Abstract
Purpose
Neurally adjusted ventilatory assist (NAVA) relies on the patient’s electrical activity of the diaphragm (EAdi) for actuating the ventilator. Thus a reliable positioning of the oesophageal EAdi catheter is mandatory. We aimed to evaluate the effects of body position (BP), positive end-expiratory pressure (PEEP) and intra-abdominal pressure (IAP) on catheter positioning.
Methods
Twenty-one patients were enrolled in this study. In six different situations [supine or 45° head of bed elevation (HBE) at PEEP 5 and 15 cmH2O; left lateral anti-decubitus at PEEP 5 cmH2O; supine at PEEP 5 cmH2O with abdominal surgical belt (ASB)] the catheter position was evaluated for the stability of the EAdi signal and information provided by a catheter positioning tool (highlighted electrical activity in central leads, absence of p waves in the distal lead).
Results
With an optimal catheter position EAdi signals were stable for all tested situations. During “45° PEEP 15” and “supine PEEP 15” absence of p waves in the distal lead revealed a difference compared with “supine PEEP 5” (p = 0.03), suggesting a caudal shift of the diaphragm relative to the oesophagus. The analysis of the highlighted electrical activity in the central leads supports this finding, revealing an influence of PEEP, BP and IAP on EAdi catheter position (p < 0.01).
Conclusion
PEEP, BP and IAP may affect the EAdi catheter position, although not compromising a stable signal. Additional information as provided by the catheter positioning tool is needed to ensure an optimal EAdi catheter position.
Keywords: Neurally adjusted ventilatory assist (NAVA), Body position, PEEP, Intra-abdominal pressure, Electrical activation of the diaphragm (EAdi)
Introduction
During neurally adjusted ventilatory assist (NAVA) the ventilator is triggered by the electrical activity of the diaphragm (EAdi), assessed by a special gastric tube (EAdi catheter) [1–5]. Pressure assistance is applied in proportion to the amplitude of the EAdi, which represents the respiratory effort of the patient controlled by his respiratory centre [6, 7]. The EAdi catheter positioning is crucial because NAVA relies on a representative signal from the diaphragm. Recently we evaluated a method to determine the optimal position of the EAdi catheter based on the quality and amplitude of the EAdi signal and the trans-oesophageal electrocardiogram (ECG) [8]. Positive end-expiratory pressure (PEEP), body position (BP) and intra-abdominal pressure (IAP) were not considered at that time, although these are factors known to influence the position of the diaphragm [9–11]. Therefore we aimed to determine their effect on the EAdi signal and the optimal EAdi catheter position.
Methods
After approval from the local ethics committee (University of Goettingen, Germany) NAVA was initiated on the basis of the clinical decision of the consultant in charge of the intensive care unit (ICU) with the intention to facilitate respirator weaning. We used a ventilator capable of NAVA (Servo-i, Maquet Critical Care, Solna, Sweden). EAdi catheter placement (16 Fr diameter, 125 cm long; Maquet Critical Care, Solna, Sweden) was performed by the study group. Patients were supine with the head of the bed elevated at 30° and pressure support ventilation with preset PEEP was used during EAdi catheter positioning. The catheter was inserted nasally to a maximum distance of 80 cm. For EAdi measurement it is mounted with an electrode array of one reference electrode and eight measuring electrode rings on the distal part of the catheter at intervals of 16 mm, starting at 120 mm from the tip.
Catheter positioning was monitored by a special tool implemented in the ventilator that displays four raw leads of electrical activity representing cranial to caudal arranged electrode pairs. The catheter positioning tool identifies the electromyographic (EMG) signal with the highest amplitude and highlights it in blue [12]. The software highlights two adjacent leads unless only the outermost pair of electrodes detects a diaphragmatic EMG signal. In addition the position of the electrodes in relation to the heart can be estimated by evaluating the presence/absence of p wave (if present) and QRS complex in different leads. Further processing of the signal (e.g. low pass filter to remove ECG) results in the EAdi signal in microvolts (μV, analogue to potential difference between electrodes) used to control the ventilator during NAVA. During the placement procedure the catheter was extracted in steps of 1 cm. At each step data were recorded over 60 s using special software (NAVA tracker, Maquet Critical Care, Solna, Sweden). The maximum EAdi value of each breathing cycle during tidal breathing was taken to calculate the median EAdimax for each step. The catheter position was considered optimal (OPT) when the highlighted EAdi appeared in the central leads, the p wave was absent in the distal lead and a stable EAdimax was found [8]. For each patient the catheter placement procedure was randomly repeated for the following six situations without completely removing/reinserting it:
Supine or 45° head of bed elevation (HBE) at PEEP 5 and 15 cmH2O
Supine, PEEP 5 cmH2O, with abdominal surgical belt (ASB)
Left lateral anti-decubitus, PEEP 5 cmH2O
The measures for catheter positioning (highlighted EAdi in the central leads, absent p wave in the distal lead, stable EAdimax) were evaluated over a range of ±5 cm from OPT.
