Abstract
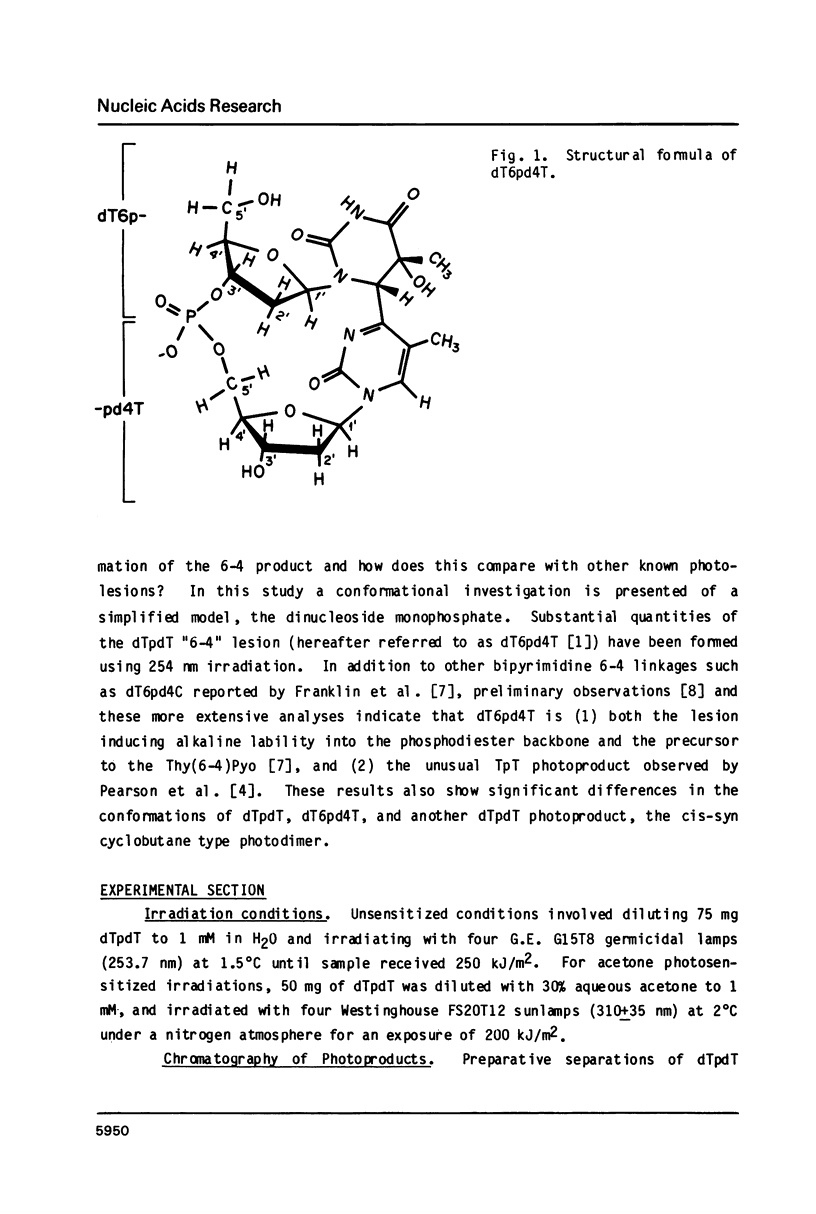
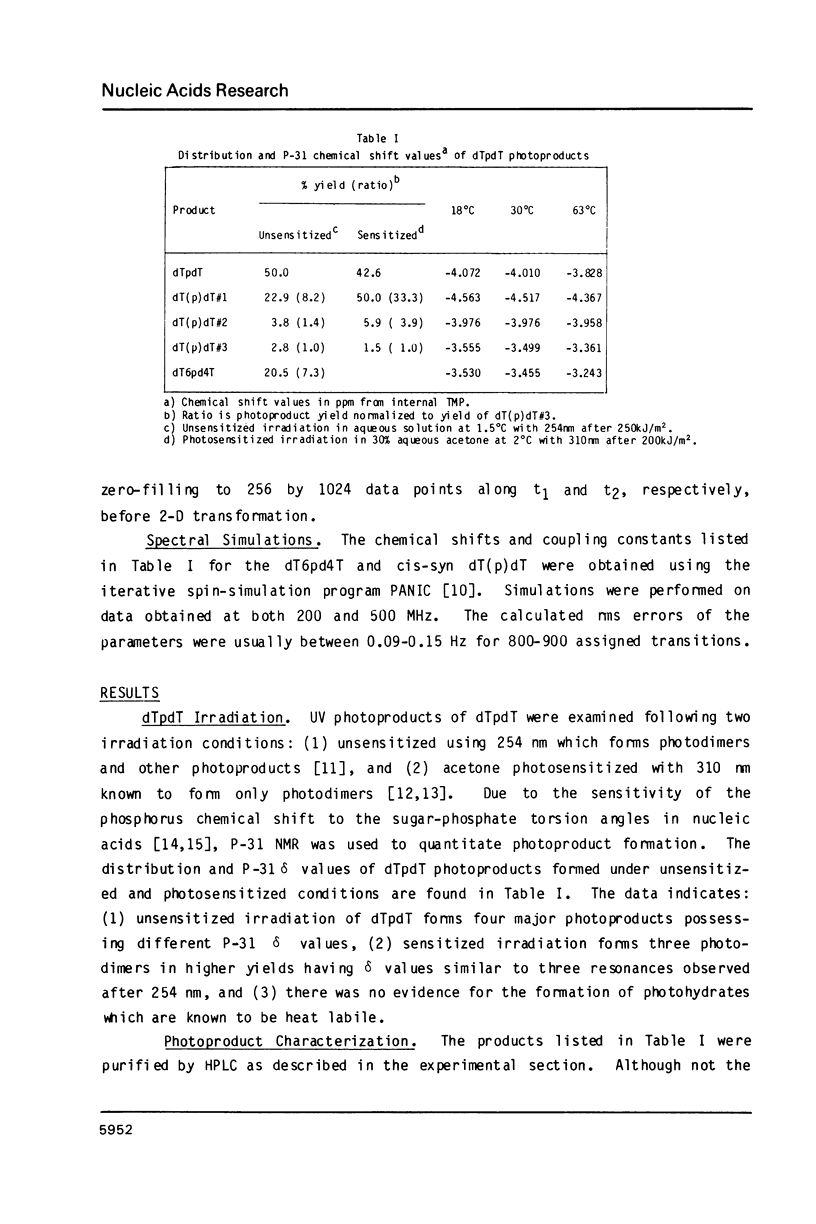
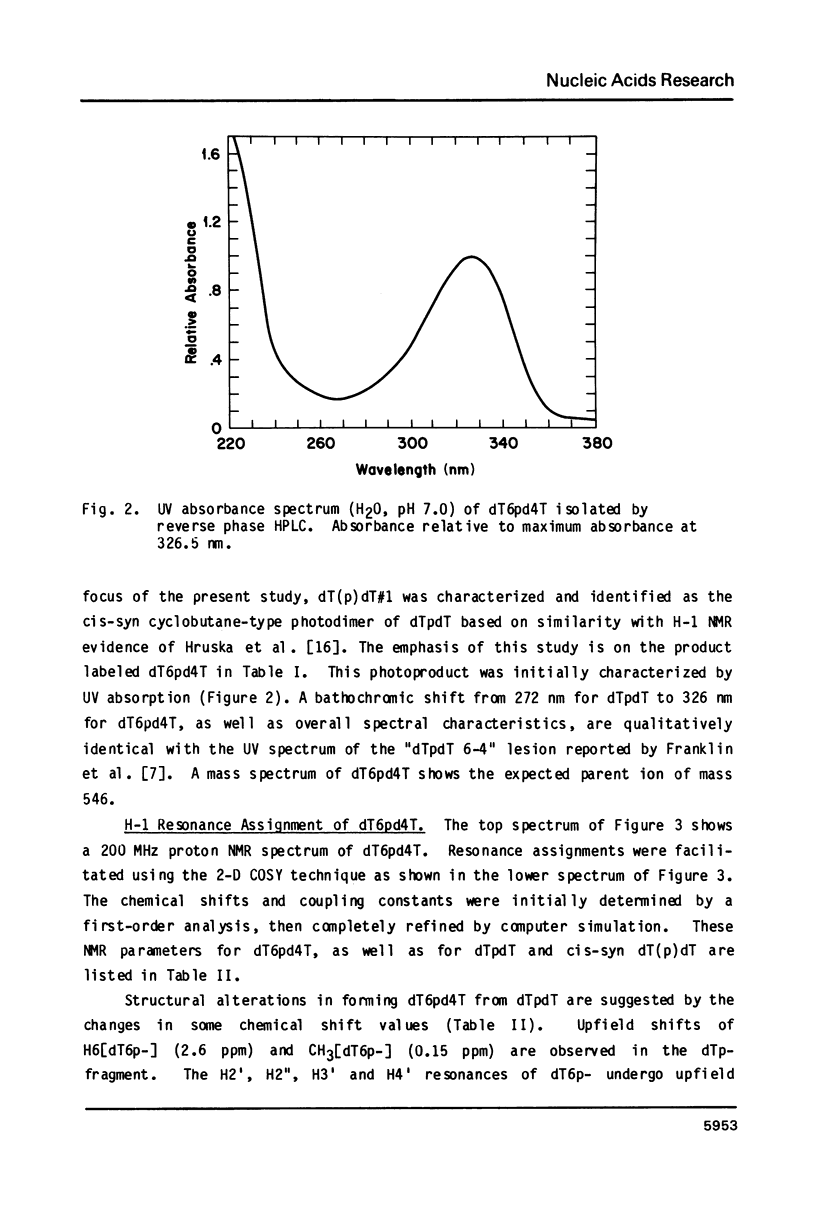
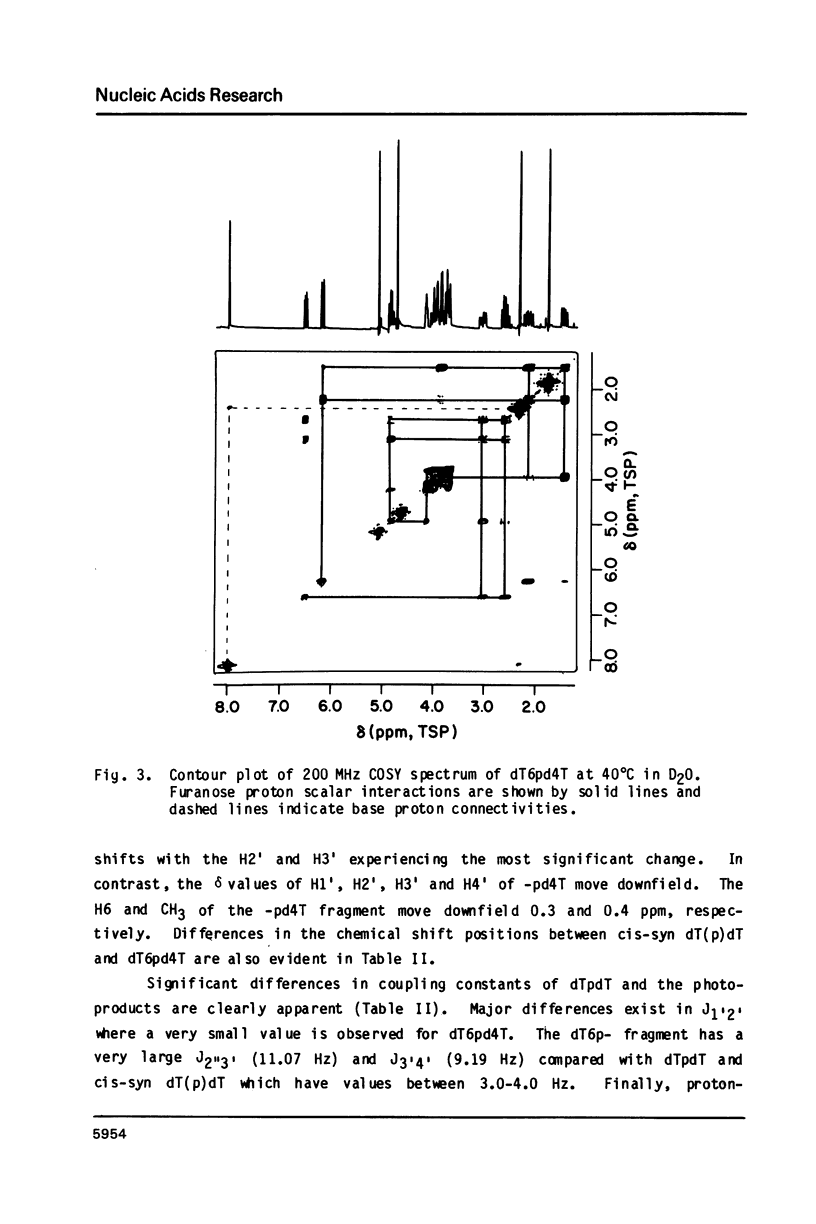
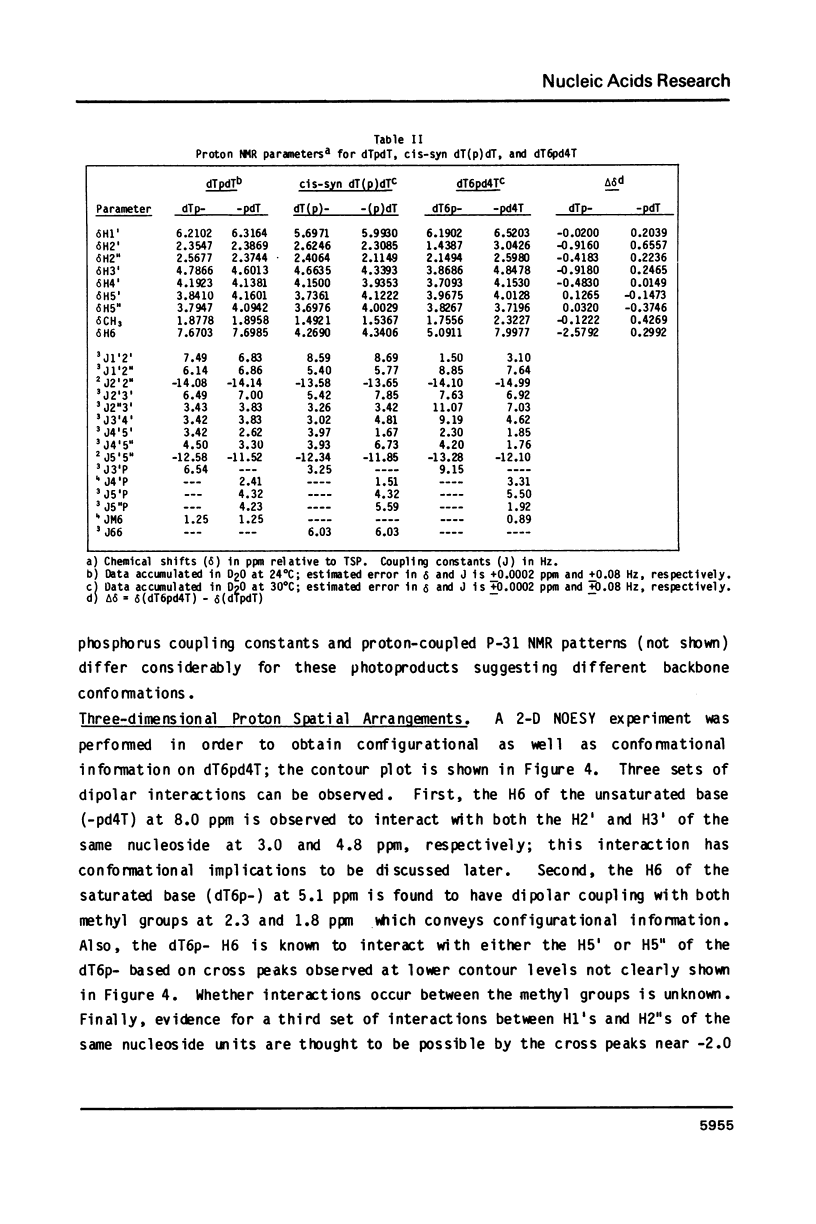
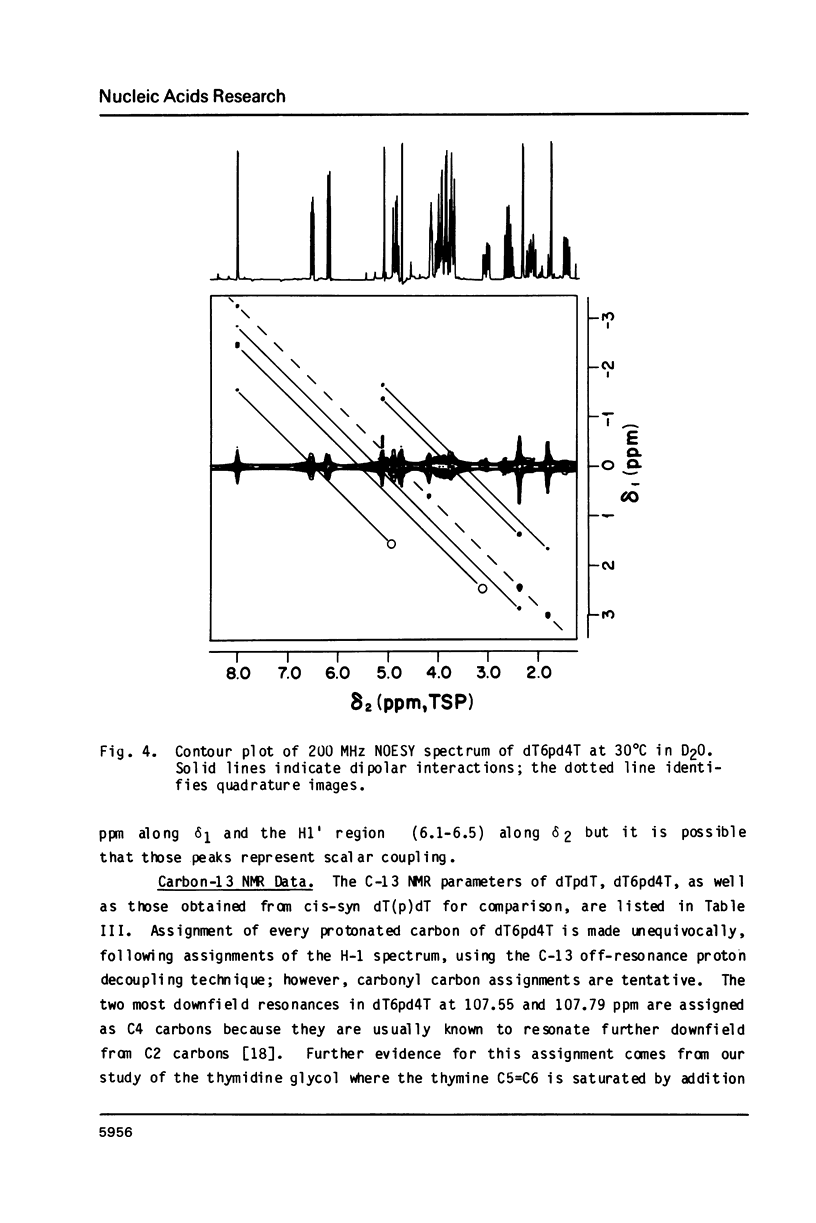
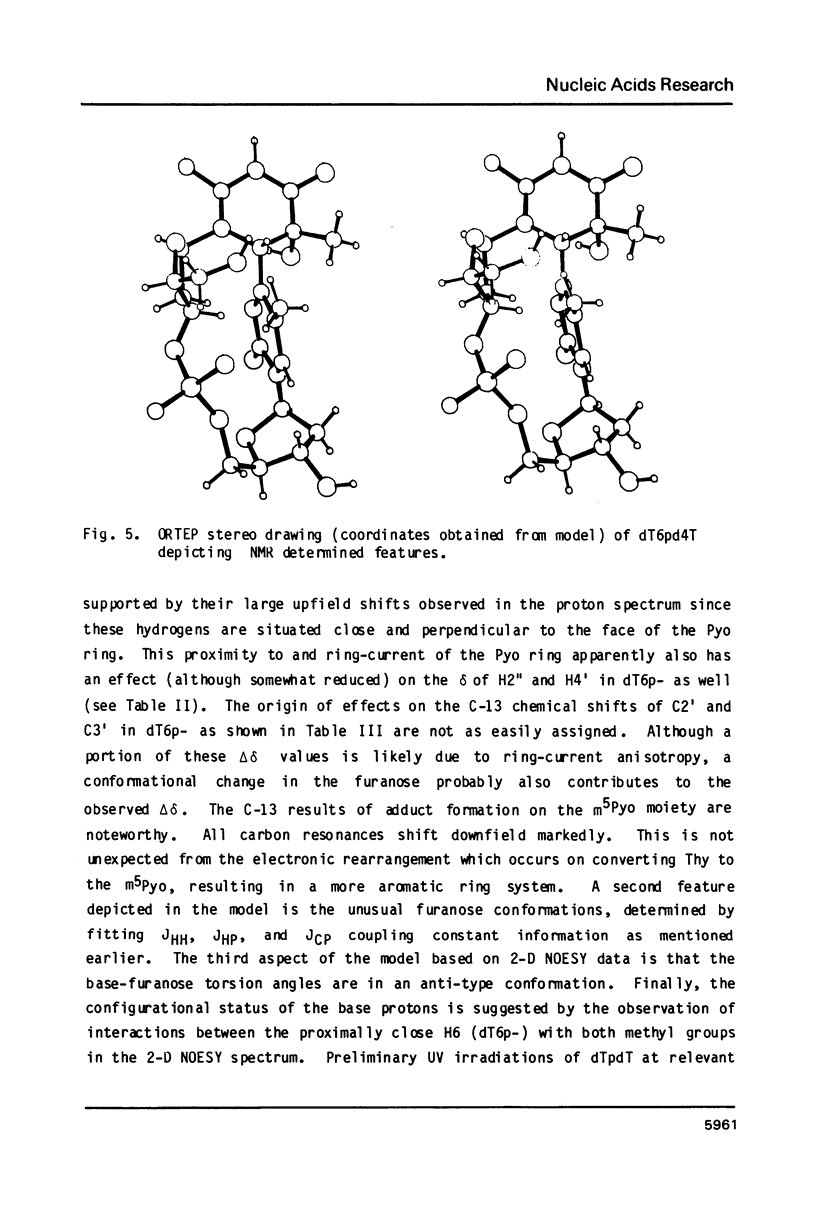
Irradiation of dTpdT with 300 kJ/m2 of 254 nm produces numerous photo-products, one of which labeled dT6pd4T[1] was purified by HPLC. dT6pd4T has a UV spectrum (H20, pH 7) with lambda max = 326 nm and lambda min = 265 nm, and a P-31 NMR resonance at -3.46 ppm (normal dTpdT occurs at -4.01 ppm; TMP, 30 degrees C). 2-D COSY NMR spectra facilitated proton resonance assignments and 2-D NOESY spectra aided analysis of spatial orientation. Carbon-13 and proton-coupled P-31 NMR spectra of dT6pd4T were