Abstract
Background
There has been increased interest in recent years in very low carbohydrate ketogenic diets (VLCKD) that, even though they are much discussed and often opposed, have undoubtedly been shown to be effective, at least in the short to medium term, as a tool to tackle obesity, hyperlipidemia and some cardiovascular risk factors. For this reason the ketogenic diet represents an interesting option but unfortunately suffers from a low compliance. The aim of this pilot study is to ascertain the safety and effects of a modified ketogenic diet that utilizes ingredients which are low in carbohydrates but are formulated to simulate its aspect and taste and also contain phytoextracts to add beneficial effects of important vegetable components.
Methods
The study group consisted of 106 Rome council employees with a body mass index of ≥ 25, age between 18 and 65 years (19 male and 87 female; mean age 48.49 ± 10.3). We investigated the effects of a modified ketogenic diet based on green vegetables, olive oil, fish and meat plus dishes composed of high quality protein and virtually zero carbohydrate but which mimic their taste, with the addition of some herbal extracts (KEMEPHY ketogenic Mediterranean with phytoextracts). Calories in the diet were unlimited. Measurements were taken before and after 6 weeks of diet.
Results
There were no significant changes in BUN, ALT, AST, GGT and blood creatinine. We detected a significant (p < 0.0001) reduction in BMI (31.45 Kg/m2 to 29.01 Kg/m2), body weight (86.15 kg to 79.43 Kg), percentage of fat mass (41.24% to 34.99%), waist circumference (106.56 cm to 97.10 cm), total cholesterol (204 mg/dl to 181 mg/dl), LDLc (150 mg/dl to 136 mg/dl), triglycerides (119 mg/dl to 93 mg/dl) and blood glucose (96 mg/dl to 91 mg/dl). There was a significant (p < 0.0001) increase in HDLc (46 mg/dl to 52 mg/dl).
Conclusions
The KEMEPHY diet lead to weight reduction, improvements in cardiovascular risk markers, reduction in waist circumference and showed good compliance.
Background
Obesity has become a health emergency in Western countries [1,2]. As is well known, obesity and in particular abdominal obesity is one of the principle risk factors for cardiovascular disease and along with dyslipidemia, hypertension and diabetes contributes to the metabolic syndrome [3]. Even though weight loss is a desired goal for most overweight individuals, and its health benefits have been clearly determined [4,5], there are still no definitive data on what dietary protocols are most effective in both the short and long term [6] or even what is the correct nutritional approach in general [7].
The most commonly accepted dietary strategy is based on relatively high levels of carbohydrates and low fat content but according to some studies these low fat diets yield only modest weight loss and suffer from low long-term compliance [8]. In fact adherence of overweight/obese individuals to high carbohydrate/low fat nutrition is often a problem because the majority have been shown to have dietary preferences for foods with a rich fat content. Furthermore rather than consume complex carbohydrates there is a tendency to prefer highly processed food containing simple sugars [9-11] such that a low fat diet can actually encourage the consumption of sugars and refined carbohydrates that can worsen weight problems and also facilitate dyslipidemia especially in insulin resistance individuals [12,13]. As a consequence of the relative inefficacy of these types of diet there has been increased interest in recent years in very low carbohydrate ketogenic diets (VLCKD) [14] that, have undoubtedly been shown to be effective, at least in the short to medium term, as a tool to tackle obesity, hyperlipidemia and some cardiovascular risk factors [15-18]. The Mediterranean diet is often proposed as the healthy standard but many of the advantages associated with it can actually be linked to life style and the true original Mediterranean diet did not contemplate the current high levels of refined carbohydrates on which the typical Italian diet is based [19,20]. For this reason the standard ketogenic diet is not associated with high compliance in populations, like the Italian, that are used to carbohydrate based diets. The objective of the present study, which was devised as a case pilot trial, is to assess the safety, compliance and effects of a "Mediterranean style" ketogenic diet that utilizes very low carbohydrate ingredients formulated to simulate the aspect and taste of common carbohydrate rich foods (e.g. pasta) and which contain phytoextracts intended to add beneficial effects of important vegetable components. The aim of using herbal extracts during the diet period was to reduce some commonly reported light side effects of ketogenic diets. The parameters measured include blood biomarkers, body composition, weight loss and compliance in a cohort of council employees in Rome, Italy.
Methods
Subjects
The pilot study group consisted of 106 Rome council employees (19 males and 87 female; mean age 48, 49 ± 10, 33). Inclusion criteria were: BMI ≥ 25, age > 18 years and < 65 years, currently on a carbohydrate rich diet (> 50% energy), [21] desire to lose weight and health status suitable for a modified ketogenic diet (Tisanoreica®) [22] i.e. normal renal function, not pregnant or lactating. After the start of the experimental protocol the subjects who began new exercise programs or pharmaceutical treatments would be excluded. A small rise in transaminase was not considered a condition for exclusion since mild alterations in GOT and GPT values are common in obese individuals. Subjects eligible for the study were invited to the IPA clinic (Istituto di Previdenza ed Assistenza - health services for public sector employees) to attend an orientation session. At the first visit it was explained that during the first three weeks it was necessary to almost totally exclude carbohydrates and a detailed menu containing permitted and non-permitted foods was provided to each participant, along with the components of the ketogenic Mediterranean with phytoextracts (KEMEPHY) diet described below. Anthropometric measures were performed and blood samples were taken from the subjects two-three days before and after they began the diet. Subjects received no monetary compensation for their participation and signed a voluntary consent form before initiating the diet. The ethical and clinical review committee of IPA and the European Nutrition Society approved the study protocol, informed consent form and information material provided to subjects.
Diet
The KEMEPHY (ketogenic Mediterranean with phytoextracts) diet protocol was ketogenic during the first 3 weeks with approximately 34 g of CHO daily, using low carbohydrate high-protein meals and herbal teas [22] (Tisanoreica® by Gianluca Mech SpA, Orgiano VI) (Table 1 and 2).
