Abstract
Resistance to phagocyte-derived reactive oxygen species is essential for Salmonella enterica serovar Typhimurium pathogenesis. Salmonella can enhance its resistance to oxidants through the induction of specific genetic pathways controlled by SoxRS, OxyR, σS, σE, SlyA, and RecA. These regulons can be found in a wide variety of pathogenic and environmental bacteria, suggesting that evolutionarily conserved mechanisms defend against oxidative stress both endogenously generated by aerobic respiration and exogenously produced by host phagocytic cells. Dps, a ferritin-like protein found in many eubacterial and archaebacterial species, appears to protect cells from oxidative stress by sequestering iron and limiting Fenton-catalyzed oxyradical formation. In Escherichia coli and some other bacterial species, Dps has been shown to accumulate during stationary phase in a σS-dependent fashion, bind nonspecifically to DNA, and form a crystalline structure that compacts and protects chromatin from oxidative damage. In the present study, we provide evidence that Dps protects Salmonella from iron-dependent killing by hydrogen peroxide, promotes Salmonella survival in murine macrophages, and enhances Salmonella virulence. Reduced numbers of dps mutant bacteria in the livers and spleens of infected mice are consistent with a role of Dps in protecting Salmonella from oxidative stress encountered during infection.
Complex mechanisms have evolved to allow bacteria to withstand the oxidative stress associated with aerobic life (13). Many regulatory and enzymatic loci required for resistance to reactive oxygen species generated during aerobic metabolism have been found to play an important role in the ability of pathogenic bacteria such as Salmonella enterica serovar Typhimurium to cause disease (5, 8, 12, 15, 20-22, 25, 26), suggesting that conserved strategies to resist oxidative stress in the environment can also allow organisms to withstand the respiratory burst of phagocytic cells.
Elegant studies of Escherichia coli by Kolter and colleagues first identified a low-molecular-weight protein that accumulates during stationary phase and binds to DNA; the protein was designated Dps (DNA binding protein in stationary phase) (2). Expression of dps in E. coli has been shown to be regulated by the stationary-phase sigma factor RpoS (σ38), OxyR, and IHF (3). As E. coli enters stationary phase, more than 180,000 Dps molecules accumulate within a single organism, making Dps the most abundant protein in the cell (1). Dps-deficient mutant E. coli strains are unable to survive long-term starvation (3, 17) and exhibit enhanced susceptibility to oxidative stress (25). E. coli Dps is able to form a microcrystalline structure on chromatin in intact stationary-phase cells or in association with purified DNA in vitro (29), leading to the suggestion that Dps physically protects DNA by sequestration. However, the crystal structure of Dps has revealed similarity to the iron storage protein ferritin (18, 30), and Zhao et al. (30) have found that Dps prevents DNA damage in E. coli through its capacity to bind Fe(II) and prevent the formation of hydroxyl radicals. Recent studies in Agrobacterium tumefaciens and Mycobacterium smegmatis indicate that Dps can prevent oxidative DNA damage even in the absence of DNA binding (10, 19).
The contribution of Dps to S. enterica pathogenesis has not been investigated previously, although dps expression appears to be induced following Salmonella ingestion by macrophages (14, 27). In this study, we report the contribution of dps to Salmonella oxidative stress resistance, survival in macrophages, and virulence in mice.
