Abstract
Background
A low level of high-density lipoprotein cholesterol (HDL-C) is strongly associated with cardiovascular events. However, the significance of HDL-C after statin therapy on the outcome of patients who have undergone percutaneous coronary intervention (PCI) with drug eluting stents (DES) is unclear.
Objectives
To investigate the significance of HDL-C after statin therapy on cardiovascular events in patients with coronary artery disease after DES implantation.
Methods
Patients who underwent PCI with DES from January 2004 to December 2009 were prospectively enrolled. The follow-up lipid panel of 2693 patients (median lab follow-up duration 225 days) who had continued using statins after PCI and who attained low-density lipoprotein cholesterol (LDL-C) <100 mg/dl was analysed. Major adverse cardiac events (MACE), including all-cause death, non-fatal myocardial infarction, and target vessel revascularisation according to follow-up HDL-C level (40 mg/dl for men or 50 mg/dl for women) were compared with the use of propensity scores matching.
Results
Median follow-up duration was 832 days. 1585 (58.9%) patients had low follow-up HDL-C and 1108 had high follow-up HDL-C. The low follow-up HDL-C group had significantly higher rates of MACE. Low follow-up HDL-C was a significant independent predictor of MACE (adjusted HR 1.404, 95% CI 1.111 to 1.774, p=0.004). In further analysis with propensity scores matching, overall findings were consistent.
Conclusions
Raising HDL-C levels may be a subsequent goal after achieving target LDL-C levels in patients with DES implantation.
Keywords: High-density lipoprotein cholesterol; statin; percutaneous coronary intervention; drug eluting stents; coronary angioplasty; lipoproteins, coronary artery disease
Introduction
Lowering low-density lipoprotein cholesterol (LDL-C) has been the primary focus in lipid modification for treatment and prevention of atherosclerosis. Lipid-lowering treatment with hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (‘statins’) which reduce LDL-C, has achieved dramatic reductions in cardiovascular events, with a RR reduction of 25–40%.1 2 However, despite attaining optimal LDL-C targets in all the statin trials, there still remains a substantial residual risk in the active treatment arms.
The Framingham Heart Study showed that low high-density lipoprotein cholesterol (HDL-C) (defined as <40 mg/dl for men and <50 mg/dl for women) was more potent as a risk factor for coronary artery disease (CAD) than high LDL-C.3 4 HDL-C levels are inversely related to cardiovascular events, even in patients receiving statin therapy.5 6 HDL-C levels continue to be inversely associated with cardiovascular events among those on statins with well controlled LDL-C levels, including those with LDL-C <70 mg/dl.5 6 Moreover, moderate increases in HDL-C in statin-treated patients are correlated with regression of coronary atherosclerosis.7 These findings support the hypothesis that HDL-C is a potent atheroprotective factor; it is considered to be a therapeutic target independent of LDL-C lowering. However, there is a paucity of data regarding the impact of HDL-C levels after statin therapy on clinical effects in patients who have undergone percutaneous coronary intervention (PCI) with drug eluting stents (DES). Accordingly, we sought to investigate the significance of HDL-C levels after statin therapy on cardiovascular events in patients treated with DES implantation for CAD.
Methods
Study population and COACT registry
COACT (Catholic University of Korea: percutaneous coronary intervention) is a large, prospective observational registry of demographic, clinical and procedural data, with short-term and long-term clinical outcome of all patients undergoing PCI with the use of DES from eight affiliated hospitals of the Catholic University of Korea between January 2004 and December 2009. The hospitals are located throughout the country, and all perform high-volume PCI (more than 500 cases per year). There was no industry involvement in the design, conduct or analysis of the study. The study protocol was approved by institutional review boards at each participating institution.
In the present study, 2693 out of the total of 9293 registered patients who had undergone successful PCI with DES had continued taking statins for more than 3 months after PCI; their follow-up LDL-C levels were below 100 mg/dl. Exclusion criteria were as follows: discontinuation of the statin during the first 3 months after PCI; development of major adverse cardiac events (MACE) within 3 months after PCI; loss of follow-up lipid panels; and estimated life expectancy of <12 months (figure 1).
Figure 1.
Study flow chart. COACT, Catholic University of Korea: Percutaneous Coronary Intervention; DES, drug eluting stents; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PCI, percutaneous coronary intervention.
PCI procedure and medical treatment
Before the PCI, all patients received aspirin 300 mg daily. Clopidogrel (600 mg loading dose) was given at least 1 day before the procedure. The procedure was performed through the femoral or radial artery after administration of unfractionated heparin (100 U/kg). During the procedure, patients received unfractionated heparin to maintain an activated clotting time between 250 and 300 s. The choice of stent was at each physician's discretion and the stent was deployed after balloon angioplasty. A successful PCI procedure was defined as a decrease in minimum stenosis diameter to <30% with thrombolysis in myocardial infarction grade III flow on coronary angiogram.
Statins were prescribed to all patients after PCI at the discretion of the operating cardiologist. Patients were given the usual starting dose of the statin, targeting LDL-C below 100 mg/dl. After discharge, patients continued receiving the same medications except for some intravenous or temporary medications.