Table 1.
diet composition in KEMEPHY (ketogenic Mediterranean with phytoextracts) diet.
| KEMEPHY Week 1-3 | KEMEPHY Week 4-6 | |
|---|---|---|
| Energy Kcal | 1098 ± 21.3 | 1186 ± 107 |
|
Protein, g/day (% daily Energy) |
99 (36) | 91 (31) |
| Carbohydrate, g/day (% daily Energy) | 34 (12) | 74 (25) |
|
Fat, g/day (% daily Energy) |
63 (52) | 58 (44) |
| Mean Kcal/die of the two phases of KEMEPHY | 1146 ± 88.8 | |
Kcal values are expressed in mean per day and SD. Other values are expressed in mean per day.
Table 2.
Plant extracts used in KEMEPHY (ketogenic Mediterranean with phytoextracts) diet
| Plant extracts | Week 1-3 | Week 4-6 | Composition |
|---|---|---|---|
| Extracts A, ml/day | 20 | 20 | Durvillea antarctica, black radish, mint, liquorice, artichoke, horsetail, burdock, dandelion, rhubarb, gentian, lemon balm, chinaroot, juniper, spear grass, elder, fucus, anise, parsley, bearberry, horehound |
| Extracts B, ml/day | 20 | 20 | Serenoa, Red clover, Chervil, Bean, Elder, Dandelion, Uncaria, Equisetum, Horehound, Rosemary |
| Extracts C, ml/day | 50 | 50 | Horsetail, asparagus, birch, cypress, couch grass, corn, dandelion, grape, fennel, elder, rosehip, anise |
| Extracts D, ml/day (only weeks 1 and 2) |
40 | 0 | Eleuthero, eurycoma longifolia, ginseng, corn, miura puama, grape, guaranà, arabic coffee, ginger |
The permitted foods were: cooked or raw green vegetables (200 g/meal), meat, fish and eggs (2 times/day), olive oil 40 g/day. Integration with a dish (PAT® i.e. porzione alimentare tisanoreica = tisanoreica nutritional portion) composed of high quality proteins (equivalent to 18 grams) and virtually zero carbohydrate (but that mimic their taste) was provided for every meal, for a maximum of four PATs per day. During the last three weeks complex carbohydrates were introduced (50-80 g/day), cheese (60 g/day), PAT was reduced from four to two, while the other indications remained unchanged. The distribution of nutrients (proteins, carbohydrates and fats) in terms of percentage of total caloric intake was 36%, 12% and 52%, respectively (weeks 1 to 3) and 31%, 25% and 44% (weeks 4 to 6). During the 6 weeks, the patients in the study group consumed 20 ml of extract A, 20 ml of extract B and 50 ml of extract C. During the first two weeks, before breakfast and lunch, they also consumed 40 ml of extract D (Tables 2 and 3).
Table 3.
Main actives ingredients of used phytoextracts, their reported beneficial effects and related references
| Extract | Main Active ingredients | Reported beneficial effects | Refs |
|---|---|---|---|
| A | Mint black radish burdock |
- indigestion - antioxidant - choleretic, increases bile secretion helping digestion |
[69] [70] |
| B | Serenoa Repens (saw palmetto) | hormonal regulating effects | [71] |
| White bean | alpha-amylase inhibitory properties and has been reported to aid weight loss and glycemic control | [72] [73] |
|
| C | Equisetum | Antioxidant diuretic glycemic control |
[74] [75] |
| Dandelion (Taraxacum officinale | diuretic | [76] | |
| D | Ginseng Miura Puama Guaranà |
Ameliorate the commonly reported symptoms of weakness and tiredness during the 1st phase of ketosis (1/2 weeks) | [77] [78] [79] |
Supplements
Subjects also took a daily (1 caplet each morning) multivitamin supplement [23] (containing Magnesium19 mg, Calcium 16 mg, Phosphorus 8 mg, Zinc 4.5 mg, Iron 4.62 mg, Manganese 1 mg, Potassium 0.5 mg, Copper 0.4 mg, Chromium 28.55 μg, Selenium 4 μg, Niacin 10 mg, Beta carotene 1.8 mg, Folic Acid 66 μg, Biotin 30 μg, Vitamin C 19.8 mg, Vitamin E 3.3 mg, Pantothenic Acid 1.98 mg, Vitamin B6 0.66 mg, Vitamin B2 0.53 mg, Vitamin B1 0.426 mg, Vitamin D3 1.65 μg, Vitamin B12 0.33 μg (Multivitaminico Balestra e Mech, Gianluca Mech SpA, Orgiano VI).
Measurements
Subjects were weighed at the same time of day at the start and after 6 weeks of the diet, using the same weighing scales (Digital Scale Joycare® Jc431). Fasting venous blood samples were collected at weeks 0 and 6 for total cholesterol (CHOLt), triacyglicerol (TG) high-density lipoprotein cholesterol (HDLc), low-density lipoprotein cholesterol (LDLc), glucose, blood urea nitrogen (BUN), uricemia, VES, creatinine, ALT, AST, GGT. Blood was collected in EDTA treated vacutainer tubes. To avoid interassay variation all blood samples were stored at -80° and analysed together at the end of the study. A separate sample of blood was clotted and serum analyzed for total cholesterol and triacylglycerols by photometric assay with an intra-assay and interassay CV values of 2% and 4%, respectively.
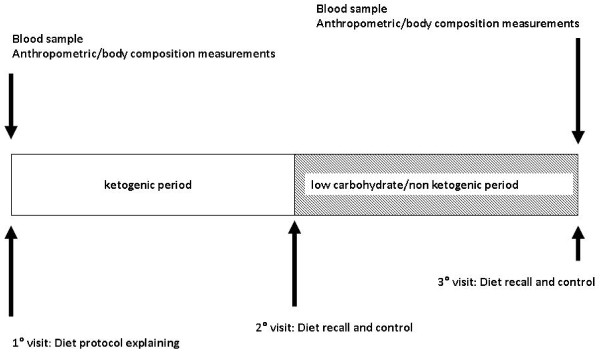
HDL cholesterol was determined using a homogenous enzyme immunoassay. The intra-assay and interassay CV values were both < 4%. Plasma glucose was determined colorimetrically using glucose oxidase methodology. Plasma urea nitrogen were measured using an enzymatic (urease), colorimetric method. Creatinine was measured colorimetrically using the picric acid assay, and uric acid was determined using a modified Trinder peroxide assay. LDLc fraction was calculated from Friedewald's formula: LDLc = TC - HDLc - (TG/5) [24]. Body composition was assessed using bioelectrical impedance analysis (BIA Akern Bioresearch, Pontassieve, FI, Italy) which is a non-invasive and portable method for the estimation of fluid compartments, fat and fat-free mass in healthy subjects. Bioelectrical impedance analysis was chosen for the analysis of body composition due to its reliability, safety, convenience and non-invasiveness making it optimal for the routine monitoring of body composition during the ketogenic diet [25,26]. The experimental design is showed in Figure 1.