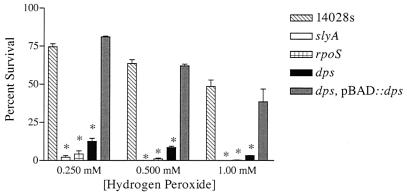
The ability of Salmonella to survive and replicate within host phagocytes is absolutely essential for Salmonella virulence (16). Phagocyte-derived reactive oxygen species generated by the NADPH phagocyte oxidase play an important role in innate immunity to Salmonella (24, 28), and a number of Salmonella mutant strains with enhanced susceptibility to oxidative stress have a reduced capacity to survive in macrophages (9, 12, 21, 26). To determine whether dps is required for oxidative stress resistance, a dps::aph mutation was constructed in S. enterica serovar Typhimurium ATCC 14028s using the method of Datsenko and Wanner, in which the dps open reading frame was replaced with the aph cassette (11). Oligonucleotide primers 5′-TTAATTACCTGGGACACAAACATCAAGAGGATATGAGATTGTGTAGGCTGGAGCCTTC and 5′-TACCTTCCTGCAACTCGAAGTATTCAGGGTAGAGATAGATATTCCGGGGATCCGTCGACC were utilized to create the dps::aph disruption, and primers 5′-CGGTGCTATACTTATTTTCG and 5′-CTGCGGATTCGCTGCGTTTG were used to confirm the expected insertion mutation. In addition, oligonucleotide primers 5′-GCCAAAACTGAAGCTACAGGTGCCAAGTGCGCACTATGTCAGGAAACAGCTATGACCATG and 5′-GAATGACCTCTTCCATCTTCCATCTCAGCGATCAGCGCGTCCGCTTTTACAACCAATAACCAATTC were used to create a slyA::aph disruption using the method of Datsenko and Wanner (11), and primers 5′-GCTTTAGTTTTAGCCAAAACTG and 5′-ACCGTCTCTCCACGCTAAAC were used to confirm the mutation. The absence of Dps protein in the dps::aph mutant strain was also confirmed by Western blot analysis using Dps antiserum (provided by R. Kolter) (data not shown). Wild-type S. enterica serovar Typhimurium, the isogenic dps::aph mutant derivative (SL3474), the dps::aph mutant complemented with plasmid pBAD::Dps (SL3476), an isogenic rpoS::pRR10 (ΔtrfA) mutant (SF1005) (15), and an isogenic slyA::aph mutant (SL3343) were compared for their susceptibilities to various concentrations of hydrogen peroxide. For Dps complementation using pBAD::dps, l-arabinose was added to a final concentration of 0.2%. Bacterial killing by hydrogen peroxide was measured in liquid medium as described by Buchmeier and Libby (7). Briefly, overnight cultures were grown in Luria-Bertani (LB) medium, diluted to 106 CFU in phosphate-buffered saline (PBS), and incubated at 37°C with a final concentration of 0.250, 0.500, or 1 mM hydrogen peroxide. Aliquots were removed after 2 h, and the number of viable cells was determined by serial dilution and plating onto LB agar. Percent survival following hydrogen peroxide challenge was calculated for each strain by dividing the number of CFU obtained from incubation in PBS alone by the number of CFU obtained from incubation in hydrogen peroxide. Each assay was repeated at least three times, and standard deviations were calculated and plotted. Salmonella carrying a dps mutation was found to be 15-fold less viable than wild type at 1 mM H2O2 and only slightly more resistant than a slyA mutant strain (Fig. 1). The hydrogen peroxide sensitivity phenotype was complemented to near-wild-type levels by providing dps on a plasmid in trans. Thus, the role of dps in the ability of S. enterica serovar Typhimurium to withstand killing by hydrogen peroxide is comparable to observations in E. coli (23).
FIG. 1.
dps mutant serovar Typhimurium has enhanced susceptibility to hydrogen peroxide. Results are expressed as percent survival after 2-h exposure to 0.250, 0.500, or 1.0 mM hydrogen peroxide at 37°C and represent the average percent survival from three independent assays. Error bars represent the standard deviations of three independent experiments. *, P < 0.01 by Student's t test, compared to wild type.
As discussed above, the antioxidant actions of Dps have been linked to its ability to sequester iron and prevent Fe(II)-dependent oxyradical formation. We therefore examined the ability of the Fe(II) chelator 2′,2-dipyridyl to inhibit killing of dps mutant Salmonella by hydrogen peroxide. Twenty-minute preincubation in 1 mM 2′,2-dipyridyl completely rescued the dps mutant strain from killing by 2 mM H2O2, but it provided only partial protection for rpoS or slyA mutant strains (Fig. 2).