also obtained. These analyses indicate: C5=C6 of dT6p- is saturated and the -pd4T base is more aromatic; the dT6p- base possesses a configuration of 5R, 6S; dT6p- and -pd4T have anti-type glycosidic conformations; furanose conformation of dT6p- is mainly C3'-endo and that of -pd4T exists in a C3'-endo in equilibrium C3'-exo; exocyclic bonds gamma (C5'-C4'), beta (05'-C5') and epsilon (C3'-03') are non-classical rotamers; dihedral angle about epsilon (C3'-03') is smaller relative to dTpdT.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alderfer J. L., Loomis R. E., Zielinski T. J. Fluorinated nucleic acid constituents: a carbon-13 nuclear magnetic resonance study of adenosine, cytidine, uridine, and their fluorinated analogues. Biochemistry. 1982 May 25;21(11):2738–2745. doi: 10.1021/bi00540a025. [DOI] [PubMed] [Google Scholar]
- Alderfer J. L., Ts'o P. O. Conformational properties of the furanose phosphate backbone in nucleic acids. A carbon-13 nuclear magnetic resonance study. Biochemistry. 1977 May 31;16(11):2410–2416. doi: 10.1021/bi00630a016. [DOI] [PubMed] [Google Scholar]
- Altona C., Sundaralingam M. Conformational analysis of the sugar ring in nucleosides and nucleotides. Improved method for the interpretation of proton magnetic resonance coupling constants. J Am Chem Soc. 1973 Apr 4;95(7):2333–2344. doi: 10.1021/ja00788a038. [DOI] [PubMed] [Google Scholar]
- Brash D. E., Haseltine W. A. UV-induced mutation hotspots occur at DNA damage hotspots. Nature. 1982 Jul 8;298(5870):189–192. doi: 10.1038/298189a0. [DOI] [PubMed] [Google Scholar]