Figure 1.
Experimental design.
Statistical analysis
We tested the normality and the assumption of homoscedasticity of all parameters at the start of the trial using the Kolmogorov-Sminorv and Shapiro-Wilk tests. To study changes over time and the effects of gender, we added an appropriate interaction analysis. As there were no significant gender interactions, the data of each group were pooled and analyzed together. A Student's t test was used to compare parameters before and after 6 weeks of the KEMEPHY diet using Statistica Software, ver. 8.0 (Tulsa, USA) and the software package GraphPad Prism version 4.00 for Windows, GraphPad Software, San Diego California USA. All data are expressed as mean ± standard deviation.
Results
Of the 106 persons recruited 87 completed the study (83, 6% compliance). Of the 19 subjects that withdrew 4 were for family/personal reasons, 8 subjects were lost to follow up and only 7 withdrew due to inadequate compliance to the diet protocol. Adjusted for causes of withdrawal only 7 of 104 were not able to follow the KEMEPHY diet for an adjusted compliance of 93.4%
Blood analysis did not reveal significant modification in ALT, AST, GGT and bilirubin values, nor were there significant variations in creatinine, uric acid, nitrogen and electrolytes (NA, K, Cl, Ca, Mg). There were significant changes in lipid profiles with reductions in triglycerides, total cholesterol and LDL along with a rise in HDL levels which all reached significance - see Table 4 and 5.
Table 4.
Blood biomarker, anthropometric and body composition values, separated for male and female, before and after 6 weeks KEMEPHY diet. Values are expressed in mean and SD.
| Pre diet female | Post diet female | Pre diet male |
Post diet male |
|
|---|---|---|---|---|
| BUN (mg/dl) | 33.4 ± 9.5 | 33.2 ± 8.7 | 35.2 ± 6.0 | 33.8 ± 7.0 |
| Uric acid (mg/dl) | 5.0 ± 1.3 | 5 ± 1.2 | 4.8 ± 1.3 | 5.0 ± 1.3 |
| VES (mm/hour) | 14 ± 7.3 | 12.7 ± 7.6 | 16 ± 7.3 | 14.4 ± 6.3 |
| Creatinine (mg/dL) | 0.84 ± 0.15 | 0.77 ± 0.11 | 0.83 ± 0.2 | 0.85 ± 0.2 |
| Total Cholesterol (mg/dl) | 206.2 ± 41.4 | 182.8 ± 34.3 | 199.2 ± 29.8 | 176.9 ± 26.1 |
| HDLc (mg/dl) | 46.7 ± 7.2 | 52.6 ± 9.5 | 43.9 ± 8.5 | 50.1 ± 9.1 |
| LDLc (mg/dl) | 151.8 ± 28.3 | 137.1 ± 24.8 | 140.9 ± 32.9 | 130.4 ± 25.9 |
| TG (mg/dl) | 119.9 ± 60.3 | 94.2 ± 41.8 | 114.1 ± 61.8 | 93.9 ± 46.2 |
| ALT (U/l) | 20.5 ± 10.9 | 17.3 ± 5.1 | 18.4 ± 4.6 | 19.1 ± 6.8 |
| AST (U/l) | 18.5 ± 5.4 | 17.1 ± 4.7 | 17.8 ± 3.8 | 17 ± 5.1 |
| GGT (U/l) | 20.5 ± 10.9 | 17.3 ± 5.1 | 21.5 ± 11.7 | 15.4 ± 4.1 |
| Glucose (mg/dl) | 95.7 ± 12.5 | 90.5 ± 9.8 | 95.9 ± 11.2 | 90.6 ± 8 |
| Weight Kg | 82.6 ± 12.7 | 76.3 ± 12.1 | 102.4 ± 22.2 | 93.4 ± 21 |
| BMI (Kg/m2) | 31 ± 4.8 | 28.7 ± 4.6 | 33.6 ± 6.2 | 30.6 ± 5.8 |
| % Fat | 42.3 ± 6, 8 | 36 ± 6, 9 | 37 ± 4.3 | 30.6 ± 4.1 |
| Waist circumference cm | 103.5 ± 14 | 94, 3 ± 10.3 | 120.8 ± 15.1 | 109.7 ± 14.1 |
| Hip circumference cm | 114.9 ± 11.6 | 107.2 ± 10.5 | 117.3 ± 9.9 | 111.2 ± 10.4 |
Table 5.
Blood biomarker values (all subjects) before and after the 6 week KEMEPHY diet. Values are expressed in mean and SD.
| Pre KEMEPHY diet | Post KEMEPHY diets | p | |
|---|---|---|---|
| BUN (mg/dl) | 33.8 ± 8.9 | 33.4 ± 8.4 | n.s. |
| Uric acid (mg/dl) | 4.9 ± 1.3 | 5 ± 1.2 | n.s. |
| VES (mm/hour) | 14.2 ± 7.2 | 12.8 ± 7.4 | n.s. |
| Creatinine (mg/dL) | 0.83 ± 0.16 | 0.78 ± 0.13 | n.s. |
| Total Cholesterol (mg/dl) | 204.2 ± 40 | 181.1 ± 33.4 | P < 0.0001 |
| HDLc (mg/dl) | 46.2 ± 7.4 | 52.1 ± 7.4 | P < 0.0001 |
| LDLc (mg/dl) | 149.7 ± 29.1 | 135.8 ± 24.8 | P < 0.0001 |
| TG (mg/dl) | 118.6 ± 59.9 | 93.8 ± 42.2 | P < 0.0001 |
| ALT (U/l) | 20.7 ± 9.1 | 18.2 ± 6.7 | n.s. |
| AST (U/l) | 18.4 ± 5.1 | 17 ± 5 | n.s. |
| GGT (U/l) | 21 ± 11 | 17 ± 5 | n.s. |
| Glucose (mg/dl) | 96 ± 12 | 91 ± 9 | P < 0.0001 |
Anthropometric and body composition measurements revealed an average weight loss of 6.72 kg or 7.8% (pre 86.15 ± 16.38 vs post 79.43 ± 15.31; p < 0.0001). Fat mass was reduced from 41.24 ± 6.74 to 34.99 ± 6.74, a fall of 15.1% (p < 0.0001). As in previous studies no significant differences were observed in total body water expressed as percent of body weight [22,27]. Waist circumference was reduced by an average of 9.46 cm from 106.56 ± 15.38 to 97.10 ± 12.69, a fall of 8.9% (p < 0.0001). There were also significant reductions in hip and thigh circumferences of 7.41 cm (from 115 ± 11.24 to 107.78 ± 10.39; p < 0.0001) and 3.32 cm respectively (from 58.65 ± 5.43 to 55.32 ± 4.90). Anthropometric and body composition results are showed in tables 6.
