Abstract
Arabinogalactan-proteins (AGPs) are a class of hyperglycosylated, hydroxyproline-rich glycoproteins that are widely distributed in the plant kingdom. AtAGP17, 18 and 19 are homologous genes encoding three classical lysine-rich AGPs in Arabidopsis. We observed subcellular localization of AtAGP18 at the plasma membrane by expressing a translational fusion gene construction of AtAGP18 attached to a green fluorescent protein (GFP) tag in Arabidopsis plants. We also overexpressed AtAGP18 without the GFP tag in Arabidopsis plants, and the resulting transgenic plants had a short, bushy phenotype. Here we discuss putative roles of AtAGP18 as a glycosylphosphatidylinositol (GPI)-anchored protein involved in a signal transduction pathway regulating plant growth and development.
Key words: Arabidopsis thaliana, arabinogalactan-proteins, co-receptor, glycosylphosphatidylinositol, lipid rafts, overexpression
Arabinogalactan-proteins (AGPs) are plant cell surface glycoproteins or proteoglycans which are thought to play important roles in various aspects of plant growth and development, such as somatic embryogenesis, cell proliferation and elongation, pattern formation and hormone signaling.1 The lysine-rich classical AGP subfamily in Arabidopsis contains three members: AtAGP17, 18 and 19. The subcellular localization of AtAGP17 and AtAGP18 was previously studied in our laboratory by expressing GFP-AtAGP17/18 fusion proteins in tobacco cell cultures.2,3 In a recent report, we used Arabidopsis plants to overexpress GFP-AtAGP17/18/19 fusion proteins to observe subcellular localization of the lysine-rich AGPs in planta, in contrast to our previous plant cell culture work.4 Moreover, the lysine-rich AGPs alone (i.e., AtAGP17/18/19 without the GFP tag) were overexpressed in Arabidopsis plants, and only AtAGP18 overexpressors had a distinctive phenotype. This phenotype included shorter stems, more branches and less seeds, indicating a role for AtAGP18 in plant growth and development.4 In this addendum, we further discuss the putative biological role of AtAGP18 on a molecular level and its possible mode of action in cellular signaling.
Classical AGPs are frequently predicted to have a glycosylphosphatidylinositol (GPI) anchor, which would allow for the localization of such AGPs to the outer surface of the plasma membrane. Biochemical analyses were carried out to support this hypothesis in tobacco, pear,5 rose6 and Arabidopsis.7 The lysine-rich classical AGPs, AtAGP17 and 18, were predicted to have a GPI anchor.8 To test this idea, tobacco cell cultures expressing GFP-AtAGP17/18 fusion proteins were plasmolyzed and GFP fluorescence was observed on the plasma membrane.2,3 To corroborate this finding in planta, GFP-AtAGP17/18 were expressed in Arabidopsis plants and leaf trichome cells were plasmolyzed. Enhanced GFP fluorescence was observed at the plasma membrane of these transgenic trichome cells, indicating the presence of GFP-AtAGP17/18 at the plasma membrane.4 The localization of these lysine-rich classical AGPs at the plasma membrane suggests possible biological roles in sensing extracellular signals. They are likely associated with lipid rafts involved in cell signaling for the following reasons. In plants as well as animals, there are sterol-enriched, detergent-resistant plasma membrane microdomains called lipid rafts. Lipid rafts are known to be involved in signal transduction and are enriched in transmembrane receptors and GPI-anchored proteins, including AGPs.9–11 The accumulation of these proteins in such microdomains may allow for interactions between these proteins in sensing extracellular signals which lead to various intracellular events. Interestingly, a recent study shows that lipid rafts from hybrid