The intervention-related catheter positions were referenced to OPT. During data analysis atrial activity was taken into account.
Urinary bladder pressure was measured after retrograde filling with 50 mL of normal saline in supine position at PEEP 5, with and without ASB.
Results are displayed as median with 25/75 percent quartiles (Q). Wilcoxon matched pairs and analysis of variance (ANOVA) were used to compare between groups.
Results
EAdi catheter placement was possible in all 21 patients (see Table 1 for patient characteristics).
Table 1.
Basic patient characteristics including the catheter position considering data from the positioning tool (OPT), intra-abdominal pressure measured with the patient supine and PEEP set to 5 cmH2O (IAP) and intra-abdominal pressure with the abdominal surgical belt in place (IAP ASB)
| No. | Gender | Diagnosis | Rhythm | Age (years) | Height (cm) | BMI | OPT (cm) | IAP (cmH2O) | IAP ASB (cmH2O) | PEEP (cmH2O) | EAdimax (μV) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | M | IAH (CHE) | SR | 67 | 160 | 25 | 61 | 25 | MD | 8 | 2.7 |
| 2 | F | SAH | SR | 69 | 160 | 16 | 62 | 6 | 10 | 5 | 3.3 |
| 3 | M | Sepsis | Afib | 71 | 178 | 39 | 67 | 14 | 18 | 8 | 8.3 |
| 4 | M | CHD (ACB) | SR | 78 | 175 | 29 | 66 | 13 | 16 | 5 | 2.1 |
| 5 | M | Aflut | Afib | 70 | 178 | 25 | 68 | 11 | 14 | 6 | 11.8 |
| 6 | F | CHD (CAB) | SR | 76 | 152 | 22 | 61 | 8 | 18 | 5 | 3.9 |
| 7 | M | AS (AVR) | Afib | 73 | 174 | 27 | 69 | 10 | 18 | 5 | 4.4 |
| 8 | M | CHD (CAB) | SR | 57 | 170 | 26 | 70 | 5 | 18 | 5 | 1.2 |
| 9 | F | ACS (CAB) | SR | 77 | 164 | 22 | 63 | 9 | 18 | 8 | 7.3 |
| 10 | F | AS (AVR) | Afib | 74 | 168 | 27 | 69 | 8 | 18 | 6 | 4.2 |
| 11 | F | Pneumonia | Afib | 82 | 165 | 37 | 67 | 10 | 14 | 10 | 5.6 |
| 12 | F | TBI | SR | 81 | 170 | 28 | 63 | 13 | 18 | 7 | 7.3 |
| 13 | F | SAH | SR | 39 | 170 | 22 | 57 | 9 | 15 | 6 | 3.2 |
| 14 | M | CHD (CAB) | SR | 60 | 178 | 25 | 66 | 9 | 14 | 6 | 13.1 |
| 15 | M | AS (AVR) | SR | 82 | 165 | 33 | 68 | 14 | 19 | 10 | 3.3 |
| 16 | M | AS (AVR) | Afib | 82 | 170 | 22 | 68 | 5 | 18 | 5 | 19.8 |
| 17 | M | CHD (CAB) | SR | 66 | 172 | 31 | 67 | 12 | 14 | 8 | 2.0 |
| 18 | F | ACS (CAB) | SR | 70 | 160 | 21 | 66 | 7 | 18 | 5 | 1.5 |
| 19 | M | pneumonia | SR | 84 | 180 | 25 | 67 | 13 | 19 | 10 | 3.7 |
| 20 | M | ACS (CAB) | SR | 69 | 174 | 30 | 69 | 17 | 21 | 5 | 1.6 |
| 21 | M | Pneumonia | SR | 65 | 175 | 27 | 69 | 8 | 18 | 8 | 6.7 |
| Median | 71 | 170 | 26 | 67 | 10* | 18* | 6 | 3.9 | |||
| Q25/Q75 | 67/78 | 165/175 | 22/29 | 63/68 | 8/13 | 15/18 | 5/8 | 2.7/7.3 | |||