Study definitions and clinical follow-up
The records of cardiovascular risk factors, past history and laboratory findings were mainly dependent on patients' electronic medical records. All follow-up laboratory tests were performed using fasting blood samples in the morning after fasting for 8 h or more. Serum lipid profiles were determined by standard enzymatic methods on a Hitachi 7600-210 (Hitachi Co., Tokyo, Japan) autoanalyser. All-cause deaths were considered to be cardiac deaths after the exclusion of non-cardiac deaths. Myocardial infarction was defined as chest pain with new ST-segment changes and elevation of cardiac markers which reflected myocardial necrosis to at least twice the upper limit of normal. Target lesion revascularisation (TLR) was defined as ischaemia-induced PCI of the target lesion resulting from restenosis or reocclusion within the stent or in the adjacent 5 mm of the distal or proximal segments. Target vessel revascularisation (TVR) was also defined as any segment of the epicardial coronary artery containing the target lesion.8
The clinical, angiographic, procedural or operative, and outcome data were collected in the dedicated PCI and surgical databases by independent research personnel. All the outcomes of interest were confirmed by source document; they were centrally adjudicated by a local events committee of the Cardiovascular Center of Seoul St Mary's Hospital, Seoul, Korea, whose members were unaware of patients' status. For validation of complete follow-up data, information on censored survival data was obtained to 31 December 2010 from telephone interviews with the corresponding patients and also from the database of the National Health Insurance Corporation, Korea with the use of a unique personal identification number.
Propensity score methods
We estimated propensity scores (PS) for follow-up HDL-C for each of the 2693 patients using a non-parsimonious multivariable logistic regression model. In this model, follow-up HDL-C was used as the dependent variable, and all measured baseline, angiographic, and procedural characteristics shown in tables 1 and 2, LDL-C at index PCI, follow-up LDL-C, and medication were included as covariates. The model was well calibrated (Hosmer–Lemeshow test: p=0.30) with reasonable discrimination (c statistic=0.66).9
Table 1.
Baseline clinical characteristics at index PCI according to follow-up HDL-C level
| Total population | Propensity-matched population | |||||
| Low HDL-C (n=1585) | High HDL-C (n=1108) | p Value | Low HDL-C (n=990) | High HDL-C (n=990) | p Value | |
| Age, years | 62.7±10.6 | 62.2±10.8 | 0.226 | 62.0±10.4 | 62.1±10.8 | 0.899 |
| Age ≥65 years | 776 (49.0) | 507 (45.8) | 0.108 | 453 (45.8) | 449 (45.4) | 0.857 |
| Male | 947 (59.7) | 833 (75.2) | <0.001 | 716 (72.3) | 717 (72.4) | 0.960 |
| Body mass index, kg/m2 | 25.0±3.1 | 24.4±3.1 | <0.001 | 24.6±2.8 | 24.6±3.0 | 0.648 |
| Clinical presentation | 0.002 | 0.757 | ||||
| Stable angina | 719 (45.4) | 576 (52.0) | 482 (48.7) | 485 (49.0) | ||
| Unstable angina | 387 (24.4) | 236 (21.3) | 238 (24.0) | 225 (22.7) | ||
| AMI | 479 (30.2) | 296 (26.7) | 270 (27.3) | 280 (28.3) | ||
| Hypertension | 993 (62.6) | 629 (56.8) | 0.002 | 579 (58.5) | 579 (58.5) | 1.000 |
| Diabetes mellitus | 650 (41.0) | 340 (30.7) | <0.001 | 349 (35.3) | 330 (33.3) | 0.368 |
| Current smoker | 373 (23.6) | 290 (26.2) | 0.123 | 249 (25.2) | 260 (26.3) | 0.572 |
| Chronic kidney disease | 68 (4.3) | 35 (3.2) | 0.153 | 28 (2.8) | 34 (3.4) | 0.439 |
| Family history of CAD | 75 (4.7) | 60 (5.4) | 0.421 | 51 (5.2) | 52 (537) | 0.919 |
| Prior MI | 69 (4.4) | 39 (3.5) | 0.318 | 40 (4.0) | 37 (3.7) | 0.727 |
| Prior PCI | 101 (6.4) | 69 (6.4) | 0.936 | 44 (4.4) | 46 (4.6) | 0.829 |
| Prior CABG | 11 (0.7) | 15 (1.4) | 0.108 | 10 (1.0) | 11 (1.1) | 0.826 |
| Prior stroke | 116 (7.3) | 73 (6.6) | 0.491 | 77 (7.8) | 67 (6.8) | 0.387 |
| LVEF, %* | 59.2 | 60.4 | 0.003 | 60.1±9.4 | 60.2±9.2 | 0.797 |
| LVEF <50% | 219 (15.6) | 119 (12.2) | 0.020 | 111 (11.2) | 109 (11.0) | 0.943 |
Data are presented as mean±SD or n (%).
Left ventricular ejection fraction (LVEF) was available for 2385 patients.
AMI, acute myocardial infarction; CABG, coronary artery bypass graft; CAD, coronary artery disease; HDL-C, high-density lipoprotein cholesterol; MI, myocardial infarction; PCI, percutaneous coronary intervention.
Table 2.