FIG. 2.
The iron chelator 2′,2-dipyridyl rescues dps mutant Salmonella challenged with hydrogen peroxide. Each strain was pretreated with 1 mM 2′,2-dipyridyl or PBS for 20 min and then incubated with 2 mM H2O2 for 20 min at 37°C. Viable bacteria were determined by serial dilution and plating on LB agar. The numbers of surviving bacteria are expressed as a percentage of the original inoculum. The average percent survival from three independent experiments is shown, with error bars indicating standard deviations. *, P < 0.05; **, P < 0.001 by Student's t test, compared to wild type.
The intracellular survival of wild-type and mutant Salmonella strains was determined in C3H/HeN (ityr) primary peritoneal macrophages essentially as described previously (6, 28). Sodium periodate-elicited peritoneal macrophages were harvested from mice and plated at a density of 4 × 105 to 6 × 105 cells/well. Macrophages were infected 24 h later at a multiplicity of infection of 5:1 (bacteria/macrophage) with wild-type, dps mutant, or slyA mutant serovar Typhimurium cells that were opsonized with normal mouse serum. Extracellular bacteria were killed by the addition of gentamicin (50 μg/ml) to the medium. Macrophages were lysed at specific time points with 0.5% deoxycholate, serially diluted in PBS, and plated onto LB agar to determine the number of surviving bacteria. The percent surviving bacteria was calculated from three separate experiments and averaged. The dps mutant strain was found to be impaired in its ability to survive in murine macrophages compared to wild type (Fig. 3), but not as severely as the slyA mutant strain. In addition, a dps mutant complemented by providing dps in trans on low-copy-number plasmid pRB3 (4) showed partial restoration of survival inside murine peritoneal macrophages.
FIG. 3.
dps mutant Salmonella exhibits reduced survival in periodate-elicited murine peritoneal macrophages. Survival of Salmonella strains was determined in C3H/HeN (ityr) peritoneal macrophages at 3, 6, and 12 h postinfection. Murine peritoneal macrophages were elicited with 5 mM sodium periodate and harvested 4 days later. Macrophages were seeded at approximately 5 × 105 macrophages per well in 48-well plates and infected with Salmonella strains at a multiplicity of infection of 5:1 (bacteria/macrophages). Results are expressed as percent survival and represent the average of three independent assays, with error bars indicating standard deviations. *, P < 0.05 by Student's t test, compared to wild type.
The virulence of dps mutant serovar Typhimurium was assessed using a C3H/HeN (ityr) murine model. Groups of four 8-week-old C3H/HeN ityr female mice (Taconic Laboratories, Germantown, N.Y.) were infected intraperitoneally with wild-type, dps mutant, or slyA mutant bacteria. Overnight cultures of bacteria were diluted in PBS, and 200 μl of each dilution (approximately 1,000 CFU) was administered intraperitoneally using a 25-gauge needle. The inoculum size was confirmed by serial dilution and plating onto LB agar (data not shown). Infected mice were monitored for survival during a 2-week period. Only one of four mice infected with the dps mutant died during the course of the experiment, and the remaining mice never showed signs of illness. The virulence assay was repeated several times, with virtually identical results (Fig. 4). All mice infected with wild-type Salmonella succumbed by 9 days postinfection, but no mice infected with the slyA mutant strain died. These results demonstrate the crucial importance of dps in the ability of serovar Typhimurium to cause lethal infection in mice.
FIG. 4.
dps mutant Salmonella is attenuated for virulence in C3H/HeN (ityr) mice. Groups of four 8-week-old C3H/HeN female mice (Taconic Laboratories) were infected intraperitoneally with wild type (14028), an avirulent slyA mutant (SL3343), or a dps mutant strain (SL3474). Approximate 1,000 CFU of each strain in PBS was administered intraperitoneally. The survival of infected mice from three independent experiments is shown. *, P < 0.001 by χ2 test, compared to wild type.