Table 6.
Anthropometric and body composition measures (all subjects) pre and post diet
| Pre KEMEPHY diet | Post KEMEPHY diets | p | |
|---|---|---|---|
| BMI (Kg/m2) | 31.5 ± 5.1 | 29. ± 4.8 | P < 0.0001 |
| Weight Kg | 86.2 ± 16.4 | 79.4 ± 15.3 | P < 0.0001 |
| % Fat | 41.2 ± 6.7 | 35 ± 6.7 | P < 0.0001 |
| Waist circumference cm | 106.6 ± 15.4 | 97.1 ± 12.7 | P < 0.0001 |
| Hip circumference cm | 115 ± 11.3 | 107.8 ± 10.4 | P < 0.0001 |
Discussion
Many weight loss diet procedures continue to focus on the reduction of fat content and controlled protein intake, however our results appear to demonstrate that the KEMEPHY diet, which is a modification of the VLCKD, is effective not only for weight and fat loss but also leads to improvements in the values of various biomarkers which are associated with increased risk of metabolic and cardiovascular disease. The weight loss effect of VLCKD diets may be caused by several factors:
1. Satiety effect of proteins leading to appetite reduction [28-36] in which also ketone bodies may have a role [37,38], although the mechanism is not clear [39];
2. Reduction in lipid synthesis and increased lipolysis mechanisms [40-44];
3. Reduction in at rest respiratory quotient and therefore an increase in fat metabolism for energy use [22,45];
4. Increased metabolic expenditure caused by gluconeogenesis and the thermic effect of proteins [46-51]
The beneficial effects on cardiovascular risk factors involve the reduction of blood triglycerides [17,18,22] and also the reduction of total and LDL cholesterol along with a rise in HDL cholesterol [17,18,22,52-54]. Furthermore the VLCKD can cause modifications in LDL-C particles leading to increased size [53] which may reduce cardiovascular risk since smaller LDL particles have been shown to be more atherogenic [55].
The cholesterol lowering effect of VLCKD is also mediated by the well known facilitating action of insulin on HMGCoA reductase and inhibition of the latter by cholesterol and fats [56]. Insulin then increases the production of endogenous cholesterol while exogenous cholesterol has the opposite effect [56].
The KEMEPHY diet protocol used in this study maintains some advantages of the Mediterranean diet such as the use of olive oil [21] and some vegetables [19] (selected to avoid stimulating insulin production) but at the same time by inducing a physiological ketosis [57] promotes beneficial modifications in cardiovascular risk factors and body composition [22]. The use of the phyoextracts in this study may have contributed to the absence of commonly reported mild effects of ketosis (e.g. weakness, constipation, bad breath, headache). During the first three weeks of the KEMEPHY diet subjects avoided fructose completely and during the second three weeks only a moderate amount of fructose, exclusively from fruit, and therefore together with starch, was permitted. As a matter of fact fructose may stimulate fat biosynthesis via mechanisms which are not yet fully characterised [58], also several studies have reported that excessive concentrations of fructose can induce some or all of the features of metabolic syndrome independently of energy intake. Clinical and epidemiologic data further suggest that excessive fructose intake can contribute to the causes of metabolic syndrome [59].
Adjusted compliance in this study was 93.4% which is higher than reported compliances of standard VLCK diets (in the 20% to 58% range). [22,60,61]. It is tempting to speculate that the inclusion of "carbohydrate-like" formulated foods is one of the reasons for high compliance - however this, along with the potential benefits of the phytoextracts, requires further verification in a future study with a matched control group.
Safety considerations
If we assimilate de facto, which is not always correct, ketogenic diets with high protein diets then the risks proposed by critics of this type of dietary approach are essentially those of possible kidney damage due to high levels of nitrogen excretion during protein metabolism which can cause an increase in glomerular pressure and hyper-filtration [27]. There is not wide agreement between studies however, some infer the possibility of renal damage from animal studies [62,63] while others, looking at both animal models and human studies propose that even high levels of protein in the diet do not damage renal function [64,65]. In subjects with intact renal function higher dietary protein levels caused some functional and morphological adaptations without negative effects [66]. Also it should be underlined that ketogenic diets are only relatively high in protein [49,67] and that some recent studies have demonstrated that VLCKD can even cause a regression of diabetic nephropathy in mice [68]. With regard to possible acidosis during VLCKD since the concentration of ketone bodies never rises above 8 mmol/l [40,42] this risk is virtually inexistent in subjects with healthy insulin function.
Conclusions
Some limitations to this study include the lack of a matched control group and the short trial period. There are though many studies that demonstrate that VLCK diets are more effective than low fat or standard Mediterranean diets, at least over the short term [21], and the main aims of the present initial study were to assess safety aspects, acceptance & palatability and weight loss & biomarker changes. We are able to conclude that at least in the short term it was able to lead to positive changes including the reduction of fasting blood glucose, improvements in lipid profiles, significant and rapid weight and fat loss with the preservation of lean mass. We also note a high level of compliance, whether this was due to specific unique features of the present diet requires confirmation in a future matched control trial.
