Abstract
Ross River virus (RRV) and Semliki Forest virus (SFV) are two alphaviruses that have a high degree of amino acid homology, as well as a very broad host range. We show here that envelope glycoproteins derived from both viruses can pseudotype human immunodeficiency virus type 1 (HIV-1)-derived lentivirus vectors. Both RRV and SFV glycoproteins considerably expand the host range of the lentivirus vector, and vectors can be efficiently concentrated by ultracentrifugation. A systematic analysis comparing the alphaviral glycoproteins to the vesicular stomatitis virus glycoprotein (VSV-G) revealed that lentivirus vectors incorporate RRV glycoproteins with an efficiency comparable to that of VSV-G. Both pseudotypes have comparable physical titers, but infectious titers with the RRV pseudotype are lower than with VSV-G. Incorporation of SFV glycoproteins into lentivirus vector is less efficient, leading to decreased physical and infectious titers. The transduction rates with VSV-G-, RRV-, and SFV-pseudotyped lentivirus vectors into adherent cell lines can be significantly increased by using a combination of Polybrene and plates coated with CH-296 recombinant fibronectin fragments. Together, our data suggest that RRV and SFV glycoproteins might be suitable as alternatives to VSV-G for pseudotyping lentivirus vectors.
Lentivirus vectors are becoming increasingly popular tools in the search for efficient gene delivery systems. A major advantage is their apparent ability to transduce genes into nondividing target cells (34). The lentivirus human immunodeficiency virus type 1 (HIV-1) has envelope glycoproteins that bind to specific receptors that mediate entry into a restricted population of target cells, generally CD4+ T cells and macrophages (16). Glycoproteins from other viruses can be incorporated into the lentiviral envelope, changing the host range of the lentivirus vector, a process known as pseudotyping (3, 28-30, 37). At present, most investigators generate a lentivirus vector pseudotype with the vesicular stomatitis virus glycoprotein (VSV-G). This glycoprotein has few species restrictions and allows the lentivirus vector to transduce most cell types. It allows production of high-titer lentivirus vectors, and its physical properties permit concentration of viral particles by centrifugation (34). Despite these attractive features, VSV-G has some properties that may limit its clinical usefulness. It has been shown to induce syncytium formation after prolonged expression on the cell surface, complicating the generation of vector packaging cell lines (5). VSV-G is also recognized by human complement, which may limit its potential for in vivo administration (11, 23, 38).
To explore alternative glycoproteins that may be used to pseudotype lentivirus vectors, we evaluated those of the alphaviruses Ross River virus (RRV) and Semliki Forest virus (SFV). Alphaviruses are a large group of enveloped viruses with an extremely broad host range that ranges from invertebrates to vertebrates (43). The alphaviral genome consists of single-stranded RNA of positive polarity (+) encapsidated in an icosahedral nucleocapsid. The virus-encoded glycoprotein components are embedded in the envelope bilayer, project from it, and form an icosahedral lattice (35). The main part of the viral glycoprotein complex consists of heterodimers of E1 and E2 glycoproteins that are further organized into trimers. A total of 80 of these trimeric spikes are distributed on the outer surface of the virion (7, 17). The E2 glycoprotein most likely mediates interactions with target cell receptors, whereas E1 is thought to mediate fusion (43). The cellular receptors used for entry by RRV and SFV are currently unknown. The broad host range of the wild-type alphaviruses indicates that the viruses might utilize either different receptors on different target cells or a protein receptor that is highly conserved among different species (43). On the other hand, although the precise nature of the cellular receptors for VSV-G has not been clearly identified, membrane lipid molecules, specifically phosphatidylserine, have been shown to be important for VSV-G binding and infection (32, 40).
Glycoproteins derived from SFV have been shown to incorporate into Moloney murine leukemia virus (MuLV) (44). Two recent studies have also demonstrated that RRV glycoproteins can be efficiently incorporated into MuLV and feline immunodeficiency virus (FIV) vectors. The resulting pseudotyped viruses can effectively transduce cells in cell culture and in vivo (27, 41). Furthermore, the RRV E1/E2-pseudotyped MuLV vectors were stable during concentration by ultracentrifugation and could be stably expressed in a vector packaging cell line, without apparent toxicity (41).
In the present study, we show for the first time that the RRV and SFV glycoproteins can be incorporated into HIV-1-derived lentivirus vectors and form infectious pseudotypes. The RRV and SFV glycoprotein-pseudotyped vectors have an expanded host range with comparable affinities for cells as the VSV-G-pseudotyped lentivirus vector. HIV vector pseudotyped by the glycoprotein of either alphavirus can be concentrated by ultracentrifugation. In addition, we conducted a comparative study of the efficiency of incorporation of the VSV, RRV, and SFV glycoproteins into lentivirus vector particles. Our findings indicate that RRV glycoproteins might be an attractive alternative to the commonly used VSV-G protein for developing safe and efficient pseudotyped lentivirus vectors for clinical gene-transfer applications.
MATERIALS AND METHODS
Cells.
The human embryonic fibroblast cell lines 293T and 293, the human fibrosarcoma cell line HT-1080, MRC-5 (human lung fibroblast), and HeLa (human cervical carcinoma), K562 (human myeloid progenitor), BeWo (human choriocarcinoma), HepG2 (human hepatocellular carcinoma), Vero (African green monkey kidney epithelial), and NIH 3T3 (mouse fibroblasts) cells were all obtained from the American Tissue Culture Collection and maintained in D-10 medium (Dulbecco modified Eagle medium (Gibco-BRL, Grand Island, N.J.) supplemented with 10% fetal calf serum (HyClone, Logan, Utah), 2 mM l-glutamine (Gibco-BRL), and 100 U of penicillin and 100 μg of streptomycin (Gibco-BRL)/ml.
Construction of glycoprotein expression plasmids.
Comparable glycoprotein expression plasmids, which differ only in the coding regions, were constructed as follows. To construct the target plasmid pCI-DestB, the Gateway destination cassette RFB (Invitrogen, Carlsbad, Calif.) that had previously been subcloned into pUC19 was digested with the restriction endonucleases XbaI and Asp718 and then ligated into the NheI and Asp718 sites of the pCI plasmid (Promega, Madison, Wis.). Then, the VSV(Indiana)-G expressing plasmid pMD.G (Cell Genesys, Foster City, Calif.) (34) was digested with SnaBI and XbaI, and the fragment containing the VSV-G coding region, together with the rabbit β-globin intron and its poly(A) signal, was ligated into the SnaBI and XbaI sites in the pCI-DestB expression vector. The resulting plasmid was designated pCI-MD.G. To insert the RRV and SFV glycoprotein expression cassettes between the rabbit β-globin control elements, pCI-MD.G was digested with EcoRI, and the free ends were filled in with T4 DNA polymerase (New England Biolabs, Beverly, Mass.). Then, pRRV-E2E1A or pSFV-E2E1A (expressing RRV and SFV glycoproteins, respectively) (41) were digested with PmeI, and the fragment encoding the RRV or SFV glycoproteins was blunt ligated into the pCI-MD.G backbone, replacing the VSV-G coding region. The resulting glycoprotein expression plasmids were designated pCI-RRV and pCI-SFV.
Vector production.