aspen cells contain callose synthase and cellulose synthase, and these enzymes are active since in vitro polysaccharide synthesis by the isolated detergent-resistant membranes was observed. These results demonstrate that lipid rafts are involved in cell wall polysaccharide biosynthesis.12 In addition, an Arabidopsis pnt mutant study shows GPI-anchored proteins are required in cell wall synthesis and morphogenesis.13 These observations, coupled with previous observations that cellulose synthases as well as AGPs interact with microtubules, suggest that AGPs in lipid rafts may have a role in signal events, including those regulating cellulose and/or callose biosynthesis or deposition.14,15
To examine the role of LeAGP-1, a lysine-rich AGP in tomato, transgenic tomato plants were produced which expressed GFP-LeAGP-1 under the control of the cauliflower mosaic virus 35S promoter.16 The tomato LeAGP-1 overexpressors and Arabidopsis AtAGP18 overexpressors both have a bushy phenotype similar to transgenic tobacco plants overproducing cytokinins.4,16,17 Cytokinins are an important class of plant hormones involved in many plant growth and development processes, such as cell growth and division, differentiation and other physiological processes.18 Therefore, Sun et al. proposed that LeAGP-1 might function in concert with the cytokinin signal transduction pathway.16 Since the overexpression phenotypes of AtAGP18 are similar to those of LeAGP-1, AtAGP18 is also likely associated with the cytokinin signal transduction pathway. The prevailing model for cytokinin signaling in Arabidopsis is similar to the two-component system in bacteria and yeast. In this model, the cytokinin receptor contains an extracellular domain, a kinase domain and a receiver domain. When the cytokinin receptor senses cytokinin signals, it auto-phosphorylates at a His residue in the kinase domain. The phosphoryl group is then transferred to an Asp residue in the receiver domain. Subsequently, the phosphoryl group is transferred to a His residue in the histidine phosphotransfer protein (Hpt) and the Hpt translocates to the nucleus and transfers the phosphoryl group to an Asp residue in a downstream response regulator to activate it.19 This model is consistent with our hypothesis since the cytokinin receptor in this model is a receptor kinase located in the plasma membrane with an extra-cellular domain that can potentially interact with AtAGP18. AtAGP18 may function as a co-receptor that first binds to cytokinins, then either directly interacts with cytokinin receptors or brings the cytokinins to cytokinin receptors in the plasma membrane. The first scenario is analogous to the interaction of contactin and contactin-associated protein (Caspr) in neurons. In this model, contactin is a GPI-anchored protein on the cell surface that binds to signal molecules and interacts with the transmembrane receptor Caspr to transmit signals to the cell interior.20 The second scenario is analogous to fibroblast growth factor (FGF) signal activation in which heparan sulfate proteoglycans bind to FGF molecules and bring them to the FGF receptor.21
Based on all the above observations and findings, a hypothetical model for AtAGP18 function is proposed in Figure 1. The model shows AtAGP18 located on the outer surface of the plasma membrane in lipid rafts where it could act as a co-receptor to sense extracellular signals (such as cytokinin) and interact with transmembrane proteins, possibly receptor kinases or ion channels, in the lipid rafts to initiate signaling by triggering various intracellular events. Interestingly, receptor tyrosine kinases and ion channels are known to be present in lipid rafts.9,22 Moreover, AGPs are likely associated with ion channels since addition of the AGP-binding reagent Yariv phenylglycoside resulted in elevated cytoplasmic calcium concentrations in tobacco cells and lily pollen tubes.15,23,24 Clearly, additional work will be required to verify such a model, and to better understand how AtAGP18 might sense extracellular signals and interact with the transmembrane proteins in the lipid rafts.