Abbreviations
KEMEPHY: ketogenic Mediterranean with phytoextracts; VLCKD: very low carbohydrate ketogenic diet; high-density lipoprotein cholesterol; LDLc: low-density lipoprotein cholesterol; TG: Triglycerides; GLU: Glucose; BUN: Blood Urea Nitrogen; UA: Uric acid; ALT: Alanine aminotransferase; AST: Aspartate Aminotransferase; GGT Gamma-glutamyl transpeptidase; Cr: Creatinine.
Competing interests
This work was partially funded by Gianluca Mech SpA, Orgiano (VI), Italy. AP and LC research activity is funded by dept. of Human Anatomy and Physiology, University of Padova; KG research activity is funded by the Biomedical Engineering Laboratory, Institute of Communication and Computer Systems, National Technical University of Athens, Athens, Greece.
LC is scientific consultant for Gianluca Mech SpA, Orgiano (VI), Italy.
Investigators conducted the study in its entirety and maintained exclusive control of all data and analyses. The funding source had no involvement in any part of the recruitment of participants, study intervention, data collection, data analyses, interpretation of the data, or preparation or review of this manuscript.
Authors' contributions
AP was the main researcher and was responsible for study design, statistical analysis and interpretation of data and draft of manuscript. conceived the study, participated in its design, drafted the manuscript and performed the statistical analysis. LC was responsible for study design and acquisition of data. KG was was responsible for analysis and interpretation of data and helped to draft the manuscript. All authors read and approved the final manuscript.
Contributor Information
Antonio Paoli, Email: antonio.paoli@unipd.it.
Lorenzo Cenci, Email: cencilorenz@libero.it.
Keith A Grimaldi, Email: keith.grimaldi@gmail.com.
Acknowledgements
The authors thank the men and women who participated in this investigation.
References
- Finkelstein EA, Ruhm CJ, Kosa KM. Economic causes and consequences of obesity. Annu Rev Public Health. 2005;26:239–257. doi: 10.1146/annurev.publhealth.26.021304.144628. [DOI] [PubMed] [Google Scholar]
- Olshansky SJ, Passaro DJ, Hershow RC, Layden J, Carnes BA, Brody J, Hayflick L, Butler RN, Allison DB, Ludwig DS. A potential decline in life expectancy in the United States in the 21st century. N Engl J Med. 2005;352:1138–1145. doi: 10.1056/NEJMsr043743. [DOI] [PubMed] [Google Scholar]
- Koh-Banerjee P, Wang Y, Hu FB, Spiegelman D, Willett WC, Rimm EB. Changes in body weight and body fat distribution as risk factors for clinical diabetes in US men. Am J Epidemiol. 2004;159:1150–1159. doi: 10.1093/aje/kwh167. [DOI] [PubMed] [Google Scholar]
- Appel LJ, Champagne CM, Harsha DW, Cooper LS, Obarzanek E, Elmer PJ, Stevens VJ, Vollmer WM, Lin PH, Svetkey LP, Stedman SW, Young DR. Writing Group of the PREMIER Collaborative Research Group. Effects of comprehensive lifestyle modification on blood pressure control: main results of the PREMIER clinical trial. JAMA. 2003;289:2083–2093. doi: 10.1001/jama.289.16.2083. [DOI] [PubMed] [Google Scholar]
- Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM. Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346:393–403. doi: 10.1056/NEJMoa012512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nordmann AJ, Nordmann A, Briel M, Keller U, Yancy WS Jr, Brehm BJ, Bucher HC. Effects of low-carbohydrate vs low-fat diets on weight loss and cardiovascular risk factors: a meta-analysis of randomized controlled trials. Arch Intern Med. 2006;166:285–293. doi: 10.1001/archinte.166.3.285. [DOI] [PubMed] [Google Scholar]
- Chahoud G, Aude YW, Mehta JL. Dietary recommendations in the prevention and treatment of coronary heart disease: do we have the ideal diet yet? Am J Cardiol. 2004;94:1260–1267. doi: 10.1016/j.amjcard.2004.07.109. [DOI] [PubMed] [Google Scholar]
- Brehm BJ, Seeley RJ, Daniels SR, D'Alessio DA. A randomized trial comparing a very low carbohydrate diet and a calorie-restricted low fat diet on body weight and cardiovascular risk factors in healthy women. J Clin Endocrinol Metab. 2003;88:1617–1623. doi: 10.1210/jc.2002-021480. [DOI] [PubMed] [Google Scholar]
- Yeomans MR. Psychological approaches to under standing satiation and satiety. Agro Food High tech. 2010;21:16–19. [Google Scholar]
- Power ML, Schulkin J. Anticipatory physiological regulation in feeding biology: cephalic phase responses. Appetite. 2008;50:194–206. doi: 10.1016/j.appet.2007.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drewnowski A, Krahn DD, Demitrack MA, Nairn K, Gosnell BA. Taste responses and preferences for sweet high-fat foods: evidence for opioid involvement. Physiol Behav. 1992;51:371–379. doi: 10.1016/0031-9384(92)90155-U. [DOI] [PubMed] [Google Scholar]
- Feinman RD, Volek JS. Carbohydrate restriction as the default treatment for type 2 diabetes and metabolic syndrome. Scand Cardiovasc J. 2008;42:256–263. doi: 10.1080/14017430802014838. [DOI] [PubMed] [Google Scholar]
- Lankinen M, Schwab U, Gopalacharyulu PV, Seppanen-Laakso T, Yetukuri L, Sysi-Aho M, Kallio P, Suortti T, Laaksonen DE, Gylling H, Poutanen K, Kolehmainen M, Oresic M. Dietary carbohydrate modification alters serum metabolic profiles in individuals with the metabolic syndrome. Nutr Metab Cardiovasc Dis. 2010;20:249–257. doi: 10.1016/j.numecd.2009.04.009. [DOI] [PubMed] [Google Scholar]