As intraperitoneal lethality is not the optimum test for virulence, since it is measuring the cumulative growth, survival, and the lethal aspects of the bacteria, quantification of bacteria from mouse tissues was performed. Groups of four mice were infected with 1,800 CFU of wild-type serovar Typhimurium 14028s, a dps mutant, and a dps mutant complemented by providing dps in trans. After 5 days, the mice were euthanized and the spleens and livers were recovered and homogenized in 3 ml of sterile water. Enumeration of viable bacteria from each organ was determined by serial dilution in PBS and plating onto xylose-lysine-desoxycholate (XLD) agar. The results are illustrated in Fig. 5. These data demonstrate that a dps mutant is required for survival and the attenuation of virulence that is seen in C3H/HeN mice is due to a defect in intramacrophage survival. A dps mutant complemented by providing dps in trans showed a partial recovery in the number of surviving bacteria in the spleen and liver. Incomplete complementation of the macrophage survival and virulence-related phenotypes by pRB3::dps may have resulted from aberrant dps expression or instability of the pRB3 episomal vector.
FIG. 5.
dps mutant Salmonella showed reduced survival in vivo. Groups of four mice were infected intraperitoneally with 1,500 CFU of wild-type serovar Typhimurium, a dps mutant, or a dps mutant complemented by providing dps in trans. After 5 days, the mice were euthanized and the spleens and livers were recovered. The organs were homogenized in 3 ml of sterile water, serially diluted in PBS, and plated onto XLD agar to enumerate viable bacteria per organ. The results are expressed as CFU per organ, and error bars are included to represent standard deviations. *, P < 0.02 as determined by Student's t test, compared to wild type.
Resistance to oxidative stress plays an important role in the ability of Salmonella to resist killing by host phagocytes and cause a productive infection. Pathogenic microorganisms can resist host-derived reactive oxygen species by avoidance, inhibiting production, production of scavengers, metabolic detoxification, or repair of damage. Iron sequestration is another important mechanism, since intracellular Fe(II) can catalyze the formation of highly toxic oxyradicals from hydrogen peroxide.
The ferritin-like protein Dps has been found in many eubacterial and archaebacterial species. Similar to ferritin, Dps can sequester iron atoms to prevent their participation in the formation of toxic reactive oxygen species (30). Additionally, some Dps homologues appear to condense chromatin into a microcrystalline array that may physically protect DNA from damage (29). In the pathogenic gram-negative bacterium Salmonella, dps expression is induced following internalization of the bacteria by macrophages (14, 27). The results of our studies unequivocally demonstrate a role of S. enterica serovar Typhimurium dps in oxidative stress resistance and virulence. The most likely mechanism is the sequestration of iron and prevention of iron-dependent oxidative DNA damage. Dps can be added to the list of evolutionarily conserved antioxidant proteins employed by Salmonella to resist killing by host phagocytes.
Acknowledgments
We thank G. Thomas, A. Treece, N. Borden, H. Hassan, and B. J. Welker for their technical assistance and R. Kolter for insightful suggestions, strains, plasmids, and polyclonal Dps antibody.
This work was supported by National Institutes of Health grants to S.J.L. (AI48622) and F.C.F. (AI50660).