Lentivirus vectors were produced by transient transfection of 293T cells. A total of 5 × 106 cells were seeded in 75-cm2 tissue culture flasks 24 h before transfection in D-10 medium. Cells were refed with fresh D-10 medium 2 h before transfection. Transfection was performed by calcium phosphate precipitation method (Profection kit; Promega) according to the manufacturer's instructions by using as a transfer vector either pRRLsin-CMV-GFP (13.2 μg; Cell Genesys) or pcDNA-HIV-CS-CGW (18 μg [gift from Phil Zoltick, Children's Hospital, Philadelphia, Pa.]; both plasmids encode for the enhanced green fluorescent protein [GFP]), and the lentiviral helper plasmids pMDLg (6.6 μg), and pRSV/Rev (3.3 μg; Cell Genesys). The 293T cells were also cotransfected with the appropriate glycoprotein expression plasmid, expressing either VSV (pMD.G [4.62 μg] or pCI-MD.G [5.13 μg]), RRV (pRRV-E2E1A [6 μg] or pCI-RRV [6.3 μg]), or SFV (pSFV-E2E1A [6 μg] or pCI-SFV [6.3 μg]) glycoproteins. After the cells were refed with fresh medium 16 to 18 h posttransfection, vector-containing supernatants were harvested 48 h after transfection, filtered through a 0.45-μm-pore-size filter, and stored at −80°C. When a concentrated vector was generated, transient transfections were scaled up to 300-cm2 flasks. For each pseudotyped vector, supernatants from two flasks were combined into a Beckman Quick-Seal ultracentrifuge tube (Beckman Coulter, Fullerton, Calif.) holding 94 ml. The supernatants were then spun at 50,000 × g for 1 h in a Beckman ultracentrifuge Optima XL-100k by using a 45-Ti fixed-angle rotor. Virus pellets were resuspended in 1 ml of D-10 and stored at −80°C.
Transduction with pseudotyped lentivirus vectors.
To determine infectious titers of concentrated and unconcentrated vector supernatants, 105 293 cells were transduced in triplicate with 1 ml each of serially diluted lentiviral supernatants plus 8 μg of Polybrene (hexadimethrine-bromide; Sigma, St. Louis, Mo.)/ml for 4 h. At 72 h after infection, cells were harvested and analyzed by flow cytometry for GFP expression (FACscan; Becton Dickinson, San Jose, Calif.). Vector dilutions were identified that showed a linear relationship between dilution of the vector and the number of transduced cells (generally between 1 and 25% GFP-expressing cells). The following formula was used to convert the percentage of GFP-expressing cells for a specific dilution into transducing units (TU): TU/milliliter = [(percent GFP-positive cells/100) × number of cells infected × dilution factor]/supernatant volume (in milliliters) used in transduction. For experiments involving transduction of cell lines other than 293, the cells were transduced under the conditions described above, except that the dilution of vector supernatants was kept constant. To determine the effect of recombinant fibronectin (CH-296 fragment; Takara Shiuzo Co., Ltd., Shiga, Japan) on cell transduction, wells in six-well plates were coated with 4 μg of CH-296/cm2 overnight at 4°C. Wells were washed with phosphate-buffered saline (PBS; Gibco) the next day, and HT-1080 or 293 cells were plated in coated or noncoated wells and transduced with or without addition of 8 μg of Polybrene/ml as described above. The effects of pseudotype, treatment, and the two-way interaction on the percentage of cells transduced were evaluated by using a two-way analysis of variance. Pairwise comparisons were made by using the Sidak adjustment method to control the overall significance level of the tests.
Determination of RNA and p24 titers.
To determine RNA titers, viral RNA was isolated from the supernatants by using the Qiagen RNeasy minikit (Qiagen, Valencia, Calif.), and a selected portion of the lentiviral RNA genome was amplified by quantitative reverse transcriptase PCR (RT-PCR) (TaqMan; ABI Prism 7700 Sequence Detector; Applied Biosystems, Foster City, Calif.) as previously described (39). The samples were quantitated by comparison to a serially diluted plasmid standard (pcDNA-HIV-CS-CGW). To control for DNA plasmid contamination in vector supernatants, each sample was amplified both with RT and without. Quantities obtained without RT were subtracted from the reaction with RT to obtain the final RNA titers. For p24 titers, supernatants were diluted 1:10,000 and assayed by enzyme-linked immunosorbent assay (ELISA; p24 ELISA kit [Beckman Coulter]) according to the manufacturer's instructions.
Western analysis of lentivirus vector particles.
293T producer cells were transiently transfected as described above under vector production. Cells were washed three times with 10 ml of PBS and then lysed with 2 ml of cell lysis buffer (20 mM Tris-Cl [pH 7.4], 0.5 M NaCl. 0.5% NP-40, 0.02% NaN3) at 37°C for 5 min. Cell lysates were centrifuged briefly to remove cell debris and then stored at −80°C. Vector supernatants were spun through a 25% sucrose cushion at 25,000 rpm for 2 h in an SW41 rotor. Samples from both cell lysates and vector supernatants were resuspended in 4× NuPAGE LDS sample buffer (Invitrogen) with reducing agent and then boiled for 10 min. Samples were then subjected to size separation in a sodium dodecyl sulfate-10% polyacrylamide gel electrophoresis precast Bis-Tris gel (Invitrogen). Separated proteins were transferred to a Novex nitrocellulose membrane by electroblotting by using the Xcell II blot module (Invitrogen) according to the manufacturer's instructions. Membranes were blocked with PBS-Tween (0.1% Tween 20) containing 5% powdered milk. Membranes were then incubated with blocking buffer containing either diluted mouse monoclonal anti-VSV glycoprotein antibody (1:20,000, clone P5D4; Sigma), diluted rabbit polyclonal anti-RRV E1 antibody (1:20,000; gift from Richard Kuhn, Purdue University), diluted rabbit polyclonal anti-SFV E1E2 antibody (1:40,000; gift from Margaret Kielian, Albert Einstein College of Medicine), or diluted mouse monoclonal anti-HIV p24 antibody (1:10,000; catalog number 183-H12-5C, obtained through the NIH AIDS Research and Reference Reagent Program, Division of AIDS, National Institute of Allergy and Infectious Disease, National Institutes of Health). After three washes with PBS-Tween, the blots were incubated with 1:40,000 dilutions of secondary antibodies consisting of species-specific horseradish peroxidase-conjugated anti-immunoglobulin G (Amersham, Piscataway, N.J.). After three additional washes, blots were developed by using the ECL-Plus chemiluminescence kit (Amersham), and bands were quantitated by using the Kodak 1D software (Kodak, Rochester, N.Y.).
RESULTS
RRV and SFV glycoproteins pseudotype lentivirus vectors.
To determine whether the glycoproteins from the alphaviruses RRV and SFV would pseudotype lentivirus vectors, we produced vector by transient transfection and compared the transduction efficiencies of RRV and SFV-pseudotyped particles to those of particles pseudotyped with VSV-G. The initial experiments utilized the pRRLsin-CMV-GFP transfer vector, which expresses the GFP; gene transfer in target cells can therefore be assessed by flow cytometric analysis. The plasmid used for VSV-G expression, pMD.G, has been used extensively for pseudotyping lentivirus vectors. The plasmids for RRV and SFV glycoprotein expression, called pRRV-E2E1A and pSFV-E2E1A, respectively, express the alphaviral glycoproteins as a polyprotein that is cleaved to form the E3, E2, 6k, and E1 proteins (41, 43). The plasmids are shown in Fig. 1A. Both the RRV and the SFV glycoproteins have been shown to successfully pseudotype oncoretroviruses and were therefore selected for use in our initial attempts to pseudotype lentivirus vectors (41, 44). In the initial evaluation, vector particles were generated by using an identical amount of glycoprotein expression plasmid with a constant amount of lentiviral packaging (pMDLg and pRSV/Rev) and transfer vector plasmid (pRRLsin-CMV-GFP). Transient transfection was performed by using calcium phosphate transfection, and vector-containing supernatants were harvested after 48 h.
