Abstract
African Americans are more susceptible to develop insulin resistance, obesity, Type 2 Diabetes, and coronary heart disease (CHD), and systemic inflammation is central to the pathophysiology of these chronic diseases. African Americans are also more likely to contract H. pylori (cagA) infections during their childhood. However, the contribution of H. pylori infection to the degree of overall systemic inflammation in these chronic diseases is not known. Therefore, we studied 46 apparently healthy African Americans, over 40 years of age who were, infected with H. pylori (cagA). These volunteers were assessed at baseline and after treatment with triple regimen drug therapy to eradicate H. pylori. All but 3 subjects were found to be free of this infection by urea breath test (UBT) after the treatment period. No hyperhomocysteinemia was found in these subjects and there were no significant changes in the level of homocysteine (tHcy), folate and B12; however, CRP levels measured by high sensitivity assay showed a significant (p=0.02) decrease 2 months after the eradication. We further stratified CRP values according to the BMI < 27 and > 27. There was more profound reduction in CRP in the more obese group (i.e., BMI>27) from 54.26 ± 23.67 to 18.73 ± 17.39 mg/l (p=0.01), compared with the leaner subjects in whom CRP decreases from 8.88 ± 6.23 to 4.94 ± 6.21 mg/L (p=0.04), after eradication of the H. pylori (cagA) infection. The level of CRP, however, remained significantly higher in the obese subjects even after the eradication of this infection, indicative of a smaller residual influence of adiposity on CRP. Thus, a major component of systemic inflammation in African Americans may be attributable to chronic H. pylori infection.
Keywords: H. pylori, Inflammation, Homocysteine, Obesity and African Americans
Introduction
Interest and investigations have been mounting to support the idea that atherosclerosis results, in part, from an inflammatory process. Systemic inflammation as a risk factor for CHD occurs in the context of insulin resistance and associated metabolic and clinical trait, which in aggregate are referred to as the Metabolic Syndrome. Ross1 postulated possible mechanisms as to how several important inflammatory markers could be used as predictors of CHD. In recent years, the association of CHD with inflammation induced by viral and/or bacterial agents, such as Chlamydia pneumoniae and Helicobacter pylori (H. pylori), is emerging as a research area of great importance2. H. pylori is a gram-negative bacterium that colonizes the gastric mucosa and can result in gastritis, gastroduodenal ulcerations, gastric adenocarcinoma, or mucosa-associated lymphoid tissue lymphoma in an estimated 10-20% of those infected3. This bacterium infects at least half of the world's population4. Recent evidence suggests that infection with H. pylori, particularly those strains containing the specific virulence factor cytotoxin-associated gene A (cagA), may be associated with CHD in humans5. H. pylori is capable of invoking a host of inflammatory responses including elevations in tumor necrosis factor-alpha (TNF-α), interleukin-1b, C-reactive protein (CRP), lipid-peroxides, homocysteinemia (tHcy) and intercellular- and vascular-cell adhesion molecules (ICAM-1 and VCAM-1, respectively)6. Farmer and Torre-Amione7 produced evidence to show that tHcy is a clinical marker of inflammation, which had already been proven to be an etiological factor for vascular diseases8.
Recently we reported9 a highly significant difference in the tHcy status between the H. pylori positive and negative groups of CHD patients (14.4±5.9 and 10.4±3.9 μmol/L, p>0.001). From a cross-sectional data analyses, Schreiner et al.10 also showed that a higher level of tHcy in African Americans is associated with several prothrombotic factors related to cardiovascular disease. Concomitantly, African Americans have also been reported to have a higher rate of infection with H. pylori (cagA) than Caucasians4. This, therefore, prompted us to conduct an intervention study in an attempt to determine if H. pylori (cagA) infection has any causal role in elevating systemic inflammation in African Americans.
