Human immunodeficiency virus (HIV)-infected persons, especially those with advanced immune suppression or uncontrolled viremia, experienced increased numbers of multimorbid non-AIDS-defining conditions compared with epidemiologically comparable HIV-uninfected persons. Many of these clinically identified chronic diseases were unrecognized and untreated.
Abstract
Background. Despite an increasing burden of age-associated non-AIDS outcomes, few studies have investigated the prevalence or correlates of multimorbidity among aging human immunodeficiency virus (HIV)–infected and epidemiologically comparable at-risk populations.
Methods. Among 1262 AIDS Linked to the IntraVenous Experience (ALIVE) study participants followed in a community-based observational cohort, we defined the prevalence of 7 non-AIDS-defining chronic conditions (diabetes, obstructive lung disease, liver disease, anemia, obesity, kidney dysfunction, and hypertension) using clinical and laboratory criteria. Ordinal logistic regression was used to model the odds of increased multimorbidity associated with demographic, behavioral, and clinical factors. Self-reported prevalence was compared with clinically defined prevalence.
Results. Participants were a median of 48.9 years of age; 65.1% were male, 87.5% were African-American, and 28.7% were HIV infected. In multivariable analysis, HIV infection (odds ratio [OR], 1.50; 95% confidence interval [CI], 1.13–1.99) was positively associated with increased multimorbidity. Among HIV-infected participants, multimorbidity was increased with lower nadir CD4 T-cell count (OR, 1.14 per 100-cell decrease; 95% CI, 1.00–1.29) and higher current HIV RNA (OR, 1.32 per log10 increase; 95% CI, 1.08–1.60). Older age, being female, not using cigarettes or drugs, and having depressive symptoms were also associated with increased multimorbidity. A substantial proportion of multimorbid conditions in HIV-infected and HIV-uninfected participants were unrecognized and untreated.
Conclusions. HIV-infected participants experienced increased numbers of multimorbid conditions; risk increased with advanced immunosuppression and higher viremia. These results underscore the heavy burden of multimorbidity associated with HIV and highlight the need for incorporating routine assessment and integrated management of chronic diseases as part of comprehensive healthcare for aging, HIV-infected persons.
Aging and immunodeficiency are associated with increasing severity, duration, and co-occurrence of chronic health conditions [1]. With the advent of highly active anti-retroviral therapy (HAART), survival of persons living with human immunodeficiency virus (HIV) infection has notably improved. However, even in patients on HAART, HIV infection can cause immunosenescence and chronic inflammation [2], which may contribute to multimorbidity (defined as “the co-occurrence of multiple chronic or acute diseases and medical conditions in one person without reference to an index condition” [3]). Having more than 1 chronic disease has been associated with poor functional status, decreased quality of life, unnecessary hospitalizations, adverse drug events, mortality, and increased medical costs [4–6].
Few studies have assessed multimorbidity among HIV-infected populations [7, 8] with even fewer specifically among injection drug users (IDUs) [9]. Given the importance of multimorbidity in aging populations [10], the increasing age of HIV-infected [11] and IDU populations [12], and the lack of information on multimorbidity in these groups, we investigated the prevalence of non-AIDS-defining multimorbidity and evaluated HIV-related and other factors as correlates of multimorbidity in a large, urban IDU cohort.
METHODS
Study Population
The AIDS Linked to the IntraVenous Experience (ALIVE) study, described in detail previously [13], is a prospective, community-recruited observational cohort of HIV-infected and at-risk current and former IDUs followed outside clinical treatment settings in Baltimore, Maryland. Enrollment of persons ≥18 years of age with a history of injection drug use began in 1988, with subsequent recruitment in 1994–1995, 1998, 2000, and 2005–2008. HIV-infected and -uninfected participants were followed under a common protocol involving biannual visits with interview, clinical examination, and biospecimen collection. All participants provided written informed consent; the study was approved by the Johns Hopkins University Institutional Review Board. With participant consent, participants and their identified providers received results from clinical assessments. Participants evaluated from 2007 to 2009 were included in this analysis.
Data Collection and Measures
At baseline and follow-up visits, sociodemographic, behavioral, and clinical information was collected or updated. Risk behavior was collected using audio computer–assisted self-interview. Trained interviewers obtained HIV and general medical history. Depressive symptoms were assessed using the Center for Epidemiologic Studies Depression Scale (CES-D) with a cut-point of 23, as suggested for drug-using populations [14].
Routine laboratory testing at each visit included measurement of HIV serology for HIV-negative participants and of T-cell subsets and HIV RNA (Roche Molecular Systems Amplicor HIV-1 Monitor test version 1.5) for HIV-positive participants; HIV RNA of ≤400 copies/mL was considered below the lower limit of detection. Since October 2007, testing included a complete blood count, serum creatinine concentration, urine protein and creatinine concentration, and glycosylated hemoglobin using high-performance liquid chromatography (boronate affinity).
At each visit, standardized assessment of blood pressure (the last of 3 seated measurements), height, and weight were performed. Since February 2006, transient elastography (FibroScan, Echosens) was used to quantify liver stiffness (in kPa) as a noninvasive assessment of liver fibrosis [15]. Since January 2007, certified technicians performed prebronchodilator spirometry using a KOKO (Pulmonary Data Services) pneumotach in accordance with American Thoracic Society guidelines [16]; percent-predicted values were calculated using standard formulas [17].
The primary outcome of interest was multimorbidity determined by presence of up to 7 non-AIDS-defining chronic conditions including diabetes, obstructive lung disease (OLD) [17], liver fibrosis [15], anemia [18], obesity, kidney dysfunction [19], and hypertension. These conditions, defined in Table 1, were chosen because clinical and laboratory data were available to systematically assess their presence; standardized clinical and laboratory measures were not available to assess other conditions.
