and coronary flow in isolated heart of diabetic rats. Acta Physiol Hung. 2001;88(3–4):207–218. doi: 10.1556/APhysiol.88.2001.3-4.3. [DOI] [PubMed] [Google Scholar]
- Nemeth J, Than M, Sari R, Peitl B, Oroszi G, Farkas B, Szolcsanyi J, Szilvassy Z. Impairment of neurogenic inflammatory and anti-inflammatory responses in diabetic rats. Eur J Pharmacol. 1999;386(1):83–88. doi: 10.1016/s0014-2999(99)00724-4. [DOI] [PubMed] [Google Scholar]
- Ness TJ, Gebhart GF. Characterization of neuronal responses to noxious visceral and somatic stimuli in the medial lumbosacral spinal cord of the rat. J Neurophysiol. 1987;57(6):1867–1892. doi: 10.1152/jn.1987.57.6.1867. [DOI] [PubMed] [Google Scholar]
- Ness TJ, Gebhart GF. Characterization of Superficial T13-L2 Dorsal Horn Neurons Encoding for Colorectal Distension in the Rat - Comparison with Neurons in Deep Laminae. Brain Research. 1989;486(2):301–309. doi: 10.1016/0006-8993(89)90516-7. [DOI] [PubMed] [Google Scholar]
- Ness TJ, Gebhart GF. Acute inflammation differentially alters the activity of two classes of rat spinal visceral nociceptive neurons. Neurosci Lett. 2000;281(2–3):131–134. doi: 10.1016/s0304-3940(00)00832-6. [DOI] [PubMed] [Google Scholar]
- Ness TJ, Gebhart GF. Inflammation enhances reflex and spinal neuron responses to noxious visceral stimulation in rats. Am J Physiol Gastrointest Liver Physiol. 2001;280(4):G649–G657. doi: 10.1152/ajpgi.2001.280.4.G649. [DOI] [PubMed] [Google Scholar]
- Pan HL, Chen SR. Myocardial ischemia recruits mechanically insensitive cardiac sympathetic afferents in cats. J Neurophysiol. 2002;87(2):660–668. doi: 10.1152/jn.00506.2001. [DOI] [PubMed] [Google Scholar]
- Pan HL, Chen SR. Sensing tissue ischemia: another new function for capsaicin receptors? Circulation. 2004;110(13):1826–1831. doi: 10.1161/01.CIR.0000142618.20278.7A. [DOI] [PubMed] [Google Scholar]
- Pan HL, Longhurst JC. Lack of a role of adenosine in activation of ischemically sensitive cardiac sympathetic afferents. Am J Physiol. 1995;269(1 Pt 2):H106–H113. doi: 10.1152/ajpheart.1995.269.1.H106. [DOI] [PubMed] [Google Scholar]
- Pertovaara A, Wei H, Kalmari J, Ruotsalainen M. Pain behavior and response properties of spinal dorsal horn neurons following experimental diabetic neuropathy in the rat: modulation by nitecapone, a COMT inhibitor with antioxidant properties. Exp Neurol. 2001;167(2):425–434. doi: 10.1006/exnr.2000.7574. [DOI] [PubMed] [Google Scholar]
- Qin C, Chandler MJ, Foreman RD. Esophagocardiac convergence onto thoracic spinal neurons: comparison of cervical and thoracic esophagus. Brain Research. 2004a;1008(2):193–197. doi: 10.1016/j.brainres.2003.12.056. [DOI] [PubMed] [Google Scholar]
- Qin C, Chandler MJ, Foreman RD, Farber JP. Upper thoracic respiratory interneurons integrate noxious somatic and visceral information in rats. J Neurophysiol. 2002;88(5):2215–2223. doi: 10.1152/jn.00120.2002. [DOI] [PubMed] [Google Scholar]
- Qin C, Chandler MJ, Miller KE, Foreman RD. Responses and afferent pathways of superficial and deeper c(1)–c(2) spinal cells to intrapericardial algogenic chemicals in rats. J Neurophysiol. 2001;85(4):1522–1532. doi: 10.1152/jn.2001.85.4.1522. [DOI] [PubMed] [Google Scholar]
- Qin C, Chandler MJ, Miller KE, Foreman RD. Chemical activation of cardiac receptors affects activity of superficial and deeper T3–T4 spinal neurons in rats. Brain Res. 2003;959(1):77–85. doi: 10.1016/s0006-8993(02)03728-9. [DOI] [PubMed] [Google Scholar]
- Qin C, Foreman RD, Farber JP. Characterization of thoracic spinal neurons with noxious convergent inputs from heart and lower airways in rats. Brain Res. 2007;1141:84–91. doi: 10.1016/j.brainres.2007.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin C, Ghorbani MLM, Wu MY, Farber JP, Ma JX, Foreman RD. Characterization of upper thoracic spinal neurons responding to esophageal distension in diabetic rats. Autonomic Neuroscience-Basic & Clinical. 2009;145(1–2):27–34. doi: 10.1016/j.autneu.2008.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin C, Kranenburg A, Foreman RD. Descending modulation of thoracic visceroreceptive transmission by C1–C2 spinal neurons. Auton Neurosci. 2004b;114(1–2):11–16. doi: 10.1016/j.autneu.2004.05.009. [DOI] [PubMed] [Google Scholar]