- Cadet J., Ducolomb R., Hruska F. E. Proton magnetic resonance studies of 5,6-saturated thymidine derivatives produced by ionizing radiation. Conformational analysis of 6-hydroxylated diastereoisomers. Biochim Biophys Acta. 1979 Jun 20;563(1):206–215. doi: 10.1016/0005-2787(79)90021-2. [DOI] [PubMed] [Google Scholar]
- Cheng D. M., Sarma R. H. Intimate details of the conformational characteristics of deoxyribodinucleoside monophosphates in aqueous solution. J Am Chem Soc. 1977 Oct 26;99(22):7333–7348. doi: 10.1021/ja00464a038. [DOI] [PubMed] [Google Scholar]
- Davies D. B., Danyluk S. S. Nuclear magnetic resonance studies of 5'-ribo- and deoxyribonucleotide structures in solution. Biochemistry. 1974 Oct 8;13(21):4417–4434. doi: 10.1021/bi00718a027. [DOI] [PubMed] [Google Scholar]
- Franklin W. A., Lo K. M., Haseltine W. A. Alkaline lability of fluorescent photoproducts produced in ultraviolet light-irradiated DNA. J Biol Chem. 1982 Nov 25;257(22):13535–13543. [PubMed] [Google Scholar]
- Giessner-Prettre C., Pullman B. On the conformational dependence of the proton chemical shifts in nucleosides and nucleotides. I. Proton shifts in the ribose ring of pyrimidine nucleosides as a function of the torsion angle about the glycosyl bond. J Theor Biol. 1977 Mar 7;65(1):171–188. doi: 10.1016/0022-5193(77)90082-0. [DOI] [PubMed] [Google Scholar]
- Greenstock C. L., Johns H. E. Photosensitized dimerization of pyrimidines. Biochem Biophys Res Commun. 1968 Jan 11;30(1):21–27. doi: 10.1016/0006-291x(68)90706-7. [DOI] [PubMed] [Google Scholar]