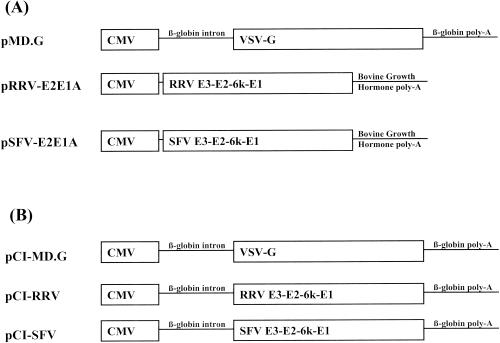
FIG. 1.
Envelope glycoprotein expression plasmids. (A) The original VSV-G expressing plasmid (pMD.G) contains both the rabbit β-globin intron and poly(A) signal. The expression plasmids for RRV and SFV glycoproteins (pRRV-E2E1A and pSFV-E2E1A, respectively) do not have an intron and also have a different poly(A) signal. (B) Standardized glycoprotein expression plasmids were constructed that have both the rabbit β-globin intron and the rabbit β-globin poly(A) signal from pMD.G to standardize expression of VSV, RRV, and SFV glycoprotein expression cassettes.
As shown in Table 1, supernatant harvested from transfections containing the RRV and SFV glycoproteins demonstrates that alphavirus glycoproteins can pseudotype HIV-1-based lentivirus vectors, although the transduction titers of both pseudotypes was below that obtained by using the VSV glycoprotein. As a control, transient transfections were performed with packaging and transfer vectors without the addition of glycoprotein expression plasmid. In this case, there was no transduction of 293 cells, confirming that the alphavirus glycoproteins were responsible for gene transfer. Transduction was also confirmed by using real-time PCR to detect vector sequences in genomic DNA of transduced cells by using our previously published technique (data not shown) (39).
TABLE 1.
Infectious titers of VSV, RRV, and SFV glycoprotein-pseudotyped lentivirus vectorsa
| Glycoproteinb | Mean infectious titer (TU/ml) ± SD |
|---|---|
| VSV (pMD.G) | (1.51 ± 1.47) × 106 |
| RRV (pRRV-E2E1A) | (1.67 ± 0.23) × 105 |
| SFV (pSFV-E2E1A) | (2.23 ± 1.88) × 104 |
Infectious titers were determined on 293 cells. Values represent the means of the results of three independent experiments.
The plasmid used for glycoprotein expression is indicated in parentheses.
RRV and SFV glycoprotein-pseudotyped lentivirus vectors display transduction properties similar to VSV-G pseudotypes.
Lentivirus vectors pseudotyped with the VSV glycoprotein transduce a broader target cell population in comparison to the limited cell populations susceptible to wild-type HIV envelope. To determine whether the RRV and SFV glycoproteins would also allow lentivirus vectors to transduce a variety of cell types, vector preparations were generated by transient transfection and incubated with eight cell lines. Prior to transduction the titers of the vector preparations were determined on 293 cells, and the amount of vector used to transduce the eight cell lines was normalized to a multiplicity of infection (MOI) of 0.2 (one infectious vector particle per five target cells). At this MOI, most transduced cells contain no more than one vector integrant, facilitating an estimate of gene transfer by flow cytometric analysis.
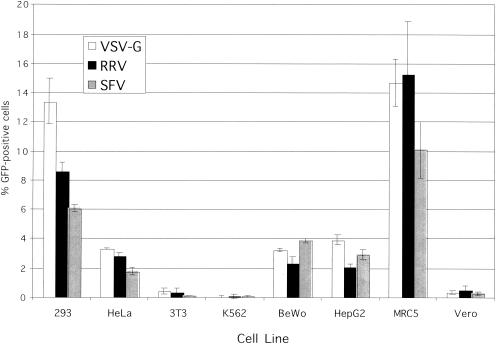
There was little difference in the susceptibility of a particular cell line to transduction by VSV, RRV, or SFV glycoprotein-pseudotyped GFP vectors (Fig. 2). There was considerable variability between the cell lines in the efficiency of lentivirus gene transfer. For the human adherent cell lines tested (HeLa, BeWo, HepG2, and MRC5 cancer cells), the transduction efficiencies of all three pseudotypes were similar to those observed with the 293 cells. Certain cell lines, such as the nonadherent K562 cell line, monkey Vero cells, and the murine NIH 3T3 cell line, showed little evidence of gene transfer at this MOI, regardless of the pseudotype. The variability in gene transfer efficiency among cell lines has been reported for VSV-G, although the mechanisms for this variability are currently unknown (5, 15, 39). Low transduction of Vero cells by VSV-G pseudotyped HIV-1-derived vector has been demonstrated before and is possibly due to an intracellular inhibitor for HIV-1 integration common to Old World monkeys (24). Conversely, the cytomegalovirus (CMV) promoter used to drive GFP transgene expression in the present study has been shown to provide only low-level transgene expression in murine cells transduced with lentivirus vectors (25). Thus, the alphavirus glycoproteins expand the host range of the lentivirus vector in a manner similar to VSV-G.
FIG. 2.
Comparison of transduction efficiencies of VSV, RRV, and SFV glycoprotein-pseudotyped lentivirus vectors for various cell lines. Infectious titers for each pseudotyped vector were first determined on 293 cells, and particles in the supernatant media were then diluted to achieve an MOI of 0.2 for the experiment. Each transduction was performed in triplicate. Error bars represent standard deviation of the mean.
Increased transduction efficiencies with a combination of Polybrene and CH-296 recombinant fibronectin fragments.
A variety of methods have been used to increase the gene transfer efficiency of oncoretroviruses. Polycations such as Polybrene and protamine sulfate have long been known to increase gene transfer efficiency, presumably by facilitating interaction between the negatively charged vector particles and target cells (8, 9, 45). More recently, colocalization of oncoretroviral particles with target cells by using fibronectin has also been shown to increase gene transfer (33, 46) by binding vector particles via a high-affinity heparin binding site and binding cells via VLA-4 and/or VLA-5 binding sites (20).
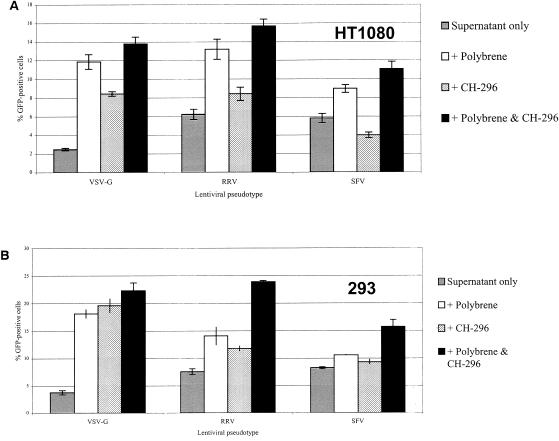
To determine whether polycations or fibronectin fragments might increase the transduction efficiency of pseudotyped lentivirus vectors, we transduced cell lines that demonstrated high- and low-level expression of VLA-4 and VLA-5. Using fluorescein isothiocyanate- and phycoerythrin-conjugated antibodies against VLA-4 and VLA-5, respectively, the HT-1080 human fibrosarcoma line was shown to be a high expresser of both antigens, whereas the 293 cell line demonstrated low expression of these antigens (data not shown). HT-1080 and 293 cells were transduced at an MOI of 0.2 for 4 h. The percentage of GFP-expressing cells was analyzed by fluorescence-activated cell sorting analysis at 72 h posttransduction. As shown in Fig. 3A, Polybrene alone significantly increased the gene transfer efficiency in HT-1080 cells for all three pseudotypes (P < 0.005). The effect of Polybrene was greater than that of the recombinant fibronectin fragment CH-296, which increased gene transfer only with the VSV-G and RRV pseudotypes (P < 0.005). The combination of Polybrene and CH-296 was superior to either agent alone for the RRV- and VSV-G-pseudotyped vectors (P < 0.05).