Table 1.
Definitions for Non-AIDS-Defining Chronic Conditions
| Condition | Definition |
| Diabetes | HbA1c concentration of >6.5% or self-reported diabetes medication taken in the prior 6 mo |
| Obstructive lung disease | Ratio of the forced expiratory volume in 1 sec to forced vital capacity of ≤70% [17] |
| Liver fibrosis | Liver stiffness ≥9.3 kPa, equivalent to Metavir F2 significant fibrosis [15] |
| Anemia | Hemoglobin concentration of <13.7, <13.2, <12.2, <12.9, <12.7, and <11.5 g/dL for white men age 20–59, white men age ≥60, white females, black men age 20–59, black men age ≥60, and black females, respectively [18] |
| Obesity | Body mass index ≥30 |
| Kidney dysfunction | Urine protein-creatinine concentration ratio >200 mg/g or an estimated glomerular filtration rate <60 mL/min/1.73 m2 using the MDRD equation [19] |
| Hypertension | Systolic BP ≥140 mm Hg, diastolic BP ≥90 mm Hg, or self-reported antihypertensive medication usage in the prior 6 mo |
Abbreviations: BP, blood pressure; HbA1c, hemoglobin A1c; MDRD, Modification of Diet and Renal Disease.
Self-reported prevalence of diseases was determined by asking participants if a healthcare provider had ever indicated that they had diabetes, OLD (chronic obstructive pulmonary disease, emphysema, or asthma), liver disease (bleeding varices, ascites, cirrhosis, or hepatic encelopathy), renal disease, or hypertension. Self-reported anemia or obesity diagnosis was not available. Self-reported prevalence was compared with the clinically determined prevalence as defined above, with the exceptions that more conservative and symptomatic definitions were used for renal (estimated glomerular filtration rate <60 mL/min/1.73 m2) and liver disease (liver stiffness ≥12.3 kPa equivalent to cirrhosis) [15].
Statistical Analysis
Summation of the number of conditions present for each participant was used to construct an ordinal multimorbidity outcome variable, categorized as 0, 1, 2, or ≥3 conditions. Although we used count data, categorization was used to account for small numbers of observations in cells with the greatest number of conditions. Analysis was restricted to participants with data available for at least 5 of the 7 conditions (n = 1262); compared with 219 excluded participants, those included were of similar sex, race, and HIV status but were slightly younger (median age, 47.4 vs 48.4 years, respectively). Our primary covariate of interest was HIV infection, further refined through examination of CD4 T-cell count, HIV RNA, and HAART use. Other covariates evaluated included sociodemographic variables (age, sex, race, marital status, homelessness, employment status), behavioral variables (alcohol consumption, cigarette or illicit drug use), and depressive symptoms.
Factors associated with multimorbidity were identified using χ2 tests for categorical variables and F-tests or median tests for continuous variables. Proportional odds models estimated 3 log odds simultaneously. Under the proportional odds assumption in the ordinal logistic regression model, the estimated odds ratio (OR) applies to any of the 3 ORs being modeled; that is, we estimated the increased likelihood associated with ≥1 relative to 0, ≥2 relative to 0–1, and ≥3 relative to 0–2 conditions. HIV-specific correlates were included in the model using interaction terms. Specifically, we investigated current CD4 T-cell count, nadir CD4 T-cell count (minimum CD4 T-cell count measured during time in study or the lowest self-reported CD4 T-cell count prior to study entry), and current HIV RNA. HAART use was evaluated as any HAART use in the prior 6 months, type of regimen, proportion or number of visits reporting HAART, time on HAART, and proportion of visits with undetectable HIV RNA to reflect optimally effective HAART.
Multicollinearity was determined using variance inflation factors. Covariates with univariate associations significant at α = .10 or with biologic significance were assessed in a multivariable proportional odds model. Covariates that were no longer significant at α = .05 were individually removed from the final model if the relationships between multimorbidity and the remaining independent variables as determined by point estimates were not changed by the removal of the nonsignificant covariates. The parallel assumption for proportional odds was formally tested. Because of differential follow-up time, a sensitivity analysis of cumulative measure variables was performed among those recruited before and after 2005. Self-reported and clinically determined prevalences were compared using Cohen’s κ test statistic. All analyses were performed using Stata software version 11.
RESULTS
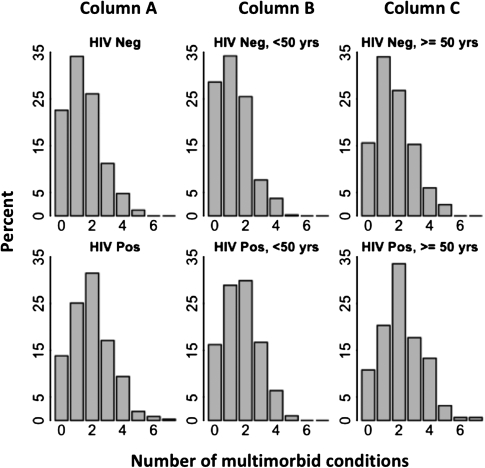
Participants’ median age was 48.9 years (interquartile range [IQR], 43.5–53.9; age range, 21–81); 65.1% were male, 87.5% were African American, and 28.7% were HIV infected (Table 2). HIV infection was positively associated with being African-American, never being married, being unemployed, and not recently drinking alcohol or using drugs. Among HIV-infected participants, the median CD4 T-cell count was 292 cells/mm3 (IQR, 165–479), 53.8% were currently receiving HAART, and 47.6% had HIV RNA ≤400 copies/mL. The median number of conditions was 1 (IQR, 1–2) for HIV-uninfected and 2 (IQR, 1–3) for HIV-infected participants. HIV-infected participants had a higher prevalence of ≥3 conditions compared with HIV-uninfected persons (29.6% vs 17.2%, respectively; P ≤ .001; Figure 1). The number of conditions was also higher for persons ≥50 years of age irrespective of HIV status.