Angiographic characteristics of patients according to follow-up HDL-C level
| Total population | Propensity-matched population | |||||
| Low HDL-C (n=1585) | High HDL-C (n=1108) | p Value | Low HDL-C (n=990) | High HDL-C (n=990) | p Value | |
| Involved vessel | 0.002 | 0.909 | ||||
| 1VD | 654 (41.3) | 532 (48.0) | 461 (46.6) | 461 (46.6) | ||
| 2VD | 517 (32.6) | 331 (29.9) | 310 (31.3) | 303 (30.6) | ||
| 3VD | 414 (26.1) | 245 (22.1) | 219 (22.1) | 226 (22.8) | ||
| Number of B2/C lesion | 1.04±0.81 | 1.02±0.82 | 0.532 | 1.02±0.82 | 1.02±0.81 | 0.868 |
| Stent number per patient | 1.40±0.83 | 1.36±0.78 | 0.193 | 1.39±0.83 | 1.37±0.80 | 0.638 |
| Mean stent diameter, mm | 3.17±0.38 | 3.23±0.38 | <0.001 | 3.21±0.39 | 3.21±0.38 | 0.914 |
| Stent length, mm | 40.9±26.1 | 39.5±26.2 | 0.146 | 40.0±26.3 | 39.6±26.1 | 0.776 |
| Stent type | ||||||
| SES | 804 (50.7) | 547 (49.4) | 0.488 | 480 (48.5) | 499 (50.4) | 0.393 |
| PES | 472 (29.8) | 327 (29.5) | 0.882 | 293 (29.6) | 288 (29.1) | 0.805 |
| ZES | 253 (16.0) | 212 (19.1) | 0.032 | 181 (18.3) | 181 (18.3) | 1.000 |
| EES | 245 (15.5) | 172 (15.5) | 0.963 | 156 (15.8) | 157 (15.9) | 0.951 |
B2/C, complex lesion; EES, everolimus-eluting stent; HDL-C, high-density lipoprotein cholesterol; PES, paclitaxel-eluting stent; SES, sirolimus-eluting stent; VD, vessel disease; ZES, zotarolimus-eluting stent.
We then applied PS, the single composite variable, to match each high follow-up HDL-C patient with a low follow-up HDL-C patient with a very similar PS, thus matching 990 pairs (62.5% of the 1585 having low follow-up HDL-C and 89.4% of the 1108 having high follow-up HDL-C) with similar PS. In our matching algorithm, we performed 1:1 match iteration by similar PS from initial eight to one digit.
We compared the balance of all baseline covariates in tables 1–4 between the groups before and after PS matching using the standardised difference, which directly quantifies the bias in the means (or proportions) of covariates across the groups, expressed as a percentage of the pooled SD.10 11 Before matching, the mean PS for low follow-up HDL-C patients (n=1585) was 0.6203; for high follow-up HDL-C patients (n=1108) it was 0.5422, with an associated standardised difference of 59.1% (t-test p value <0.0001). After matching, the mean PS for the matched patients with low follow-up HDL-C (n=990) was 0.5645; in those with high follow-up HDL-C (n=990) it was 0.5637, which yields a standardised difference of 0.65% (t-test p value=0.88).
Table 3.
Laboratory findings at index PCI and at follow-up according to follow-up HDL-C level
| Total population | Propensity-matched population | |||||
| Low HDL-C (n=1585) | High HDL-C (n=1108) | p Value | Low HDL-C (n=990) | High HDL-C (n=990) | p Value | |
| At index PCI | ||||||
| Glucose, mg/dl | 137.2±65.3 | 126.9±51.1 | <0.001 | 132.7±61.1 | 128.7±53.1 | 0.133 |
| HbA1C (%) | 6.7±1.5 | 6.4±1.4 | 0.001 | 6.6±1.5 | 6.5±1.4 | 0.290 |
| Creatinine, mg/dl | 1.13±0.96 | 1.09±0.90 | 0.325 | 1.11±0.86 | 1.09±0.84 | 0.595 |
| Total cholesterol, mg/dl | 173.5±40.6 | 176.6±39.4 | 0.049 | 173.0±40.2 | 177.6±39.8 | 0.012 |
| Triglyceride, mg/dl | 154.9±138.8 | 138.8±120.1 | <0.001 | 159.0±122.0 | 137.4±104.6 | <0.001 |
| LDL-C, mg/dl | 107.0±33.4 | 105.9±36.5 | 0.432 | 107.0±30.3 | 106.9±34.7 | 0.962 |
| HDL-C, mg/dl | 38.9±9.5 | 48.3±11.4 | <0.001 | 38.5±8.7 | 48.2±11.2 | <0.001 |
| hs-CRP, mg/l | 1.68±6.55 | 1.09±5.93 | 0.025 | 1.51±7.18 | 1.15±6.224 | 0.261 |
| Follow-up lab after PCI | ||||||
| Glucose, mg/dl | 123.8±52.3 | 118.3±42.3 | 0.004 | 120.3±45.1 | 119.6±43.7 | 0.758 |
| HbA1C (%) | 6.8±1.5 | 0.001 | 6.7±1.4 | 6.6±1.3 | 0.483 | |
| Creatinine, mg/dl | 1.18±1.13 | 1.11±0.91 | 0.061 | 1.14±1.0 | 1.10±0.87 | 0.428 |
| Total cholesterol, mg/dl | 129.0±28.0 | 140.1±23.2 | <0.001 | 129.6±28.6 | 140.0±23.2 | <0.001 |
| Triglyceride, mg/dl | 151.0±114.0 | 114.1±77.9 | <0.001 | 152.4±117.0 | 113.0±74.6 | <0.001 |
| LDL-C, mg/dl | 70.8±27.5 | 69.3±16.7 | 0.005 | 68.8±16.7 | 68.9±16.8 | 0.924 |
| HDL-C, mg/dl | 37.5±6.04 | 52.3±9.7 | <0.001 | 35.3±6.0 | 52.4±9.6 | <0.001 |
| hs-CRP, mg/l | 1.10±9.46 | 0.59±2.07 | 0.072 | 1.31±11.8 | 0.59±2.03 | 0.086 |
Data are presented as mean±SD.
HDL-C, high-density lipoprotein cholesterol; hs-CRP, high sensitivity C reactive protein; LDL-C, low-density lipoprotein cholesterol; PCI, percutaneous coronary intervention.
Table 4.