FIG. 3.
Effect of Polybrene and the fibronectin fragment CH-296 on transduction of cells low or high in expression of VLA-4 and VLA-5 cell adhesion markers. HT-1080 cells (high expresser) (A) and 293 cells (low expresser) (B) were each transduced with VSV-, RRV-, and SFV-glycoprotein pseudotyped lentivirus vectors at an MOI of 0.2 by using various combinations of Polybrene and CH-296. Transductions were performed in triplicate. Error bars represent standard deviation of the mean.
Similarly, in 293 cells the combination of Polybrene and CH-296 was superior to either treatment alone or no treatment (Fig. 3B, P < 0.05). In 293 cells, Polybrene only and CH-296 only treatment groups were not significantly different from each other at P = 0.05, whereas in HT-1080 cells treatment with Polybrene above led to higher transduction levels than seen with CH-296 alone (P < 0.001). For the SFV pseudotype, either Polybrene or fibronectin alone was not significantly better than the control but, together, they enhanced gene transfer.
Optimized production of lentivirus pseudotypes.
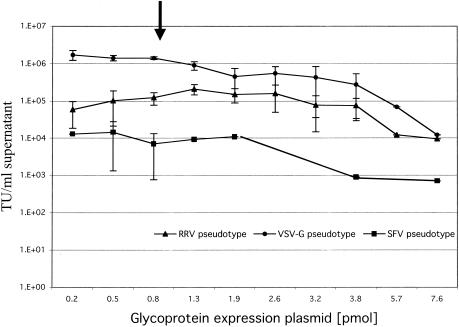
Despite the structural differences between the alphavirus glycoproteins and VSV-G, our initial observations demonstrate similarities in their ability to transduce a variety of cell lines and their gene transfer potential in the presence of polycations and CH-296. The major difference we observed related to the titer of vector produced by transient transfection (Table 1). To evaluate this further, the concentration of glycoprotein plasmid used in vector production was analyzed. Our initial experiments utilized concentrations of glycoprotein, transfer, and packaging plasmids optimized for production of VSV-G-pseudotyped vector. It is known that efficient incorporation of heterologous proteins, specifically SFV glycoproteins, into oncoretrovirus vector particles is only possible when the heterologous proteins are expressed at high densities at the cell surface (44). To determine whether the lower titers observed for RRV and SFV glycoprotein-pseudotyped vectors were due to suboptimal concentrations of glycoprotein expression plasmids, transient transfections were performed by using a range of glycoprotein plasmid concentrations. As shown in Fig. 4, varying the amount of VSV, RRV, and SFV glycoprotein plasmids between 0.2 and 7.6 pmol (corresponding to a 0.3- to 10-fold change in the original plasmid amounts) did not significantly improve vector titer. We found that infectious titers of all three pseudotyped vectors were fairly constant, except at the higher plasmid concentrations when the titers markedly decreased (Fig. 4). Titers of RRV and SFV glycoprotein-pseudotyped vectors at all glycoprotein plasmid dilutions were always lower than the titers of the VSV-G pseudotype. Therefore, the amount of glycoprotein plasmid used in transient-transfection assays was not a major factor in the different titers observed with the various alphaviral glycoproteins.
FIG. 4.
Effect of transfecting various amounts of VSV, RRV, and SFV glycoprotein expression plasmids on the production of pseudotyped lentivirus vectors. Pseudotyped GFP lentivirus vectors were generated by transient transfection with various amounts of VSV, RRV, and SFV glycoprotein expression plasmids, and then the titers were determined on 293 cells. The arrow indicates the amount of glycoprotein plasmid initially used for vector production (see Table 1). Points without error bars represent the mean value of triplicate determinations in a single experiment. Values with error bars represent two to four separate experiments; the mean of triplicate samples was determined in each experiment, and the mean was then used to calculate the standard deviation (shown as error bars).
Another potential variable that may influence vector production is the level of glycoprotein expression. In our initial experiments, VSV-G was expressed from the plasmid pMD.G, which contains a β-globin intron that is thought to increase RNA stability and thereby protein expression (4). The RRV and SFV glycoproteins were expressed from the pcDNA3.1-Zeo(+) backbone that does not contain this intron (41). To standardize the vector backbone and regulatory elements driving glycoprotein expression, the VSV, RRV, and SFV glycoprotein expression cassettes were subcloned into the high-copy plasmid backbone pCI-DestB (Fig. 1b). The rabbit β-globin intron and the poly(A) addition signal from the VSV-G expression vector pMD.G were used in all three constructs. We expected that the inclusion of the β-globin intron would result in significantly higher expression levels for RRV and SFV glycoproteins. Surprisingly, infectious titers for all three pseudotyped vectors dropped compared to titers obtained with the original plasmid backbones (compare the results in Table 2 with those in Table 1). Since all three pseudotypes had lower titers, this suggests that the pCI backbone was responsible for the modestly lower titers. Interestingly, the RRV titers decreased less than VSV-G, suggesting that the original plasmid constructs may have played a partial role in the titer differences observed. Therefore, we believe that the standardization of plasmid backbone and regulatory elements is important, and the pCI-based glycoprotein expression plasmids were utilized in subsequent experiments.
TABLE 2.
Effect of standardized glycoprotein expression and type of transfer vector used on infectious titersa
| Glycoproteinb | Mean infectious titer (TU/ml) ± SD with transfer vector:
|
|
|---|---|---|
| pRRLsinCMV-GFP | pcDNA-HIV-CS-CGW | |
| VSV (pCI-MD.G) | (4.6 ± 1.6) × 105 | (3.4 ± 1.6) × 106 |
| RRV (pCI-RRV) | (9 ± 3) × 104 | (9 ± 2) × 105 |
| SFV (pCI-SFV) | (8 ± 6) × 103 | (5 ± 0.1) × 104 |
Infectious titers were determined on 293 cells. Values represent the means of the results of three independent experiments.
The plasmid used for glycoprotein expression is indicated in parentheses.
To offset the decrease in titer in the pCI-based plasmid constructs, we investigated the use of a modified transfer vector (pHIV-CS-CGW) that contains the central polypurine tract of HIV and the woodchuck hepatitis virus posttranscriptional regulatory element (48, 49). For the three pseudotyped vectors, the addition of these regulatory elements to the transfer vector led to titers that were up to 10-fold higher (Table 2, right column). This transfer plasmid was therefore used in subsequent experiments.
Incorporation of glycoproteins into the lentivirus vector.