Table 2.
Participant Characteristics, Stratified by HIV Status (N = 1262)
| HIV negative (n = 900) | HIV positive (n = 362) | P valuea | |
| Age, median in years (IQR) | 49.1 (43.3–54.1) | 48.7 (44.2–53.0) | .663 |
| Sex | |||
| Male | 589 (65.4) | 232 (64.1) | .648 |
| Female | 311 (34.6) | 130 (35.9) | |
| Race | |||
| White/other | 133 (14.8) | 24 (6.63) | <.001 |
| African-American | 767 (85.2) | 338 (93.4) | |
| Marital statusa | |||
| Never married | 557 (62.0) | 264 (73.5) | <.001 |
| Ever married | 342 (38.0) | 95 (26.5) | |
| Employedb | |||
| No | 662 (73.6) | 305 (85.0) | <.001 |
| Yes | 238 (26.4) | 54 (15.0) | |
| Homelessb | |||
| No | 766 (85.2) | 310 (86.4) | .602 |
| Yes | 133 (14.8) | 49 (13.7) | |
| Cigarette smokerb | |||
| No | 147 (16.3) | 64 (17.7) | .546 |
| Yes | 756 (83.7) | 298 (82.3) | |
| No. of alcoholic drinks/dayb | |||
| 0 | 406 (45.1) | 210 (58.0) | <.001 |
| 1–2 | 266 (29.6) | 80 (22.1) | |
| 3–4 | 152 (16.9) | 41 (11.3) | |
| ≥5 | 76 (8.44) | 31 (8.56) | |
| Drug userb | |||
| No | 349 (38.7) | 188 (51.8) | <.001 |
| Noninjection only | 177 (19.6) | 52 (14.3) | |
| Injection only | 124 (13.7) | 54 (14.9) | |
| Both | 253 (28.0) | 69 (19.0) | |
| Depressive symptoms | |||
| CES-D <23 | 706 (78.4) | 282 (78.1) | .898 |
| CES-D ≥23 | 194 (21.6) | 79 (21.9) |
Data represent no. (%) unless otherwise indicated.
Abbreviations: CES-D, Center for Epidemiologic Studies Depression Scale; HIV, human immunodeficiency virus; IQR, interquartile range.
P values are from Pearson’s χ2 for categorical variables or from a Hodges-Lehmann nonparametric k-sample test on equality of medians testing the null hypothesis that samples are drawn from a population with the same median.
At enrollment.
c In prior 6 months.
Figure 1.
Number of multimorbid conditions by human immunodeficiency virus (HIV) status and age group (N = 1262). Histograms display the proportion of participants having each number of multimorbid conditions, stratified by HIV status and presented as all age groups combined (A) and within groups <50 years of age (B) or ≥50 years of age (C).
The overall prevalence of specific conditions ranged from 8.4% having diabetes to 38% with hypertension (Table 3). Except for anemia and obesity, all conditions were more common among persons ≥50 years of age. Accounting for age group, HIV infection was positively associated with liver disease (P ≤ .001), anemia (P ≤ .001), obesity (P = .003), and kidney dysfunction (P ≤ .001) but was not significantly associated with diabetes (P = .349), OLD (P = .672), or hypertension (P = .582).
Table 3.
Prevalence of Specific Multimorbid Conditions by HIV Status and Age Group
| HIV negative |
HIV positive |
|||||
| Overall | <50 years | ≥50 years | <50 years | ≥50 years | P valuea | |
| Diabetes (n = 1262) | 106 (8.40) | 35 (7.26) | 45 (10.8) | 12 (5.88) | 14 (8.86) | .349 |
| Obstructive lung disease (n = 1158) | 225 (19.4) | 76 (17.4) | 87 (22.6) | 28 (14.7) | 34 (23.5) | .672 |
| Liver disease (n = 1121) | 249 (22.2) | 48 (11.7) | 95 (25.5) | 53 (28.0) | 53 (36.0) | <.001 |
| Anemia (n = 1262) | 301 (23.9) | 95 (19.7) | 74 (17.7) | 78 (38.2) | 54 (34.2) | <.001 |
| Obesity (n = 1255) | 326 (26.0) | 141 (29.4) | 112 (27.1) | 42 (20.6) | 31 (19.6) | .003 |
| Kidney dysfunction (n = 1176) | 321 (27.3) | 67 (15.3) | 84 (21.8) | 85 (42.9) | 85 (54.8) | <.001 |
| Hypertension (n = 1262) | 480 (38.0) | 137 (28.4) | 211 (50.5) | 59 (28.9) | 73 (46.2) | .582 |
Data represent no. (%) of the sample with the condition.
P values for human immunodeficiency virus (HIV) were calculated from coefficients in a logistic regression for the effects of HIV on presence of specific disease adjusted for age grouping and are based on the asymptotic normality theory.
In unadjusted analysis, several factors were associated with increasing numbers of multimorbid conditions (Table 4), including age (median age increased >5 years from those with no conditions to those with ≥3 conditions), being African-American, and not smoking or using illicit drugs. In additional models that excluded obesity and hypertension from the definition of multimorbidity, the protective effect of cigarette use and injection drug use was lost. In other models that included pharmacologic treatment for drug use, buprenorphine was not associated with multimorbidity. Whereas methadone maintenance or detoxification was associated with multimorbidity, largely through an association with obesity and liver disease, including this variable in an adjusted model did not alter the protective effect of drug use. HIV infection was significantly associated with increased multimorbidity; the proportion of participants with HIV increased from 19.8% of those with no conditions to 40.8% of those with ≥3 conditions.