Medications according to follow-up HDL-C level
| Total population | Propensity-matched population | |||||
| Low HDL-C (n=1585) | High HDL-C (n=1108) | p Value | Low HDL-C (n=990) | High HDL-C (n=990) | p Value | |
| Prior PCI | ||||||
| Aspirin | 776 (49.0) | 507 (45.8) | 0.108 | 377 (42.4) | 389 (42.8) | 0.852 |
| Statin | 304 (21.7) | 226 (22.2) | 0.771 | 184 (20.7) | 200 (22.2) | 0.454 |
| At discharge | ||||||
| Aspirin | 1561 (98.5) | 1097 (99.0) | 0.381 | 982 (99.3) | 980 (99.0) | 0.998 |
| Thienophyridine | 1583 (99.9) | 1105 (99.7) | 0.655 | 877 (99.9) | 892 (99.7) | 0.625 |
| β-blocker | 1099 (69.3) | 729 (65.8) | 0.054 | 695 (70.2) | 662 (66.9) | 0.110 |
| ACEI or ARB | 1184 (74.7) | 809 (73.0) | 0.326 | 719 (72.6) | 738 (74.5) | 0.333 |
| CCB | 350 (22.1) | 316 (28.5) | <0.001 | 259 (26.2) | 253 (25.6) | 0.758 |
ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin II receptor blocker; CCB, calcium channel blocker; HDL-C, high-density lipoprotein cholesterol; PCI, percutaneous coronary intervention.
Statistical analysis
Continuous variables were expressed as mean±SD and compared with the Student t test. Discrete variables were expressed as percentages and compared with the χ2 test or Fisher's exact test. A multivariable Cox regression analysis (after confirming the appropriateness of the proportional hazards assumption) was performed in order to identify independent predictors for MACE. Variables which were evaluated in the multivariable Cox regression analysis included using those with significant association in the univariate analysis and also those without statistical significance in the univariate analysis, but with prognostic impact demonstrated in previous studies. The cumulative survival was estimated by Kaplan–Meier survival curves, and compared using log-rank tests. All analyses were two-tailed, with clinical significance defined as values of p<0.05. All statistical analyses were performed with SAS V.9.1.
Results
Characteristics of the study populations
The study flow chart is summarised in figure 1. Among 9292 patients registered, we selected 3239 subjects who had been prescribed statins for more than 3 months after PCI, with follow-up lipid panels. We excluded patients with follow-up LDL-C >100 mg/dl (n=546). A total of 2693 patients were selected in this study.
Baseline demographic, clinical, laboratory and angiographic characteristics between the two groups are shown in tables 1–3. The low follow-up HDL-C group had less male gender, lower left ventricular ejection fraction, higher body mass index, and a higher prevalence of acute coronary syndrome (ACS), hypertension and diabetes mellitus. These patients with low follow-up HDL-C were more likely to have higher baseline triglycerides, higher baseline high sensitivity C reactive protein (hs-CRP), higher follow-up triglycerides, and lower baseline HDL-C. Patients with low follow-up HDL-C had higher incidences of multi-vessel disease and less mean stent diameter on coronary angiogram.
Statin therapy reduced LDL-C levels by 28.3% and increased HDL-C levels by 2.0% during 7.5 months on average. Most study subjects (1416 patients, 52.6%) achieved LDL-C levels ≤70 mg/dl after statin therapy. At index PCI, 1592 patients (59.1%) had low HDL-C levels. Among these patients, 373 (23.4% of patients with low HDL-C, 13.9% of total patients) achieved high HDL-C levels and 370 patients with high HDL-C levels at baseline (33.3% of patients with high HDL-C, 13.7% of total patients) attained low HDL-C levels after statin therapy.
Characteristics of patients matched for propensity scores
In the matched cohorts, there were no significant differences in baseline demographic, clinical and angiographic characteristics, and medications between two groups (tables 1, 2 and 4). The only differences were in lipid profiles (table 3).
Clinical outcomes for the study populations
The median follow-up duration was 804 days (IQR 423–1255) in the low follow-up HDL-C group and 861 days (IQR 454–1253) in the high follow-up HDL-C group, which was not significantly different (p=0.272). The mean compliance of statin during entire follow-up duration was 96.3% (n=1526) in the low follow-up HDL-C group and 97.2% (n=1077) in the high follow-up HDL-C group, which was not significantly different (p=0.189). Complete follow-up data for major clinical events were obtained in 97.9% of the overall cohort. The follow-up coronary angiography was performed in 77.4% (n=2084) of the overall cohort at median follow-up 239 days (IQR 192–299). The follow-up lipid panels were performed at median follow-up 228 days (IQR 181–299) in the low follow-up HDL-C group and 223 days (IQR 181–287) in the high follow-up HDL-C group, which was not significantly different (p=0.236).
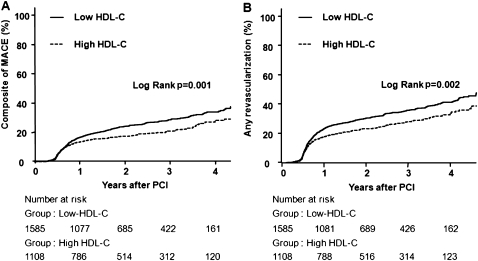
During follow-up days, 399 patients (25.2%) with low HDL-C levels and 213 patients (19.2%) with high HDL-C levels had MACE. The incidence of MACE was significantly higher in patients with low follow-up HDL-C than in those with high follow-up HDL-C (table 5, figure 2A). Although both groups had comparable incidences of all-cause death or myocardial infarction, the low follow-up HDL-C group had a significantly higher incidence of TLR and TVR (table 5, figure 2B). Multivariate analysis showed that low follow-up HDL-C was associated with significantly higher incidence rates of MACE, TLR and TVR (table 5). Age, renal disease, total stent length, angiographically complex lesion and follow-up triglyceride levels were also associated with MACE. However, levels of baseline triglycerides, baseline HDL, baseline LDL, follow-up LDL, baseline hs-CRP and follow-up hs-CRP did not influence the occurrence of MACE.