No. number of patient, BMI body mass index, NEX mod catheter position calculated according to the modified “nose–ear–xiphoid” formula, OPT optimal catheter position, IAP intra-abdominal pressure, ASB abdominal surgical belt, M male, F female, IAH intra-abdominal hypertension, CHE cholecystectomy, SAH subarachnoid haemorrhage, CHD coronary heart disease, CAB coronary artery bypass, Aflut atrial flutter, AS aortic valve stenosis, AVR aortic valve replacement, ACS acute coronary syndrome, TBI traumatic brain injury, SR sinus rhythm, Afib atrial fibrillation, MD missing data (no ASB applied due to IAH)
*p < 0.001 (IAP compared to IAP ASB)
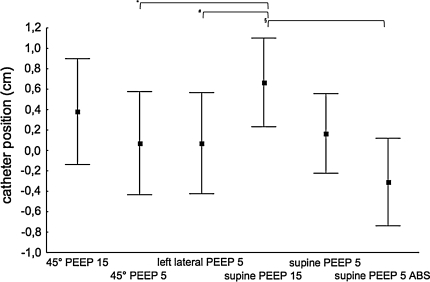
Over all six situations we found a stable EAdimax from −5 cm to OPT to 5 cm with a trend to weaker signals at −5 cm (p = 0.12). The central leads of the catheter positioning tool were highlighted in more than 95% of patients at OPT in all tested situations, but a shift of the highlighted central leads could be detected in all 21 patients (Fig. 1). The tested situations influenced the optimal catheter position (p < 0.01).
Fig. 1.
Optimal catheter positions determined by “highlighted central leads” displayed as mean (filled squares) with 95% confidence interval (whiskers) for six different situations; y-axis catheter position in cm from OPT. Significant differences in post hoc paired comparison (Tukey–Kramer) for 45° PEEP 5 vs supine PEEP 15 (*p = 0.002), left lateral PEEP 5 vs supine PEEP 15 (# p = 0.006) and supine PEEP 15 vs supine PEEP 5 ASB (§ p = 0.0005) (n = 21)
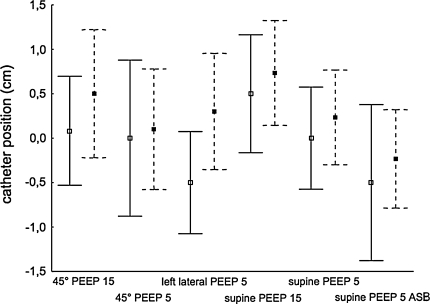
P waves could be evaluated in 15 patients (six patients with atrial fibrillation). The OPT condition “absence of p wave in distal lead” was fulfilled at OPT during “left lateral PEEP 5” and “supine PEEP 5” in 93%, during “supine PEEP 5 ASB” in 87%, “45° PEEP 5” in 80% and “45° PEEP 15” and “supine PEEP 15” in 53% of the patients (p = 0.03 compared to “supine PEEP 5”). With the EAdi catheter at OPT + 1 cm, p waves were absent in 93% (“45° PEEP 15”) and 87% (“supine PEEP 15”) of patients. Comparing patients with sinus rhythm and atrial fibrillation neither a main effect of heart rhythm (p = 0.41) nor an interaction effect with the tested situations (p = 0.57) could be found (Fig. 2).
Fig. 2.
Optimal catheter positions determined by “highlighted central leads” for patients with sinus rhythm (filled squares and dashed lines; n = 15) and atrial fibrillation (open squares and solid lines; n = 6) displayed as mean (squares) with 95% confidence interval (whiskers) for six different situations; y-axis catheter position in cm from OPT
Considering EAdimax in patients with atrial fibrillation, we could not determine a difference between catheter positions in the evaluated range (±5 cm to OPT; p = 0.44). In patients with sinus rhythm the EAdimax changed (p = 0.04) with smaller values at −5 cm.