- Atkins RC. Dr. Atkins' diet revolution; the high calorie way to stay thin forever. New York: D. McKay Co; 1972. [Google Scholar]
- Al-Khalifa A, Mathew TC, Al-Zaid NS, Mathew E, Dashti HM. Therapeutic role of low-carbohydrate ketogenic diet in diabetes. Nutrition. 2009;25:1177–1185. doi: 10.1016/j.nut.2009.04.004. [DOI] [PubMed] [Google Scholar]
- Dashti HM, Mathew TC, Khadada M, Al-Mousawi M, Talib H, Asfar SK, Behbahani AI, Al-Zaid NS. Beneficial effects of ketogenic diet in obese diabetic subjects. Mol Cell Biochem. 2007;302:249–256. doi: 10.1007/s11010-007-9448-z. [DOI] [PubMed] [Google Scholar]
- Adam-Perrot A, Clifton P, Brouns F. Low-carbohydrate diets: nutritional and physiological aspects. Obes Rev. 2006;7:49–58. doi: 10.1111/j.1467-789X.2006.00222.x. [DOI] [PubMed] [Google Scholar]
- Sharman MJ, Kraemer WJ, Love DM, Avery NG, Gomez AL, Scheett TP, Volek JS. A ketogenic diet favorably affects serum biomarkers for cardiovascular disease in normal-weight men. J Nutr. 2002;132:1879–1885. doi: 10.1093/jn/132.7.1879. [DOI] [PubMed] [Google Scholar]
- Bellisle F. Infrequently asked questions about the Mediterranean diet. Public Health Nutr. 2009;12:1644–1647. doi: 10.1017/S1368980009990498. [DOI] [PubMed] [Google Scholar]
- Cordain L, Eaton SB, Sebastian A, Mann N, Lindeberg S, Watkins BA, O'Keefe JH, Brand-Miller J. Origins and evolution of the Western diet: health implications for the 21st century. Am J Clin Nutr. 2005;81:341–354. doi: 10.1093/ajcn.81.2.341. [DOI] [PubMed] [Google Scholar]
- Perez-Guisado J, Munoz-Serrano A, Alonso-Moraga A. Spanish Ketogenic Mediterranean Diet: a healthy cardiovascular diet for weight loss. Nutr J. 2008;7:30. doi: 10.1186/1475-2891-7-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paoli A, Cenci L, Fancelli M, Parmagnani A, Fratter A, Cucchi A, Bianco A. Ketogenic diet and phytoextracts Comparison of the efficacy of Mediterranean, zone and tisanoreica diet on some health risk factors. Agro Food Ind Hi-Tech. 2010;21:24–29. [Google Scholar]
- Zupec-Kania B, Zupanc ML. Long-term management of the ketogenic diet: seizure monitoring, nutrition, and supplementation. Epilepsia. 2008;49(Suppl 8):23–26. doi: 10.1111/j.1528-1167.2008.01827.x. [DOI] [PubMed] [Google Scholar]
- Gazi IF, Elisaf M. LDL-cholesterol calculation formulas in patients with or without the metabolic syndrome. Int J Cardiol. 2007;119:414–415. doi: 10.1016/j.ijcard.2006.07.139. [DOI] [PubMed] [Google Scholar]
- Saunders NH, al-Zeibak S, Ryde SJ, Birks JL. The composition of weight loss in dieting obese females by electrical methods. Int J Obes Relat Metab Disord. 1993;17:317–322. [PubMed] [Google Scholar]
- Piccoli A, Pastori G, Codognotto M, Paoli A. Equivalence of information from single frequency v. bioimpedance spectroscopy in bodybuilders. Br J Nutr. 2007;97:182–192. doi: 10.1017/S0007114507243077. [DOI] [PubMed] [Google Scholar]
- Kreider RB, Rasmussen C, Kerksick CM, Wilborn C, Taylor L, Campbell B, Magrans-Courtney T, Fogt D, Ferreira M, Li R, Galbreath M, Iosia M, Cooke M, Serra M, Gutierrez J, Byrd M, Kresta JY, Simbo S, Oliver J, Greenwood M. A carbohydrate-restricted diet during resistance training promotes more favorable changes in body composition and markers of health in obese women with and without insulin resistance. Phys Sportsmed. 2011;39:27–40. doi: 10.3810/psm.2011.05.1893. [DOI] [PubMed] [Google Scholar]
- Westerterp-Plantenga MS, Nieuwenhuizen A, Tome D, Soenen S, Westerterp KR. Dietary protein, weight loss, and weight maintenance. Annu Rev Nutr. 2009;29:21–41. doi: 10.1146/annurev-nutr-080508-141056. [DOI] [PubMed] [Google Scholar]
- Paddon-Jones D, Westman E, Mattes RD, Wolfe RR, Astrup A, Westerterp-Plantenga M. Protein, weight management, and satiety. Am J Clin Nutr. 2008;87:1558S–1561S. doi: 10.1093/ajcn/87.5.1558S. [DOI] [PubMed] [Google Scholar]
- Soenen S, Westerterp-Plantenga MS. Proteins and satiety: implications for weight management. Curr Opin Clin Nutr Metab Care. 2008;11:747–751. doi: 10.1097/MCO.0b013e328311a8c4. [DOI] [PubMed] [Google Scholar]
- Veldhorst M, Smeets A, Soenen S, Hochstenbach-Waelen A, Hursel R, Diepvens K, Lejeune M, Luscombe-Marsh N, Westerterp-Plantenga M. Protein-induced satiety: effects and mechanisms of different proteins. Physiol Behav. 2008;94:300–307. doi: 10.1016/j.physbeh.2008.01.003. [DOI] [PubMed] [Google Scholar]
- Vogels N, Westerterp-Plantenga MS. Successful long-term weight maintenance: a 2-year follow-up. Obesity (Silver Spring) 2007;15:1258–1266. doi: 10.1038/oby.2007.147. [DOI] [PubMed] [Google Scholar]
- Bravata DM, Sanders L, Huang J, Krumholz HM, Olkin I, Gardner CD, Bravata DM. Efficacy and safety of low-carbohydrate diets: a systematic review. JAMA. 2003;289:1837–1850. doi: 10.1001/jama.289.14.1837. [DOI] [PubMed] [Google Scholar]
- Astrup A. The satiating power of protein--a key to obesity prevention? Am J Clin Nutr. 2005;82:1–2. doi: 10.1093/ajcn.82.1.1. [DOI] [PubMed] [Google Scholar]