- Schultz HD. Cardiac vagal chemosensory afferents - Function in pathophysiological states. Neuro-Cardiovascular Regulation: from Molecules to Man. 2001;940:59–73. [PubMed] [Google Scholar]
- Schultz HD. The spice of life is at the root of cardiac pain. J Physiol. 2003;551(Pt 2):400. doi: 10.1113/jphysiol.2003.050104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selvarajah D, Wilkinson ID, Emery CJ, Harris ND, Shaw PJ, Witte DR, Griffiths PD, Tesfaye S. Early involvement of the spinal cord in diabetic peripheral neuropathy. Diabetes Care. 2006;29(12):2664–2669. doi: 10.2337/dc06-0650. [DOI] [PubMed] [Google Scholar]
- Song JX, Wang LH, Yao L, Xu C, Wei ZH, Zheng LR. Impaired transient receptor potential vanilloid 1 in streptozotocin-induced diabetic hearts. International Journal of Cardiology. 2009;134(2):290–292. doi: 10.1016/j.ijcard.2007.12.081. [DOI] [PubMed] [Google Scholar]
- Suzuki C, Ozaki I, Tanosaki M, Suda T, Baba M, Matsunaga M. Peripheral and central conduction abnormalities in diabetes mellitus. Neurology. 2000;54(10):1932–1937. doi: 10.1212/wnl.54.10.1932. [DOI] [PubMed] [Google Scholar]
- Tanabe M, Shimizu S, Takabayashi K, Honda M, Ono H. Functional alteration of inhibitory influences on spinal motor output in painful diabetic neuropathy in rats. Neurosci Lett. 2005;389(3):152–156. doi: 10.1016/j.neulet.2005.07.039. [DOI] [PubMed] [Google Scholar]
- Thomas PK, Tomlinson DR. Diabetic and hypglycemic neuropathy, chapter 64. In: Dyck PJ, Thomas PK, Griffin JW, Low PA, Poduslo JF, editors. Peripheral Neuropathy. 3. Saunders; Philadelphia: 1993. pp. 1219–1250. [Google Scholar]
- Vinik AI, Maser RE, Mitchell BD, Freeman R. Diabetic autonomic neuropathy. Diabetes Care. 2003;26(5):1553–1579. doi: 10.2337/diacare.26.5.1553. [DOI] [PubMed] [Google Scholar]
- Vinik AI, Ziegler D. Diabetic cardiovascular autonomic neuropathy. Circulation. 2007;115(3):387–397. doi: 10.1161/CIRCULATIONAHA.106.634949. [DOI] [PubMed] [Google Scholar]
- Walker D, Carrington A, Cannan SA, Sawicki D, Sredy J, Boulton AJ, Malik RA. Structural abnormalities do not explain the early functional abnormalities in the peripheral nerves of the streptozotocin diabetic rat. J Anat. 1999;195(Pt 3):419–427. doi: 10.1046/j.1469-7580.1999.19530419.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei ZH, Wang LH, Han J, Song JX, Yao L, Shao L, Sun ZH, Zheng LR. Decreased Expression of Transient Receptor Potential Vanilloid 1 Impairs the Postischemic Recovery of Diabetic Mouse Hearts. Circulation Journal. 2009;73(6):1127–1132. doi: 10.1253/circj.cj-08-0945. [DOI] [PubMed] [Google Scholar]
- Woolf CJ, Mannion RJ. Neuropathic pain: aetiology, symptoms, mechanisms, and management. Lancet. 1999;353(9168):1959–1964. doi: 10.1016/S0140-6736(99)01307-0. [DOI] [PubMed] [Google Scholar]
- Woolf CJ, Shortland P, Coggeshall RE. Peripheral nerve injury triggers central sprouting of myelinated afferents. Nature. 1992;355(6355):75–78. doi: 10.1038/355075a0. [DOI] [PubMed] [Google Scholar]
- Zahner MR, Li DP, Chen SR, Pan HL. Cardiac vanilloid receptor 1-expressing afferent nerves and their role in the cardiogenic sympathetic reflex in rats. J Physiol. 2003;551(Pt 2):515–523. doi: 10.1113/jphysiol.2003.048207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zochodne DW. Is early diabetic neuropathy a disorder of the dorsal root ganglion? A hypothesis and critique of some current ideas on the etiology of diabetic neuropathy. J Peripher Nerv Syst. 1996;1(2):119–130. [PubMed] [Google Scholar]