Figure 1.
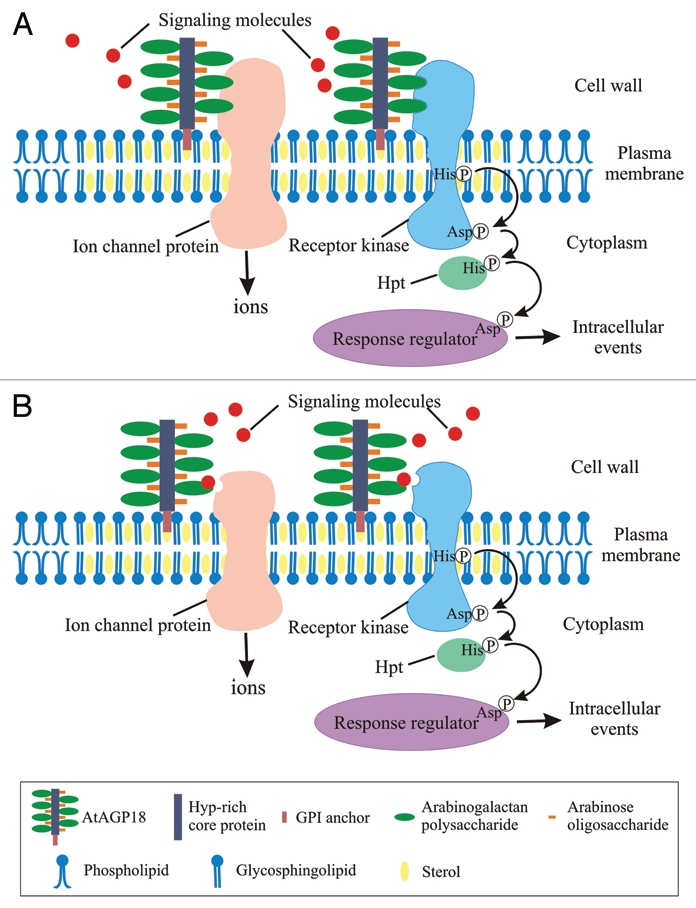
Model for atAGP18 functioning in cellular signaling to control plant growth and development. In this model, lipid rafts are enriched in glycosphingolipids, sterols, transmembrane proteins (such as receptors, receptor kinases and ion channel proteins) and GPI-anchored proteins including AtAGP18. (a) AtAGP18 acts as a co-receptor by binding to signaling molecules and directly interacting with transmembrane proteins in the lipid rafts. (B) AtAGP18 acts as a co-receptor by binding to signaling molecules and bringing the signaling molecules to transmembrane proteins in the lipid rafts. Upon activation by the extracellular signals, the transmembrane proteins initiate signaling and lead to various intracellular events (e.g., phosphorylation similar to the two-component signaling system, influx of calcium ions). The different components of the AtAGP18 molecule and the various lipid components of lipid rafts and plasma membrane are shown in the boxed inset. Hpt, histidine phosphotransfer protein.
Acknowledgements
This work was supported by the National Science Foundation (IBN-0110413).
Abbreviations
- AGPs
arabinogalactan-proteins
- Caspr
contactin-associated protein
- FGF
fibroblast growth factor
- GFP
green fluorescent protein
- GPI
glycosylphosphatidylinositol
- Hpt
histidine phosphotransfer protein
References
- 1.Seifert GJ, Roberts K. The biology of arabinogalactan proteins. Annu Rev Plant Biol. 2007;58:137–161. doi: 10.1146/annurev.arplant.58.032806.103801. [DOI] [PubMed] [Google Scholar]
- 2.Sun W, Xu J, Yang J, Kieliszewski MJ, Showalter AM. The lysine-rich arabinogalactan-protein subfamily in Arabidopsis: gene expression, glycoprotein purification and biochemical characterization. Plant Cell Physiol. 2005;46:975–984. doi: 10.1093/pcp/pci106. [DOI] [PubMed] [Google Scholar]
- 3.Yang J, Showalter AM. Expression and localization of AtAGP18, a lysine-rich arabinogalactan-protein in Arabidopsis. Planta. 2007;226:169–179. doi: 10.1007/s00425-007-0478-2. [DOI] [PubMed] [Google Scholar]
- 4.Zhang Y, Yang J, Showalter AM. AtAGP18 is localized at the plasma membrane and functions in plant growth and development. Planta. 2011;233:675–683. doi: 10.1007/s00425-010-1331-6. [DOI] [PubMed] [Google Scholar]
- 5.Youl JJ, Bacic A, Oxley D. Arabinogalactan-proteins from Nicotiana alata and Pyrus communis contain glycosylphosphatidylinositol membrane anchors. Proc Natl Acad Sci USA. 1998;95:7921–7926. doi: 10.1073/pnas.95.14.7921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Svetek J, Yadav MP, Nothnagel EA. Presence of a glycosylphosphatidylinositol lipid anchor on rose arabinogalactan proteins. J Biol Chem. 1999;274:14724–14733. doi: 10.1074/jbc.274.21.14724. [DOI] [PubMed] [Google Scholar]
- 7.Sherrier DJ, Prime TA, Dupree P. Glycosylphosphatidylinositol-anchored cell-surface proteins from Arabidopsis. Electrophoresis. 1999;20:2027–2035. doi: 10.1002/(SICI)1522-2683(19990701)20:10<2027::AID-ELPS2027>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 8.Schultz CJ, Rumsewicz MP, Johnson KL, Jones BJ, Gaspar YM, Bacic A. Using genomic resources to guide research directions. The arabinogalactan protein gene family as a test case. Plant Physiol. 2002;129:1448–1463. doi: 10.1104/pp.003459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pike LJ. Lipid rafts: bringing order to chaos. J Lipid Res. 2003;44:655–667. doi: 10.1194/jlr.R200021-JLR200. [DOI] [PubMed] [Google Scholar]