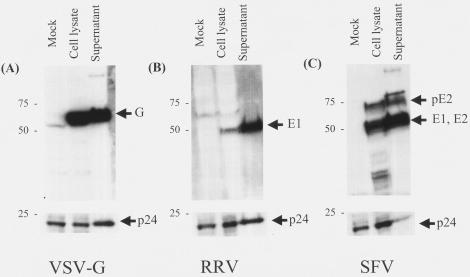
After we determined that the glycoprotein plasmid constructs and their concentration did not play a major role in the titer differences observed, we assessed the efficiency of glycoprotein incorporation into vector particles and the release of particles into the supernatant. Transient transfection of 293T cells was performed and, after 48 h, the amounts of virus glycoproteins were analyzed in vector supernatants and in producer cell lysates. Western blot analysis was performed by using monoclonal antibodies for the VSV glycoprotein, the RRV E1 glycoprotein, and a polyclonal antibody for the SFV glycoproteins. Glycoproteins were detected in both cell lysates and in the vector-particle-containing supernatant medium (Fig. 5). To compare the amount of glycoprotein detected with the number of potential viral particles in the test material, the lower portion of the blot (less than 30 kDa) was probed with a monoclonal antibody specific for the HIV-1 p24 capsid protein. Strikingly, levels of p24 capsid protein were significantly decreased in supernatants of SFV-pseudotyped vector productions compared to the corresponding cell lysate (Fig. 5C). This difference was confirmed by an additional immunoblot analysis of the original vector preparation and analysis of an independently generated preparation of pseudotyped vectors (data not shown).
FIG. 5.
Incorporation of VSV, RRV, and SFV glycoproteins into HIV-1-derived lentivirus vectors. Protein was isolated from VSV-G-, RRV-, and SFV-pseudotyped vector producer cell lysates and corresponding supernatant media. Samples were run on a sodium dodecyl sulfate-10% polyacrylamide gel electrophoresis Bis-Tris gel and then blotted onto a nitrocellulose membrane. Blots were cut into strips and probed with monoclonal antibodies to VSV (A) and RRV E1 (B) glycoproteins and with polyclonal antibodies to SFV glycoproteins (C). Blots were also cut below 30 kDa and probed with a monoclonal antibody against p24 lentivirus capsid protein. The mock lanes are lysates from 293T cells transfected with all lentiviral producer plasmids, except for the glycoprotein expression plasmid. The lanes marked cell lysate and supernatant correspond to transfections that also include the appropriate glycoprotein expression plasmid. Sizes are shown in kilodaltons.
To further characterize the vector particles that are generated after transient transfection, the physical titer of vector particles was compared to the infectious titers. Quantitative RT-PCR amplification of vector RNA was performed to estimate the number of vector genomes per ml of vector supernatant (Table 3). It was observed that the amount of vector RNA was similar between VSV and RRV glycoprotein-pseudotyped vector preparations. This suggests that the lower infectious titers observed with RRV glycoprotein-pseudotyped particles results from their decreased capacity to transduce target cells. Physical titers for SFV glycoprotein-pseudotyped particles were ∼10-fold lower than those for VSV-G-pseudotyped particles, which suggests that the lower infectious titer observed when SFV glycoprotein- and VSV-G-pseudotyped particles (almost 100-fold) are compared is due both to lower vector production and decreased ability to transduce target cells.
TABLE 3.
Physical titers of VSV, RRV, and SFV glycoprotein-pseudotyped lentivirus vectorsa
| Glycoproteinb | Mean p24gag level (ng/ml) ± SD | Mean RNA level (RNA molecules/ml) ± SD |
|---|---|---|
| VSV (pCI-MD.G) | 327 ± 85 | (1.35 ± 0.63) × 1010 |
| RRV (pCI-RRV) | 308 ± 8 | (1.40 ± 0.49) × 1010 |
| SFV (pCI-SFV) | 147 ± 2 | (2.50 ± 0.53) × 109 |
Values represent the means of the results of three independent experiments.
The plasmid used for glycoprotein expression is indicated in parentheses.
The relatively large difference between the infectious titer and physical titer is consistent with previously reported results and probably derives from the large number of defective particles generated during the production process (39). The RNA titers were consistent with physical titers calculated by using the p24 ELISA assay (Table 3). Together, these results suggest that differential incorporation of the respective glycoproteins into lentivirus vectors, as well as excess or virus free envelope which can serve as defective interfering particles, may contribute to the observed titer differences between VSV-G-, RRV-, and SFV-pseudotyped vectors.
RRV and SFV-glycoprotein pseudotyped vectors are stable during ultraconcentration.
In addition to the broad target cell tropism conveyed by VSV-G, the ability of this glycoprotein to survive ultracentrifugation permits the concentration of vector. Sharkey et al. have shown that RRV-pseudotyped MuLV vectors are stable during ultracentrifugation, suggesting that concentration may also be possible with HIV-1-based lentivirus vectors (41). Lentivirus vector supernatants containing VSV, RRV, and SFV glycoprotein-pseudotyped GFP vectors were subjected to ultracentrifugation, as described in Materials and Methods. The RRV and SFV glycoprotein-pseudotyped vectors can be successfully concentrated ∼90-fold (Table 4); this is similar to the concentration achievable with VSV-G-pseudotyped vectors (5). This concentration is accomplished with relatively little loss of infectious particles (yields of 79 and 83% for the RRV and SFV pseudotypes, respectively). These results demonstrate that each of the alphavirus glycoproteins is stable during ultracentrifugation and permit efficient concentration of vector particles pseudotyped with them. For RRV glycoprotein-pseudotyped vectors, ultracentrifugation can generate titers suitable for in vivo use or other applications requiring highly concentrated material.
TABLE 4.
Concentrations of pseudotyped lentivirus vectors as determined by ultracentrifugationa
| Glycoproteinb | Mean titer (TU/ml) ± SD
|
Mean yield (%) ± SD | |
|---|---|---|---|
| Initial | Final | ||
| VSV (pCI-MD.G) | (4.4 ± 1.5) × 106 | (1.8 ± 1.0) × 108 | 48 ± 42 |
| RRV (pCI-RRV) | (7.1 ± 1.5) × 105 | (5.2 ± 1.4) × 107 | 79 ± 16 |
| SFV (pCI-SFV) | (4.7 ± 0.1) × 104 | (5.6 ± 4.0) × 106 | 83 ± 24 |
Initial and final titers were determined before and after ultracentrifugation respectively. Values represent the means of the results of four independent experiments. Yield is the percentage of vector remaining after centrifugation.
The plasmid used for glycoprotein expression is indicated in parentheses.
DISCUSSION
In the present study, we demonstrate that glycoproteins derived from the alphaviruses RRV and SFV can pseudotype lentivirus vectors derived from HIV-1. Our data show that incorporation of either RRV or SFV glycoproteins into pseudotyped lentiviral particles can render them capable of infecting a range of human cell types. The particles are also stable during ultracentrifugation and can be concentrated efficiently and with high yields. Although the titer of vectors pseudotyped with the SFV glycoproteins was consistently lower than that obtained with those pseudotyped with VSV-G, the titer obtained with RRV glycoprotein-pseudotyped HIV-1 vectors is sufficient to warrant further studies to determine the potential of these pseudotypes for animal and human gene therapy applications.