Editor: A. D. O'Brien
REFERENCES
- 1.Ali Azam, T., A. Iwata, A. Nishimura, S. Ueda, and A. Ishihama. 1999. Growth phase-dependent variation in protein composition of the Escherichia coli nucleoid. J. Bacteriol. 181:6361-6370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Almiron, M., A. J. Link, D. Furlong, and R. Kolter. 1992. A novel DNA-binding protein with regulatory and protective roles in starved Escherichia coli. Genes Dev. 6:2646-2654. [DOI] [PubMed] [Google Scholar]
- 3.Altuvia, S., M. Almiron, G. Huisman, R. Kolter, and G. Storz. 1994. The dps promoter is activated by OxyR during growth and by IHF and sigma S in stationary phase. Mol. Microbiol. 13:265-272. [DOI] [PubMed] [Google Scholar]
- 4.Berggren, R. E., A. Wunderlich, E. Ziegler, M. Schleicher, R. C. Duke, D. Looney, and F. C. Fang. 1995. HIV gp120-specific cell-mediated immune responses in mice after oral immunization with recombinant Salmonella. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 10:489-495. [PubMed] [Google Scholar]
- 5.Buchmeier, N., S. Bossie, C. Y. Chen, F. C. Fang, D. G. Guiney, and S. J. Libby. 1997. SlyA, a transcriptional regulator of Salmonella typhimurium, is required for resistance to oxidative stress and is expressed in the intracellular environment of macrophages. Infect. Immun. 65:3725-3730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Buchmeier, N. A., and F. Heffron. 1989. Intracellular survival of wild-type Salmonella typhimurium and macrophage-sensitive mutants in diverse populations of macrophages. Infect. Immun. 57:1-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Buchmeier, N. A., and S. J. Libby. 1997. Dynamics of growth and death within a Salmonella typhimurium population during infection of macrophages. Can. J. Microbiol. 43:29-34. [DOI] [PubMed] [Google Scholar]
- 8.Buchmeier, N. A., S. J. Libby, Y. Xu, P. C. Loewen, J. Switala, D. G. Guiney, and F. C. Fang. 1995. DNA repair is more important than catalase for Salmonella virulence in mice. J. Clin. Investig. 95:1047-1053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Buchmeier, N. A., C. J. Lipps, M. Y. So, and F. Heffron. 1993. Recombination-deficient mutants of Salmonella typhimurium are avirulent and sensitive to the oxidative burst of macrophages. Mol. Microbiol. 7:933-936. [DOI] [PubMed] [Google Scholar]
- 10.Ceci, P., A. Ilari, E. Falvo, and E. Chiancone. 2003. The Dps protein of Agrobacterium tumefaciens does not bind to DNA but protects it toward oxidative cleavage: X-ray crystal structure, iron binding, and hydroxyl-radical scavenging properties. J. Biol. Chem. 278:20319-20326. [DOI] [PubMed] [Google Scholar]
- 11.Datsenko, K. A., and B. L. Wanner. 2000. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA 97:6640-6645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.De Groote, M. A., U. A. Ochsner, M. U. Shiloh, C. Nathan, J. M. McCord, M. C. Dinauer, S. J. Libby, A. Vazquez-Torres, Y. Xu, and F. C. Fang. 1997. Periplasmic superoxide dismutase protects Salmonella from products of phagocyte NADPH-oxidase and nitric oxide synthase. Proc. Natl. Acad. Sci. USA 94:13997-14001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dukan, S., and T. Nystrom. 1999. Oxidative stress defense and deterioration of growth-arrested Escherichia coli cells. J. Biol. Chem. 274:26027-26032. [DOI] [PubMed] [Google Scholar]
- 14.Eriksson, S., S. Lucchini, A. Thompson, M. Rhen, and J. C. Hinton. 2003. Unravelling the biology of macrophage infection by gene expression profiling of intracellular Salmonella enterica. Mol. Microbiol. 47:103-118. [DOI] [PubMed] [Google Scholar]