Table 4.
Sociodemographic, Behavioral, and Clinical Characteristics by Number of Multimorbid Conditions (N = 1262)
| No. of multimorbid conditions |
|||||
| 0 (n = 253) | 1 (n = 398) | 2 (n = 349) | ≥3 (n = 262) | ||
| (20.0%) | (31.5%) | (27.7%) | (20.8%) | P valuea | |
| Age, median in years (IQR) | 46.7 (40.3–51.0) | 48.6 (43.0–53.3) | 49.5 (44.5–54.0) | 52.0 (46.5–55.7) | <.001 |
| Sex | |||||
| Male | 167 (66.0) | 267 (67.1) | 218 (62.5) | 169 (64.5) | .596 |
| Female | 86 (34.0) | 131 (32.9) | 131 (37.5) | 93 (35.5) | |
| Race | |||||
| White/other | 49 (19.4) | 54 (13.6) | 28 (8.02) | 26 (9.92) | <.001 |
| African-American | 204 (80.6) | 344 (86.4) | 321 (92.0) | 236 (90.1) | |
| Marital statusb | |||||
| Never married | 170 (67.5) | 267 (67.1) | 228 (65.7) | 156 (59.8) | .202 |
| Ever married | 82 (32.5) | 131 (32.9) | 119 (34.3) | 105 (40.2) | |
| Employedc | |||||
| No | 194 (76.7) | 298 (75.1) | 275 (79.0) | 200 (76.6) | .649 |
| Yes | 59 (23.3) | 99 (24.9) | 73 (21.0) | 61 (23.4) | |
| Homelessc | |||||
| No | 211 (83.7) | 341 (85.9) | 289 (82.8) | 235 (90.4) | .052 |
| Yes | 41 (16.3) | 56 (14.1) | 60 (17.2) | 25 (9.62) | |
| Cigarette smokerc | |||||
| No | 30 (11.9) | 58 (14.8) | 61 (17.7) | 62 (23.9) | .046 |
| Yes | 63 (25.0) | 88 (22.4) | 82 (23.8) | 61 (23.6) | |
| No. of alcoholic drinks/dayc | |||||
| 0 | 112 (44.3) | 180 (45.2) | 191 (54.7) | 133 (50.8) | .158 |
| 1–2 | 80 (31.6) | 117 (29.4) | 84 (24.1) | 65 (24.8) | |
| 3–4 | 44 (17.4) | 63 (15.8) | 48 (13.8) | 38 (14.5) | |
| ≥5 | 17 (6.72) | 38 (9.55) | 26 (7.45) | 26 (7.45) | |
| Drug userc | |||||
| No | 80 (31.6) | 148 (37.2) | 174 (49.9) | 134 (51.2) | <.001 |
| Noninjection only | 51 (20.2) | 75 (18.8) | 58 (16.6) | 45 (17.2) | |
| Injection only | 36 (14.2) | 59 (14.8) | 52 (14.9) | 30 (11.5) | |
| Both | 86 (34.0) | 116 (29.2) | 65 (18.6) | 53 (20.2) | |
| Depressive symptoms | |||||
| CES-D <23 | 202 (79.8) | 317 (79.7) | 272 (77.9) | 197 (75.5) | .566 |
| CES-D ≥23 | 51 (20.2) | 81 (20.4) | 77 (22.1) | 64 (24.5) | |
| HIV status | |||||
| HIV negative | 203 (80.2) | 307 (77.1) | 235 (67.3) | 155 (59.2) | <.001 |
| HIV positive | 50 (19.8) | 91 (22.9) | 114 (32.7) | 107 (40.8) | |
Data represent no. (%) unless otherwise indicated.
Abbreviations: CES-D, Center for Epidemiologic Studies Depression Scale; HIV, human immunodeficiency virus; IQR, interquartile range.
P values are from Pearson’s χ2 for categorical variables and from a Hodges-Lehmann nonparametric k-sample test on equality of medians that tests the null hypothesis that the samples are drawn from a population with the same median.
At enrollment.
In prior 6 months.
Compared to an HIV-uninfected person and controlling for other covariates (Table 5), an HIV-infected person with nadir CD4 T-cell count of 200 cells/mm3 and with current virologic suppression had >50% higher odds of multimorbidity (adjusted OR, 1.50; 95% CI, 1.13–1.99). Among HIV-infected persons, greater multimorbidity was associated with lower nadir CD4 T-cell count (adjusted OR, 1.14; 95% CI, 1.00–1.29 per 100-cell decrease) and higher current HIV RNA (adjusted OR, 1.32; 95% CI, 1.08–1.60 per log10 copies/mL increase). However, the number of multimorbid conditions was not significantly associated with any measure of HAART (data not shown). Among persons undergoing HAART, neither current protease inhibitor use (χ2 P = .597) nor any use of a protease inhibitor during time in study (χ2 P = .532) was associated with diabetes. Tenofovir use during any time in study was not associated with kidney dysfunction (χ2 P = .850). However, clinicians may selectively avoid prescribing specific antiretroviral medicines to persons with or at risk for comorbid conditions (eg, avoiding tenofovir in patients with renal dysfunction); this selection bias may have contributed to our observed lack of association. When analysis was restricted to earlier-recruited participants with longer follow-up with adjustment for age and sex, the risk estimates for current and nadir CD4 T-cell count and current HIV RNA were accentuated, while inferences from cumulative measures for virologic suppression or HAART were not substantively changed (data not shown).
Table 5.