To date, VSV-G has been the glycoprotein of choice for many investigators developing lentivirus vectors, mainly due to its wide host range, high infectious titers, and stability in ultraconcentration protocols. On the other hand, prolonged VSV-G expression has been shown to be toxic to cells (5, 37). Therefore, the construction of stable packaging cell lines with VSV-G has been hindered. We show here that RRV and SFV glycoproteins form infectious pseudotypes with HIV-1 lentiviral particles and that they considerably broaden the host range of the lentivirus vector. Previously, SFV glycoproteins have been shown to incorporate into MuLV particles, but the particles were not evaluated for infectivity (44). Our results provide the first estimates of infectious titers of SFV glycoprotein-pseudotyped retroviruses. We also add HIV to the list of viruses (specifically, MuLV and FIV) pseudotyped by RRV with infectious titers comparable to those obtained with VSV-G (27, 41). In addition, Sharkey et al. have demonstrated successful construction of a stable packaging cell line for RRV-pseudotyped MuLV vector, suggesting that RRV glycoproteins can be stably expressed on the cell surface for long periods of time, without inducing cell toxicity (41). Thus, our results, as well as previous work, reveal characteristics of the RRV glycoproteins that may make them an attractive alternative to VSV-G for pseudotyping lentivirus vectors.
Alphaviruses occur throughout the world, and several members of this large viral family are known to pose a health risk to humans. RRV is endemic to Australia and is responsible for outbreaks of epidemic polyarthritis in humans (21). SFV has been found mainly in West Africa and there caused fever, headaches, arthralgia, and myalgia (26). Both viruses are transmitted by mosquitoes; the primary vertebrate reservoir of RRV seems to be marsupials and other small mammals (43). Despite their different geographic distribution and pathogenicity, the viruses share extensive amino acid identity: 73% in structural and 78% in nonstructural proteins (7). Nevertheless, our results show significant differences in the ability of their glycoproteins to pseudotype HIV-1-derived lentivirus vectors. Using expression plasmids with identical control elements, the infectious titers of RRV and SFV glycoprotein-pseudotyped vectors were about 5-fold and 65-fold lower, respectively, than the titers of vectors pseudotyped with VSV-G.
Western blot analysis revealed differing incorporation efficiencies of the VSV versus the alphaviral glycoproteins. There were only moderate differences in the ratios of glycoproteins to p24 capsid protein when we compared VSV-G- and RRV-pseudotyped vector supernatants. Also, vector particles, as measured by p24 and RNA titer assays, were found to be similar. This suggests that the difference in titer observed may relate to factors at the level of the target cell rather than to vector production. Strikingly, we observed a consistent decrease in p24 capsid protein in supernatants of SFV-pseudotyped vector compared to cell lysates. There could be several possible explanations for this. It is possible that SFV glycoproteins do not incorporate efficiently into lentivirus vector particles, leading to decreased budding of infectious virus vector containing the p24 capsid. Instead, SFV glycoproteins may simply be shed into the supernatant medium, similar to VSV-G, which can be secreted into conditioned medium (1). It has been shown that increased levels of the 4070A glycoprotein in the supernatant medium of pseudotyped MuLV vector can interfere with transduction by presumably blocking receptor binding sites, and SFV glycoproteins may potentially inhibit transduction by a similar mechanism (42). Alternatively, expression of SFV glycoproteins may inhibit the processing or transport of p24 capsid proteins to the sites of virus budding, thus preventing efficient vector particle assembly. Lastly, SFV glycoproteins could be sequestered after secretion by binding glycosaminoglycans on the surface of producer cells. Alphaviruses, such as Sindbis virus, that have been exposed to prolonged passage in tissue culture often acquire the ability to attach to cell surface heparan sulfate. In contrast to SFV, the RRV strain used in the present study has a relatively short passage history and has been shown not to bind heparan sulfate unless mutations are introduced within the RRV glycoproteins (6, 22). Taken together, our data indicates that decreased amounts of capsid protein and viral genomic RNA in supernatants of SFV glycoprotein-pseudotyped vector could partly account for its lower infectious titers.
The efficiency of gene transfer for oncoretrovirus and lentivirus vectors has been enhanced by using a variety of agents that promote interactions between target cells and vector particles (8, 9, 45). A number of studies have demonstrated the utility of polycations and fibronectin fragments to increase transduction with mainly amphotropic or gibbon ape leukemia virus-pseudotyped oncoretrovirus vectors (19, 36). Our results demonstrate that when VSV-G-pseudotyped lentivirus vectors are used, both Polybrene and the fibronectin fragment CH-296 alone can significantly increase transduction into adherent cells. These effects were also seen when RRV glycoproteins were used and were less pronounced when SFV glycoproteins were used, suggesting that the alphavirus glycoproteins may differ somewhat in their net charge and their binding affinity for fibronectin. In all cases, the highest transduction was achieved with both Polybrene and CH-296 in combination. This finding contrasts with studies with oncoretroviruses pseudotyped with the amphotropic envelope, which have shown that Polybrene can actually inhibit the effect of fibronectin fragments on transduction (2, 31). Most investigators have reported that fibronectin does not increase gene transfer into either human CD34+ hematopoietic progenitor cells or human T lymphocytes by VSV-G-pseudotyped lentivirus vector (10, 18, 38, 47), although one study showed increased gene transfer into nonhuman primate CD34+ cells (12). Fibronectin is believed to enhance gene transfer by two mechanisms, (i) the colocalization of target cells with vector particles and (ii) by binding vector particles, thereby increasing their local concentration (19). In our study, the high expression of VLA-4 and VLA-5 did not correlate with higher gene transfer compared to low-expression cells. It may be that the concentrating ability is more important for the pseudotypes tested. Also, VLA-4 and VLA-5 binding sites on fibronectin may be more important for nonadherent cells than the adherent cells used in the present study. Given the variability of benefit from CH-296 reported with lentivirus vectors, it appears that transduction conditions need to be carefully optimized for each pseudotyped vector and target cell.
A recent study using pseudotyped FIV-based lentivirus vectors has demonstrated the ability of RRV glycoproteins to mediate efficient transduction of target cells in vivo (27). When injected into mice, this vector transduced liver cells with greater efficiency and less toxicity than a similar vector pseudotyped with VSV-G. Also, the study demonstrated preferential transduction of neuroglial cells rather than neurons by RRV glycoprotein-pseudotyped FIV in the mouse brain. On the other hand, recombinant SFV vectors have been shown to preferentially transduce neurons in rat hippocampal slice cultures (13, 14). Thus, RRV glycoproteins might be useful for gene transfer into the liver, and both RRV and SFV glycoproteins might have possible applications in the area of neurodegenerative diseases. Recently, we have also noted differences in transduction efficiency of certain target cell populations when we compared VSV-G and alphavirus envelopes. Although VSV-G-pseudotyped HIV vectors can transduce most human hematopoietic cell lines efficiently, RRV and SFV glycoproteins appear to transduce primary and immortalized hematopoietic cells at very low efficiency (C. Kahl et al., Am. Soc. Gene Ther. 6th Annu. Meet., abstr. 90, 2003).
In conclusion, lentivirus vectors pseudotyped with either RRV or SFV glycoproteins can successfully pseudotype HIV-1-derived lentivirus vector and considerably expand its host range. Both are stable enough to permit efficient concentration of vector by ultracentrifugation, and the RRV glycoproteins permit generation of titers sufficient for clinical applications. Since alphavirus glycoproteins can be stably expressed without any visible cytotoxicity, their role in the construction of stable producer cell lines warrants further investigation. Our findings indicate that RRV glycoproteins may serve as an attractive alternative to VSV-G for pseudotyping lentivirus vectors intended for basic and clinical gene transfer applications.