Materials and Methods
For this intervention study, asymptomatic male and female African American subjects were recruited who were greater than 40 years of age, and who were infected with H. pylori (cagA). Exclusion criteria included alcohol abuse and cigarette smoking; gastric diseases or symptomatology; treatment with antibiotics less than 8 weeks prior to screening, having any other infections, cancer, diabetes, and CHD. About 781 subjects were screened and 218 African Americans satisfied inclusion/exclusion criteria; out of these, 46 subjects were detected to be positive for H. pylori (cagA) infection. This represents a prevalence rate of about 21%, which falls within the range of prevalence rate of H. pylori infection in Caucasians in developed countries4. The IgG antibodies to H. pylori were measured by an enzyme linked immunosorbent assay (ELISA) method and cagA was assessed by the Western blot analysis. The protocol was approved by the IRB at UAB and informed consent was obtained from each volunteer,
A triple regimen drug therapy that includes omeprazole, 20 mg BID for 2 weeks then once a day for 2 weeks; amoxycillin, 500 mg 4 times daily for 2 weeks, and tinidazole (tindamax, an analogue of metronidazole) 500 mg BID for 2 weeks, was dispensed to each subject. This drug regimen was reported to be effective and less expensive11. Their BMI was obtained using TANITA body composition analyzer on the day of the first visit. Subjects reported back to the clinic 2 months after completing the treatment; a time period good enough to eliminate any influence of antibiotics12. Urea breath test (UBT) was performed using urea breath test to kit by metabolic solution Inc. Nashua, NH to make sure a complete eradication of the H. pylori (cagA) infection. Only 3 subjects were identified still to be H. pylori positive, giving about 94% cure rate in agreement with the previous studies11. The pre- and post-treatment blood samples were analyzed for folate and vitamin B12 levels using the SimulTRAC-SNB radioassay kit by MP diagnostics division Orangeburg, NY. The level of tHcy was measured by fluorescence polarization immunoassay on an Abbott IMX analyzer, while the CRP was measured using a high sensitivity C-reactive protein ELISA method manufactured by ALPCO Diagnostics, Salem, NH. Data were analyzed using SAS (SAS Institute Inc., Cary, NC, USA). Patient's pre and post treatment CRP-values were compared in two BMI groups using the unpaired t-test or Fisher's exact test. P-values of less than 0.05 were considered to indicate statistical significance.
Results
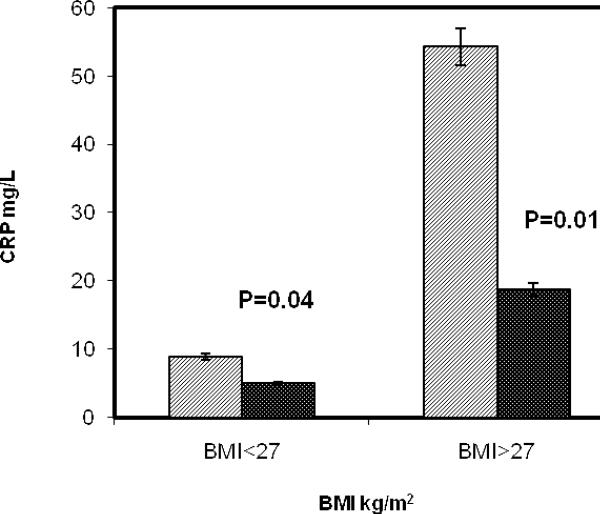
Demographic, inflammatory, and nutritional parameters of African Americans, obtained before and 2 months after the eradication of their H. pylori (cagA) infection, are listed in Table 1. It is interesting to note that the tHcy level falls within the normal range (6.7-12 μmol/L) for the H. pylori (cagA) seropositive African Americans. The levels of folate and B12 also did not change significantly upon the eradication of H. pylori. The baseline blood level of high- sensitivity CRP was found to be significantly higher (see Table 1) in African Americans infected with this organism. The CRP values for the non-obese and obese groups prior to H. pylori eradication (shown in Figure 1) were respectively 8.88 ± 6.23 and 54.26 ± 23.67 mg/L. Following treatment of H. pylori, the level of CRP in the non-obese subjects was decreased to 4.94 ± 6.21 mg/L, (p=0.04). The level of CRP in the obese group after eradication, on the other hand, decreased significantly and profoundly from 54.26 ± 23.67 to 18.73 ± 17.39 mg/l (p=0.01).
Table 1.
Biochemical Parameters in African Americans Before and After H. pylori (cagA) Eradication.
| Variables | Pre-Treatment | Post- treatment | p-values |
|---|---|---|---|
| Age (years) : Mean± SD | 55.82±10.89 | N/A | |
| BMI (kg/m2): Mean± SD | 32.6±9.2 | 31.9±8.8 | |
| Gender: Male (%) | 11(24%) | N/A | |
| Female (%) | 35 (75%) | ||
| P.folate (ng/ml): Mean± SD | 22.75±14.95 | 19.40±12.90 | 0.63 |
| RBC folate (ng/ml) : Mean± SD | 686.11±322.77 | 660.56±278.40 | NS |
| B-12 (pg/ml) Mean± SD | 607.01±246.29 | 628.122±214.03 | NS |
| CRP (mg/l) : Mean± SD | 30.24±28.45 | 11.43±14.21 | 0.03 |
| tHcy (μmol/l): Mean± SD | 9.29±3.15 | 8.52±2.49 | 0.25 |
Figure 1.
Blood level of high-sensitivity CRP in African Americans before (gray stripe) and 2 months after (black stripe) the eradication of their H. pylori (cagA) infection.