Correlates of Increasing Multimorbidity Among ALIVE Study Participants (N = 1262)
| Unadjusted OR (95% CI)a | P value | Adjusted OR (95% CI)a | P value | |
| Age (per 1-year increase) | 1.06 (1.04–1.07) | <.001 | 1.06 (1.04–1.07) | <.001 |
| Sex | ||||
| Male | ref | ref | ||
| Female | 1.10 (.890–1.35) | .388 | 1.29 (1.04–1.60) | .023 |
| Race | ||||
| White/Other | ref | |||
| African-American | 1.86 (1.37–2.53) | <.001 | ||
| Marital statusb | ||||
| Never married | ref | |||
| Ever married | 1.22 (0.989–1.50) | .064 | ||
| Employedc | ||||
| No | ref | |||
| Yes | 0.941 (.744–1.19) | .612 | ||
| Homelessc | ||||
| No | ref | |||
| Yes | 0.813 (.615–1.07) | .144 | ||
| Cigarette smokerc | ||||
| No | ref | ref | ||
| Yes | 0.599 (.458–.784) | <.000 | 0.697 (.527–.923) | .012 |
| No. alcoholic drinks/dayc | ||||
| 0 | ref | |||
| 1–2 | 0.734 (.579–.930) | .011 | ||
| 3–4 | 0.770 (.575–1.03) | .079 | ||
| ≥5 | 1.00 (.695–1.45) | .981 | ||
| Drug userc | ||||
| No | ref | ref | ||
| Noninjection only | 0.639 (.483–.845) | .002 | 0.672 (.502–.900) | .008 |
| Injection only | 0.652 (.481–.883) | .006 | 0.753 (.550–1.03) | .075 |
| Both | 0.478 (.371–.616) | <.001 | 0.600 (.458–.787) | <.001 |
| Depressive symptoms | ||||
| CES-D <23 | ref | ref | ||
| CES-D ≥23 | 1.18 (.926–1.50) | .180 | 1.44 (1.11–1.85) | .005 |
| HIV status | ||||
| HIV negative | ref | ref | ||
| HIV positive | 1.99 (1.60–2.49) | <.001 | 1.50 (1.13–1.99) | .005 |
| Current CD4 T-cell countd | 1.11 (1.03–1.20) | .007 | ||
| CD4 T-cell nadird | 1.18 (1.04–1.34) | .003 | 1.14 (1.00–1.29) | .046 |
| HIV log10 HIV RNAe | 1.19 (0.988–1.44) | .067 | 1.32 (1.08–1.60) | .006 |
Abbreviations: ALIVE, AIDS Linked to the IntraVenous Experience study; CES-D, Center for Epidemiologic Studies Depression Scale; CI, confidence interval; HIV, human immunodeficiency virus; OR, odds ratio.
Univariate and multivariate proportional odds model: Under the proportional odds assumption, the estimated OR applies to any of the 3 ORs being modeled; that is, we estimate the OR for ≥1 conditions relative to 0, the OR for ≥2 conditions relative to 0–1, or the OR for ≥3- conditions relative to 0–2.
At enrollment.
In prior 6 months.
Per 100-cell decrease (centered at 200 cells/mm3).
Per log10 copies/mL increase (centered at 2.60, level of detection).
In addition to HIV infection, greater multimorbidity was associated with increased age, female sex, not smoking cigarettes, not using injection or noninjection drugs, and having depressive symptoms. For these covariates in the multivariable model (Table 5), an approximate likelihood ratio test (χ2 P = .990) and a Wald test by Brandt (χ2 P value for all variables = .979, and χ2 P-value range for individual variables = .102–.972) confirmed that the proportional odds assumption holds.
The majority of participants who did not have a clinically defined disease also self-reported never being diagnosed with that disease by a healthcare provider, with the percent agreement ranging from 74.3% for OLD to 99.5% for renal disease (Table 6). However, a majority of participants with clinically defined disease self-reported never being previously diagnosed. Among those with renal or liver disease, >80% reported never being diagnosed, while one-quarter to one-third with clinical evidence for diabetes or hypertension reported never being diagnosed. The κ statistics suggested slight to moderate agreement between clinical and self-reported prevalence. Agreement was similar by HIV status.
Table 6.
Specific Multimorbid Conditions Defined Clinically by Self-Reported Disease Status, Stratified by HIV Status
| HIV negative |
HIV positive |
|||||
| Clinically defined status | Self-report No | Self-report Yes | κa | Self-report No | Self-report Yes | κa |
| Diabetes (n = 1256)b | ||||||
| No | 768 (94.4) | 46 (5.65) | 0.581 | 322 (95.8) | 14 (4.17) | 0.637 |
| Yes | 23 (28.8) | 57 (71.3) | 6 (23.1) | 20 (76.9) | ||
| Obstructive lung disease (n = 1155) | ||||||
| No | 503 (76.4) | 155 (23.6) | 0.201 | 188 (69.1) | 84 (30.9) | 0.189 |
| Yes | 86 (53.1) | 76 (46.9) | 27 (43.6) | 35 (56.5) | ||
| Cirrhosis (n = 1121) | ||||||
| No | 699 (99.4) | 4 (0.57) | 0.214 | 267 (97.8) | 6 (2.20) | 0.195 |
| Yes | 66 (85.7) | 11 (14.3) | 52 (83.9) | 10 (16.1) | ||
| Renal disease (n = 1168) | ||||||
| No | 769 (99.4) | 5 (0.65) | 0.248 | 307 (99.0) | 3 (0.97) | 0.229 |
| Yes | 39 (83.0) | 8 (17.0) | 31 (83.8) | 6 (16.2) | ||
| Hypertension (n = 1224)c | ||||||
| No | 424 (77.5) | 123 (22.5) | 0.467 | 185 (81.1) | 43 (18.9) | 0.442 |
| Yes | 105 (30.4) | 241 (69.7) | 49 (37.4) | 82 (62.6) | ||
Data represent no. (%) unless otherwise indicated.
Using Cohen’s unweighted κ.