- JOHNS H. E., PEARSON M. L., LEBLANC J. C., HELLEINER C. W. THE ULTRAVIOLET PHOTOCHEMISTRY OF THYMIDYLYL-(3'-5')-THYMIDINE. J Mol Biol. 1964 Aug;9:503–524. doi: 10.1016/s0022-2836(64)80223-0. [DOI] [PubMed] [Google Scholar]
- Karle I. L. Crystal structure of a thymine-thymine adduct from irradiated thymine. Acta Crystallogr B. 1969 Oct 15;25(10):2119–2126. doi: 10.1107/s056774086900522x. [DOI] [PubMed] [Google Scholar]
- Karle I. L., Wang S. Y., Varghese A. J. Crystal and molecular structure of a thymine-thymine adduct. Science. 1969 Apr 11;164(3876):183–184. doi: 10.1126/science.164.3876.183. [DOI] [PubMed] [Google Scholar]
- Kućan Z., Waits H. P., Chambers R. W. Acetone-sensitized photochemistry of some pyrimidine dinucleoside phosphates. Biochemistry. 1972 Aug 15;11(17):3290–3295. doi: 10.1021/bi00767a026. [DOI] [PubMed] [Google Scholar]
- Lankhorst P. P., Haasnoot C. A., Erkelens C., Altona C. Carbon-13 NMR in conformational analysis of nucleic acid fragments. 2. A reparametrization of the Karplus equation for vicinal NMR coupling constants in CCOP and HCOP fragments. J Biomol Struct Dyn. 1984 Jun;1(6):1387–1405. doi: 10.1080/07391102.1984.10507527. [DOI] [PubMed] [Google Scholar]
- Pearson M. L., Ottensmeyer F. P., Johns H. E. Properties of an unusual photoproduct of U.V. irradiated thymidylyl-thymidine. Photochem Photobiol. 1965 Sep;4(4):739–747. doi: 10.1111/j.1751-1097.1965.tb07916.x. [DOI] [PubMed] [Google Scholar]
- Varghese A. J., Wang S. Y. Thymine-thymine adduct as a photoproduct of thymine. Science. 1968 Apr 12;160(3824):186–187. doi: 10.1126/science.160.3824.186. [DOI] [PubMed] [Google Scholar]