Table 5.
Clinical events in patients with low follow-up HDL-C compared with high follow-up HDL-C
| Low HDL-C | High HDL-C | Unadjusted HR (95% CI) | p Value | Adjusted HR* (95% CI) | p Value | |
| Total population (n=2693) | (n=1585) | (n=1108) | ||||
| All-cause death | 45 (2.8) | 16 (1.4) | 2.02 (1.14 to 3.58) | 0.016 | 1.35 (0.59 to 3.08) | 0.471 |
| Cardiac death | 21 (1.3) | 9 (0.8) | 1.66 (0.76 to 3.63) | 0.201 | 1.34 (0.43 to 4.18) | 0.618 |
| MI | 19 (1.2) | 10 (0.9) | 1.34 (0.62 to 2.88) | 0.455 | 1.31 (0.47 to 3.67) | 0.605 |
| Stroke | 28 (1.8) | 18 (1.6) | 1.10 (0.61 to 2.00) | 0.750 | 1.50 (0.61 to 3.68) | 0.382 |
| TLR | 278 (17.5) | 160 (14.4) | 1.22 (1.01 to 1.48) | 0.044 | 1.45 (1.10 to 1.91) | 0.008 |
| TVR | 360 (22.7) | 194 (17.5) | 1.33 (1.11 to 1.58) | 0.002 | 1.46 (1.14 to 1.86) | 0.003 |
| Composite of MACE | 399 (25.2) | 213 (19.2) | 1.34 (1.14 to 1.58) | 0.001 | 1.40 (1.11 to 1.77) | 0.004 |
| Propensity-matched population (n=1980) | (n=990) | (n=990) | ||||
| All-cause death | 15 (1.5) | 11 (1.1) | 1.38 (0.64 to 3.01) | 0.415 | 1.08 (0.28 to 4.14) | 0.915 |
| Cardiac death | 4 (0.4) | 7 (0.7) | 0.57 (0.17 to 1.95) | 0.372 | 0.65 (0.25 to 2.80) | 0.851 |
| MI | 11 (1.1) | 10 (1.0) | 1.09 (0.46 to 2.58) | 0.837 | 0.85 (0.23 to 3.11) | 0.805 |
| Stroke | 18 (1.8) | 18 (1.8) | 0.99 (0.52 to 1.91) | 0.986 | 0.59 (0.22 to 1.60) | 0.298 |
| TLR | 186 (18.8) | 136 (13.7) | 1.37 (1.10 to 1.71) | 0.005 | 1.64 (1.22 to 2.19) | 0.001 |
| TVR | 237 (23.9) | 168 (17.0) | 1.44 (1.18 to 1.75) | <0.001 | 1.65 (1.27 to 2.13) | <0.001 |
| Composite of MACE | 248 (25.1) | 183 (18.5) | 1.38 (1.14 to 1.67) | 0.001 | 1.55 (1.21 to 2.00) | 0.001 |
Adjusted covariates included age, sex, hypertension, diabetes mellitus, chronic renal disease, current smoker, acute coronary syndrome, ejection fraction, baseline high-density lipoprotein (HDL), baseline low-density lipoprotein (LDL), baseline triglycerides (TG), baseline high sensitivity C reactive protein (hs-CRP), follow-up LDL, follow-up TG, follow-up hs-CRP, total stent length, mean stent diameter, number of stent, number of B2/C lesions.
HDL-C, high-density lipoprotein cholesterol; MACE, major adverse cardiac events; MI, myocardial infarction; TLR, target lesion revascularisation; TVR, target vessel revascularisation.
Figure 2.
Kaplan–Meier curves in all study populations. (A) Kaplan–Meier curves for major adverse cardiac events (MACE) in the all-study population with low follow-up high-density lipoprotein cholesterol (HDL-C) (solid line) versus high follow-up HDL-C (dashed line). (B) Kaplan–Meier curves for any revascularisation in the all-study population with low follow-up HDL-C (solid line) versus high follow-up HDL-C (dashed line). PCI, percutaneous coronary intervention.
Clinical outcome for patients matched for propensity scores
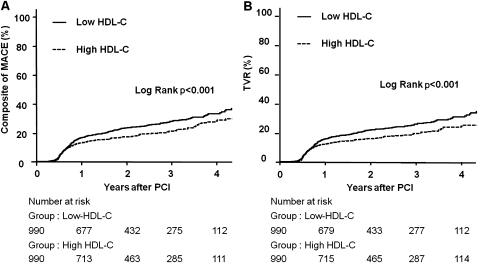
For the 990 matched pairs, there was no significant difference between two groups in all-cause death and cardiac death during a median follow-up of 861 days. The incidence of MACE was significantly higher in patients with low follow-up HDL-C than in those with high follow-up HDL-C (table 5, figure 3A). The rate of TLR and TVR was significantly higher in the low follow-up HDL-C group (table 5, figure 3B). In multivariate analysis, low follow-up HDL-C was associated with the significantly higher incidence of MACE, TLR and TVR (table 5).
Figure 3.
Kaplan–Meier curves in propensity matched populations. (A) Kaplan–Meier curves for major adverse cardiac events (MACE) in the propensity matched population with low follow-up high-density lipoprotein cholesterol (HDL-C) (solid line) versus high follow-up HDL-C (dashed line). (B) Kaplan-Meier curves for any revascularisation in the propensity matched population with low follow-up HDL-C (solid line) versus high follow-up HDL-C (dashed line). PCI, percutaneous coronary intervention.