Acknowledgments
We thank Robert Getty and Jing Yao for performing the RNA titer assays and Yi Xu for performing the p24 assays. Also, we thank Lakshmi Sastry for technical assistance. We thank Karen Pollok for assistance with the fibronectin experiments, Richard Kuhn for providing antibodies against the RRV E1 glycoprotein, Margaret Kielian for providing antibodies against the SFV envelope glycoproteins, and Phil Zoltick for providing the pcDNA-HIV-CS-CGW plasmid.
The patent for CH-296 is owned by Indiana University and licensed to Takara Shuzo (Shiga, Japan). None of the authors has any direct financial interest in the material.
The Indiana University Vector Production Facility is an NIH-designated National Gene Vector Laboratory (U42 RR11148), and this study was supported, in part, by a Core Centers of Excellence in Molecular Hematology grant (PHS P50 DK49218) and a core laboratory supporting grant (PHS P01 HL53586).
REFERENCES
- 1.Abe, A., S.-T. Chen, A. Miyanohara, and T. Friedmann. 1998. In vitro cell-free conversion of noninfectious Moloney retrovirus particles to an infectious form by the addition of the vesicular stomatitis virus surrogate envelope G protein. J. Virol. 72:6356-6361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bajaj, B., P. Lei, and S. T. Andreadis. 2001. High efficiencies of gene transfer with immobilized recombinant retrovirus: kinetics and optimization. Bio/Technol. Prog. 17:587-596. [DOI] [PubMed] [Google Scholar]
- 3.Beyer, W. R., M. Westphal, W. Ostertag, and D. von Laer. 2002. Oncoretrovirus and lentivirus vectors pseudotyped with lymphocytic choriomeningitis virus glycoprotein: generation, concentration, and broad host range. J. Virol. 76:1488-1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Buchman, A. R., and P. Berg. 1988. Comparison of intron-dependent and intron-independent gene expression. Mol. Cell. Biol. 8:4395-4405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Burns, J. C., T. Friedmann, W. Driever, M. Burrascano, and J.-K. Yee. 1993. Vesicular stomatitis virus G glycoprotein pseudotyped retroviral vectors: concentration to very high titer and efficient gene transfer into mammalian and nonmammalian cells. Proc. Natl. Acad. Sci. USA 90:8033-8037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Byrnes, A. P., and D. E. Griffin. 1998. Binding of Sindbis virus to cell surface heparan sulfate. J. Virol. 72:7349-7356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cheng, R. H., R. J. Kuhn, N. H. Olson, M. G. Rossmann, H.-K. Choi, T. J. Smith, and T. S. Baker. 1995. Nucleocapsid and glycoprotein organization in an enveloped virus. Cell 80:621-630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Conti, C., P. Mastromarino, A. Riccioli, and N. Orsi. 1991. Electrostatic interactions in the early events of VSV infection. Res. Virol. 142:17-24. [DOI] [PubMed] [Google Scholar]
- 9.Cornetta, K., and W. F. Anderson. 1989. Protamine sulfate as an effective alternative to Polybrene in retroviral-mediated gene-transfer: implications for human gene therapy. J. Virol. Methods 23:187-194. [DOI] [PubMed] [Google Scholar]
- 10.Costello, E., M. Munoz, E. Buetti, P. Meylan, H. Diggelmann, and M. Thali. 2000. Gene transfer into stimulated and unstimulated T lymphocytes by HIV-1-derived lentiviral vectors. Gene Ther. 7:596-604. [DOI] [PubMed] [Google Scholar]
- 11.DePolo, N. J., J. D. Reed, P. L. Sheridan, K. Townsend, S. L. Sauter, D. J. Jolly, and T. W. Dubensky, Jr. 2000. VSV-G pseudotyped lentiviral vector particles produced in human cells are inactivated by human serum. Mol. Ther. 2:218-222. [DOI] [PubMed] [Google Scholar]
- 12.Donahue, R. E., B. P. Sorrentino, R. G. Hawley, D. S. An, I. S. Chen, and R. P. Wersto. 2001. Fibronectin fragment CH-296 inhibits apoptosis and enhances ex vivo gene transfer by murine retrovirus and human lentivirus vectors independent of viral tropism in nonhuman primate CD34+ cells. Mol. Ther. 3:359-367. [DOI] [PubMed] [Google Scholar]
- 13.Ehrengruber, M. U., S. Hennou, H. Bueler, H. Y. Naim, N. Deglon, and K. Lundstrom. 2001. Gene transfer into neurons from hippocampal slices: comparison of recombinant Semliki Forest Virus, adenovirus, adeno-associated virus, lentivirus, and measles virus. Mol. Cell. Neurosci. 17:855-871. [DOI] [PubMed] [Google Scholar]
- 14.Ehrengruber, M. U., K. Lundstrom, C. Schweitzer, C. Heuss, S. Schlesinger, and B. H. Gahwiler. 1999. Recombinant Semliki Forest virus and Sindbis virus efficiently infect neurons in hippocampal slice cultures. Proc. Natl. Acad. Sci. USA 96:7041-7046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Emi, N., T. Friedmann, and J.-K. Yee. 1991. Pseudotype formation of murine leukemia virus with the G protein of vesicular stomatitis virus. J. Virol. 65:1202-1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Freed, E. O., and M. A. Martin. 2001. HIVs and their replication, p. 1971-2041. In D. M. Knipe and P. M. Howley (ed.), Fields virology, 4th ed. Lippincott/The Williams & Wilkins Co., Philadelphia, Pa.
- 17.Fuller, S. D., J. A. Berriman, S. J. Butcher, and B. E. Gowen. 1995. Low pH induces the swivelling of the glycoprotein heterodimers in the Semliki Forest virus spike complex. Cell 81:715-725. [DOI] [PubMed] [Google Scholar]
- 18.Haas, D. L., S. S. Case, G. M. Crooks, and D. B. Kohn. 2000. Critical factors influencing stable transduction of human CD34+ cells with HIV-1-derived lentiviral vectors. Mol. Ther. 2:71-80. [DOI] [PubMed] [Google Scholar]
- 19.Hanenberg, H., K. Hashino, H. Konishi, R. A. Hock, I. Kato, and D. A. Williams. 1997. Optimization of fibronectin-assisted retroviral gene transfer into human CD34+ hematopoietic cells. Hum. Gene Ther. 8:2193-2206. [DOI] [PubMed] [Google Scholar]
- 20.Hanenberg, H., X. L. Xiao, D. Dilloo, K. Hashino, I. Kato, and D. A. Williams. 1996. Colocalization of retrovirus and target cells on specific fibronectin fragments increases genetic transduction of mammalian cells. Nat. Med. 2:876-882. [DOI] [PubMed] [Google Scholar]
- 21.Harley, D., A. Sleigh, and S. Ritchie. 2001. Ross River virus transmission, infection, and disease: a cross-disciplinary review. Clin. Microbiol. Rev. 14:909-932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Heil, M. L., A. Albee, J. H. Strauss, and R. J. Kuhn. 2001. An amino acid substitution in the coding region of the E2 glycoprotein adapts Ross River virus to utilize heparan sulfate as an attachment moiety. J. Virol. 75:6303-6309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Higashikawa, F., and L.-J. Chang. 2001. Kinetic analysis of stability of simple and complex retroviral vectors. Virology 280:124-131. [DOI] [PubMed] [Google Scholar]
- 24.Hofmann, W., D. Schubert, J. LaBonte, L. Munson, S. Gibson, J. Scammell, P. Ferrigno, and J. Sodroski. 1999. Species-specific, postentry barriers to primate immunodeficiency virus infection. J. Virol. 73:10020-10028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ikeda, Y., M. K. Collins, P. A. Radcliffe, K. A. Mitrophanous, and Y. Takeuchi. 2002. Gene transduction efficiency in cells of different species by HIV and EIAV vectors. J. Virol. 9:932-938. [DOI] [PubMed] [Google Scholar]
- 26.Johnston, R. E., and C. J. Peters. 2001. Alphaviruses, p. 843-890. In D. M. Knipe and P. M. Howley (ed.), Fields virology, 4th ed. Lippincott/The Williams & Wilkins Co., Philadelphia, Pa.