Discussion
Our present findings in contradiction to our previous report9 on the occurrence of hyperhomocysteinemia (HHcy) in CHD patients suggest that in African Americans H. pylori infection may not be a high-impact causal factor for Hcy elevations nor play a major role in explaining individual variation in Hcy levels. However, further confirmation of these observations should be made in a larger population.
The baseline blood level of high- sensitivity CRP was found to be significantly higher (see Table 1) in African Americans infected with this organism. High levels of CRP have previously been found to be associated with arterial inflammation, central to the initiation and progression of atherosclerosis13, and to be elevated in subjects with obesity and with the Metabolic Syndome14. We, therefore, stratified our subjects according to BMI values of less than 27, indicating a leaner subgroup with relatively normal weight for height (non-obese), and BMI greater than 27, indicating a more obese subgroup with overweight and obesity. The BMI of 27 as a cut-off value was chosen since most of our subjects (75%) were female18. These interesting results are graphically presented in Figure 1. It is generally accepted that a CRP value of more than 6 mg/L is indicative of an elevated state of inflammation14. The CRP values for the non-obese and obese groups prior to H. pylori eradication (shown in Figure 1) were respectively 8.88 ± 6.23 and 54.26 ± 23.67 mg/L, suggesting a state of inflammation in these subjects as expected. Following treatment of H. pylori, the level of CRP in the non-obese subjects was decreased to 4.94 ± 6.21 mg/L, (p=0.04), a value which then fell within the normal range. The level of CRP in the obese group after eradication, on the other hand, decreased significantly and profoundly from 54.26 ± 23.67 to 18.73 ± 17.39 mg/l (p=0.01). Although the CRP was lowered significantly in the obese group, it remained elevated above normal (> 6 mg/L) indicating perhaps the underlying influence of obesity or insulin resistance as reported earlier14. It is important to note, however, that the high baseline levels of CRP (54.26 ± 23.67 mg/L) in the obese subjects were dramatically reduced by treatment of the H. pylori (cagA) infection. Consequently, H. pylori (cagA) infection could be considered as an important explanation for systemic manifestation of inflammation in these African Americans, and one of the potential causal factors of inducing and/or aggravating arterial inflammation in obese individuals. In fact, in infected individuals, the contribution of H. pylori infection to overall systemic inflammation can be much greater than that attributable to obesity and/or insulin resistance.
The above observation is consistent with the increased inflammation due to H. pylori (cagA) infection may have a significant bearing on the pathogenesis of atherosclerosis. Although an association of this infection with CHD is controversial15,16, Kanbay, et al.17 hypothesized that this organism could be a major risk factor for endothelial dysfunction by virtue of its association in the elevation of some proinflammatory cytokins, particularly CRP. A thorough search of the literature also revealed that a handful of studies reported a possible link between H. pylori and CHD in subjects with higher BMI. For example, Lip et al.18 found a significant association with CHD in patients with BMI 28.4 ± 4.2 infected with H. pylori compared to subjects with BMI of 26.2 ± 3.2 (p= 0.019) who did not harbor this infection. Wald et al.19, on the other hand, found no association of CHD with H. pylori infected subjects with BMI of 25.9 ± 3.2. Therefore, a synergistic influence of adiposity (BMI) and H. pylori infection, particularly with cagA, may be operative in the initiation and/or progression of arterial inflammation, an important contributor to the pathogenesis of atherosclerosis. This warrants further prospective clinical investigations in order to verify the role of H. pylori infection, or other chronic/acute infections, in obese humans as an etiological factor for the development of CHD.
Acknowledgments
We thank Drs. Chandrika Piyathilake and Barbara Gower for their laboratory support in the estimation of biochemical parameters.
Sources of Funding
This study was partially funded by a grant from the American Heart Association (AHA), from NIH (DK-38764), and a merit review grant from the Department of Veterans Affairs, and core laboratory support from the UAB Diabetes Research and Training Center (P60 DK079626).