- 15.Fang, F. C., S. J. Libby, N. A. Buchmeier, P. C. Loewen, J. Switala, J. Harwood, and D. G. Guiney. 1992. The alternative sigma factor katF (rpoS) regulates Salmonella virulence. Proc. Natl. Acad. Sci. USA 89:11978-11982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fields, P. I., R. V. Swanson, C. G. Haidaris, and F. Heffron. 1986. Mutants of Salmonella typhimurium that cannot survive within the macrophage are avirulent. Proc. Natl. Acad. Sci. USA 83:5189-5193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Frenkiel-Krispin, D., S. Levin-Zaidman, E. Shimoni, S. G. Wolf, E. J. Wachtel, T. Arad, S. E. Finkel, R. Kolter, and A. Minsky. 2001. Regulated phase transitions of bacterial chromatin: a non-enzymatic pathway for generic DNA protection. EMBO J. 20:1184-1191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Grant, R. A., D. J. Filman, S. E. Finkel, R. Kolter, and J. M. Hogle. 1998. The crystal structure of Dps, a ferritin homolog that binds and protects DNA. Nat. Struct. Biol. 5:294-303. [DOI] [PubMed] [Google Scholar]
- 19.Gupta, S., and D. Chatterji. 2002. Bimodal protection of DNA by Mycobacterium smegmatis Dps. J. Biol. Chem. 278:5235-5241. [DOI] [PubMed] [Google Scholar]
- 20.Humphreys, S., A. Stevenson, A. Bacon, A. B. Weinhardt, and M. Roberts. 1999. The alternative sigma factor, σE, is critically important for the virulence of Salmonella typhimurium. Infect. Immun. 67:1560-1568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Libby, S. J., W. Goebel, A. Ludwig, N. Buchmeier, F. Bowe, F. C. Fang, D. G. Guiney, J. G. Songer, and F. Heffron. 1994. A cytolysin encoded by Salmonella is required for survival within macrophages. Proc. Natl. Acad. Sci. USA 91:489-493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lundberg, B. E., R. E. Wolf, Jr., M. C. Dinauer, Y. Xu, and F. C. Fang. 1999. Glucose 6-phosphate dehydrogenase is required for Salmonella typhimurium virulence and resistance to reactive oxygen and nitrogen intermediates. Infect. Immun. 67:436-438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Martinez, A., and R. Kolter. 1997. Protection of DNA during oxidative stress by the nonspecific DNA-binding protein Dps. J. Bacteriol. 179:5188-5194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mastroeni, P., A. Vazquez-Torres, F. C. Fang, Y. Xu, S. Khan, C. E. Hormaeche, and G. Dougan. 2000. Antimicrobial actions of the NADPH phagocyte oxidase and inducible nitric oxide synthase in experimental salmonellosis. II. Effects on microbial proliferation and host survival in vivo. J. Exp. Med. 192:237-248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Testerman, T. L., A. Vazquez-Torres, Y. Xu, J. Jones-Carson, S. J. Libby, and F. C. Fang. 2002. The alternative sigma factor σE controls antioxidant defences required for Salmonella virulence and stationary-phase survival. Mol. Microbiol. 43:771-782. [DOI] [PubMed] [Google Scholar]
- 26.Tsolis, R. M., A. J. Baumler, and F. Heffron. 1995. Role of Salmonella typhimurium Mn-superoxide dismutase (SodA) in protection against early killing by J774 macrophages. Infect. Immun. 63:1739-1744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Valdivia, R. H., and S. Falkow. 1996. Bacterial genetics by flow cytometry: rapid isolation of Salmonella typhimurium acid-inducible promoters by differential fluorescence induction. Mol. Microbiol. 22:367-378. [DOI] [PubMed] [Google Scholar]
- 28.Vazquez-Torres, A., J. Jones-Carson, P. Mastroeni, H. Ischiropoulos, and F. C. Fang. 2000. Antimicrobial actions of the NADPH phagocyte oxidase and inducible nitric oxide synthase in experimental salmonellosis. I. Effects on microbial killing by activated peritoneal macrophages in vitro. J. Exp. Med. 192:227-236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wolf, S. G., D. Frenkiel, T. Arad, S. E. Finkel, R. Kolter, and A. Minsky. 1999. DNA protection by stress-induced biocrystallization. Nature 400:83-85. [DOI] [PubMed] [Google Scholar]
- 30.Zhao, G., P. Ceci, A. Ilari, L. Giangiacomo, T. M. Laue, E. Chiancone, and N. D. Chasteen. 2002. Iron and hydrogen peroxide detoxification properties of DNA-binding protein from starved cells. A ferritin-like DNA-binding protein of Escherichia coli. J. Biol. Chem. 277:27689-27696. [DOI] [PubMed] [Google Scholar]