A total of 9.43% of those with diabetes self-reported taking diabetes medication in prior 6 months.
A total of 10.6% of those with hypertension self-reported taking antihypertension medication in prior 6 months.
DISCUSSION
In developed countries, HIV infection has evolved into a chronic disease with increasing morbidity and mortality associated with non-AIDS-defining conditions. In aging HIV-uninfected populations, the consequences of multimorbidity are considerable and include poorer functioning with reduced quality of life, increased healthcare utilization and costs, and reduced survival [4–6]. Our study documents a heavy burden of non-AIDS multimorbidity among aging HIV-infected persons, especially those with advanced immune suppression or uncontrolled HIV viremia. Extending prior prevalence estimates [7, 8, 20], these data identify factors associated with multimorbidity in HIV-infected and at-risk persons, provide insight into the pathogenesis of non-AIDS-defining conditions, and draw attention to the substantial prevalence of unrecognized and untreated multimorbid conditions in this population.
Estimates of multimorbidity prevalence in the general population are sparse [5, 21] but suggest that 10%–13% of adults have ≥2 and 3%–5% have ≥3 comorbid diseases [22]. Similar estimates in HIV-infected populations are limited and use disparate definitions of multimorbidity [7, 8, 20], making comparisons difficult. We found that 61% and approximately 30% of HIV-infected persons had ≥2 or ≥3 multimorbid conditions, respectively, which is roughly 6–10-fold higher than reported from the general population and 1.5–2 times the prevalence observed in our HIV-uninfected comparison group. The disease-specific prevalence observed among our HIV-infected participants was substantially higher than prior estimates among US adults for chronic lung disease [23], liver disease [24], anemia [25], kidney dysfunction [26], and hypertension [27], and comparable to estimates for diabetes [28] and obesity [29]; the same trends were also apparent in comparison to other HIV-infected populations [7, 8, 30, 31]. Recognizing that comparison of disease prevalence between populations is challenging (our current or former IDUs were generally younger and more likely to be African-American and male), our data strongly suggest that non-AIDS conditions, considered in terms of multimorbidity or individually, are extremely prevalent and represent a primary health concern for this population.
HIV infection was associated with an increasing number of non-AIDS-defining conditions and a >50% increased likelihood of multimorbidity; immunosuppression and higher HIV RNA further enhanced this effect. These data are consistent with prior findings that showed increased development of liver, renal, and cardiovascular disease among persons on antiretroviral therapy (ART) with structured treatment interruption, suggesting a role for immune deficiency and chronic virally mediated inflammation in development of individual non-AIDS events [32]. Several biomarkers of inflammation, immune activation, and coagulation have been associated with chronic disease outcomes in the general population [33]. Many of these markers appear elevated in HIV-infected persons and have been associated with non-AIDS events and mortality [34, 35]. Thus, evidence is mounting for an underlying physiological pathway of chronic inflammation attributable to chronic HIV infection that contributes to susceptibility to chronic disease and poorer health.
Based on this pathway, effective ART with viral suppression would be expected to have a protective effect on multimorbidity. We nevertheless found no relationship between HAART and multimorbidity, even when considering effective ART with CD4 T-cell recovery or virological suppression. Competing risks related to HAART may be present, as other studies link specific antiretroviral drugs to metabolic changes and chronic disease [36]. If disease outcomes are heterogeneous depending on ART use and drug class, then the lack of association in our study may be explained partly by our evaluation of multimorbidity rather than specific diseases. Alternatively, multimorbidities associated with more advanced HIV disease (and lower nadir CD4 T-cell count) may have developed prior to HAART initiation, consistent with data supporting improved non-AIDS outcomes with earlier HAART initiation [37].
In addition to older age, female sex and depressive symptoms were correlated with increased multimorbidity. These findings are consistent with cross-sectional and longitudinal studies of noninstitutionalized older adults [38], although the effects of age and sex vary by condition [39]. Using cigarettes or illicit drugs appeared protective against multimorbidity in our analyses. However, this protective effect appears to be explained largely by smoking and drug use lowering body mass index and blood pressure, as exclusion of these 2 conditions from our definition of multimorbidity resulted in a null association with cigarette and injection drug use. Further, a “‘healthy drug user” effect, in which individuals who are more ill may no longer be using harmful substances, may also have contributed to these cross-sectional findings.
Multimorbid conditions were common among both HIV-infected and -uninfected participants, yet were largely unrecognized by participants. For liver and kidney disease, which tend to be asymptomatic until advanced stages, this was particularly true. Among our HIV-infected IDUs, approximately 90% are coinfected with hepatitis C virus and 83% are current smokers; therefore, evaluation for these conditions would be of high priority. Notably, despite increased provider encounters and a smaller proportion without any insurance coverage (14% vs 35%), HIV-infected participants did not have improved recognition or treatment of multimorbid conditions compared with HIV-uninfected IDUs. The large proportion of unrecognized and untreated disease in this population could represent barriers to access to appropriate medical care, inadequate evaluation for persons in care, or, among those that may have been diagnosed, a failure of patient-provider communication resulting in a lack of patient comprehension and inaccurate self-report. Each of these explanations supports evolving approaches to providing comprehensive HIV care that routinely incorporates chronic disease management strategies while emphasizing self-management and patient-centered approaches [7, 40].
This study has several limitations. For our analysis, we used a summation measure of multimorbidity that did not account for disease severity. Currently no standard list of diseases or approaches for measuring multimorbidity exists. A summation of multimorbidity is useful to assess prevalence and provide insight without giving weight to any specific condition [1]. Accounting for other conditions such as cardiovascular, bone, or neurocognitive disease, however, would have provided a more comprehensive view of multimorbidity. The cross-sectional analysis precludes against inferring causality between risk factors and multimorbidity. Because our data derive from a largely African-American, male, urban, IDU population, results may not be generalizable to other HIV-infected populations. Self-report data may be subject to reporting bias; however, this bias is likely nondifferential. Furthermore, because data are collected in a nonclinical setting, we may avoid reporting bias specific to clinic-based data collection.