Footnotes
Disclosures
None
References
- 1.Ross R. Atherosclerosis – an inflammatory disease. New Engl J Med. 1999;340:115–126. doi: 10.1056/NEJM199901143400207. [DOI] [PubMed] [Google Scholar]
- 2.Libby P Egan D. Skarlatos S. Roles of infectious agents in atherosclerosis and restenosis: an assessment of the evidence and need for future research. Circulation. 1997;96:4095–4103. doi: 10.1161/01.cir.96.11.4095. [DOI] [PubMed] [Google Scholar]
- 3.Hocker M, Hohenberger P. Helicobacter pylori virulence factors-one part of a big picture. Lancet. 2003;362:1231–1233. doi: 10.1016/S0140-6736(03)14547-3. [DOI] [PubMed] [Google Scholar]
- 4.Graham DY, Adam E, Reddy GT, Agarwal JP, Agarwal R, Evans DJ, Jr, Malaty HM, Evans DG. Seroepidemiology of H. pylori infection in India: comparison of developing and developed countries. Dig Dis Sci. 1991;36:1084–88. doi: 10.1007/BF01297451. [DOI] [PubMed] [Google Scholar]
- 5.Singh RK, McMohan AD, Patel H, Packard CJ, Rathbone BJ, Samani NJ. Prospective analysis of the association of infection with cagA bearing strains of Helicobacter pylori and coronary heart disease. Heart. 2002;88:43–46. doi: 10.1136/heart.88.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pasceri V, Cammarota G, Patti G, Cuoco L, Gasbarrini A, Grillo RL, Fedeli G, Gasbarrini G, Maseri A. Association of Virulent Helicobacter pylori Strains With Ischemic Heart Disease. Circulation. 1998;97:1675–1679. doi: 10.1161/01.cir.97.17.1675. [DOI] [PubMed] [Google Scholar]
- 7.Farmer JA, Torre-Amione G. Atherosclerosis and inflammation. Curr Atherosclero Rep. 2002;4:92–98. doi: 10.1007/s11883-002-0031-5. [DOI] [PubMed] [Google Scholar]
- 8.Graham IM, Daly LE, Refsum HM, Robinson K, et al. Plasma homocysteine as risk factors for vascular disease. JAMA. 1997;277:1775–1781. doi: 10.1001/jama.1997.03540460039030. [DOI] [PubMed] [Google Scholar]
- 9.Khaled MA, Cornwell PE. Hyperhomocysteinemia due to H. pylori. Atherosclerosis. 2004;172:199–200. doi: 10.1016/j.atherosclerosis.2003.09.023. [DOI] [PubMed] [Google Scholar]
- 10.Schreiner PJ, Wu KK, Malinow MR, et al. Hyperhomocyst(e)inemia and Homeostatic factors: The Atherosclerosis Risk in Community Studies. Ann Epidemiol. 2002;12:228–236. doi: 10.1016/s1047-2797(01)00311-8. [DOI] [PubMed] [Google Scholar]
- 11.Abu-Mahfuz MZ, Prasad VM, Santogade P, et al. H. pylori recurrence after successful eradication. Am J Gastroenterol. 1997;92:2025–2028. [PubMed] [Google Scholar]
- 12.Roe DA. Drug and nutrient interactions. In: Bodwell CE, Erdman JW Jr, editors. Nutrient Interactions. Marcel Dekker, Inc.; New York: 1988. pp. 365–377. Ift Basic Symposium Series. [Google Scholar]
- 13.Ridker PM. High-Sensitivity C-reactive protein: potential adjunct for global risk assessment in the primary prevention of cardiovascular disease. Circullation. 2001;103:1813–1818. doi: 10.1161/01.cir.103.13.1813. [DOI] [PubMed] [Google Scholar]
- 14.Aronson D, Bartha P, Zinder O, et al. Obesity is the major determinant of elevated C-reactive protein in subjects with the metabolic syndrome. Internat J Obesity. 2004;28:674–679. doi: 10.1038/sj.ijo.0802609. [DOI] [PubMed] [Google Scholar]
- 15.Danesh J, Peto R. Risk factors for coronary heart disease and infection with Helicobacter pylori: meta-analysis of 18 studies. BMJ. 1998;316:1130–1132. doi: 10.1136/bmj.316.7138.1130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wolf SC, Brehm BR, Mayer O, et al. Infectious risk for atherosclerotic vascular disease in hemodialysis patients-Chlamydia pneumoniae but not Helicobacter pylori or cytomegalovirus is associated with increased C-reactive protein. Ren Fail. 2004;26:279–287. doi: 10.1081/jdi-120039527. [DOI] [PubMed] [Google Scholar]
- 17.Kanbay M, Kasapoglu B, Turgut F, et al. Helicobacter pylori: A major risk factor for endothelial dysfunction. Med Hypoth. 2007;69:227–228. doi: 10.1016/j.mehy.2006.12.001. [DOI] [PubMed] [Google Scholar]
- 18.Lip GH, Wise R, Beevers G. Association of Helicobacter pylori with coronary heart disease. BMJ. 1996;312:250–251. doi: 10.1136/bmj.312.7025.250b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wald NJ, Law MR, Morris JK, Bagnall AM. Helicobacter pylori and mortality from ischaemic heart disease: negative result from a large, prospective study. BMJ. 1997;315:1199–1201. doi: 10.1136/bmj.315.7117.1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gibson RS. Book: Principles of nutritional assessment. 1990. Vol. 315. Oxford Univ. Press; 1997. pp. 1199–1201. [Google Scholar]