- Soenen S, Plasqui G, Smeets AJ, Westerterp-Plantenga MS. Protein intake induced an increase in exercise stimulated fat oxidation during stable body weight. Physiol Behav. 2010;101:770–774. doi: 10.1016/j.physbeh.2010.08.019. [DOI] [PubMed] [Google Scholar]
- Soenen S, Westerterp-Plantenga MS. Changes in body fat percentage during body weight stable conditions of increased daily protein intake vs. control. Physiol Behav. 2010;101:635–638. doi: 10.1016/j.physbeh.2010.09.014. [DOI] [PubMed] [Google Scholar]
- Johnstone AM, Horgan GW, Murison SD, Bremner DM, Lobley GE. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am J Clin Nutr. 2008;87:44–55. doi: 10.1093/ajcn/87.1.44. [DOI] [PubMed] [Google Scholar]
- McClernon FJ, Yancy WS Jr, Eberstein JA, Atkins RC, Westman EC. The effects of a low-carbohydrate ketogenic diet and a low-fat diet on mood, hunger, and other self-reported symptoms. Obesity (Silver Spring) 2007;15:182–187. doi: 10.1038/oby.2007.516. [DOI] [PubMed] [Google Scholar]
- Cabeza De Vaca S, Holiman S, Carr KD. A search for the metabolic signal that sensitizes lateral hypothalamic self-stimulation in food-restricted rats. Physiol Behav. 1998;64:251–260. doi: 10.1016/S0031-9384(98)00050-X. [DOI] [PubMed] [Google Scholar]
- Cahill GF Jr. Fuel metabolism in starvation. Annu Rev Nutr. 2006;26:1–22. doi: 10.1146/annurev.nutr.26.061505.111258. [DOI] [PubMed] [Google Scholar]
- Sato K, Kashiwaya Y, Keon CA, Tsuchiya N, King MT, Radda GK, Chance B, Clarke K, Veech RL. Insulin, ketone bodies, and mitochondrial energy transduction. FASEB J. 1995;9:651–658. doi: 10.1096/fasebj.9.8.7768357. [DOI] [PubMed] [Google Scholar]
- Robinson AM, Williamson DH. Physiological roles of ketone bodies as substrates and signals in mammalian tissues. Physiol Rev. 1980;60:143–187. doi: 10.1152/physrev.1980.60.1.143. [DOI] [PubMed] [Google Scholar]
- Veldhorst MA, Westerterp-Plantenga MS, Westerterp KR. Gluconeogenesis and energy expenditure after a high-protein, carbohydrate-free diet. Am J Clin Nutr. 2009;90:519–526. doi: 10.3945/ajcn.2009.27834. [DOI] [PubMed] [Google Scholar]
- Vazquez JA, Kazi U. Lipolysis and gluconeogenesis from glycerol during weight reduction with very-low-calorie diets. Metabolism. 1994;43:1293–1299. doi: 10.1016/0026-0495(94)90225-9. [DOI] [PubMed] [Google Scholar]
- Langfort J, Pilis W, Zarzeczny R, Nazar K, Kaciuba-Uscilko H. Effect of low-carbohydrate-ketogenic diet on metabolic and hormonal responses to graded exercise in men. J Physiol Pharmacol. 1996;47:361–371. [PubMed] [Google Scholar]
- Feinman RD, Fine EJ. Nonequilibrium thermodynamics and energy efficiency in weight loss diets. Theor Biol Med Model. 2007;4:27. doi: 10.1186/1742-4682-4-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinman RD, Fine EJ. Whatever happened to the second law of thermodynamics? Am J Clin Nutr. 2004;80:1445–6. doi: 10.1093/ajcn/80.5.1445. author reply 1446. [DOI] [PubMed] [Google Scholar]
- Feinman RD, Fine EJ. "A calorie is a calorie" violates the second law of thermodynamics. Nutr J. 2004;3:9. doi: 10.1186/1475-2891-3-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinman RD, Fine EJ. Thermodynamics and metabolic advantage of weight loss diets. Metab Syndr Relat Disord. 2003;1:209–219. doi: 10.1089/154041903322716688. [DOI] [PubMed] [Google Scholar]
- Fine EJ, Feinman RD. Thermodynamics of weight loss diets. Nutr Metab (Lond) 2004;1:15. doi: 10.1186/1743-7075-1-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jequier E. Pathways to obesity. Int J Obes Relat Metab Disord. 2002;26(Suppl 2):S12–17. doi: 10.1038/sj.ijo.0802123. [DOI] [PubMed] [Google Scholar]
- Lofgren I, Zern T, Herron K, West K, Sharman MJ, Volek JS, Shachter NS, Koo SI, Fernandez ML. Weight loss associated with reduced intake of carbohydrate reduces the atherogenicity of LDL in premenopausal women. Metabolism. 2005;54:1133–1141. doi: 10.1016/j.metabol.2005.03.019. [DOI] [PubMed] [Google Scholar]
- Volek JS, Sharman MJ, Forsythe CE. Modification of lipoproteins by very low-carbohydrate diets. J Nutr. 2005;135:1339–1342. doi: 10.1093/jn/135.6.1339. [DOI] [PubMed] [Google Scholar]
- Volek JS, Sharman MJ, Gomez AL, Scheett TP, Kraemer WJ. An isoenergetic very low carbohydrate diet improves serum HDL cholesterol and triacylglycerol concentrations, the total cholesterol to HDL cholesterol ratio and postprandial pipemic responses compared with a low fat diet in normal weight, normolipidemic women. J Nutr. 2003;133:2756–2761. doi: 10.1093/jn/133.9.2756. [DOI] [PubMed] [Google Scholar]
- Austin MA, King MC, Vranizan KM, Krauss RM. Atherogenic lipoprotein phenotype. A proposed genetic marker for coronary heart disease risk. Circulation. 1990;82:495–506. doi: 10.1161/01.CIR.82.2.495. [DOI] [PubMed] [Google Scholar]
- Siliprandi N, Tettamanti G. Biochimica medica: strutturale, metabolica e funzionale. Padova: Piccin; 2005. [Google Scholar]
- Krebs HA. The regulation of the release of ketone bodies by the liver. Adv Enzyme Regul. 1966;4:339–354. doi: 10.1016/0065-2571(66)90027-6. [DOI] [PubMed] [Google Scholar]
- Samuel VT. Fructose induced lipogenesis: from sugar to fat to insulin resistance. Trends Endocrinol Metab. 2011;22:60–65. doi: 10.1016/j.tem.2010.10.003. [DOI] [PubMed] [Google Scholar]