Immune changes and inflammation may underlie the increased vulnerability to age-associated morbidity we observed among HIV-infected persons. The heavy burden of age-associated multimorbid conditions indicates that appropriate diagnosis and management of these conditions is essential to preventing disease progression and maintaining health for this population. The aging and increasing medical complexity of HIV-infected populations challenges HIV care providers to move beyond focusing on ART and virological suppression to integrating evaluation and care of comorbid diseases into comprehensive healthcare services for HIV-infected persons. Improved understanding of the burden, mechanisms, and consequences of multimorbidity could contribute to improved management and care of aging HIV-infected persons.
Notes
Acknowledgments.
We thank the participants and staff of the ALIVE study for their contributions to this research.
Financial support.
This work was supported by the National Institutes of Health (grants RC1-AI-086053, R01-DA-04334, R01-DA-12568, R01-HL-90483, and R01-DA-16078; and grant K01-AI071754 to B. L.) and the American Cancer Society (MRSG-07-284-01-CCE to G. K.).
Potential conflicts of interest.
All authors: No reported conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Guralnik JM. Assessing the impact of comorbidity in the older population. Ann Epidemiol. 1996;6:376–80. doi: 10.1016/s1047-2797(96)00060-9. [DOI] [PubMed] [Google Scholar]
- 2.Effros RB, Fletcher CV, Gebo K, et al. Aging and infectious diseases: workshop on HIV infection and aging: what is known and future research directions. Clin Infect Dis. 2008;47:542–53. doi: 10.1086/590150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.van den Akker M, Buntinx F, Metsemakers JF, Roos S, Knottnerus JA. Multimorbidity in general practice: prevalence, incidence, and determinants of co-occurring chronic and recurrent diseases. J Clin Epidemiol. 1998;51:367–75. doi: 10.1016/s0895-4356(97)00306-5. [DOI] [PubMed] [Google Scholar]
- 4.Parekh AK, Barton MB. The challenge of multiple comorbidity for the US health care system. JAMA. 2010;303:1303–4. doi: 10.1001/jama.2010.381. [DOI] [PubMed] [Google Scholar]
- 5.Gijsen R, Hoeymans N, Schellevis FG, Ruwaard D, Satariano WA, van den Bos GA. Causes and consequences of comorbidity: a review. J Clin Epidemiol. 2001;54:661–74. doi: 10.1016/s0895-4356(00)00363-2. [DOI] [PubMed] [Google Scholar]
- 6.Wolff JL, Starfield B, Anderson G. Prevalence, expenditures, and complications of multiple chronic conditions in the elderly. Arch Intern Med. 2002;162:2269–76. doi: 10.1001/archinte.162.20.2269. [DOI] [PubMed] [Google Scholar]
- 7.Chu C, Umanski G, Blank A, Meissner P, Grossberg R, Selwyn PA. Comorbidity-related treatment outcomes among HIV-infected adults in the Bronx, NY. J Urban Health. 2011;88:507–16. doi: 10.1007/s11524-010-9540-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Goulet JL, Fultz SL, Rimland D, et al. Aging and infectious diseases: do patterns of comorbidity vary by HIV status, age, and HIV severity? Clin Infect Dis. 2007;45:1593–601. doi: 10.1086/523577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Klein RS. Trends related to aging and co-occurring disorders in HIV-Infected drug users. Subst Use Misuse. 2011;46:233–44. doi: 10.3109/10826084.2011.522843. [DOI] [PubMed] [Google Scholar]
- 10.Yancik R, Ershler W, Satariano W, Hazzard W, Cohen HJ, Ferrucci L. Report of the national institute on aging task force on comorbidity. J Gerontol A Biol Sci Med Sci. 2007;62:275–80. doi: 10.1093/gerona/62.3.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Smith G. Statement of Senator Gordon H. Smith. Aging hearing: HIV over fifty: exploring the new threat. Senate Special Committee on aging, 2005. Washington, DC: Available at: http://www.hivatis.org. http://www.aging.senate.gov/public/_files/hr141gs.pdf. Accessed 13 April 2006. [Google Scholar]
- 12.Armstrong GL. Injection drug users in the United States, 1979–2002: an aging population. Arch Intern Med. 2007;167:166–73. doi: 10.1001/archinte.167.2.166. [DOI] [PubMed] [Google Scholar]
- 13.Vlahov D, Anthony JC, Munoz A, et al. The ALIVE study, a longitudinal study of HIV-1 infection in intravenous drug users: description of methods and characteristics of participants. NIDA Res Monogr. 1991;109:75–100. [PubMed] [Google Scholar]
- 14.Weissman MM, Sholomskas D, Pottenger M, Prusoff BA, Locke BZ. Assessing depressive symptoms in five psychiatric populations: a validation study. Am J Epidemiol. 1977;106:203–14. doi: 10.1093/oxfordjournals.aje.a112455. [DOI] [PubMed] [Google Scholar]
- 15.Kirk GD, Astemborski J, Mehta SH, et al. Assessment of liver fibrosis by transient elastography in persons with hepatitis C virus infection or HIV-hepatitis C virus coinfection. Clin Infect Dis. 2009;48:963–72. doi: 10.1086/597350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J. 2005;26:319–38. doi: 10.1183/09031936.05.00034805. [DOI] [PubMed] [Google Scholar]