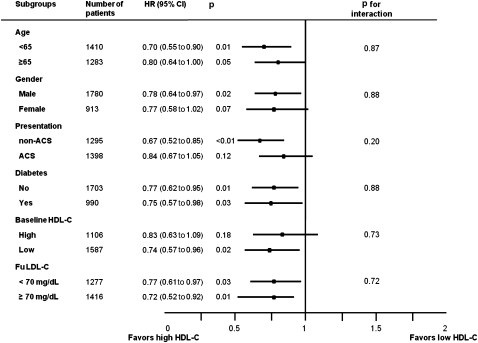
Subgroup analysis
We calculated the unadjusted HR for MACE in various subgroups (figure 4). The rate of MACE was numerically lower in the high follow-up HDL-C group than in the low follow-up HDL-C group in all subgroups, although statistical significance was not found in patients with old age, female gender, ACS presentation and high baseline HDL-C. There were no significant interactions between follow-up HDL-C levels and MACE among the six subgroups. Subgroup analysis in the propensity matched population had similar findings except that statistical significance was found in patients with old age.
Figure 4.
Comparative unadjusted HRs of major adverse cardiac events (MACE) for subgroups. ACS, acute coronary syndrome; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.
Discussion
This study provides evidence that the HDL-C level after adequate application of statin therapy is an important risk factor predicting TLR and TVR in patients with CAD after PCI. The HDL-C level after LDL-C reduction with statin therapy at 7.5 months is a significant predictor even after all other risk factors, including baseline HDL-C, follow-up LDL-C level and follow-up hs-CRP level, have been taken into account.
In a pooled analysis of four statin random controlled trials, substantial reduction of LDL-C accompanied by an approximate increase of 7.5% in HDL-C with statin therapy, was associated with coronary atherosclerotic regression.7 Another study analysed the effect of HDL-C increment after lipid therapy in patients with metabolic syndrome; results showed that HDL-C increasing therapies significantly decrease coronary artery progression and cardiovascular events.12 In patients who received PCI, the HDL-C level before PCI was an independent predictor of in-stent restenosis and first-year mortality.13–15 After carotid artery stenting, post-procedural HDL-C levels predicted stent patency at first year.16 A meta-analysis of statin trials showed that statin treatment has a beneficial impact on TVR.17 Our study is the first toshow the clinical impact of HDL-C levels after statin therapy in patients undergoing PCI.
Our study suggests that achieving certain levels of HDL-C after statin therapy is associated with a diminished rate of TVR. In this study, percentage change in HDL-C from baseline to 7.5 months after statin therapy was not related to cardiovascular events. Interestingly, a follow-up LDL-C level that is currently a major target for secondary prevention after PCI is not a risk factor in this present study. A possible explanation for this that all the patients registered in this study maintained low LDL-C levels (<100 mg/dl). The superiority of HDL-C compared with LDL-C as a predictor in patients with CAD was reported in the Framingham Heart Study and the Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering (MIRACL) trial.3 4 18 In general, the effect of statins on raising levels of HDL-C was modest (5–15%).19 In the total cohort of our study, HDL-C levels increased by an average of 2.0% and LDL-C levels decreased by an average of 28.3% after statin therapy. In the propensity scores matched cohort, HDL-C levels increased by 3.6% and LDL-C levels decreased by an average of 29.7%. This change of LDL-C level is consistent with previous studies, but that of HDL-C is much lower. There was no heterogeneity in the efficacy of follow-up HDL-C among patients in subgroups characterised by various baseline characteristics, including age, gender, presentation of ACS, diabetes mellitus and other cholesterol levels.
The most acceptable explanation for the protective effects of HDL-C on cardiovascular disease is its action to mediate the uptake of peripheral cholesterol and return it to the liver for excretion into the bile and gut, termed ‘reverse cholesterol transport’,20 a process which has been postulated to explain how HDL-C protects against atherosclerosis.21 However, emerging experimental studies have identified that HDL-C modifies endothelial cell adhesion protein expression, inhibits endothelial cell apoptosis, promotes re-endothelialisation, stimulates the production of prostacyclin, decreases platelet aggregability, inhibits LDL-C oxidation, and has anti-inflammatory effects, all of which may contribute to its anti-atherosclerotic properties.22–24
It is not yet established whether very aggressive reductions in LDL-C are enough to offset the increased risk associated with very low serum levels of HDL-C. Previous studies indicated that total cholesterol to HDL ratio could be a target for high risk patients, which could be achieved by more aggressive LDL-C lowering or potentially by increasing HDL-C.6 25–28 A recent meta-analysis of statin therapy reported that statin monotherapy did not alter the correlation between HDL-C level and cardiovascular risk, such that low levels of HDL-C remained significantly and independently associated with increased risk despite statin treatment.29 A substudy of JUPITER showed that HDL-C level was not predictive of residual vascular risk among patients treated with potent statin therapy who attained very low concentrations of LDL-C.30 Although it is unclear why the JUPITER study showed a different result from previous studies, this primary prevention study investigated patients with low to intermediate risk who had never experienced cardiovascular disease or diabetes.