- 10.Johnson KL, Ingram GC. Sending the right signals: regulating receptor kinase activity. Curr Opinion Plant Biol. 2005;8:648–656. doi: 10.1016/j.pbi.2005.09.007. [DOI] [PubMed] [Google Scholar]
- 11.Borner GH, Sherrier DJ, Weimar T, Michaelson LV, Hawkins ND, Macaskill A, et al. Analysis of detergent-resistant membranes in Arabidopsis. Evidence for plasma membrane lipid rafts. Plant Physiol. 2005;137:104–116. doi: 10.1104/pp.104.053041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bessueille L, Sindt N, Guichardant M, Djerbi S, Teeri TT, Bulone V. Plasma membrane microdomains from hybrid aspen cells are involved in cell wall polysaccharide biosynthesis. Biochem J. 2009;420:93–103. doi: 10.1042/BJ20082117. [DOI] [PubMed] [Google Scholar]
- 13.Gillmor CS, Lukowitz W, Brininstool G, Sedbrook JC, Hamann T, Poindexter P, et al. Glycosylphosphatidylinositol-anchored proteins are required for cell wall synthesis and morphogenesis in Arabidopsis. Plant Cell. 2005;17:1128–1140. doi: 10.1105/tpc.105.031815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sardar HS, Yang J, Showalter AM. Molecular interactions of arabinogalactan proteins with cortical microtubules and F-actin in bright yellow-2 tobacco cultured cells. Plant Physiol. 2006;142:1469–1479. doi: 10.1104/pp.106.088716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Driouich A, Baskin TI. Intercourse between cell wall and cytoplasm exemplified by arabinogalactan proteins and cortical microtubules. Am J Bot. 2008;95:1491–1497. doi: 10.3732/ajb.0800277. [DOI] [PubMed] [Google Scholar]
- 16.Sun W, Kieliszewski MJ, Showalter AM. Overexpression of tomato LeAGP-1 arabinogalactan-protein promotes lateral branching and hampers reproductive development. Plant J. 2004;40:870–881. doi: 10.1111/j.1365-313X.2004.02274.x. [DOI] [PubMed] [Google Scholar]
- 17.Li Y, Hagen G, Guilfoyle TJ. Altered morphology in transgenic tobacco plants that overproduce cytokinins in specific tissues and organs. Dev Biol. 1992;153:386–395. doi: 10.1016/0012-1606(92)90123-x. [DOI] [PubMed] [Google Scholar]
- 18.Sakakibara H. Cytokinins: activity, biosynthesis and translocation. Annu Rev Plant Biol. 2006;57:431–449. doi: 10.1146/annurev.arplant.57.032905.105231. [DOI] [PubMed] [Google Scholar]
- 19.Ferreira FJ, Kieber JJ. Cytokinin signaling. Curr Opinion Plant Biol. 2005;8:518–525. doi: 10.1016/j.pbi.2005.07.013. [DOI] [PubMed] [Google Scholar]
- 20.Peles E, Nativ M, Lustig M, Grumet M, Schilling J, Martinez R, et al. Identification of a novel contactinassociated transmembrane receptor with multiple domains implicated in protein-protein interactions. EMBO J. 1997;16:978–988. doi: 10.1093/emboj/16.5.978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schlessinger J, Lax I, Lemmon M. Regulation of growth factor activation by proteoglycans: what is the role of the low affinity receptors? Cell. 1995;83:357–360. doi: 10.1016/0092-8674(95)90112-4. [DOI] [PubMed] [Google Scholar]
- 22.Maguy A, Hebert TE, Nattel S. Involvement of lipid rafts and caveolae in cardiac ion channel function. Cardiovasc Res. 2006;69:798–807. doi: 10.1016/j.cardiores.2005.11.013. [DOI] [PubMed] [Google Scholar]
- 23.Pickard BG, Fujiki M. Ca2+ pulsation in BY-2 cells and evidence for control of mechanosensory calcium-selective channels by the plasmalemmal reticulum. Funct Plant Biol. 2005;32:863–879. doi: 10.1071/FP05045. [DOI] [PubMed] [Google Scholar]
- 24.Roy SJ, Holdaway-Clarke TL, Hackett GR, Kunkel JG, Lord EM, Hepler PK. Uncoupling secretion and tip growth in lily pollen tubes: Evidence for the role of calcium in exocytosis. Plant J. 1999;19:379–386. doi: 10.1046/j.1365-313x.1999.00515.x. [DOI] [PubMed] [Google Scholar]