- 27.Kang, Y., C. S. Stein, J. A. Heth, P. L. Sinn, A. K. Penisten, P. D. Staber, K. L. Ratliff, H. Shen, C. K. Barker, I. Martins, C. M. Sharkey, D. A. Sanders, P. B. McCray, Jr., and B. L. Davidson. 2002. In vivo gene transfer using a nonprimate lentiviral vector pseudotyped with Ross River virus glycoproteins. J. Virol. 76:9378-9388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kobinger, G. P., D. J. Weiner, Q.-C. Yu, and J. M. Wilson. 2001. Filovirus-pseudotyped lentiviral vector can efficiently and stably transduce airway epithelia in vivo. Nat. Bio/Technol. 19:225-230. [DOI] [PubMed] [Google Scholar]
- 29.Kowolik, C. M., and J.-K. Yee. 2002. Preferential transduction of human hepatocytes with lentiviral vectors pseudotyped by Sendai virus F protein. Mol. Ther. 5:762-769. [DOI] [PubMed] [Google Scholar]
- 30.Landau, N. R., K. A. Page, and D. R. Littman. 1991. Pseudotyping with human T-cell leukemia virus type I broadens the human immunodeficiency virus host range. J. Virol. 65:162-169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lei, P., B. Bajaj, and S. T. Andreadis. 2002. Retrovirus-associated heparan sulfate mediates immobilization and gene transfer on recombinant fibronectin. J. Virol. 76:8722-8728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mastromarino, P., C. Conti, P. Goldoni, B. Hauttecoeur, and N. Orsi. 1987. Characterization of membrane components of the erythrocyte involved in vesicular stomatitis virus attachment and fusion at acidic pH. J. Gen. Virol. 68:2359-2369. [DOI] [PubMed] [Google Scholar]
- 33.Moritz, T., P. Dutt, X. Xiao, D. Carstanjen, T. Vik, H. Hanenberg, and D. A. Williams. 1996. Fibronectin improves transduction of reconstituting hematopoietic stem cells by retroviral vectors: evidence of direct viral binding to chymotryptic carboxy-terminal fragments. Blood 88:855-862. [PubMed] [Google Scholar]
- 34.Naldini, L., U. Bloemer, P. Gallay, D. Ory, R. Mulligan, F. H. Gage, I. M. Verma, and D. Trono. 1996. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 272:263-267. [DOI] [PubMed] [Google Scholar]
- 35.Paredes, A. M., D. T. Brown, R. Rothnagel, W. Chiu, R. J. Schoepp, R. E. Johnston, and B. V. Prasad. 1993. Three-dimensional structure of a membrane-containing virus. Proc. Natl. Acad. Sci. USA 90:9095-9099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Relander, T., A. Brun, R. G. Hawley, S. Karlsson, and J. Richter. 2001. Retroviral transduction of human CD34+ cells on fibronectin fragment CH-296 is inhibited by high concentrations of vector containing medium. J. Gene Med. 3:207-218. [DOI] [PubMed] [Google Scholar]
- 37.Sanders, D. A. 2002. No false start for novel pseudotyped vectors. Curr. Opin. Bio/Technol. 13:437-442. [DOI] [PubMed] [Google Scholar]
- 38.Sandrin, V., B. Boson, P. Salmon, W. Gay, D. Nègre, R. Le Grand, D. Trono, and F.-L. Cosset. 2002. Lentiviral vectors pseudotyped with a modified RD114 envelope glycoprotein show increased stability in sera and augmented transduction of primary lymphocytes and CD34+ cells derived from human and nonhuman primates. Blood 100:823-832. [DOI] [PubMed] [Google Scholar]
- 39.Sastry, L., T. Johnson, M. J. Hobson, B. Smucker, and K. Cornetta. 2002. Titering lentiviral vectors: comparison of DNA, RNA and marker expression methods. Gene Ther. 9:1155-1162. [DOI] [PubMed] [Google Scholar]
- 40.Schlegel, R., T. S. Tralka, M. C. Willingham, and I. Pastan. 1983. Inhibition of VSV binding and infectivity by phosphatidylserine: is phosphatidylserine a VSV-binding site? Cell 32:639-646. [DOI] [PubMed] [Google Scholar]
- 41.Sharkey, C. M., C. L. North, R. J. Kuhn, and D. A. Sanders. 2001. Ross River virus glycoprotein-pseudotyped retroviruses and stable cell lines for their production. J. Virol. 75:2653-2659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Slingsby, J. H., D. Baban, J. Sutton, M. Esapa, T. Price, S. M. Kingsman, A. J. Kingsman, and A. Slade. 2000. Analysis of 4070A envelope levels in retroviral preparations and effect on target cell transduction efficiency. Hum. Gene Ther. 11:1439-1451. [DOI] [PubMed] [Google Scholar]
- 43.Strauss, J. H., and E. G. Strauss. 1994. The alphaviruses: gene expression, replication, and evolution. Microbiol. Rev. 58:491-562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Suomalainen, M., and H. Garoff. 1994. Incorporation of homologous and heterologous proteins into the envelope of Moloney murine leukemia virus. J. Virol. 68:4879-4889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Toyoshima, K., and P. K. Vogt. 1969. Enhancement and inhibition of avian sarcoma viruses by polycations and polyanions. Virology 38:414-426. [DOI] [PubMed] [Google Scholar]
- 46.Traycoff, C. M., E. F. Srour, P. Dutt, Y. Fan, and K. Cornetta. 1997. The 30/35 kDa chymotryptic fragment of fibronectin enhances retroviral-mediated gene transfer in purified chronic myelogenous leukemia bone marrow progenitors. Leukemia 11:159-167. [DOI] [PubMed] [Google Scholar]
- 47.von Laer, D., A. Corovic, B. Vogt, U. Herwig, S. Roscher, B. Fehse, and C. Baum. 2000. Amphotropic and VSV-G-pseudotyped retrovirus vectors transduce human hematopoietic cells with similar efficiency. Bone Marrow Transplant. 25(Suppl. 2):S75-S79. [DOI] [PubMed] [Google Scholar]
- 48.Zennou, V., C. Petit, D. Guetard, U. Nehrbass, L. Montagnier, and P. Charneau. 2000. HIV-1 genome nuclear import is mediated by a central DNA flap. Cell 101:173-185. [DOI] [PubMed] [Google Scholar]
- 49.Zufferey, R., J. E. Donello, D. Trono, and T. J. Hope. 1999. Woodchuck hepatitis virus posttranscriptional regulatory element enhances expression of transgenes delivered by retroviral vectors. J. Virol. 73:2886-2892. [DOI] [PMC free article] [PubMed] [Google Scholar]