Discussion
NAVA relies on a stable EAdi signal for respirator control. Therefore correct placement of the EAdi catheter is mandatory. The EAdi catheter derives the signal from the part of the diaphragm close to the oesophagus. The position of the diaphragm depends on the application of PEEP, BP and IAP [9–11]. Six different situations with regard to PEEP, BP and IAP were evaluated and their effects on the catheter position relative to the diaphragm via the information provided by the catheter positioning tool (highlighted central leads and absence of p waves in the distal lead) were found. The changes of the EAdi signal were not significant comparing catheter positions from −5 to OPT to +5 cm and would not have impaired running the NAVA mode. The electrode array on the EAdi catheter is 14.4 cm wide and the ventilator software always uses the diaphragmatic EMG with the highest amplitude found for calculating the EAdi signal used for NAVA. Thus even with a catheter that is displaced ±5 cm from OPT there are still electrodes close to the diaphragm, thereby ensuring a constant signal quality. This result is in accordance with our previous study and demonstrates the limits of safety implemented in the EAdi signal acquisition [8]. EAdimax therefore is only suitable to detect large changes in the EAdi catheter position relative to the diaphragm.
Further limitations have to be considered when looking at our results. Our measurement technique relies on the detection of the electromyographic activity of the diaphragmatic tunnel covering 2–3 cm of the oesophagus [13] and the assumption that the signal originates from its centre [12]. The accuracy of measurement is limited by the electrode distance of 16 mm. Nevertheless we were able to demonstrate changes.
The evaluation of the p waves revealed a difference between “supine PEEP 5” and “45° PEEP 15” and “supine PEEP 15”. The fact that the distal lead still had proximity to the right atrium in both PEEP 15 situations may be explained by the fact that the increase in PEEP displaces the diaphragm and thoracic content, including the heart, to a more caudal position. Froese and Byran [10] reported that a PEEP level of 10 cmH2O results in a 2-cm caudal shift of the middle part of the diaphragm compared with zero PEEP. Comparing patients with and without p waves we did not find a statistically significant difference although Fig. 2 suggests a shift to deeper catheter positions in patients with sinus rhythm. Figure 2 considers the highlighted central leads only and not the additional information provided by the p waves. Therefore the figure allows the interpretation that OPT was determined at a more cranial position in patients with sinus rhythm compared with patients with atrial fibrillation. This observation is confirmed by the smaller EAdimax at −5 cm from OPT in patients with sinus rhythm but not in patients with atrial fibrillation. A possible explanation lies in the catheter positioning procedure that defined OPT (pulling the catheter out stepwise) and the different prioritization of the variables (first, EAdi highlighted in central leads; second, p waves absent in the distal lead; third, stable EAdimax). Although we consider only a small collective it might be assumed by the evaluation of the EAdimax data that catheter positioning was more accurate in patients in whom p waves were unavailable.
The analysis of the highlighted central leads reveals effects of PEEP, BP and IAP on the diaphragm, with a high PEEP displacing it caudally and an increased IAP displacing it to a more cranial position (Fig. 1). The changes are consistent over all tested situations and similar to the results described above regarding p waves. A 30° HBE increases IAP by 3.7 mmHg [14] and a 45° HBE increases IAP by 9 mmHg [15]. This might explain why the caudal shift in “supine PEEP 15” seems to be more pronounced than in “45° PEEP 15”. Furthermore it explains a caudal shift in “supine PEEP 5” as OPT was established at 30° HBE at a median PEEP of 6 cmH2O.
Conclusion
PEEP, BP and IAP affect the optimal EAdi catheter position. NAVA ventilation is not affected due to the wide electrode array compensating for the small diaphragmatic shift seen with such changes. However this only holds true if an optimal catheter position is ensured in advance.
A verification of the optimal catheter position should be performed after major changes in ventilator settings, patient positioning or clinical condition e.g. increases of abdominal pressure. Catheter positioning is not impaired by the lack of p waves.