Because of residual cardiovascular risk seen in statin monotherapy, treatment may be intensified with the use of combination therapy aiming at either further reducing the LDL-C level or raising the HDL-C level. It is an important issue because some studies have reported that nearly 80% of statin-treated patients with low LDL-C levels still have low HDL-C levels.31 A recent randomised trial, the ARBITER 6-HALTS study, demonstrated the superiority of extended-release niacin over ezetimibe when it is combined with statin therapy. Niacin therapy led to regression of carotid intima–media thickness and fewer clinical cardiovascular events over 14 months among patients with LDL-C levels of <100 mg/dl and HDL-C levels of <50 or 55 mg/dl. Niacin elevated mean HDL-C levels by 18.4% over the 14 months. The ARBITER 6-HALTS study was not a large sample prospective study, but it implied that raising HDL-C may be the next target to ameliorate the progression of coronary atherosclerosis in the treatment of statin.32 A meta-analysis of 23 trials of various lipid-altering drugs suggested that favourably altered LDL-C and HDL-C might be more effective in preventing cardiovascular events.33
Lipid modulation treatment, reducing LDL-C levels or raising HDL-C levels, requires attention. It is well known that patients taking statins are at increased risk of adverse effects, including liver dysfunction, myopathy, acute renal failure and cataracts. A population-based cohort study has developed and validated new risk prediction algorithms that can be used to identify patients at high risk of adverse effects.34 Clinicians should assess the balance of risks and benefits at the start of statin treatment, and check whether the patients have adverse effects. Meta-regression analysis and the ILLUMINATE study showed an association between HDL-C increase and non-cardiovascular mortality.35 36 The increase in adverse events observed in these studies where HDL-C was considerably elevated could be related to the mechanism of action of torcetrapib rather than the HDL-C increase itself. Raising HDL-C is a potential therapeutic goal for cardiovascular disease prevention, but the impact on non-cardiovascular events remains uncertain.
Our study has some limitations. First, our findings are subject to selection bias and confounding factors because of its non-randomised, observational design. To minimise these biases, we used propensity-score matching, but hidden bias may still remain because of the influence of unmeasured confounders. Our findings should be confirmed by an adequately powered, randomised prospective trial. Second, coronary angiography was analysed qualitatively, not quantitatively. Detailed quantitative coronary analysis may be helpful in further interpreting our findings. Third, the name and dosage of the statins prescribed to the study population were not reported in this study. However, the lipid modulating effect on lowering of LDL-C levels was adequate—it was below 100 mg/dl. Forth, we routinely recommend lifestyle modification, which potentially influenced HDL-C levels. However, non-pharmacological factors such as diet, regular physical exercise, smoking cessation, weight loss and alcohol use were not evaluated precisely.
Conclusions
HDL-C level after statin therapy was an independent risk factor for TVR in patients who underwent PCI with DES. Raising the HDL-C level with statin treatment may be a subsequent goal after achieving target LDL-C levels in patients with coronary artery stents.
Footnotes
Competing interests: None.
Ethics approval: This study was conducted with the approval of the Seoul St Mary's Hospital, Catholic University of Korea.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.LaRosa JC, He J, Vupputuri S. Effect of statins on risk of coronary disease: a meta-analysis of randomized controlled trials. JAMA 1999;282:2340–6 [DOI] [PubMed] [Google Scholar]
- 2.Grundy SM, Cleeman JI, Merz CN, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 2004;110:227–39 [DOI] [PubMed] [Google Scholar]
- 3.Castelli WP, Garrison RJ, Wilson PW, et al. Incidence of coronary heart disease and lipoprotein cholesterol levels. JAMA 1986;256:2835–8 [PubMed] [Google Scholar]
- 4.Gordon T, Castelli WP, Hjortland MC, et al. High density lipoprotein as a protective factor against coronary heart disease. The Framingham Study. Am J Med 1977;62:707–14 [DOI] [PubMed] [Google Scholar]
- 5.Sacks FM, Tonkin AM, Shepherd J, et al. Effect of pravastatin on coronary disease events in subgroups defined by coronary risk factors: the Prospective Pravastatin Pooling Project. Circulation 2000;102:1893–900 [DOI] [PubMed] [Google Scholar]
- 6.Barter P, Gotto AM, LaRosa JC, et al. HDL cholesterol, very low levels of LDL cholesterol, and cardiovascular events. N Engl J Med 2007;357:1301–10 [DOI] [PubMed] [Google Scholar]
- 7.Nicholls SJ, Tuzcu EM, Sipahi I, et al. Statins, high-density lipoprotein cholesterol, and regression of coronary atherosclerosis. JAMA 2007;297:499–508 [DOI] [PubMed] [Google Scholar]
- 8.Cutlip DE, Windecker S, Mehran R, et al. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation 2007;115:2344–51 [DOI] [PubMed] [Google Scholar]
- 9.Weitzen S, Lapane KL, Toledano AY, et al. Principles for modeling propensity scores in medical research: a systematic literature review. Pharmacoepidemiol Drug Saf 2004;13:841–53 [DOI] [PubMed] [Google Scholar]
- 10.D'Agostino RB., Jr Propensity score methods for bias reduction in the comparison of a treatment to a non-randomized control group. Stat Med 1998;17:2265–81 [DOI] [PubMed] [Google Scholar]