- Madero M, Arriaga JC, Jalal D, Rivard C, McFann K, Perez-Mendez O, Vazquez A, Ruiz A, Lanaspa MA, Jimenez CR, Johnson RJ, Lozada LG. The effect of two energy-restricted diets, a low-fructose diet versus a moderate natural fructose diet, on weight loss and metabolic syndrome parameters: a randomized controlled trial. Metabolism; 2011. in press . [DOI] [PubMed] [Google Scholar]
- Klein P, Janousek J, Barber A, Weissberger R. Ketogenic diet treatment in adults with refractory epilepsy. Epilepsy Behav. 2010;19:575–579. doi: 10.1016/j.yebeh.2010.09.016. [DOI] [PubMed] [Google Scholar]
- Mosek A, Natour H, Neufeld MY, Shiff Y, Vaisman N. Ketogenic diet treatment in adults with refractory epilepsy: a prospective pilot study. Seizure. 2009;18:30–33. doi: 10.1016/j.seizure.2008.06.001. [DOI] [PubMed] [Google Scholar]
- Jia Y, Hwang SY, House JD, Ogborn MR, Weiler HA, O K, Aukema HM. Long-term high intake of whole proteins results in renal damage in pigs. J Nutr. 2010;140:1646–1652. doi: 10.3945/jn.110.123034. [DOI] [PubMed] [Google Scholar]
- Wakefield AP, House JD, Ogborn MR, Weiler HA, Aukema HM. A diet with 35% of energy from protein leads to kidney damage in female Sprague-Dawley rats. Br J Nutr. 2011. pp. 656–63. [DOI] [PubMed]
- Skov AR, Haulrik N, Toubro S, Molgaard C, Astrup A. Effect of protein intake on bone mineralization during weight loss: a 6-month trial. Obes Res. 2002;10:432–438. doi: 10.1038/oby.2002.60. [DOI] [PubMed] [Google Scholar]
- Martin WF, Armstrong LE, Rodriguez NR. Dietary protein intake and renal function. Nutr Metab (Lond) 2005;2:25. doi: 10.1186/1743-7075-2-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welle S, Nair KS. Relationship of resting metabolic rate to body composition and protein turnover. Am J Physiol. 1990;258:E990–998. doi: 10.1152/ajpendo.1990.258.6.E990. [DOI] [PubMed] [Google Scholar]
- Westerterp-Plantenga MS. How are normal, high- or low-protein diets defined? Br J Nutr. 2007;97:217–218. doi: 10.1017/S0007114507381348. [DOI] [PubMed] [Google Scholar]
- Poplawski MM, Mastaitis JW, Isoda F, Grosjean F, Zheng F, Mobbs CV. Reversal of diabetic nephropathy by a ketogenic diet. PLoS One. 2011;6:e18604. doi: 10.1371/journal.pone.0018604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lugasi A, Blazovics A, Hagymasi K, Kocsis I, Kery A. Antioxidant effect of squeezed juice from black radish (Raphanus sativus L. var niger) in alimentary hyperlipidaemia in rats. Phytother Res. 2005;19:587–591. doi: 10.1002/ptr.1655. [DOI] [PubMed] [Google Scholar]
- Lou Z, Wang H, Li J, Chen S, Zhu S, Ma C, Wang Z. Antioxidant activity and chemical composition of the fractions from burdock leaves. J Food Sci. 2010;75:C413–419. doi: 10.1111/j.1750-3841.2010.01616.x. [DOI] [PubMed] [Google Scholar]
- Di Silverio F, D'Eramo G, Lubrano C, Flammia GP, Sciarra A, Palma E, Caponera M, Sciarra F. Evidence that Serenoa repens extract displays an antiestrogenic activity in prostatic tissue of benign prostatic hypertrophy patients. Eur Urol. 1992;21:309–314. doi: 10.1159/000474863. [DOI] [PubMed] [Google Scholar]
- Barrett ML, Udani JK. A proprietary alpha-amylase inhibitor from white bean (Phaseolus vulgaris): a review of clinical studies on weight loss and glycemic control. Nutr J. 2011;10:24. doi: 10.1186/1475-2891-10-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Celleno L, Tolaini MV, D'Amore A, Perricone NV, Preuss HG. A Dietary supplement containing standardized Phaseolus vulgaris extract influences body composition of overweight men and women. Int J Med Sci. 2007;4:45–52. doi: 10.7150/ijms.4.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mimica-Dukic N, Simin N, Cvejic J, Jovin E, Orcic D, Bozin B. Phenolic compounds in field horsetail (Equisetum arvense L.) as natural antioxidants. Molecules. 2008;13:1455–1464. doi: 10.3390/molecules13071455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Safiyeh S, Fathallah FB, Vahid N, Hossine N, Habib SS. Antidiabetic effect of Equisetum arvense L. (Equisetaceae) in streptozotocin-induced diabetes in male rats. Pak J Biol Sci. 2007;10:1661–1666. doi: 10.3923/pjbs.2007.1661.1666. [DOI] [PubMed] [Google Scholar]
- Clare BA, Conroy RS, Spelman K. The diuretic effect in human subjects of an extract of Taraxacum officinale folium over a single day. J Altern Complement Med. 2009;15:929–934. doi: 10.1089/acm.2008.0152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pieralisi G, Ripari P, Vecchiet L. Effects of a standardized ginseng extract combined with dimethylaminoethanol bitartrate, vitamins, minerals, and trace elements on physical performance during exercise. Clin Ther. 1991;13:373–382. [PubMed] [Google Scholar]
- Piato AL, Detanico BC, Linck VM, Herrmann AP, Nunes DS, Elisabetsky E. Anti-stress effects of the "tonic"Ptychopetalum olacoides (Marapuama) in mice. Phytomedicine. 2010;17:248–253. doi: 10.1016/j.phymed.2009.07.001. [DOI] [PubMed] [Google Scholar]
- Lima WP, Carnevali LC Jr, Eder R, Costa Rosa LF, Bacchi EM, Seelaender MC. Lipid metabolism in trained rats: effect of guarana (Paullinia cupana Mart.) supplementation. Clin Nutr. 2005;24:1019–1028. doi: 10.1016/j.clnu.2005.08.004. [DOI] [PubMed] [Google Scholar]