Acknowledgments
We thank Katharina Lange, Dep. Medical Statistics, Georg-August University of Göttingen, Germany for her support.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Sinderby C, Navalesi P, Beck J, Skrobik Y, Comtois N, Friberg S, Gottfried SB, Lindstrom L. Neural control of mechanical ventilation in respiratory failure. Nat Med. 1999;5:1433–1436. doi: 10.1038/71012. [DOI] [PubMed] [Google Scholar]
- 2.Colombo D, Cammarota G, Bergamaschi V, De Lucia M, Corte FD, Navalesi P. Physiologic response to varying levels of pressure support and neurally adjusted ventilatory assist in patients with acute respiratory failure. Intensive Care Med. 2008;34:2010–2018. doi: 10.1007/s00134-008-1208-3. [DOI] [PubMed] [Google Scholar]
- 3.Brander L, Leong-Poi H, Beck J, Brunet F, Hutchison SJ, Slutsky AS, Sinderby C. Titration and implementation of neurally adjusted ventilatory assist in critically ill patients. Chest. 2009;135:695–703. doi: 10.1378/chest.08-1747. [DOI] [PubMed] [Google Scholar]
- 4.Allo JC, Beck JC, Brander L, Brunet F, Slutsky AS, Sinderby CA. Influence of neurally adjusted ventilatory assist and positive end-expiratory pressure on breathing pattern in rabbits with acute lung injury. Crit Care Med. 2006;34:2997–3004. doi: 10.1097/01.CCM.0000242520.50665.9F. [DOI] [PubMed] [Google Scholar]
- 5.Beck J, Campoccia F, Allo JC, Brander L, Brunet F, Slutsky AS, Sinderby C. Improved synchrony and respiratory unloading by neurally adjusted ventilatory assist (NAVA) in lung-injured rabbits. Pediatr Res. 2007;61:289–294. doi: 10.1203/01.pdr.0000257324.22406.93. [DOI] [PubMed] [Google Scholar]
- 6.Moerer O, Beck J, Brander L, Costa R, Quintel M, Slutsky AS, Brunet F, Sinderby C. Subject-ventilator synchrony during neural versus pneumatically triggered non-invasive helmet ventilation. Intensive Care Med. 2008;34:1615–1623. doi: 10.1007/s00134-008-1163-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Moerer O, Barwing J, Quintel M. Neurally adjusted ventilatory assist (NAVA). A new mode of assisted mechanical ventilation. Anaesthesist. 2008;57:998–1005. doi: 10.1007/s00101-008-1412-0. [DOI] [PubMed] [Google Scholar]
- 8.Barwing J, Ambold M, Linden N, Quintel M, Moerer O. Evaluation of the catheter positioning for neurally adjusted ventilatory assist. Intensive Care Med. 2009;35:1809–1814. doi: 10.1007/s00134-009-1587-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Malbrain ML. Is it wise not to think about intra-abdominal hypertension in the ICU? Curr Opin Crti Care. 2004;10:132–145. doi: 10.1097/00075198-200404000-00010. [DOI] [PubMed] [Google Scholar]
- 10.Froese AB, Bryan AC. Effects of anesthesia and paralysis on diaphragmatic mechanics in man. Anesthesiology. 1974;41:242–255. doi: 10.1097/00000542-197409000-00006. [DOI] [PubMed] [Google Scholar]
- 11.Mutoh T, Lamm WJ, Embree LJ, Hildebrandt J, Albert RK. Abdominal distension alters regional pleural pressures and chest wall mechanics in pigs in vivo. J Appl Physiol. 1991;70:2611–2618. doi: 10.1152/jappl.1991.70.6.2611. [DOI] [PubMed] [Google Scholar]
- 12.Beck J, Sinderby C, Lindström L, Grassino A. Influence of bipolar esophageal electrode positioning on measurements of human crural diaphragm electromyogram. J Appl Physiol. 1996;81:1434–1449. doi: 10.1152/jappl.1996.81.3.1434. [DOI] [PubMed] [Google Scholar]
- 13.Delattre JF, Palot JP, Ducasse A, Flament JB, Hureau J. The crura of the diaphragm and diaphragmatic passage. Applications to gastroesophageal reflux, its investigation and treatment. Anat Clin. 1985;7:271–283. doi: 10.1007/BF01784644. [DOI] [PubMed] [Google Scholar]
- 14.Cheatham ML, De Waele JJ, De Laet I, De Keulenaer B, Widder S, Kirkpatrick AW, Cresswell AB, Malbrain M, Bodnar Z, Mejia-Mantilla JH, Reis R, Parr M, Schulze R, Puig S. The impact of body position on intra-abdominal pressure measurement: a multicenter analysis. Crit Care Med. 2009;37:2187–2190. doi: 10.1097/CCM.0b013e3181a021fa. [DOI] [PubMed] [Google Scholar]
- 15.De Keulenaer BL, De Waele JJ, Powell B, Malbrain M. What is normal intra-abdominal pressure and how is it affected by positioning, body mass and positive end-expiratory pressure? Intensive Care Med. 2009;35:969–976. doi: 10.1007/s00134-009-1445-0. [DOI] [PubMed] [Google Scholar]