- 11.Normand ST, Landrum MB, Guadagnoli E, et al. Validating recommendations for coronary angiography following acute myocardial infarction in the elderly: a matched analysis using propensity scores. J Clin Epidemiol 2001;54:387–98 [DOI] [PubMed] [Google Scholar]
- 12.Zhao XQ, Krasuski RA, Baer J, et al. Effects of combination lipid therapy on coronary stenosis progression and clinical cardiovascular events in coronary disease patients with metabolic syndrome: a combined analysis of the Familial Atherosclerosis Treatment Study (FATS), the HDL-Atherosclerosis Treatment Study (HATS), and the Armed Forces Regression Study (AFREGS). Am J Cardiol 2009;104:1457–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shah PK, Amin J. Low high density lipoprotein level is associated with increased restenosis rate after coronary angioplasty. Circulation 1992;85:1279–85 [DOI] [PubMed] [Google Scholar]
- 14.Sukhija R, Aronow WS, Sureddi R, et al. Predictors of in-stent restenosis and patient outcome after percutaneous coronary intervention in patients with diabetes mellitus. Am J Cardiol 2007;100:777–80 [DOI] [PubMed] [Google Scholar]
- 15.Ghazzal ZB, Dhawan SS, Sheikh A, et al. Usefulness of serum high-density lipoprotein cholesterol level as an independent predictor of one-year mortality after percutaneous coronary interventions. Am J Cardiol 2009;103:902–6 [DOI] [PubMed] [Google Scholar]
- 16.Topakian R, Sonnberger M, Nussbaumer K, et al. Postprocedural high-density lipoprotein cholesterol predicts carotid stent patency at 1 year. Eur J Neurol 2008;15:179–84 [DOI] [PubMed] [Google Scholar]
- 17.Zhang ZJ, Cheng Q, Jiang GX, et al. Statins in prevention of repeat revascularization after percutaneous coronary intervention–a meta-analysis of randomized clinical trials. Pharmacol Res 2010;61:316–20 [DOI] [PubMed] [Google Scholar]
- 18.Olsson AG, Schwartz GG, Szarek M, et al. High-density lipoprotein, but not low-density lipoprotein cholesterol levels influence short-term prognosis after acute coronary syndrome: results from the MIRACL trial. Eur Heart J 2005;26:890–6 [DOI] [PubMed] [Google Scholar]
- 19.Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet 2005;366:1267–78 [DOI] [PubMed] [Google Scholar]
- 20.Glomset JA. The plasma lecithins:cholesterol acyltransferase reaction. J Lipid Res 1968;9:155–67 [PubMed] [Google Scholar]
- 21.Ross R, Glomset JA. Atherosclerosis and the arterial smooth muscle cell: Proliferation of smooth muscle is a key event in the genesis of the lesions of atherosclerosis. Science 1973;180:1332–9 [DOI] [PubMed] [Google Scholar]
- 22.Mineo C, Deguchi H, Griffin JH, et al. Endothelial and antithrombotic actions of HDL. Circ Res 2006;98:1352–64 [DOI] [PubMed] [Google Scholar]
- 23.Barter PJ, Nicholls S, Rye KA, et al. Antiinflammatory properties of HDL. Circ Res 2004;95:764–72 [DOI] [PubMed] [Google Scholar]
- 24.Toth PP. High-density lipoprotein and cardiovascular risk. Circulation 2004;109:1809–12 [DOI] [PubMed] [Google Scholar]
- 25.Frost PH, Davis BR, Burlando AJ, et al. Serum lipids and incidence of coronary heart disease. Findings from the Systolic Hypertension in the Elderly Program (SHEP). Circulation 1996;94:2381–8 [DOI] [PubMed] [Google Scholar]
- 26.Lamarche B, Després JP, Moorjani S, et al. Triglycerides and HDL-cholesterol as risk factors for ischemic heart disease. Results from the Québec cardiovascular study. Atherosclerosis 1996;119:235–45 [DOI] [PubMed] [Google Scholar]
- 27.Manninen V, Tenkanen L, Koskinen P, et al. Joint effects of serum triglyceride and LDL cholesterol and HDL cholesterol concentrations on coronary heart disease risk in the Helsinki Heart Study. Implications for treatment. Circulation 1992;85:37–45 [DOI] [PubMed] [Google Scholar]
- 28.Wang TD, Chen WJ, Chien KL, et al. Efficacy of cholesterol levels and ratios in predicting future coronary heart disease in a Chinese population. Am J Cardiol 2001;88:737–43 [DOI] [PubMed] [Google Scholar]
- 29.Jafri H, Alsheikh-Ali AA, Karas RH. Meta-analysis: statin therapy does not alter the association between low levels of high-density lipoprotein cholesterol and increased cardiovascular risk. Ann Intern Med 2010;153:800–8 [DOI] [PubMed] [Google Scholar]
- 30.Ridker PM, Genest J, Boekholdt SM, et al. HDL cholesterol and residual risk of first cardiovascular events after treatment with potent statin therapy: an analysis from the JUPITER trial. Lancet 2010;376:333–9 [DOI] [PubMed] [Google Scholar]
- 31.Alsheikh-Ali AA, Lin JL, Abourjaily P, et al. Prevalence of low high-density lipoprotein cholesterol in patients with documented coronary heart disease or risk equivalent and controlled low-density lipoprotein cholesterol. Am J Cardiol 2007;100:1499–501 [DOI] [PubMed] [Google Scholar]
- 32.Taylor AJ, Villines TC, Stanek EJ, et al. Extended-release niacin or ezetimibe and carotid intima-media thickness. N Engl J Med 2009;361:2113–22 [DOI] [PubMed] [Google Scholar]
- 33.Brown BG, Stukovsky KH, Zhao XQ. Simultaneous low-density lipoprotein-C lowering and high-density lipoprotein-C elevation for optimum cardiovascular disease prevention with various drug classes, and their combinations: a meta-analysis of 23 randomized lipid trials. Curr Opin Lipidol 2006;17:631–6 [DOI] [PubMed] [Google Scholar]
- 34.Hippisley-Cox J, Coupland C. Individualising the risks of statins in men and women in England and Wales: population-based cohort study. Heart 2010;96:939–47 [DOI] [PubMed] [Google Scholar]
- 35.Burillo E, Andres EM, Mateo-Gallego R, et al. High-density lipoprotein cholesterol increase and non-cardiovascular mortality: a meta-analysis. Heart 2010;96:1345–51 [DOI] [PubMed] [Google Scholar]
- 36.Barter PJ, Caulfield M, Eriksson M, et al. Effects of torcetrapib in patients at high risk for coronary events. N Engl J Med 2007;357:2109–22 [DOI] [PubMed] [Google Scholar]