- 17.Drummond MB, Kirk GD, Astemborski J, et al. Prevalence and risk factors for unrecognized obstructive lung disease among urban drug users. Int J Chronic Obstruct Pulmon Dis. 2011;6:89–95. doi: 10.2147/COPD.S15968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Beutler E, Waalen J. The definition of anemia: what is the lower limit of normal of the blood hemoglobin concentration? Blood. 2006;107:1747–50. doi: 10.1182/blood-2005-07-3046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yanik EL, Lucas GM, Vlahov D, Kirk GD, Mehta SH. HIV and proteinuria in an injection drug user population. Clin J Am Soc Nephrol. 2010;5:1836–43. doi: 10.2215/CJN.01030210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Oursler KK, Goulet JL, Leaf DA, et al. Association of comorbidity with physical disability in older HIV-infected adults. AIDS Patient Care STDs. 2006;20:782–91. doi: 10.1089/apc.2006.20.782. [DOI] [PubMed] [Google Scholar]
- 21.Fortin M, Lapointe L, Hudon C, Vanasse A. Multimorbidity is common to family practice: is it commonly researched? Can Fam Physician. 2005;51:244–5. [PMC free article] [PubMed] [Google Scholar]
- 22.Fortin M, Hudon C, Haggerty J, Akker M, Almirall J. Prevalence estimates of multimorbidity: a comparative study of two sources. BMC Health Serv Res. 2010;10:111. doi: 10.1186/1472-6963-10-111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mannino DM, Homa DM, Akinbami LJ, Ford ES, Redd SC. Chronic obstructive pulmonary disease surveillance–United States, 1971–2000. MMWR Surveill Summ. 2002;51:1–16. [PubMed] [Google Scholar]
- 24.Pleis JR, Lucas JW. Summary health statistics for U.S. adults: National Health Interview Survey, 2007. Vital Health Stat 10. 2009:1–159. [PubMed] [Google Scholar]
- 25.Looker AC, Cogswell ME, Gunter MT. Iron deficiency—United States, 1999–2000. MMWR Morb Mortal Wkly Rep. 2002;51:897–9. [PubMed] [Google Scholar]
- 26.Saydah S, Eberhardt M, Rios-Burrows N, Williams D, Geiss L, Dorsey R. Prevalence of chronic kidney disease and associated risk factors—United States, 1999-2004. MMWR Morb Mortal Wkly Rep. 2007;56:161–5. [PubMed] [Google Scholar]
- 27.Egan BM, Zhao Y, Axon RN. US trends in prevalence, awareness, treatment, and control of hypertension, 1988–2008. JAMA. 2010;303:2043–50. doi: 10.1001/jama.2010.650. [DOI] [PubMed] [Google Scholar]
- 28.National Center for Health Statistics. Health, United States, 2008 with Chartbook. Hyattsville, MD: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, and National Center for Health Statistics; 2009. [Google Scholar]
- 29.Flegal KM, Carroll MD, Ogden CL, Curtin LR. Prevalence and trends in obesity among US adults, 1999–2008. JAMA. 2010;303:235–41. doi: 10.1001/jama.2009.2014. [DOI] [PubMed] [Google Scholar]
- 30.Justice AC, McGinnis KA, Skanderson M, et al. Towards a combined prognostic index for survival in HIV infection: the role of ‘non-HIV’ biomarkers. HIV Med. 2010;11:143–51. doi: 10.1111/j.1468-1293.2009.00757.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Oursler KK, Goulet JL, Crystal S, et al. Association of age and comorbidity with physical function in HIV-infected and uninfected patients: results from the veterans aging cohort study. AIDS Patient Care STDS. 2011;25:13–20. doi: 10.1089/apc.2010.0242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.El-Sadr WM, Lundgren JD, Neaton JD, et al. CD4+ count-guided interruption of antiretroviral treatment. N Engl J Med. 2006;355:2283–96. doi: 10.1056/NEJMoa062360. [DOI] [PubMed] [Google Scholar]
- 33.Ferrucci L, Ble A, Bandinelli S, Lauretani F, Suthers K, Guralnik JM. A flame burning within. Aging Clin Exp Res. 2004;16:240–3. doi: 10.1007/BF03327390. [DOI] [PubMed] [Google Scholar]
- 34.Deeks SG. HIV infection, inflammation, immunosenescence, and aging. Annu Rev Med. 2011;62:141–55. doi: 10.1146/annurev-med-042909-093756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nixon DE, Landay AL. Biomarkers of immune dysfunction in HIV. Curr Opin HIV AIDS. 2010;5:498–503. doi: 10.1097/COH.0b013e32833ed6f4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Roling J, Schmid H, Fischereder M, Draenert R, Goebel FD. HIV-associated renal diseases and highly active antiretroviral therapy-induced nephropathy. Clin Infect Dis. 2006;42:1488–95. doi: 10.1086/503566. [DOI] [PubMed] [Google Scholar]
- 37.Kitahata MM. When to start antiretroviral therapy. Top HIV Med. 2010;18:121–6. [PubMed] [Google Scholar]
- 38.Marengoni A, Angleman S, Melis R, et al. Ageing with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430–9. doi: 10.1016/j.arr.2011.03.003. [DOI] [PubMed] [Google Scholar]
- 39.Fillenbaum GG, Pieper CF, Cohen HJ, Cornoni-Huntley JC, Guralnik JM. Comorbidity of five chronic health conditions in elderly community residents: determinants and impact on mortality. J Gerontol A Biol Sci Med Sci. 2000;55:M84–9. doi: 10.1093/gerona/55.2.m84. [DOI] [PubMed] [Google Scholar]
- 40.Swendeman D, Ingram BL, Rotheram-Borus MJ. Common elements in self-management of HIV and other chronic illnesses: an integrative framework. AIDS Care. 2009;21:1321–4. doi: 10.1080/09540120902803158. [DOI] [PMC free article] [PubMed] [Google Scholar]